应用化学 ›› 2023, Vol. 40 ›› Issue (1): 116-125.DOI: 10.19894/j.issn.1000-0518.220128
含1,2,4-三唑双席夫碱衍生物的合成、抗菌活性及分子对接
- 西北大学化工学院,西安市特种能源重点实验室,西安 710069
-
收稿日期:2022-04-12接受日期:2022-10-08出版日期:2023-01-01发布日期:2023-01-28 -
通讯作者:任莹辉 -
基金资助:陕西省自然科学基金重点项目(2016JZ003)
Synthesis, Antifungal Activity and Molecular Docking Study of 1,2,4-Triazole Bis-Schiff Base Derivatives
Mou-Cui LI, Yang-Ming DONG, Ying-Hui REN( ), Hai-Xia MA, Le QI
), Hai-Xia MA, Le QI
- Schoolof Chemical Engineering,Northwest University,Xi'an Key Laboratory of Special Energy Materials,Xi'an 710069,China
-
Received:2022-04-12Accepted:2022-10-08Published:2023-01-01Online:2023-01-28 -
Contact:Ying-Hui REN -
About author:nwuryh@163.com
-
Supported by:the Provincial Key-point Natural Science Foundation of Shaanxi, China(2016JZ003)
摘要:
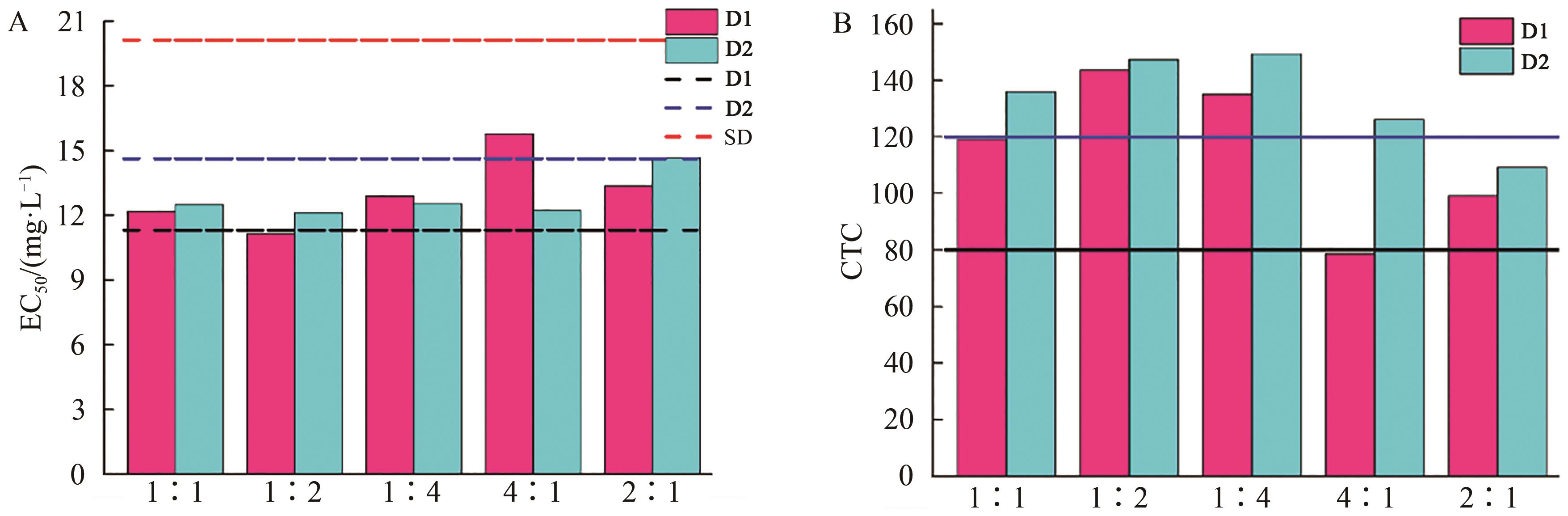
以3, 5-二氨基-1,2,4-三唑与取代苯甲醛为原料成功合成了5种1,2,4-三唑双席夫碱衍生物(D1-D5)。采用菌丝生长速率法和分子对接技术研究了化合物对小麦赤霉病菌的体外抗真菌活性。结果表明,除化合物D3外,其余4种化合物的活性均优于对照药物氟康唑,其中化合物D1的抑制效果约为对照药物的1.8倍,可作为抗小麦赤霉病菌的先导化合物进一步研究。分子对接的结果与生物活性一致,即对接结合能越小,抗小麦赤霉病菌的活性越强,且氢键是维持药物分子与受体蛋白稳定结合的关键因素。为克服病原菌的耐药性问题,研究了活性最强的化合物D1和D2与氟康唑复配体系的抑菌作用,结果发现,m(D1)∶m(氟康唑)=1∶2、m(D2)∶m(氟康唑)=1∶1、1∶2、1∶4和4∶1时,共毒系数(CTC)值大于120,表现为增效作用,故化合物D2有望成为一种具有应用前景的复配用剂。
中图分类号:
引用本文
李谋翠, 董洋铭, 任莹辉, 马海霞, 齐乐. 含1,2,4-三唑双席夫碱衍生物的合成、抗菌活性及分子对接[J]. 应用化学, 2023, 40(1): 116-125.
