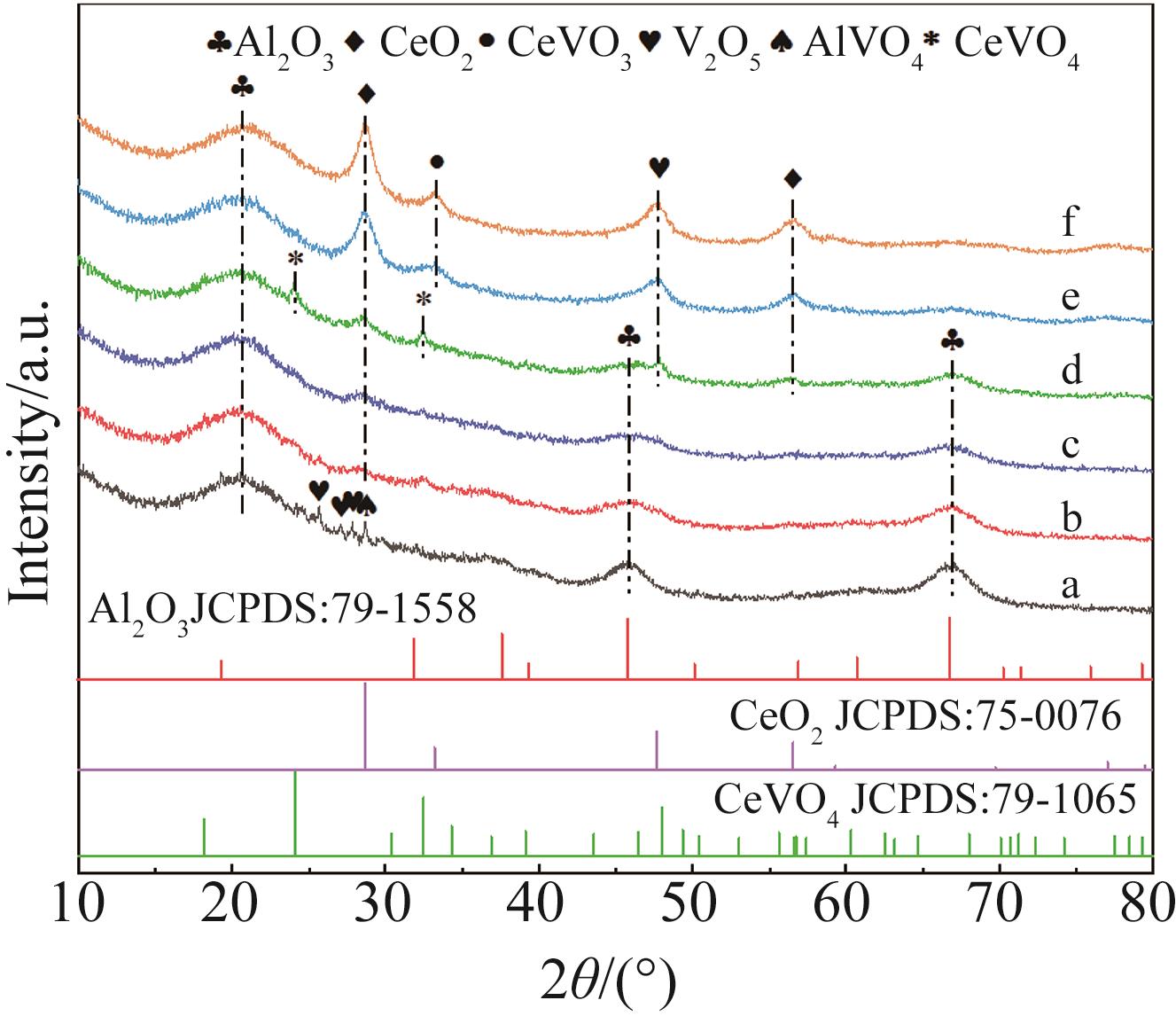
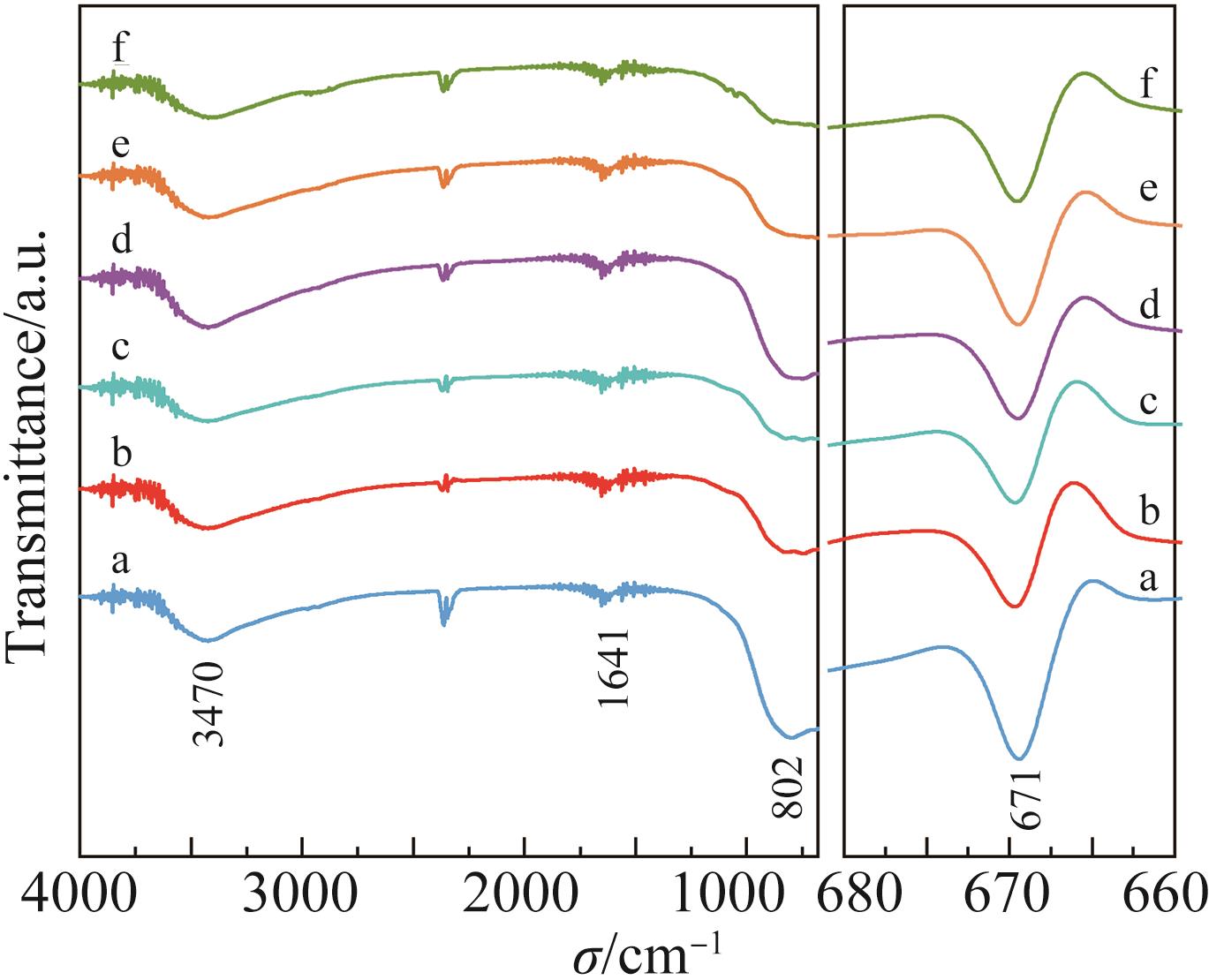
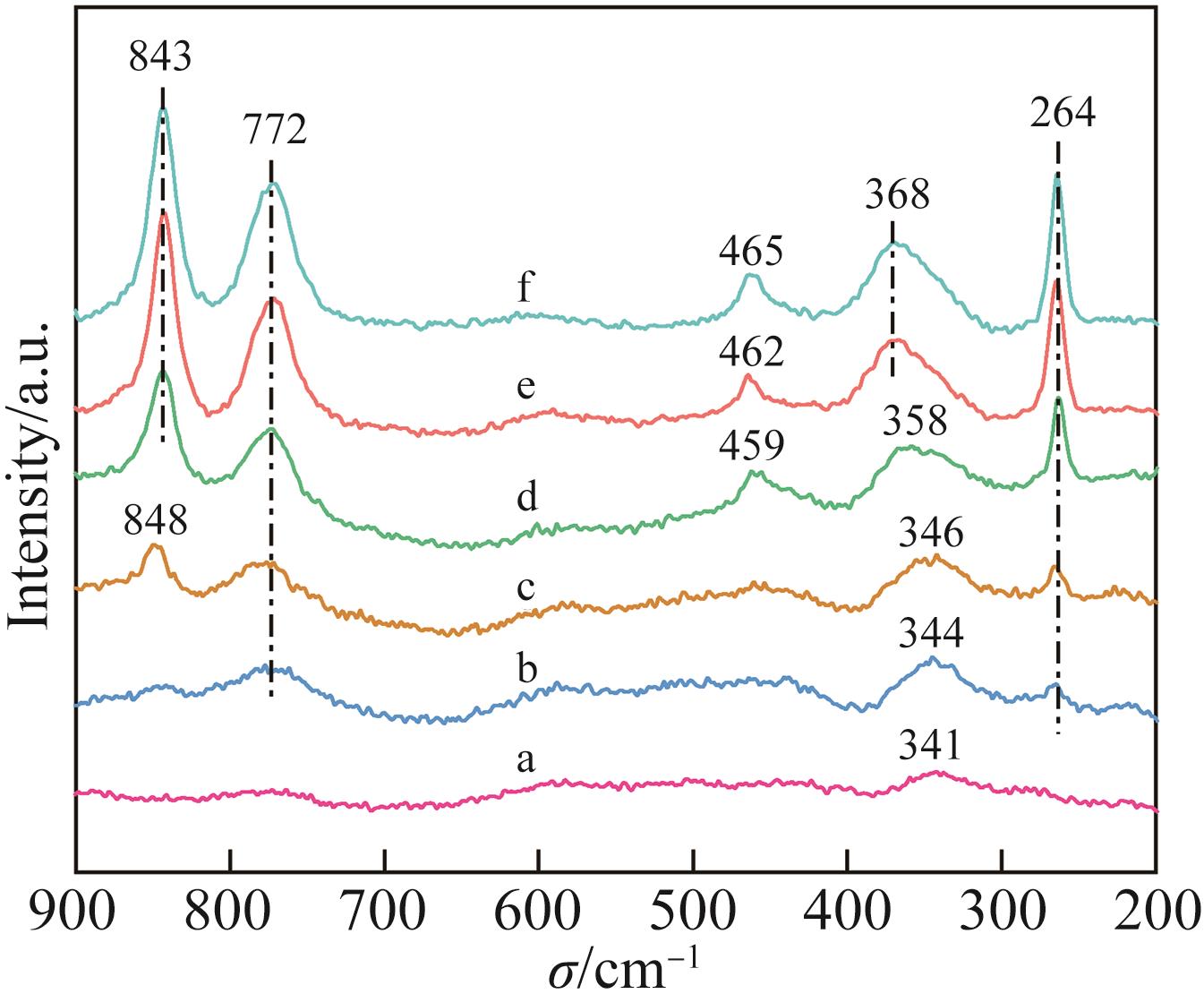
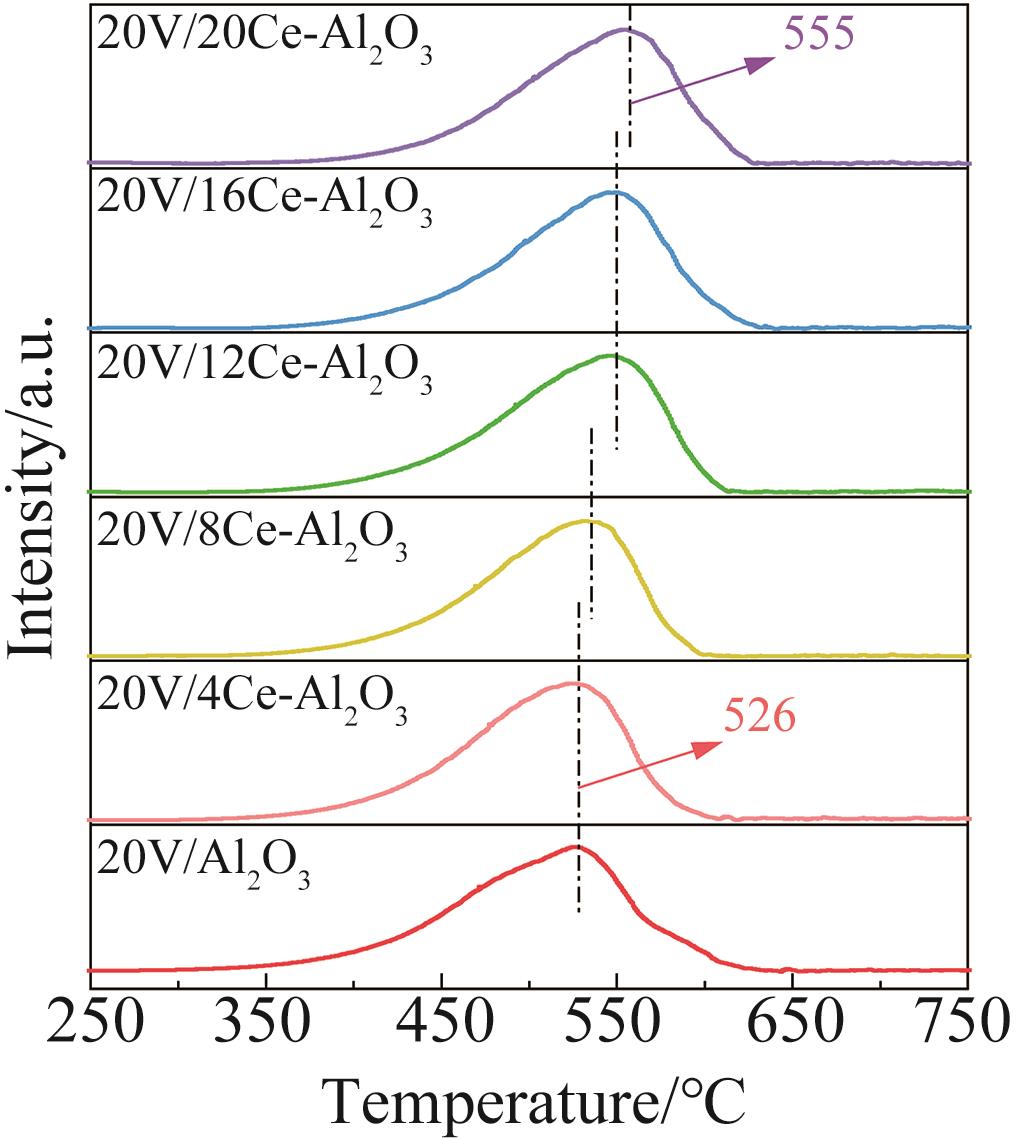
| 1 |
YUAN M, CHE Y, TANG R, et al. One-step synthesis of methylal via methanol oxidation by Mo∶Fe(x)/HZSM-5 bifunctional catalyst[J]. Fuel, 2020, 261: 116416.1-116416.9.
|
| 2 |
武建兵, 吴志伟, 王瑞义, 等. 甲醇制备甲酸甲酯、甲缩醛和聚甲醛二甲醚的催化反应研究进展[J]. 燃料化学学报, 2015, 43(7): 816-828.
|
|
