应用化学 ›› 2024, Vol. 41 ›› Issue (3): 349-364.DOI: 10.19894/j.issn.1000-0518.230238
• 综合评述 • 上一篇
水系锌离子电池宽温域性能的电解质改性策略研究进展
程羽1, 何灵均1, 林楚园1, 林慧1, 肖富玉1, 赖文斌1, 钱庆荣1,2, 黄晓霞3( ), 陈庆华1,2, 曾令兴1,2(
), 陈庆华1,2, 曾令兴1,2( )
)
- 1.福建师范大学环境与资源学院,聚合物资源绿色循环利用教育部工程研究中心,福州 350007
2.南开大学化学学院,先进能源材料化学教育部重点实验室,天津 300071
3.福建水利电力职业技术学院,三明 366000
-
收稿日期:2023-08-09接受日期:2024-01-01出版日期:2024-03-01发布日期:2024-04-09 -
通讯作者:黄晓霞,曾令兴 -
作者简介:zenglingxing@fjnu.cdu.cn
hxx202308@163.com;
-
基金资助:国家重点研发计划项目(2023YFC3906300);国家自然科学基金(21801251);福建省雏鹰计划青年拔尖人才、福建省重点基金(2023J02013);福建省级科技创新重点项目(2022G02022);福州市对外科技合作项目(2022-Y-004)
Progress Research on Electrolyte Modification Strategy to Improve the Performance of Aqueous Zinc-Ion Batteries Within the Wide Temperature Range
Yu CHENG1, Ling-Jun HE1, Chu-Yuan LIN1, Hui LIN1, Fu-Yu XIAO1, Wen-Bin LAI1, Qing-Rong QIAN1,2, Xiao-Xia HUANG3( ), Qing-Hua CHEN1,2, Ling-Xing ZENG1,2(
), Qing-Hua CHEN1,2, Ling-Xing ZENG1,2( )
)
- 1.Engineering Research Center of Polymer Green Recycling of Ministry of Education,College of Environment and Resources,Fujian Normal University,Fuzhou 350007,China
2.(Key Laboratory of Advanced Energy Materials Chemistry (Ministry of Education),College of Chemistry,Nankai University,Tianjin 300071,China )
3.Fujian College of Water Conservancy and Electric Power,Sanming 366000,China
-
Received:2023-08-09Accepted:2024-01-01Published:2024-03-01Online:2024-04-09 -
Contact:Xiao-Xia HUANG,Ling-Xing ZENG -
Supported by:the National Key R&D Program Project of China(2023YFC3906300);the National Natural Science Foundation of China(21801251);Fujian Eagle Programme for Young Top Talents, Fujian Provincial Key Fund(2023J02013);the Provincial Science and Technology Innovation Key Project(2022G02022);Fuzhou City Foreign Science and Technology Cooperation Project(2022-Y-004)
摘要:
在资源短缺、能源需求倍增的当今世界,水系锌离子电池(AZIBs)作为一种大规模储能技术以其具有高安全性、低成本、高容量和快速充放电等优势脱颖而出。随着对能源多元化应用场景的增加,AZIBs被开发并应用于多种极端环境。然而,电池中的自由水分子会引发一系列的不良反应,导致电池出现容量下降和寿命缩短的问题。在低温条件下,溶剂水的冻结会引起AZIBs的离子电导率降低、电荷转移阻抗增加,导致电池速率性能下降。在高温条件下,溶剂水的快速蒸发会产生气泡和气体膨胀,水诱导的副反应加剧,同时电极材料也会出现腐蚀和溶解,从而影响电池寿命。针对这些挑战,在这篇综述中,分别总结了针对水系锌离子电池在高温与低温下的研究进展,提出了适用于低温、高温以及同时适用于高低温的电解质策略,重点研究了高浓度电解质、凝胶电解质、电解质添加剂和共晶电解质降低电解质凝固点、提高低温电化学性能的机理,并对进一步提高水系锌离子电池的宽温域性能和工业应用进行了展望。
中图分类号:
引用本文
程羽, 何灵均, 林楚园, 林慧, 肖富玉, 赖文斌, 钱庆荣, 黄晓霞, 陈庆华, 曾令兴. 水系锌离子电池宽温域性能的电解质改性策略研究进展[J]. 应用化学, 2024, 41(3): 349-364.
Yu CHENG, Ling-Jun HE, Chu-Yuan LIN, Hui LIN, Fu-Yu XIAO, Wen-Bin LAI, Qing-Rong QIAN, Xiao-Xia HUANG, Qing-Hua CHEN, Ling-Xing ZENG. Progress Research on Electrolyte Modification Strategy to Improve the Performance of Aqueous Zinc-Ion Batteries Within the Wide Temperature Range[J]. Chinese Journal of Applied Chemistry, 2024, 41(3): 349-364.
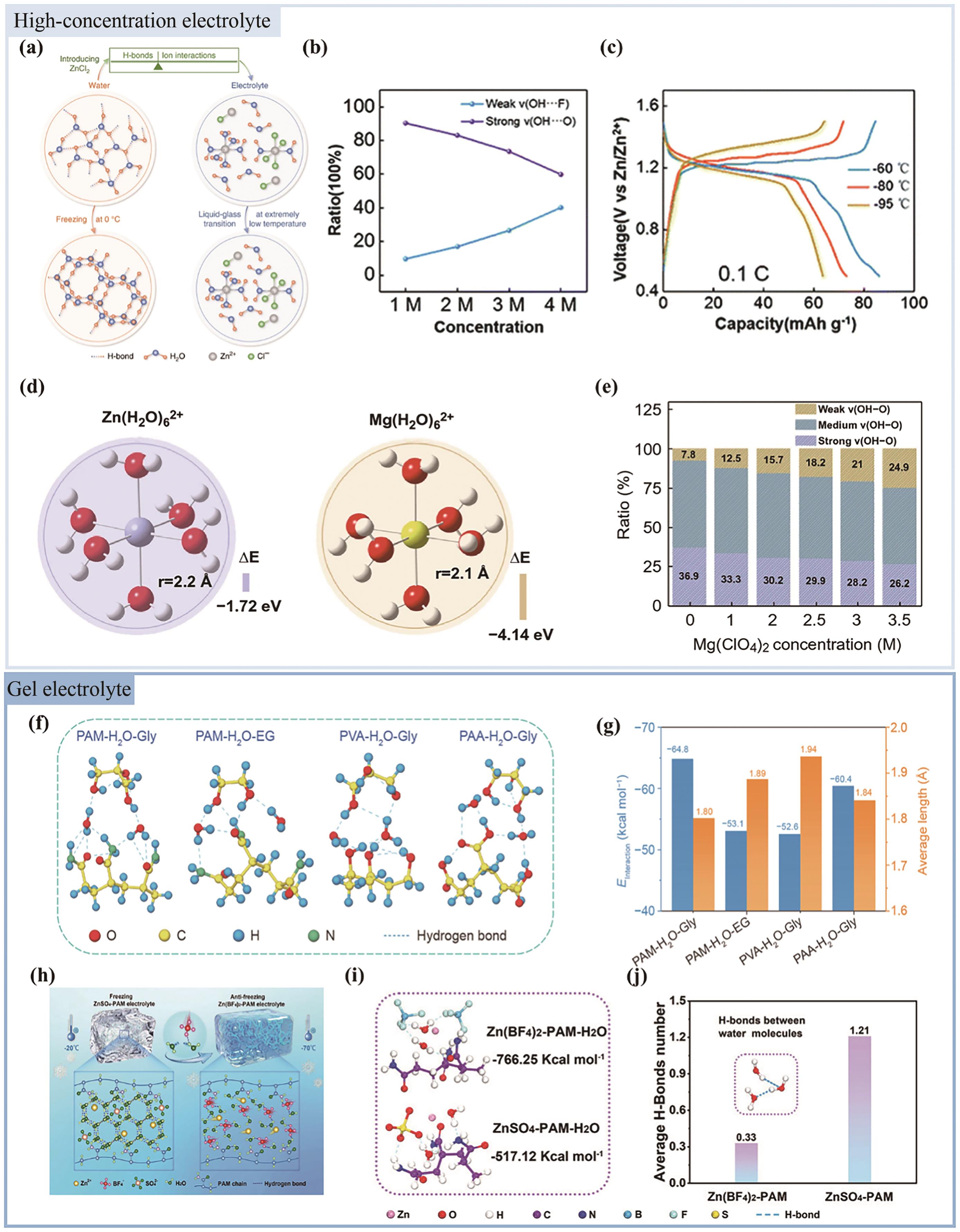
图2 (a)“盐包水”的作用机理[29]; (b)不同浓度Zn(BF4)2中2种氢键的占比[38]; (c) Zn//TCBQ电池在-95~-60 ℃温度范围内的充放电曲线[38]; (d)计算Zn2+溶剂化构型和Mg2+溶剂化构型的形成能和水合半径[53]; (e)不同类型氢键的比例[37]; (f) PAM-H2O-Gly、PAM-H2O-EG、PVA-H2O-Gly和PAA-H2O-Gly的DFT优化结构[53]; (g) PAM-H2O-Gly、PAM-H2O-EG、PVA-H2O-Gly和PAA-H2O-Gly的相互作用能和氢键的平均长度[51]; (h)防冻水凝胶电解质的原理图和机理[54]; (i)通过DFT模拟计算得到Zn(BF4)2-PAM-H2O体系和ZnSO4-PAM-H2O体系的结合能[54]; (j) MD模拟得到的水分子间形成氢键的平均数[54]
Fig.2 (a) The mechanism of “water-in-salt” electrolyte action[29]; (b) The proportion of two hydrogen bonds in different concentrations of Zn(BF4)2[38]; (c) The charge-discharge curves of Zn//TCBQ battery in the temperature range from -95 ℃ to -60 ℃ were obtained[38]; (d) The calculated formation energy and hydrated radius of Zn2+ solvation configuration and Mg2+ solvation configuration[51]; (e) The ratio of different types of HBs[37]; (f) DFT optimized structures of PAM-H2O-Gly, PAM-H2O-EG, PVA-H2O-Gly and PAA-H2O-Gly were obtained; (g) The interaction energies and the average length of hydrogen bonds of PAM-H2O-Gly, PAM-H2O-EG, PVA-H2O-Gly and PAA-H2O-Gly were calculated[51]; (h) The principle diagram and mechanism of antifreeze hydrogel electrolyte[54]; (i) The binding energy of Zn(BF4)2-PAM-H2O system and ZnSO4-PAM-H2O system obtained by DFT simulation[54]; (j) The average number of H-bonds formed between water molecules obtained by MD simulation[54]
