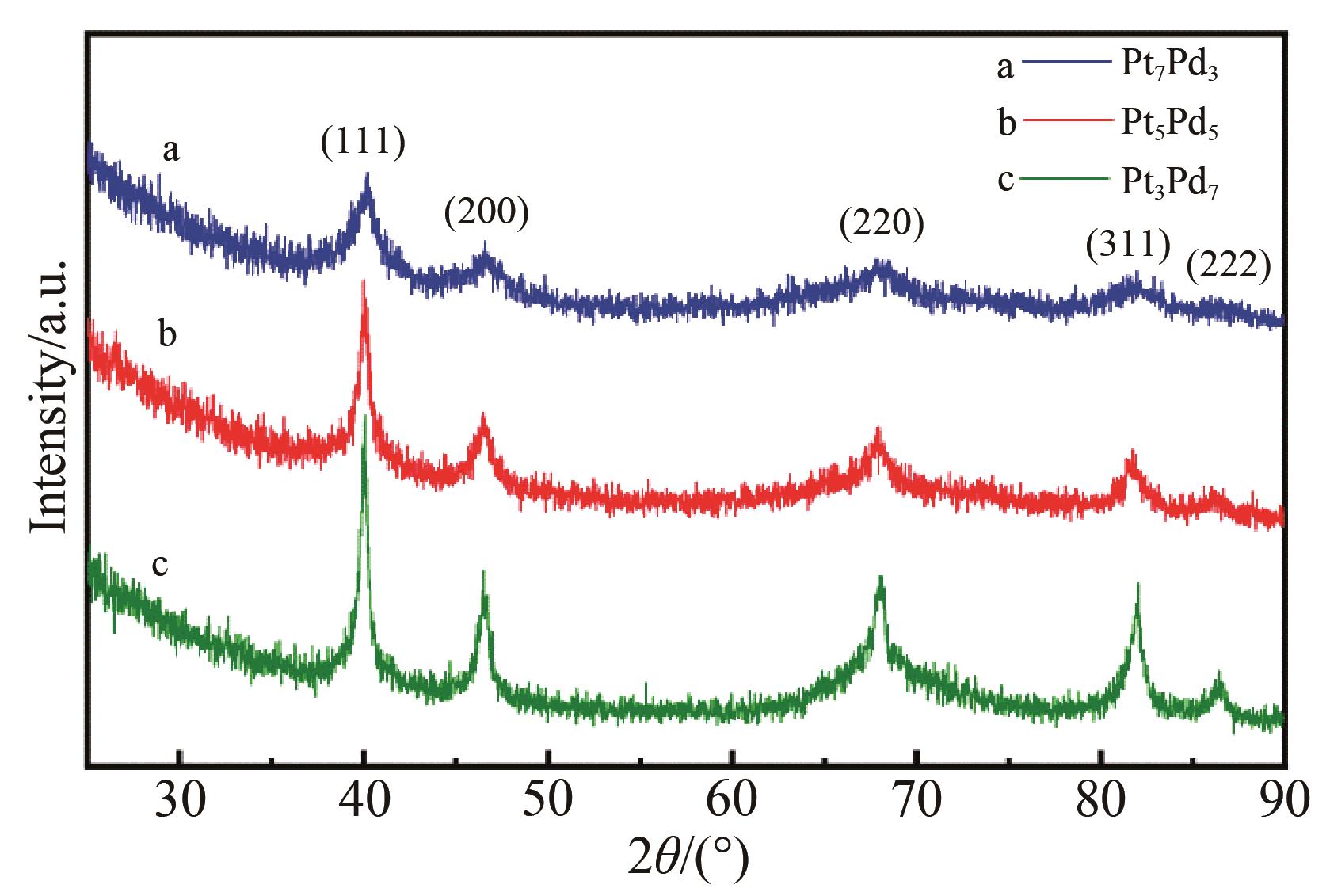
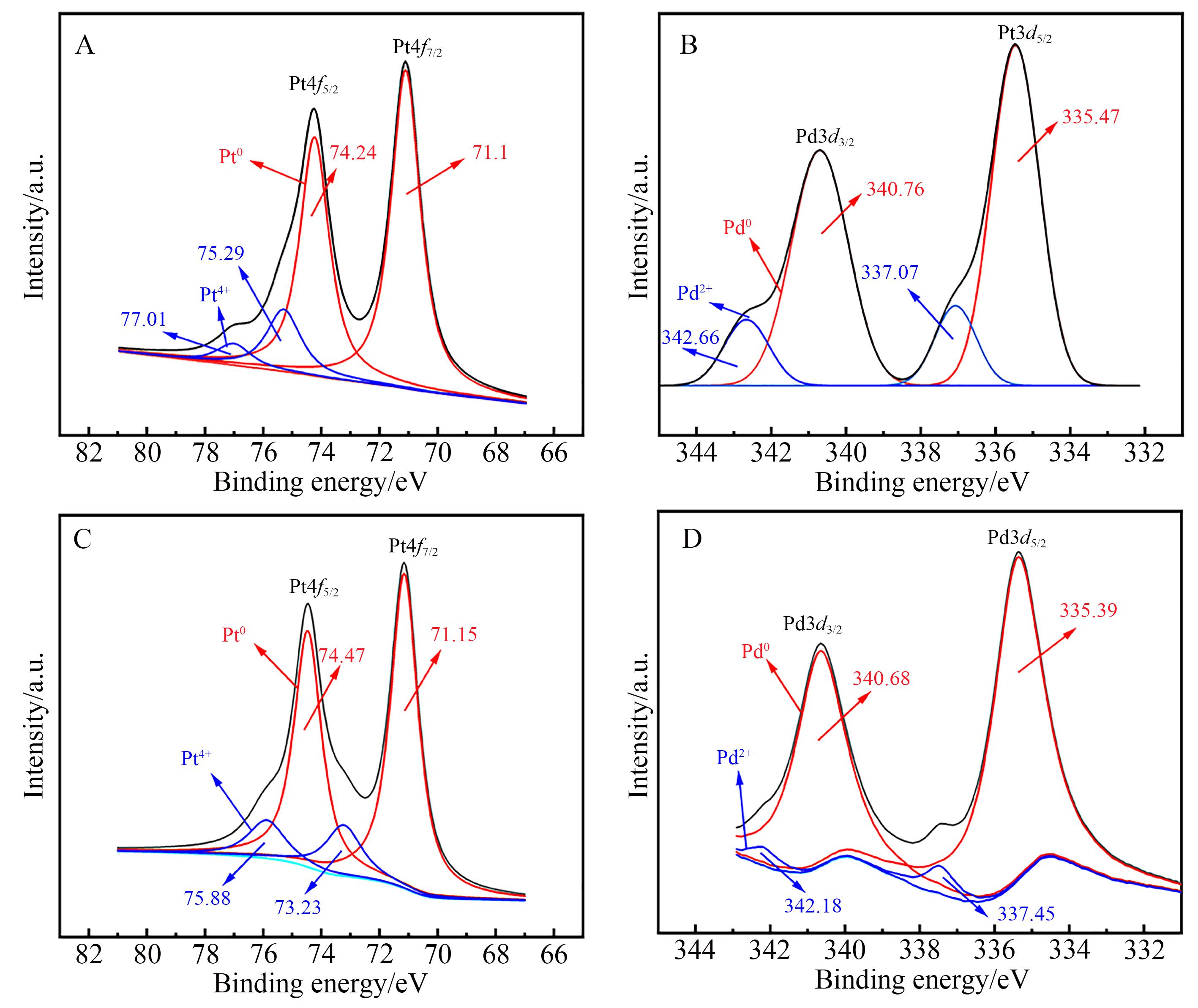
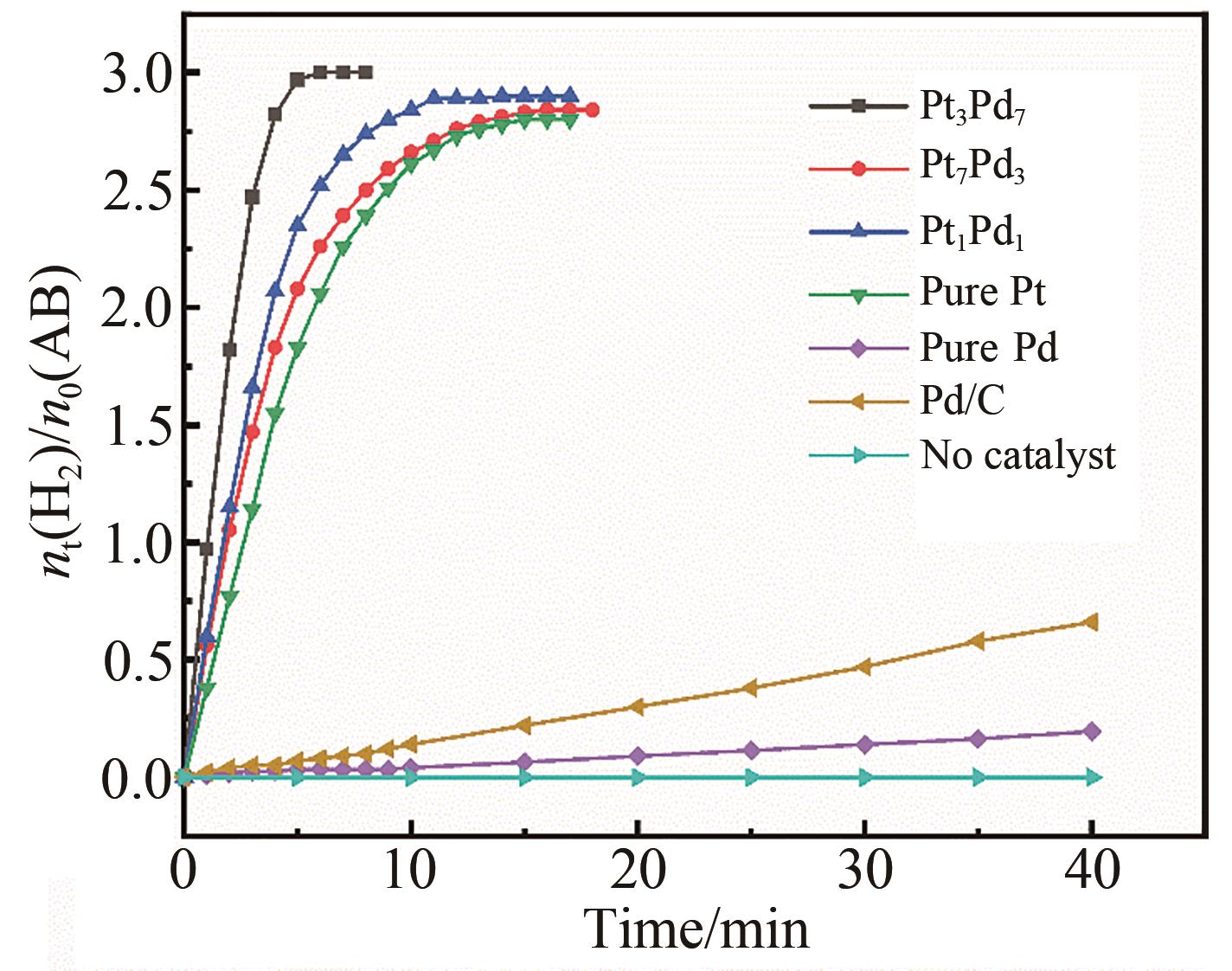
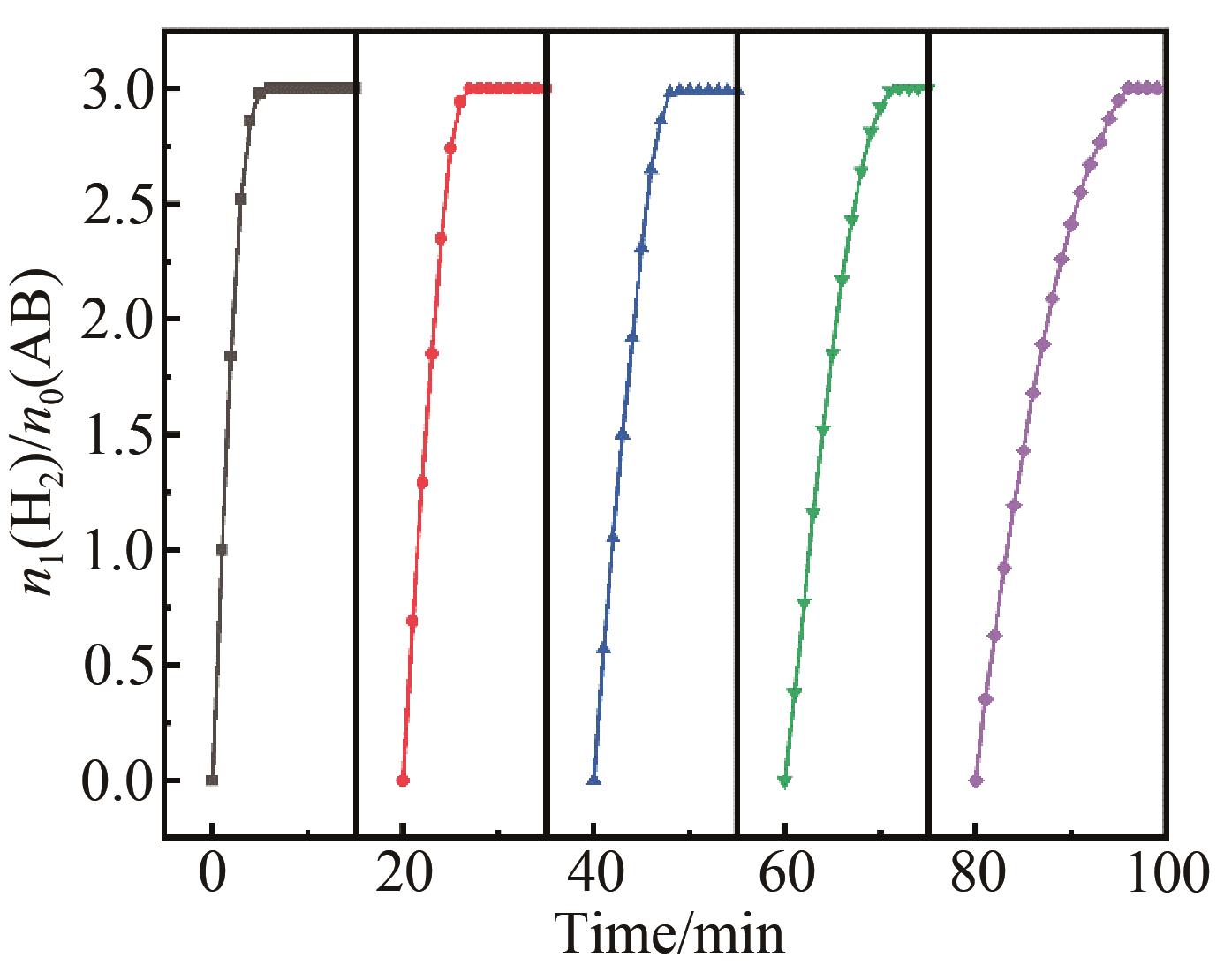
Chinese Journal of Applied Chemistry ›› 2023, Vol. 40 ›› Issue (4): 597-609.DOI: 10.19894/j.issn.1000-0518.220321
• Energy Materials • Previous Articles Next Articles
Ionic Liquid-Modulated Synthesis of Pt-Pd Bimetallic Nanomaterials and Their Catalytic Performance for Ammonia Borane Hydrolysis to Generate Hydrogen
Hui-Hui LI, Kai-Sheng YAO( ), Ya-Nan ZHAO, Li-Na FAN, Yu-Lin TIAN, Wei-Wei LU(
), Ya-Nan ZHAO, Li-Na FAN, Yu-Lin TIAN, Wei-Wei LU( )
)
- Chemical Engineering & Pharmaceutics School,Henan University of Science & Technology,Luoyang 471023,China
-
Received:2022-10-07Accepted:2023-02-22Published:2023-04-01Online:2023-04-17 -
Contact:Kai-Sheng YAO,Wei-Wei LU -
About author:luwei1980@126.com
ksyao@haust.edu.cn
-
Supported by:the National Natural Science Foundation of China(21673067);the Key Science and Technology Project of Henan Province(192102210043)
CLC Number:
Cite this article
Hui-Hui LI, Kai-Sheng YAO, Ya-Nan ZHAO, Li-Na FAN, Yu-Lin TIAN, Wei-Wei LU. Ionic Liquid-Modulated Synthesis of Pt-Pd Bimetallic Nanomaterials and Their Catalytic Performance for Ammonia Borane Hydrolysis to Generate Hydrogen[J]. Chinese Journal of Applied Chemistry, 2023, 40(4): 597-609.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.220321
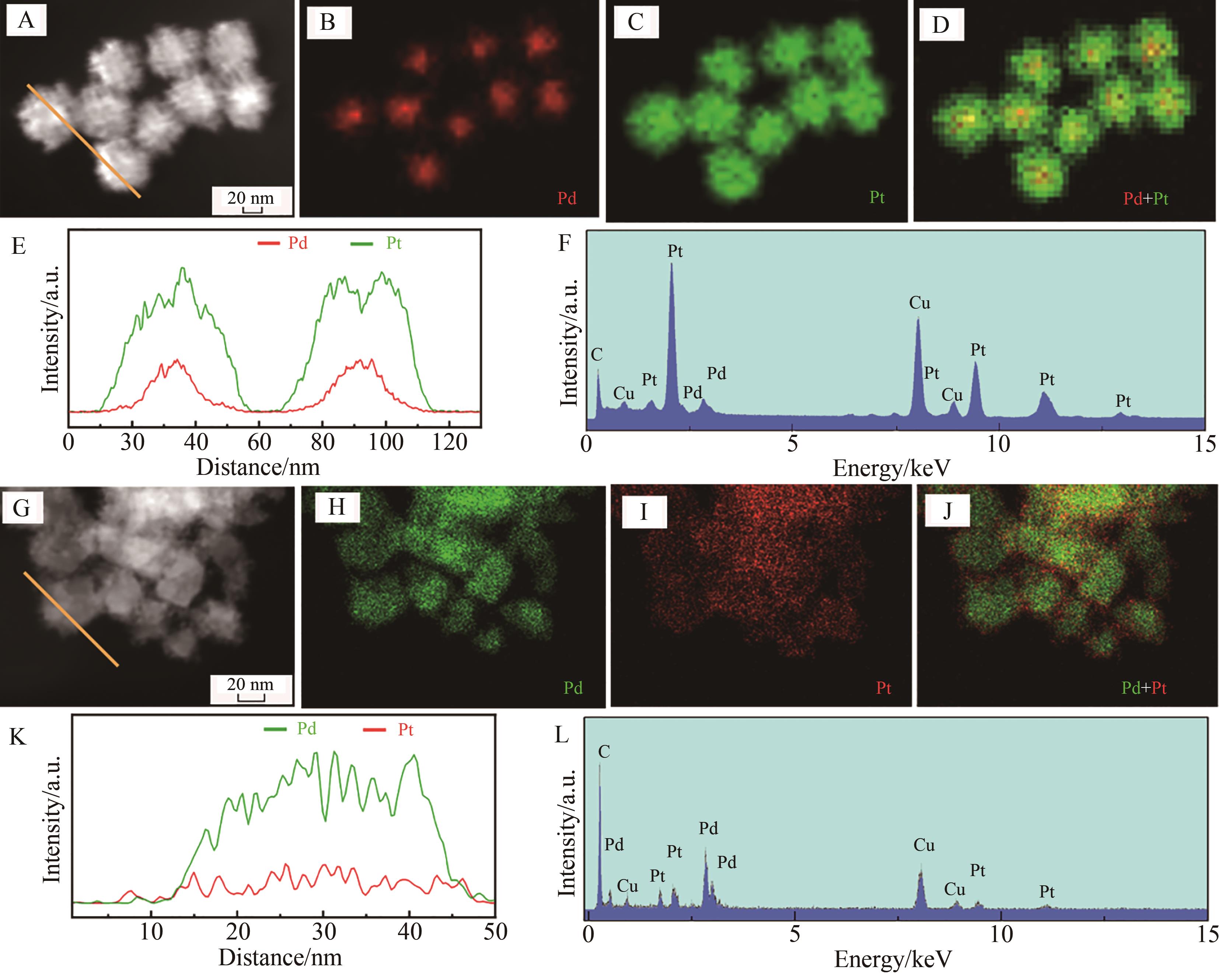
Fig.2 The as-prepared Pt7Pd3 and Pt3Pd7 samples: (A) HAADF-STEM image, (B-D) elemental mapping images, (E) HAADF-STEM compositional curves of Pd (red lines) and Pt (green lines) along with the orange line in (A), (F) EDS spectra of Pt7Pd3 sample; (G) HAADF-STEM image, (H-J) elemental mapping images, (K) HAADF-STEM compositional curves of Pd (green lines) and Pt (red lines) along with the orange line in (G) and (L) EDS spectra of Pt3Pd7 sample
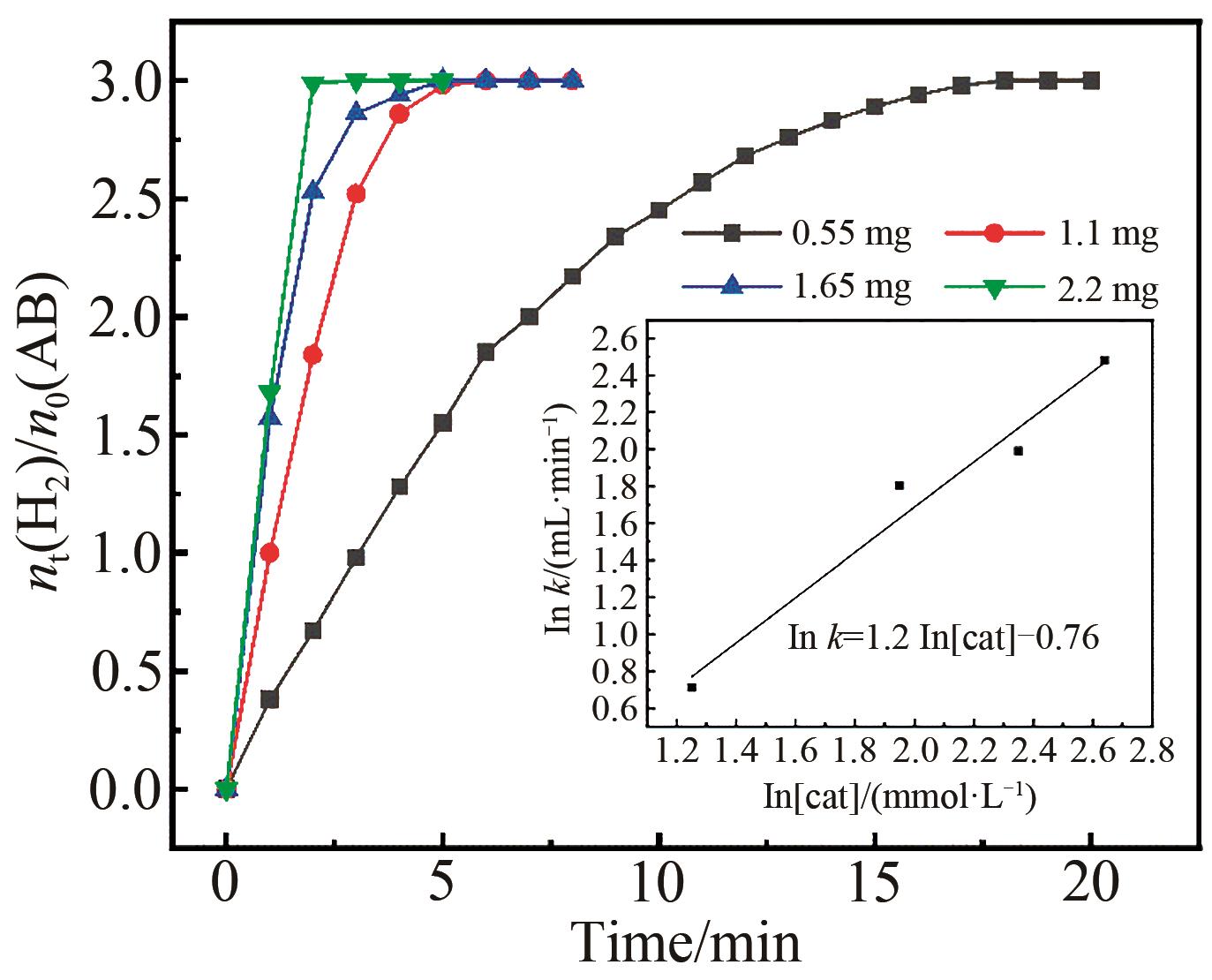
