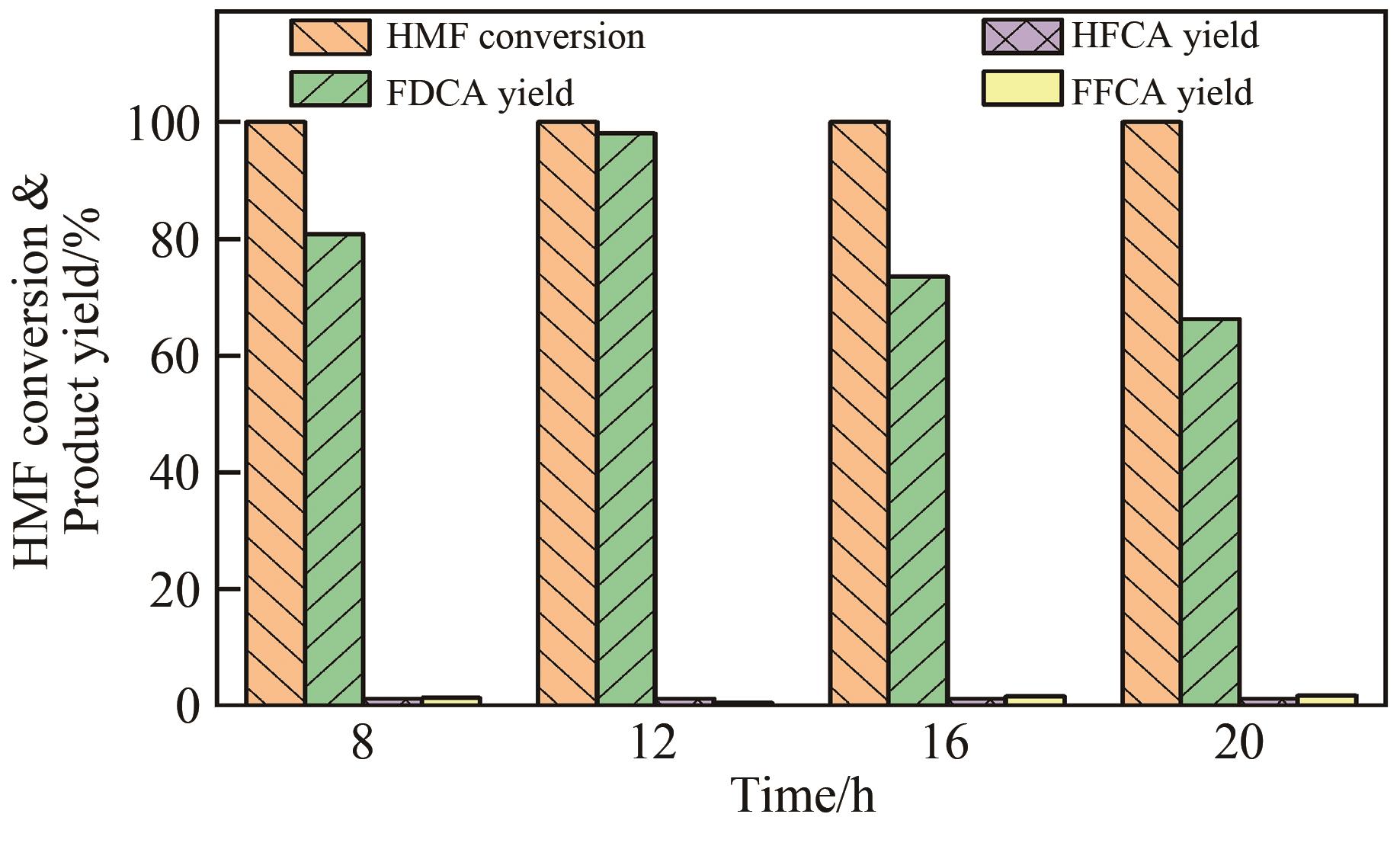
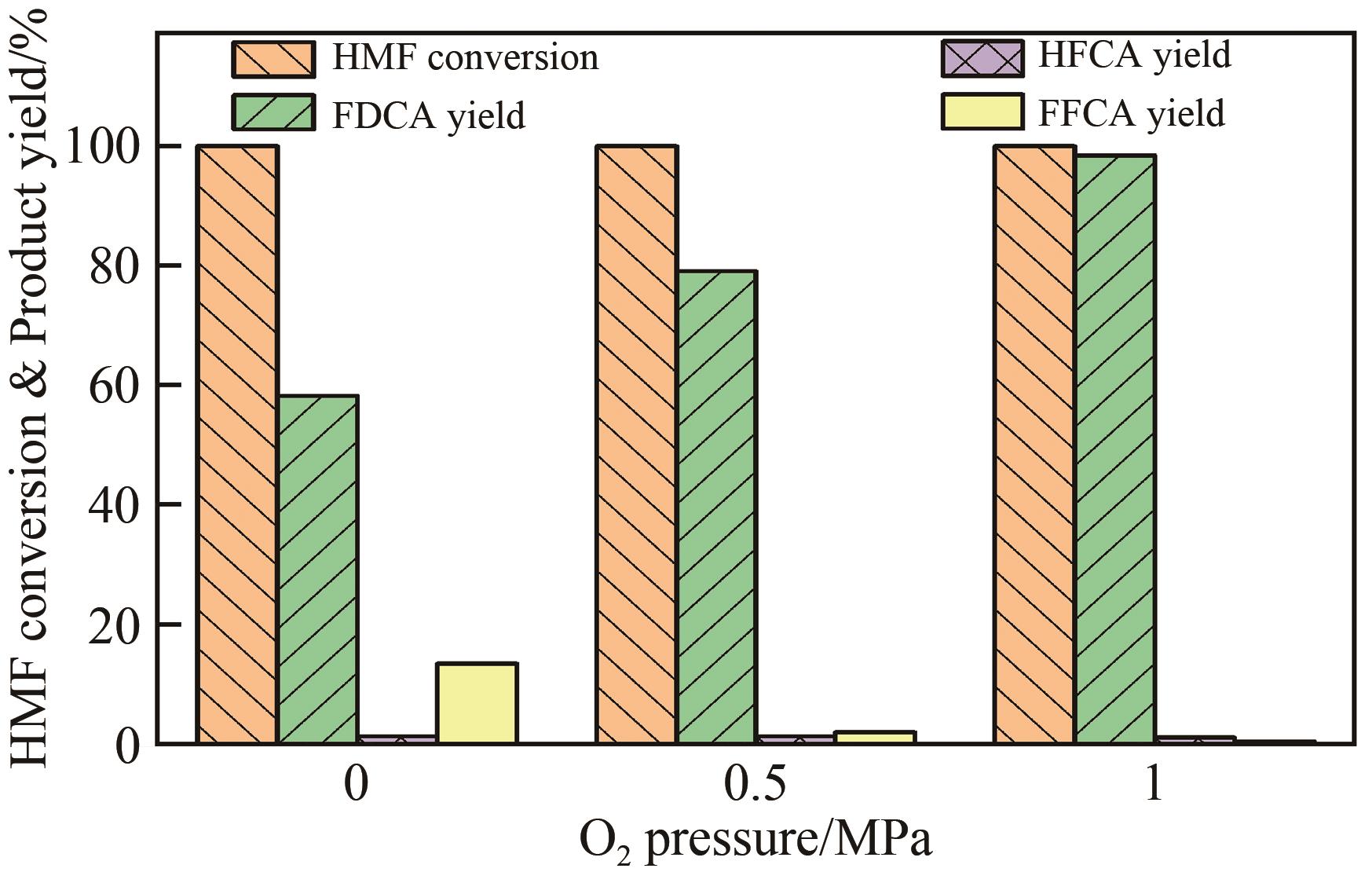
