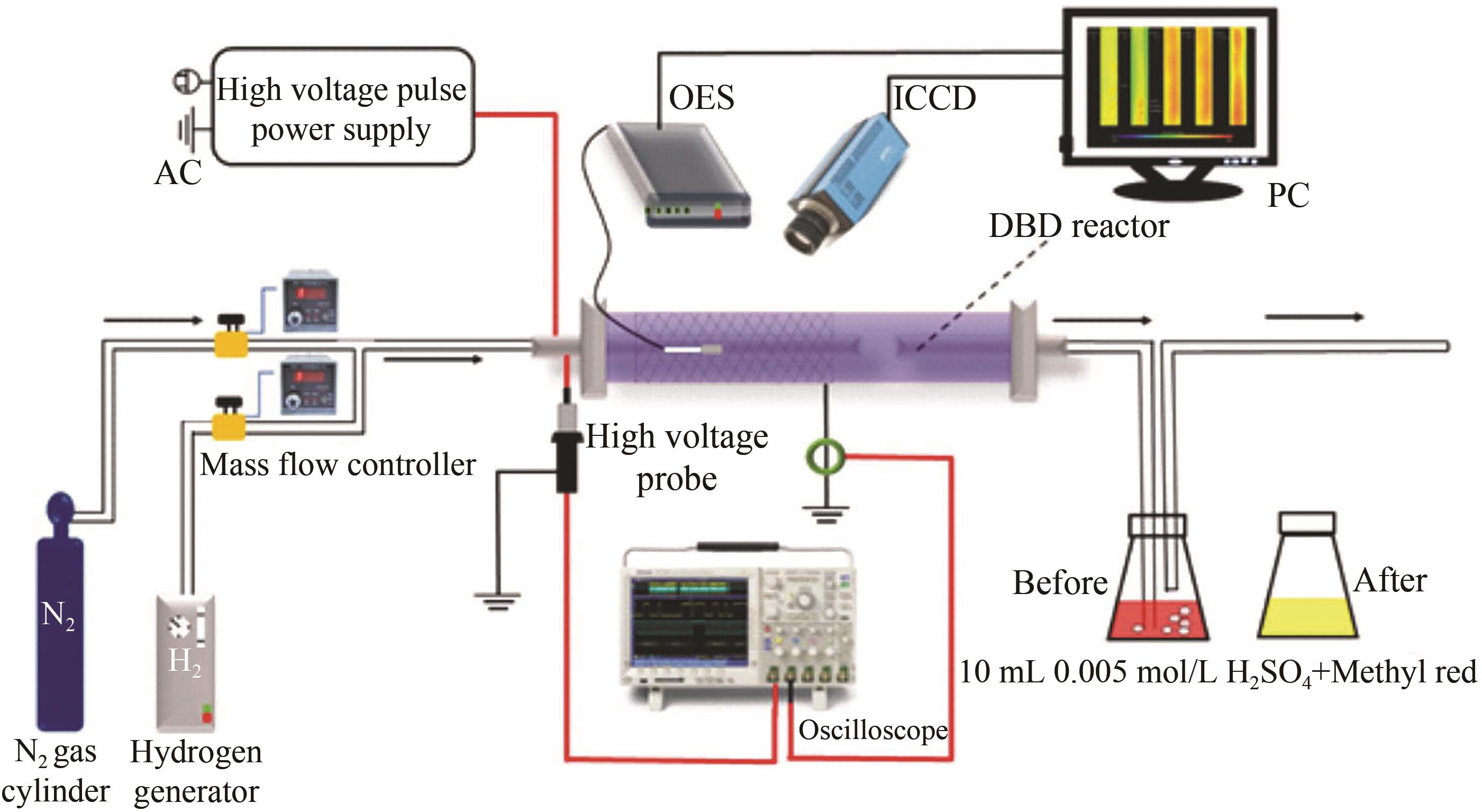
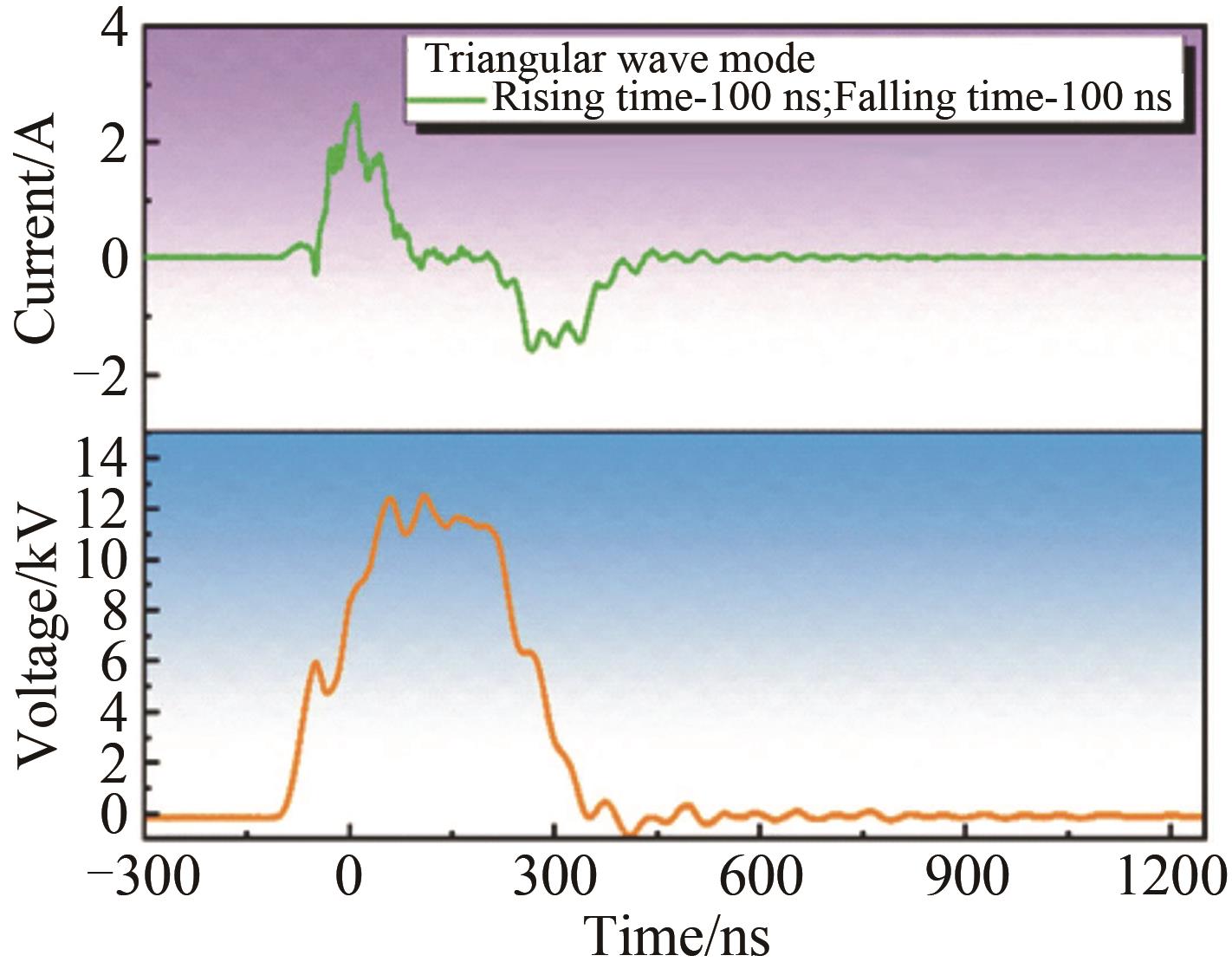
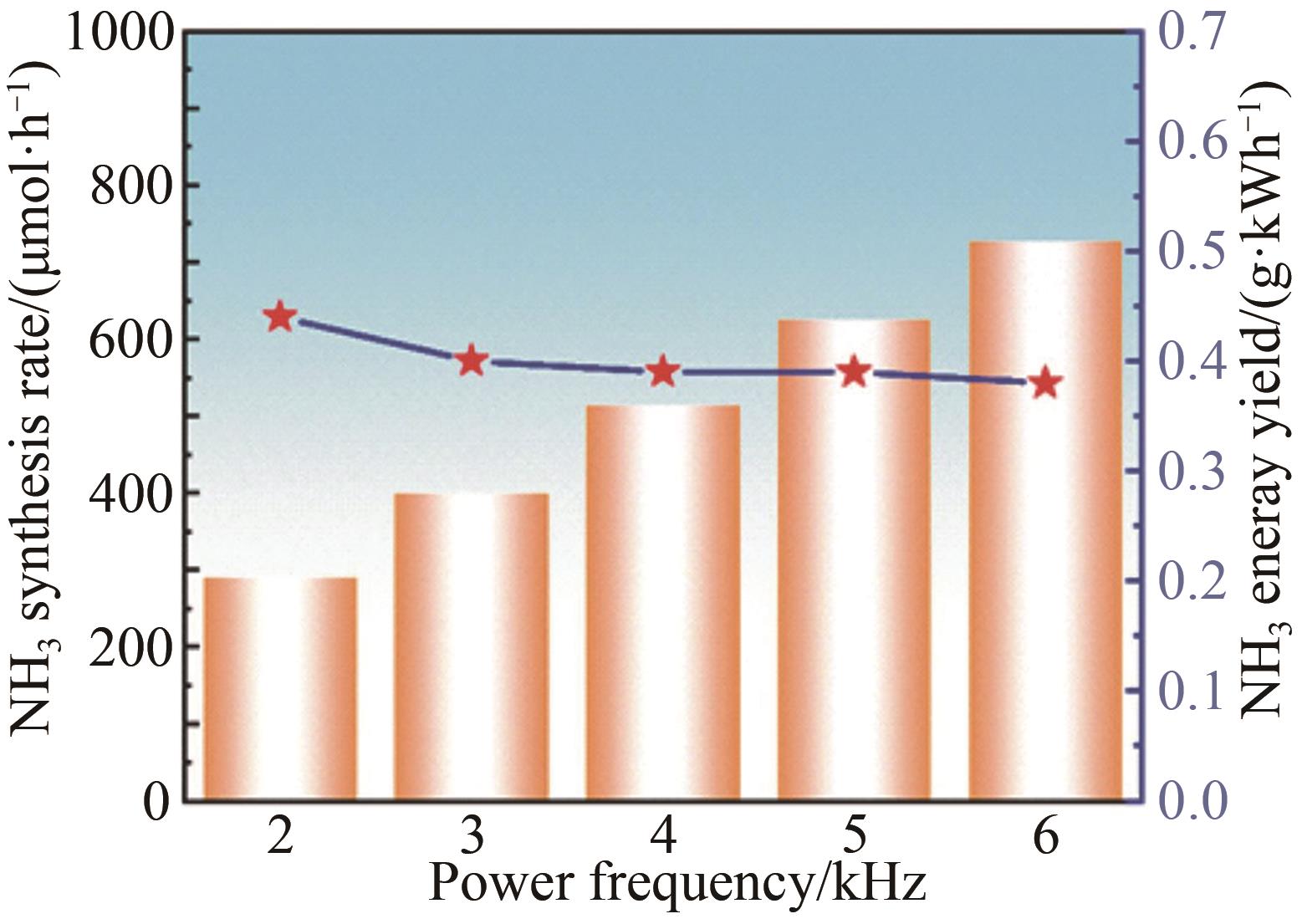
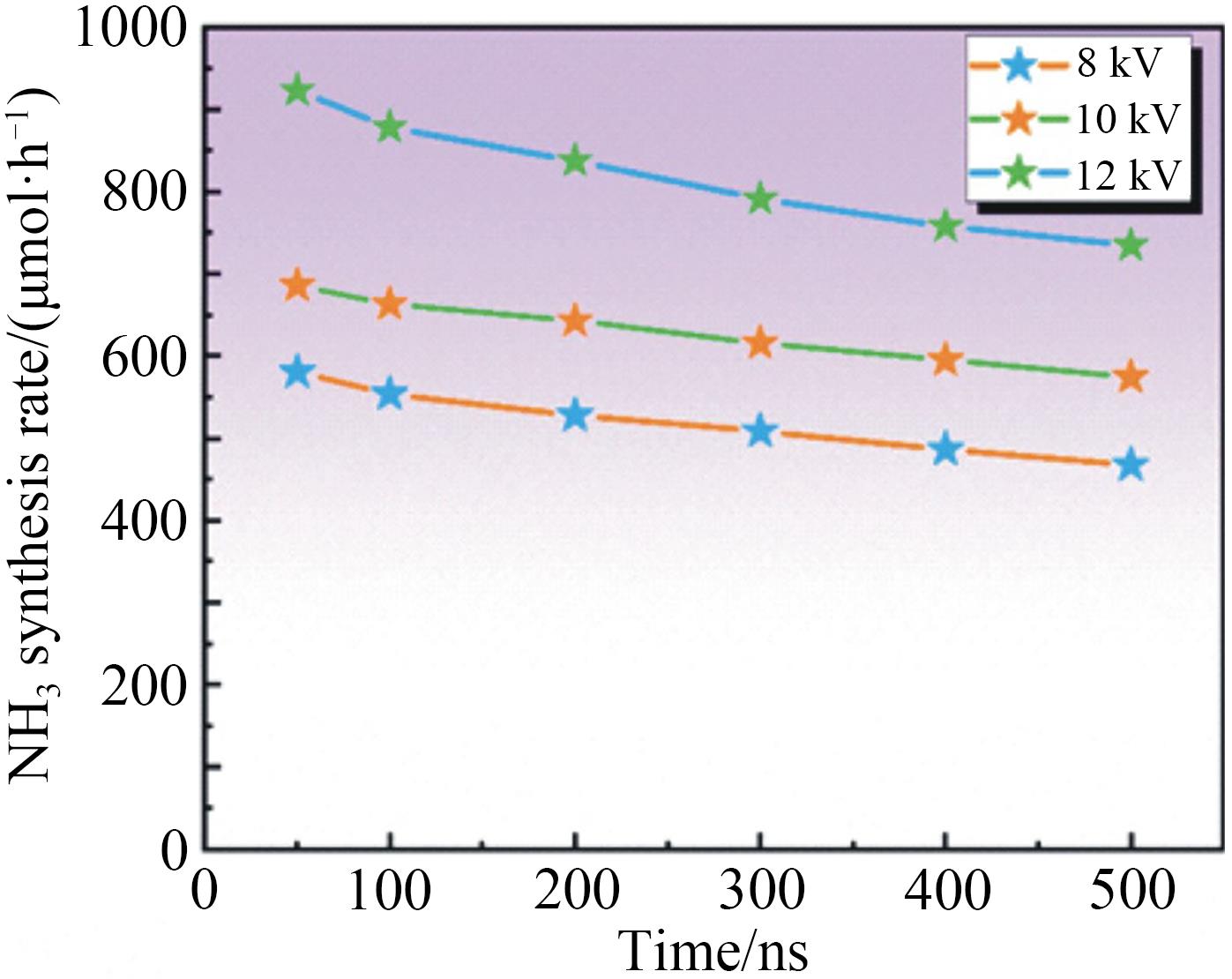
应用化学 ›› 2023, Vol. 40 ›› Issue (2): 268-276.DOI: 10.19894/j.issn.1000-0518.220204
纳秒脉冲介质阻挡放电等离子体合成氨工艺条件优化
- 北京印刷学院等离子体物理与材料实验室,北京 102600
-