Fig.8 Plots of time versus hydrogen volume generated from AB catalyzed by Pt3Pd7 with different catalyst amounts; Inset: plot of hydrogen generation rate versus Pt3Pd7 concentration (both in logaritmic scale)
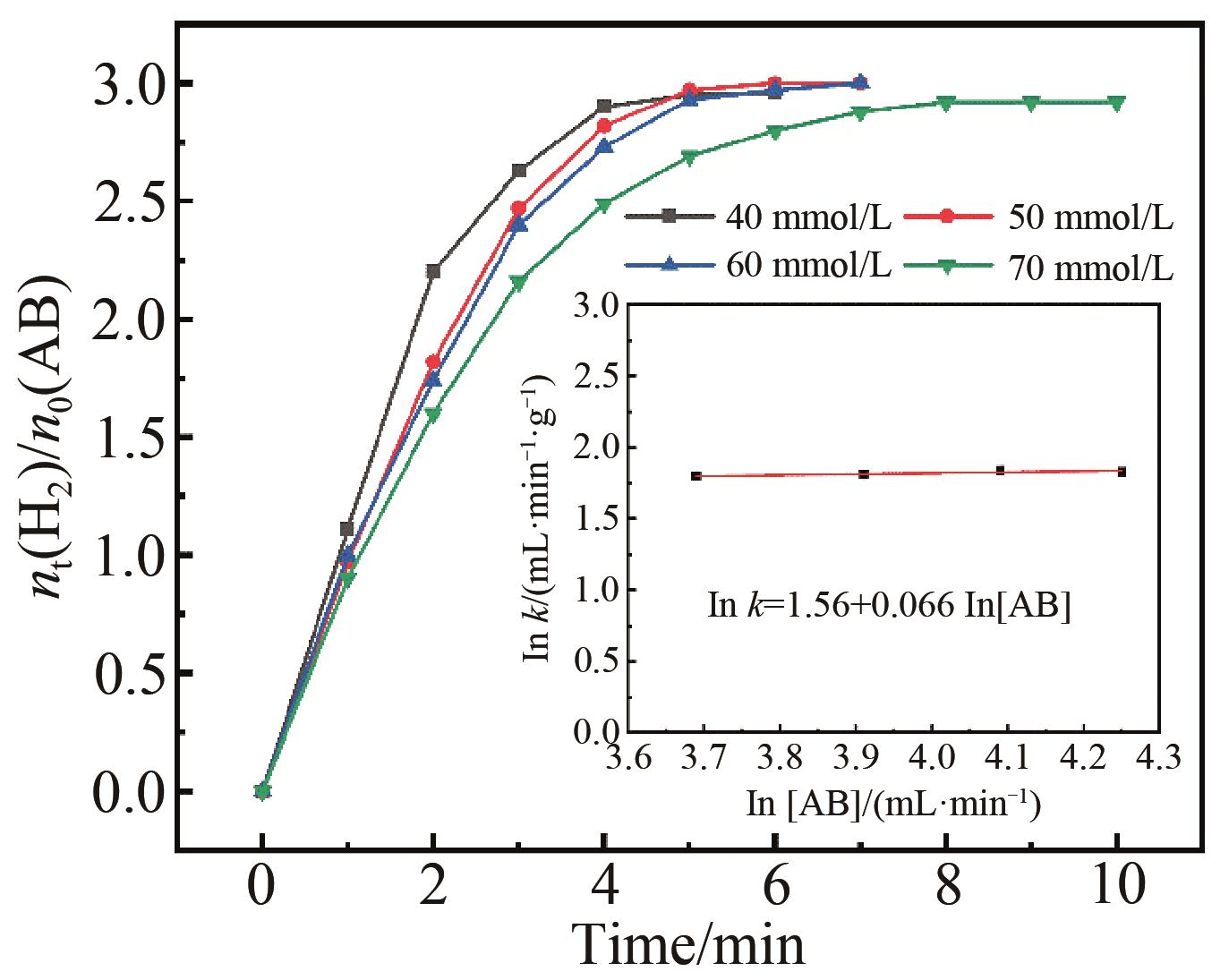
Fig.9 Plots of moles of H2 versus time for the hydrolysis of AB at different concentrations (40,50,60,70 mmol/L) keeping Pt3Pd7 catalyst concentration at 7.0 mmol/L; Inset: plot of hydrogen generation rate versus AB concentration (both in logaritmic scale)
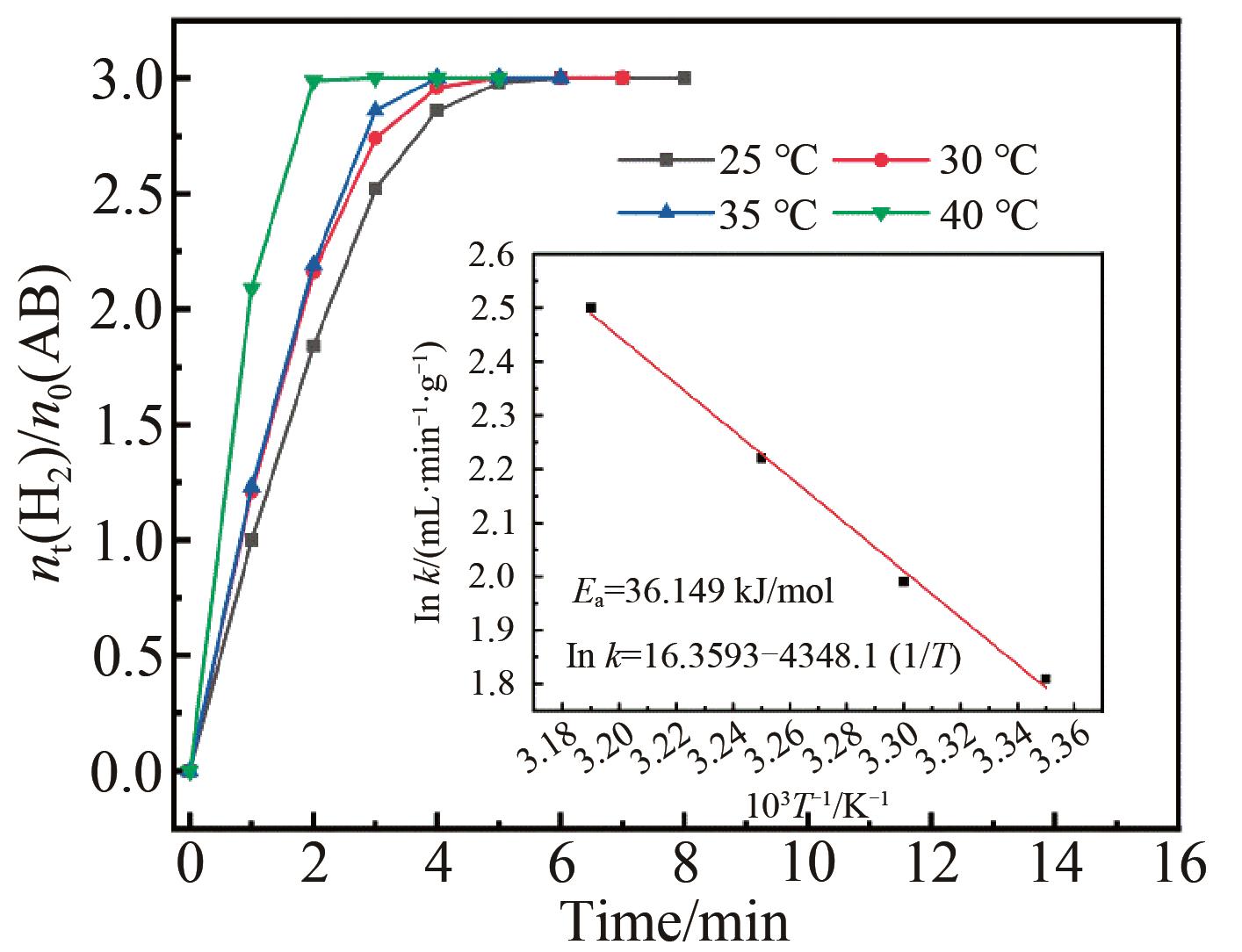
Fig.10 Plots of equivalent H2 produced by per mole of AB hydrolysis versus time in the presence of porous Pt3Pd7 catalyst at different temperature; Inset: the linear fitting of Arrhenius equation between ln k and 1000/T
| Catalysts | Temperature/℃ | TOF/(mol·mol-1·min-1) | Ea/(kJ·mol-1) | Ref. |
|---|---|---|---|---|
| Pt3Pd7 | 25 | 35.72 | 36.15 | This work |
| PdPt spherical NPs | 25 | 22.5 | 57.3 | [ |
| AuCo/CNT-2 | 25 | 8.47 | 34.83 | [ |
| Au@AuCo/CNT | 25 | 13.24 | 41.91 | [ |
| 1/1000Pt+Ni/CNT | 25 | 17.8 | - | [ |
| 1/100Pt+Ni/CNT | 25 | 33.3 | - | [ |
| Pd80Ni20-CeO2/rGO | 25 | 30.5 | - | [ |
| Ru1Co1/γ-Al2O3 | 65 | 32.9 | 47 | [ |
Table 1 TOF and Ea values for AB hydrolysis to produce hydrogen with different catalysts
| Catalysts | Temperature/℃ | TOF/(mol·mol-1·min-1) | Ea/(kJ·mol-1) | Ref. |
|---|---|---|---|---|
| Pt3Pd7 | 25 | 35.72 | 36.15 | This work |
| PdPt spherical NPs | 25 | 22.5 | 57.3 | [ |
| AuCo/CNT-2 | 25 | 8.47 | 34.83 | [ |
| Au@AuCo/CNT | 25 | 13.24 | 41.91 | [ |
| 1/1000Pt+Ni/CNT | 25 | 17.8 | - | [ |
| 1/100Pt+Ni/CNT | 25 | 33.3 | - | [ |
| Pd80Ni20-CeO2/rGO | 25 | 30.5 | - | [ |
| Ru1Co1/γ-Al2O3 | 65 | 32.9 | 47 | [ |
| 1 | ZHAN W W, ZHU Q L, XU Q. Dehydrogenation of ammonia borane by metal nanoparticle catalysts[J]. ACS Catal, 2016, 6(10): 6892-6905. |
| 2 | 刘林昌, 郭亚君, 朱红林, 等. 负载型超细纳米材料催化氨硼烷水解制氢的研究进展[J]. 应用化学, 2021, 38(11): 1405-1422. |
| LIU L C, GUO Y J, ZHU H L, et al. Research progress on supported ultrafine nano-catalysts for hydrolytic dehydrogenation of ammonia borane[J]. Chin J Appl Chem, 2021, 38(11): 1405-1422. | |
