
应用化学 ›› 2024, Vol. 41 ›› Issue (2): 217-229.DOI: 10.19894/j.issn.1000-0518.230310
• 综合评述 • 上一篇
单原子催化剂在类芬顿水处理领域的研究进展
孙天礼1, 朱国1, 何海1, 黄炳坤2, 熊兆锟2( ), 赖波2(
), 赖波2( )
)
- 1.中国石化西南油气分公司采气二厂,阆中 637400
2.四川大学建筑与环境学院,中德水环境与健康研究中心,成都 610065
-
收稿日期:2023-10-10接受日期:2023-12-24出版日期:2024-02-01发布日期:2024-03-05 -
通讯作者:熊兆锟,赖波 -
基金资助:国家自然科学基金(52200105);四川省自然科学基金(2023NSFSC0344)
Research Prospect of Single-Atom Catalysts for Fenton-Like Water Treatment
Tian-Li SUN1, Guo ZHU1, Hai HE1, Bing-Kun HUANG2, Zhao-Kun XIONG2( ), Bo LAI2(
), Bo LAI2( )
)
- 1.The Second Gas Production Plant of Southwest Branch,Sinopec,Langzhong 637400,China
2.Sino-German Centre for Water and Health Research,College of Architecture and Environment,Sichuan University,Chengdu 610065,China
-
Received:2023-10-10Accepted:2023-12-24Published:2024-02-01Online:2024-03-05 -
Contact:Zhao-Kun XIONG,Bo LAI -
About author:laibo@scu.edu.cn
scuxzk@scu.edu.cn;
-
Supported by:the National Natural Science Foundation of China(52200105);the Natural Science Foundation of Sichuan Province(2023NSFSC0344)
摘要:
单原子催化剂(SACs)以其独特的结构和特性,特别是在最大限度地利用原子和提高内在催化活性方面受到了广泛的关注。 近年来,基于SACs的高级氧化技术(AOPs)已成为水污染控制研究中的一个新兴领域,广泛应用于去除各种难降解有机污染物。 本文分析了SACs的合成及表征方法,着重介绍了SACs在不同类芬顿催化反应中的性能及其作用机理。 此外,还介绍了将SACs固定在膜状或柱状过滤器上连续流的测试,以探究SACs在类芬顿反应中实际应用的潜力。 最后,对合理的SACs设计、类芬顿反应机理探索等方向进行了展望,旨在提高SACs应用于类芬顿反应在实际废水处理中的应用潜力。
中图分类号:
引用本文
孙天礼, 朱国, 何海, 黄炳坤, 熊兆锟, 赖波. 单原子催化剂在类芬顿水处理领域的研究进展[J]. 应用化学, 2024, 41(2): 217-229.
Tian-Li SUN, Guo ZHU, Hai HE, Bing-Kun HUANG, Zhao-Kun XIONG, Bo LAI. Research Prospect of Single-Atom Catalysts for Fenton-Like Water Treatment[J]. Chinese Journal of Applied Chemistry, 2024, 41(2): 217-229.

图1 (a) Pd-纳米颗粒@ZIF-8、中间物Ⅰ、中间物Ⅱ和Pd单原子的高角环形暗场像-扫描透射电子显微镜(HAADF-STEM)图像和高分辨率HAADF-STEM图像(插图)[24];(b) ALD技术在石墨烯纳米片上沉积Pt的示意图[27]
Fig.1 (a) High-angle annular dark field scanning transmission electron microscopy (HAADF-STEM) images and high-resolution HAADF-STEM images (insets) of Pd-nanoparticles@ZIF-8, intermediate Ⅰ, intermediate Ⅱ and Pd single atoms[24]; (b) Schematic illustrations of Pt ALD mechanism on graphene nanosheets[27]

图2 (a)通过SAS-Fe观测到的HAADF-STEM。 比例尺: 2 nm[29]; (b) FeSA-MNC、Fe箔、Fe2O3和FePc的EXAFS[30]
Fig.2 (a) HAADF-STEM obtained from SAS-Fe. Scale bar: 2 nm[29]; (b) EXAFS of FeSA-MNC, Fe foil, Fe2O3 and FePc[30]
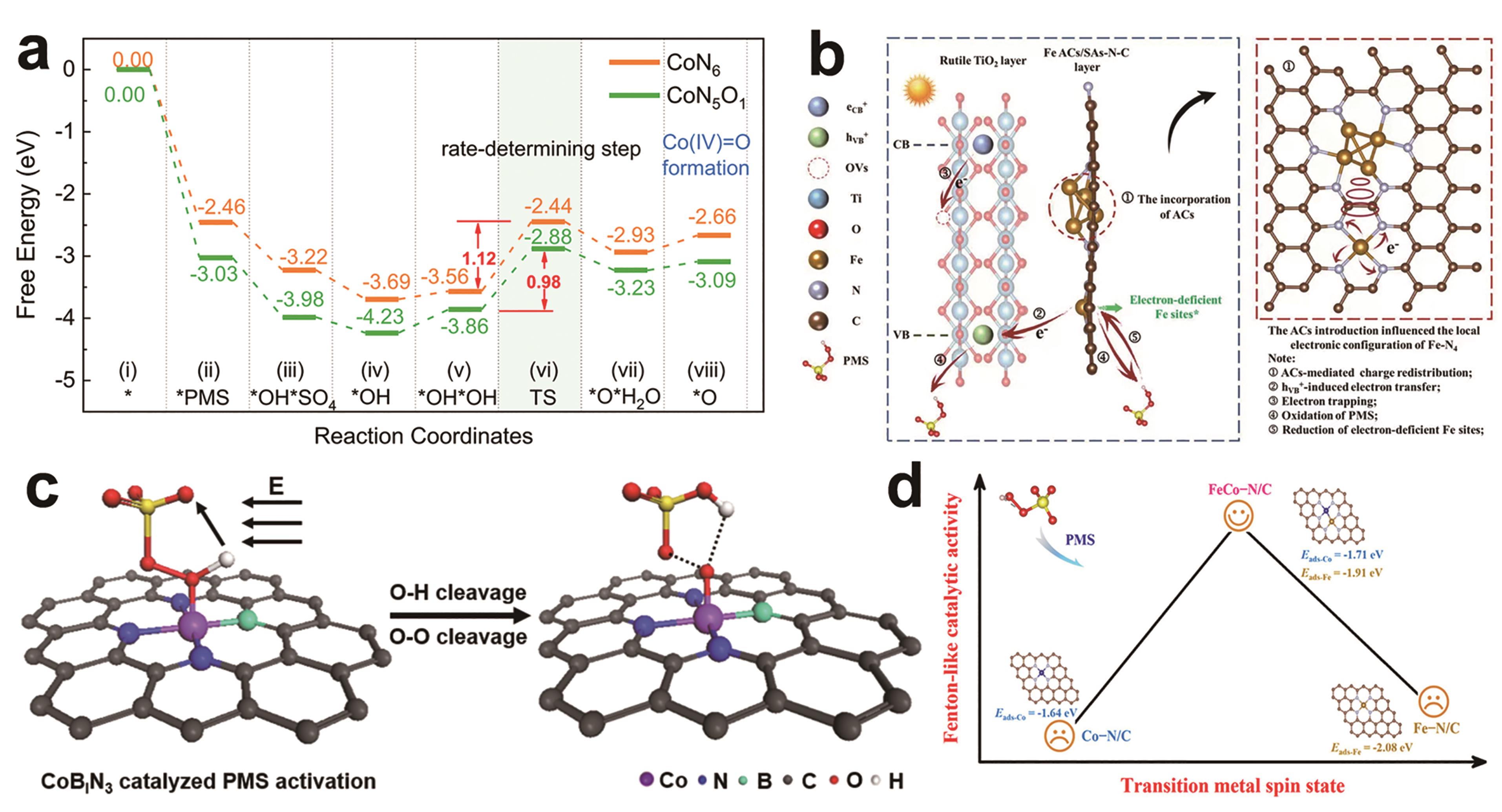
