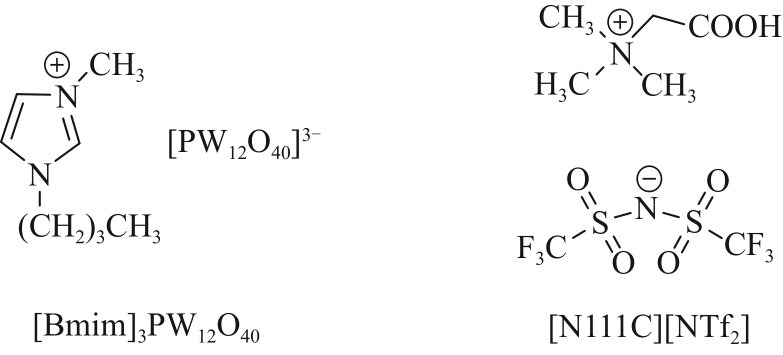应用化学 ›› 2024, Vol. 41 ›› Issue (6): 800-812.DOI: 10.19894/j.issn.1000-0518.240035
• 综合评述 • 上一篇
盐湖卤水萃取法提锂的研究进展
- 1.中国科学院过程工程研究所,绿色过程与工程重点实验室,北京 100190
2.上饶师范学院化学与环境科学学院,上饶 334001
3.景德镇陶瓷大学材料科学与工程学院,景德镇 333403
-
收稿日期:2024-01-31接受日期:2024-04-21出版日期:2024-06-01发布日期:2024-07-09 -
通讯作者:杨良嵘 -
基金资助:国家重点研发计划(2021YFC2901500);国家自然科学基金(22138012);中国科学院基础研究青年团队(YSBR-038);中国科学院青促会优秀会员(Y202014)
Research Progress of Lithium Extraction from Salt Lake Brines
Jian-Feng ZHANG1, Hui-Yong WU1,2,3, Liang-Rong YANG1( )
)
- 1.Key Laboratory of Green Process and Engineering,Institute of Process Engineering,Chinese Academy of Sciences,Beijing 100190,China
2.College of Chemistry and Environmental Science,Shangrao Normal University,Shangrao 334001,China
3.School of Materials Science and Engineering,Jingdezhen Ceramic Institute,Jingdezhen 333403,China
-
Received:2024-01-31Accepted:2024-04-21Published:2024-06-01Online:2024-07-09 -
Contact:Liang-Rong YANG -
About author:lryang@ipe.ac.cn
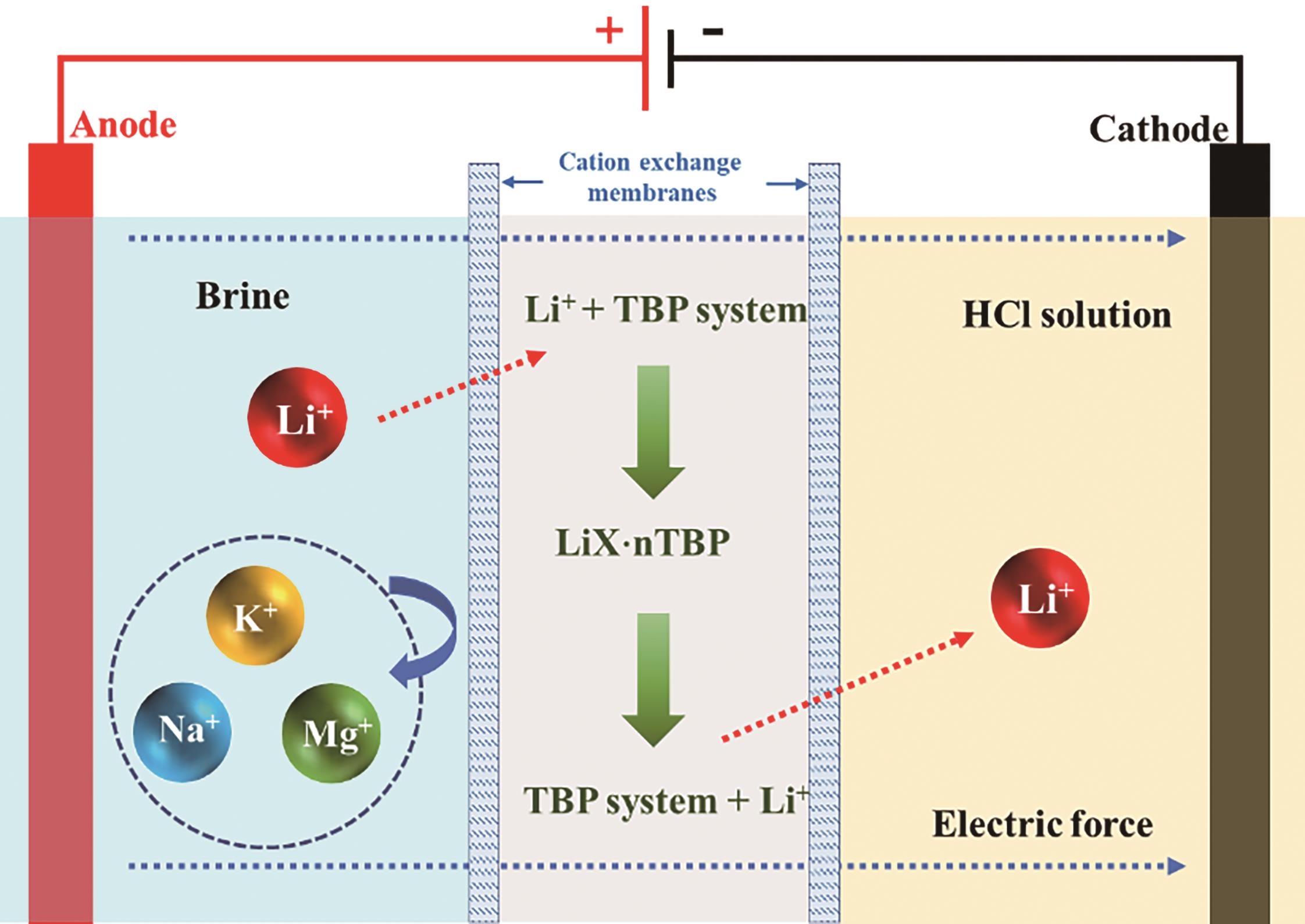
-
Supported by:the National Key R&D Program of China(2021YFC2901500);the National Natural Science Foundation of China(22138012);the CAS Project for Young Scientists in Basic Research(YSBR-038);the Excellent Member in Youth Innovation Promotion Association, Chinese Academy of Sciences(Y202014)
摘要:
随着全球锂电行业的迅猛发展,作为原材料的锂需求量也日益攀升。盐湖卤水中蕴含着丰富的锂资源,但浓度低、成分复杂。开发高效锂分离技术,从卤水中提取锂资源具有重要的经济价值和战略意义。溶剂萃取法具有处理量大、萃取效率高、优异选择性以及易于规模化操作等优点,具有良好的工业应用前景。本文主要综述了近5年常用的萃取体系,包括β-二酮、有机磷化合物、大环分子、磷酰基吡唑啉酮、离子液体以及低共熔溶剂。系统地分析了各种萃取体系的作用机理、适用的环境以及存在的优缺点,对于萃取体系存在的问题进一步介绍了萃取过程强化技术,如膜萃取强化、电场强化。最后,对未来各种萃取体系的研究重点和改进提出了观点和评述,同时对萃取体系的发展前景进行了展望。
中图分类号:
引用本文
张建锋, 吴辉勇, 杨良嵘. 盐湖卤水萃取法提锂的研究进展[J]. 应用化学, 2024, 41(6): 800-812.
