Chinese Journal of Applied Chemistry ›› 2021, Vol. 38 ›› Issue (10): 1362-1370.DOI: 10.19894/j.issn.1000-0518.210356
• Full Papers • Previous Articles Next Articles
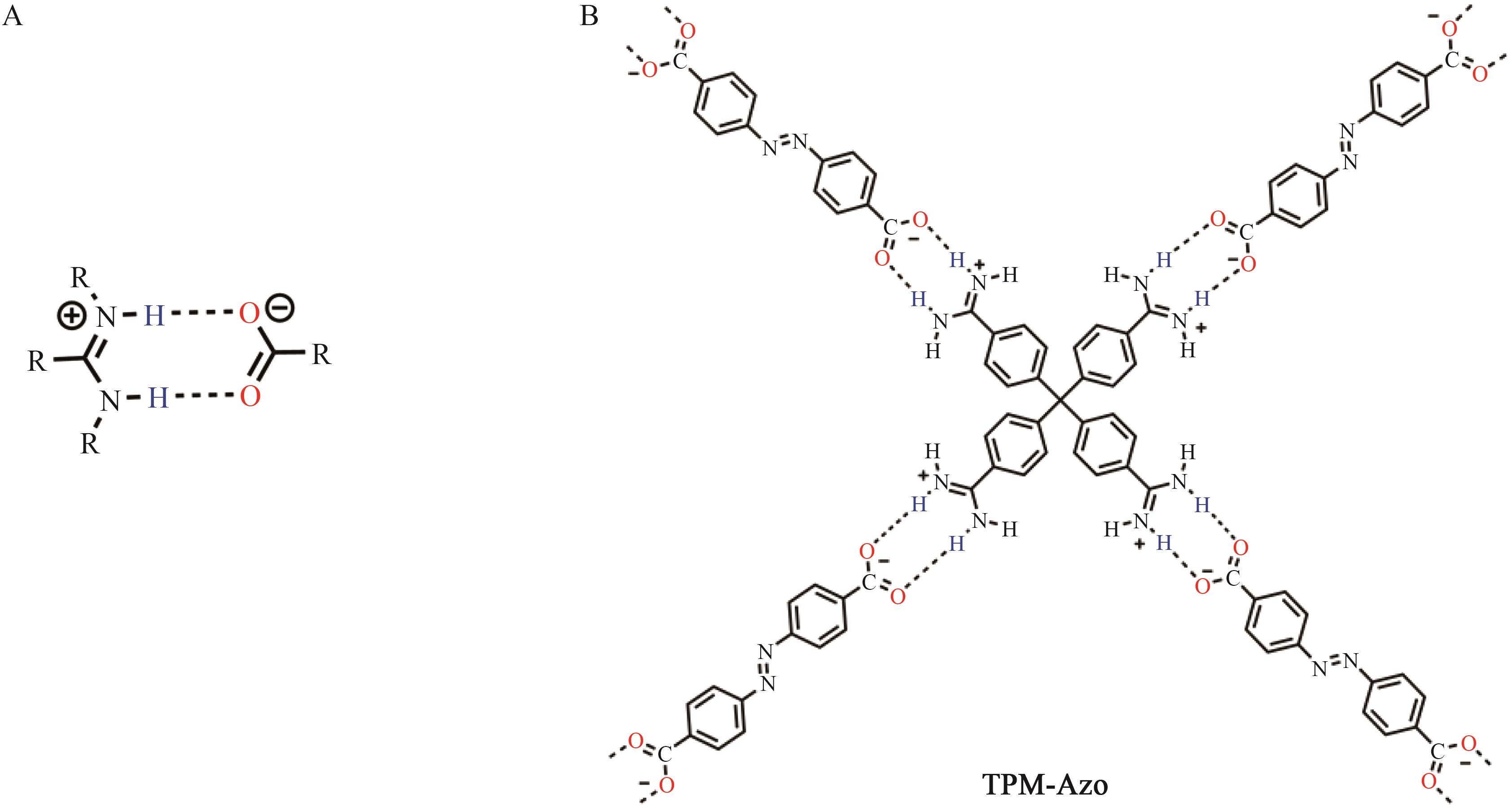
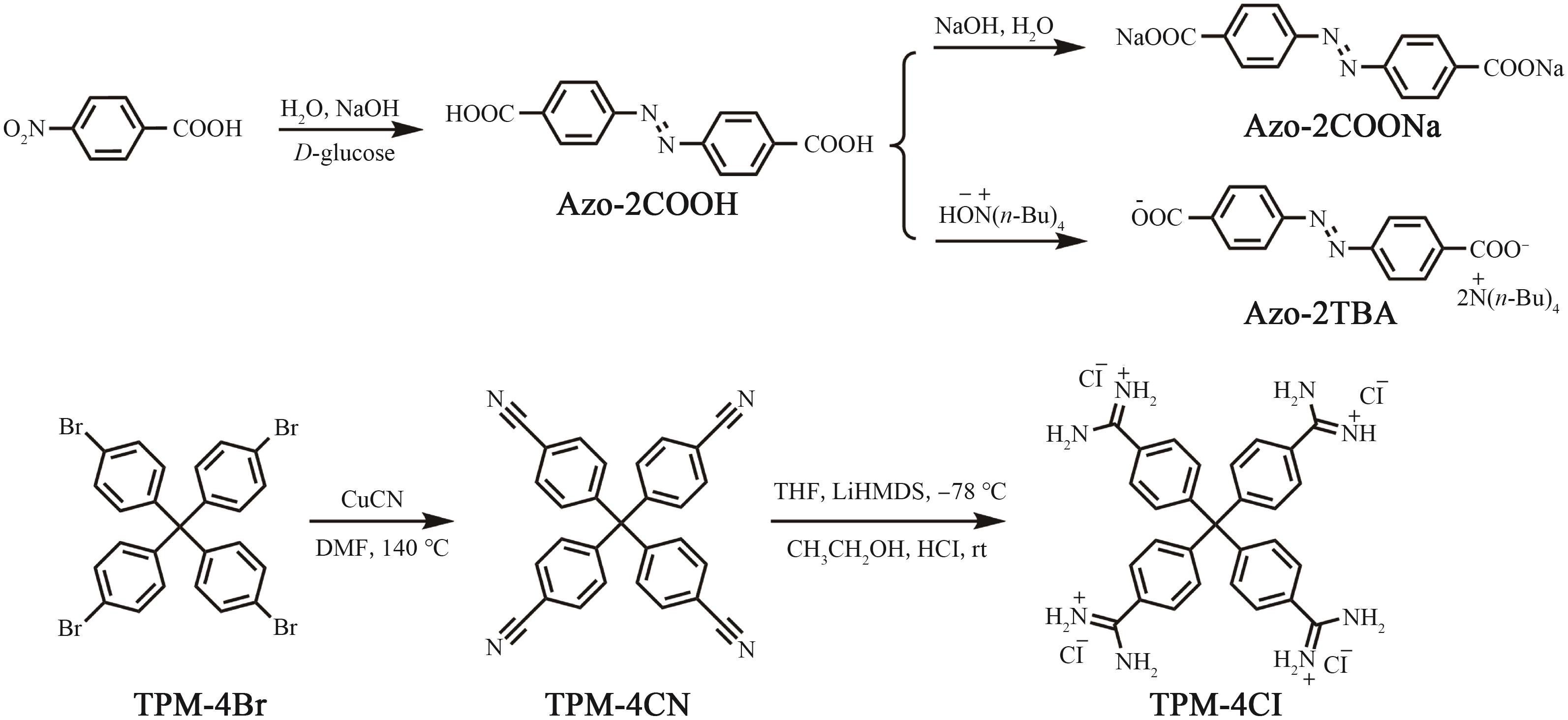
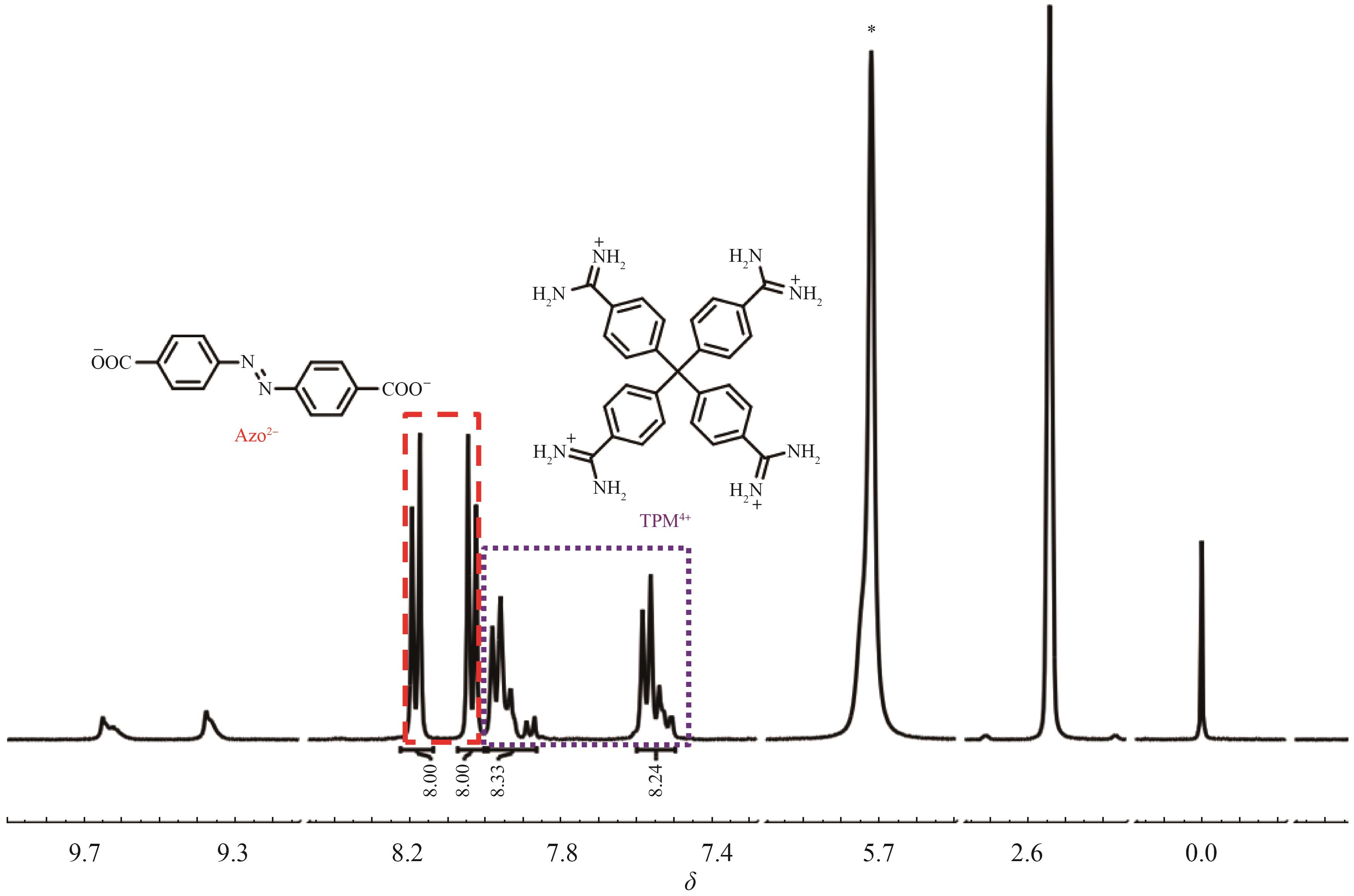
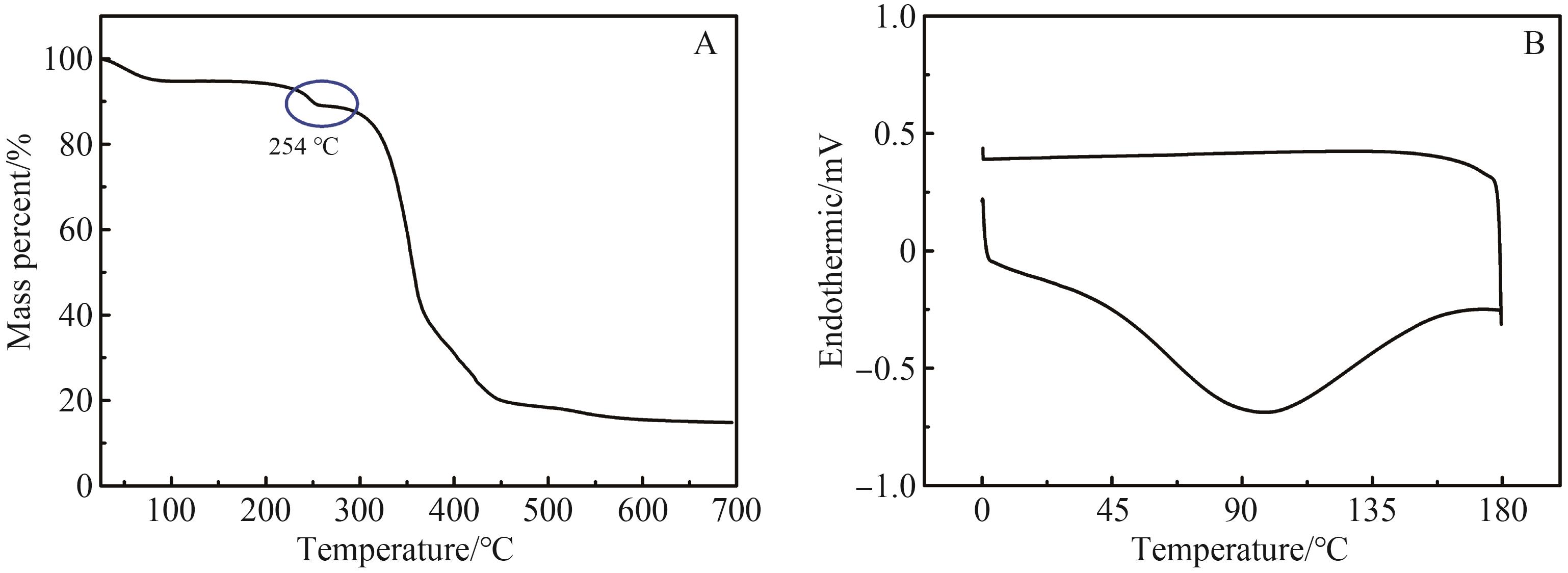
Supramolecular Self⁃Assembly of Azobenzene⁃4,4'⁃dicarboxylic Acid Derivatives in Water
Meng-Wei LI1( ), Hong-Yan WANG1, Ying-Ying WANG1, Xiao-Ying KANG1, Mei-Fang LIU2(
), Hong-Yan WANG1, Ying-Ying WANG1, Xiao-Ying KANG1, Mei-Fang LIU2( ), Yu-Lan CHEN1
), Yu-Lan CHEN1
- 1Tianjin Key Laboratory of Molecular Optoelectronic Science,Institute of Molecular Plus,Tianjin University,Tianjin 300354,China
2College of Chemistry-Chemical & Environmental Engineering,Weifang University,Weifang 261061,China
-
Received:2021-07-21Accepted:2021-08-31Published:2021-10-01Online:2021-10-15 -
Contact:Meng-Wei LI,Mei-Fang LIU -
About author:liumf@iccas.ac.cn; lmw@tju.edu.cn;
-
Supported by:National Key Research and Development Program of China(2017YFA0204503);the National Natural Science Foundation of China(51903187);the China Postdoctoral Science Foundation(2020M670642)
CLC Number:
Cite this article
