
应用化学 ›› 2024, Vol. 41 ›› Issue (7): 998-1009.DOI: 10.19894/j.issn.1000-0518.240029
氮杂环咪唑离子液体用于水系锌离子电池负极无枝晶保护
徐磊1,2, 王龙洋1,2, 桃李1,2( ), 张浩男1,2, 贾鑫旺1,2, 万厚钊1,2, 张军1,2, 王浩1,2(
), 张浩男1,2, 贾鑫旺1,2, 万厚钊1,2, 张军1,2, 王浩1,2( )
)
- 1.湖北大学微电子学院,武汉 430062
2.湖北江城实验室,武汉 430205
-
收稿日期:2024-01-29接受日期:2024-05-15出版日期:2024-07-01发布日期:2024-08-03 -
通讯作者:桃李,王浩 -
基金资助:湖北省自然科学基金项目(2022CFB402);国家自然科学基金项目(52272198)
Long-Term Aqueous Zinc-Ion Batteries without Dendrites Protected by Nitrogen Heterocyclic Imidazole Ionic Liquid
Lei XU1,2, Long-Yang WANG1,2, Li TAO1,2( ), Hao-Nan ZHANG1,2, Xin-Wang JIA1,2, Hou-Zhao WAN1,2, Jun ZHANG1,2, Hao WANG1,2(
), Hao-Nan ZHANG1,2, Xin-Wang JIA1,2, Hou-Zhao WAN1,2, Jun ZHANG1,2, Hao WANG1,2( )
)
- 1.School of Microelectronics,Hubei University,Wuhan 430062,China
2.Hubei Yangtze Memory Laboratories,Wuhan 430205,China
-
Received:2024-01-29Accepted:2024-05-15Published:2024-07-01Online:2024-08-03 -
Contact:Li TAO,Hao WANG -
About author:wangh@hubu.edu.cn
litao@hubu.edu.cn
-
Supported by:the Natural Science Foundation of Hubei Province(2022CFB402);the National Natural Science Foundation of China(52272198)
摘要:
根据可持续发展的需要以及更好地去实现碳达峰、碳中和,水系锌离子电池由于其安全可靠、成本低及离子电导率高等特点逐渐进入人们的视线。 相比于传统的锂离子电池,可充电的水系锌离子电池具有很高的安全稳定性,同时,锌金属资源丰富、理论容量(820 mA·h/g)高、氧化还原电位(-0.762 V(vs.SHE))低,这也缓解了金属锂的资源压力。 但是,电解质中的活性水会严重腐蚀锌负极,发生析氢反应(HER),并产生羟基硫酸锌(ZHS)等副产物以及引起枝晶生长,从而使电池循环性能大大降低,达不到储能要求。 结果表明,通过离子液体添加剂优化水系锌离子电解液,实现了循环过程中无枝晶的锌负极,其中氯化1-氰丁基-3-甲基咪唑(MCBI)添加剂在最佳添加浓度条件下,Zn//Cu半电池200圈库伦效率可达99.37%,Zn//Zn对称电池在小电流密度下可稳定循环1600 h以上,即使在10 mA/cm2、5 mA·h/cm2下也能稳定循环1000 h以上; 全电池循环500次后依旧有88.5%的高容量保持率。 这项工作解决了水系锌离子电池枝晶问题,并在优化电解质体系方面提供了一个全新的视角。
中图分类号:
引用本文
徐磊, 王龙洋, 桃李, 张浩男, 贾鑫旺, 万厚钊, 张军, 王浩. 氮杂环咪唑离子液体用于水系锌离子电池负极无枝晶保护[J]. 应用化学, 2024, 41(7): 998-1009.
Lei XU, Long-Yang WANG, Li TAO, Hao-Nan ZHANG, Xin-Wang JIA, Hou-Zhao WAN, Jun ZHANG, Hao WANG. Long-Term Aqueous Zinc-Ion Batteries without Dendrites Protected by Nitrogen Heterocyclic Imidazole Ionic Liquid[J]. Chinese Journal of Applied Chemistry, 2024, 41(7): 998-1009.
