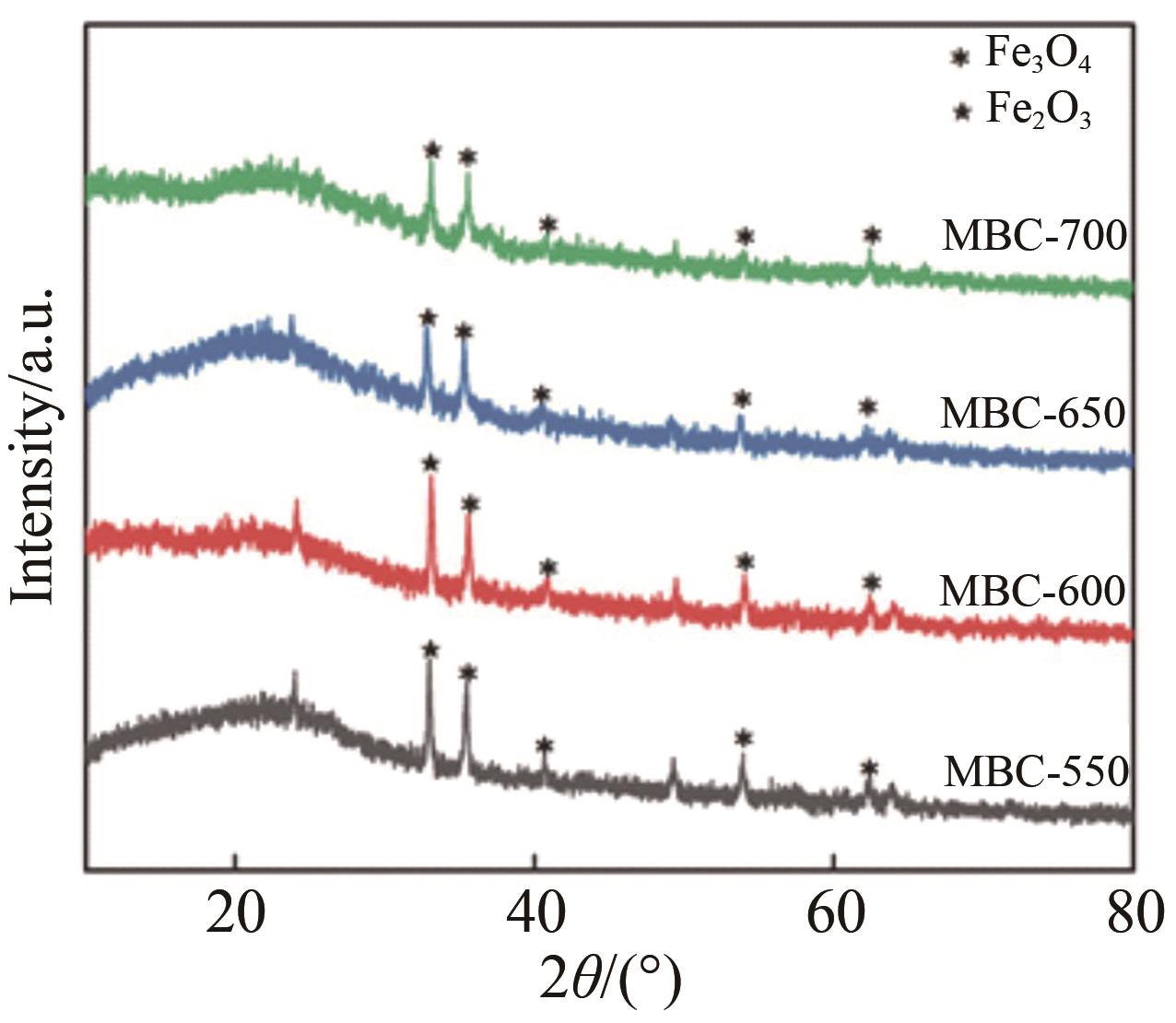
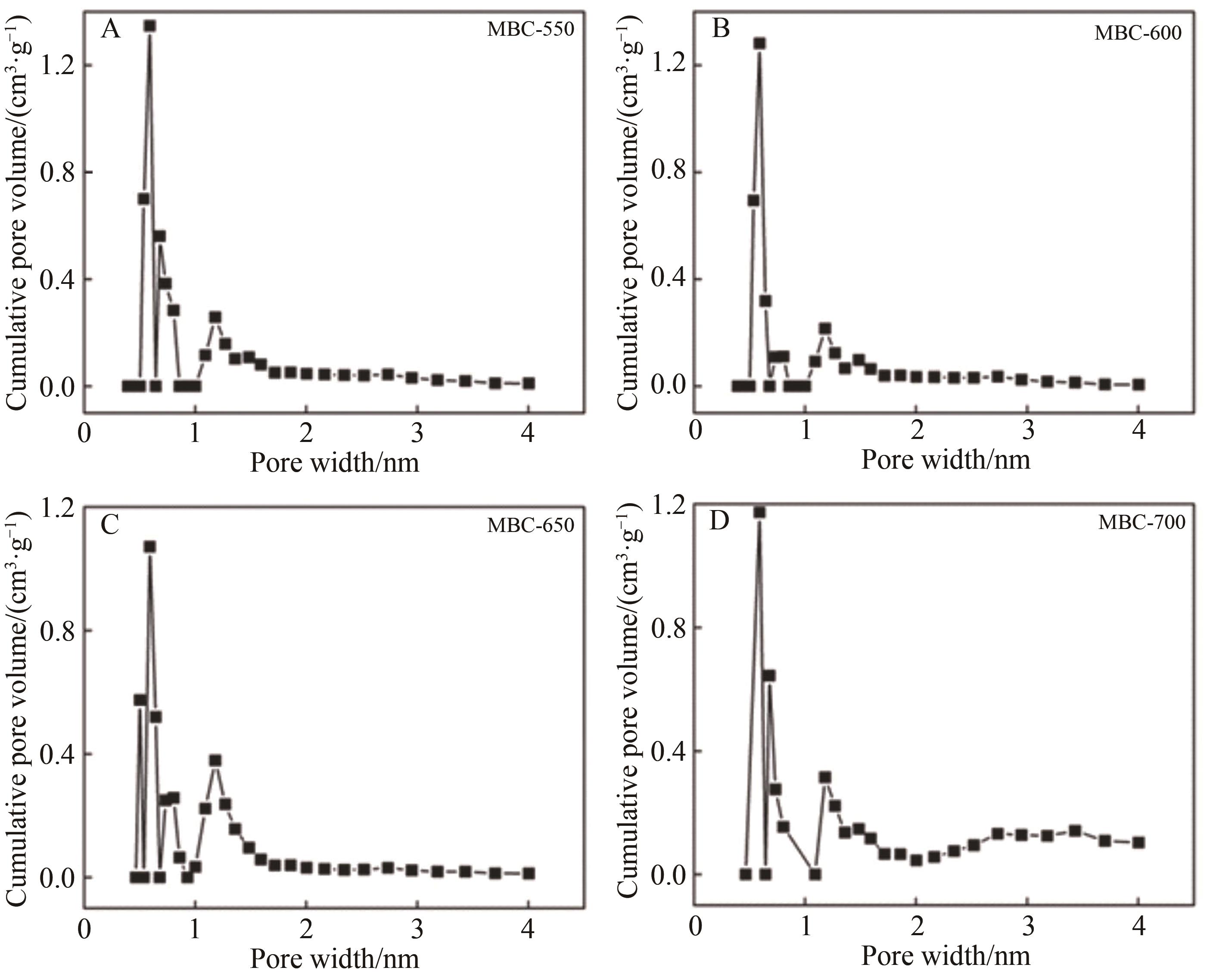
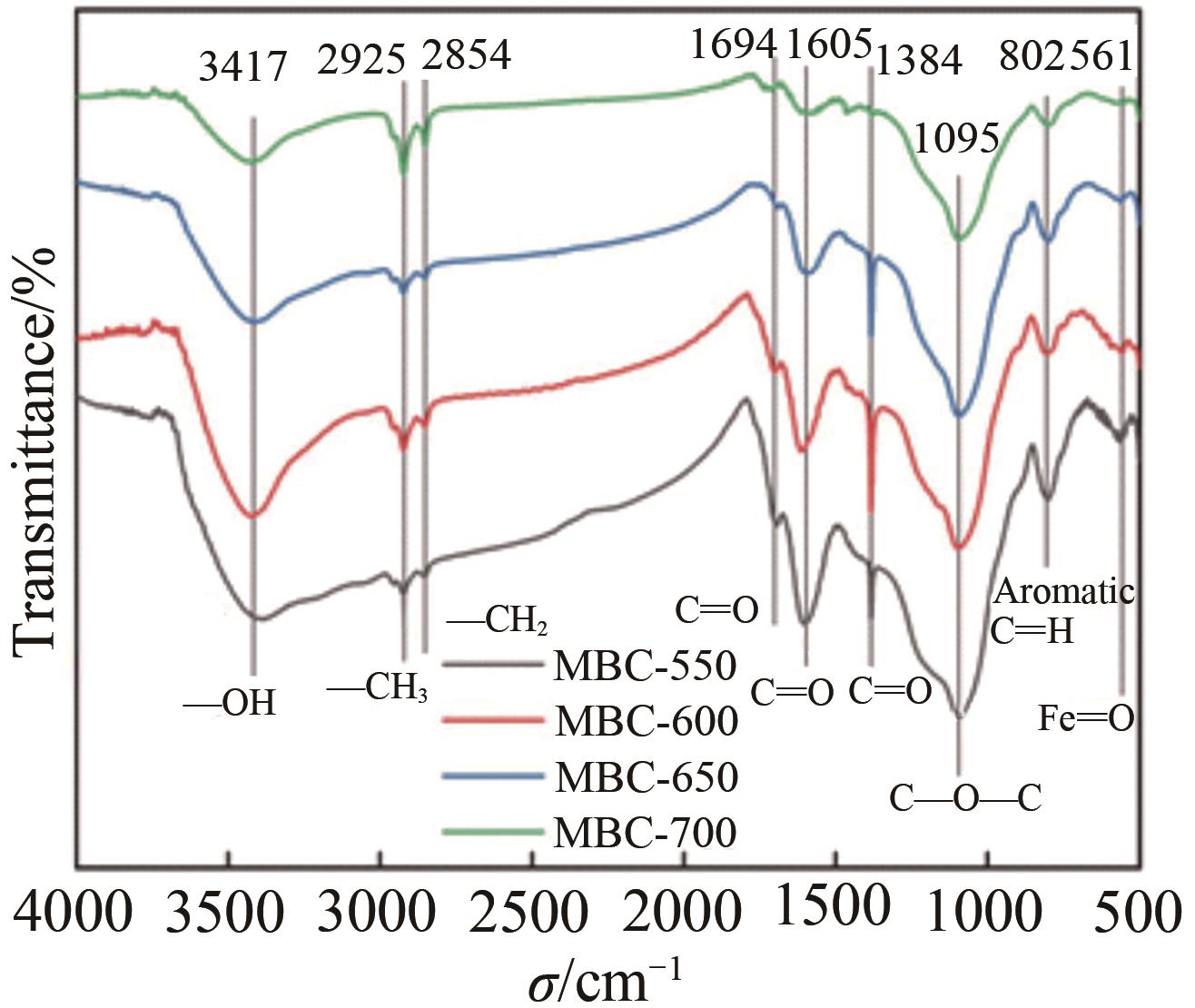
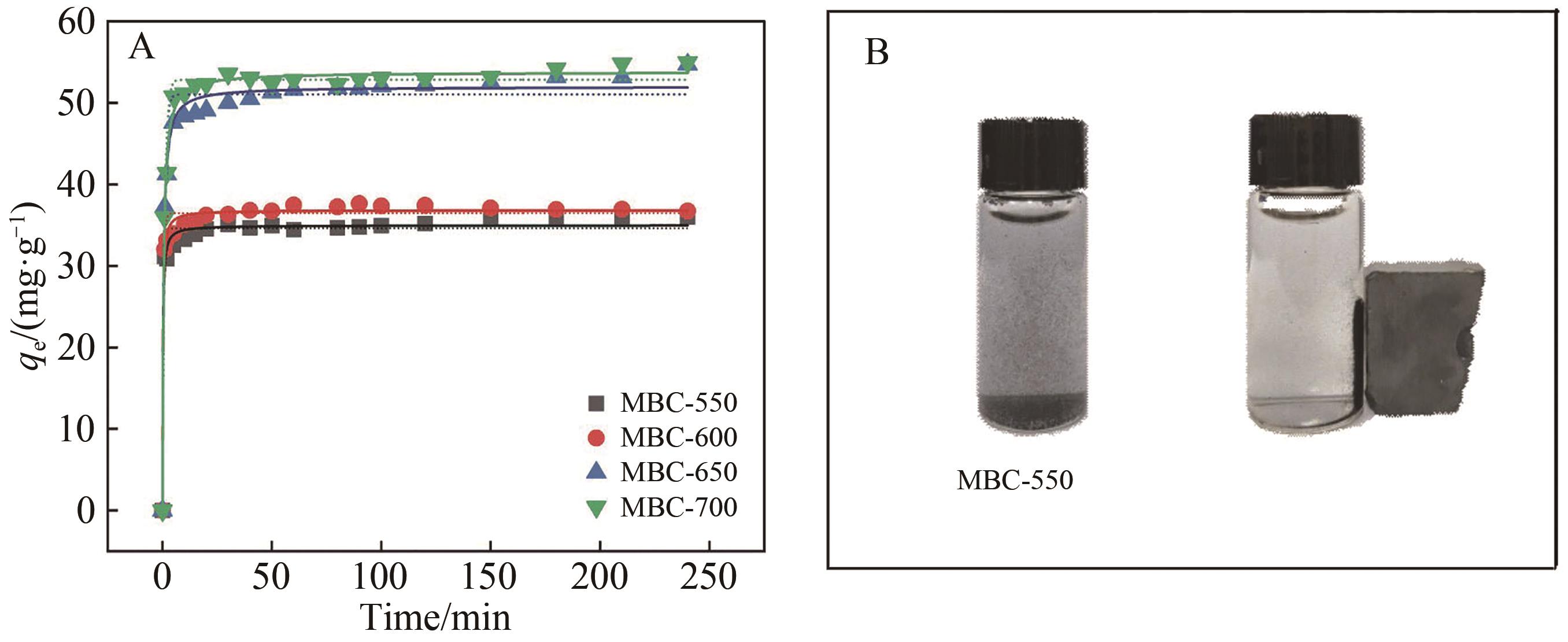
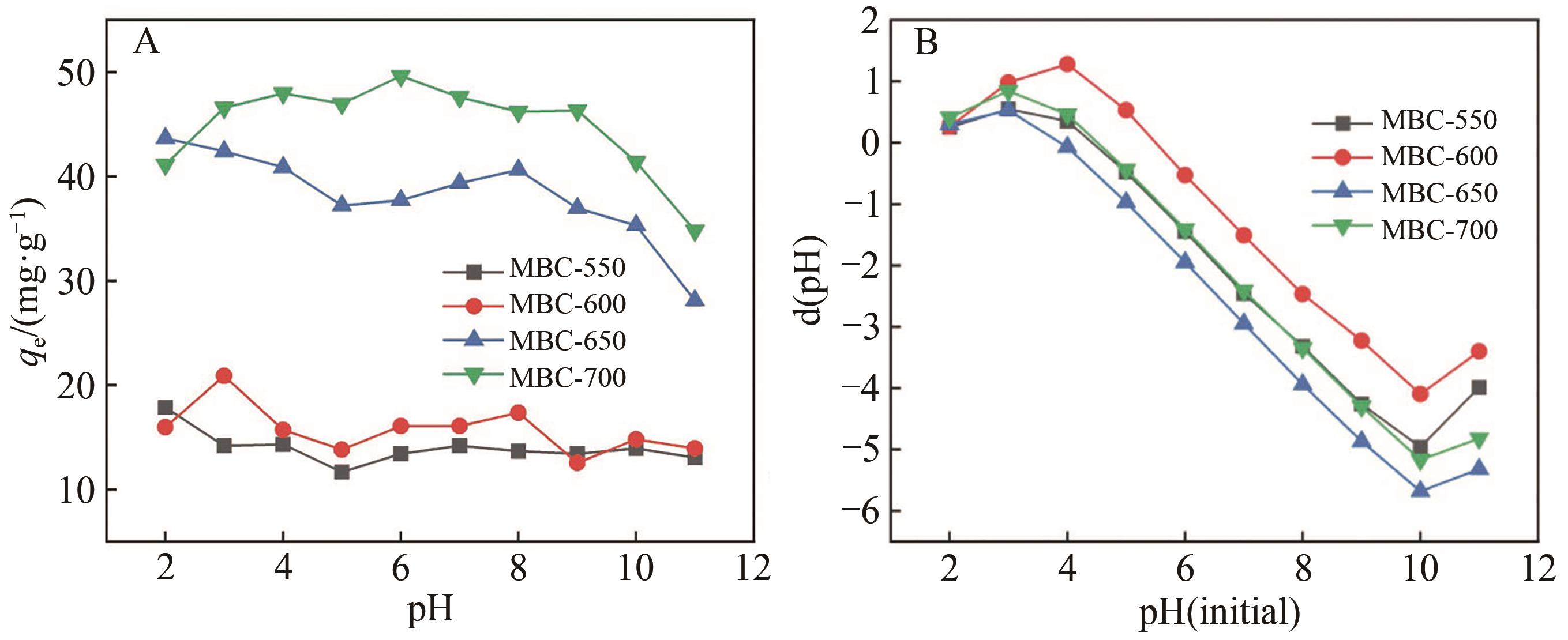
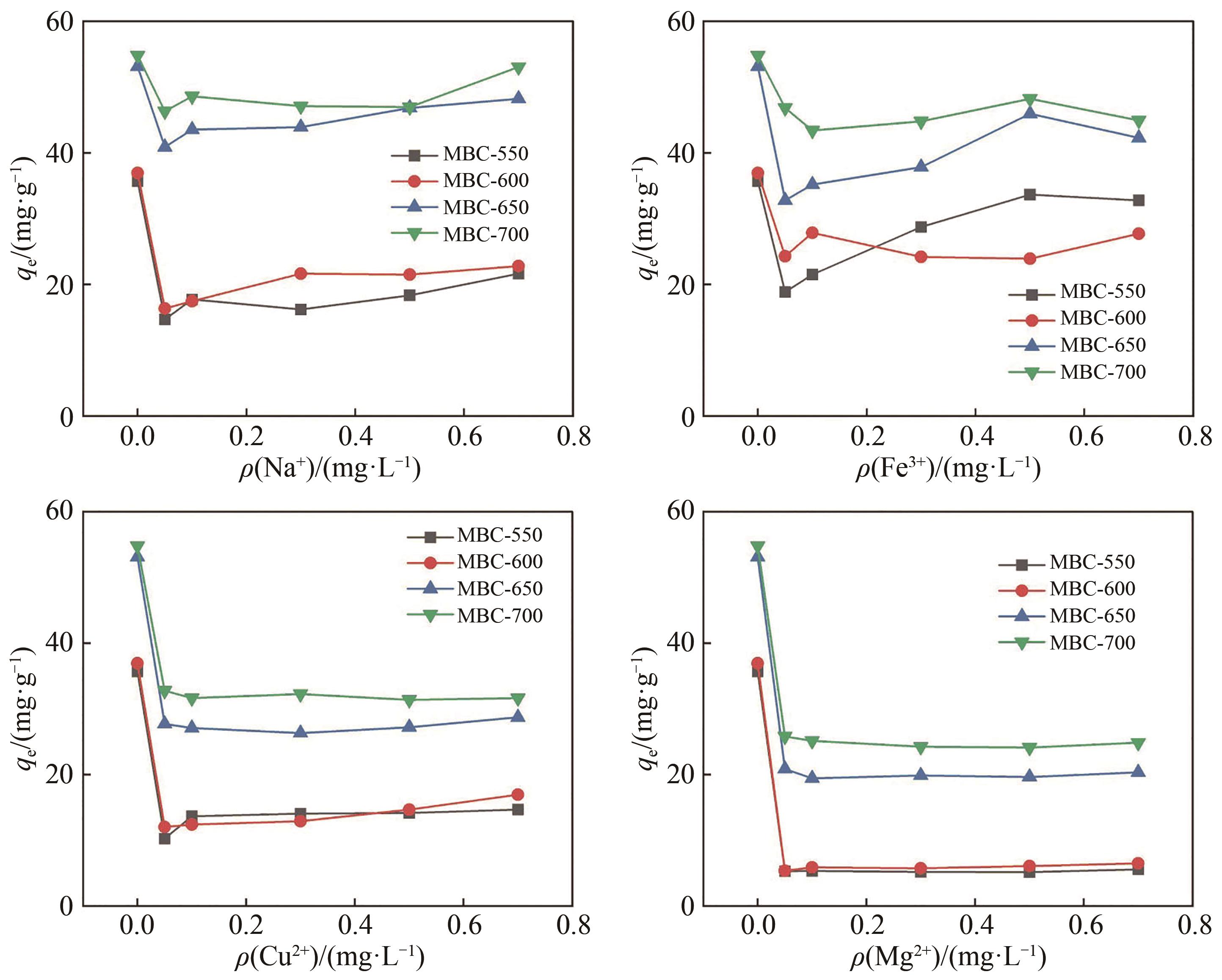
Chinese Journal of Applied Chemistry ›› 2023, Vol. 40 ›› Issue (2): 288-298.DOI: 10.19894/j.issn.1000-0518.220169
• Full Papers • Previous Articles Next Articles
Adsorption Performance of Rhodamine B on Magnetic Biochar Prepared at Different Pyrolysis Temperatures
Qian YANG, Yang ZHANG, Ming-Yang LAI, Lin GUO, Jian-Bo JIA( )
)
- School of Biotechnology and Health Sciences,Wuyi University,Jiangmen 529000,China
-
Received:2022-05-06Accepted:2022-11-06Published:2023-02-01Online:2023-02-27 -
Contact:Jian-Bo JIA -
About author:jbjiagu@163.com
-
Supported by:the National Natural Science Foundation of China(21974097);the Education Department of Guangdong Province(2020KSYS004)
CLC Number:
Cite this article
Qian YANG, Yang ZHANG, Ming-Yang LAI, Lin GUO, Jian-Bo JIA. Adsorption Performance of Rhodamine B on Magnetic Biochar Prepared at Different Pyrolysis Temperatures[J]. Chinese Journal of Applied Chemistry, 2023, 40(2): 288-298.
share this article
Pyrolysis temperature/℃ | BET surface area/(m2·g-1) | Total pore volume/(cm3·g-1) | Micropore volume/(cm3·g-1) | Micropore area/(m2·g-1) |
|---|---|---|---|---|
| 550 | 314.779 | 0.123 | 0.121 | 260.450 |
| 600 | 331.710 | 0.137 | 0.119 | 257.683 |
| 650 | 418.436 | 0.172 | 0.151 | 327.129 |
| 700 | 427.418 | 0.211 | 0.120 | 261.633 |
Table 1 Variation of pores of MBC prepared at different pyrolysis temperatures
Pyrolysis temperature/℃ | BET surface area/(m2·g-1) | Total pore volume/(cm3·g-1) | Micropore volume/(cm3·g-1) | Micropore area/(m2·g-1) |
|---|---|---|---|---|
| 550 | 314.779 | 0.123 | 0.121 | 260.450 |
| 600 | 331.710 | 0.137 | 0.119 | 257.683 |
| 650 | 418.436 | 0.172 | 0.151 | 327.129 |
| 700 | 427.418 | 0.211 | 0.120 | 261.633 |
| MBC | qe(exp)/(mg·g-1) | Pseudo first-order kinetic model | Pseudo second-order kinetic model | ||||
|---|---|---|---|---|---|---|---|
| qe(cal)/(mg·g-1) | K1/min-1 | R2 | qe(cal)/(mg·g-1) | K2/min-1 | R2 | ||
| MBC-550 | 35.696 | 34.604 | 2.096 | 0.978 | 34.967 | 0.167 | 0.990 |