Meng-Wei LI, Hong-Yan WANG, Ying-Ying WANG, Xiao-Ying KANG, Mei-Fang LIU, Yu-Lan CHEN. Supramolecular Self⁃Assembly of Azobenzene⁃4,4'⁃dicarboxylic Acid Derivatives in Water[J]. Chinese Journal of Applied Chemistry, 2021, 38(10): 1362-1370.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.210356
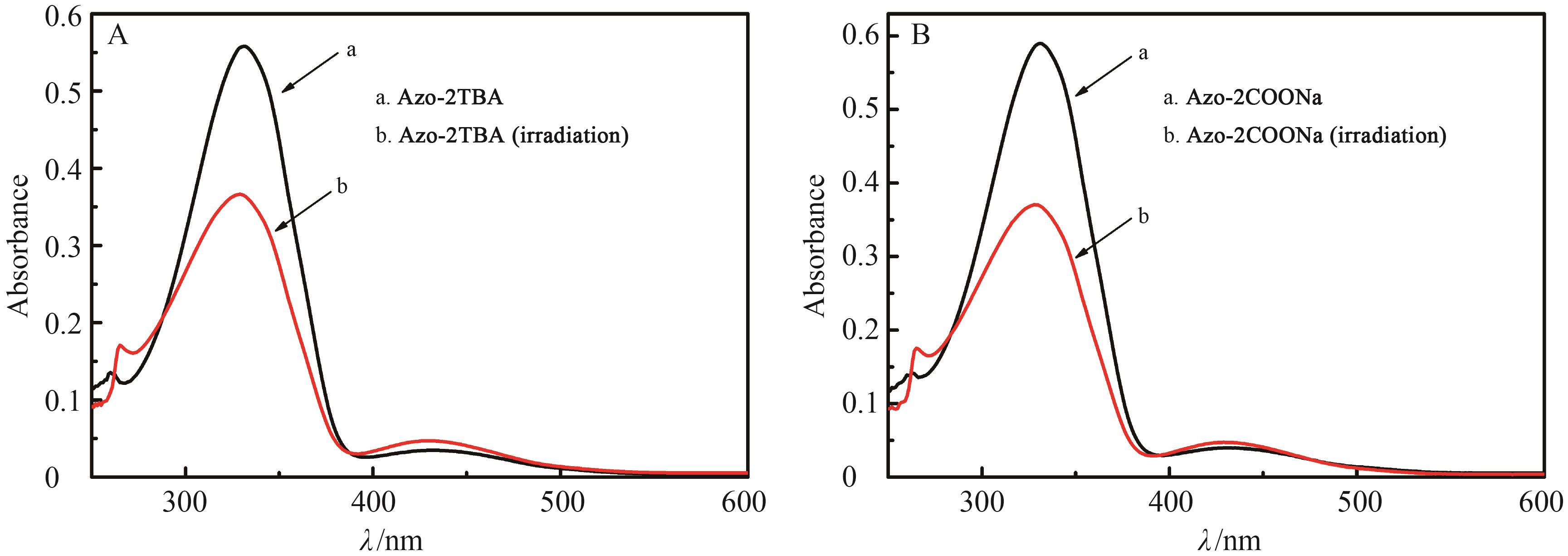
Fig.8 UV-Vis absorption spectra of the aqueous solutions of (a) Azo?2TBA and (b) Azo?2COONa (2.0×105 mol/L) before and after the 365 nm UV irradiation for 8 min
| 1 | WANG H, ZHANG D, ZHAO X, et al. Supramolecular organic frameworks (SOFs): water-phase periodic porous self-assembled architectures[J]. Acta Chim Sin, 2015, 73(6): 471. |
| 2 | TIAN T, QIAN T, JIANG T, et al. A donor-acceptor type macrocycle: toward photolyzable self-assembly[J]. Chem Commun, 2020, 56(28): 3939-3942. |
| 3 | XIA D, WANG P, JI X, et al. Functional supramolecular polymeric networks: the marriage of covalent polymers and macrocycle-based host-guest interactions[J]. Chem Rev, 2020, 120(13): 6070-6123. |
| 4 | XIE T, YUAN W, LI X, et al. Circularly polarized luminescence from chiral π-terphenylene-based supramolecular aggregates[J]. Chinese J Chem, 2021, doi: 10.1002/cjoc.202100139. |
| 5 | YASHIMA E, OUSAKA N, TAURA D, et al. Supramolecular helical systems: helical assemblies of small molecules, foldamers, and polymers with chiral amplification and their functions[J]. Chem Rev, 2016, 116(22): 13752-13990. |
