应用化学 ›› 2024, Vol. 41 ›› Issue (4): 547-556.DOI: 10.19894/j.issn.1000-0518.230346
原子层沉积法制备限域型Pt基催化剂及其苯酚催化性能
王眉花1, 冯禹1, 王运坤1, 赵旭东1, 杨雯1( ), 董川2(
), 董川2( )
)
- 1.太原科技大学材料科学与工程学院,分析测试中心,太原 030024
2.山西大学环境科学研究所,太原 030006
-
收稿日期:2023-11-06接受日期:2024-02-20出版日期:2024-04-01发布日期:2024-04-28 -
通讯作者:杨雯,董川 -
基金资助:山西省科技创新人才团队专项(202204051001002);山西省基础研究计划资助项目(20210302124471);国家自然科学基金青年项目(22208230);山西省留学回国人员科技活动择优资助项目(20230033)
Preparation and Catalytic Performance of the Confined Pt Catalyst by Atomic Layer Deposition
Mei-Hua WANG1, Yu FENG1, Yun-Kun WANG1, Xu-Dong ZHAO1, Wen YANG1( ), Chuan DONG2(
), Chuan DONG2( )
)
- 1.Instrumental Analysis Center,School of Material Science and Engineering,Taiyuan University of Science and Technology,Taiyuan 030024,China
2.Institute of Environmental Science,Shanxi University,Taiyuan 030006,China
-
Received:2023-11-06Accepted:2024-02-20Published:2024-04-01Online:2024-04-28 -
Contact:Wen YANG,Chuan DONG -
About author:dc@sxu.edu.cn
yangwen@tyust.edu.cn;
-