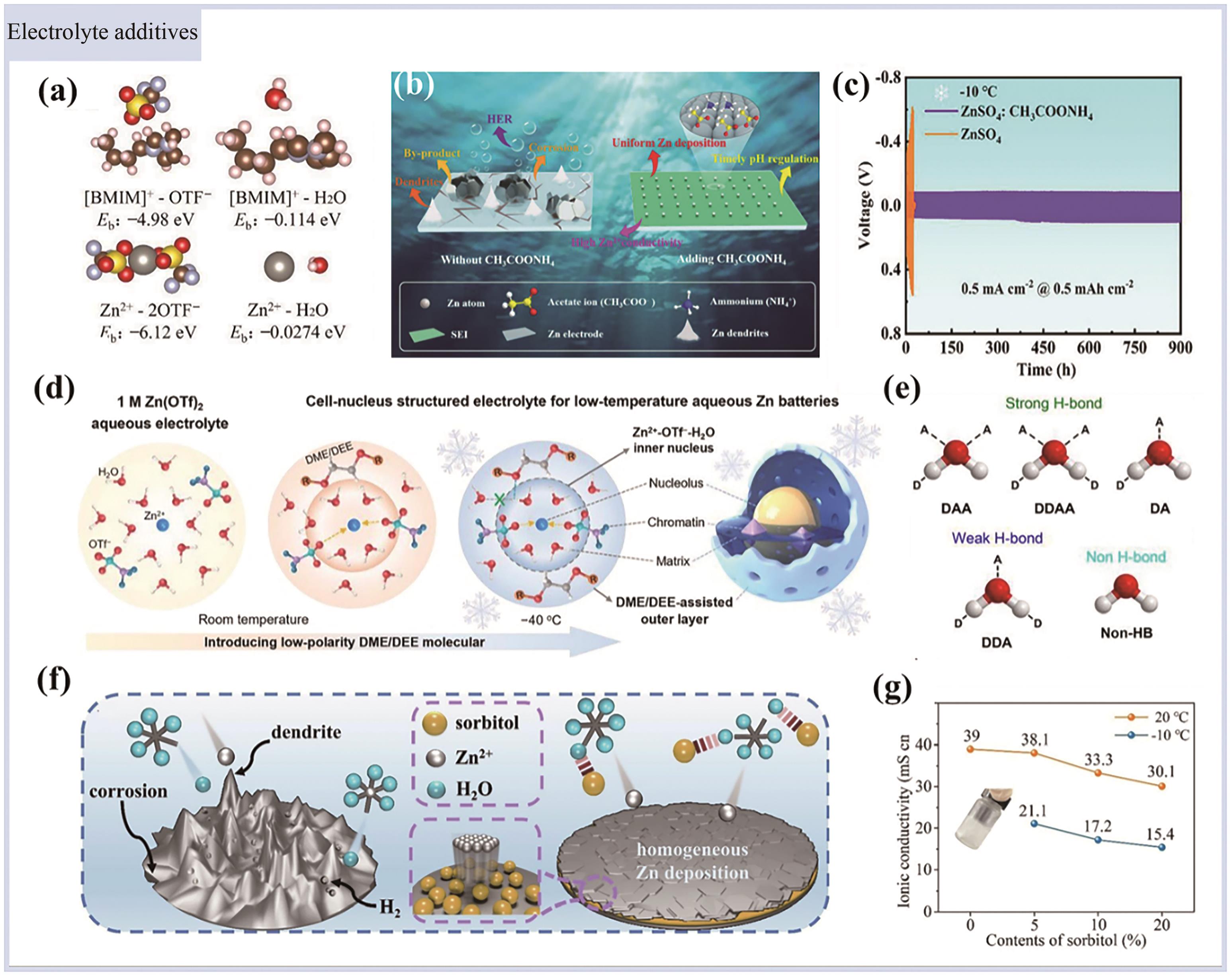
图3 (a)不同组合电解质的Eb值[67]; (b)含/不含CH3COONH4添加剂的电解质中Zn表面演变的示意图[68]; (c)锌对称电池-10 ℃下在ZnSO4∶CH3COONH4电解质和ZnSO4电解质中的长寿命循环性能[68]; (d)低温水系锌电池的细胞-核结构电解质设计示意图[69]; (e)表示具有DAA、DDAA、DA、DDA和非HB的H2O分子的示意模式[69]; (f)有无山梨醇添加剂的锌沉积过程对比[70]; (g)不同山梨醇添加量电解液的离子电导率值, g中插图为未添加山梨醇的2 mol/L ZnSO4水系电解液在-10 ℃的照片[70]
Fig.3 (a) Eb values of different couples[67]; (b) A schematic diagram of Zn surface evolution in electrolytes with/without CH3COONH4 additives[68]; (c) Long-life cycling performance of zinc symmetric cells in ZnSO4∶CH3COONH4 and ZnSO4 electrolytes at -10 ℃[68]; (d) Schematic illustration of the cell-nucleus structured electrolyte design for low temperature aqueous Zn batteries[69]; (e) A schematic pattern of H2O molecules with DAA, DDAA, DA, DDA and non-HB[69]; (f) Comparison of Zn deposition process with or without sorbitol additive[70]; (g) Ionic conductivity values of the electrolytes with different amounts of sorbitol, The inset in g shows the photograph of 2 mol/L ZnSO4 aqueous electrolyte without sorbitol at -10 ℃[70]

图4 (a)共晶溶剂化结构的结构示意图[8]; (b)环丁砜的加入破坏了本体H—O—H…O—H网络,并形成一种新型的环丁砜-H2O网络[72]; (c)基于1∶1电解质的Zn-VOH电池在-20 ℃下的倍率性能[72]; (d) -20 ℃不同溶剂化结构对Zn镀层影响的示意图[74]
Fig.4 (a) Structure diagram of cocrystal solvation structure[8]; (b) The addition of sulfolane destroyed the bulk H—O—H…O—H hydrogen bond network and formed a new type of sulfolane-H2O hydrogen bond[72]; (c) The rate performance of Zn-VOH battery based on 1∶1 electrolyte at -20 ℃[72]; (d) The schematic diagram of the effect of different solvation structures on Zn coating at -20 ℃[74]
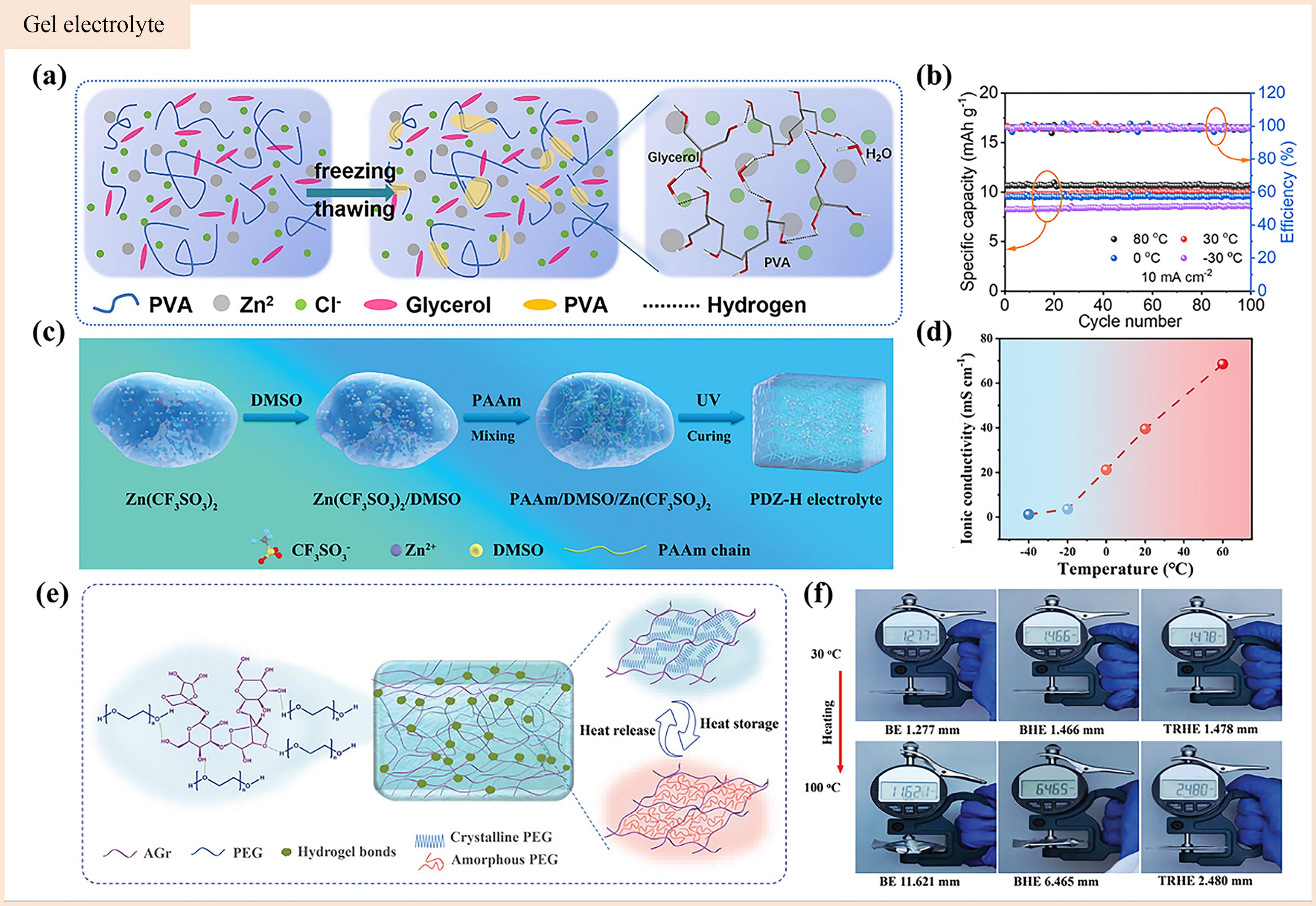
图5 (a) PVA基凝胶电解质示意图[80]; (b)组装的防冻FZHSC在80、30、0和-30 ℃下的循环性能[80]; (c)合成PDZ-H电解质的过程示意图[65]; (d)不同温度下PDZ-H电解质的离子电导率[65]; (e)温度调节水凝胶电解质机制示意图[81]; (f)温度从30 ℃上升到100 ℃时,空白电解质(BE)、基线水凝胶电解质(BHE)和TRHE的袋装电池的表观体积膨胀[81]
Fig.5 (a) The PVA-based gel electrolyte schematic[80]; (b) The cycle performance of the assembled antifreeze FZHSC at 80, 30, 0 and -30 ℃[80]; (c) Schematic diagram of synthesis process of the PDZ-H electrolyte[65]; (d) The ionic conductivity of the PDZ-H electrolyte at different temperature[65]; (e) The schematic diagram of the temperature-regulated hydrogel electrolyte mechanism[81]; (f) When the temperature rises from 30 ℃ to 100 ℃, the apparent volume expansion of the blank electrolyte (BE), the baseline hydrogel electrolyte (BHE) and the TRHE bagged cell[81]

图6 (a) 25 ℃下Zn(OTf)2-H2O和Zn(OTf)2-H2O/PD电解质中不同试剂与Zn2+配位的比率[82]; (b) Zn(OTf)2-PD和Zn(OTf)2-H2O/PD电解质的点火试验[82]; (c) pH值低至3.23的2 mol/L-P5W5共溶剂电解质的酸性环境可能有利于在较宽的温度条件下抑制副产物的生长[76]; 在不含(d)或与(e)多羟基聚合物共溶剂的水电解质中镀锌演示图[76]; (f) 1 mol/L ZnSO4和30 mol/L ZnCl2电解质的电化学反应示意图[83]; (g) V2O5阴极在各种电解质中高温储存(55 ℃)后的电荷保持和电荷回收[83]
Fig.6 (a) The coordination ratios of different reagents to Zn2+ in Zn(OTf)2-H2O and Zn(OTf)2-H2O/PD electrolytes at 25 ℃[82]; (b) Ignition test of Zn(OTf)2-PD electrolyte and Zn (OTf)2-H2O/PD electrolyte[82]; (c) The acidic environment of the 2 mol/L-P5W5 cosolvent electrolyte with a pH value as low as 3.23 may be beneficial to inhibit the growth of by-products at a wide temperature[76]; the zinc plating demonstration diagram in a water electrolyte without (d) or with (e) polyhydroxy polymer cosolvent[76]; (f) The electrochemical reaction diagram of 1 mol/L ZnSO4 and 30 mol/L ZnCl2 electrolyte[83]; (g) The charge retention and charge recovery of V2O5 cathode after high temperature storage (55 ℃) in various electrolytes[83]