Mou-Cui LI, Yang-Ming DONG, Ying-Hui REN, Hai-Xia MA, Le QI. Synthesis, Antifungal Activity and Molecular Docking Study of 1,2,4-Triazole Bis-Schiff Base Derivatives[J]. Chinese Journal of Applied Chemistry, 2023, 40(1): 116-125.
| Compounds | Functional group | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| ν(N—H) | ν(C N) N) | ν(C—N) | ν(Ar—H) | ν(C C) C) | ν(N—N) | ν(C—Cl) | ν(CH3) | ν(O—H) | ν(C—F) | |
| D1 | 3 098 | 1 602 | 1 349 | 2 934 | 1 477 | 1 022 | 674 | — | 3 027 | — |
| D2 | 3 104 | 1 599 | 1 348 | 3 007 | 1 538 | 1 031 | — | 2 971 | — | — |
| D3 | 2 988 | 1 595 | 1 349 | 2 912 | 1 493 | 1 027 | — | 2 919 | 3 359 | — |
| D4 | 3 352 | 1 615 | 1 331 | 3 040 | 1 441 | 1 028 | — | — | — | 1 110 |
| D5 | 3 125 | 1 610 | 1 331 | 2 959 | 1 487 | 1 033 | 670 | — | — | — |
表1 化合物红外光谱信息
Table 1 Infrared spectral information of compounds
| Compounds | Functional group | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| ν(N—H) | ν(C N) N) | ν(C—N) | ν(Ar—H) | ν(C C) C) | ν(N—N) | ν(C—Cl) | ν(CH3) | ν(O—H) | ν(C—F) | |
| D1 | 3 098 | 1 602 | 1 349 | 2 934 | 1 477 | 1 022 | 674 | — | 3 027 | — |
| D2 | 3 104 | 1 599 | 1 348 | 3 007 | 1 538 | 1 031 | — | 2 971 | — | — |
| D3 | 2 988 | 1 595 | 1 349 | 2 912 | 1 493 | 1 027 | — | 2 919 | 3 359 | — |
| D4 | 3 352 | 1 615 | 1 331 | 3 040 | 1 441 | 1 028 | — | — | — | 1 110 |
| D5 | 3 125 | 1 610 | 1 331 | 2 959 | 1 487 | 1 033 | 670 | — | — | — |
| Compounds | Inhibition rate/% | Toxicity equation | R | EC50 | |||
|---|---|---|---|---|---|---|---|
| 2 mg/L | 4 mg/L | 8 mg/L | 16 mg/L | ||||
| D1 | 16.33 | 19.33 | 39.67 | 62.33 | y=1.491x+3.429 | 0.938 | 11.31 |
| D2 | 12.22 | 18.52 | 35.93 | 52.96 | y=1.412x+3.351 | 0.985 | 14.63 |
| D3 | 9.37 | 13.45 | 24.56 | 43.86 | y=1.299x+3.206 | 0.967 | 24.28 |
| D4 | 8.66 | 15.22 | 27.46 | 53.73 | y=1.593x+3.078 | 0.972 | 16.09 |
| D5 | 10.80 | 18.95 | 34.88 | 50.00 | y=1.414x+3.306 | 0.997 | 15.80 |
| SD | 3.33 | 14.00 | 19.67 | 44.16 | y=1.769x+2.693 | 0.965 | 20.13 |
表2 目标化合物的抗小麦赤霉病菌活性结果
Table 2 The results of the target compounds against Fusarium graminearum
| Compounds | Inhibition rate/% | Toxicity equation | R | EC50 | |||
|---|---|---|---|---|---|---|---|
| 2 mg/L | 4 mg/L | 8 mg/L | 16 mg/L | ||||
| D1 | 16.33 | 19.33 | 39.67 | 62.33 | y=1.491x+3.429 | 0.938 | 11.31 |
| D2 | 12.22 | 18.52 | 35.93 | 52.96 | y=1.412x+3.351 | 0.985 | 14.63 |