| MBC-600 | 36.962 | 36.455 | 1.955 | 0.981 | 36.838 | 0.146 | 0.993 |
| MBC-650 | 53.118 | 51.039 | 1.110 | 0.967 | 51.971 | 0.042 | 0.990 |
| MBC-700 | 54.810 | 52.827 | 0.972 | 0.984 | 53.755 | 0.036 | 0.995 |
Table 2 Kinetics parameters for adsorption of RhB by MBC
| MBC | qe(exp)/(mg·g-1) | Pseudo first-order kinetic model | Pseudo second-order kinetic model | ||||
|---|---|---|---|---|---|---|---|
| qe(cal)/(mg·g-1) | K1/min-1 | R2 | qe(cal)/(mg·g-1) | K2/min-1 | R2 | ||
| MBC-550 | 35.696 | 34.604 | 2.096 | 0.978 | 34.967 | 0.167 | 0.990 |
| MBC-600 | 36.962 | 36.455 | 1.955 | 0.981 | 36.838 | 0.146 | 0.993 |
| MBC-650 | 53.118 | 51.039 | 1.110 | 0.967 | 51.971 | 0.042 | 0.990 |
| MBC-700 | 54.810 | 52.827 | 0.972 | 0.984 | 53.755 | 0.036 | 0.995 |
| MBC | T/℃ | Freundlich isotherm model | Langmuir isotherm model | ||||
|---|---|---|---|---|---|---|---|
| KF | N | R2 | qmax/(mg·g-1) | KL | R2 | ||
| MBC-550 | 20 | 2.224 | 2.496 | 0.999 | 20.594 | 0.026 | 0.962 |
| 30 | 2.272 | 1.995 | 0.993 | 36.558 | 0.015 | 0.973 | |
| 40 | 2.962 | 3.322 | 0.979 | 14.637 | 0.052 | 0.954 | |
| MBC-600 | 20 | 2.704 | 2.645 | 0.995 | 21.566 | 0.030 | 0.935 |
| 30 | 3.684 | 2.402 | 0.993 | 40.203 | 0.026 | 0.976 | |
| 40 | 2.956 | 3.067 | 0.981 | 16.961 | 0.043 | 0.871 | |
| MBC-650 | 20 | 11.283 | 3.313 | 0.990 | 49.978 | 0.090 | 0.929 |
| 30 | 13.760 | 3.547 | 0.991 | 54.975 | 0.116 | 0.899 | |
| 40 | 14.795 | 3.906 | 0.971 | 49.343 | 0.180 | 0.935 | |
| MBC-700 | 20 | 12.554 | 3.318 | 0.991 | 53.779 | 0.102 | 0.893 |
| 30 | 16.993 | 3.496 | 0.990 | 68.877 | 0.108 | 0.937 | |
| 40 | 15.660 | 3.509 | 0.980 | 59.61 | 0.146 | 0.940 | |
Table 3 Langmuir and Freundlich adsorption isotherm constants for adsorption of RhB by MBC
| MBC | T/℃ | Freundlich isotherm model | Langmuir isotherm model | ||||
|---|---|---|---|---|---|---|---|
| KF | N | R2 | qmax/(mg·g-1) | KL | R2 | ||
| MBC-550 | 20 | 2.224 | 2.496 | 0.999 | 20.594 | 0.026 | 0.962 |
| 30 | 2.272 | 1.995 | 0.993 | 36.558 | 0.015 | 0.973 | |
| 40 | 2.962 | 3.322 | 0.979 | 14.637 | 0.052 | 0.954 | |
| MBC-600 | 20 | 2.704 | 2.645 | 0.995 | 21.566 | 0.030 | 0.935 |
| 30 | 3.684 | 2.402 | 0.993 | 40.203 | 0.026 | 0.976 | |