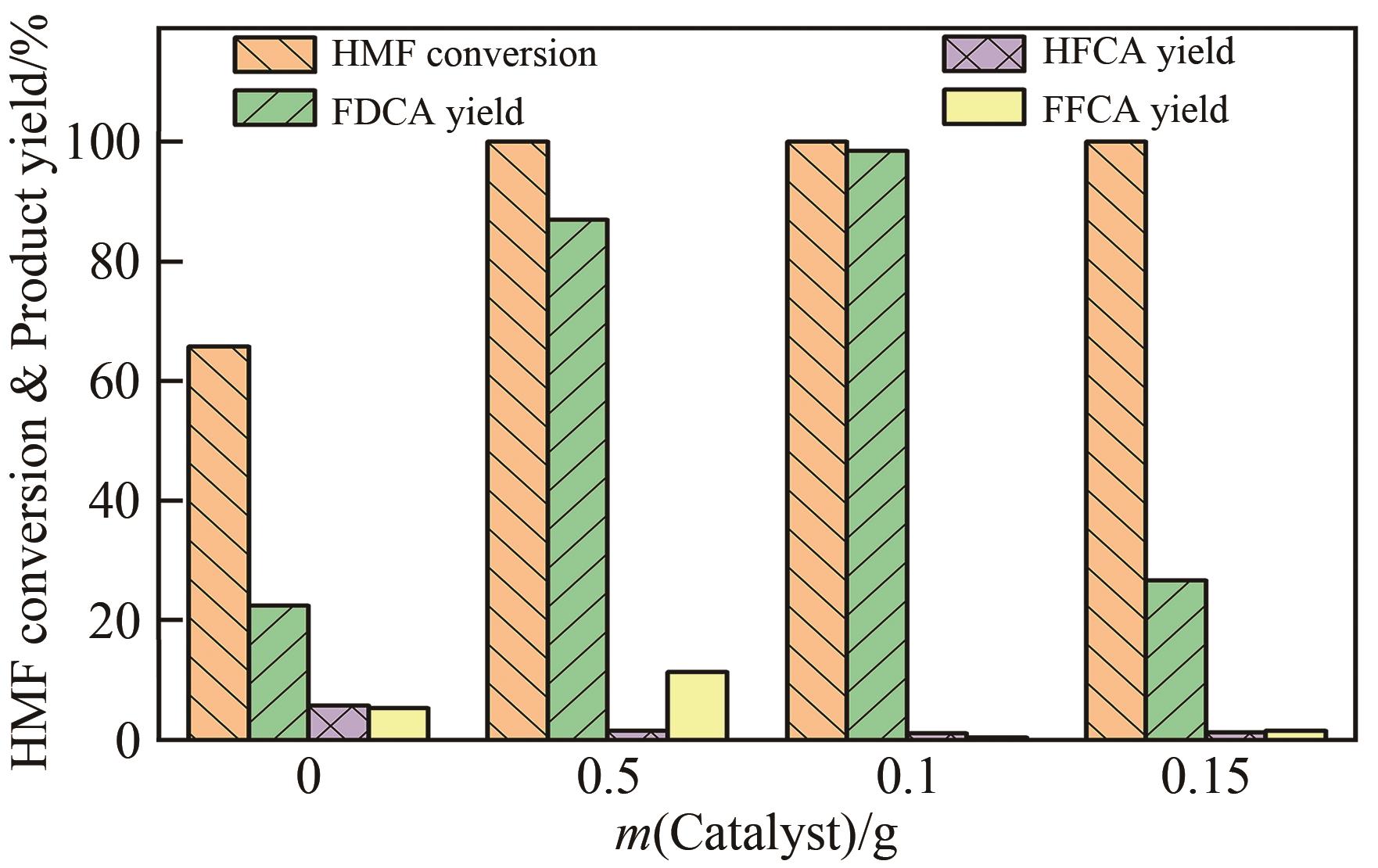
| 1 |
BEASSON M, GALLEZOT P, PINEL C. Conversion of biomass into chemicals over metal catalysts[J]. Chem Rev, 2014, 114(3): 1827-1870.