| D3 | 9.37 | 13.45 | 24.56 | 43.86 | y=1.299x+3.206 | 0.967 | 24.28 |
| D4 | 8.66 | 15.22 | 27.46 | 53.73 | y=1.593x+3.078 | 0.972 | 16.09 |
| D5 | 10.80 | 18.95 | 34.88 | 50.00 | y=1.414x+3.306 | 0.997 | 15.80 |
| SD | 3.33 | 14.00 | 19.67 | 44.16 | y=1.769x+2.693 | 0.965 | 20.13 |
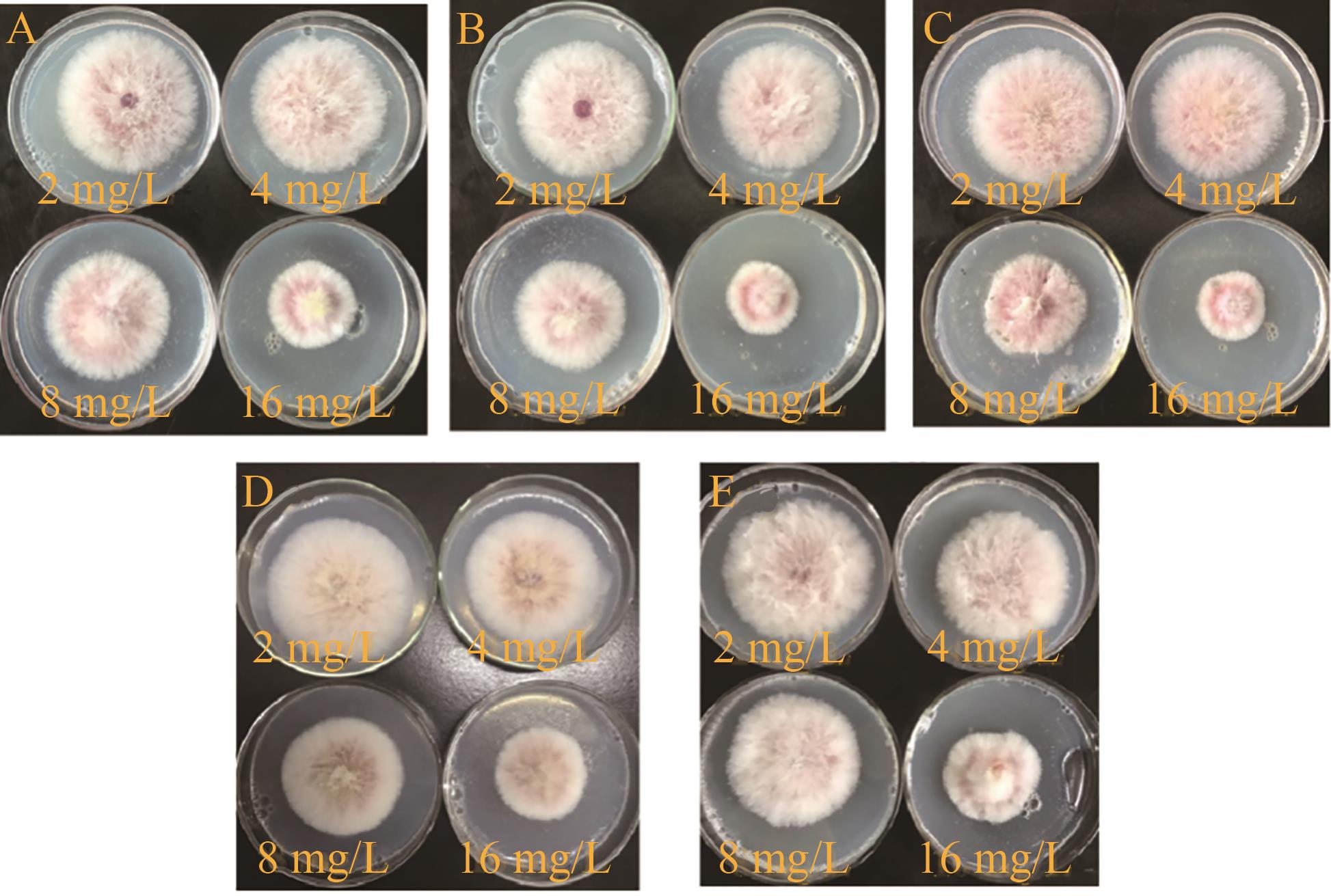
图2 化合物D1与SD在5种比例下复配的抑制效果图: (A) 1∶1, (B) 1∶2, (C) 1∶4, (D)4∶1和(E) 2∶1(28 ℃, 72 h)
Fig.2 Inhibition effect of D1 compounded with fluconazole at 5 different m(D1)∶m(SD) of 1∶1 (A), 1∶2 (B), 1∶4 (C), 4∶1 (D) and 2∶1 (E) (28 ℃, 72 h)
| Compounds | Mass ratio | Toxicity equation | R | EC50 | ATI | TTI | CTC |
|---|---|---|---|---|---|---|---|
| D1∶SD | 1∶0 | y=1.491x+3.429 | 0.938 | 11.31 | — | — | — |
| 0∶1 | y=1.769x+2.693 | 0.965 | 20.13 | — | — | — | |
| 1∶1 | y=1.444x+3.423 | 0.992 | 12.17 | 165.41 | 138.99 | 119.00 | |
| 1∶2 | y=1.150x+3.796 | 0.958 | 11.14 | 180.70 | 125.99 | 143.42 | |
| 1∶4 | y=1.124x+3.751 | 0.900 | 12.89 | 156.17 | 115.60 | 135.10 | |
| 4∶1 | y=1.036x+3.759 | 0.955 | 15.78 | 127.57 | 162.38 | 78.56 | |
| 2∶1 | y=1.463x+3.353 | 0.999 | 13.36 | 150.67 | 151.98 | 99.14 | |
| D2∶SD | 1∶0 | y=1.412x+3.351 | 0.985 | 14.63 | — | — | — |
| 0∶1 | y=1.769x+2.693 | 0.965 | 20.13 | — | — | — | |
| 1∶1 | y=1.622x+3.222 | 0.976 | 12.47 | 161.43 | 118.80 | 135.89 | |
| 1∶2 | y=1.895x+2.919 | 0.987 | 12.11 | 166.23 | 112.53 | 147.22 | |
| 1∶4 | y=1.555x+3.315 | 0.990 | 12.54 | 160.53 | 105.52 | 149.30 | |
| 4∶1 | y=1.445x+3.428 | 0.999 | 12.23 | 164.60 | 130.07 | 126.22 | |
| 2∶1 | y=1.369x+3.423 | 0.991 | 14.672 | 137.22 | 125.06 | 109.21 |
表3 复配实验的毒力回归方程、EC50、ATI、TTI和CTC值
Table 3 The toxicity equation, values of EC50, ATI, TTI and CTC of the combinations experiment
| Compounds | Mass ratio | Toxicity equation | R | EC50 | ATI | TTI | CTC |
|---|---|---|---|---|---|---|---|
| D1∶SD | 1∶0 | y=1.491x+3.429 | 0.938 | 11.31 | — | — | — |
| 0∶1 | y=1.769x+2.693 | 0.965 | 20.13 | — | — | — | |
| 1∶1 | y=1.444x+3.423 | 0.992 | 12.17 | 165.41 | 138.99 | 119.00 | |
| 1∶2 | y=1.150x+3.796 | 0.958 | 11.14 | 180.70 | 125.99 | 143.42 | |
| 1∶4 | y=1.124x+3.751 | 0.900 | 12.89 | 156.17 | 115.60 | 135.10 | |
| 4∶1 | y=1.036x+3.759 | 0.955 | 15.78 | 127.57 | 162.38 | 78.56 | |
| 2∶1 | y=1.463x+3.353 | 0.999 | 13.36 | 150.67 | 151.98 | 99.14 | |