Jian-Feng ZHANG, Hui-Yong WU, Liang-Rong YANG. Research Progress of Lithium Extraction from Salt Lake Brines[J]. Chinese Journal of Applied Chemistry, 2024, 41(6): 800-812.
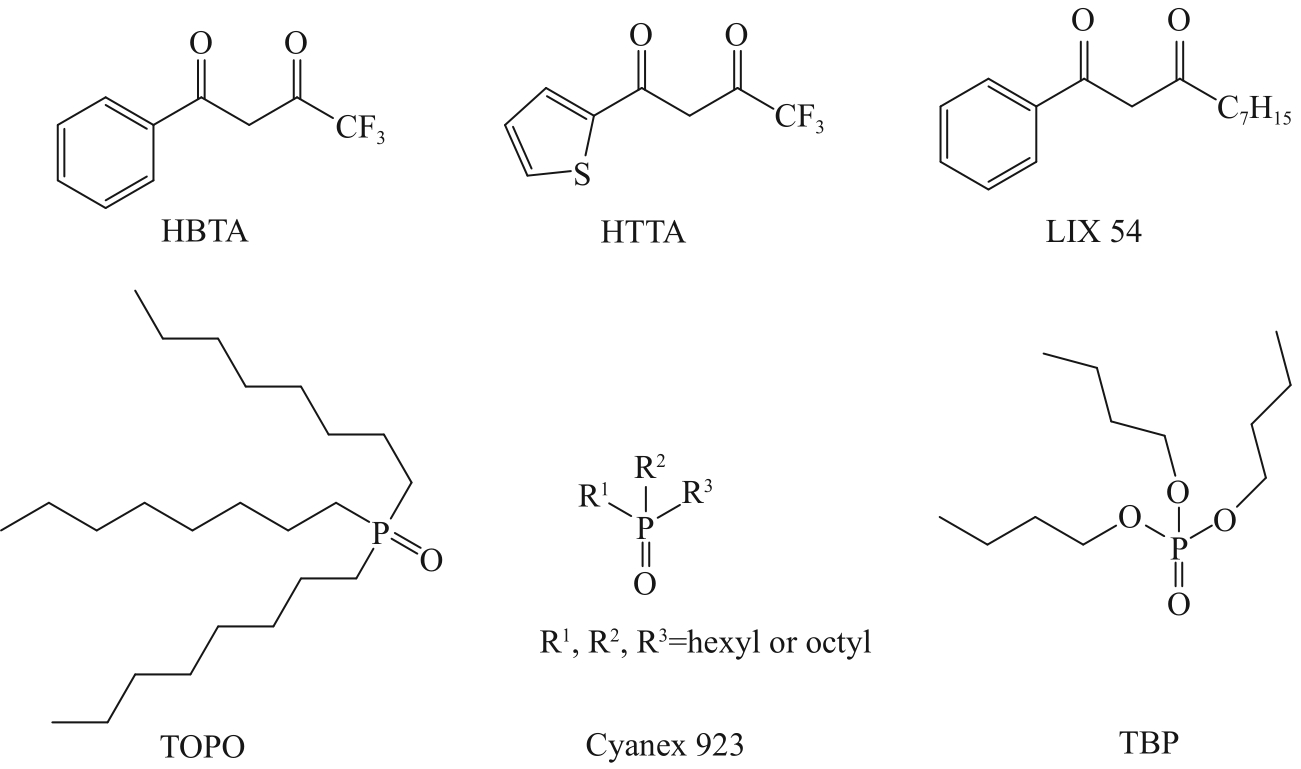
| Extractants | Organic phase | Aqueous phase | Extraction/% | Ref. |
|---|---|---|---|---|
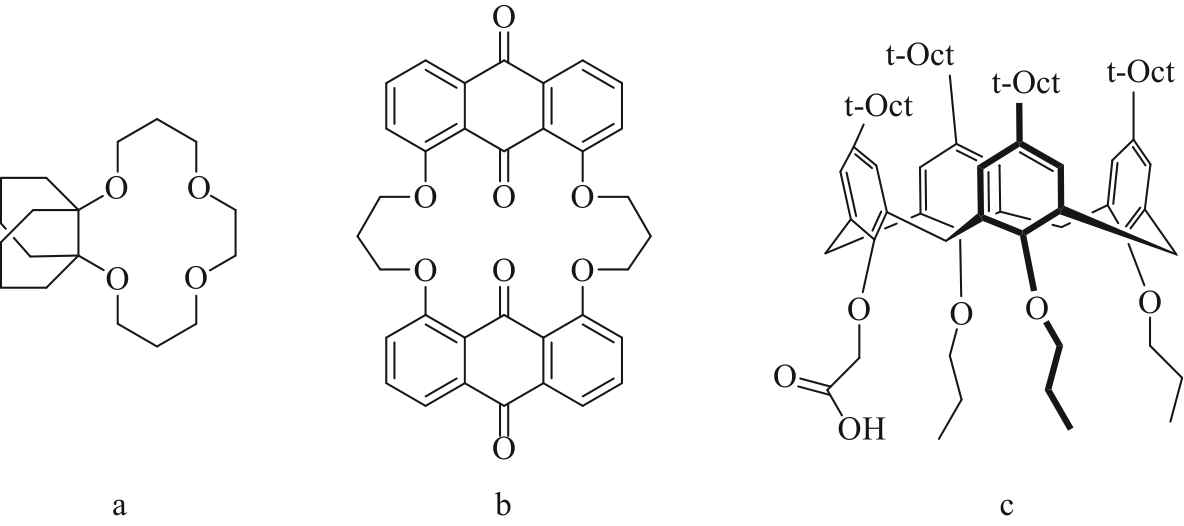
| β-Diketone | HBTA-TOPO, NaOH Pre-saponification | 1.85 g/L Li+, 31.2 g/L Na+ | Li+: 80% | [ |
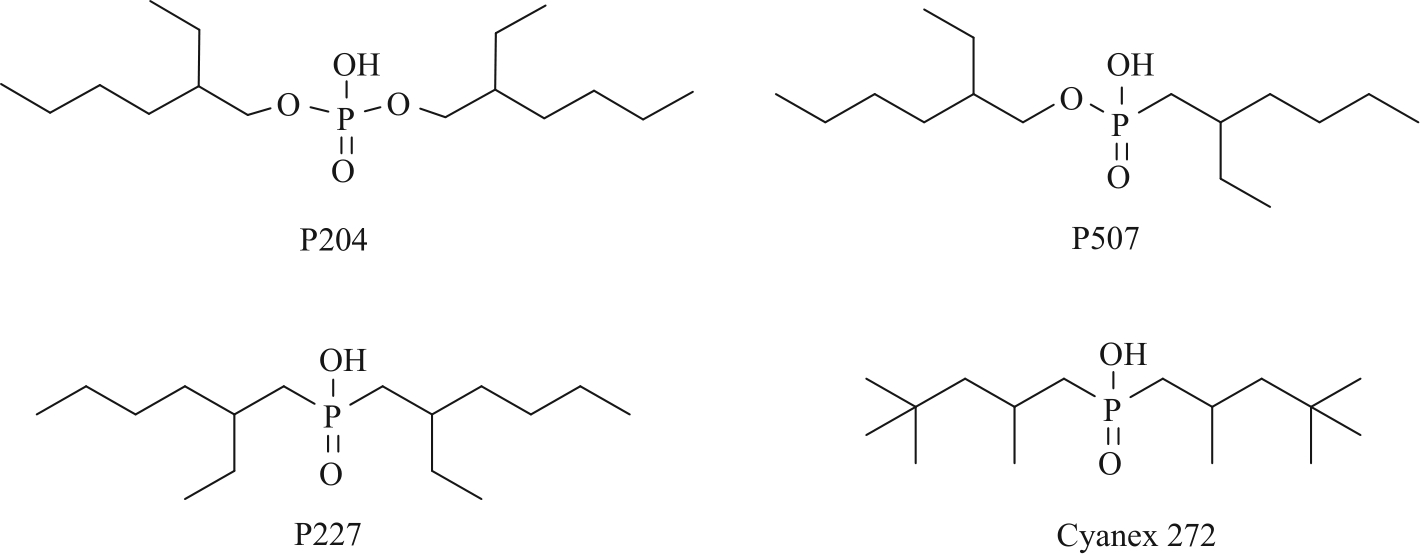
| Acidic organophosphates | P204/P507 | 1.5 g/L Ca2+, 1.5 g/L Mg2+, 30 g/L Li+ | Mg2+: 98.48% Ca2+: 99.05% Li+: 5.22% | [ |
| Cyanex 272-TBP | 0.06 mol/L Li+, 0.02 mol/L Co2+ | Li+: 5% Co2+: 90% | [ | |
| P227 | 30 g/L Li+ | Li+: 2.09% | [ | |
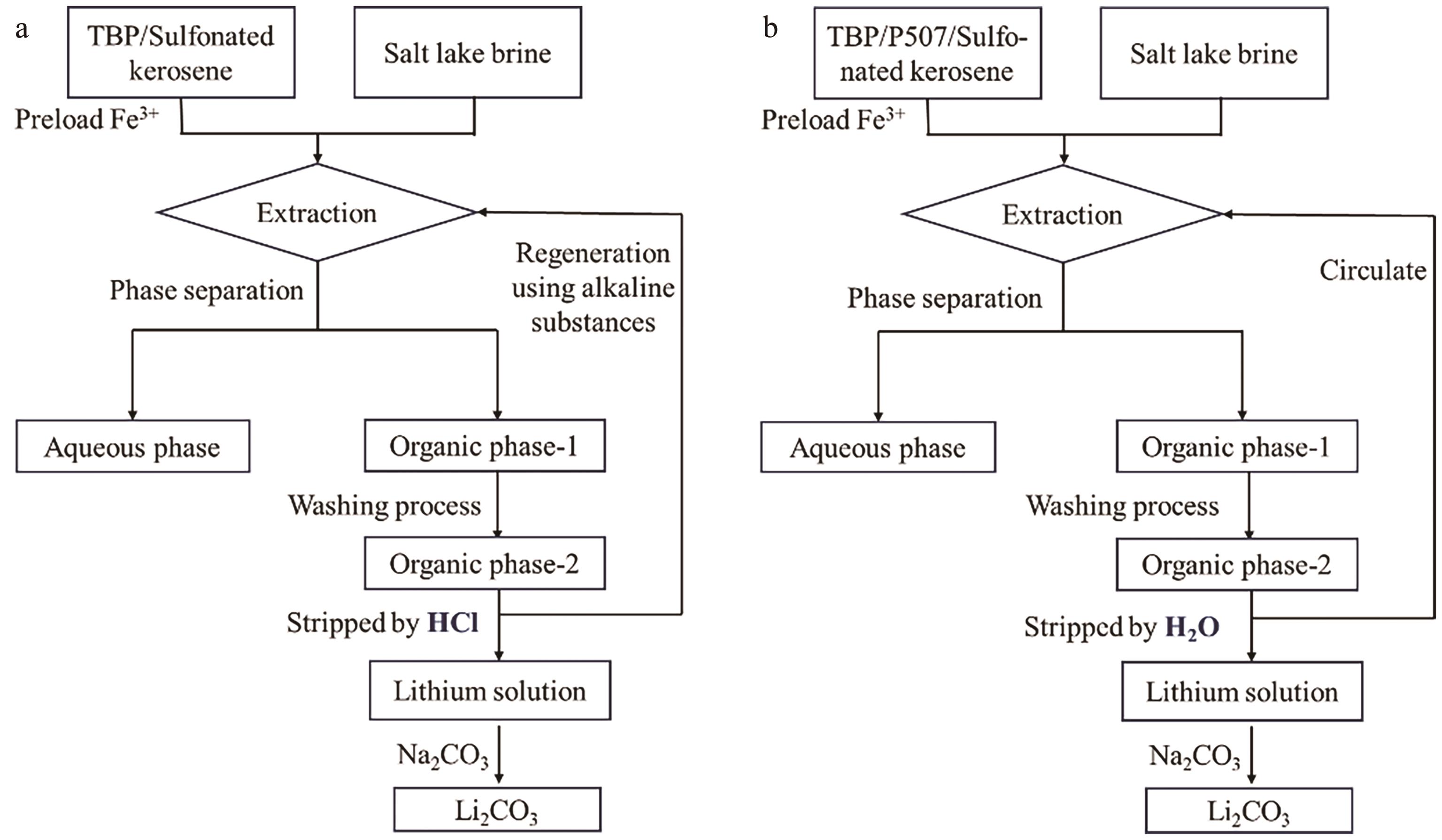
| Neutral organophosphates | TBP/P507-FeCl3 | 97 g/L Mg2+, 0.69 g/L Li+ | Mg2+: 2.6% Li+: 84.5% | [ |
| TBP/NaBPh4 | 0.35 g/L Li+, 96 g/L Mg2+ | Li+: 85.73% Mg2+: 0.44% | [ | |
| PVC/c-TBP | 0.2 mol/L Li+ | Li+: 27% | [ | |
| Macrocyclic molecules | Decahydronaphthalene-14-crown-4 | 0.01 mol/L Li+, Na+, K+, Rb+, Cs+ | Li+: 81%, K+, Rb+, Cs+: <1% | [ |
| Macrocyclic ionophore | 1 mol/L Li+ | Li+: 27% | [ | |
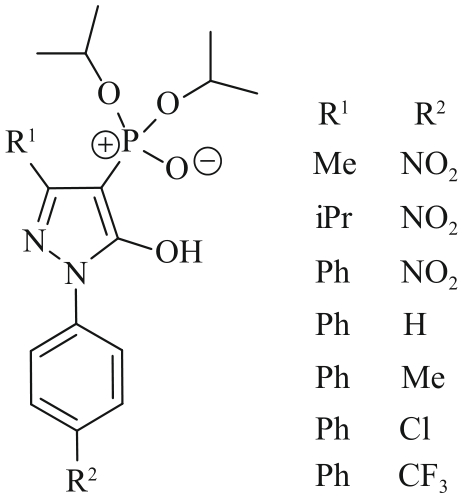
| Pyrazolone | Trifluoromethyl-4-phosphoryl -pyrazolones-TOPO | 0.01 mol/L Li+, Na+, K+, Mg2+, Ca2+ | Li+: 94% Na+, K+:<3.6% (pH=6.0) | [ |
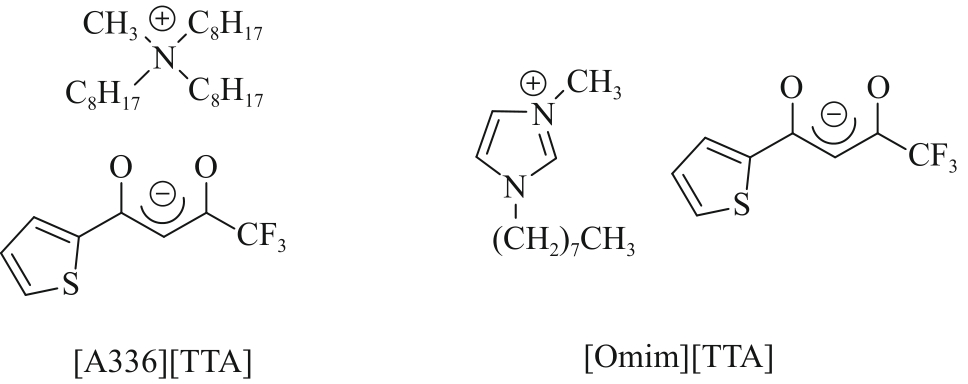
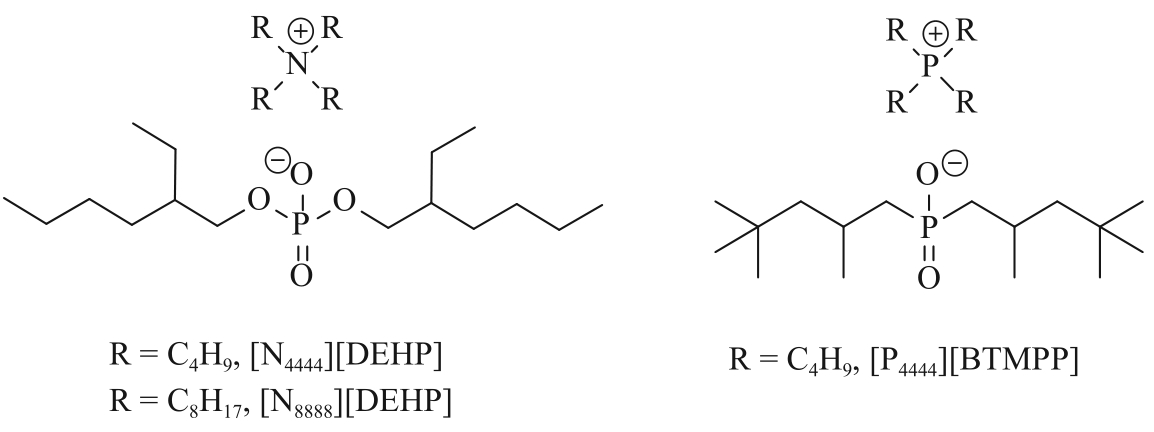
| Ionic liquids | [A336][TTA]-Cyanex 923 | 0.5 g/L Li+, 23 g/L Na+ | Li+: 98.52% Na+: 5.58% | [ |
| [Omim][TTA]-[Omim][NTf2] | 32 mmol/L Li+ | Li+: 95% | [ | |
| Deep eutectic solvents | HTTA-TOPO | 1.17 g/L Li+, 129.6 g/L Na+, 40.9 g/L K+ | Li+: 95.7% Na+: 1.1% K+:<0.01% | [ |
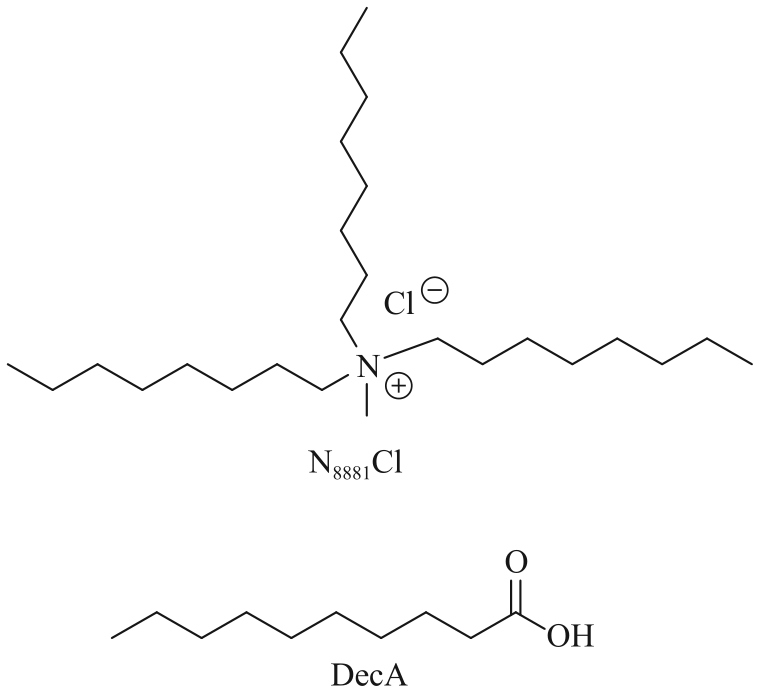
| N8881Cl/2DecA-P204 | 1 mg/L Li+ | Li+: 80% | [ |
表1 常用萃取体系
Table 1 Commonly used extraction systems
| Extractants | Organic phase | Aqueous phase | Extraction/% | Ref. |
|---|---|---|---|---|
| β-Diketone | HBTA-TOPO, NaOH Pre-saponification | 1.85 g/L Li+, 31.2 g/L Na+ | Li+: 80% | [ |
| Acidic organophosphates | P204/P507 | 1.5 g/L Ca2+, 1.5 g/L Mg2+, 30 g/L Li+ | Mg2+: 98.48% Ca2+: 99.05% Li+: 5.22% | [ |
| Cyanex 272-TBP | 0.06 mol/L Li+, 0.02 mol/L Co2+ | Li+: 5% Co2+: 90% | [ | |