Supported by:Shanxi Province Science and Technology Innovation Talent Team Special Project(202204051001002);Shanxi Province Basic Research Program Funding Project(20210302124471);the National Natural Science Foundation of China(22208230);Shanxi Provincial Excellent Support Program for Returned Overseas Scholars(20230033)
摘要:
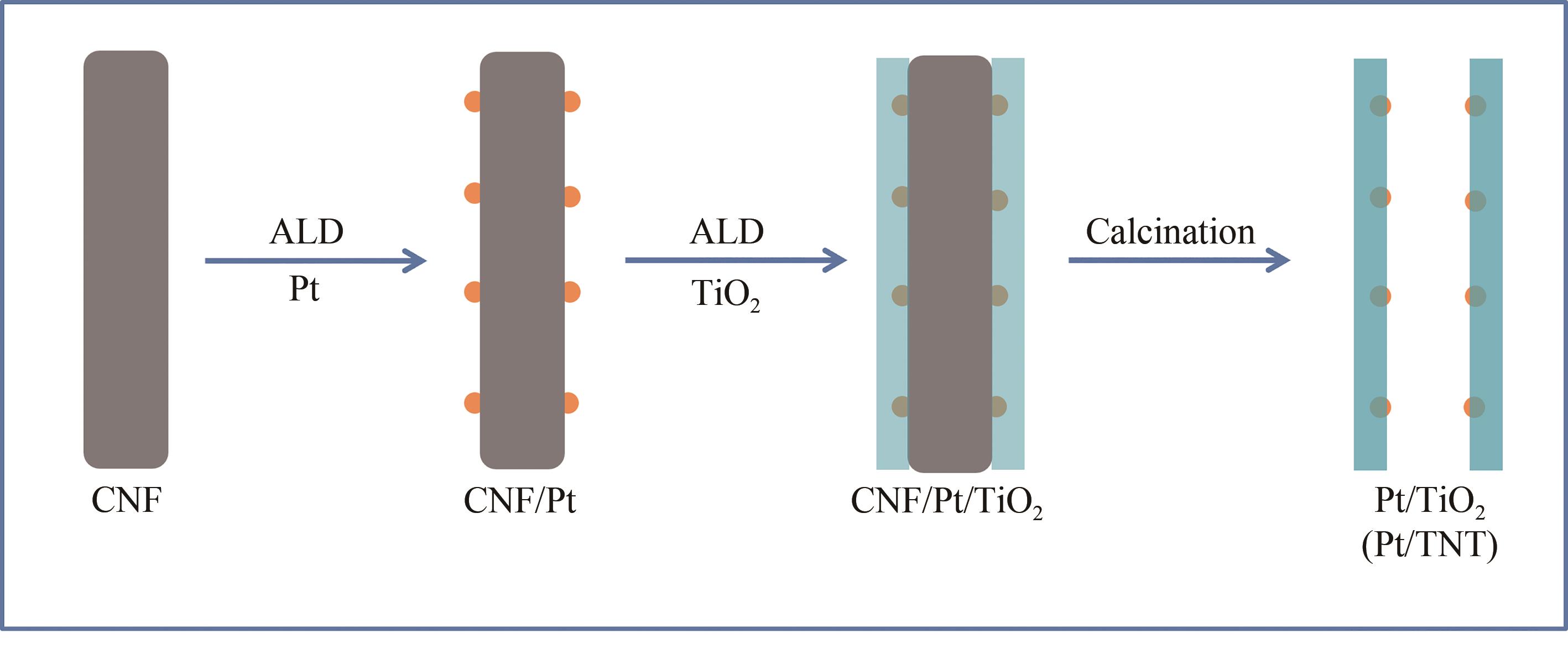
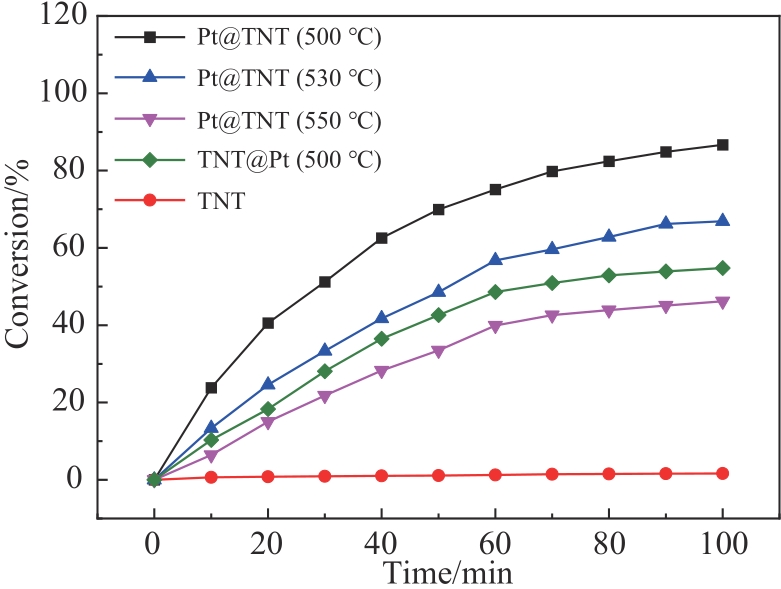
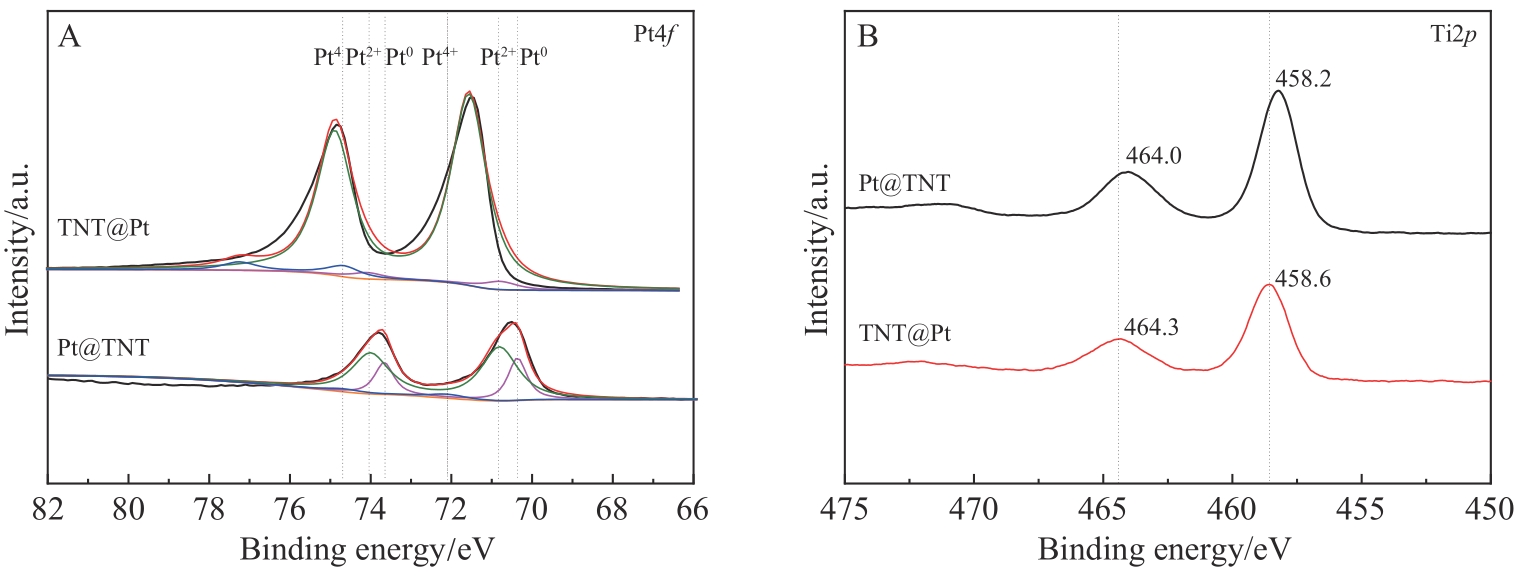
选择合适的载体负载贵金属Pt纳米粒子,对于制备高效的苯酚加氢催化剂具有重要意义。使用原子层沉积技术,以碳纳米纤维作为模板,在其表面依次沉积Pt纳米粒子,厚的氧化钛壳层,通过高温烧结除去模板,得到限域在氧化钛纳米管内壁的Pt纳米粒子。 在500 ℃烧结温度下Pt纳米粒子的粒径在2.0~3.2 nm之间,平均粒径为2.6 nm,尺寸高度一致,均匀地分散并且嵌入到氧化钛纳米管的中空管道内壁。与常规负载在氧化钛纳米管外壁的Pt纳米粒子相比,该限域催化剂的催化转化率更高,在500 ℃烧结温度下转化频率(TOF)值为482.1 h-1,表现出更好的催化活性,而且由于氧化钛纳米管的保护作用,可有效防止Pt纳米粒子在反应过程中的聚集和脱落,在反应后Pt的质量分数仅降低了4.52%,其形貌也没有发生明显改变,从而表现出优异的催化稳定性。
中图分类号:
引用本文
王眉花, 冯禹, 王运坤, 赵旭东, 杨雯, 董川. 原子层沉积法制备限域型Pt基催化剂及其苯酚催化性能[J]. 应用化学, 2024, 41(4): 547-556.
Mei-Hua WANG, Yu FENG, Yun-Kun WANG, Xu-Dong ZHAO, Wen YANG, Chuan DONG. Preparation and Catalytic Performance of the Confined Pt Catalyst by Atomic Layer Deposition[J]. Chinese Journal of Applied Chemistry, 2024, 41(4): 547-556.