图3 (a) CoN6/PMS和CoN5O1/PMS体系Co(Ⅳ)O生成的能量谱,*O表示Co(Ⅳ)O的反应种类[36]; (b)提议的Vis/TiFeAS/PMS体系的反应机制[37]; (c)局部电场诱导耦合电子-质子转移过程促进高价金属转化示意图[38]; (d)过渡金属自旋态与类芬顿催化活性的关系[39]
Fig.3 (a) Energy profiles of Co(Ⅳ)O formation for CoN6/PMS and CoN5O1/PMS systems; *O represents the reactive species of Co(?Ⅳ??)?O[36]; (b) The proposed reaction mechanisms of Vis/TiFeAS/PMS system[37]; (c) Schematic illustration of a local electric field-induced coupled electron-proton transfer process promoting the conversion of high-valent metal-oxo species[38]; (d) Relationship between spin states and Fenton-like catalytic activity of transition metals[39]
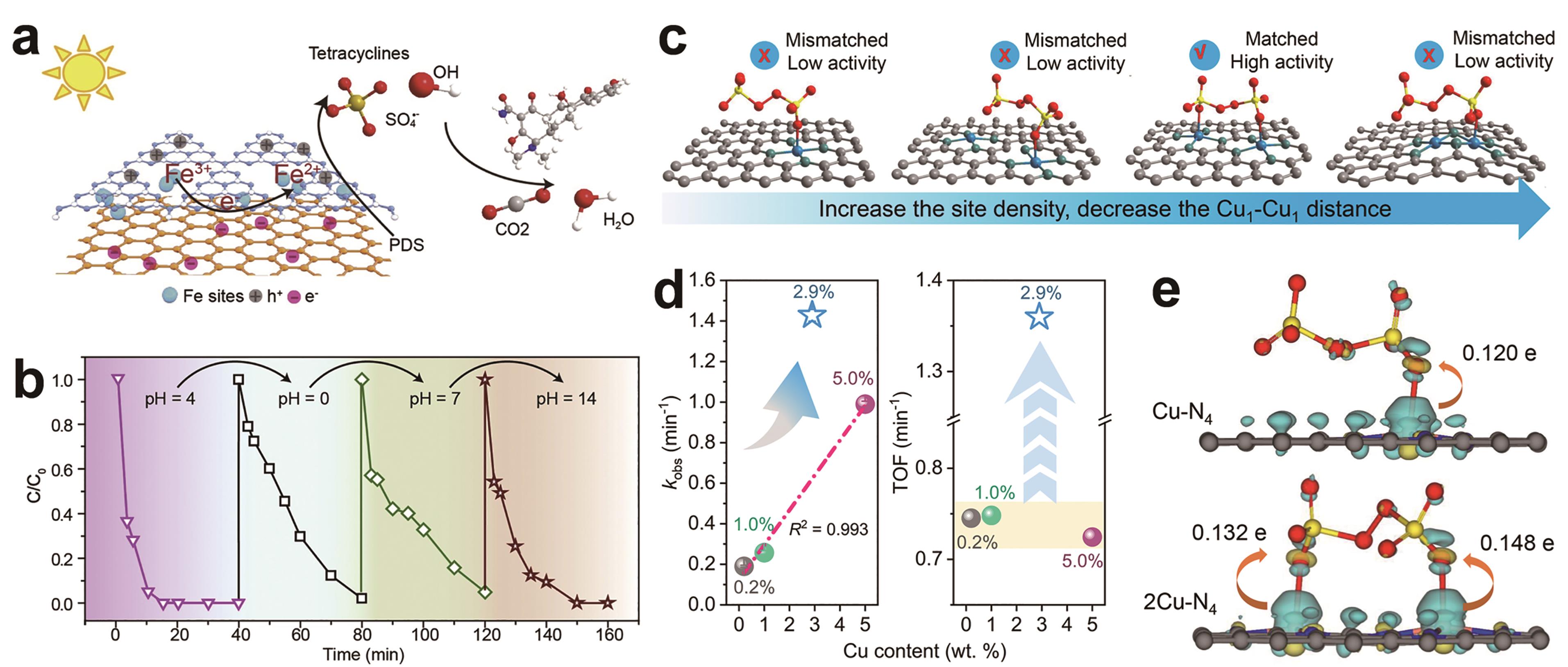
图4 (a) C3N4-Fe-rGO/PDS类芬顿反应机理的示意图[4]; (b)以C3N4-Fe-rGO为催化剂,考察了不同pH值条件下溶液的循环降解性能[4]; (c) Cu1-Cu1距离对PDS吸附和活化的影响[41]; (d)不同Cu位密度的Cu1/NG活化PDS去除BPA的一级速率常数和TOF[41]; (e) PDS吸附在2Cu-N4和Cu-N4的电子密度差及相应的电荷转移。 黄色和青色轮廓分别代表电子积累和缺失[41]
Fig.4 (a) Scheme diagram proposed for the Fenton-like reaction mechanism of C3N4-Fe-rGO/PDS[4]?; (b) The degradation performance during cycles of solution with different pH conditions, with C3N4-Fe-rGO as the catalyst[4]; (c) Depiction of Cu1-Cu1 distance manipulation for PDS adsorption and activation[41]; (d) First-order rate constants and TOFs of BPA removal by Cu1/NG with different Cu site densities activating PDS[41]??; (e) Electron density difference for PDS adsorption on 2Cu-N4 and Cu-N4 and the corresponding charge transfer. Yellow and cyan contours stand for electron accumulation and deletion, respectively[41]

图5 (a)芬顿过滤器的图像及过滤介质的SEM图像。 比例尺: 100 μm。 内页: 涂在碳纤维表面的Cu-C3N4催化剂放大的SEM图像。 比例尺: 5 μm[44]; (b)染料去除率和废水中的铜浓度与过滤时间的关系[44]; (c) FeN5和FeN4模型的差分电荷图。 黄色和天蓝色区域分别代表电子积累和电子消耗[45]; (d) FeN5和FeN4模型反应过程能垒图[45]; (e) FeN5/NG+H2O2、FeN4/NG+H2O2和常规均相Fenton对不同有机污染物的降解[45]
Fig.5 (a) Photo and SEM image of the filter medium. Scale bar: 100 μm. Inset, magnified SEM image showing the Cu-C3N4 catalyst coated on the surface of a carbon fibre. Scale bar: 5 μm[44]?; (b) Dye removal and Cu concentration in effluent as functions of filtration time[44]?; (c) Charge density differences of FeN5 and FeN4 models. The yellow and skyblue regions represent electron accumulation and electron depletion, respectively[45]; (d) Energy diagram of the reaction process for FeN5 and FeN4 models[45]?; (e) Degradation of select organic pollutants in FeN4/NG+H2O2, FeN5/NG+H2O2, conventional homogeneous Fenton (Fe2++H2O2) and control (H2O2) systems[45]