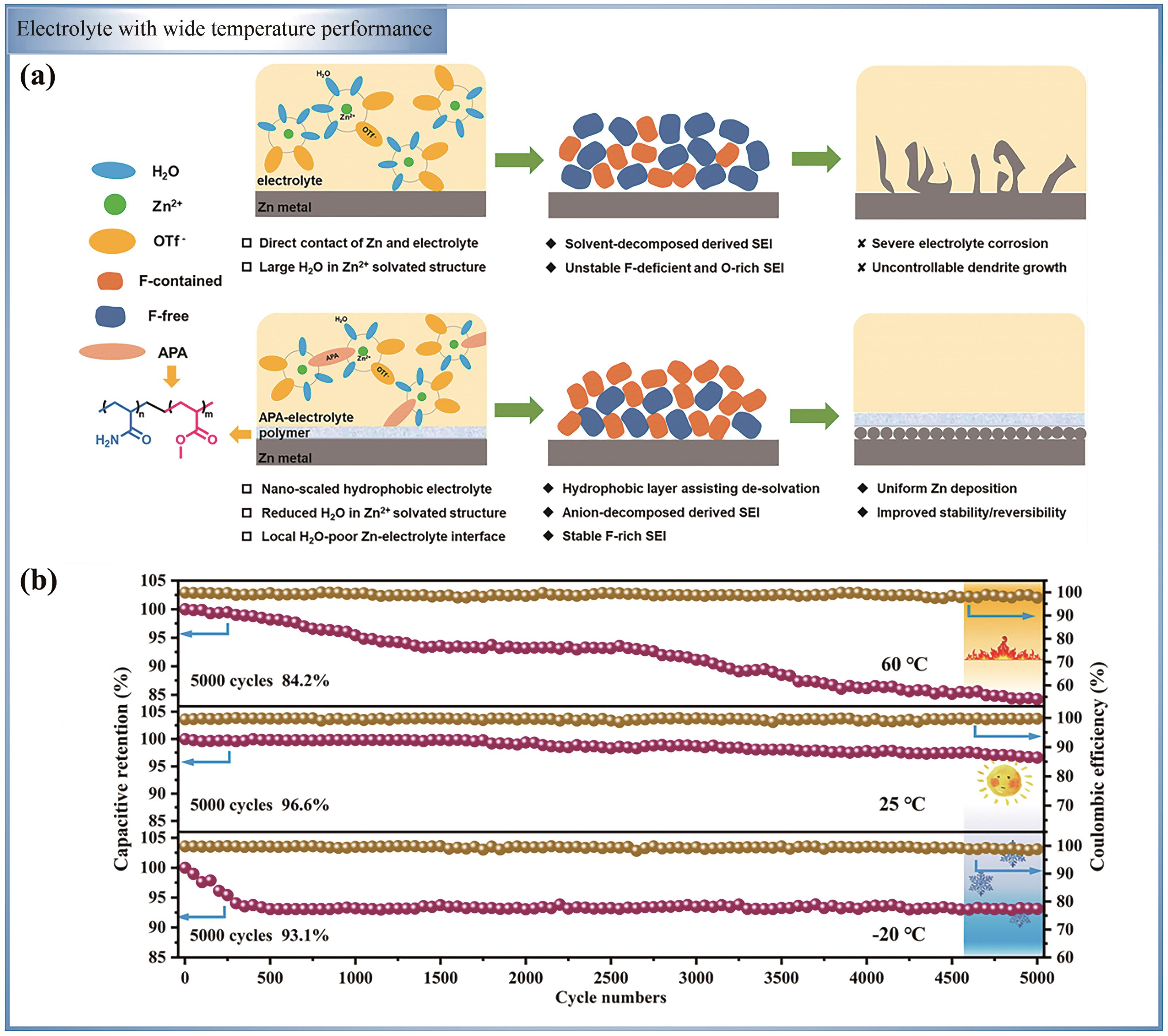
图7 (a)不含和含APA的Zn(OTf)2水性电解质中形态演变的示意图[85]; (b)基于PMPG-25 GPE储能装置的ZIHC在5 A/g的循环性能[86]
Fig.7 (a) The schematic diagram of the morphological evolution of Zn(OTf)2 aqueous electrolytes without and with APA[85]; (b) The cycle performance of ZIHC based on PMPG-25 GPE energy storage device at 5 A/g is studied[86]
| Electrolyte | Category | Cathode | Ionicconductivity/ (mS·cm-1·℃-1) | Operating temperature range/℃ | Freezing point/℃ | Capacity/((mA·h·g-1)|(A·g-1)) | Year |
|---|---|---|---|---|---|---|---|
| 4 mol/L Zn(BF4)2 | High-concentration electrolyte | TCBQ | 1.47/-70 | -95~25 | -122 | -95 ℃:63.5/0.22 | 2021[ |
| 3.5 mol/L Mg(ClO4)2+1 mol/L Zn(ClO4)2 | High-concentration electrolyte | PTO | 1.41/-70 | -70~25 | -121 | -70 ℃:101.5/0.2 | 2021[ |
| PAM-H2O-Gly-20% | Gel electrolyte | SWCNTs/PANI | 0.096 5/-40 | -40~20 | * | -40 ℃:52/1 | 2022[ |
| 3 mol/L Zn(BF4)2-PAM | Gel electrolyte | PANI | 2.38/-70 | -70~25 | * | -70 ℃:33.5/0.25 | 2023[ |
| 0.5 mol/L [BMIM]OTF+3 mol/L Zn(OTF)2 | Electrolyte additives | H11Al2V6O23.2(HAVO) | 27.7/-40 | -40~25 | * | -30 ℃:165/2 | 2022[ |
| ZnSO4∶CH3COONH4 | Electrolyte additives | Zn | * | -10~25 | * | * | 2022[ |
| DME(DME∶DME+H2O=0.15)+1 mol/L Zn(OTf)2 | Electrolyte additives | V2O5 | 1.06/-40 | -40~25 | -52.4 | -40 ℃:212.4/0.5 | 2023[ |
| 10% C6H14O6 | Electrolyte additives | MnO2 | 17.2/-10 | -10~20 | -18 | -10 ℃:101.9/5 | 2023[ |
| 30% 2-propanol/H2O/Zn(OTf)2 | Eutectic electrolyte | V2O5 | * | -20~25 | <-100 | * | 2022[ |
| Zn(TFSI)2-sulfolane-H2O(1∶1) | Eutectic electrolyte | V2O5·nH2O | * | -20~25 | <-80 | * | 2023[ |
| 3 mol/L ZnOAc1.2Cl1.8-DOL | Eutectic electrolyte | AC | * | -20~20 | * | * | 2023[ |
| PVA/Gly/ZnCl2 | Gel electrolyte | δ-MnO2 | 0.21/-50 | -30~80 | -105.73 | * | 2022[ |
| PAAm/DMSO/Zn(CF3SO3)2 | Gel electrolyte | Zn3V2O8 | * | -40~60 | <-40 | * | 2022[ |
| PEG+AGr | Gel electrolyte | Na5V12O32(NVO) | * | 30~100 | * | * | 2022[ |
| Zn(OTf)2-H2O/PD | Electrolyte additives | Te | 12.8/100 | 30~100 | * | 100 ℃:195.7/2C | 2022[ |
| 50%PEG+50%H2O+2 mol/L Zn(OTf)2 | Electrolyte additives | PANI@V2O5 | 87/100 | -20~80 | -93.04 | 60 ℃:310/2 | 2022[ |
| 30 mol/L ZnCl2 | High-concentration electrolyte | V2O5 | * | 55 | * | * | 2021[ |
| 1% APA+3 mol/L Zn(OTf)2 | Electrolyte additives | Zn | * | -30~50 | * | * | 2023[ |
| PMPG-25 GPE+2 mol/L ZnCl2 | Gel electrolyte | AC/CC | * | -20~60 | * | * | 2023[ |
表1 AZIBs物理化学性能及电化学性能对比表
Table 1 Comparison of physical and chemical properties and electrochemical properties of aqueous zinc ion batteries
| Electrolyte | Category | Cathode | Ionicconductivity/ (mS·cm-1·℃-1) | Operating temperature range/℃ | Freezing point/℃ | Capacity/((mA·h·g-1)|(A·g-1)) | Year |
|---|---|---|---|---|---|---|---|
| 4 mol/L Zn(BF4)2 | High-concentration electrolyte | TCBQ | 1.47/-70 | -95~25 | -122 | -95 ℃:63.5/0.22 | 2021[ |
| 3.5 mol/L Mg(ClO4)2+1 mol/L Zn(ClO4)2 | High-concentration electrolyte | PTO | 1.41/-70 | -70~25 | -121 | -70 ℃:101.5/0.2 | 2021[ |
| PAM-H2O-Gly-20% | Gel electrolyte | SWCNTs/PANI | 0.096 5/-40 | -40~20 | * | -40 ℃:52/1 | 2022[ |
| 3 mol/L Zn(BF4)2-PAM | Gel electrolyte | PANI | 2.38/-70 | -70~25 | * | -70 ℃:33.5/0.25 | 2023[ |
| 0.5 mol/L [BMIM]OTF+3 mol/L Zn(OTF)2 | Electrolyte additives | H11Al2V6O23.2(HAVO) | 27.7/-40 | -40~25 | * | -30 ℃:165/2 | 2022[ |
| ZnSO4∶CH3COONH4 | Electrolyte additives | Zn | * | -10~25 | * | * | 2022[ |