| D2∶SD | 1∶0 | y=1.412x+3.351 | 0.985 | 14.63 | — | — | — |
| 0∶1 | y=1.769x+2.693 | 0.965 | 20.13 | — | — | — | |
| 1∶1 | y=1.622x+3.222 | 0.976 | 12.47 | 161.43 | 118.80 | 135.89 | |
| 1∶2 | y=1.895x+2.919 | 0.987 | 12.11 | 166.23 | 112.53 | 147.22 | |
| 1∶4 | y=1.555x+3.315 | 0.990 | 12.54 | 160.53 | 105.52 | 149.30 | |
| 4∶1 | y=1.445x+3.428 | 0.999 | 12.23 | 164.60 | 130.07 | 126.22 | |
| 2∶1 | y=1.369x+3.423 | 0.991 | 14.672 | 137.22 | 125.06 | 109.21 |
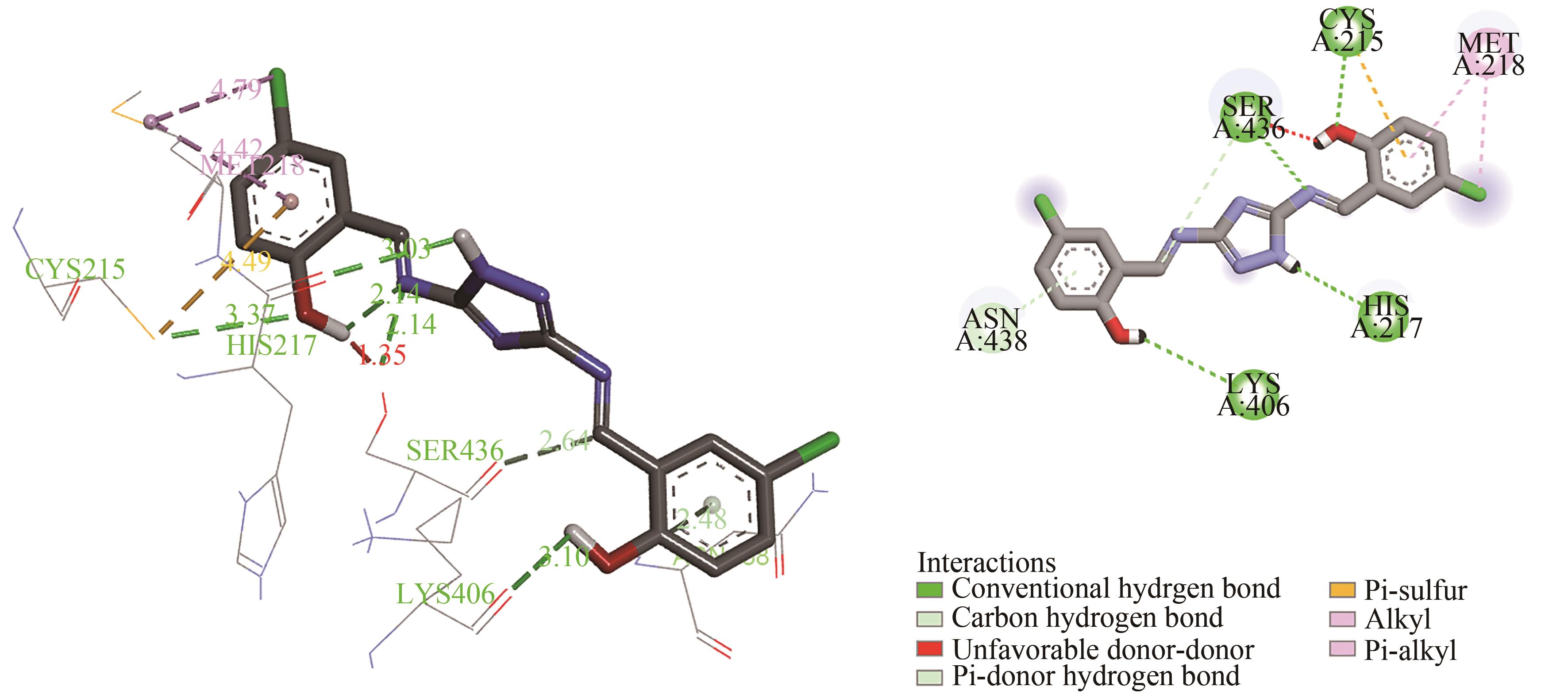
| Compounds | ΔG/ (kJ·mol-1) | Ki/ (μmol·L-1) | ΔGvdW+ΔGHbond+ΔGdesolv/ (kJ·mol-1) | ΔGele/ (kJ·mol-1) | ΔGTor/ (kJ·mol-1) | Hydrogen bond |
|---|---|---|---|---|---|---|
| D1 | -32.30 | 3.71 | -38.74 | -1.05 | 7.49 | CYS A:215, SER A:436, HIS A:217, LYS A:406 |
| D2 | -32.17 | 2.30 | -36.74 | -0.46 | 4.98 | SER A:436 |
| D3 | -23.89 | 65.07 | -30.84 | -0.54 | 7.49 | - |
| D4 | -28.62 | 9.63 | -35.19 | -0.96 | 7.49 | LYS A:221, SER A:436 |
| D5 | -28.66 | 9.60 | -32.84 | -0.79 | 4.98 | SER A:436, CYS A:215 |
表4 化合物与5E9H的对接结果
Table 4 Docking results of compounds with 5E9H
| Compounds | ΔG/ (kJ·mol-1) | Ki/ (μmol·L-1) | ΔGvdW+ΔGHbond+ΔGdesolv/ (kJ·mol-1) | ΔGele/ (kJ·mol-1) | ΔGTor/ (kJ·mol-1) | Hydrogen bond |
|---|---|---|---|---|---|---|
| D1 | -32.30 | 3.71 | -38.74 | -1.05 | 7.49 | CYS A:215, SER A:436, HIS A:217, LYS A:406 |
| D2 | -32.17 | 2.30 | -36.74 | -0.46 | 4.98 | SER A:436 |
| D3 | -23.89 | 65.07 | -30.84 | -0.54 | 7.49 | - |
| D4 | -28.62 | 9.63 | -35.19 | -0.96 | 7.49 | LYS A:221, SER A:436 |
| D5 | -28.66 | 9.60 | -32.84 | -0.79 | 4.98 | SER A:436, CYS A:215 |
| 1 | 吾尔尼沙·哈也尔肯. 小麦赤霉病、白粉病的发生与防治[J]. 农家参谋, 2022(5): 58-60. |
| URNISHA H. Occurrence and control of fusarium graminearum and powdery mildew in wheat[J]. Farm Consult, 2022(5): 58-60. | |
| 2 | SHARMA A, SHUKLA A, ATTRI K, et al. Global trends in pesticides: a looming threat and viable alternatives[J]. Ecotoxicol Environ Saf, 2020, 201: 110812. |
| 3 | 陈明桂, 华学文, 魏巍, 等. 含有苯磺酰胺基团1,2,4-三唑和噻二唑类化合物的设计、合成及抑菌活性[J]. 应用化学, 2017, 34(10): 1161-1171. |
| CHEN M G, HUA X W, WEI W, et al. Design, synthesis and fungicidal bioactivity of 1,2,4-triazole or thiadiazoles containing a benzenesulfonamide moiety[J]. Chin J Appl Chem, 2017, 34(10): 1161-1171. | |