图6 (a) g-C3N4和FeCN的表观速率常数比较(插图: 总有机碳(TOC)结果)[49]; (b) CH3COO?-TEMPO全扫描色谱图[49]; (c) FeCN+PAA体系中18O标记或未标记PMSO2的质谱分析[49]; (d)不同活化剂活化IO4-降解4-CP[50]; (e)溶液初始pH值对N-rGO-CoSA/IO4-体系中4-CP降解的影响[50]; (f)在N-rGO-CoSA粉末包覆的工作电极上依次加入IO4-和4-CP后的电流响应。 插图显示了N-rGO-CoSA和N-rGO-800电极在电解质溶液中的EIS谱图[50]; (g) Fe5-NC/O3体系中底物依赖性示意图[51]; (h) AIMD模拟的动态过程[51]; (i) DFT计算的O3与Fe-N4位点相互作用的相对能量分布[51]
Fig.6 (a) Comparison between the apparent rate constants of g-C3N4 and FeCN (inset: TOC result)?[49]; (b) Full-scan chromatogram of CH3COO?-TEMPO[49]; (c) Mass spectral analyses of the 18O-labeled or unlabeled PMSO2 generated in the FeCN+PAA system[49]; (d) Degradation of 4-CP by IO4- activated using different activators[50]; (e) Initial solution pH on the degradation of 4-CP in the N-rGO-CoSA IO4- system[50]; (f) Current response after the sequential addition of IO4- and 4-CP at the working electrode coated with the N-rGO-CoSA powder. The inset shows the EIS profiles of the N-rGO-CoSA and N-rGO-800 electrodes in the electrolyte solution[50]; (g) Substrate dependence diagram in Fe5-NC/O3 system[51]; (h) Dynamic processes simulated by AIMD[51]; (i) Relative energy profile calculated by DFT in the interaction of O3 and the Fe-N4 site[51]
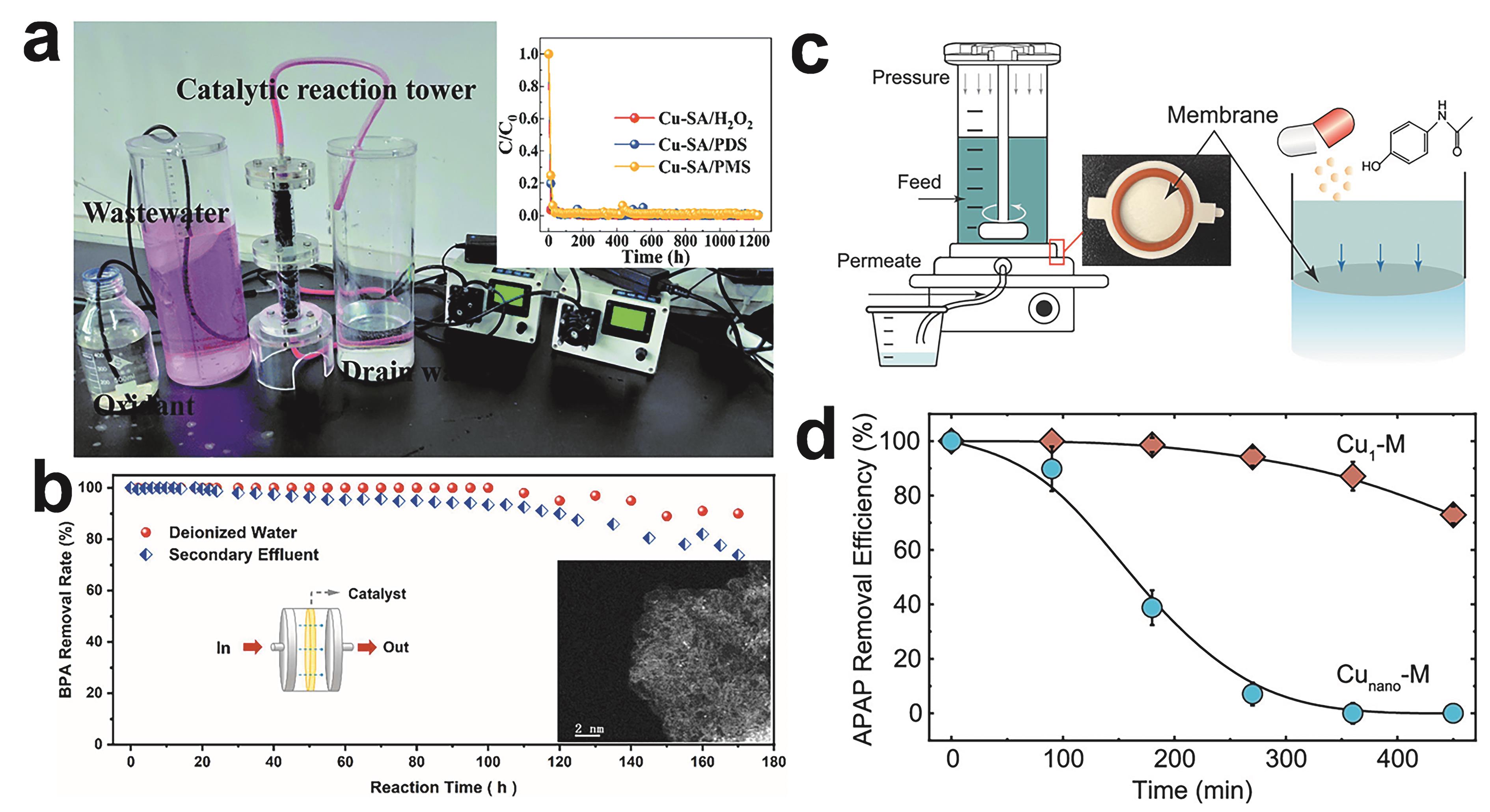
图7 (a) Cu-SA的连续流装置及效果[55]; (b) Fe-CNW3包覆膜过滤器连续流去去离子水和除污水处理厂二级出水中的BPA,插图为Fe-CNW3反应后的HADDF-STEM图像[59]; (c)安装了有效面积为3.1 cm2的催化膜和用于去除微污染物的终端过滤池的示意图和照片[60]; (d)过滤过程中催化膜对APAP的去除效率[60]
Fig.7 (a) Pilot device and effect of Cu-SA in continuous flow unit[55]; (b) BPA removal in deionized water and secondary effluent of WWTP with Fe-CNW3 coated membrane filter under continuous flow. Inset is the HADDF-STEM image of Fe-CNW3 after the reaction[59]; (c) Schematic and photograph of the catalytic membrane (effective area of 3.1 cm2) are installed and the dead-end filtration cell for micropollutant removal. APAP is selected as a model micropollutant[60]; (c) APAP removal efficiency by the catalytic membranes during filtration[60]
| 1 | HUANG L Z, WEI X, GAO E, et al. Single Fe atoms confined in two-dimensional MoS2 for sulfite activation: a biomimetic approach towards efficient radical generation[J]. Appl Catal B: Environ, 2020, 268: 118459. |
| 2 | HUANG B, XIONG Z, ZHOU P, et al. Ultrafast degradation of contaminants in a trace cobalt(Ⅱ) activated peroxymonosulfate process triggered through borate: indispensable role of intermediate complex[J]. J Hazard Mater, 2022, 424: 127641. |