| 40 | 2.956 | 3.067 | 0.981 | 16.961 | 0.043 | 0.871 | |
| MBC-650 | 20 | 11.283 | 3.313 | 0.990 | 49.978 | 0.090 | 0.929 |
| 30 | 13.760 | 3.547 | 0.991 | 54.975 | 0.116 | 0.899 | |
| 40 | 14.795 | 3.906 | 0.971 | 49.343 | 0.180 | 0.935 | |
| MBC-700 | 20 | 12.554 | 3.318 | 0.991 | 53.779 | 0.102 | 0.893 |
| 30 | 16.993 | 3.496 | 0.990 | 68.877 | 0.108 | 0.937 | |
| 40 | 15.660 | 3.509 | 0.980 | 59.61 | 0.146 | 0.940 | |
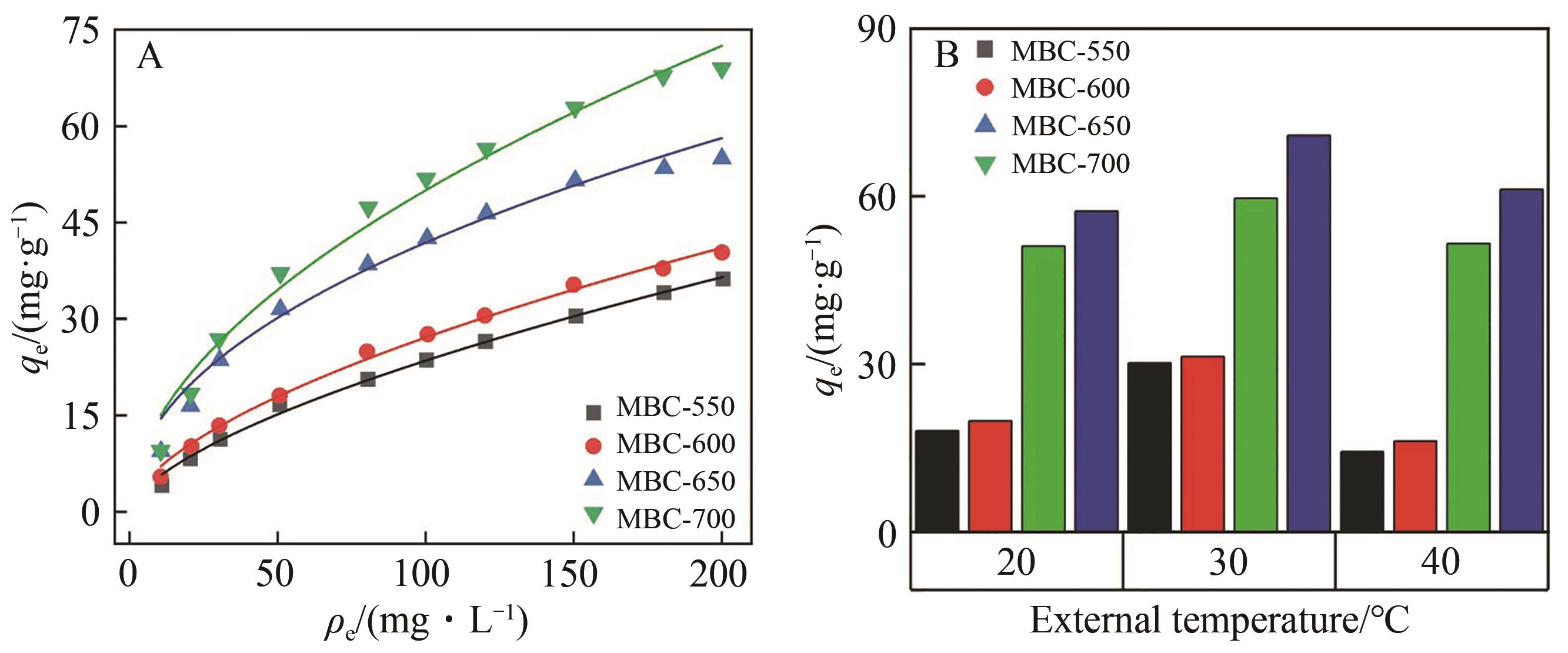
Fig.6 Fitting of the adsorption isotherms of RhB by MBC at different pyrolysis temperatures (A); The influence of external temperature on the adsorption of MBC (B)
| MBC | ΔG/(kJ·mol-1) | ΔH/(kJ·mol-1) | ΔS/(kJ·mol-1) | ||
|---|---|---|---|---|---|
| 20 ℃ | 30 ℃ | 40 ℃ | |||
| MBC-550 | -3.807 | -4.175 | -5.950 | -38.345 | 24.545 |
| MBC-600 | -4.793 | -5.492 | -5.737 | -59.762 | 43.872 |
| MBC-650 | -8.821 | -9.794 | -10.557 | -23.631 | 4.717 |
| MBC-700 | -9.085 | -10.289 | -10.426 | -12.266 | 6.742 |
Table 4 Thermodynamic parameters of RhB adsorption onto MBC
| MBC | ΔG/(kJ·mol-1) | ΔH/(kJ·mol-1) | ΔS/(kJ·mol-1) | ||
|---|---|---|---|---|---|
| 20 ℃ | 30 ℃ | 40 ℃ | |||
| MBC-550 | -3.807 | -4.175 | -5.950 | -38.345 | 24.545 |
| MBC-600 | -4.793 | -5.492 | -5.737 | -59.762 | 43.872 |
| MBC-650 | -8.821 | -9.794 | -10.557 | -23.631 | 4.717 |
| MBC-700 | -9.085 | -10.289 | -10.426 | -12.266 | 6.742 |
| 1 | YAN S C, LI Z S, ZOU Z G. Photodegradation of rhodamine B and methyl orange over boron-doped g-C3N4 under visible light irradiation[J]. Langmuir, 2010, 26(6): 3894-3901. |
| 2 | AL-KAZRAGI M A, Al-HEETIMI D T A. Removal of toxic dye (rhodamine B) from aqueous solutions by natural smectite (SMC) and SMC-nanoTiO2[J]. Desalin Water Treat, 2021, 230: 276-287. |
| 3 | CADENBACH T, BENITEZ M J, TIRADO S A, et al. Adsorption enhanced photocatalytic degradation of rhodamine B using GdxBi1- xFeO3@SBA-15 (x=0, 0.05, 0.10, 0.15) nanocomposites under visible light irradiation[J]. Ceram Int, 2021, 47(20): 29139-29148. |
| 4 | REN Z G, CHEN F, WANG B, et al. Magnetic biochar from alkali-activated rice straw for removal of rhodamine B from aqueous solution[J]. Environ Eng Res, 2020, 25(4): 536-544. |