收稿日期:2022-06-08接受日期:2022-10-12出版日期:2023-02-01发布日期:2023-02-27 -
通讯作者:张海宝 -
基金资助:国家自然科学基金(11875090);北京市自然科学基金(1192008);北京市教委项目(KM202010015003)
Optimization of Process Parameters for Ammonia Synthesis by Nanosecond Pulsed Dielectric Barrier Discharge Plasma
Yang LIU, Hai-Bao ZHANG( ), Qiang CHEN
), Qiang CHEN
- Laboratory of Plasma Physics and Materials,Beijing Institute of Graphic Communication,Beijing 102600,China
-
Received:2022-06-08Accepted:2022-10-12Published:2023-02-01Online:2023-02-27 -
Contact:Hai-Bao ZHANG -
About author:hbzhang@bigc.edu.cn
-
Supported by:the National Natural Science Foundation of China(11875090);Beijing Municipal National Science Foundation(1192008);Beijing Municipal Education Commission Project(KM202010015003)
摘要:
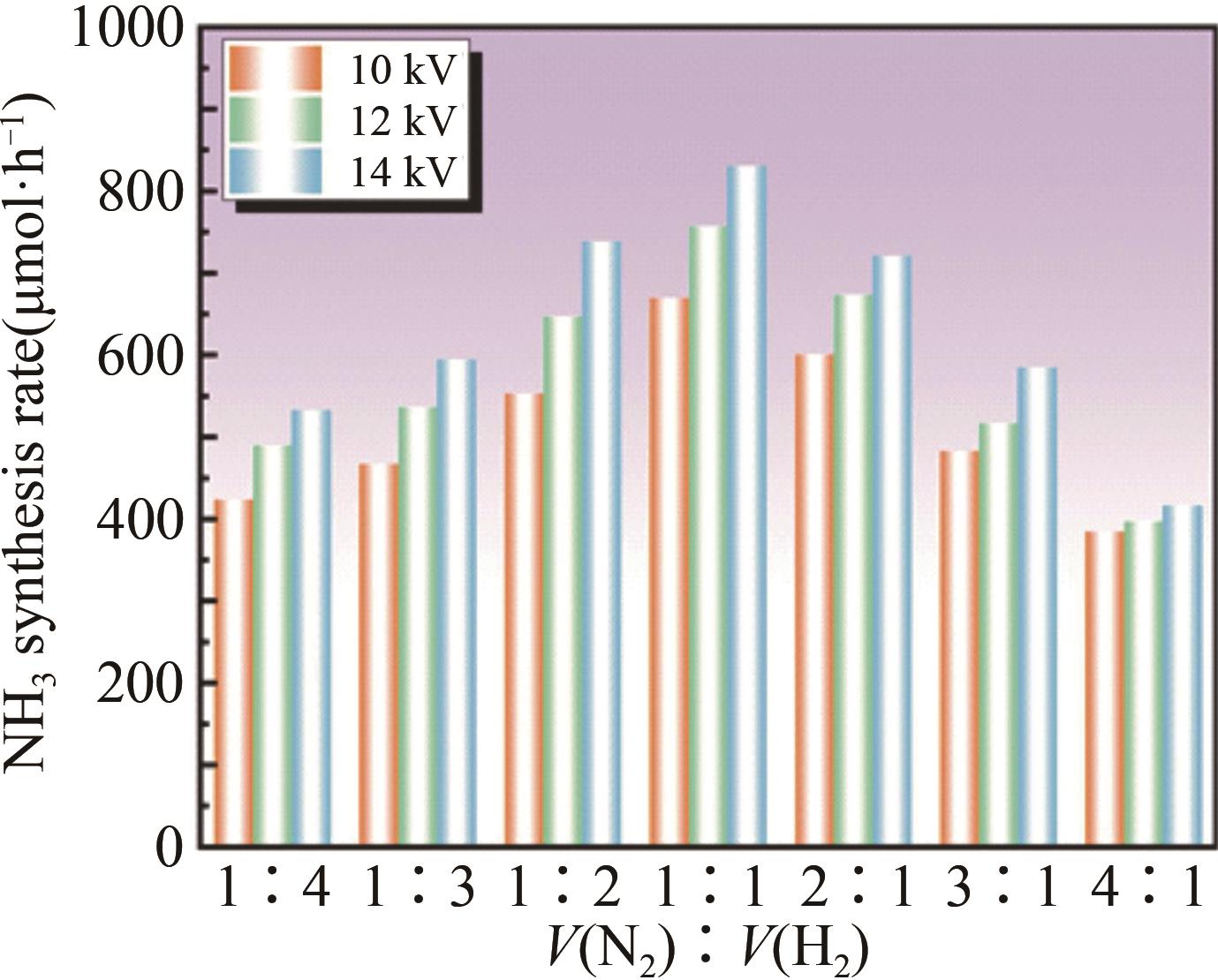
氨(NH3)作为重要的化工原料对农业及国计民生发展有直接影响。工业合成氨需高温高压、能耗高和污染重。低温等离子体技术是一种可持续,有潜力的合成氨途径,已成为国内外研究热点。