| 3 | CHENG Y, HE S, LU S, et al. Iron single atoms on graphene as nonprecious metal catalysts for high-temperature polymer electrolyte membrane fuel cells[J]. Adv Sci, 2019, 6(10): 1802066. |
| 4 | ZUO S, JIN X, WANG X, et al. Sandwich structure stabilized atomic Fe catalyst for highly efficient Fenton-like reaction at all pH values[J]. Appl Catal B: Environ, 2021, 282: 119551. |
| 5 | QIAO B, WANG A, YANG X, et al. Single-atom catalysis of CO oxidation using Pt1/FeOx[J]. Nat Chem, 2011, 3(8): 634-641. |
| 6 | 曹蓉, 夏杰桢, 廖漫华, 等. 单原子催化剂在电化学合成氨中的理论研究进展[J]. 应用化学, 2023, 40(1): 9-23. |
| CAO R, XIA J Z, LIAO M H, et al. Theoretical research progress of single atom catalysts in electrochemical synthesis of ammonia[J]. Chin J Appl Chem, 2023, 40(1): 9-23. | |
| 7 | 张超. 单原子催化剂电催化还原二氧化碳研究进展[J]. 应用化学, 2022, 39(6): 871-887. |
| ZHANG C. Research prospect of single atom catalysts towards electrocatalytic reduction of carbon dioxide[J]. Chin J Appl Chem, 2022, 39(6): 871-887. | |
| 8 | 王欣, 张冬, 杜菲. 单原子催化剂在锂硫电池中的研究进展[J]. 应用化学, 2022, 39(4): 513-527. |
| WANG X, ZHANG D, DU F. Recent progress of single⁃atom catalytic materials for lithium sulfur batteries[J]. Chin J Appl Chem, 2022, 39(4): 513-527. | |
| 9 | WEON S, HUANG D, RIGBY K, et al. Environmental materials beyond and below the nanoscale: single-atom catalysts[J]. ACS EST Eng, 2020, 1(2): 157-172. |
| 10 | WU X, KIM J. Outlook on single atom catalysts for persulfate-based advanced oxidation[J]. ACS EST Eng, 2022, 2(10): 1776-1796. |
| 11 | HUANG B, WU Z, ZHOU H, et al. Recent advances in single-atom catalysts for advanced oxidation processes in water purification[J]. J Hazard Mater, 2021, 412: 125253. |
| 12 | LI W H, YANG J, WANG D, et al. Striding the threshold of an atom era of organic synthesis by single-atom catalysis[J]. Chem, 2022, 8(1): 119-140. |
| 13 | YAN H, SU C, HE J, et al. Single-atom catalysts and their applications in organic chemistry[J]. J Mater Chem A, 2018, 6(19): 8793-8814. |
| 14 | GIANNAKAKIS G, MITCHELL S, PÉREZ-RAMíREZ J. Single-atom heterogeneous catalysts for sustainable organic synthesis[J]. Trends Chem, 2022, 4(4): 264-276. |
| 15 | LIU Y, YAO M, HAN W, et al. Construction of a single-atom nanozyme for enhanced chemodynamic therapy and chemotherapy[J]. Chem Eur J, 2021, 27(53): 13418-13425. |