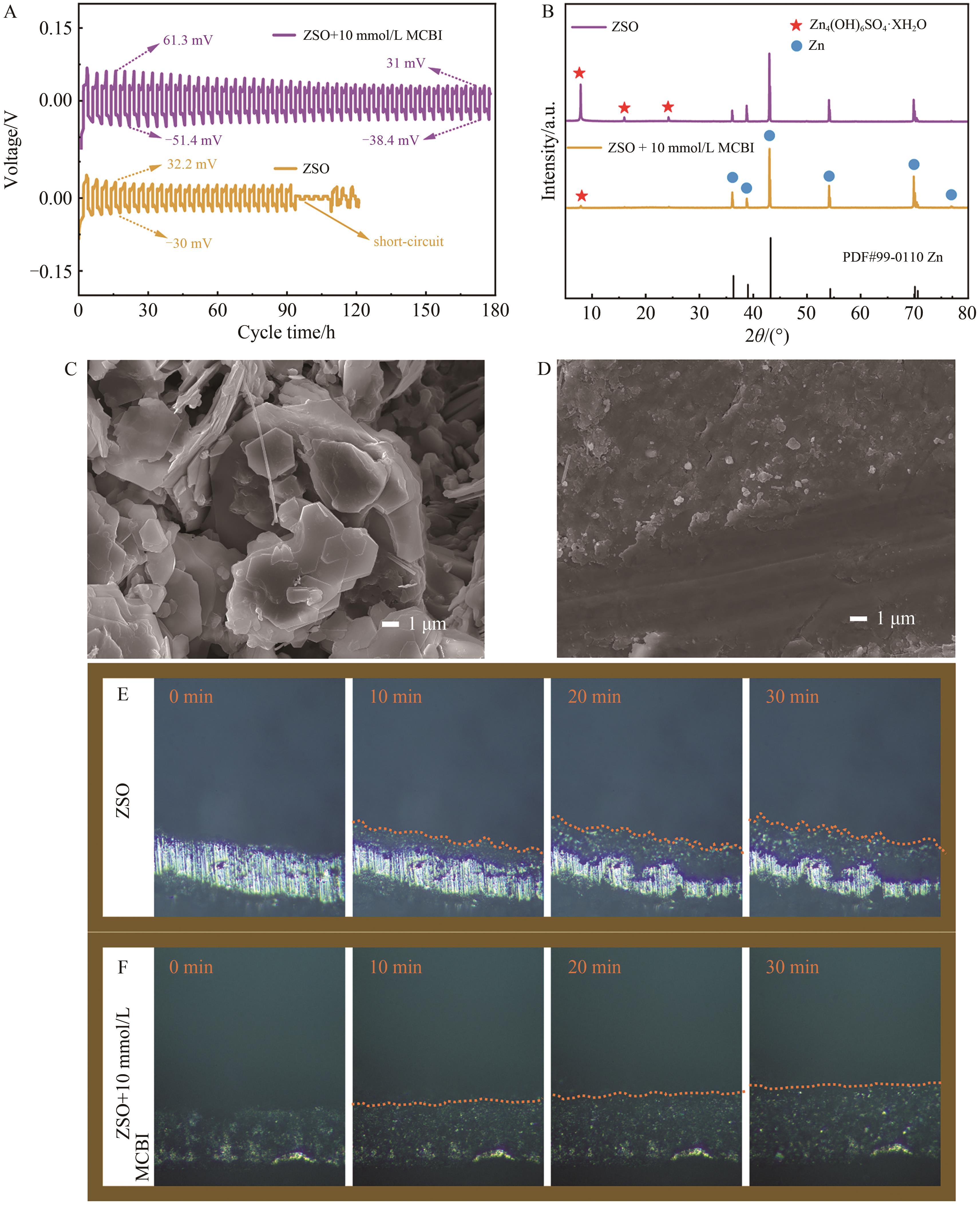
图2 锌离子电池的测试与负极表征图像A. Comparison of Zn//Zn symmetric battery cycling performance in electrolyte with or without ionic liquid additives; B. XRD pattern of zinc anode after electroplating/stripping cycle in different solutions; C. SEM image of Zn anode after circulating in pure ZSO electrolyte; D. SEM image of Zn anode after adding 10 mmol/L MCBI; E, F. Deposition of zinc foil in different electrolytes by in-situ optical microscopy
Fig.2 Test and characterization images of zinc-ion batteries
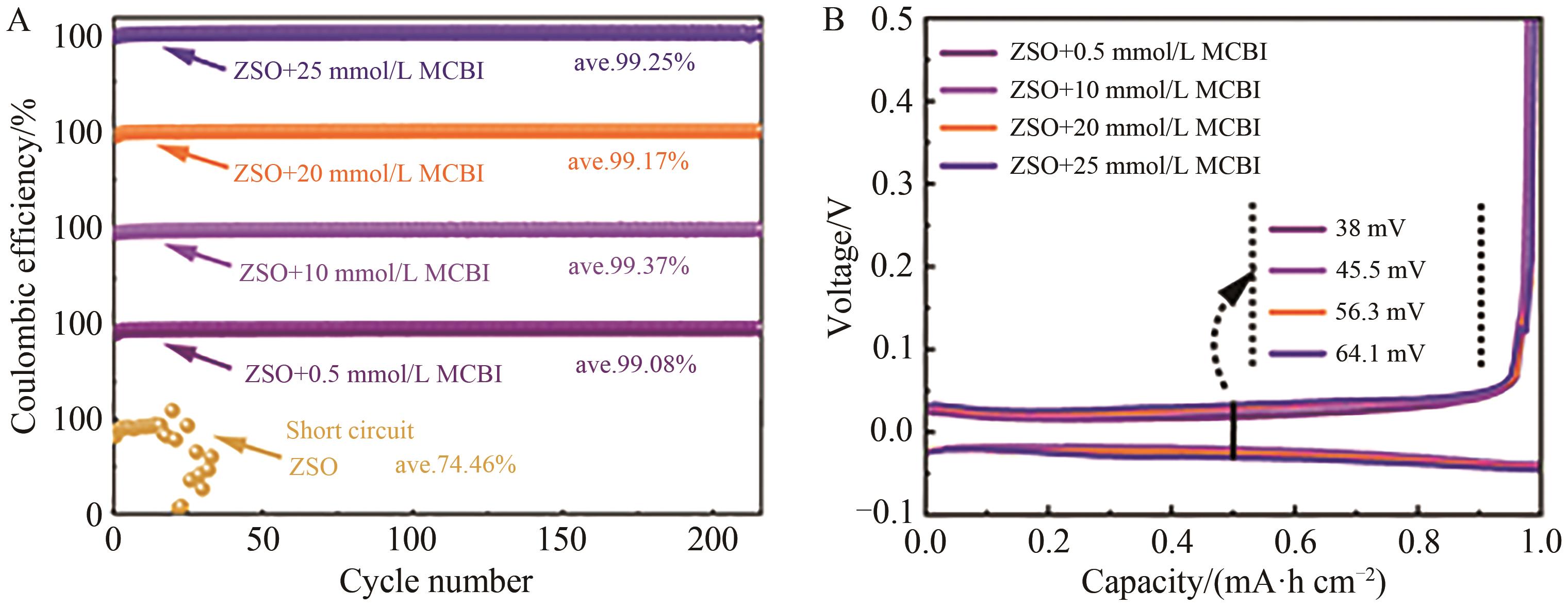
图3 扣式半电池的循环性能A. Coulomb efficiency of Zn//Cu asymmetric batteries under the action of MCBI additive with different concentration; B. Performance curve of Zn//Cu half battery 20th turn galvanizing/stripping under different concentration of MCBI additives
Fig.3 Cycle performance of asymmetric batteries
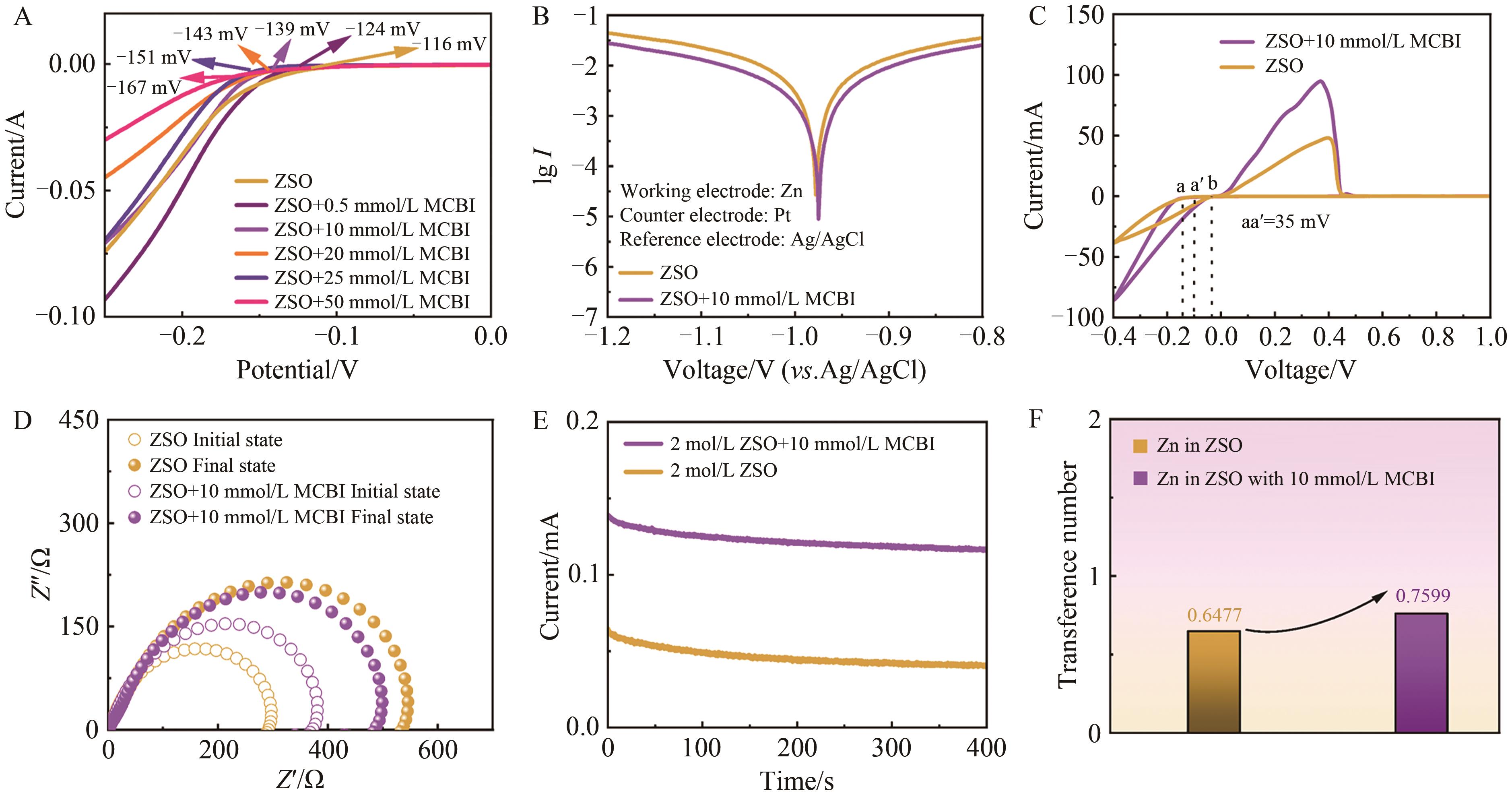
图4 锌离子电池负极的电化学性能A. LSV performance curves of different concentrations of MCBI additive electrolyt; B. Tafel corrosion polarization curve of MCBI electrolyte; C. CV curves of Zn//Cu batteries in different electrolytes were determined by cyclic voltammetry; D. Electrochemical impedance spectroscopy was used to measure the impedance of pure ZSO and MCBI additives before and after timing current; E. CA curve measured by chronoamperometry; F. Zinc ion transfer number fitting diagram
Fig.4 Electrochemical properties of anode of zinc ion battery
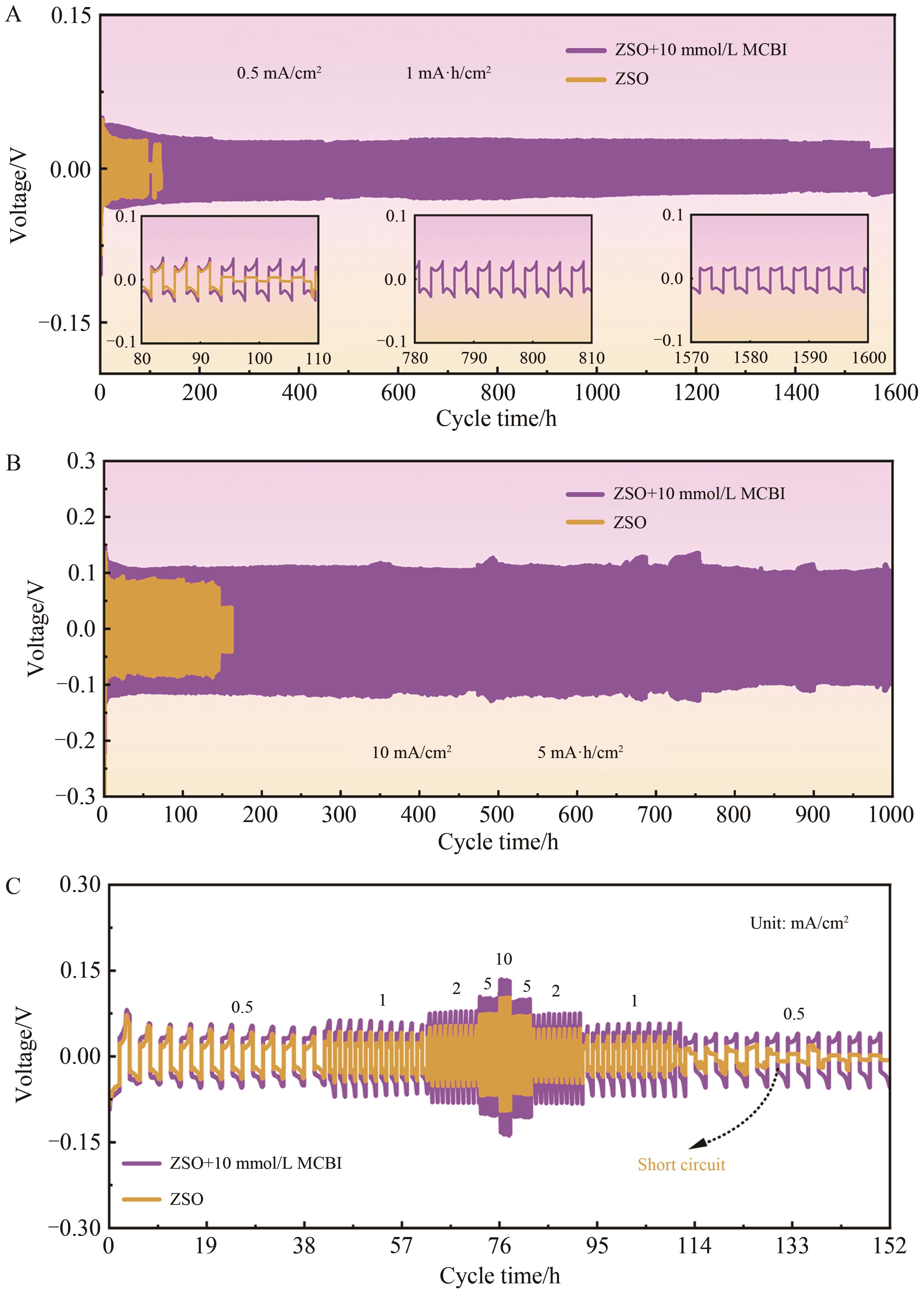
图5 Zn//Zn对称扣式电池的循环性能曲线A. Cyclic performance curve of symmetrical battery under 0.5 mA/cm2,1 mA·h/cm2 condition; B. Cyclic performance curve of symmetrical battery under 10 mA/cm2, 5 mA·h/cm2 condition; C. Rate performance of symmetrical batteries
Fig.5 Cycle performance curve of Zn//Zn symmetric battery