| DME(DME∶DME+H2O=0.15)+1 mol/L Zn(OTf)2 | Electrolyte additives | V2O5 | 1.06/-40 | -40~25 | -52.4 | -40 ℃:212.4/0.5 | 2023[ |
| 10% C6H14O6 | Electrolyte additives | MnO2 | 17.2/-10 | -10~20 | -18 | -10 ℃:101.9/5 | 2023[ |
| 30% 2-propanol/H2O/Zn(OTf)2 | Eutectic electrolyte | V2O5 | * | -20~25 | <-100 | * | 2022[ |
| Zn(TFSI)2-sulfolane-H2O(1∶1) | Eutectic electrolyte | V2O5·nH2O | * | -20~25 | <-80 | * | 2023[ |
| 3 mol/L ZnOAc1.2Cl1.8-DOL | Eutectic electrolyte | AC | * | -20~20 | * | * | 2023[ |
| PVA/Gly/ZnCl2 | Gel electrolyte | δ-MnO2 | 0.21/-50 | -30~80 | -105.73 | * | 2022[ |
| PAAm/DMSO/Zn(CF3SO3)2 | Gel electrolyte | Zn3V2O8 | * | -40~60 | <-40 | * | 2022[ |
| PEG+AGr | Gel electrolyte | Na5V12O32(NVO) | * | 30~100 | * | * | 2022[ |
| Zn(OTf)2-H2O/PD | Electrolyte additives | Te | 12.8/100 | 30~100 | * | 100 ℃:195.7/2C | 2022[ |
| 50%PEG+50%H2O+2 mol/L Zn(OTf)2 | Electrolyte additives | PANI@V2O5 | 87/100 | -20~80 | -93.04 | 60 ℃:310/2 | 2022[ |
| 30 mol/L ZnCl2 | High-concentration electrolyte | V2O5 | * | 55 | * | * | 2021[ |
| 1% APA+3 mol/L Zn(OTf)2 | Electrolyte additives | Zn | * | -30~50 | * | * | 2023[ |
| PMPG-25 GPE+2 mol/L ZnCl2 | Gel electrolyte | AC/CC | * | -20~60 | * | * | 2023[ |
| 1 | WANG Y, XIAO F, CHEN X, et al. Extraordinarily stable and wide‐temperature range sodium/potassium-ion batteries based on 1D SnSe2‐SePAN composite nanofibers[J]. Info Mat, 2023, 5: e12467. |
| 2 | 王丹, 侯现飚, 汪兴坤, 等. 应用于锌空气电池的碳包覆铁基纳米颗粒电催化剂研究进展[J]. 应用化学, 2022, 39(10): 1488-1500. |
| WANG D, HOU X B, WANG X K, et al. Research progress of carbon-encapsulated iron‑based nanoparticles electrocatalysts for zinc‑air batteries[J]. Chin J Appl Chem, 2022, 39(10): 1488-1500. | |
| 3 | 宋林虎, 李世友, 王洁, 等. 锂离子电池电解液除酸除水添加剂的研究进展[J]. 应用化学, 2022, 39(5): 697-706. |
| SONG L H, LI S Y, WANG J, et al. Research progress of additives for acid and water removal in electrolyte of lithium ion battery[J]. Chin J Appl Chem, 2022, 39(5): 697-706. | |
| 4 | ZHENG B, LIN X, ZHANG X, et al. Emerging functional porous polymeric and carbonaceous materials for environmental treatment and energy storage[J]. Adv Funct Mater, 2019, 30(41): 1907006. |
| 5 | LIN X, ZHOU G, LIU J, et al. Rechargeable battery electrolytes capable of operating over wide temperature windows and delivering high safety[J]. Adv Energy Mater, 2020, 10(43): 2001235. |
| 6 | LIN X, ZHOU G, ROBSON M J, et al. Hydrated deep eutectic electrolytes for high‐performance Zn-ion batteries capable of low‐temperature operation[J]. Adv Funct Mater, 2021, 32(14): 2109322. |
| 7 | YUAN Z, XIAO F, FANG Y, et al. Defect engineering on VO2(B) nanoleaves/graphene oxide for the high performance of cathodes of zinc-ion batteries with a wide temperature range[J]. J Power Sources, 2023, 559: 232688. |
| 8 | MA Q, GAO R, LIU Y, et al. Regulation of outer solvation shell toward superior low-temperature aqueous zinc-ion batteries[J]. Adv Mater, 2022, 34(49): 2207344. |
| 9 | YUAN Z, YANG X, LIN C, et al. Progressive activation of porous vanadium nitride microspheres with intercalation-conversion reactions toward high performance over a wide temperature range for zinc-ion batteries[J]. J Colloid Interface Sci, 2023, 640(15): 487-497. |
| 10 | XIONG P, ZHANG Y, ZHANG J, et al. Recent progress of artificial interfacial layers in aqueous Zn metal batteries[J]. Energy Chem, 2022, 4(4): 100076. |
| 11 | YAO Q, XIAO F, LIN C, et al. Regeneration of spent lithium manganate into cation-doped and oxygen-deficient MnO2 cathodes toward ultralong lifespan and wide-temperature-tolerant aqueous Zn-ion batteries[J]. Battery Energy, 2023, 2(4): 20220065. |