| 16 | LU X, GAO S, LIN H, et al. Bioinspired copper single-atom catalysts for tumor parallel catalytic therapy[J]. Adv Mater, 2020, 32(36): 2002246. |
| 17 | LIANG J, JOHANNESSEN B, WU Z, et al. Regulating the coordination environment of mesopore-confined single atoms from metalloprotein-MOFs for highly efficient biocatalysis[J]. Adv Mater, 2022, 34(44): 2205674. |
| 18 | JIAO L, YAN H, WU Y, et al. When nanozymes meet single-atom catalysis[J]. Angew Chem Int Ed, 2020, 59(7): 2565-2576. |
| 19 | DOHERTY F, WANG H, YANG M, et al. Nanocluster and single-atom catalysts for thermocatalytic conversion of CO and CO2[J]. Catal Sci Technol, 2020, 10(17): 5772-5791. |
| 20 | LIU L, CORMA A. Metal catalysts for heterogeneous catalysis: from single atoms to nanoclusters and nanoparticles[J]. Chem Rev, 2018, 118(10): 4981-5079. |
| 21 | ZHANG H, TIAN W, DUAN X, et al. Catalysis of a single transition metal site for water oxidation: from mononuclear molecules to single atoms[J]. Adv Mater, 2020, 32(18): 1904037. |
| 22 | REN W, CHENG C, SHAO P, et al. Origins of electron-transfer regime in persulfate-based nonradical oxidation processes[J]. Environ Sci Technol, 2022, 56(1): 78-97. |
| 23 | SHANG Y, XU X, GAO B, et al. Single-atom catalysis in advanced oxidation processes for environmental remediation[J]. Chem Soc Rev, 2021, 50(8): 5281-322. |
| 24 | WEI S, LI A, LIU J C, et al. Direct observation of noble metal nanoparticles transforming to thermally stable single atoms[J]. Nat Nanotechnol, 2018, 13(9): 856-861. |
| 25 | YIN P, YAO T, WU Y, et al. Single cobalt atoms with precise N-coordination as superior oxygen reduction reaction catalysts[J]. Angew Chem Int Ed, 2016, 55(36): 10800. |
| 26 | HAN A, WANG B, KUMAR A, et al. Recent advances for MOF-derived carbon-supported single-atom catalysts[J]. Small Methods, 2019, 3(9): 1800471. |
| 27 | SUN S, ZHANG G, GAUQUELIN N, et al. Single-atom catalysis using Pt/graphene achieved through atomic layer deposition[J]. Sci Rep, 2013, 3(1): 1775. |
| 28 | DUAN S, WANG R, LIU J. Stability investigation of a high number density Pt1/Fe2O3 single-atom catalyst under different gas environments by HAADF-STEM[J]. Nanotechnology, 2018, 29(20): 204002. |