| 5 | WANG J, LIAO Z W, IFTHIKAR J, et al. One-step preparation and application of magnetic sludge-derived biochar on acid orange 7 removal via both adsorption and persulfate based oxidation[J]. RSC Adv, 2017, 7(30): 18696-18706. |
| 6 | FROHLICH A C, FOLETTO E L, DOTTO G L. Preparation and characterization of NiFe2O4/activated carbon composite as potential magnetic adsorbent for removal of ibuprofen and ketoprofen pharmaceuticals from aqueous solutions[J]. J Clean Prod, 2019, 229: 828-837. |
| 7 | YAO X X, JI L L, GUO J, et al. Magnetic activated biochar nanocomposites derived from wakame and its application in methylene blue adsorption[J]. Bioresour Technol, 2020, 302: 8. |
| 8 | 陈芳, 赵国平, 莫尊理. 石墨烯/Fe3O4/TiO2磁性纳米复合材料的光催化性能研究[J]. 人工晶体学报, 2016, 45(12): 2795-2800, 2806. |
| CHEN F, ZHAO G P, MO Z L. Photocatalytic performance study of graphene/Fe3O4/TiO2 magnetic nanocomposites[J]. J Synth Cryst, 2016, 45 (12): 2795-2800, 2806. | |
| 9 | SUN P, HUI C, AZIM K R, et al. Efficient removal of crystal violet using Fe3O4-coated biochar: the role of the Fe3O4 nanoparticles and modeling study their adsorption behavior[J]. Sci Rep, 2015, 5: 12638. |
| 10 | NGUYEN V H, NGUYEN D T, NGUYEN T T, et al. Activated carbon with ultrahigh surface area derived from sawdust biowaste for the removal of rhodamine B in water[J]. Environ Technol Innov, 2021, 24: 101811. |
| 11 | CHEN F H, LIANG W W, QIN X Y, et al. Preparation and recycled simultaneous adsorption of methylene blue and Cu2+ co-pollutants over carbon layer encapsulated Fe3O4/graphene oxide nanocomposites rich in amino and thiol groups[J]. Colloids Surf A, 2021, 625: 13. |
| 12 | WANG Z X, ZHOU Q H, CHEN S J, et al. Preparation of a quinoa straw-derived porous carbon material and a Fe3O4-contained composite material for removal of rhodamine B from water[J]. Mater Res Express, 2020, 7(12): 16. |
| 13 | OLADIPO A A, IFEBAJO A O. Highly efficient magnetic chicken bone biochar for removal of tetracycline and fluorescent dye from wastewater: two-stage adsorber analysis[J]. J Environ Manage, 2018, 209: 9-16. |
| 14 | SAFARIK I, BALDIKOVA E, PROCHAZKOVA J, et al. Magnetically modified agricultural and food waste: preparation and application[J]. J Agric Food Chem, 2018, 66 (11): 2538-2552. |