| 3 | MBOYI C D, POINSOT D, ROGER J, et al. The hydrogen-storage challenge: nanoparticles for metal-catalyzed ammonia borane dehydrogenation[J]. Small, 2021, 17: 2102759. |
| 4 | HE T, PACHFULE P, WU H, et al. Hydrogen carriers[J]. Nat Rev Mater, 2016, 1: 16059. |
| 5 | WEN M C, CUI Y W, KUWAHARA Y, et al. Non-noble-metal nanoparticle supported on metal-organic framework as an efficient and durable catalyst for promoting H2 production from ammonia borane under visible light irradiation[J]. ACS Appl Mater Interfaces, 2016, 8: 21278-21284. |
| 6 | ZHANG J K, CHEN C Q, YAN W J, et al. Ni nanoparticles supported on CNTs with excellent activity produced by atomic layer deposition for hydrogen generation from the hydrolysis of ammonia borane[J]. Catal Sci Technol, 2017, 7: 322-329. |
| 7 | ZOU H, JIN B, WANG R W, et al. Iodide-mediated templating synthesis of highly porous rhodium nanospheres for enhanced dehydrogenation of ammonia borane[J]. J Mater Chem A, 2018, 6(47): 24166-24174. |
| 8 | ÖZHAVA D, KILIÇASLAN N Z, ÖZKAR S. PVP-stabilized nickel(o) nanoparticles as catalyst in hydrogen generation from the methanolysis of hydrazine borane or ammonia borane[J]. Appl Catal B: Environ, 2015, 162: 573-582. |
| 9 | HU H W, XIN J H, HU H, et al. Synthesis and stabilization of metal nanocatalysts for reduction reactions-a review[J]. J Mater Chem A, 2015, 3(21): 11157-11182. |
| 10 | YAN Y C, DU J S, GILROY K D, et al. Intermetallic nanocrystals: syntheses and catalytic applications[J]. Adv Mater, 2017, 29(14): 1605997. |
| 11 | HONG W, WANG J, WANG E K. Facile synthesis of highly active PdAu nanowire networks as self-supported electrocatalyst for ethanol electrooxidation[J]. ACS Appl Mater Interfaces, 2014, 6(12): 9481-9487. |
| 12 | ZHANG J, LI H B, ZHANG H, et al. Porously hierarchical Cu@Ni cubic-cage microstructure: very active and durable catalyst for hydrolytically liberating H2 gas from ammonia borane[J]. Renew Energy, 2016, 99: 1038-1045. |