| 6 | SANG Y, HAN J, ZHAO T, et al. Circularly polarized luminescence in nanoassemblies: generation, amplification, and application[J]. Adv Mater, 2019, 32(41): 1900110. |
| 7 | MOLINA P, ZAPATA F, CABALLERO A. Anion recognition strategies based on combined noncovalent interactions[J]. Chem Rev, 2017, 117(15): 9907-9972. |
| 8 | LI J, WANG J, LI H, et al. Supramolecular materials based on AIE luminogens (AIEgens): construction and applications[J]. Chem Soc Rev, 2020, 49(4): 1144-1172. |
| 9 | ČONKOV M, DROŻDŻ W, MIŁOSZ Z, et al. Influencing prototropy by metal ion coordination: supramolecular transformation of a dynamer into a Zn-based toroidal species[J]. J Mater Chem C, 2021, 9(9): 3065-3069. |
| 10 | LIN Z J, CAO R. Porous hydrogen-bonded organic frameworks (HOFs): status and challenges[J]. Acta Chim Sin, 2020, 78(12): 1309. |
| 11 | WHITE N G. Recent advances in self-assembled amidinium and guanidinium frameworks[J]. Dalton Trans, 2019, 48(21): 7062-7068. |
| 12 | LIN R B, HE Y, LI P, et al. Multifunctional porous hydrogen-bonded organic framework materials[J]. Chem Soc Rev, 2019, 48(5): 1362-1389. |
| 13 | ROQUES N, MOUCHAHAM G, DUHAYON C, et al. A robust nanoporous supramolecular metal-organic framework based on ionic hydrogen bonds[J]. Chem Eur J, 2014, 20(37): 11690-11694. |