| P227 | 30 g/L Li+ | Li+: 2.09% | [ | |
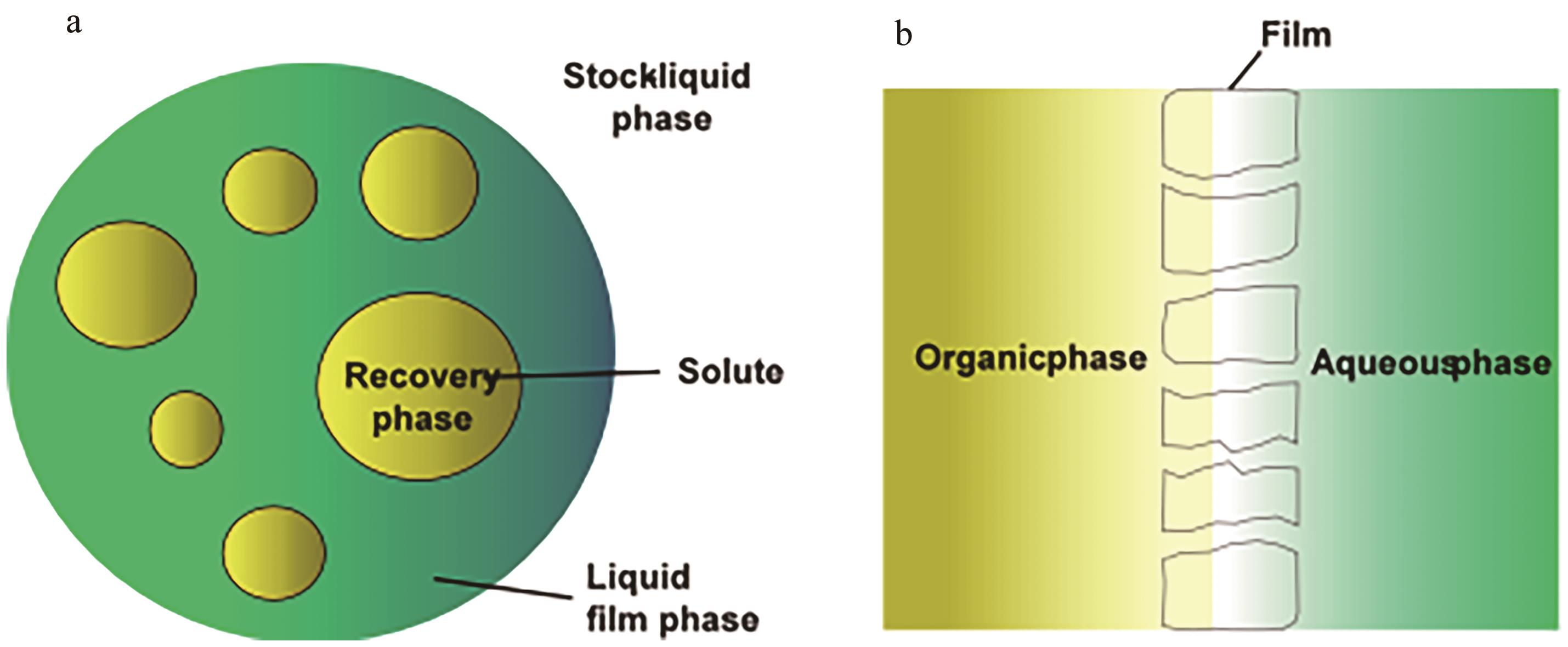
| Neutral organophosphates | TBP/P507-FeCl3 | 97 g/L Mg2+, 0.69 g/L Li+ | Mg2+: 2.6% Li+: 84.5% | [ |
| TBP/NaBPh4 | 0.35 g/L Li+, 96 g/L Mg2+ | Li+: 85.73% Mg2+: 0.44% | [ | |
| PVC/c-TBP | 0.2 mol/L Li+ | Li+: 27% | [ | |
| Macrocyclic molecules | Decahydronaphthalene-14-crown-4 | 0.01 mol/L Li+, Na+, K+, Rb+, Cs+ | Li+: 81%, K+, Rb+, Cs+: <1% | [ |
| Macrocyclic ionophore | 1 mol/L Li+ | Li+: 27% | [ | |
| Pyrazolone | Trifluoromethyl-4-phosphoryl -pyrazolones-TOPO | 0.01 mol/L Li+, Na+, K+, Mg2+, Ca2+ | Li+: 94% Na+, K+:<3.6% (pH=6.0) | [ |
| Ionic liquids | [A336][TTA]-Cyanex 923 | 0.5 g/L Li+, 23 g/L Na+ | Li+: 98.52% Na+: 5.58% | [ |
| [Omim][TTA]-[Omim][NTf2] | 32 mmol/L Li+ | Li+: 95% | [ | |
| Deep eutectic solvents | HTTA-TOPO | 1.17 g/L Li+, 129.6 g/L Na+, 40.9 g/L K+ | Li+: 95.7% Na+: 1.1% K+:<0.01% | [ |
| N8881Cl/2DecA-P204 | 1 mg/L Li+ | Li+: 80% | [ |
| 1 | GOODENOUGH J B, PARK K S. The Li-ion rechargeable battery: a perspective[J]. J Am Chem Soc, 2013, 135(4): 1167-1176. |
| 2 | DONALD I W, METCALFE B L, GERRARD L A. Interfacial reactions in glass-ceramic-to-metal seals[J]. J Am Ceram Soc, 2008, 91(3): 715-720. |
| 3 | PARIKH V P, AHMADI A, PAREKH M H, et al. Upcycling of spent lithium cobalt oxide cathodes from discarded lithium-ion batteries as solid lubricant additive[J]. ACS ES&T, 2019, 53(7): 3757-3763. |
| 4 | MJOS K D, ORVIG C. Metallodrugs in medicinal inorganic chemistry[J]. Chem Rev, 2014, 114(8): 4540-4563. |
| 5 | BIRCH N J. Inorganic pharmacology of lithium[J]. Chem Rev, 1999, 99(9): 2659-2682. |
| 6 | Mineral commodity summaries 2022[R]. US. Geological Survey, 2022. |
| 7 | KAVANAGH L, KEOHANE J, GARCIA-CABELLOS G, et al. Global lithium sources-industrial use and future in the electric vehicle industry: a review[J]. Resources, 2018, 7(3): 57. |
| 8 | 蒋晨啸, 陈秉伦, 张东钰, 等. 我国盐湖锂资源分离提取进展[J]. 化工学报, 2022, 73(2): 481-503. |
| JIANG C X, CHEN B L, ZHANG D Y, et al. Progress in isolating lithium resources from China salt lake brine[J]. CIESC J, 2022, 73(2): 481-503. | |
| 9 | YAKSIC A, TILTON J E. Using the cumulative availability curve to assess the threat of mineral depletion: the case of lithium[J]. Resour Policy, 2009, 34(4): 185-194. |