本工作以氮气和氢气为原料,在低温常压下采用纳秒脉冲介质阻挡放电等离子体合成氨,通过单因素实验系统研究脉冲峰值电压、脉冲重复频率、气体总流量、N2和H2体积比(V(N2)∶V(H2))等因素对合成氨速率及能量产率的影响规律。进一步通过正交实验评价确定影响合成氨反应速率因素的主次顺序为:脉冲峰值电压>脉冲重复频率>气体体积比>气体总流量。影响合成氨能量产率因素的主次顺序为:脉冲峰值电压>气体体积比>脉冲重复频率>气体总流量。结合两部分实验,最终得到合成氨的优选条件:脉冲峰值电压16 kV、脉冲重复频率6 kHz、脉冲上升沿100 ns、V(N2)∶V(H2)=1∶1、气体总流量200 mL/min。此时NH3合成速率最高为923.08 μmol/h,能量产率为0.30 g/kWh。
中图分类号:
引用本文
刘洋, 张海宝, 陈强. 纳秒脉冲介质阻挡放电等离子体合成氨工艺条件优化[J]. 应用化学, 2023, 40(2): 268-276.
Yang LIU, Hai-Bao ZHANG, Qiang CHEN. Optimization of Process Parameters for Ammonia Synthesis by Nanosecond Pulsed Dielectric Barrier Discharge Plasma[J]. Chinese Journal of Applied Chemistry, 2023, 40(2): 268-276.