| 14 | MOUCHAHAM G, ROQUES N, KHODJA W, et al. Hydrogen-bonded open-framework with pyridyl-decorated channels: straightforward preparation and insight into its affinity for acidic molecules in solution[J]. Chem Eur J, 2017, 23(49): 11818-11826. |
| 15 | BOER S A, MORSHEDI M, TARZIA A, et al. Molecular tectonics: a node-and-linker building block approach to a family of hydrogen-bonded frameworks[J]. Chem Eur J, 2019, 25(42): 10006-10012. |
| 16 | MORSHEDI M, THOMAS M, TARZIA A, et al. Supramolecular anion recognition in water: synthesis of hydrogen-bonded supramolecular frameworks[J]. Chem Sci, 2017, 8(4): 3019-3025. |
| 17 | CULLEN D A, GARDINER M G, WHITE N G. A three dimensional hydrogen bonded organic framework assembled through antielectrostatic hydrogen bonds[J]. Chem Commun, 2019, 55(80): 12020-12023. |
| 18 | YAMAUCHI M, YOKOYAMA K, ARATANI N, et al. Crystallization-induced emission of azobenzene derivatives[J]. Angew Chem Int Ed, 2019, 58(40): 14173-14178. |
| 19 | WANG H, HAN Y, YUAN W, et al. Self-assembly of azobenzene derivatives into organogels and photoresponsive liquid crystals[J]. Chem Asian J, 2018, 13(9): 1173-1179. |
| 20 | LIU D, XIE Y, SHAO H, et al. Using azobenzene-embedded self-assembled monolayers to photochemically control cell adhesion reversibly[J]. Angew Chem Int Ed, 2009, 48(24): 4406-4408. |
| 21 | SHI Z M, CHEN S G, ZHAO X, et al. Meta-substituted benzamide oligomers that complex mono-, di- and tricarboxylates: folding-induced selectivity and chirality[J]. Org Biomol Chem, 2011, 9(23): 8122-81129. |