| 10 | MURPHY O, HAJI M N. A review of technologies for direct lithium extraction from low Li+ concentration aqueous solutions[J]. Front Chem Eng, 2022, 4: 1008680. |
| 11 | 苏慧, 朱兆武, 王丽娜, 等. 从盐湖卤水中提取与回收锂的技术进展及展望[J]. 材料导报, 2019, 33(13): 2119-2126. |
| SU H, ZHU Z W, WANG L N, et al. Advances and prospects of extracting and recovering lithium from salt lake brines[J]. Mater Rep, 2019, 33(13): 2119-2126. | |
| 12 | 黄维农, 孙之南, 王学魁, 等. 盐湖提锂研究和工业化进展[J]. 现代化工, 2008, 28(2): 14-19. |
| HUANG W N,SUN Z N,WANG X K, et al. Progress in industrialization for lithium extraction from salt lake[J]. Mod Chem Ind, 2008, 28(2): 14-19. | |
| 13 | KUMAR A, FUKUDA H, HATTON T A, et al. Lithium recovery from oil and gas produced water: a need for a growing energy industry[J]. ACS Energy Lett, 2019, 4(6): 1471-1474. |
| 14 | BAI Y, MURALIDHARAN N, SUN Y K, et al. Energy and environmental aspects in recycling lithium-ion batteries: concept of battery identity global passport[J]. Mater Today, 2020, 41: 304-315. |
| 15 | MASMOUDI A, ZANTE G, TRÉBOUET D, et al. Solvent extraction of lithium ions using benzoyltrifluoroacetone in new solvents[J]. Sep Purif Technol, 2021, 255: 117653. |
| 16 | ZHANG L, LI L, RUI H, et al. Lithium recovery from effluent of spent lithium battery recycling process using solvent extraction[J]. J Hazard Mater, 2020, 398: 122840. |
| 17 | FERNÁNDEZ-ESCALANTE E, IBAÑEZ R, SAN-ROMÁN M F. Selective lithium separation from desalination concentrates via the synergy of extractant mixtures[J]. Desalination, 2023, 556: 116525. |
| 18 | SUN Q, CHEN H, YU J. A density functional theory insight into the extraction mechanism of lithium recovery from alkaline brine by β-diketones[J]. AIChE J, 2023, 69(7): e18094. |
| 19 | HANADA T, GOTO M. Synergistic deep eutectic solvents for lithium extraction[J]. ACS Sustainable Chem Eng, 2021, 9(5): 2152-2160. |
| 20 | MENG X, LONG Y, TIAN Y, et al. Electro-membrane extraction of lithium with D2EHPA/TBP compound extractant[J]. Hydrometallurgy, 2021, 202: 105615. |
| 21 | MAHMOUDI S, BABAKHANI A, NOORI M, et al. Synergistic extraction and separation of cobalt and lithium using D2EHPA and Cyanex 272[J]. Min Metall Explor, 2022, 39(2): 777-792. |