| Horizontal factor | A | B | C | D |
|---|---|---|---|---|
| Peak voltage/ kV | Power frequency/ kHz | Total gas flow/ (mL·min-1) | V(N2)∶V(H2) | |
| 1 | 8 | 2 | 200 | 1∶3 |
| 2 | 10 | 3 | 180 | 1∶2 |
| 3 | 12 | 4 | 160 | 1∶1 |
| 4 | 14 | 5 | 140 | 2∶1 |
| 5 | 16 | 6 | 120 | 3∶1 |
表1 正交实验因素和水平表
Table 1 Levels of the factors used in the orthogonal experiment
| Horizontal factor | A | B | C | D |
|---|---|---|---|---|
| Peak voltage/ kV | Power frequency/ kHz | Total gas flow/ (mL·min-1) | V(N2)∶V(H2) | |
| 1 | 8 | 2 | 200 | 1∶3 |
| 2 | 10 | 3 | 180 | 1∶2 |
| 3 | 12 | 4 | 160 | 1∶1 |
| 4 | 14 | 5 | 140 | 2∶1 |
| 5 | 16 | 6 | 120 | 3∶1 |
| Experiment number | A | B | C | D | Ammonia synthesis rate/ (μmol·h-1) | Energy yield/ (g?kWh-1) |
|---|---|---|---|---|---|---|
| Peak voltage/kV | Power frequency/kHz | Total gas flow/(mL·min-1) | V(N2)∶V(H2) | |||
| 1 | 8 | 2 | 200 | 1∶3 | 135.75 | 0.28 |
| 2 | 8 | 3 | 160 | 2∶1 | 107.14 | 0.30 |
| 3 | 8 | 4 | 120 | 1∶2 | 190.48 | 0.36 |
| 4 | 8 | 5 | 180 | 3∶1 | 239.20 | 0.30 |
| 5 | 8 | 6 | 140 | 1∶1 | 350.88 | 0.28 |
| 6 | 10 | 2 | 120 | 2∶1 | 180.45 | 0.32 |
| 7 | 10 | 3 | 180 | 1∶2 | 272.73 | 0.23 |
| 8 | 10 | 4 | 140 | 3∶1 | 194.81 | 0.20 |
| 9 | 10 | 5 | 200 | 1∶1 | 483.22 | 0.30 |
| 10 | 10 | 6 | 160 | 1∶3 | 441.72 | 0.26 |
| 11 | 12 | 2 | 140 | 1∶2 | 221.13 | 0.26 |
| 12 | 12 | 3 | 200 | 3∶1 | 218.45 | 0.20 |
| 13 | 12 | 4 | 160 | 1∶1 | 442.26 | 0.29 |
| 14 | 12 | 5 | 120 | 1∶3 | 413.79 | 0.25 |
| 15 | 12 | 6 | 180 | 2∶1 | 545.45 | 0.26 |
| 16 | 14 | 2 | 160 | 3∶1 | 244.90 | 0.29 |
| 17 | 14 | 3 | 120 | 1∶1 | 371.13 | 0.38 |
| 18 | 14 | 4 | 180 | 1∶3 | 413.79 | 0.18 |
| 19 | 14 | 5 | 140 | 2∶1 | 383.80 | 0.13 |
| 20 | 14 | 6 | 200 | 1∶2 | 666.67 | 0.27 |
| 21 | 16 | 2 | 180 | 1∶1 | 423.53 | 0.26 |
| 22 | 16 | 3 | 140 | 1∶3 | 365.48 | 0.20 |
| 23 | 16 | 4 | 200 | 2∶1 | 456.85 | 0.20 |
| 24 | 16 | 5 | 160 | 1∶2 | 692.31 | 0.24 |
| 25 | 16 | 6 | 120 | 3∶1 | 264.71 | 0.09 |
| NH3 synthesis rate/(μmol·h-1) | ||||||
| k1 | 204.69 | 241.15 | 392.19 | 354.11 | ||
| k2 | 314.59 | 266.99 | 378.94 | 408.70 | ||
| k3 | 368.22 | 339.64 | 364.24 | 414.20 | ||
| k4 | 416.06 | 442.46 | 303.22 | 334.74 | ||
| k5 | 440.58 | 453.89 | 246.02 | 232.41 | ||