| 22 | HE Y, XIANG S, CHEN B. A microporous hydrogen-bonded organic framework for highly selective C2H2/C2H4 separation at ambient temperature[J]. J Am Chem Soc, 2011, 133(37): 14570-14573. |
| 23 | NORIKANE Y, UCHIDA E, TANAKA S, et al. Photoinduced crystal-to-liquid phase transitions of azobenzene derivatives and their application in photolithography processes through a solid-liquid patterning[J]. Org Lett, 2014, 16(19): 5012-5015. |
| [1] | Yu-Zhu CHEN, Si-Si LIU, Meng-Meng ZHANG, Xiang-De LIN, Dong-Dong ZENG. Polyurethane Dressing Based on Antibacterial Chitosan/Carboxymethyl Cellulose Composite Drug Coating [J]. Chinese Journal of Applied Chemistry, 2023, 40(2): 252-260. |
| [2] | Kai WANG, Hai-Kuan YANG, Hui-Lan LIU, Jia-Min LU, Chen ZHANG. Synthesis and Gelation Properties of Stigmasterol‑Based Supramolecular Gelators [J]. Chinese Journal of Applied Chemistry, 2022, 39(9): 1453-1463. |
| [3] | Xin HE, Cai-Yun JIANG, Tao DING, Yu-Ping WANG. Reserch Progress of Preparation of Ordered Surface Enhanced Raman Scattering Substrate [J]. Chinese Journal of Applied Chemistry, 2022, 39(8): 1167-1176. |
| [4] | Wei-Qiang ZHANG, Chen WANG, Yu-Rong ZHAO, Dong WANG, Ji-Qian WANG, Hai XU. Research Progress of Regulation of Driving Forces in Short Peptide Supramolecular Self‑Assembly [J]. Chinese Journal of Applied Chemistry, 2022, 39(8): 1190-1201. |