| 15 | 汪燕南, 李文文, 孙运飞, 等. 改性甘蔗渣去除模拟废水中孔雀石绿的研究[J]. 环境工程技术学报, 2016, 6(5): 434-439. |
| WANG Y L, LI W W, SUN Y F, et al. Removal of malachite green from simulated wastewater by modified sugarcane bagasse[J]. J Environ Eng Technol, 2016, 6(5): 434-439. | |
| 16 | 江美琳, 金辉, 邓聪, 等. 生物炭负载Fe3O4纳米粒子的制备与表征[J]. 农业环境科学学报, 2018, 37(3): 592-597. |
| JIANG M L, JIN H, DENG C, et al. Preparation and characterization of biochar-loaded Fe3O4 nanoparticles[J]. J Agro Environ Sci, 2018, 37(0): 592-597. | |
| 17 | 魏太庆, 王博, 艾丹, 等. 磁性生物炭的制备及其在环境修复中的研究进展[J]. 功能材料, 2021, 52(10): 10039-10047. |
| WEI T Q, WANG B, AI D, et al. Preparation of magnetic biochar and its research progress in environmental remediation[J]. J Funct Mater, 2021, 52(10): 10039-10047. | |
| 18 | 林珈羽, 张越, 刘沅, 等. 不同原料和炭化温度下制备的生物炭结构及性质[J]. 环境工程学报, 2016, 10(6): 3200-3206. |
| LIN J Y, ZHANG Y, LIU Y, et al. Structure and properties of biochar prepared with different raw materials and charring temperatures[J]. J Environ Eng, 2016, 10(6): 3200-3206. | |
| 19 | 简敏菲, 高凯芳, 余厚平. 不同裂解温度对水稻秸秆制备生物炭及其特性的影响[J]. 环境科学学报, 2016, 36(5): 1757-1765. |
| JIAN M F, GAO K F, YU H P. Effects of different cracking temperatures on the preparation and properties of biochar from rice straw[J]. Acta Sci Circum, 2016, 36(5): 1757-1765. | |
| 20 | 韦思业, 宋建中, 彭平安, 等. 不同温度制备生物炭的热解产物特征[J]. 地球化学, 2019, 48(5): 511-520. |
| WEI S Y, SONG J C, PENG P A, et al. Characterization of pyrolysis products of biochar prepared at different temperatures[J]. Geochimica, 2019, 48(5): 511-520. | |
| 21 | 徐亮, 王豹祥, 汪健, 等. 不同热解温度制备的水稻秸秆生物炭理化特性分析[J]. 土壤通报, 2020, 51(1):136-143. |
| XU L, WANG B X, WANG J, et al. Analysis of physicochemical properties of rice straw biochar prepared at different pyrolysis temperatures[J]. Chin J Soil Sci, 2020, 51(1): 136-143. | |
| 22 | 任爱玲, 王启山, 郭斌. 污泥活性炭的结构特征及表面分形分析[J]. 化学学报, 2006, 64(10): 1068-1072. |
| REN A L, WANG Q S, GUO B. Structure characterization and surface fractal analysis of sludge activated carbon[J]. Acta Chim Sin, 2006, 64 (10): 1068-1072. | |
| 23 | NGUYEN B T, LEHMANN J. Black carbon decomposition undervarying water regimes[J]. Org Geochem, 2009, 40(8): 846-853. |
| 24 | 高凯芳, 简敏菲, 余厚平, 等. 裂解温度对稻秆与稻壳制备生物炭表面官能团的影响[J]. 环境化学, 2016, 35(8): 1663-1669. |