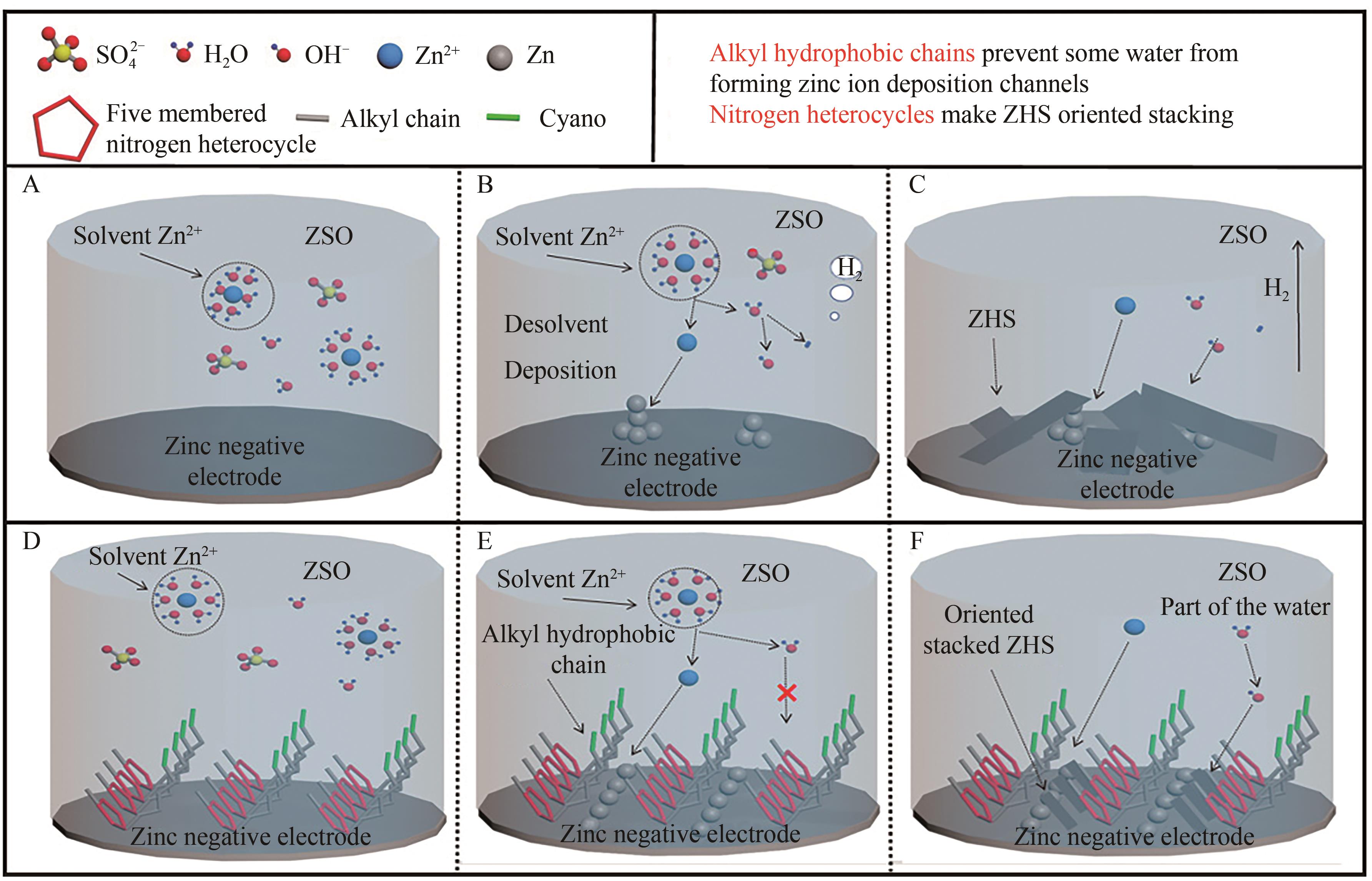
图6 Zn沉积的反应过程示意图以及对应Zn2+迁移和沉积的示意图A-C. Reaction diagram when the electrolyte is 2 mol/L ZnSO4; D-F. Reaction diagram when the electrolyte is 2 mol/L ZnSO4+10 mmol/L MCBI
Fig. 6 Diagram of reaction process of Zn deposition and corresponding Zn2+ migration and deposition
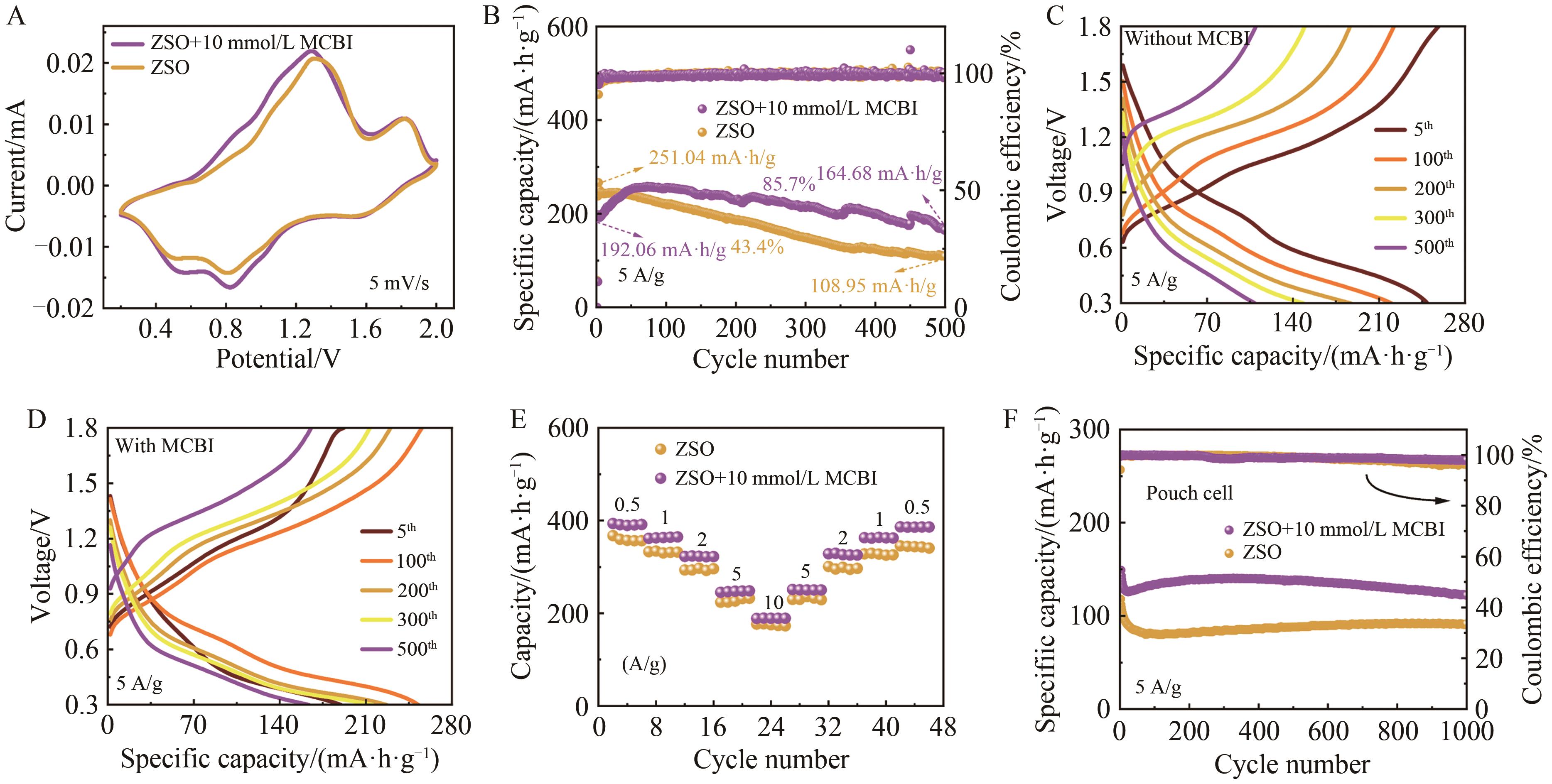
图7 Zn//VO2全电池的测试A. CV image of Zn//VO2 button full battery at 5 mV/s sweep speed in two electrolytes; B. Full battery cycle performance of Zn//VO2 button full battery at 5 A/g current density; C. GCD curves of Zn//VO2 full battery with different cycles in pure ZnSO4 electrolyte; D. GCD curves of Zn//VO2 full battery with different cycles under the action of MCBI additive; E. Rate performance of Zn//VO2 button full battery under two electrolytes; F. Zn//VO2 package full battery cycle performance
Fig.7 Zn//VO2 whole cell test
| 1 | CHEN M Z, ZHANG Y Y, XING G C, et al. Electrochemical energy storage devices working in extreme conditions[J]. Energ Environ Sci, 2021,14(6): 3323-3351. |
| 2 | 衡永丽, 谷振一, 郭晋芝, 等. 水系锌离子电池用钒基正极材料的研究进展[J]. 物理化学学报, 2021, 37(3): 2005013. |
| HENG Y L, GU Z Y, GUO J Z, et al. Research progresses on vanadium-based cathode mate‐rials for aqueous zinc-ion batteries[J]. Acta Phys Chim Sin,2021, 37(3): 2005013. | |
| 3 | LU X, WANG Y, XU X, et al. Polymer-based solid-state electrolytes for high-energy-density lithium-ion batteries-review[J]. Adv Energy Mater, 2023, 13(38): 2301746. |
| 4 | WANG H, LI X, ZENG Q, et al. A novel hyperbranched polyurethane solid electrolyte for room temperature ultra-long cycling lithium-ion batteries[J]. Energy Storage Mater, 2024, 66: 103188. |
| 5 | DONG N, ZHANG F L, PAN H L. Towards the practical application of Zn metal anodes for mild aqueous rechargeable Zn batteries[J]. Chem Sci, 2022, 13(28): 8243-8252. |