| 12 | KUNDU D, HOSSEINI VAJARGAH S, WAN L, et al. Aqueous vs. nonaqueous Zn-ion batteries: consequences of the desolvation penalty at the interface[J]. Energy Environ Sci, 2018, 11(4): 881-892. |
| 13 | SU Z, GUO H, ZHAO C. Rational design of electrode-electrolyte interphase and electrolytes for rechargeable proton batteries[J]. Nano-Micro Lett, 2023, 15(1): 96. |
| 14 | ZHANG Y, CHEN Z, QIU H, et al. Pursuit of reversible Zn electrochemistry: a time-honored challenge towards low-cost and green energy storage[J]. NPG Asia Mater, 2020, 12(1): 4. |
| 15 | ZHU C, ZHOU J, WANG Z, et al. Phase diagrams guided design of low-temperature aqueous electrolyte for Zn metal batteries[J]. Chem Eng J, 2023, 454: 140413. |
| 16 | HAN M M, LI T C, CHEN X, et al. Electrolyte modulation strategies for low-temperature Zn batteries[J]. Small, 2023: 2304901. |
| 17 | WANG N, DONG X, WANG B, et al. Zinc-organic battery with a wide operation-temperature window from -70 to 150 ℃[J]. Angew Chem Int Ed, 2020, 59(34): 14577-14583. |
| 18 | ZHANG W, DOONG Q, WANG J, et al. Failure mechanism, electrolyte design, and electrolyte/electrode interface regulation for low-temperature zinc-based batteries[J]. Small Methods, 2023: 2300324. |
| 19 | YUE F, TIE Z W, DENG S Z, et al. An ultralow temperature aqueous battery with proton chemistry[J]. Angew Chem Int Ed, 2021, 60(25): 13882-13886. |
| 20 | LI M, YANG J, SHI Y, et al. Soluble organic cathodes enable long cycle life, high rate, and wide-temperature lithium-ion batteries[J]. Adv Mater, 2022, 34(5): 2107226. |
| 21 | NIAN Q, LIU S, LIU J, et al. All-climate aqueous dual-ion hybrid battery with ultrahigh rate and ultralong life performance[J]. ACS Appl Energy Mater, 2019, 2(6): 4370-4378. |
| 22 | JIANG L, LU Y, ZHAO C, et al. Building aqueous K-ion batteries for energy storage[J]. Nat Energy, 2019, 4(6): 495-503. |
| 23 | YOU Y, YAO H, XIN S, et al. Subzero-temperature cathode for a sodium-ion battery[J]. Adv Mater, 2016, 28(33): 7243-7248. |
| 24 | NIAN Q, WANG J, LIU S, et al. Aqueous batteries operated at -50 ℃[J]. Angew Chem Int Ed, 2019, 58(47): 16994-16999. |
| 25 | GUO Z, WANG T, WEI H, et al. Ice as solid electrolyte to conduct various kinds of ions[J]. Angew Chem Int Ed, 2019, 58(36): 12569-12573. |
| 26 | WANG X, ZHANG Z, XI B, et al. Advances and perspectives of cathode storage chemistry in aqueous zinc-ion batteries[J]. ACS Nano, 2021, 15(6): 9244-9272. |
| 27 | VERMA V, KUMAR S, MANALASTAS W, et al. Undesired reactions in aqueous rechargeable zinc ion batteries[J]. ACS Energy Lett, 2021, 6(5): 1773-1785. |
| 28 | LIU C, XIE X, LU B, et al. Electrolyte strategies toward better zinc-ion batteries[J]. ACS Energy Lett, 2021, 6(3): 1015-1033. |
| 29 | LIU Z, LUO X, QIN L, et al. Progress and prospect of low-temperature zinc metal batteries[J]. Adv Powder Mater, 2022, 1(2): 100011. |
| 30 | TAMTOGL A, BAHN E, SACCHI M, et al. Motion of water monomers reveals a kinetic barrier to ice nucleation on graphene[J]. Nat Commun, 2021, 12(1): 3120. |
| 31 | RAMANUJAPURAM A, YUSHIN G. Understanding the exceptional performance of lithium-ion battery cathodes in aqueous electrolytes at subzero temperatures[J]. Adv Energy Mater, 2018, 8(35): 1802624. |
| 32 | NIAN Q, SUN T, LIU S, et al. Issues and opportunities on low-temperature aqueous batteries[J]. Chem Eng J, 2021, 423: 130253. |
| 33 | ZHANG Q, MA Y, LU Y, et al. Modulating electrolyte structure for ultralow temperature aqueous zinc batteries[J]. Nat Commun, 2020, 11(1): 4463. |
| 34 | LEONARD D P, WEI Z, CHEN G, et al. Water-in-salt electrolyte for potassium-ion batteries[J]. ACS Energy Lett, 2018, 3(2): 373-374. |