| 22 | LIU T, CHEN J, LI H, et al. High-efficiency removal of calcium and magnesium from lithium-concentrated solution via counter-current extraction using di-(2-ethylhexyl)phosphinic acid[J]. ACS Sustainable Chem Eng, 2022, 10(2): 967-974. |
| 23 | LI Z, BINNEMANS K. Opposite selectivities of tri-n-butyl phosphate and Cyanex 923 in solvent extraction of lithium and magnesium[J]. AIChE J, 2021, 67(7): e17219. |
| 24 | SUN Q, CHEN H, YU J. Investigation on the lithium extraction process with the TBP-FeCl3 solvent system using experimental and DFT methods[J]. Ind Eng Chem Res, 2022, 61(13): 4672-4682. |
| 25 | SU H, LI Z, ZHANG J, et al. Recovery of lithium from salt lake brine using a mixed ternary solvent extraction system consisting of TBP, FeCl3 and P507[J]. Hydrometallurgy, 2020, 197: 105487. |
| 26 | LI Z, BINNEMANS K. Mechanism of ferric chloride facilitating efficient lithium extraction from magnesium-rich brine with tri-n-butyl phosphate[J]. Ind Eng Chem Res, 2021, 60(23): 8538-8547. |
| 27 | SU H, TAN B, ZHANG J, et al. Modelling of lithium extraction with TBP/P507-FeCl3 system from salt-lake brine[J]. Sep Purif Technol, 2022, 282: 120110. |
| 28 | GOHIL H, CHATTERJEE S, YADAV S, et al. An ionophore for high lithium loading and selective capture from brine[J]. Inorg Chem, 2019, 58(11): 7209-7219. |
| 29 | CUI L, LI S, KANG J, et al. A novel ion-pair strategy for efficient separation of lithium isotopes using crown ethers[J]. Sep Purif Technol, 2021, 274: 118989. |
| 30 | SADAMATSU H, HANADA T, MORISADA S, et al. Comprehensive comparison of alkali metal extraction with a series of calix[4]arene derivatives with propyl and/or acetic acid groups[J]. J Inclusion Phenom Macrocyclic Chem, 2016, 84(1): 87-97. |
| 31 | KURNIAWAN Y S, SATHULURI R R, OHTO K, et al. A rapid and efficient lithium-ion recovery from seawater with tripropyl-monoacetic acid calix[4]arene derivative employing droplet-based microreactor system[J]. Sep Purif Technol, 2019, 211: 925-934. |
| 32 | KURNIAWAN Y S, SATHULURI R R, OHTO K, et al. New concept for the study of the fluid dynamics of lithium extraction using Calix[4]arene derivatives in T-type microreactor systems[J]. Separations, 2021, 8(5): 70. |