| 13 | JIANG B, LI C, ÖMER D, et al. Mesoporous metallic rhodium nanoparticles[J]. Nat Commun, 2017, 8: 15581. |
| 14 | LAMY C. Electrocatalytic oxidation of organic compounds on noble metals in aqueous solution[J]. Electrochim Acta, 1984, 29(11): 1581-1588. |
| 15 | ZHANG H, JIN M, XIA Y. Enhancing the catalytic and electrocatalytic properties of Pt-based catalysts by forming bimetallic nanocrystals with Pd[J]. Chem Soc Rev, 2012, 41(24): 8035-8049. |
| 16 | YIN A X, MIN X Q, ZHANG Y W, et al. Shape-selective synthesis and facet-dependent enhanced electrocatalytic activity and durability of monodisperse sub-10 nm Pt-Pd tetrahedrons and cubes[J]. J Am Chem Soc, 2011, 133(11): 3816-3819. |
| 17 | ARLIN J A, KENGO A, TAKEYUKI U, et al. PdPt Nanocubes: a high-performance catalyst for hydrolytic dehydrogenation of ammonia borane[J]. Part Part Syst Char, 2013, 30(10): 888. |
| 18 | DINSMORE A D, HSU M F, NIKOLAIDES M G, et al. Colloidosomes: selectively permeable capsules composed of colloidal particles[J]. Science, 2002, 298(5595): 1006-1009. |
| 19 | BERTHOD A, RUIZ-ÁNGEL M J, CARDA-BROCH S. Recent advances on ionic liquid uses in separation techniques[J]. J Chromatogr A, 2018, 1559: 2-16. |
| 20 | NASIR R, MOHSHIM D F, MANNAN H A, et al. A perspective on ionic liquid-based membranes for CO2 separation[J]. Chem Pap, 2021, 75(3): 839-852. |
| 21 | CORREIA D M, FERNANDES L C, MARTINS P M, et al. Ionic liquid-polymer composites: a new platform for multifunctional applications[J]. Adv Funct Mater, 2020, 30: 1909736. |
| 22 | ZHAO Y, ZHAO Y, QIU J, et al. Facile grafting of imidazolium salt in covalent organic frameworks with enhanced catalytic activity for CO2 fixation and the knoevenagel reaction[J]. ACS Sustainable Chem Eng, 2020, 8: 18413-18419. |
| 23 | LI Z, SHI Y, ZHU A, et al. Light-responsive, reversible emulsification and demulsification of oil-in-water pickering emulsions for catalysis[J]. Angew Chem Int Ed, 2021, 60: 3928-3933. |
| 24 | ZHANG G R, ETZOLD B J M. Emerging applications of solid catalysts with ionic liquid layer concept in electrocatalysis[J]. Adv Funct Mater, 2021, 31: 2010977. |