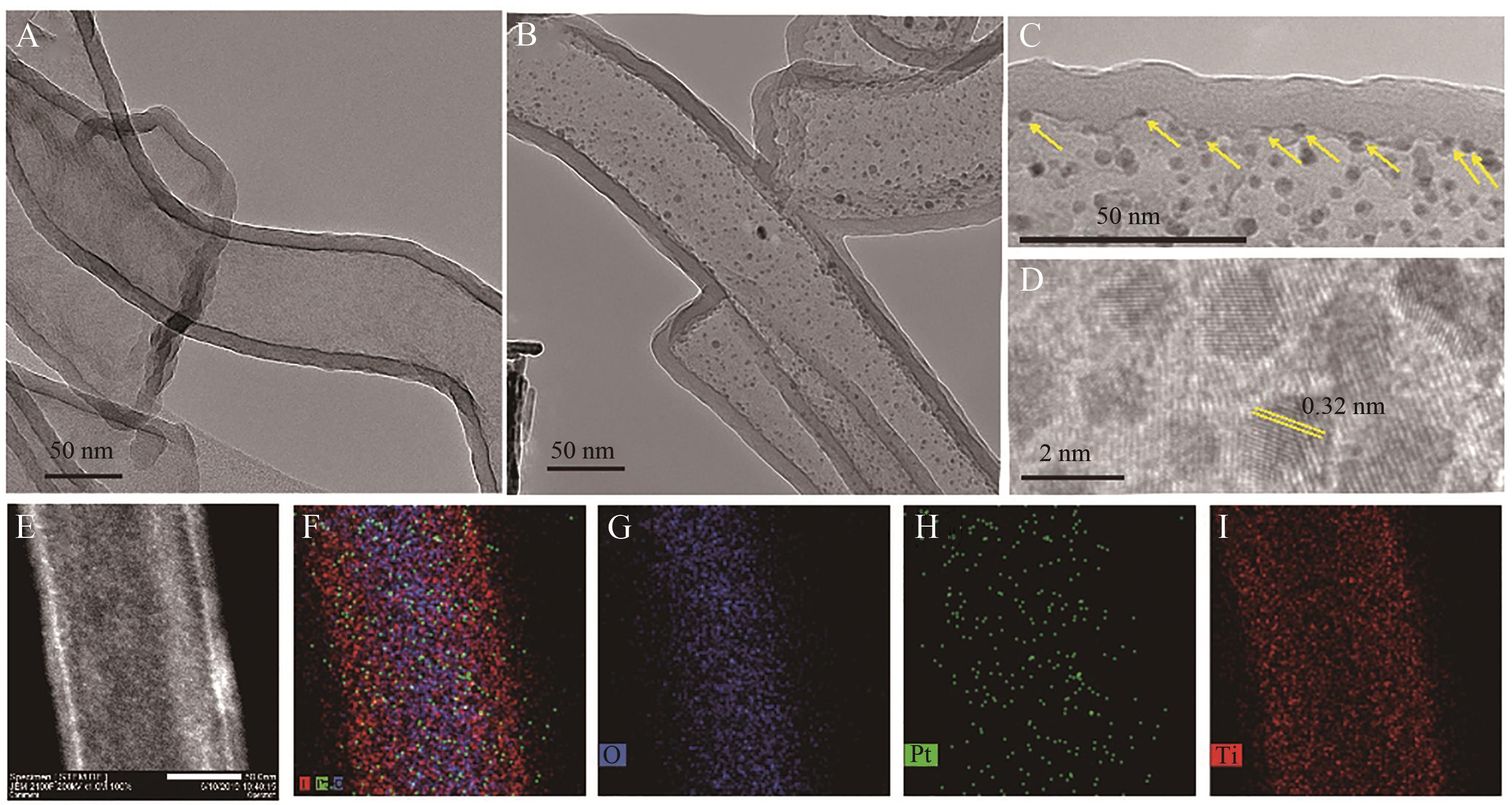
图2 氧化钛纳米管(A)和氧化钛纳米管内的Pt纳米粒子催化剂(B)的低倍透射电子显微镜图。 Pt纳米粒子的高分辨率透射电子显微镜图(C、D)。 氧化钛纳米管内的Pt纳米粒子的扫描透射电子显微镜图(STEM图)(E)和相应的Mapping元素图谱,元素代表分别为O(G)、Pt(H)、Ti(I)和这些元素的叠加(F)
Fig.2 TEM images of (A) TiO2 nanotubes and (B) Pt nanoparticles in TiO2 nanotubes. High resolution transmission electron microscopy of Pt nanoparticles (C, D). The STEM (E) and Mapping element diagram of Pt nanoparticles in TiO2 nanotubes, representing O (G), Pt (H), Ti (I) and their overlap (F)
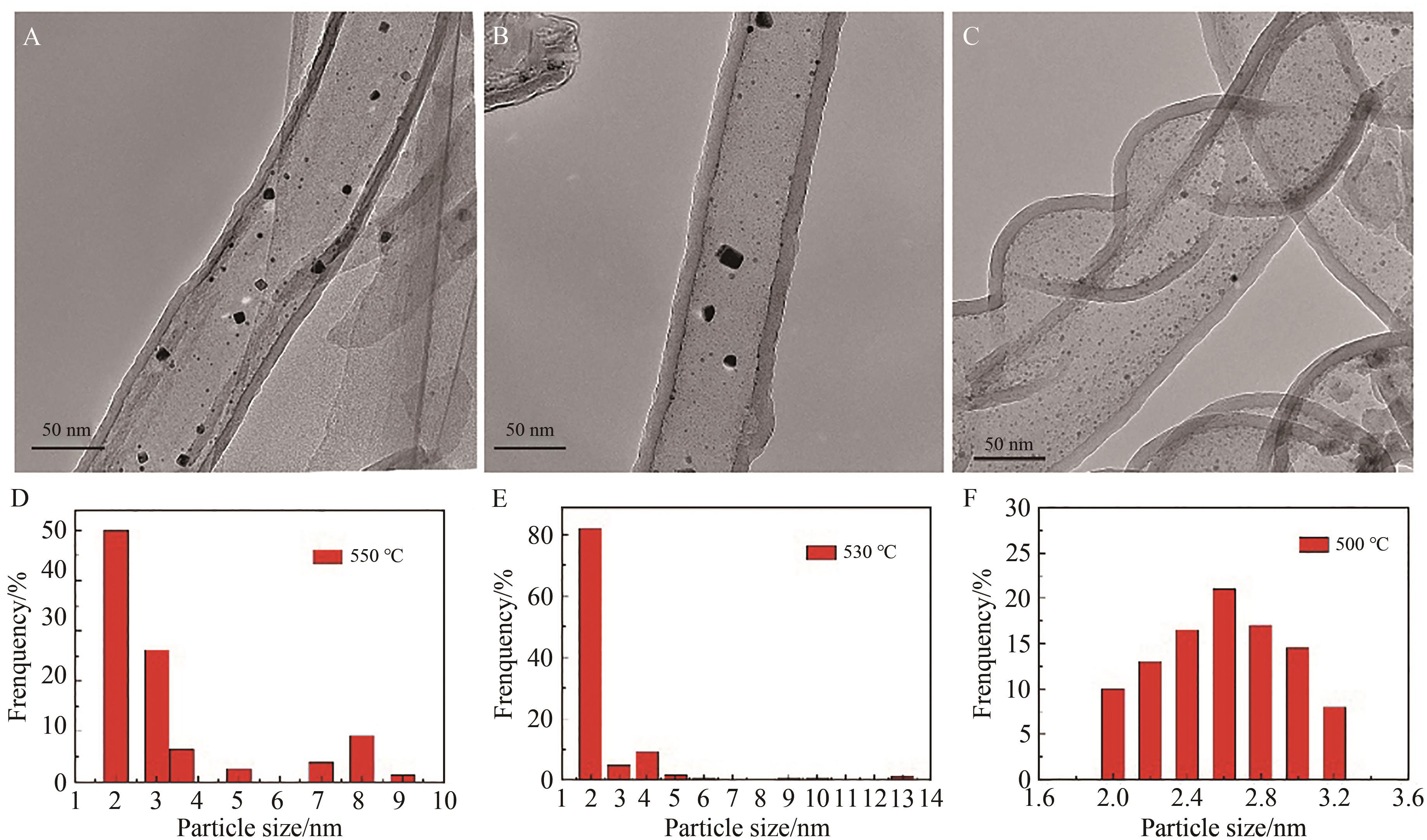