| 33 | ZHANG J, WENZEL M, STEUP J, et al. 4-Phosphoryl pyrazolones for highly selective lithium separation from alkali metal ions[J]. Chem-Eur J, 2022, 28(1): e202103640. |
| 34 | ZHANG J, TANJEDREW N, WENZEL M, et al. Selective separation of lithium, magnesium and calcium using 4-phosphoryl pyrazolones as pH-regulated receptors[J]. Angew Chem Int Ed, 2023, 62(13): e202216011. |
| 35 | WANG J, YANG S, ZHANG X, et al. Extraction mechanism of lithium from the alkali solution with diketonate-based ionic liquid extractants[J]. Energy Fuels, 2020, 34(9): 11581-11589. |
| 36 | CAI C, HANADA T, FAJAR A T N, et al. An ionic liquid extractant dissolved in an ionic liquid diluent for selective extraction of Li(Ⅰ) from salt lakes[J]. Desalination, 2021, 509: 115073. |
| 37 | SHI C, JING Y, XIAO J, et al. Solvent extraction of lithium from aqueous solution using non-fluorinated functionalized ionic liquids as extraction agents[J]. Sep Purif Technol, 2017, 172: 473-479. |
| 38 | SHI C, JING Y, XIAO J, et al. Liquid-liquid extraction of lithium using novel phosphonium ionic liquid as an extractant[J]. Hydrometallurgy, 2017, 169: 314-320. |
| 39 | WANG Y, LIU H, FAN J, et al. Recovery of lithium ions from salt lake brine with a high magnesium/lithium ratio using heteropolyacid ionic liquid[J]. ACS Sustainable Chem Eng, 2019, 7(3): 3062-3072. |
| 40 | ZHENG H, DONG T, SHA Y, et al. Selective extraction of lithium from spent lithium batteries by functional ionic liquid[J]. ACS Sustainable Chem Eng, 2021, 9(20): 7022-7029. |
| 41 | 白瑞兵, 王均凤, 王道广, 等. 离子液体基萃取体系用于卤水中锂分离的研究进展[J]. 化工进展, 2021, 40(6): 3224-3238. |
| BAI R B, WANG J F, WANG D G, et al. Research progress of ionic liquid-based extraction separation of lithium from brine[J]. Chem Ind Eng Prog, 2021, 40(6): 3224-3238. | |
| 42 | LUO H, YAO H, WANG X, et al. Selective recovery of lithium from mother liquor of Li2CO3 by synergistic hydrophobic deep eutectic solvents: performance and mechanistic insight[J]. Sep Purif Technol, 2023, 313: 123353. |
| 43 | YU L Y, WU K J, HE C H, Tailoring hydrophobic deep eutectic solvent for selective lithium recovery from dilute aqueous solutions[J]. Sep Purif Technol, 2022, 281: 119928. |