|
| 2 |
XU C, PAONE E, RODRÍGUEZ-PADRÓN D, et al. Recent catalytic routes for the preparation and the upgrading of biomass derived furfural and 5-hydroxymethylfurfural[J]. Chem Soc Rev, 2020, 49(13): 4273-4306.
|
| 3 |
HAYASHI E, YAMAGUCHI Y, KAMATA K, et al. Effect of MnO2 crystal structure on aerobic oxidation of 5-hydroxymethylfurfural to 2, 5-furandicarboxylic acid[J]. J Am Chem Soc, 2019, 141(2): 890-900.
|
| 4 |
陈英, 姜敏, 孙长江, 等. 聚2, 5-呋喃二甲酸乙二醇酯/聚丁二酸丁二醇酯共混物的制备与表征[J]. 应用化学, 2015, 32(9): 1022-1027.
|
|
CHEN Y, JIANG M, SUN C J, et al. Preparation and characterization of poly(ethylene 2, 5-furandicarboxylate)/poly(butylene succinate) blends[J]. Chin J Appl Chem, 2015, 32(9): 1022-1027.
|
| 5 |
ADVANI J H, MORE G S, SRIVASTAVA R. Spinel-based catalysts for the biomass valorisation of platform molecules via oxidative and reductive transformations[J]. Green Chem, 2022, 24(9): 3574-3604.
|
| 6 |
WAN X, TANG N, XIE Q, et al. A CuMn2O4 spinel oxide as a superior catalyst for the aerobic oxidation of 5-hydroxymethylfurfural toward 2, 5-furandicarboxylic acid in aqueous solvent[J]. Catal Sci Technol, 2021, 11(4): 1497-1509.
|
| 7 |
ZHANG S, SUN X, ZHENG Z, et al. Nanoscale center-hollowed hexagon MnCo2O4 spinel catalyzed aerobic oxidation of 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid[J]. Catal Commun, 2018, 113: 19-22.
|
| 8 |
MISHRA D K, LEE H J, KIM J, et al. MnCo2O4 spinel supported ruthenium catalyst for air-oxidation of HMF to FDCA under aqueous phase and base-free conditions[J]. Green Chem, 2017, 19(7): 1619-1623.
|
| 9 |
GAWADE A B, NAKHATE A V, YADAV G D. Selective synthesis of 2, 5-furandicarboxylic acid by oxidation 5-hydroxymethylfurfural over MnFe2O4 catalyst[J]. Catal Today, 2018, 309: 119-125.
|
| 10 |
陈文龙. 碳纳米管负载铂、钌催化剂的制备及其催化羰基化合物加氢性能研究[D]. 兰州: 西北师范大学, 2016.
|
|
CHEN W L. Preparation and catalytic properties of carbon nanotubes supported platinum and ruthenium catalysts in the hydrogenation of carbonyl compounds[D]. Lanzhou: Northwest Normal University, 2016.
|
| 11 |
刘帅, 黄振, 何方, 等. NiFe2O4为载氧体的生物质半焦化学链燃烧热力学模拟研究[J]. 新能源进展, 2016, 4(3): 172-178.
|
|
LIU S, HUANG Z, HE F, et al. Thermodynamic simulation of chemical chain combustion of biomass char with NiFe2O4 as oxygen carrier[J]. Adv New Renew Energy, 2016, 4(3): 172-178.
|
| 12 |
陈康伟, 熊文婷, 符继乐, 等. 合成气费托合成制重质烃Ru-Co/SiC催化剂的制备及性能[J]. 化工学报, 2021, 72(7): 3648-3657.
|
|
CHEN K W, XIONG W T, FU J L, et al. Preparation and performance of Ru-Co/SiC catalyst for Fischer-Tropsch synthesis of syngas to heavy hydrocarbons[J]. CIESC J, 2021, 72(7): 3648-3657.
