| 1 |
WANG J, CHENG G F, ZHANG J H, et al. Feasibility and mechanism of recycling carbon resources from waste cyanobacteria and reducing microcystin toxicity by dielectric barrier discharge plasma[J]. J Hazard Mater, 2023, 460: 132333.
|
| 2 |
JETHVA S, BHABHOR F, PATIL C, et al. Studies of physio-chemical changes of dielectric barrier discharge plasma treated aramid fibers[J]. Vacuum, 2023, 215: 112313.
|
| 3 |
黑雪婷, 高远, 窦立广, 等. 纳秒脉冲介质阻挡放电等离子体驱动CH4-CH3OH转化制备液态化学品的特性研究[J]. 电工技术学报, 2022, 37(15): 3941-3950.
|
|
HEI X T, GAO Y, DOU L G, et al. Study on plasma enhanced CH4-CH3OH conversion to liquid chemicals by nanosecond pulsed dielectric barrier discharge[J]. Trans China Electrotech Soc, 2022, 37(15): 3941-3950.
|
| 4 |
WANG X J, XU P, YANG C Y, et al. Enhanced 4-FP removal with MnFe2O4 catalysts under dielectric barrier discharge plasma: economical synthesis, catalytic performance and degradation mechanism[J]. J Hazard Mater, 2021, 414: 125602.
|
| 5 |
刘鑫, 刘建奇, 陈佳尧, 等. 催化剂协同介质阻挡放电等离子体对不同VOCs的催化选择性[J]. 环境工程学报, 2022, 16(6): 1862-1871.
|
|
LIU X, LIU J Q, CHEN J Y, et al. Catalytic selectivity of catalyst in the degradation of mixed VOCs by dielectric barrier discharge plasma[J]. Chin J Environ Eng, 2022, 16(6): 1862-1871.
|
| 6 |
ZHANG A, ZHOU Y, LI Y, et al. Motivation of reactive oxygen and nitrogen species by a novel non-thermal plasma coupled with calcium peroxide system for synergistic removal of sulfamethoxazole in waste activated sludge[J]. Water Res, 2022, 212: 118128.
|
| 7 |
BUMROONGSAKULSAWAT P, KHONGTHON W, PAVARAJARN V. Degradation of diuron in water by electrochemical advanced oxidation in a microreactor: effects of anion contamination on degradation and toxicity[J]. J Environ Chem Eng, 2020, 8(4): 103824.
|
| 8 |