| 35 | LUKATSKAYA M R, FELDBLYUM J I, MACKANIC D G, et al. Concentrated mixed cation acetate “water-in-salt” solutions as green and low-cost high voltage electrolytes for aqueous batteries[J]. Energy Environ Sci, 2018, 11(10): 2876-2883. |
| 36 | ZHANG H, LIU X, LI H, et al. High-voltage operation of a V2O5 cathode in a concentrated gel polymer electrolyte for high-energy aqueous zinc batteries[J]. ACS Appl Mater Interfaces, 2020, 12(13): 15305-15312. |
| 37 | SUN T, ZHENG S, DU H, et al. Synergistic effect of cation and anion for low-temperature aqueous zinc-ion battery[J]. Nanomicro Lett, 2021, 13(1): 204. |
| 38 | SUN T, YUAN X, WANG K, et al. An ultralow-temperature aqueous zinc-ion battery[J]. J Mater Chem A, 2021, 9(11): 7042-7047. |
| 39 | PU Y, WANG C, FENG J, et al. Organics-free aqueous hybrid electrolyte for high-performance zinc ion hybrid capacitors operating at low temperature[J]. J Power Sources, 2023, 571: 233061. |
| 40 | BORODIN O, SELF J, PERSSON K A, et al. Uncharted waters: super-concentrated electrolytes[J]. Joule, 2020, 4(1): 69-100. |
| 41 | WANG Z, LI H, TANG Z, et al. Hydrogel electrolytes for flexible aqueous energy storage devices[J]. Adv Funct Mater, 2018, 28(48): 1804560. |
| 42 | ZHAO S, ZUO Y, LIU T, et al. Multi-functional hydrogels for flexible zinc-based batteries working under extreme conditions[J]. Adv Energy Mater, 2021, 11(34): 2101749. |
| 43 | LOU Z, SHEN G. Flexible image sensors with semiconducting nanowires for biomimic visual applications[J]. Small Struct, 2021, 2(7): 2000152. |
| 44 | MA L, CHEN S, WANG D, et al. Super-stretchable zinc-air batteries based on an alkaline-tolerant dual-network hydrogel electrolyte[J]. Adv Energy Mater, 2019, 9(12): 1803046. |
| 45 | MA L, ZHAO Y, JI X, et al. A usage scenario independent “air chargeable” flexible zinc ion energy storage device[J]. Adv Energy Mater, 2019, 9(19): 1900509. |
| 46 | MO F, LIANG G, MENG Q, et al. A flexible rechargeable aqueous zinc manganese-dioxide battery working at -20 ℃[J]. Energy Environ Sci, 2019, 12(2): 706-715. |
| 47 | HAN L, LIU K, WANG M, et al. Mussel-inspired adhesive and conductive hydrogel with long-lasting moisture and extreme temperature tolerance[J]. Adv Funct Mater, 2018, 28(3): 1704195. |
| 48 | LIU T, LIU M, DOU S, et al. Triboelectric-nanogenerator-based soft energy-harvesting skin enabled by toughly bonded elastomer/hydrogel hybrids[J]. ACS Nano, 2018, 12(3): 2818-2826. |
| 49 | YANG J, GAO L, LIU M, et al. Advanced biotechnology for cell cryopreservation[J]. Trans Tianjin Univ, 2019, 26(6): 409-423. |
| 50 | YUK H, ZHANG T, PARADA G A, et al. Skin-inspired hydrogel-elastomer hybrids with robust interfaces and functional microstructures[J]. Nat Commun, 2016, 7: 12028. |
| 51 | WANG R, YAO M, HUANG S, et al. An anti-freezing and anti-drying multifunctional gel electrolyte for flexible aqueous zinc-ion batteries[J]. Sci China Mater, 2022, 65(8): 2189-2196. |
| 52 | LIU L, DOU Q, SUN Y, et al. A moisture absorbing gel electrolyte enables aqueous and flexible supercapacitors operating at high temperatures[J]. J Mater Chem A, 2019, 7(35): 20398-20404. |
| 53 | SUN Y, MA H, ZHANG X, et al. Salty ice electrolyte with superior ionic conductivity towards low‐temperature aqueous zinc ion hybrid capacitors[J]. Adv Funct Mater, 2021, 31(28): 2101277. |
| 54 | SHI Y, WANG R, BI S, et al. An anti‐freezing hydrogel electrolyte for flexible zinc‐ion batteries operating at -70 ℃[J]. Adv Funct Mater, 2023, 33(24): 2214546. |
| 55 | CHEN M, CHEN J, ZHOU W, et al. Realizing an all-round hydrogel electrolyte toward environmentally adaptive dendrite-free aqueous Zn-MnO2 batteries[J]. Adv Mater, 2021, 33(9): 2007559. |