| 6 | ZHOU J H, WU F, MEI Y, et al. Establishing thermal infusion method for stable zinc metal anodes in aqueous zinc-ion batteries[J]. Adv Mater, 2022, 34(21): 2200782. |
| 7 | LI C, JIN S, ARCHER L A, et al. Toward practical aqueous zinc-ion batteries for electrochemical energy storage[J]. Joule,2022, 6(8): 1733-1738. |
| 8 | WANG J D, ZHANG B, CAI Z, et al. Stable interphase chemistry of textured Zn anode for rechargeable aqueous batteries[J]. Sci Bull, 2022, 67(7): 716-724. |
| 9 | ZHAO J, YING Y P, WANG G L, et al. Covalent organic framework film protected zinc anode for highly stable rechargeable aqueous zinc-ion batteries[J]. Energy Storage Mater, 2022, 48: 82-89. |
| 10 | WANG S N, LI T Y, YIN Y B, et al. High-energy-density aqueous zinc-based hybrid supercapacitor-battery with uniform zinc deposition achieved by multifunctional decoupled additive[J]. Nano Energy, 2022, 96: 107120. |
| 11 | ZHANG X Q, CHEN J, CAO H, et al. Efficient suppression of dendrites and side reactions by strong electrostatic shielding effect via the additive of Rb2SO4 for anodes in aqueous zinc-ion batteries[J]. Small, 2023, 19(52): 2303906. |
| 12 | CHEN R W, ZHANG W, HUANG Q B, et al. Trace amounts of triple-functional additives enable reversible aqueous zinc-ion batteries from a comprehensive perspective[J]. Nano-Micro Lett, 2023, 15(1): 81. |
| 13 | TAO L, GUAN K L, YANG R, et al. Dual-protected zinc anodes for long-life aqueous zinc ion battery with bifunctional interface constructed by zwitterionic surfactants[J]. Energy Storage Mater, 2023, 63: 102981. |
| 14 | SHI M, WANG R, HE J, et al. Multiple redox-active cyano-substituted organic compound integrated with MXene for high-performance flexible aqueous K-ion battery[J]. Chem Eng J, 2022, 450: 138238. |