| GAO K F, KAN M F, YU H P, et al. Effect of cracking temperature on the surface functional groups of biochar prepared from rice straw and rice husk[J]. Environ Chem, 2016, 35(8): 1663-1669. | |
| 25 | SHRESTHA D. Efficiency of wood-dust of dalbergia sisoo as low-cost adsorbent for rhodamine-B dye removal[J]. Nanomaterials-Basel, 2021, 11 (9): 2217. |
| 26 | SADEGH N, HADDADI H, ARABKHANI P, et al. Simultaneous elimination of rhodamine B and malachite green dyes from the aqueous sample with magnetic reduced graphene oxide nanocomposite: optimization using experimental design[J]. J Mol Liq, 2021, 343: 117710. |
| 27 | 常帅帅, 张学杨, 王洪波, 等. 秸秆生物炭对Pb2+的吸附性能及影响因素研究[J]. 环境科技, 2019, 32(6): 23-28. |
| CHANG S S, ZHANG X Y, WANG H B, et al. Study on adsorption performance of straw biochar on Pb2+ and influencing factors[J]. Environl Tech, 2019, 32(6): 23-28. | |
| 28 | 李艳春, 张鹏会, 张强, 等. 4种生物炭对阳离子染料吸附性能[J]. 环境科学与技术, 2020, 43(7): 101-110. |
| LI Y C, ZHANG P H, ZHANG Q, et al. Adsorption performance of four biochar species on cationic dyes[J]. Environ Sci Tech, 2020, 43(7): 101-110. | |
| 29 | 温嘉伟, 王辉, 张浩, 等. 改性棕榈树纤维生物质炭的制备及其对溶液中Pb2+的吸附性能分析[J]. 农业环境科学学报, 2021, 40(5): 1088-1096. |
| WEN J W, WANG H, ZHANG H, et al. Preparation of modified palm fiber biochars and their adsorption of Pb2+ in solution[J]. J Agro-Environ Sci, 2021, 40(5): 1088-1096. | |
| 30 | 白书立, 陈凯, 李换英, 等. ZIF-8吸附罗丹明B模拟废水热力学和动力学研究[J]. 河南师范大学学报(自然科学版), 2020, 48(4): 73-77. |
| BAI S L, CHEN K, LI H Y, et al. Thermodynamic and kinetic study of ZIF-8 adsorption on rhodamine B simulated wastewater[J]. J Henan Norm Univ (Nat Sci Ed), 2020, 48(4): 73-77. | |
| 31 | DHINA F K B, NASSRALLAH N, CHOUKCHOU-BRAHAM A, et al. Use of Pistacia lentiscus leaves, after extraction of their oil, as a new biosorbent for the removal of dyes from water[J]. Euro-Mediterr J Environ Integrat, 2021, 6(2): 18. |
| 32 | 李艳春, 张鹏会, 王德乾, 等. 磁性银杏叶生物炭对罗丹明B的吸附特性[J]. 功能材料, 2019, 50(5): 5121-5127. |
| LI Y H, ZHANG P H, WANG D Q, et al. Adsorption characteristics of rhodamine B by magnetic ginkgo biloba biochar[J]. J Funct Mater, 2019, 50(5): 5121-5127. | |
| 33 | 王章鸿, 郭海艳, 沈飞, 等. 热解条件对生物炭性质和氮、磷吸附性能的影响[J]. 环境科学学报, 2015, 35(9), 2805-2812. |