| 29 | XIONG Y, SUN W, XIN P, et al. Gram-scale synthesis of high-loading single-atomic-site Fe catalysts for effective epoxidation of styrene[J]. Adv Mater, 2020: e2000896. |
| 30 | HUANG B, WU Z, WANG X, et al. Coupled surface-confinement effect and pore engineering in a single-Fe-atom catalyst for ultrafast Fenton-like reaction with high-valent iron-oxo complex oxidation[J]. Environ Sci Technol, 2023, 57(41): 15667-15679. |
| 31 | LI X, CAO C S, HUNG S F, et al. Identification of the electronic and structural dynamics of catalytic centers in single-Fe-atom material[J]. Chem, 2020, 6(12): 3440-3454. |
| 32 | HUANG B, REN X, ZHAO J, et al. Modulating electronic structure engineering of atomically dispersed cobalt catalyst in Fenton-like reaction for efficient degradation of organic pollutants[J]. Environ Sci Technol, 2023, 57(37): 14071-14081. |
| 33 | LAI L, YAN J, LI J, et al. Co/Al2O3-EPM as peroxymonosulfate activator for sulfamethoxazole removal: performance, biotoxicity, degradation pathways and mechanism[J]. Chem Eng J, 2018, 343: 676-688. |
| 34 | XIONG Z, JIANG Y, WU Z, et al. Synthesis strategies and emerging mechanisms of metal-organic frameworks for sulfate radical-based advanced oxidation process: a review[J]. Chem Eng J, 2021, 421: 127863. |
| 35 | LI X, HUANG X, XI S, et al. Single cobalt atoms anchored on porous N-doped graphene with dual reaction sites for efficient Fenton-like catalysis[J]. J Am Chem Soc, 2018, 140(39): 12469-12475. |
| 36 | WU Q Y, YANG Z W, WANG Z W, et al. Oxygen doping of cobalt-single-atom coordination enhances peroxymonosulfate activation and high-valent cobalt-oxo species formation[J]. Proc Natl Acad Sci USA, 2023, 120(16): e2219923120. |
| 37 | MO F, SONG C, ZHOU Q, et al. The optimized Fenton-like activity of Fe single-atom sites by Fe atomic clusters-mediated electronic configuration modulation[J]. Proc Natl Acad Sci USA, 2023, 120(15): e2300281120. |
| 38 | SONG J, HOU N, LIU X, et al. Asymmetrically coordinated CoB1N3 moieties for selective generation of high-valent co-oxo species via coupled electron-proton transfer in Fenton-like reactions[J]. Adv Mater, 2023: e2209552. |
| 39 | ZHAO Z, HU M, NIE T, et al. Improved electronic structure from spin-state reconstruction of a heteronuclear Fe-Co diatomic pair to boost the Fenton-like reaction[J]. Environ Sci Technol, 2023, 57(11): 4556-4567. |