| 25 | VENTURA S P M, SILVA F A E, QUENTAL M V, et al. Ionic-liquid-mediated extraction and separation processes for bioactive compounds: past, present, and future trends[J]. Chem Rev, 2017, 117: 6984-7052. |
| 26 | WANG H, CUI J, ZHAO Y, et al. Highly efficient separation of 5-hydroxymethylfurfural from imidazolium-based ionic liquids[J]. Green Chem, 2021, 23: 405-411. |
| 27 | LI Z, FENG Y, LIU X, et al. Light triggered switchable ionic liquid aqueous two-phase systems[J]. ACS Sustainable Chem Eng, 2020, 8: 15327-15335. |
| 28 | ZHANG T, DOERT T, WANG H, et al. Inorganic synthesis based on reactions of ionic liquids and deep eutectic solvents[J]. Angew Chem Int Ed, 2021, 60: 22148-22165. |
| 29 | SEITKALIEVA M M, SAMOYLENKO D E, LOTSMAN K A, et al. Metal nanoparticles in ionic liquids: synthesis and catalytic applications[J]. Coordin Chem Rev, 2021, 445: 213982. |
| 30 | YAO K S, WANG N, LI Z Y, et al. Ionic liquid-modulated synthesis of porous worm-like gold with strong SERS response and superior catalytic activities[J]. Nanomaterials, 2019, 9: 1772. |
| 31 | 王楠, 姚开胜, 赵晨晨, 等. 离子液体辅助合成AuPd纳米海绵及催化性能[J]. 高等学校化学学报, 2020, 41(1): 62-69. |
| WANG N, YAO K S, ZHAO C C, et al. Ionnic liquid-assisted synthesis of AuPd nanosponges and their catalytic performance[J]. Chem J Chin Univ, 2020, 41(1): 62-69. | |
| 32 | YAO K S, LI T J, ZHAO C C, et al. Au3Pd1 nanodendrites with hyperbranched architectures: green synthesis at room temperature and highly selective hydrogenation for 4-nitrophenylacetylene[J]. ACS Sustainable Chem Eng, 2020, 8: 14914-14926. |
| 33 | 李旺. 类沸石咪唑酯骨架材料-8的形貌调控及其催化制氢性能[J]. 应用化学, 2022, 39(7): 1065-1072. LI W. Morphology control and catalytic dehydrogenation performance of zeolitic imidazolate frameworks-8[J]. Chin J Appl Chem, 2022, 39(7): 1065-1072. |
| 34 | WANG L, YAMAUCHI Y. Metallic nanocages: synthesis of bimetallic Pt-Pd hollow nanoparticles with dendritic shells by selective chemical etching[J] . J Am Chem Soc, 2013, 135(45): 16762-16765. |