| 4 | KAPRON B, LUSZCZKI J J, PLAZISKA A, et al. Development of the 1,2,4-triazole-based anticonvulsant drug candidates acting on the voltage-gated sodium channels. Insights from in-vivo, in-vitro, and in-silico studies[J]. Eur J Pharm Sci, 2019, 129: 42-57. |
| 5 | KAUR P, CHAWL A A. 1,2,4-Triazole: a review of pharmacological activities[J]. Int Res J Pharm, 2017, 8(7): 10-29. |
| 6 | 杨清翠, 孙晓红, 齐亚兵, 等. 3位取代1,2,4-三唑席夫碱的物性及杀菌活性研究[J]. 化学工程, 2020, 48(11): 1-5. |
| YANG Q C, SUN X H, QI Y B, et al. Physical properties and antibacterial activities on three substitutions of 1,2,4-triazole Schiff bases[J]. Chem Eng, 2020, 48(11): 1-5. | |
| 7 | YANG P, LUO J B, WANG Z Z, et al. Synthesis, molecular docking, and evaluation of antibacterial activity of 1,2,4-triazole-norfloxacin hybrids[J]. Bioorg Chem, 2021, 115: 105270. |
| 8 | HOLANDA V N, SILVA W V, NASCIMENTO P H, et al. Antileishmanial activity of 4-phenyl-1-[2-(phthalimido-2-yl) ethyl]-1H-1,2,3-triazole (PT4) derivative on Leishmania amazonensis and Leishmania braziliensis: in silico ADMET, in vitro activity, docking and molecular dynamic simulations[J]. Bioorg Chem, 2020, 105: 104437. |
| 9 | ZHANG J, ZHANG H, CAI W, et al. ‘Click’ D1 receptor agonists with a 5-HT1A receptor pharmacophore producing D2 receptor activity[J]. Bioorg Med Chem, 2009, 17(14): 4873-4880. |
| 10 | HUBER D, HUBNER H, GMEINER P. 1′-Disubstituted ferrocenes as molecular hinges in mono-and bivalent dopamine receptor ligands[J]. J Med Chem, 2009, 52(21): 6860-6870. |
| 11 | WU S J, QI L, REN Y H, et al.1,2,4-Triazole-3-thione Schiff bases compounds: crystal structure, hirshfeld surface analysis, DFT studies and biological evaluation[J].J Mol Struct, 2020, 1219: 128591. |
| 12 | CHENG Y N, JIANG Z H, SUN L S, et al. Synthesis of 1,2,4-triazole benzoyl arylamine derivatives and their high antifungal activities[J]. Eur J Med Chem, 2020, 200: 112463. |
| 13 | LI C, LIU J C, LI Y R, et al. Synthesis and antimicrobial evaluation of 5-aryl-1,2,4-triazole-3-thione derivatives containing a rhodanine moiety[J]. Bioorg Med Chem Lett, 2015, 25(15): 3052-3056. |
| 14 | 暴金平, 樊素芳, 杨国玉, 等. 壳寡糖氨基硫脲席夫碱铜的制备及其抗真菌活性[J]. 应用化学, 2019, 36(5): 500-508. |
| BAO J P, FAN S F, YANG G Y, et al. Synthesis and antifungal activities of chitosan oligosaccharide thiosemicarbazone Schiff base Cu(Ⅱ) complex[J]. Chin J Appl Chem, 2019, 36(5): 500-508. | |