图3 不同烧结温度下(A) 550、(B) 530和(C) 500 ℃的TiO2纳米管内的Pt纳米粒子催化剂的TEM图和相应的Pt纳米粒子的粒径分布图(D、E、F)
Fig.3 TEM images of Pt nanoparticles in TiO2 nanotubes sintered at (A) 550, (B) 530 and (C) 500 ℃ and size distribution of corresponding Pt nanoparticles (D, E, F)
| Sample | w( Pt)/% a | Conversion/% b | Selectivity/% c | TOF/h-1 d |
|---|---|---|---|---|
| TNT@Pt | 2.55 | 46.2 | 98.7 | 281.5 |
| Pt@TNT(500 ℃) | 2.44 | 75.1 | 97.0 | 482.1 |
Pt@TNT(530 ℃) Pt@TNT(550 ℃) Pt@TNT4th | 2.36 2.49 2.33 | 56.8 39.9 70.3 | 97.2 96.8 98.5 | 377.1 249.9 472.7 |
| TNT@Pt4th | 1.01 | 10.3 | 94.3 | 159.1 |
表1 催化剂对苯酚加氢的催化性能比较
Table 1 Catalytic performance of these samples for the hydrogenation of phenol
| Sample | w( Pt)/% a | Conversion/% b | Selectivity/% c | TOF/h-1 d |
|---|---|---|---|---|
| TNT@Pt | 2.55 | 46.2 | 98.7 | 281.5 |
| Pt@TNT(500 ℃) | 2.44 | 75.1 | 97.0 | 482.1 |
Pt@TNT(530 ℃) Pt@TNT(550 ℃) Pt@TNT4th | 2.36 2.49 2.33 | 56.8 39.9 70.3 | 97.2 96.8 98.5 | 377.1 249.9 472.7 |
| TNT@Pt4th | 1.01 | 10.3 | 94.3 | 159.1 |
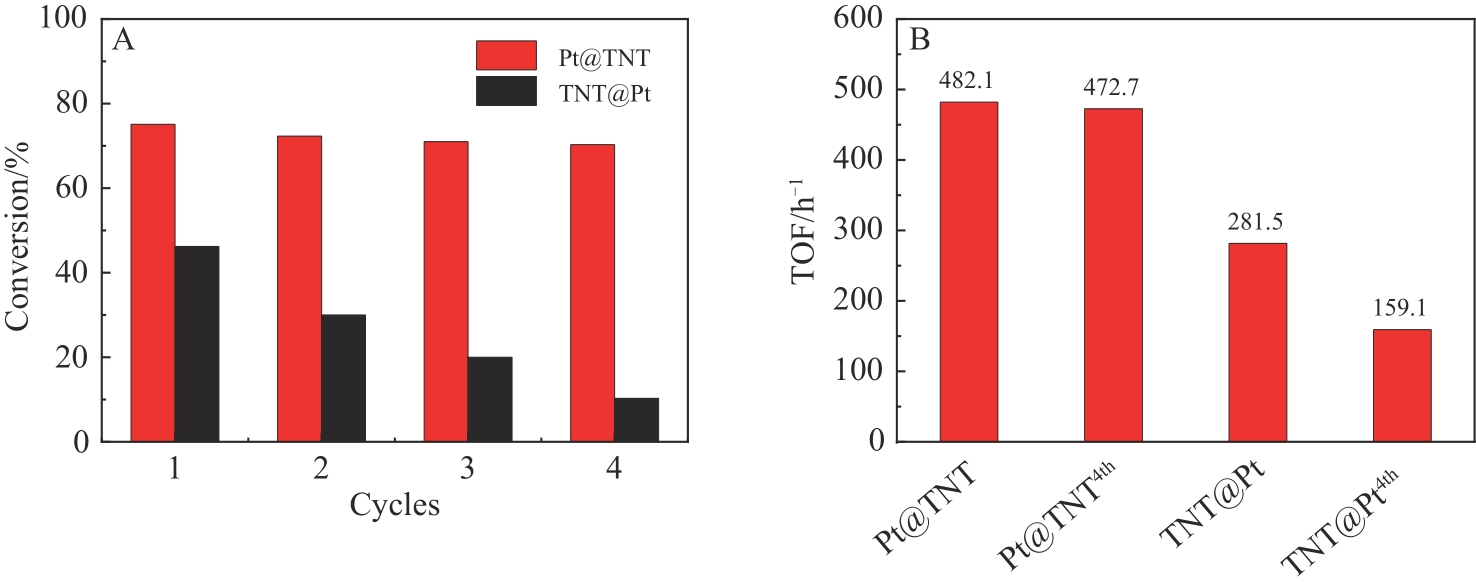
图5 (A)催化剂的循环稳定性(每个循环的反应时间为1 h); (B)催化剂对于苯酚加氢TOF值的比较
Fig.5 (A) The recycling stability of phenol hydrogenation for the catalysts (the reaction