| 15 | CHEN J, ZHOU W, QUAN Y, et al. Ionic liquid additive enabling anti-freezing aqueous electrolyte and dendrite-free Zn metal electrode with organic/inorganic hybrid solid electrolyte interphase layer[J]. Energy Storage Mater, 2022, 53: 629-637. |
| 16 | YAN Q, HU Z, LIU Z, et al. Synergistic interaction between amphiphilic ion additive groups for stable long-life zinc ion batteries[J]. Energy Storage Mater, 2024, 67: 103299. |
| 17 | 刘欢, 马宇, 曹斌, 等. MXenes 在水系锌离子电池中的应用研究进展[J]. 物理化学学报, 2023, 39(5): 2210027. |
| LIU H, MA Y, CAO B, et al. Recent progress of MXenes in aqueous zinc-ion batteries[J]. Acta Phys Chim Sin, 2023, 39(5): 2210027. | |
| 18 | LIU Z X, WANG R, MA Q W, et al. A dual-functional organic electrolyte additive with regulating suitable overpotential for building highly reversible aqueous zinc ion batteries[J]. Adv Funct Mater, 2023: 2214538. |
| 19 | ZHENG H, HUANG Y, XIAO J, et al. Multi-protection of zinc anode via employing a natural additive in aqueous zinc ion batteries[J]. Chem Eng J, 2023, 468: 143834. |
| 20 | JI H J, HAN Z Q, LIN Y H, et al. Stabilizing zinc anode for high-performance aqueous zinc ion batteries via employing a novel inositol additive[J]. J Alloy Compd, 2022, 914: 165231. |
| 21 | YANG J Z, YIN B S, SUN Y, et al. Zinc anode for mild aqueous zinc-ion batteries: challenges, strategies, and perspectives[J]. Nano-Micro Lett, 2022, 14: 1-47. |
| 22 | GUO S, QIN L P, ZHANG T S, et al. Fundamentals and perspectives of electrolyte additives for aqueous zinc-ion batteries[J]. Energy Storage Mater, 2021, 34: 545-562. |
| 23 | CAO H, HUANG X M, LIU Y, et al. An efficient electrolyte additive of tetramethylammonium sulfate hydrate for dendritic-free zinc anode for aqueous zinc-ion batteries[J]. J Colloid Interface Sci, 2022, 627: 367-374. |
| 24 | GENG Y F, PAN L, PENG Z Y, et al. Electrolyte additive engineering for aqueous Zn ion batteries[J]. Energy Storage Mater, 2022, 51: 733-755. |