| 15 | MESBAH M, DOUADI T, SAHLI F, et al. Synthesis, characterization, spectroscopic studies and antimicrobial activity of three new schiff bases derived from heterocyclic moiety[J]. J Mol Struct, 2018, 1151: 41-48. |
| 16 | XU R B, AOTEGEN B, ZHONG Z M. Synthesis, characterization and biological activity of C6-Schiff bases derivatives of chitosan[J].Int J Biol Macromol, 2017, 105: 1563-1571. |
| 17 | ABOSEADA H A, HASSANIEN M M, EL-SAYED I H, et al. Schiff base 4-ethyl-1-(pyridin-2-yl) thiosemicarbazide up-regulates the antioxidant status and inhibits the progression of Ehrlich solid tumor in mice[J]. Biochem Biophys Res Commun, 2021, 573: 42-47. |
| 18 | QI L, LI M C, BAI J C, et al. In vitro antifungal activities, molecular docking, and DFT studies of 4-amine-3-hydrazino-5-mercapto-1,2,4-triazole derivatives[J]. Bioorg Med Chem Lett, 2021, 40: 127902. |
| 19 | WU S J, ZHANG W H, QI L, et al. Investigation on 4-amino-5-substituent-1,2,4-triazole-3-thione Schiff bases an antifungal drug by characterization (spectroscopic, XRD), biological activities, molecular docking studies and electrostatic potential (ESP)[J]. J Mol Struct, 2019, 1197: 171-182. |
| 20 | WANG B L, ZHANG L Y, LIU X H, et al. Synthesis, biological activities and SAR studies of new 3-substitutedphenyl-4-substitutedbenzylideneamino-1,2,4-triazole Mannich bases and bis-Mannich bases as ketol-acid reductoisomerase inhibitors[J]. Bioorg Med Chem Lett, 2017, 27(24): 5457-5462. |
| 21 | 宣芳, 徐鉴, 张翠玲, 等. 一种新型1,2,4-三唑席夫碱衍生物的合成及其抑菌活性的研究[J]. 化工时刊, 2017, 31(8): 16-18. |
| XUAN F, XU J, ZHANG C L, et al. Synthesis and antibacterial activity of a novel 1,2,4-triazole Schiff base derivatives[J]. Chem Ind Times, 2017, 31(8): 16-18. | |
| 22 | 袁阳, 刘旭川, 胡先奇. 石榴枯萎病防治药剂筛选和复配作用的初步研究[J]. 农药, 2018, 57(1): 71-74. |
| YUAN Y, LIU X C, HU X Q. Preliminary study on screening and compounding effects of insecticides for control of pomegranate fusarium wilt[J]. Chin J Pestic, 2018, 57(1): 71-74. | |
| 23 | 杨丽娜, 张亮, 韦永淑, 等. 吡唑醚菌酯及与生物农药复配防治桃枝枯病[J]. 农药, 2022, 61(1): 65-69. |
| YANG L N, ZHANG L, WEI Y S, et al. Control of peach shoot blight by pyrazole pyrazole and its combination with biopesticides[J]. Chin J Pestic, 2022, 61(1): 65-69. | |
| 24 | 张培芬, 林晓铧, 陆楚文, 等. 四种植物提取物及复配的抑菌效果[J]. 广州化工, 2021, 49(15): 89-92. |
| ZHANG P F, LIN X H, LU C W, et al. Antibacterial effects of four plant extracts and their complexes[J]. Guangzhou Chem Ind, 2021, 49(15): 89-92. | |