time is 1 h per run for both catalysts); (B) The comparison of TOF values of phenol hydrogenation
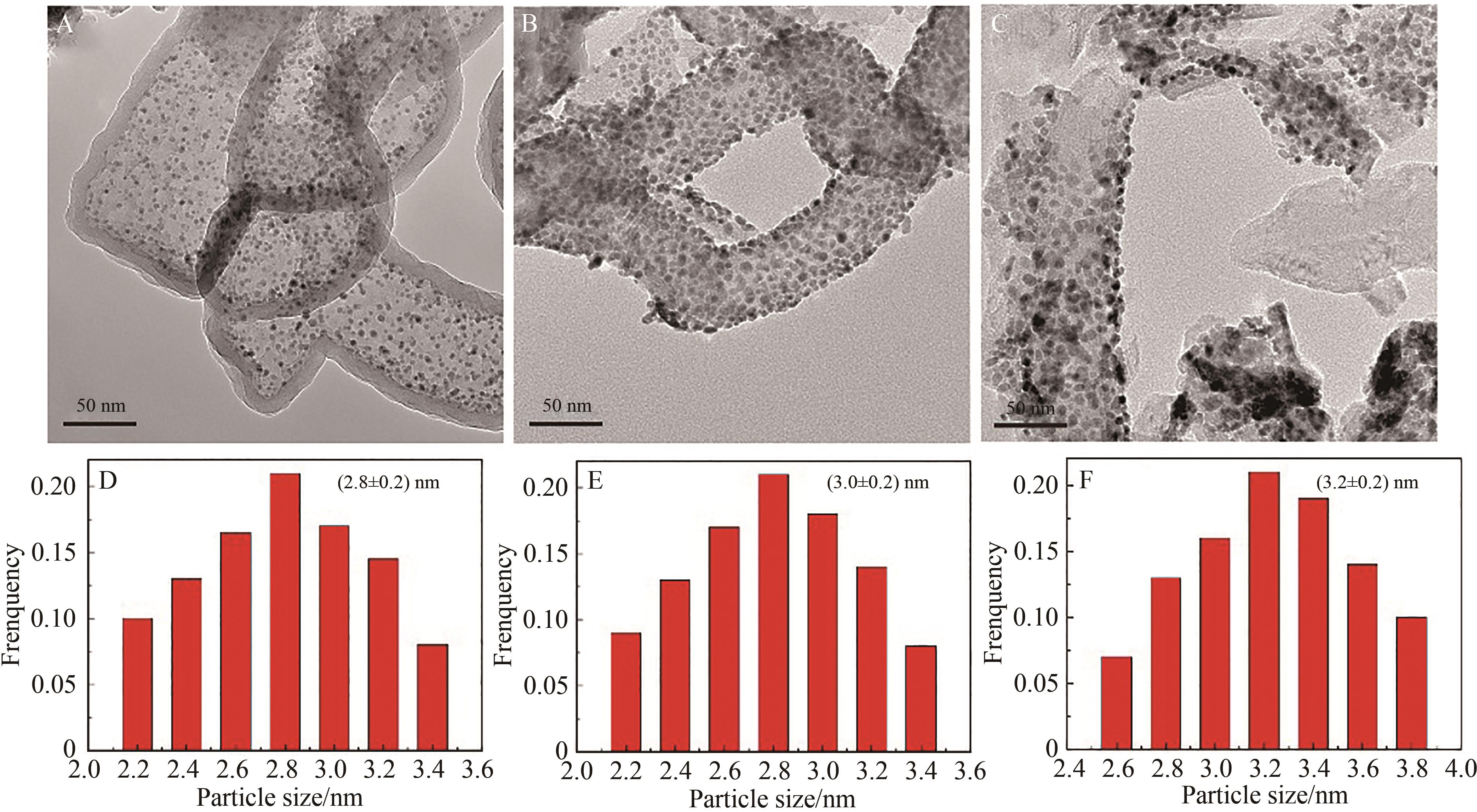
图6 Pt@TNT(A)反应后、TNT@Pt反应前(B)和(C)反应后的TEM图和相应的Pt纳米粒子的粒径分布图(D、E、F)
Fig.6 TEM images of (A) Pt@TNT after reaction, (B) TNT@Pt before, after reaction (C) and size distribution of corresponding Pt nanoparticles (D, E, F)
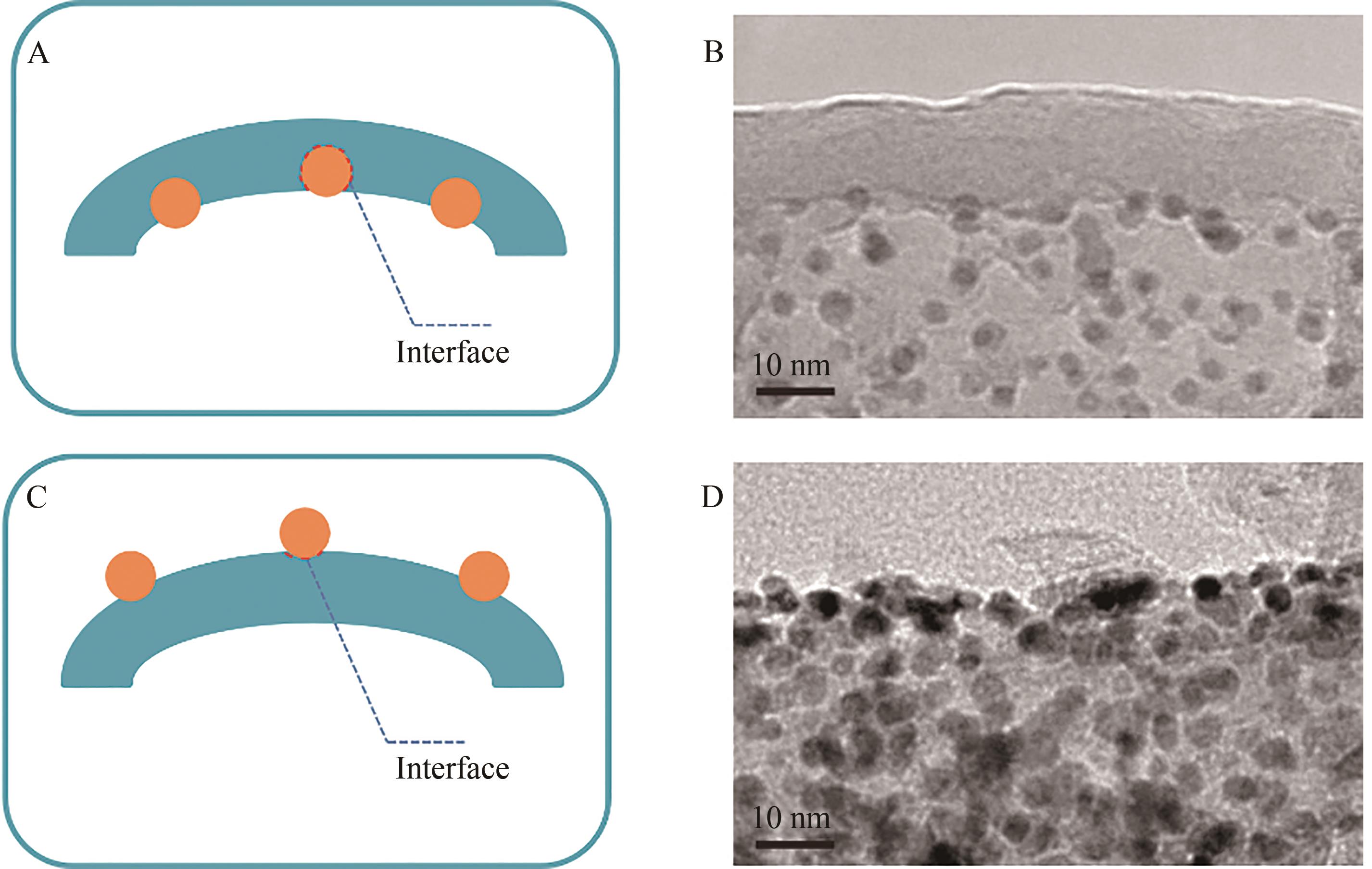
图8 Pt@TNT(A、B)和TNT@Pt(C、D)催化剂的Pt-TiO2界面结构模型示意图及TEM图
Fig.8 The schematic diagram of the structure models and TEM images of Pt-TiO2 interfaces of Pt@TNT (A, B) and TNT@Pt (C,D) catalysts.