| 35 | YAO K S, ZHAO C C, WANG N, et al. An aqueous synthesis of porous PtPd nanoparticles with reversed bimetallic structures for highly efficient hydrogen generation from ammonia borane hydrolysis[J]. Nanoscale, 2020, 125(2): 638-647. |
| 36 | 王晓俊, 许文, 刘润路, 等. 水凝胶负载的纳米银/氮化碳光催化剂的制备及性能研究[J]. 无机材料学报,2022, 37(7): 731-740. |
| WANG X J, XU W, LIU R L, et al. Preparation and properties of Ag@C3N4 photocatalyst supported by hydrogel[J]. J Inoge Mater, 2022, 37(7): 731-740. | |
| 37 | GOODCHILD I, COLLIER L, MILLAR S L, et al. Structural studies of the phase, aggregation and surface behaviour of 1-alkyl-3-methylimidazolium halide+water mixtures[J]. J Colloid Interface Sci, 2007, 307(2): 455-468. |
| 38 | BLESIC M, MARQUES M H, PLECHKOVA N V, et al. Self-aggregation of ionic liquids: micelle formation in aqueous solution[J]. Green Chem, 2007, 9: 481-490. |
| 39 | BHARGAVA B L, KLEIN M L. Formation of micelles in aqueous solutions of a room temperature ionic liquid: a study using coarse grained molecular dynamics[J]. Soft Matter, 2009, 107(4/5/6): 393-401. |
| 40 | SHI L, WEI Y, SUN N, et al. First observation of rich lamellar structures formed by a single-tailed amphiphilic ionic liquid in aqueous solutions[J]. Chem Commun, 2013, 49: 113880-11390. |
| 41 | XU Q, CHANDRA M J. Catalytic activities of non-noble metals for hydrogen generation from aqueous ammonia-borane at room temperature[J]. J Power Sources, 2006, 163(1): 364-370. |
| 42 | ZHOU Q, XU C. Nanoporous PtRu alloys with unique catalytic activity toward hydrolytic dehydrogenation of ammonia borane[J]. Chem Asian J, 2016, 11(5): 705-712. |
| 43 | KANG K, GU X, GUO L, et al. Efficient catalytic hydrolytic dehydrogenation of ammonia borane over surfactant-free bimetallic nanoparticles immobilized on amine-functionalized carbon nanotubes[J]. Int J Hydrogen Energy, 2015, 40: 12315-12324. |
| 44 | LI Z, HE T, MATSUMURA D, et al. Atomically dispersed Pt on the surface of Ni particles: synthesis and catalytic function in hydrogen generation from aqueous ammonia-borane[J]. ACS Catal, 2017, 7(10): 6762-6769. |