| WANG Z H, GUO H Y, SHEN F, et al. Effect of pyrolysis conditions on the properties and nitrogen and phosphorus adsorption performance of biochar[J]. Acta Sci Circum, 2015, 35(9): 2805-2812 | |
| 34 | 常帅帅, 张学杨, 王洪波, 等. 木屑生物炭的制备及其对Pb2+的吸附特性研究[J]. 生物质化学工程, 2020, 54(3): 37-44. |
| CHANG S S, ZHANG X Y, WANG H B, et al. Preparation of wood chip biochar and its adsorption characteristics on Pb2+[J]. Biomass Chem Eng, 2020, 54(3): 37-44. | |
| 35 | BELLO O S, ADEGOKE K A, INYINBOR A A, et al. Trapping rhodamine B dye using functionalized mango (Mangifera indica) pod[J]. Water Environ Res, 2021, 93(10): 2308-2328. |
| [1] | HU Xumin, LI Guishui, CHENG Lijun, HAO Liang. Preparation and Photocatalytic Properties of Bismuth Subcarbonate Modified with Surfactants [J]. Chinese Journal of Applied Chemistry, 2018, 35(6): 692-699. |
| [2] | WANG Dejia, XU Yongqian, SUN Shiguo, LI Hongjuan. Advanced in Fluorescent Probes Based on Excited State Intramolecular Proton Transfer [J]. Chinese Journal of Applied Chemistry, 2018, 35(1): 1-20. |
| [3] | WANG Cui, ZHANG Feiyun, LYU Rongwen, ZHANG Shufen. Gold Nanopaticle Surface Energy Transfer and Its Application for Thiols Detection [J]. Chinese Journal of Applied Chemistry, 2018, 35(1): 60-67. |
| [4] | WANG Song, LI Yang, LI Fei, CHENG Xiaohong. Microwave Hydrothermal Synthesis and Photocatalytic Properties of ZnO Nano-/Microparticles [J]. Chinese Journal of Applied Chemistry, 2017, 34(2): 220-224. |
| [5] | HUANG Qi, SONG Haohan, WANG Xiao, PANG Lanfang, ZHOU Yanmei. Synthesis of a New Rhodamine-Based Probe for Selective Detection of Ni2+ in Aqueous Solution [J]. Chinese Journal of Applied Chemistry, 2017, 34(12): 1468-1473. |
| [6] | WANG Haoran, YANG Weicheng, TU Yahui, YANG Chao, FANG Chao, LI Meihua, LUO Yong, WU Fanhong. Synthesis and Properties of Stable Isotope Deuterium Labeled D4-Rhodamine B [J]. Chinese Journal of Applied Chemistry, 2016, 33(8): 939-944. |
| [7] | ZOU Xiaomei, CHEN Yan, ZHU Xingwang, LIU Gaopeng, GUO Xinwei, LEI Qin, KE Xiaoxue, LI Shuaixing, HUA Yingjie, WANG Chongtai. Preparation of the Keggin Type Chromium Substituted Phosphotungstates/Titanium Dioxide Nano Film and Its Visible Photocatalytic Performance [J]. Chinese Journal of Applied Chemistry, 2016, 33(3): 320-329. |