| 40 | JIANG N, XU H, WANG L, et al. Nonradical oxidation of pollutants with single-atom-Fe(Ⅲ)-activated persulfate: Fe(V) being the possible intermediate oxidant[J]. Environ Sci Technol, 2020, 54(21): 14057-14065. |
| 41 | WANG B, CHENG C, JIN M, et al. A site distance effect induced by reactant molecule matchup in single-atom catalysts for Fenton-like reactions[J]. Angew Chem Int Ed Engl, 2022, 61(33): e202207268. |
| 42 | FU H, WEI J, CHEN G, et al. Axial coordination tuning Fe single-atom catalysts for boosting H2O2 activation[J]. Appl Catal B: Environ, 2023, 321: 122012. |
| 43 | ZHANG C, BAI L, CHEN M, et al. Modulating the site density of Mo single atoms to catch adventitious O atoms for efficient H2O2 oxidation with light[J]. Adv Mater, 2023, 35(5): e2208704. |
| 44 | XU J, ZHENG X, FENG Z, et al. Organic wastewater treatment by a single-atom catalyst and electrolytically produced H2O2[J]. Nat Sustainable, 2021, 4: 233-241. |
| 45 | FU H, WEI J, CHEN G, et al. Axial coordination tuning Fe single-atom catalysts for boosting H2O2 activation[J]. Appl Catal B, 2023, 321: 122012. |
| 46 | AN S, ZHANG G, WANG T, et al. High-density ultra-small clusters and single-atom Fe sites embedded in graphitic carbon nitride (g-C3N4) for highly efficient catalytic advanced oxidation processes[J]. ACS Nano, 2018, 12(9): 9441-9450. |
| 47 | ZHOU P, ZHANG J, XIONG Z, et al. C60 Fullerol promoted Fe(Ⅲ)/H2O2 Fenton oxidation: role of photosensitive Fe(Ⅲ)-Fullerol complex[J]. Appl Catal B: Environ, 2020, 265: 118264. |
| 48 | SHENG B, DENG C, LI Y, et al. In situ hydroxylation of a single-atom iron catalyst for preferential 1O2 production from H2O2[J]. ACS Catal, 2022, 12(23): 14679-14688. |
| 49 | CHEN F, LIU L L, WU J H, et al. Single-atom iron anchored tubular g-C3N4 catalysts for ultrafast Fenton-like reaction: roles of high-valency iron-oxo species and organic radicals[J]. Adv Mater, 2022, 34(31): 2202891. |
| 50 | LONG Y, DAI J, ZHAO S, et al. Atomically dispersed cobalt sites on graphene as efficient periodate activators for selective organic pollutant degradation[J]. Environ Sci Technol, 2021, 55: 5357-5370. |
| 51 | REN T, YIN M, CHEN S, et al. Single-atom Fe-N4 sites for catalytic ozonation to selectively induce a nonradical pathway toward wastewater purification[J]. Environ Sci Technol, 2023, 57(9): 3623-3633. |