| 45 | ZHOU Y H, ZHANG Z, WANG S, et al. rGO supported PdNi-CeO2 nanocomposite as an efficient catalyst for hydrogen evolution from the hydrolysis of NH3BH3[J]. Int J Hydrogen Energy, 2018, 43(41): 18745-18753. |
| 46 | RACHIERO G P, DEMIRCI U B, MIELE P. Bimetallic RuCo and RuCu catalysts supported on gamma-Al2O3. a comparative study of their activity in hydrolysis of ammonia-borane[J]. Int J Hydrogen Energy, 2011, 36(12): 7051-7065. |
| [1] | Qi-Hang CHEN, Fei-Jian XU, Feng WANG, Shuang-Chan FU, Ying-Hao YU. Research Progress of Deep Eutectic Solvent and Its Application Prospects as Antistatic Agents [J]. Chinese Journal of Applied Chemistry, 2023, 40(3): 341-359. |
| [2] | Xiao-Hu LIU, Xiao-Juan LAI, Hong-Yan CAO, Ting-Ting WANG, Zhi-Qiang DANG. Synergistic Performance of Foaming Agent/Stabilizer/SiO2 Composite Foam Retarded Acid System [J]. Chinese Journal of Applied Chemistry, 2023, 40(1): 91-99. |
| [3] | Hui DU, Chen-Yang YAO, Hao PENG, Bo JIANG, Shun-Xiang LI, Jun-Lie YAO, Fang ZHENG, Fang YANG, Ai-Guo WU. Applications of Transition Metal⁃doped Iron⁃based Nanoparticles in Biomedicine [J]. Chinese Journal of Applied Chemistry, 2022, 39(3): 391-406. |
| [4] | NIU Zhan-Ning, TANG Hao-Qing, ZHENG Chao, TIAN Tian, ZHENG Li-Yun. Study on [RESA]Br@-COOH@Fe3O4 with Density Functional Theory [J]. Chinese Journal of Applied Chemistry, 2021, 38(7): 825-835. |
| [5] | HUANG Xiao-Tong, CHEN Ying-Xin, ZHU Ze-Bin, ZHOU Li-Hua. Research Progress on Detection of Ascorbic Acid by Nanomaterial-Based Spectral Analysis Method [J]. Chinese Journal of Applied Chemistry, 2021, 38(6): 637-650. |
| [6] | LIU Jiao, ZOU Peng-Fei, LI Ping, ZHANG Xiao, WANG Xin-Xin, GAO Yuan-Yuan, LI Li-Li. Research Progress of the Peptide-Based Self-assembled Nanomaterials Against Microbial Resistance [J]. Chinese Journal of Applied Chemistry, 2021, 38(5): 546-558. |