ANSARI M, SHARIFIAN M, EHRAMPOUSH M H, et al. Dielectric barrier discharge plasma with photocatalysts as a hybrid emerging technology for degradation of synthetic organic compounds in aqueous environments: a critical review[J]. Chemosphere, 2021, 263: 128065.
|
| 9 |
SHANG K F, LI W F, WANG X J, et al. Degradation of p-nitrophenol by DBD plasma/Fe2+/persulfate oxidation process[J]. Sep Purif Technol, 2019, 218: 106-112.
|
| 10 |
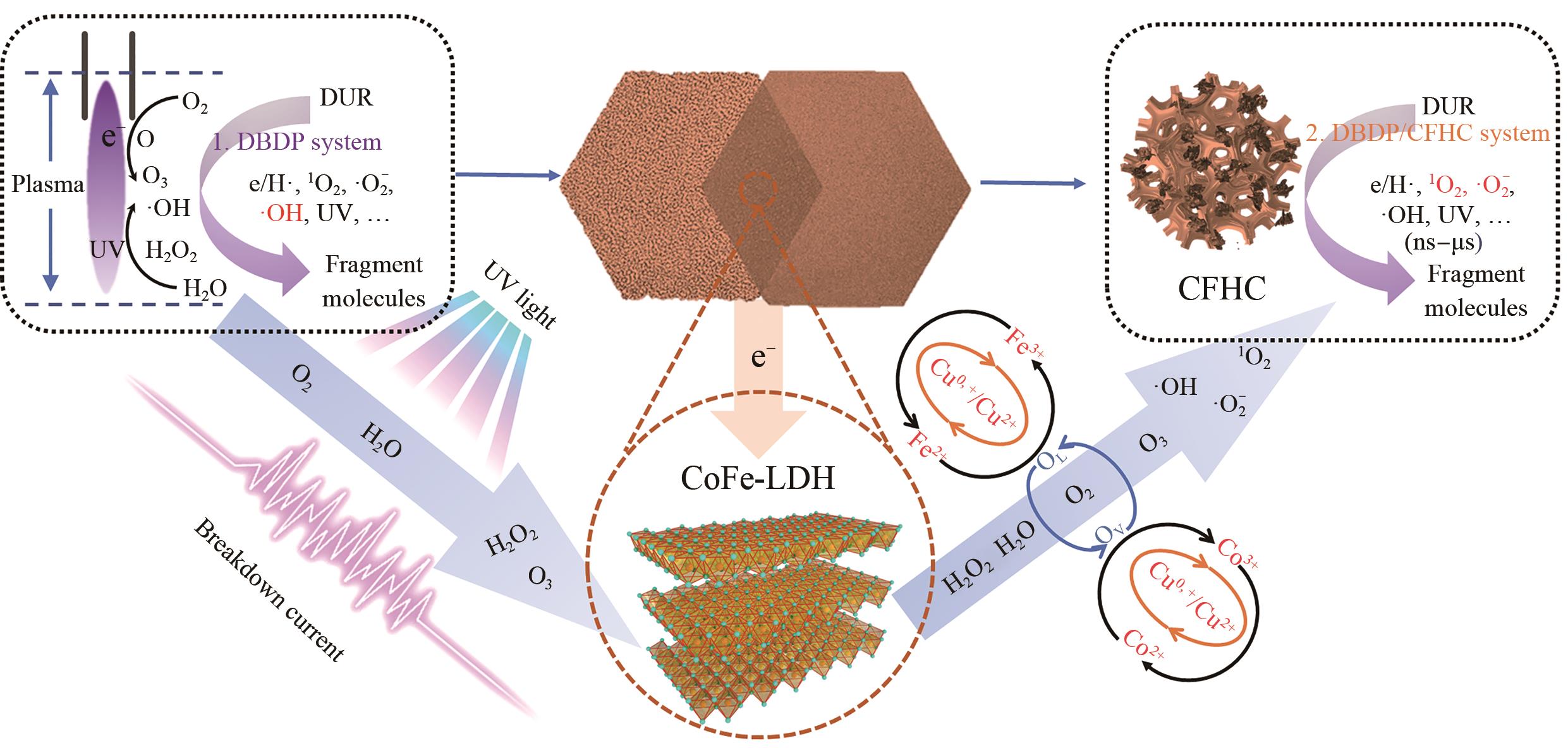
SHEN T Y, WANG X J, LI J Q, et al. Construction of 3D ternary layered double hydroxides on nickel foam for enhancing dielectric barrier discharge plasma to degrade DUR: performance, mechanism and energy efficiency[J]. Chem Eng J, 2023, 455: 140790.
|
| 11 |
GUO H, JIANG N, WANG H J, et al. Degradation of flumequine in water by pulsed discharge plasma coupled with reduced graphene oxide/TiO2 nanocomposites[J]. Sep Purif Technol, 2019, 218: 206-216.
|
| 12 |
YANG Z Z, ZHANG C, ZENG G, et al. Design and engineering of layered double hydroxide based catalysts for water depollution by advanced oxidation processes: a review[J]. J Mater Chem A, 2020, 8(8): 4141-4173.
|
| 13 |
ZENG H X, DENG L, SHI Z, et al. Heterogeneous degradation of carbamazepine by Prussian blue analogues in the interlayers of layered double hydroxides: performance, mechanism and toxicity evaluation[J]. J Mater Chem A, 2019, 7(1): 342-352.
|
| 14 |
WANG H, JING M M, WU Y, et al. Effective degradation of phenol via Fenton reaction over CuNiFe layered double hydroxides[J]. J Hazard Mater, 2018, 353: 53-61.
|
| 15 |
HU L M, ZHANG G S, LIU M, et al. Application of nickel foam-supported Co3O4-Bi2O3 as a heterogeneous catalyst for BPA removal by peroxymonosulfate activation[J]. Sci Total Environ, 2019, 647: 352-361.
|
| 16 |
WANG X J, XU P, YANG C Y, et al. Removal of 4-fluorophenol by dielectric barrier discharge plasma in three different structures: comparison, optimization and mechanism[J]. J Environ Chem Eng, 2021, 9(2): 105160.