| [5] | Zi-Li LI, Xing-Ran XU, Jiang-Hao ZHAN, Xiao-Hua HU, Zi-Ying ZHANG, Shi-Sheng XIONG. Advanced Materials for Lithography [J]. Chinese Journal of Applied Chemistry, 2022, 39(6): 859-870. |
| [6] | HU Xiao-Hua, XIONG Shi-Sheng. Advanced Lithography: Directed Self-Assembly [J]. Chinese Journal of Applied Chemistry, 2021, 38(9): 1029-1078. |
| [7] | TIAN Xin, LAI Han-Wen, LIU Ya-Dong, JI Sheng-Xiang. Analysis of Defects in Block Copolymer Films by a Convolution Algorithm [J]. Chinese Journal of Applied Chemistry, 2021, 38(9): 1199-1208. |
| [8] | WANG Hou-Chen, ZHOU Li, LIU Yang, CHEN Ming-Yue, SHI Tie-Sheng. Research Progress on the Molecular Designs of Building Blocks for Supramolecular Aggregates and Self-Assemblies [J]. Chinese Journal of Applied Chemistry, 2021, 38(6): 615-621. |
| [9] | LIU Jiao, ZOU Peng-Fei, LI Ping, ZHANG Xiao, WANG Xin-Xin, GAO Yuan-Yuan, LI Li-Li. Research Progress of the Peptide-Based Self-assembled Nanomaterials Against Microbial Resistance [J]. Chinese Journal of Applied Chemistry, 2021, 38(5): 546-558. |
| [10] | CHEN Song-Hua, CHEN Xin, LIU Yong-Qi, HE Mei-Yun, FU Shan-Shan, PENG Li, YANG Ji-En. Preparation and Optical Waveguide Property of Benzothiadiazole-Containing One Dimensional Micro-nanowires [J]. Chinese Journal of Applied Chemistry, 2021, 38(11): 1479-1485. |