| 25 | 刘召阳, 宋艳艳, 冯浩, 等. 3种复配杀菌剂对苹果褐斑病菌的室内生物活性及田间防效评价[J]. 农药, 2021, 60(1): 66-69. |
| LIU Z Y, SONG Y Y, FENG H, et al. Evaluation of indoor biological activity and field control efficiency of three compound fungicides against apple brown spot[J]. Chin J Pestic, 2021, 60(1): 66-69. | |
| 26 | 赵茜. 一种防治小菜蛾植物源农药的研发[J]. 黑龙江农业科学, 2017(8): 53-56. |
| ZHAO Q. Development of a plant derived pesticide for control of Plutella xylostella[J]. Heilongjiang Agric Sci, 2017(8): 53-56. | |
| 27 | SUMRRA S H, CHOHAN Z H. In vitro antibacterial, antifungal and cytotoxic activities of some triazole Schiff bases and their oxovanadium (IV) complexes[J]. J Enzyme Inhib Med Chem, 2013, 28(6): 1291-1299. |
| 28 | SAYED H A, SAYED H, TAMANY M, et al. Regioselective synthesis, characterization and antimicrobial evaluation of S-glycosides and S,N-diglycosides of 1,2-dihydro-5-(1H-indol-2-yl)-1,2,4-triazole-3-thione[J]. Eur J Med Chem, 2013, 66(15): 106-113. |
| 29 | SUN Y P, JOHNSON E R. Analysis of joint action of insecticides against house flies[J]. J Econ Entomol, 1960, 53(5): 887-892. |
| 30 | HARGROVE T Y, WAWRZAK Z, LAMB D C, et al. Structure-functional characterization of cytochrome P450 sterol 14α-demethylase (CYP51B) from aspergillus fumigatus and molecular basis for the development of antifungal drugs[J]. J Biol Chem, 2015, 290(39): 23916-23934. |
| 31 | AMERI A, KHODARAHMI G, HASSANZADEH F, et al. Novel aldimine-type Schiff bases of 4-amino-5-[(3,4,5-trimethoxyphenyl)methyl]-1,2,4-triazole-3-thione/thiol: docking study, synthesis, biological evaluation, and anti-tubulin activity[J]. Arch Pharm, 2016, 349(8): 662-681. |
| 32 | ABDELAZEEM A H, EL-SAADI M T, SAID E G, et al. Novel diphenylthiazole derivatives with multi-target mechanism: synthesis, docking study, anticancer and anti-inflammatory activities[J]. Bioorg Chem, 2017, 75: 127-138. |
| 33 | STUDIO D. Dassault systemes BIOVIA, discovery studio modelling environment, Release 4.5[J]. Accelrys Softw Inc, 2015: 98-104. |
| 34 | IDNURM A, HOWLETT B J. Isocitrate lyase is essential for pathogenicity of the fungus leptosphaeria maculans to canola (Brassica napus)[J]. Eukaryotic Cell, 2002, 1(5): 719-724. |
| 35 | DUNN M F, RAMIREZ-TRUJILLO J A, HERNANDEZ-LUCAS I. Major roles of isocitrate lyase and malate synthase in bacterial and fungal pathogenesis[J]. Microbiol, 2009, 155(10): 3166-3175. |