| 1 | LIU S, HAN J, WU Q. Hydrogenation of phenol to cyclohexanone over bifunctional Pd/C-heteropoly acid catalyst in the liquid phase[J]. Catal Lett, 2019, 149(9): 2383-2389. |
| 2 | PORWA G, GUPTA S, SREEDHALA S, et al. Mechanistic insights into the pathways of phenol hydrogenation on Pd nanostructures[J]. ACS Sustainable Chem Eng, 2019, 7(20): 17126-17136. |
| 3 | LI F, CAO B, ZHU W X, et al. Hydrogenation of phenol over Pt/CNTs: the effects of Pt loading and reaction solvents[J]. Catalysts, 2017, 7(5): 145. |
| 4 | 罗二桂, 唐涛, 王艺, 等. 两电子氧还原制备过氧化氢: 贵金属催化剂的几何与电子结构调控的研究进展[J]. 应用化学, 2023, 40(8): 1063-1076. |
| LUO E G, TANG T, WANG Y, et al. Progress on tuning the geometric and electronic structure of precious metal catalysts for hydrogen peroxide production via two-electron oxygen reduction[J]. Chin J Appl Chem, 2023, 40(8): 1063-1076. | |
| 5 | SONG Y, GUTIERREZ O Y, HERRANZ J, et al. Aqueous phase electrocatalysis and thermal catalysis for the hydrogenation of phenol at mild conditions[J]. Appl Catal B: Environ, 2016, 182: 236-246. |
| 6 | YANG X, YU X, LONG L Z, et al. Pt nanoparticles entrapped in titanate nanotubes (TNT) for phenol hydrogenation: the confinement effect of TNT[J]. Chem Commun, 2014, 50(21): 2794-2796. |
| 7 | ZHANG H C, XU B, WANG X. Engineering nanointerfaces for nanocatalysis[J]. Chem Soc Rev, 2014, 43(22): 7870-7886 |
| 8 | ZHANG F, JIAO F, PAN X L, et al. Tailoring the oxidation activity of Pt nanoclusters via encapsulation[J]. ACS Catal, 2015, 5(2): 1381-1385. |
| 9 | 朱诗尧, 李平, 叶黎城, 等. 基于Pt/CNTs催化剂的燃料电池Pt/Buckypaper催化层的制备与表征[J]. 材料工程,2018, 46(6): 27-35. |
| ZHU S Y, LI P, YE L C, et al. Preparation and characterization of Pt/Buckypaper catalytic layer based on Pt/CNTs catalyst for fuel cells[J]. J Mater Eng, 2018, 46 (6): 27-35. | |
| 10 | 杨春巍, 胡信国, 张亮, 等. 多壁碳纳米管的超声处理对PtRu/MWCNTs电催化性能的影响[J]. 材料工程, 2008(7): 79-82. |
| YANG C W, HU X G, ZHANG L, et al. Study of functionalization multiwall carbon nanotubes by ultrasound[J]. J Mater Eng, 2008(7): 79-82. | |
| 11 | NAKAMURA I, MANTOKU H, FURUKAWA T, et al. Active sites for hydrogen dissociation over TiOx/Au(111) surfaces[J]. J Phys Chem C, 2011, 115(32): 16074-16080. |
| 12 | ZHAO W X, WANG X T, SANG H X, et al. Synthesis of Bi-doped TiO2 nanotubes and enhanced photocatalytic activity for hydrogen evolution from glycerol solution[J]. Chin J Chem, 2013, 31(3): 415-420. |
| 13 | JIANG D W, ZHOU T S, SUN Q, et al. Enhanced visible-light-induced photoelectrocatalytic degradation of methyl orange by CdS sensitized TiO2 nanotube arrays electrode[J]. Chin J Chem, 2011, 29(11): 2505-2510. |
| 14 | DONG J N, HUANG J Y, WANG A, et al. Vertically-aligned Pt-decorated MoS2 nanosheets coated on TiO2 nanotube arrays enable high-efficiency solar-light energy utilization for photocatalysis and self-cleaning SERS devices[J]. Nano Energy, 2020, 71: 104-579. |
| 15 | MAITY S, RANA M, BEJ S, et al. TiO2-ZrO2 mixed oxide as a support for hydrotreating catalyst[J]. Catal Lett, 2001, 72: 115-119. |
| 16 | WU Z F, LI Y Y, HUANG W X. Size-dependent Pt-TiO2 strong metal-support interaction[J]. J Phys Chem Lett, 2020, 11(12): 4603-4607. |
| 17 | HAN B, GUO Y L, HUANG Y K, et al. Strong metal-support interactions between Pt single atoms and TiO2[J]. Angew Chem Int Ed, 2020, 59(29): 11824-11829. |
| 18 | LOU B H, CHEN C L, LIU J J, et al. Selectively depositing Bi2O3 quantum dots on TiO2 nanotubes for efficient visible-light-driven photocatalysis[J]. Mater Lett, 2021, 288: 129-413. |
| 19 | TSUCHIYA H, SCHMUKI P. Less known facts and findings about TiO2 nanotubes[J]. Nanoscale, 2020, 12(15): 8119-8132. |
| 20 | ZHAO Y X, ZHU L J, YU Y M, et al. Facile one-pot preparation of Ti3+, N co-doping TiO2 nanotube arrays and enhanced photodegradation activities by tuning tube lengths and diameters[J]. Catal Today, 2020, 355: 563-572. |