| 56 | YAN C, WANG Y, DENG X, et al. Cooperative chloride hydrogel electrolytes enabling ultralow-temperature aqueous zinc ion batteries by the hofmeister effect[J]. Nanomicro Lett, 2022, 14(1): 98. |
| 57 | GAO J, XIE X, LIANG S, et al. Inorganic colloidal electrolyte for highly robust zinc-ion batteries[J]. Nanomicro Lett, 2021, 13(1): 69. |
| 58 | LI C, XIE X, LIU H, et al. Integrated 'all-in-one' strategy to stabilize zinc anodes for high-performance zinc-ion batteries[J]. Natl Sci Rev, 2022, 9(3): nwab177. |
| 59 | TANG Y, LIU C, ZHU H, et al. Ion-confinement effect enabled by gel electrolyte for highly reversible dendrite-free zinc metal anode[J]. Energy Storage Mater, 2020, 27: 109-116. |
| 60 | CURSARU B, STANESCU P O, TEODORESCU M. The states of water in hydrogels synthesized from diepoxy-terminated poly(ethylene glycol)s and aliphatic polyamines[J]. UPB Sci Bull Ser B, 2010, 72(4): 99-15. |
| 61 | JIANG H, SHIN W, MA L, et al. A high‐rate aqueous proton battery delivering power below -78 ℃ via an unfrozen phosphoric acid[J]. Adv Energy Mater, 2020, 10(28): 2000968. |
| 62 | LU C, CHEN X. All-temperature flexible supercapacitors enabled by antifreezing and thermally stable hydrogel electrolyte[J]. Nano Lett, 2020, 20(3): 1907-1914. |
| 63 | WANG A, ZHOU W, HUANG A, et al. Developing improved electrolytes for aqueous zinc-ion batteries to achieve excellent cyclability and antifreezing ability[J]. J Colloid Interface Sci, 2021, 586: 362-370. |
| 64 | HUANG S, HE S, LI Y, et al. Hydrogen bond acceptor lined hydrogel electrolyte toward dendrite-free aqueous Zn ion batteries with low temperature adaptability[J]. Chem Eng J, 2023, 464: 142607. |
| 65 | LU H, HU J, WANG L, et al. Multi-component crosslinked hydrogel electrolyte toward dendrite-free aqueous Zn ion batteries with high temperature adaptability[J]. Adv Funct Mater, 2022, 32(19): 2112540. |
| 66 | WANG J, ZHU Q, LI F, et al. Low-temperature and high-rate Zn metal batteries enabled by mitigating Zn2+ concentration polarization[J]. Chem Eng J, 2022, 433: 134589. |
| 67 | CHEN J, ZHOU W, QUAN Y, et al. Ionic liquid additive enabling anti-freezing aqueous electrolyte and dendrite-free Zn metal electrode with organic/inorganic hybrid solid electrolyte interphase layer[J]. Energy Storage Mater, 2022, 53: 629-637. |
| 68 | LIN C, YANG X, XIONG P, et al. High-rate, large capacity, and long life dendrite-free Zn metal anode enabled by trifunctional electrolyte additive with a wide temperature range[J]. Adv Sci, 2022, 9(21): 2201433. |
| 69 | DONG Y, ZHANG N, WANG Z, et al. Cell-nucleus structured electrolyte for low-temperature aqueous zinc batteries[J]. J Energy Chem, 2023, 83: 324-332. |
| 70 | QUAN Y, YANG M, CHEN M, et al. Electrolyte additive of sorbitol rendering aqueous zinc-ion batteries with dendrite-free behavior and good anti-freezing ability[J]. Chem Eng J, 2023, 458: 141392. |
| 71 | JIANG L, DONG D, LU Y. Design strategies for low temperature aqueous electrolytes[J]. Nano Res Energy, 2022, 1(1): 9120003. |
| 72 | LI M, WANG X, HU J, et al. Comprehensive H2O molecules regulation via deep eutectic solvents for ultra-stable zinc metal anode[J]. Angew Chem Int Ed, 2023, 62(8): e202215552. |
| 73 | ZHAO Y, LIU H, HUYAN Y, et al. In situ construction of a stable composite solid electrolyte interphase for dendrite-free Zn batteries[J]. J Energy Chem, 2023, 79: 450-458. |
| 74 | LU X, LIU Z, AMARDEEP A, et al. Ultra-stable zinc metal anodes at -20 ℃ through eutectic solvation sheath in chlorine‐functionalized eutectic electrolytes with 1,3‐dioxolane[J]. Angew Chem Int Ed, 2023, 62(33): e202307475. |