| 25 | THIEU N A, LI W, CHEN X J, et al. Synergistically stabilizing zinc anodes by molybdenum dioxide coating and Tween 80 electrolyte additive for high-performance aqueous zinc-ion batteries[J]. ACS Appl Mater Interfaces, 2023, 15: 55570-55586. |
| 26 | LIU Z X, WANG R, GAO Y C, et al. Low-cost multi-function electrolyte additive enabling highly stable interfacial chemical environment for highly reversible aqueous zinc ion batteries[J]. Adv Funct Mater, 2023, 33: 2308463. |
| 27 | ZHOU W J, CHEN M F, TIAN Q H, et al. Stabilizing zinc deposition with sodium lignosulfonate as an electrolyte additive to improve the life span of aqueous zinc-ion batteries[J]. J Colloid Interface Sci, 2021, 601: 486-494. |
| 28 | DONG H Y, YAN S X, LI T F, et al. Chelating dicarboxylic acid as a multi-functional electrolyte additive for advanced Zn anode in aqueous Zn-ion batteries[J]. J Power Sources, 2023, 585: 233593. |
| 29 | YIN J Y, LIU H L, LI P, et al. Integrated electrolyte regulation strategy: trace trifunctional tranexamic acid additive for highly reversible Zn metal anode and stable aqueous zinc ion battery[J]. Energy Storage Mater, 2023, 59: 102800. |
| 30 | HONG L, WU X M, WANG L Y, et al. Highly reversible zinc anode enabled by a cation-exchange coating with Zn-ion selective channels[J]. ACS Nano, 2022, 16(4): 6906-6915. |
| 31 | GUAN Q L, LI J H, LI L J, et al. In situ construction of organic anion-enriched interface achieves ultra-long life aqueous zinc-ion battery[J]. Chem Eng J, 2023, 476: 146534. |
| 32 | YAO R, QIAN L, SUI Y M, et al. A versatile cation additive enabled highly reversible zinc metal anode[J]. Adv Energy Mater, 2022, 12(2): 2102780. |
| 33 | CAO P H, ZHOU X Y, WEI A R, et al. Fast-charging and ultrahigh-capacity zinc metal anode for high-performance aqueous zinc-ion batteries[J]. Adv Funct Mater, 2021, 31(20): 2100398. |