WU J B, WU Z W, WANG R Y, et al. Recent research progresses in the catalytic synthesis of methyl formate, dimethoxymethane and polyoxymethylene dimethyl ethers from methanol[J]. J Fuel Chem Technol, 2015, 43(7): 816-828.
|
| 3 |
陈欣, 贾雯雯, 商永臣. V2O5/MoO3-SnO2催化氧化二甲醚氧化制备甲缩醛的研究[J]. 精细石油化工进展, 2018, 19(6): 42-44, 53.
|
|
CHEN X, JIA W W, SAHGN Y C. Oxidation of dimethyl ether to dimethoxymethane over V2O5/MoO3-SnO2 catalyst[J]. Adv Fine Petrochem, 2018, 19(6): 42-44, 53.
|
| 4 |
BADMAEV S D, SOBYANIN V A. Steam reforming of dimethoxymethane to hydrogen-rich gas over bifunctional CuO-ZnO/γ-Al2O3 catalyst-coated FeCrAl wire mesh[J]. Catal Today, 2020, 348: 9-14.
|
| 5 |
司马蕊. 甲醇一步氧化制DMM高性能催化剂合成与机理研究[D]. 上海: 上海大学, 2017.
|
|
SIMA R. Synthesis and mechanism investigation on high efficient catalysts for methanol one-step oxidation to DMM[D]. Shanghai: Shanghai University, 2017.
|
| 6 |
CHEN S, MENG Y, ZHAO Y, et al. Selective oxidation of methanol to dimethoxymethane over mesoporous Al-P-V-O catalysts[J]. AIChE J, 2013, 59(7): 2587-2593.
|
| 7 |
程金燮, 王科, 胡志彪, 等. 甲醇定向转化制二甲氧基甲烷催化剂研究进展[J]. 天然气化工(C1化学与化工), 2018, 43(3): 95-103.
|
|
CHENG J X, WANG K, HU Z H, et al. Research progress in catalysts for direct conversion of methanol to dimethoxymethane[J]. Nat Gas Chem Ind, 2018, 43(3): 95-103.