| 44 | ZHANG J, LIU Y, LIU W, et al. Mechanism study on the synergistic effect and emulsification formation of phosphine oxide with β-diketone for lithium extraction from alkaline systems[J]. Sep Purif Technol, 2021, 279: 119648. |
| 45 | HANO T, MATSUMOTO M, OHTAKE T, et al. Recovery of lithium from geothermal water by solvent extraction technique[J]. Solvent Extr Ion Exch, 1992, 10(2): 195-206. |
| 46 | SHI D, CUI B, LI L, et al. Removal of calcium and magnesium from lithium concentrated solution by solvent extraction method using D2EHPA[J]. Desalination, 2020, 479: 114306. |
| 47 | LU J, STEVENS G W, MUMFORD K A. Development of heterogeneous equilibrium model for lithium solvent extraction using organophosphinic acid[J]. Sep Purif Technol, 2021, 276: 119307. |
| 48 | NELLI J R, ARTHUR T E. Recovery of lithium from bitterns: US, US3537813A[P]. 1970. |
| 49 | SHI D, CUI B, LI L, et al. Lithium extraction from low-grade salt lake brine with ultrahigh Mg/Li ratio using TBP-kerosene- FeCl3 system[J]. Sep Purif Technol, 2019, 211: 303-309. |
| 50 | SU H, LI Z, ZHANG J, et al. Combining selective extraction and easy stripping of lithium using a ternary synergistic solvent extraction system through regulation of Fe3+ coordination[J]. ACS Sustainable Chem Eng, 2020, 8(4): 1971-1979. |
| 51 | LI R, WANG Y, DUAN W, et al. Selective extraction of lithium ions from salt lake brines using a tributyl phosphate-sodium tetraphenyl boron-phenethyl isobutyrate system[J]. Desalination, 2023, 555: 116543. |
| 52 | 任其龙, 邢华斌, 关怡新. 萃取过程强化技术[M]. 北京: 化学工业出版社, 2020: 383. |
| REN Q L, XING H B, GUAN Y X. Extraction process intensification[M]. Beijing: Chemical Industry Press, 2020: 383. | |
| 53 | SONG J, LI X M, ZHANG Y, et al. Hydrophilic nanoporous ion-exchange membranes as a stabilizing barrier for liquid-liquid membrane extraction of lithium ions[J]. J Membr Sci, 2014, 471: 372-380. |
| 54 | ZHANG C, MU Y, ZHAO S, et al. Lithium extraction from synthetic brine with high Mg2+/Li+ ratio using the polymer inclusion membrane[J]. Desalination, 2020, 496: 114710. |
| 55 | THORNTON J D. Science and practice of liquid-liquid extraction[J]. Chem Environ Sci Med,1992, 2: 1039. |
| 56 | ZHAO Z, LIU G, JIA H, et al. Sandwiched liquid-membrane electrodialysis: lithium selective recovery from salt lake brines with high Mg/Li ratio[J]. J Membr Sci, 2020, 596: 117685. |