| 34 | TIAN Z, ZOU Y, LIU G, et al. Electrolyte solvation structure design for sodium ion batteries[J]. Adv Sci, 2022, 9(22): 2201207. |
| 35 | CHENG H, SUN Q, LI L, et al. Emerging era of electrolyte solvation structure and interfacial model in batteries[J]. ACS Energy Lett, 2022, 7(1): 490-513. |
| 36 | LI L, CHENG H, ZHANG J, et al. Quantitative chemistry in electrolyte solvation design for aqueous batteries[J]. ACS Energy Lett, 2023, 8(2): 1076-1095. |
| 37 | XIE C L, LI Y H, WANG Q, et al. Issues and solutions toward zinc anode in aqueous zinc-ion batteries: a mini review[J]. Carbon Energy, 2020, 2(4): 540-560. |
| 38 | HAN C, LI W J, LIU H K, et al. Principals and strategies for constructing a highly reversible zinc metal anode in aqueous batteries[J]. Nano Energy, 2020, 74: 104880. |
| 39 | YANG S, CHEN A, TANG Z J, et al. Regulating the electrochemical reduction kinetics by the steric hindrance effect for a robust Zn metal anode[J]. Energ Environ Sci, 2024, 17(3): 1095-1106. |
| 40 | SU K L M, ZHANG X Y, ZHANG X Q, et al. Polar small molecular electrolyte additive for stabilizing Zn anode[J]. Chem Eng J, 2023, 474: 145730. |
| 41 | WU C, SUN C, REN K, et al. 2-Methyl imidazole electrolyte additive enabling ultra-stable Zn anode[J]. Chem Eng J, 2023, 452: 139465. |
| 42 | ZHAO Y, HONG H, ZHONG L, et al. Zn-rejuvenated and SEI-regulated additive in zinc metal battery via the iodine post-functionalized zeolitic imidazolate framework-90[J]. Adv Energy Mater, 2023, 13(28): 2300627. |
| 43 | ZHANG Q, MA Y, LU Y, et al. Designing anion-type water-free Zn2+ solvation structure for robust Zn metal anode[J]. Angew Chem, 2021, 133(43): 23545-23552. |