| 21 | 于亚洲, 赵先亮, 谭宁宁, 等. 聚烯丙胺改性磺酸催化剂催化苯甲酸正丁酯的合成[J]. 应用化学, 2023, 40(10): 1430-1436. |
| YU Y Z, ZHAO X L, TAN N N, et al. Polyallylamine modified sulfonic acid catalyst catalyse synthesis of n-butyl benzoate[J]. Chin J Appl Chem, 2023, 40(10): 1430-1436. | |
| 22 | WANG M H, GAO Z, ZHANG B, et al. Ultrathin coating of confined Pt nanocatalysts by atomic layer deposition for enhanced catalytic performance in hydrogenation reactions[J]. Chem Eur J, 2016, 22(25): 8438-8443. |
| 23 | ZHANG J K, YU Z B, GAO Z, et al. Porous TiO2 nanotubes with spatially separated platinum and CoOx cocatalysts produced by atomic layer deposition for photocatalytic hydrogen production[J]. Angew Chem Int Ed, 2017, 56(3): 816-820. |
| 24 | KIM M, BAE C, KIM H, et al. Confined crystallization of anatase TiO2 nanotubes and their implications on transport properties[J]. J Mater Chem A, 2013, 1(45): 14080-14088. |
| 25 | QIN Y, ZHANG Z K, CUI Z L. Helical carbon nanofibers with a symmetric growth mode[J]. Carbon, 2004, 42(10): 1917-1922. |
| 26 | WANG M H, LI Y, GAO X L, et al. TiO2 ultrathin coating of SBA-15/Au by atomic layer deposition for enhanced catalytic performance in epoxidation of styrene[J]. J Phys Chem C, 2020, 124(19): 10530-10540. |
| 27 | QIN Y, ZHANG Z K, CUI Z L. Helical carbon nanofibers prepared by pyrolysis of acetylene with a catalyst derived from the decomposition of copper tartrate[J]. Carbon, 2003, 41(15): 3072-3074. |
| 28 | YAO Q, WANG C, WANG H, et al. Revisiting the Au particle size effect on TiO2-coated Au/TiO2 catalysts in CO oxidation reaction[J]. J Phys Chem C, 2016, 120(17): 9174-9183. |
| 29 | SU X, LU K L, CHANG Y L, et al. Immersion deposition of Pt nanoparticles on porous silicon for methanol oxidation[J]. Chin J Chem, 2009, 27(9):1682-1686. |
| 30 | GAO Z, DONG M, WANG G Z, et al. Multiply confined nickel nanocatalysts produced by atomic layer deposition for hydrogenation reactions[J]. Angew Chem Int Ed, 2015, 54: 9006-9100. |
| 31 | WANG C J, LIU X, HE W J, et al. All-solid-state Z-scheme photocatalysts of g-C3N4/Pt/macroporous-(TiO2@carbon) for selective boosting visible-light-driven conversion of CO2 to CH4[J]. J Catal, 2020, 389: 440-449. |
| 32 | XIE J, YU Z H, XIA D G. Electrochemical sensor for oxidation of NO based on Au-Pt nanoparticles self-assembly film[J]. Chin J Chem, 2009, 27(5):887-890. |
| [1] | 孙天礼, 朱国, 何海, 黄炳坤, 熊兆锟, 赖波. 单原子催化剂在类芬顿水处理领域的研究进展[J]. 应用化学, 2024, 41(2): 217-229. |
| [2] | 沈天瑶, 杨怿, 于海鹤, 徐鹏, 张广山, 王鹏. CoFe-LDH/泡沫铜的制备及催化介质阻挡放电等离子体降解水中敌草隆性能与机制[J]. 应用化学, 2024, 41(2): 243-255. |
| [3] | 曹梦寒, 徐鹏, 石凤银, 李桂鄂, 张国栋, 郑庆柱, 张广山. CoFe2O4/MXene复合材料的构筑及活化过氧单硫酸盐降解阿特拉津[J]. 应用化学, 2024, 41(2): 256-267. |
| [4] | 张涛, 张贺, 杜雅欣, 展思辉. Co掺杂Mn2O3复合材料的构筑及活化过氧单硫酸盐降解医药废水[J]. 应用化学, 2024, 41(2): 268-278. |
| [5] | 陈淑敏, 吕子全, 邹旋, 桂水清, 卢雪梅. 新冠常态下功能性口罩研究进展[J]. 应用化学, 2023, 40(11): 1504-1517. |
| [6] | 于亚洲, 赵先亮, 谭宁宁, 肖洋, 张拥军. 聚烯丙胺改性磺酸催化剂催化苯甲酸正丁酯的合成[J]. 应用化学, 2023, 40(10): 1430-1436. |
| [7] | 梁锦辉, 梁乐程, 崔志明. 金属间化合物电催化剂在氢电转换器件应用中的研究进展[J]. 应用化学, 2023, 40(8): 1140-1157. |
| [8] | 王伟, 李家源. 电解水析氢反应磷化钴异质结催化剂的研究进展[J]. 应用化学, 2023, 40(8): 1175-1186. |
| [9] | 刘嘉欣, 范家禾, 李曙辉, 马亮. Rh@Pt内凹立方体核壳催化剂的制备及其乙醇电氧化性能[J]. 应用化学, 2023, 40(8): 1195-1204. |
| [10] | 武彦彬, 李利珍, 李俊华, 许志锋. 基于MoS2纳米花的表面离子印迹材料的制备及性能[J]. 应用化学, 2023, 40(7): 1024-1033. |
| [11] | 张毅城, 查飞, 唐小华, 常玥, 田海锋, 郭效军. 非均相催化制备有机过氧化物的研究进展[J]. 应用化学, 2023, 40(6): 769-788. |
| [12] | 于宜辰, 张雨宸, 张耀远, 吴芹, 史大昕, 陈康成, 黎汉生. 本体金属氧化物在丙烷无氧脱氢中的研究进展[J]. 应用化学, 2023, 40(6): 789-805. |
| [13] | 杨玉雯, 齐婧瑶, 李林, 楚国宁, 王赛, 张钰, 张爽. 磁性NiFe2O4负载Ru催化5-羟甲基糠醛选择性氧化合成2,5-呋喃二甲酸[J]. 应用化学, 2023, 40(6): 879-887. |
| [14] | 沈方燮, 万翔, 王卫超. 基于锰基YMn2O5催化剂室温降解挥发性有机化合污染物[J]. 应用化学, 2023, 40(6): 888-895. |
| [15] | 伍凡, 田贺元, 刘鹏, 孙立伟, 张一波, 杨向光. 高氧空位尖晶石型锰基催化剂用于低温NH3-SCR反应[J]. 应用化学, 2023, 40(5): 697-707. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||