| [11] | WU Yu, LIU Jia-Cheng. Preparation and Characterization of Dye-sensitized Solar Cells by Inverted Coordination Self-assembly [J]. Chinese Journal of Applied Chemistry, 2021, 38(11): 1494-1502. |
| [12] | Long-Fei LUO, Yu-Jie LI, Zhi-Hao SHEN, Shi-Jun ZHENG, Xing-He FAN. Progress in Self⁃assembly and Photo⁃responsiveness of Thin Films of Azobenzene⁃Based Liquid Crystalline Block Copolymers [J]. Chinese Journal of Applied Chemistry, 2021, 38(10): 1238-1254. |
| [13] | Ai-Hua CHEN, Cheng-Yun ZHANG, Zi-Chao DENG, Ya-Lan SUN. Structure Control of Liquid Crystalline Block Copolymers in Liquid⁃Phase Self⁃assembly [J]. Chinese Journal of Applied Chemistry, 2021, 38(10): 1255-1267. |
| [14] | Yuan DENG, Yao-Jian FAN, Lei TAO, Zhi-Wang LUO, He-Lou XIE. Liquid Crystal Nanoparticles Containing Azobenzene:Synthesis, Supramolecular Structure and Their Applications in Light⁃Controlled Orientation [J]. Chinese Journal of Applied Chemistry, 2021, 38(10): 1353-1361. |
| [15] | Sha-Man LUO, Hao-Zhe SUN, Shi-Qiang YAN, Hui HUANG, Wei-Jia ZHANG, Jia WEI, Yan-Lei YU. Cell Biocompatibility of Photodeformable Azobenzene⁃containing Liquid Crystal Polymers [J]. Chinese Journal of Applied Chemistry, 2021, 38(10): 1371-1381. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||