| 44 | CHEN Y M, GONG F C, DENG W J, et al. Dual-function electrolyte additive enabling simultaneous electrode interface and coordination environment regulation for zinc-ion batteries[J]. Energy Storage Mater, 2023, 58: 20-29. |
| 45 | QUAN Y H, YANG M, CHEN M F, et al. Electrolyte additive of sorbitol rendering aqueous zinc-ion batteries with dendrite-free behavior and good anti-freezing ability[J]. Chem Eng J, 2023, 458: 141392. |
| [1] | 张润, 战宏梅, 赵晨阳, 程延祥, 秦川江. 含氟膦氧衍生物修饰的纯蓝光钙钛矿发光二极管[J]. 应用化学, 2024, 41(6): 830-838. |
| [2] | 程羽, 何灵均, 林楚园, 林慧, 肖富玉, 赖文斌, 钱庆荣, 黄晓霞, 陈庆华, 曾令兴. 水系锌离子电池宽温域性能的电解质改性策略研究进展[J]. 应用化学, 2024, 41(3): 349-364. |
| [3] | 李远杰, 范冰冰, 张君丽, 周艳梅. 氨基酸离子液体在能量存储和生物质资源化中的研究进展[J]. 应用化学, 2024, 41(3): 391-404. |
| [4] | 李慧慧, 姚开胜, 赵亚南, 范李娜, 田钰琳, 卢伟伟. 离子液体调控合成Pt-Pd双金属纳米材料及其催化氨硼烷水解释氢[J]. 应用化学, 2023, 40(4): 597-609. |
| [5] | 陈启航, 许飞健, 王锋, 傅双婵, 于英豪. 低共熔溶剂及其在抗静电领域中应用的研究进展[J]. 应用化学, 2023, 40(3): 341-359. |
| [6] | 王路飞, 甄蒙蒙, 沈伯雄. 贫电解液下电催化剂对调控锂硫电池性能的研究进展[J]. 应用化学, 2023, 40(2): 188-209. |
| [7] | 石雪建, 刘万强, 王春丽, 程勇, 王立民. 钾离子电池用Sb基负极材料研究进展[J]. 应用化学, 2023, 40(2): 210-228. |
| [8] | 刘成员, 于江玉, 李奉翠, 刘智伟. 拉曼光谱测试技术在可充电铝离子电池储能机理的研究进展[J]. 应用化学, 2023, 40(10): 1347-1358. |
| [9] | 宋林虎, 李世友, 王洁, 张晶晶, 张宁霜, 赵冬妮, 徐菲. 锂离子电池电解液除酸除水添加剂的研究进展[J]. 应用化学, 2022, 39(5): 697-706. |
| [10] | 吴双, 赵德扬, 吴胜寒, 魏立纲, 刘娜, 安庆大. 1-丁基-3-甲基咪唑甲基磺酸盐水溶液中酚型木质素单体溶解的二维相关红外光谱分析[J]. 应用化学, 2022, 39(10): 1600-1609. |
| [11] | 何欧文, 孙长富, 于宏兵. 廉价离子液体体系中玉米芯生成糠醛优化[J]. 应用化学, 2022, 39(02): 272-282. |
| [12] | 李小欧, 顾雪松, 刘亚栋, 季生象. 193 nm化学放大光刻胶研究进展[J]. 应用化学, 2021, 38(9): 1105-1118. |
| [13] | 钮占宁, 唐好庆, 郑超, 田甜, 郑立允. 密度泛函理论研究烷基乙醇仲胺溴离子液体表面改性四氧化三铁[J]. 应用化学, 2021, 38(7): 825-835. |
| [14] | 周超, 生程钜, 闻林林. 咪唑盐类聚离子液体抗菌剂的制备及其在水凝胶敷料中的应用[J]. 应用化学, 2021, 38(1): 51-59. |
| [15] | 刘宁, 王丹凤, 伍素云, 刘水林, 付琳, 刘跃进. 短孔道介孔分子筛Zr-Ce-SBA-15固载酸性离子液体催化合成双酚F[J]. 应用化学, 2020, 37(9): 1038-1047. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