| [7] | LIU Lin-Chang, GUO Ya-Jun, ZHU Hong-Lin, MA Jing-Jing, LI Zhong-Yi, SHUI Miao, ZHENG Yue-Qing. Research Progress on Supported Ultrafine Nano-catalysts for Hydrolytic Dehydrogenation of Ammonia Borane [J]. Chinese Journal of Applied Chemistry, 2021, 38(11): 1405-1422. |
| [8] | ZHOU Chao, SHENG Cheng-Ju, WEN Lin-Lin. Preparation of Imidazolium Salt-based Poly(ionic liquids) Antibacterial Agent and Its Application in Hydrogel Dressing [J]. Chinese Journal of Applied Chemistry, 2021, 38(1): 51-59. |
| [9] | LIU Ning, WANG Danfeng, WU Suyun, LIU Shuilin, FU Lin, LIU Yuejin. Catalytic Synthesis of Bisphenol F over Short-Channeled Mesoporous Molecular Sieve Zr-Ce-SBA-15 Supported Acidic Ionic Liquids [J]. Chinese Journal of Applied Chemistry, 2020, 37(9): 1038-1047. |
| [10] | MENG Yang, YANG Chan, PENG Juan. Progress in Iron, Cobalt and Nickel-Based Metal Phosphide Nano-catalysts for Hydrogen Production under Alkaline Conditions [J]. Chinese Journal of Applied Chemistry, 2020, 37(7): 733-745. |
| [11] | FAN Zhe,ZHANG Shengsheng,TANG Jiahao,FAN Ping. Structure, Preparation and Application of Graded Nanomaterials [J]. Chinese Journal of Applied Chemistry, 2020, 37(5): 489-501. |
| [12] | WANG Chunli,SUN Lianshan,ZHONG Ming,WANG Limin,CHENG Yong. Research Progress of Transition Metal and Compounds for Lithium-Sulfur Batteries [J]. Chinese Journal of Applied Chemistry, 2020, 37(4): 387-404. |
| [13] | WANG Qiushi, HE Junhui. Synthesis of Magnetic CuS Composite Nanomaterial and Its Specific Adsorption of Hg(II) in Water [J]. Chinese Journal of Applied Chemistry, 2020, 37(11): 1316-1323. |
| [14] | GAO Bo, YANG Hongwei, TIAN Shaopeng, ZHAO Yuzhen, TIAN Tian. Solubility Properties of 1-Alkyl-4-amino-1,2,4-triazolium Energetic Ionic Liquids [J]. Chinese Journal of Applied Chemistry, 2019, 36(9): 1044-1052. |
| [15] | SU Fengmei, ZHANG Da, LIANG Feng. Progress in Preparation and Modification of Nano-catalytic Materials by Low-Temperature Plasma [J]. Chinese Journal of Applied Chemistry, 2019, 36(8): 882-891. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||