|
| 8 |
刘焕君, 高腾飞, 施达, 等. 甲醇一步催化转化制甲缩醛和甲酸甲酯的双功能催化剂[J]. 化学进展, 2016, 28(6):942-953.
|
|
LIU H J, GAO T F, SHI D, et al. Bifunctional catalysts of methanol catalytic conversion to dimethoxymethane and methyl formate[J]. Prog Chem, 2016, 28(6): 942-953.
|
| 9 |
黄昶, 王志强, 龚学庆. VO3/CeO2(111)催化剂上丙烷氧化脱氢反应活性和选择性的密度泛函理论研究[J]. 催化学报, 2018, 39(9): 1520-1526.
|
|
HUANG C, WANG Z Q, GONG X Q. Activity and selectivity of propane oxidative dehydrogenation over VO3/CeO2(111) catalysts:A density functional theory study[J]. Chinese J Catal, 2018, 39(9): 1520-1526.
|
| 10 |
邢鹏, 王淑芳, 王延吉. 载体和焙烧温度对VOx/AC催化甲醇氧化反应性能的影响[J]. 河北工业大学学报, 2021, 50(3): 31-38.
|
|
XING P, WANG S F, WANG Y J. Effect of supports and calcinations temperatures on VOx/AC catalyzed methanol oxidation [J]. J Hebei Univ Technol, 2021, 50(3): 31-38.
|
| 11 |
FAN Z, GUO H, FANG K, et al. Efficient V2O5/TiO2 composite catalysts for dimethoxymethane synthesis from methanol selective oxidation[J]. RSC Adv, 2015, 5(31): 24795-24802.
|
| 12 |
TAO M, WANG H, LU B, et al. Highly selective oxidation of methanol to dimethoxymethane over SO4 2-/V2O5-ZrO2[J]. New J Chem, 2017, 41(16): 8370-8376.
|
| 13 |
ZHAO H, BENNICI S, CAI J, et al. Influence of the metal oxide support on the surface and catalytic properties of sulfated vanadia catalysts for selective oxidation of methanol[J]. J Catal, 2010, 274(2): 259-272.
|
| 14 |
WANG T, MENG Y L, ZENG L, et al. Selective oxidation of methanol to dimethoxymethane over V2O5/TiO2-Al2O3 catalysts[J].Sci Bull, 60(11): 1009-1018.
|
| 15 |
王亚敏. 并流沉淀法制备活性氧化铝及其热稳定性的改性研究[D]. 沈阳: 沈阳工业大学, 2015.
|
|
WANG Y M. Synthesizing active alumina by parallel flow coprecipitating method and thermal stability[D]. Shenyang:Shenyang University of Technology, 2015.
|
| 16 |
BAKE H, ZAMAN S F, ALHAMED Y A, et al. Partial oxidation of methanol over Au/CeO2-ZrO2 and Au/CeO2-ZrO2-TiO2 catalysts[J]. RSC Adv, 2016, 6(27): 22555-22562.
|
| 17 |
IORDANOVA R, DIMITRIEV Y, DIMITROV V. Structure of V2O5-MoO3-Fe2O3 glasses[J]. J Non-cryst Solids, 1994, 167(1/2): 74-80.
|
| 18 |
REDDY B M, KHAN A, YAMADA Y, et al. Raman and X-ray photoelectron spectroscopy study of CeO2-ZrO2 and V2O5/CeO2-ZrO2 catalysts[J]. Langmuir, 2003, 19(7): 3025-3030.
|
| 19 |
LIU J, SUN Q, FU Y, et al. Preparation of mesoporous V-Ce-Ti-O for the selective oxidation of methanol to dimethoxymethane[J]. Catal Lett, 2008, 126(1): 155-163.
|
| 20 |
GU X, GE J, ZHANG H, et al.Structural redox and acid-base properties of V2O5/CeO2 catalysts[J]. Thermochim Acta, 2006, 451(1/2): 84-93.
|
| 21 |
武建兵, 张晓艳, 孙泽平, 等. NaOH质量分数对HZSM-5分子筛性质及其催化甲缩醛气相羰基化性能的影响[J]. 燃料化学学报, 2019, 47(10): 1226-1234.
|
|
WU J B, ZHANG X Y, SUN Z P, et al. Effect of NaOH content for the properties of HZSM-5 zeolites and its catalytic performance on gas phase carbonylation of dimethoxymethane[J]. J Fuel Chem Technol, 2019, 47(10): 1226-1234.
|

 ), 邢锦娟2
), 邢锦娟2
 ), Jin-Juan XING2
), Jin-Juan XING2