| 36 | WANG Z Y, THORNTON C R, KERSHAW M J, et al. The glyoxylate cycle is required for temporal regulation of virulence by the plant pathogenic fungus Magnaporthe grisea[J]. Mol Microbiol, 2003, 47(6): 1601-1612. |
| 37 | 武爱波. 禾谷镰刀菌(Fusarium graminearum)致病力鉴定、毒素检测及其分子生物学研究[D]. 武汉: 华中农业大学, 2005. |
| WU A B. Virulence identification, toxin detection and molecular biology study of Fusarium graminearum[D]. Wuhan: Huazhong Agricultural University, 2005. | |
| 38 | 杨森. 三唑类杀菌剂作用机理和残留检测技术分析研究[J]. 农药科学与管理, 2020, 41(6): 27-32. |
| YANG S. Study on the mechanism and residue detection technology of triazole fungicides[J]. Pestic Sci Adm, 2020, 41(6): 27-32. | |
| 39 | PARK Y, CHO Y, LEE Y H, et al. Crystal structure and functional analysis of isocitrate lyases from Magnaporthe oryzae and Fusarium graminearum[J]. J Struct Biol, 2016, 194(3): 395-403. |
| [1] | 杨玉雯, 齐婧瑶, 李林, 楚国宁, 王赛, 张钰, 张爽. 磁性NiFe2O4负载Ru催化5-羟甲基糠醛选择性氧化合成2,5-呋喃二甲酸[J]. 应用化学, 2023, 40(6): 879-887. |
| [2] | 元宁, 马洁, 张晋玲, 张建胜. 蒸气辅助合成PCN-6(M)双金属有机框架材料及其CH4和CO2吸附性能[J]. 应用化学, 2023, 40(6): 896-903. |
| [3] | 修海祥, 刘万强, 尹东明, 程勇, 王春丽, 王立民. AB2型Laves相储氢合金研究进展[J]. 应用化学, 2023, 40(5): 640-652. |
| [4] | 杨林, 潘卉, 郜定峰, 王晓冬. 芳纶改性聚合物基纳米复合皮革涂饰剂的制备和性能[J]. 应用化学, 2023, 40(5): 708-719. |
| [5] | 方愈, 况王强, 邝圣庭, 廖伍平. Cextrant 230从低品位铜矿H2SO4-NaCl浸出液中选择性提取回收铜[J]. 应用化学, 2023, 40(5): 758-768. |
| [6] | 成燕琴, 黎卓熹, 王有娣, 徐娟娟, 卞证. 结构简单的4-羟基脯氨酰胺高效催化醛和硝基烯不对称迈克尔加成反应[J]. 应用化学, 2023, 40(1): 146-154. |
| [7] | 郭佳昕, 刘洋, 徐长山, 刘晓男, 程亮. 不同pH值下MgO NPs悬浮液中Mg2+质量浓度的动态变化及其对小麦生长的影响[J]. 应用化学, 2022, 39(9): 1401-1411. |
| [8] | 王恩通, 杨林芳. 高比容量锂离子电池正极材料LiNi0.6Co0.2Mn0.2O2的制备及性能[J]. 应用化学, 2022, 39(8): 1209-1215. |
| [9] | 李颖, 张云, 林良良, 许虎君. N‑月桂酰基甲基丙氨酸钠三元复配体系的协同效应[J]. 应用化学, 2022, 39(8): 1262-1273. |
| [10] | 唐连波, 付大友, 陈琦, 奉阳润, 熊桠林, 王竹青. 碳量子点增强气液相化学发光检测二氧化碳[J]. 应用化学, 2022, 39(8): 1294-1302. |
| [11] | 王兵, 唐敏, 王颖, 刘志光. 微氧化烧结制备掺杂Y2O3的SiC陶瓷及含镉模拟废水处理[J]. 应用化学, 2022, 39(8): 1312-1318. |
| [12] | 杨雪贤, 张健, 谷志刚. 表面配位金属-有机框架薄膜HKUST-1在光电应用中的研究进展[J]. 应用化学, 2022, 39(7): 1013-1025. |
| [13] | 张晓丽, 彭玉美, 王庆伟, 秦利霞, 刘肖霞, 康诗钊, 李向清. 纳米Ag/TiO2纳米管阵列基底构建及表面增强拉曼散射光谱检测与降解盐酸四环素[J]. 应用化学, 2022, 39(7): 1147-1156. |
| [14] | 张超. 单原子催化剂电催化还原二氧化碳研究进展[J]. 应用化学, 2022, 39(6): 871-887. |
| [15] | 尹广婷, 周雪健, 姚红柳, 付金凤, 曹洪玉, 郑学仿, 苏丽红. 多光谱法与分子对接模型研究西维来司钠与弹性蛋白酶的相互作用[J]. 应用化学, 2022, 39(6): 960-968. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||