| [8] | TUERDI Ailijiang, CHEN Pei, ABDUKAYUM Abdukader, TUERHONG Muhetaer. Preparation and Photocatalytic Kinetic of Bismuth Doped Mesoporous Titanium Dioxide [J]. Chinese Journal of Applied Chemistry, 2016, 33(2): 213-220. |
| [9] | SUN Linlin, ZHOU Yehong, WANG Fei, SHUANG Shaomin, DONG Chuan. Adsorption Properties of Carboxymethyl-β-cyclodextrin Functionalized Ferroferric Oxide Magnetic Nanocomposites on Rhodamine B [J]. Chinese Journal of Applied Chemistry, 2015, 32(1): 110-117. |
| [10] | LI Mengting, LIU Haicheng, ZENG Xing, XU Xiaonan, ZOU Xiaomei, HUA Yingjie*, WANG Chongtai*. Visible Light Catalysis of Keggin-type Cr-Substituted Heteropolyanion/D301R [J]. Chinese Journal of Applied Chemistry, 2014, 31(08): 965-970. |
| [11] | GONG Guizhen*, CAO Hong. Analysis of Petroleum Ether and Ethanol Extractable Species from Rice Straw [J]. Chinese Journal of Applied Chemistry, 2014, 31(06): 726-731. |
| [12] | WANG Ruizhi, WANG Zhenzhen, ZHANG Ming, LI Fangfang, GUO Han, WANG Aijun, FENG Jiuju*. Room-temperature Synthesis and Photocatalytic Activity of Lamella Flower-like ZnO [J]. Chinese Journal of Applied Chemistry, 2014, 31(03): 316-322. |
| [13] | WU Yunxia1,2, HUANG Jing1,2, YIN Zhengzhi2, ZENG Yanbo2, ZHANG Zulei2, LI Lei2*. Properties of Rhodamine B′ Magnetic Molecularly Imprinted Polymers Prepared from the Suspension Polymerization Method [J]. Chinese Journal of Applied Chemistry, 2013, 30(12): 1481-1488. |
| [14] | HAN Liyan1, FAN Xiaoqiang2, ZHANG Zhe2, WANG Hongxuan2, LIU Jian2*. Catalytic Effect of Configuration Structures of Transition Metal Ions in Catalysts with Highly Dispersed and Isolated Active Sites on the Selective Oxidation of Propane [J]. Chinese Journal of Applied Chemistry, 2013, 30(05): 516-522. |
| [15] | TAN Kejun*, WU Fei, DAI Xu, ZHENG Li. Determination of Perfluorooctane Sulfonate by Light Scattering-Fluorescence Ratio Method [J]. Chinese Journal of Applied Chemistry, 2013, 30(02): 220-224. |
| Viewed | ||||||||||||||||||||||||||||||||||||||||||||||||||
|
Full text 655
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
Abstract 434
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||