| 75 | ZHANG C, ZHANG L, YU G. Eutectic electrolytes as a promising platform for next-generation electrochemical energy storage[J]. Acc Chem Res, 2020, 53(8): 1648-1659. |
| 76 | ZHOU J, YUAN H, LI J, et al. Highly reversible and stable Zn metal anode under wide temperature conditions enabled by modulating electrolyte chemistry[J]. Chem Eng J, 2022, 442: 136218. |
| 77 | HOU Z, LU Z, CHEN Q, et al. Realizing wide-temperature Zn metal anodes through concurrent interface stability regulation and solvation structure modulation[J]. Energy Storage Mater, 2021, 42: 517-525. |
| 78 | XIE X, FU H, FANG Y, et al. Manipulating ion concentration to boost two‐electron Mn4+/Mn2+ redox kinetics through a colloid electrolyte for high‐capacity zinc batteries[J]. Adv Energy Mater, 2021, 12(5): 2102393. |
| 79 | WANG H, CHEN Z, JI Z, et al. Temperature adaptability issue of aqueous rechargeable batteries[J]. Mater Today Energy, 2021, 19: 100577. |
| 80 | JIANG Y, MA K, SUN M, et al. All‐climate stretchable dendrite‐free Zn-ion hybrid supercapacitors enabled by hydrogel electrolyte engineering[J]. Energy Environ Mater, 2022, 6(2): e12357. |
| 81 | MENG Y, ZHANG L, PENG M, et al. Developing thermoregulatory hydrogel electrolyte to overcome thermal runaway in zinc-ion batteries[J]. Adv Funct Mater, 2022, 32(46): 2206653. |
| 82 | WANG J, YANG Y, WANG Y, et al. Working aqueous Zn metal batteries at 100 ℃[J]. ACS Nano, 2022, 16(10): 15770-15778. |
| 83 | TANG X, WANG P, BAI M, et al. Unveiling the reversibility and stability origin of the aqueous V2O5-Zn batteries with a ZnCl2 “Water-in-Salt” electrolyte[J]. Adv Sci, 2021, 8(23): 2102053. |
| 84 | SUN L, SONG Z, DENG C, et al. Electrolytes for aqueous Zn-ion batteries working in wide-temperature range: progress and perspective[J]. Batteries, 2023, 9(7): 386. |
| 85 | NIU B, LI Z, LUO D, et al. Nano-scaled hydrophobic confinement of aqueous electrolyte by a nonionic amphiphilic polymer for long-lasting and wide-temperature Zn-based energy storage[J]. Energy Environ Sci, 2023, 16(4): 1662-1675. |
| 86 | WAN L, ZHANG H, QU M, et al. Polyanion-induced single zinc-ion gel polymer electrolytes for wide-temperature and interfacial stable zinc-ion hybrid capacitors[J]. Energy Storage Mater, 2023, 63: 102982. |
| [1] | 伍凡, 田贺元, 刘鹏, 孙立伟, 张一波, 杨向光. 高氧空位尖晶石型锰基催化剂用于低温NH3-SCR反应[J]. 应用化学, 2023, 40(5): 697-707. |
| [2] | 韩志鹏, 秦冲, 周金向, 余明, 陈兆彬. 浇注型环氧树脂基耐高温中子屏蔽复合材料[J]. 应用化学, 2023, 40(10): 1420-1429. |
| [3] | 熊玉华, 周蕾, 杨世忠, 牟伯中. 低温β-甘露聚糖酶的酶学性质及破胶性能[J]. 应用化学, 2023, 40(1): 134-145. |
| [4] | 宋林虎, 李世友, 王洁, 张晶晶, 张宁霜, 赵冬妮, 徐菲. 锂离子电池电解液除酸除水添加剂的研究进展[J]. 应用化学, 2022, 39(5): 697-706. |
| [5] | 李小欧, 顾雪松, 刘亚栋, 季生象. 193 nm化学放大光刻胶研究进展[J]. 应用化学, 2021, 38(9): 1105-1118. |
| [6] | 刘洋, 张海宝, 陈强. 低温等离子体合成氨研究进展[J]. 应用化学, 2021, 38(6): 622-636. |
| [7] | 胡晨,金翼,朱少青,徐晔,水江澜. 磷酸铁锂电池低温性能的改性方法简述[J]. 应用化学, 2020, 37(4): 380-386. |
| [8] | 潘鸽, 刘芳, 付志磊, 李双双, 许东华, 徐朝华, 石彤非, 王晓伟, 马蕊. 不同色浆质量分数的水性聚氨酯涂料对聚氯乙烯表皮低温爆破性能的影响[J]. 应用化学, 2020, 37(2): 182-189. |
| [9] | 刘西德, 周迪, 张兆顺. 1,2,3,9-四氢咔唑-4-酮生产废液处理及氯化锌回收利用[J]. 应用化学, 2020, 37(1): 117-122. |
| [10] | 苏风梅, 张达, 梁风. 低温等离子体制备与改性纳米催化材料的研究进展[J]. 应用化学, 2019, 36(8): 882-891. |
| [11] | 盛磊,李廷鱼,郭丽芳,李刚,张文栋. 功能化多壁碳纳米管填充的凝胶电解质在染料敏化太阳能电池的应用[J]. 应用化学, 2019, 36(7): 815-822. |
| [12] | 郝斌,赵文武,郁建元,刘进强,刘剑,董秀珍,王秀文. 荧光粉Ba5-3x/2B4O11∶xEu3+的制备及发光性能[J]. 应用化学, 2019, 36(5): 548-553. |
| [13] | 朱云, 洪亮, 金葆康. 高温红外光谱电化学薄层池的制作与表征[J]. 应用化学, 2019, 36(1): 107-113. |
| [14] | 张庆轩, 李金涛, 张梦. 马瑞原油在空气和氮气中的热解-氧化性能[J]. 应用化学, 2018, 35(12): 1470-1477. |
| [15] | 郇彦, 李肖肖, 田雨濛, 王杰, 杨小牛. 多元醇对对苯二异氰酸酯基聚氨酯微孔弹性体的形态与性能影响[J]. 应用化学, 2017, 34(10): 1110-1116. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||