|
| 13 |
孙景玉. 直接甲醇燃料电池碳纳米管负载的阳极催化剂研究[D]. 乌鲁木齐: 新疆大学, 2007.
|
|
SUN J Y. Studies on anode catalysts supported on carbon nanotubes for direct methanol fuel Cell[D]. Urumqi: Xinjiang University, 2007.
|
| 14 |
SEEKHIAW P, PINTHONG P, PRASERTHDAM P, et al. Optimal conditions for butanol production from ethanol over MgAlO catalyst derived from Mg-Al layer double hydroxides[J]. J Oleo Sci, 2022, 71(1): 141-149.
|
| 15 |
LIU M, ZHANG J, LIU J, et al. Synthesis of PVP-stabilized Pt/Ru colloidal nanoparticles by ethanol reduction and their catalytic properties for selective hydrogenation of ortho-chloronitrobenzene[J]. J Catal, 2011, 278(1): 1-7.
|
| 16 |
YANG Y J, FENG Z, LI X, et al. One step synthesis of 2, 5-furandicarboxylic acid from fructose catalyzed by Ce modified Ru/HAP[J]. J Fuel Chem Technol, 2020, 48(8): 942-948.
|
| 17 |
BENRABAA R, BOUKHLOUF H, LÖFBERG A, et al. Nickel ferrite spinel as catalyst precursor in the dry reforming of methane: synthesis, characterization and catalytic properties[J]. J Nat Gas Chem, 2012, 21(5): 595-604.
|
| 18 |
张丹, 尚润梅, 赵振涛, 等. V/Ce-Al2O3催化甲醇选择性氧化制备二甲氧基甲烷[J]. 应用化学, 2022, 39(9): 1429-1436.
|
|
ZHANG D, SHANG R M, ZHAO Z T, et al. Selective oxidation of methanol to dimethoxymethane over V/Ce-Al2O3[J]. Chin J Appl Chem, 2022, 39(9): 1429-1436.
|
| 19 |
DAS S, THAKUR S, BAG A, et al. Support interaction of Ni nanocluster based catalysts applied in CO2 reforming[J]. J Catal, 2015, 330: 46-60.
|
| 20 |
REDDY G K,BOOLCHAND P,SMIRNIOTIS P G. Unexpected behavior of copper in modified ferrites during high temperature WGS reaction aspects of Fe3+↔Fe2+ redox chemistry from mössbauer and XPS studies[J]. J Phys Chem C, 2012, 116(20): 11019-11031.
|
| 21 |
GAO T, YIN Y, ZHU G, et al. Co3O4 NPs decorated Mn-Co-O solid solution as highly selective catalyst for aerobic base-free oxidation of 5-HMF to 2, 5-FDCA in water[J]. Catal Today, 2020, 355: 252-262.
|
| 22 |
GAO T, YIN Y, FANG W, et al. Highly dispersed ruthenium nanoparticles on hydroxyapatite as selective and reusable catalyst for aerobic oxidation of 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid under base-free conditions[J]. Mol Catal, 2018, 450: 55-64.
|
| 23 |
ZHU M M, DU X L, ZHAO Y, et al. Ring-opening transformation of 5-hydroxymethylfurfural using a golden single-atomic-site palladium catalyst[J]. ACS Catal, 2019, 9(7): 6212-6222.
|
| 24 |
VANDARKUZHALI S A A, KARTHIKEYAN G, PACHAMUTHU M P. Efficient oxidation of 5-hydroxymethylfurfural to 2, 5-furandicarboxylic acid over FeNPs@NH2-SBA-15 catalyst in water[J]. Mol Catal, 2021, 16: 111951.
|
| 25 |
KANDASAMY P, GOGOI P, VENUGOPALAN A T, et al. A highly efficient and reusable Ru-NaY catalyst for the base free oxidation of 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid[J]. Catal Today, 2021, 375: 145-154.
|

 )
)
 )
)