|
| 17 |
HAO Y J, MA Y G, ZHANG X, et al. Unraveling the importance between electronic intensity and oxygen vacancy on photothermocatalytic toluene oxidation over CeO2[J]. Chem Eng J, 2022, 433: 134619.
|
| 18 |
ZHU K Y, SHI F, ZHU X F, et al. The roles of oxygen vacancies in electrocatalytic oxygen evolution reaction[J]. Nano Energy, 2020, 73: 104761.
|
| 19 |
WANG X Y, LI X Y, MU J C, et al. Oxygen vacancy-rich porous Co3O4 nanosheets toward boosted NO reduction by CO and CO oxidation: insights into the structure-activity relationship and performance enhancement mechanism[J]. ACS Appl Mater Interfaces, 2019, 11(45): 41988-41999.
|
| 20 |
YANG W C, XI D D, LI C F, et al. “In-situ synthesized” iron-based bimetal promotes efficient removal of Cr(Ⅵ) in by zero-valent iron-loaded hydroxyapatite[J]. J Hazard Mater, 2021, 420: 126540.
|
| 21 |
CAI Z, ZHOU D J, WANG M Y, et al. Introducing Fe2+ into nickel-iron layered double hydroxide: local structure modulated water oxidation activity[J]. Angew Chem Int Ed, 2018, 57(30): 9392-9396.
|
| 22 |
ZHANG Y T, LIU C, XU B B, et al. Degradation of benzotriazole by a novel Fenton-like reaction with mesoporous Cu/MnO2: combination of adsorption and catalysis oxidation[J]. Appl Catal B: Environ, 2016, 199: 447-457.
|
| 23 |
孙立智, 吕浩, 闵晓文, 等. 介孔钯-硼合金纳米颗粒的制备和甲醇氧化电催化性能[J]. 应用化学, 2022, 39(4): 673-691.
|
|
SUN L Z, LV H, MIN X W, et al. Mesoporous palladium boron alloy nanocatalysts:synthesis and performance in methanol oxidation electrocatalysis[J]. Chin J Appl Chem, 2022, 39(4): 673-691.
|
| 24 |
SHEN T Y, WANG X J, LI J Q, et al. Introduction of oxygen vacancy to Bi2Mn4O10 supported by nickel foam for 1O2 dominated metronidazole degradation under dielectric barrier discharge plasma[J]. Appl Catal B: Environ, 2023, 328: 122518.
|
| 25 |
MAHYAR A, MIESSNER H, MUELLER S, et al. Development and application of different non-thermal plasma reactors for the removal of perfluorosurfactants in water: a comparative study[J]. Plasma Chem Plasma P, 2019, 39(3): 531-544.
|
| 26 |
DONG C, QU Z P, JIANG X, et al. Tuning oxygen vacancy concentration of MnO2 through metal doping for improved toluene oxidation[J]. J Hazard Mater, 2020, 391: 122181.
|
| 27 |
XU P, LI X, WEI R, et al. High adaptability and stability FeCo2O4/diatomite composite for efficient peroxymonosulfate activation: performance, water matrix impact, and mechanism[J]. Chem Eng J, 2023, 462: 142204.
|
| 28 |
YAO X S, LIN Q T, ZENG L Z, et al. Degradation of humic acid using hydrogen peroxide activated by CuO-Co3O4@AC under microwave irradiation[J]. Chem Eng J, 2017, 330: 783-791
|
| 29 |
YAN Y T, ZHANG H Y, WANG W, et al. Synthesis of Fe0/Fe3O4@porous carbon through a facile heat treatment of iron-containing candle soots for peroxymonosulfate activation and efficient degradation of sulfamethoxazole[J]. J Hazard Mater, 2021, 411: 124952.
|
| 30 |
LEE H, LEE H J, SEO J, et al. Activation of oxygen and hydrogen peroxide by copper(Ⅱ) coupled with hydroxylamine for oxidation of organic contaminants[J]. Environ Sci Technol, 2016, 50(15): 8231-8238.
|

 ), 王鹏1(
), 王鹏1( )
)
 ), Peng WANG1(
), Peng WANG1( )
)