| 52 | GUO Z, XIE Y, XIAO J, et al. Single-atom Mn-N4 site-catalyzed peroxone reaction for the efficient production of hydroxyl radicals in an acidic solution[J]. J Am Chem Soc, 2019, 141(30): 12005-12010. |
| 53 | LAN S, JING B, YU C, et al. Protrudent iron single-atom accelerated interfacial piezoelectric polarization for self-powered water motion triggered Fenton-like reaction[J]. Small, 2022, 18(2): 2105279. |
| 54 | WANG Z, ALMATRAFI E, WANG H, et al. Cobalt single atoms anchored on oxygen-doped tubular carbon nitride for efficient peroxymonosulfate activation: simultaneous coordination structure and morphology modulation[J]. Angew Chem Int Ed, 2022, 61(29): e202202338. |
| 55 | ZUO S, GUAN Z, YANG F, et al. Reactive oxygen species regulation and synergistic effect for effective water purification through Fenton-like catalysis on single-atom Cu-N sites[J]. J Mater Chem A, 2022, 10(19): 10503-10513. |
| 56 | LIANG X, WANG D, ZHAO Z, et al. Coordination number dependent catalytic activity of single-atom cobalt catalysts for Fenton-like reaction[J]. Adv Funct Mater, 2022, 32: 2203001. |
| 57 | CHU C, YANG J, ZHOU X, et al. Cobalt single atoms on tetrapyridomacrocyclic support for efficient peroxymonosulfate activation[J]. Environ Sci Technol, 2021, 55(2): 1242-1250. |
| 58 | WU X, RIGBY K, HUANG D, et al. Single-atom cobalt incorporated in a 2D graphene oxide membrane for catalytic pollutant degradation[J]. Environ Sci Technol, 2022, 56(2): 1341-1351. |
| 59 | ZHANG Y, CHEN X, LIANG C, et al. Reconstructing the coordination environment of single atomic Fe-catalysts for boosting the Fenton-like degradation activities[J]. Appl Catal B: Environ, 2022, 315: 121536. |
| 60 | MA W, SUN M, HUANG D, et al. Catalytic membrane with copper single-atom catalysts for effective hydrogen peroxide activation and pollutant destruction[J]. Environ Sci Technol, 2022: 8733-8745. |
| 61 | YANG M, HOU Z, ZHANG X, et al. Unveiling the origins of selective oxidation in single-atom catalysis via Co-N4-C intensified radical and nonradical pathways[J]. Environ Sci Technol, 2022, 56(16): 11635-11645. |
| [1] | 曹蓉, 夏杰桢, 廖漫华, 赵路超, 赵晨, 吴琪. 单原子催化剂在电化学合成氨中的理论研究进展[J]. 应用化学, 2023, 40(1): 9-23. |
| [2] | 王显, 杨小龙, 马荣鹏, 刘长鹏, 葛君杰, 邢巍. 单原子分散的Ir-N-C燃料电池阳极抗中毒催化剂[J]. 应用化学, 2022, 39(8): 1202-1208. |
| [3] | 张超. 单原子催化剂电催化还原二氧化碳研究进展[J]. 应用化学, 2022, 39(6): 871-887. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||