| 57 | LUO Y, MARETS N, KATO T. Selective lithium ion recognition in self-assembled columnar liquid crystals based on a lithium receptor[J]. Chem Sci, 2018, 9(3): 608-616. |
| 58 | KOBIRO K, HIRO T, MATSUOKA T, et al. Decalino-14-crown-4. new type of lithium ion selective ionophore[J]. Bull Chem Soc Jpn, 1988, 61(11): 4164-4166. |
| 59 | ROGERS R D, SEDDON K R. Ionic liquids-solvents of the future?[J]. Sci, 2003, 302(5646): 792-793. |
| 60 | SUN X, LUO H, DAI S. Ionic liquids-based extraction: a promising strategy for the advanced nuclear fuel cycle[J]. Chem Rev, 2012, 112(4): 2100-2128. |
| 61 | ABBOTT A P, CAPPER G, DAVIES D L, et al. Novel solvent properties of choline chloride/urea mixtures[J]. Chem Commun, 2003(1): 70-71. |
| [1] | 张轶, 陈宇童, 师靖宇, 黄科科. 石榴石Li7La3Zr2O12固态电解质电导率优化策略研究进展[J]. 应用化学, 2024, 41(4): 484-495. |
| [2] | 陈胜, 胡祖飞, 曹洪美, 赵振华, 张宇栋. 镁掺杂高镍三元正极材料LiNi0.90Co0.05Mn0.05O2的合成与性能[J]. 应用化学, 2024, 41(4): 568-576. |
| [3] | 罗二桂, 唐涛, 王艺, 张俊明, 常宇虹, 胡天军, 贾建峰. 两电子氧还原制备过氧化氢:贵金属催化剂的几何与电子结构调控的研究进展[J]. 应用化学, 2023, 40(8): 1063-1076. |
| [4] | 陈兵帅, 卓海涛, 黄书, 陈少军. 高性能硅基负极聚合物粘结剂的研究进展[J]. 应用化学, 2023, 40(5): 625-639. |
| [5] | 伍凡, 田贺元, 刘鹏, 孙立伟, 张一波, 杨向光. 高氧空位尖晶石型锰基催化剂用于低温NH3-SCR反应[J]. 应用化学, 2023, 40(5): 697-707. |
| [6] | 胡方正, 高兴, 刘雷, 袁天恒, 曹宁, 李凯, 王亚涛, 李建华, 连慧琴, 汪晓东, 崔秀国. 锂离子电池黑磷负极的储能优势及其优化的研究进展[J]. 应用化学, 2023, 40(4): 571-582. |
| [7] | 王路飞, 甄蒙蒙, 沈伯雄. 贫电解液下电催化剂对调控锂硫电池性能的研究进展[J]. 应用化学, 2023, 40(2): 188-209. |
| [8] | 潘阳, 鲁惠玲, 李浩, 潘建明. 贵金属离子印迹聚合物的制备及其应用的研究进展[J]. 应用化学, 2023, 40(10): 1359-1375. |
| [9] | 高静霞, 王子安, 张连明, 李建平. 大环化合物在高选择性分子印迹识别体系中的研究进展[J]. 应用化学, 2023, 40(1): 24-39. |
| [10] | 林锦, 王芳珠, 吕灵灵. 工业原料制备大孔拟薄水铝石及在异佛尔酮选择性加氢中的应用[J]. 应用化学, 2023, 40(1): 79-90. |
| [11] | 郭佳昕, 刘洋, 徐长山, 刘晓男, 程亮. 不同pH值下MgO NPs悬浮液中Mg2+质量浓度的动态变化及其对小麦生长的影响[J]. 应用化学, 2022, 39(9): 1401-1411. |
| [12] | 张丹, 尚润梅, 赵振涛, 李君华, 邢锦娟. V/Ce-Al2O3催化甲醇选择性氧化制备二甲氧基甲烷[J]. 应用化学, 2022, 39(9): 1429-1436. |
| [13] | 王恩通, 杨林芳. 高比容量锂离子电池正极材料LiNi0.6Co0.2Mn0.2O2的制备及性能[J]. 应用化学, 2022, 39(8): 1209-1215. |
| [14] | 宋林虎, 李世友, 王洁, 张晶晶, 张宁霜, 赵冬妮, 徐菲. 锂离子电池电解液除酸除水添加剂的研究进展[J]. 应用化学, 2022, 39(5): 697-706. |
| [15] | 王欣, 张冬, 杜菲. 单原子催化剂在锂硫电池中的研究进展[J]. 应用化学, 2022, 39(4): 513-527. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||