| Range R1 | 235.89 | 212.74 | 146.17 | 181.79 | ||
| Order of factors | A>B>D>C | |||||
| Energy yield/(g?kWh-1) | ||||||
| k1 | 0.30 | 0.28 | 0.25 | 0.23 | ||
| k2 | 0.26 | 0.26 | 0.25 | 0.27 | ||
| k3 | 0.25 | 0.25 | 0.22 | 0.30 | ||
| k4 | 0.25 | 0.24 | 0.21 | 0.24 | ||
| k5 | 0.20 | 0.23 | 0.21 | 0.22 | ||
| Range R2 | 0.10 | 0.05 | 0.04 | 0.08 | ||
| Order of factors | A>D>B>C | |||||
表2 纳秒脉冲介质阻挡放电等离子体合成氨正交实验结果和极差分析
Table 2 Orthogonal experimental results and rang analysis of NH3 synthesis by nanosecond pulsed dielectric barrier discharge plasma
| Experiment number | A | B | C | D | Ammonia synthesis rate/ (μmol·h-1) | Energy yield/ (g?kWh-1) |
|---|---|---|---|---|---|---|
| Peak voltage/kV | Power frequency/kHz | Total gas flow/(mL·min-1) | V(N2)∶V(H2) | |||
| 1 | 8 | 2 | 200 | 1∶3 | 135.75 | 0.28 |
| 2 | 8 | 3 | 160 | 2∶1 | 107.14 | 0.30 |
| 3 | 8 | 4 | 120 | 1∶2 | 190.48 | 0.36 |
| 4 | 8 | 5 | 180 | 3∶1 | 239.20 | 0.30 |
| 5 | 8 | 6 | 140 | 1∶1 | 350.88 | 0.28 |
| 6 | 10 | 2 | 120 | 2∶1 | 180.45 | 0.32 |
| 7 | 10 | 3 | 180 | 1∶2 | 272.73 | 0.23 |
| 8 | 10 | 4 | 140 | 3∶1 | 194.81 | 0.20 |
| 9 | 10 | 5 | 200 | 1∶1 | 483.22 | 0.30 |
| 10 | 10 | 6 | 160 | 1∶3 | 441.72 | 0.26 |
| 11 | 12 | 2 | 140 | 1∶2 | 221.13 | 0.26 |
| 12 | 12 | 3 | 200 | 3∶1 | 218.45 | 0.20 |
| 13 | 12 | 4 | 160 | 1∶1 | 442.26 | 0.29 |
| 14 | 12 | 5 | 120 | 1∶3 | 413.79 | 0.25 |
| 15 | 12 | 6 | 180 | 2∶1 | 545.45 | 0.26 |
| 16 | 14 | 2 | 160 | 3∶1 | 244.90 | 0.29 |
| 17 | 14 | 3 | 120 | 1∶1 | 371.13 | 0.38 |
| 18 | 14 | 4 | 180 | 1∶3 | 413.79 | 0.18 |
| 19 | 14 | 5 | 140 | 2∶1 | 383.80 | 0.13 |
| 20 | 14 | 6 | 200 | 1∶2 | 666.67 | 0.27 |
| 21 | 16 | 2 | 180 | 1∶1 | 423.53 | 0.26 |
| 22 | 16 | 3 | 140 | 1∶3 | 365.48 | 0.20 |
| 23 | 16 | 4 | 200 | 2∶1 | 456.85 | 0.20 |
| 24 | 16 | 5 | 160 | 1∶2 | 692.31 | 0.24 |
| 25 | 16 | 6 | 120 | 3∶1 | 264.71 | 0.09 |
| NH3 synthesis rate/(μmol·h-1) | ||||||
| k1 | 204.69 | 241.15 | 392.19 | 354.11 | ||
| k2 | 314.59 | 266.99 | 378.94 | 408.70 | ||
| k3 | 368.22 | 339.64 | 364.24 | 414.20 | ||
| k4 | 416.06 | 442.46 | 303.22 | 334.74 | ||
| k5 | 440.58 | 453.89 | 246.02 | 232.41 | ||
| Range R1 | 235.89 | 212.74 | 146.17 | 181.79 | ||
| Order of factors | A>B>D>C | |||||
| Energy yield/(g?kWh-1) | ||||||
| k1 | 0.30 | 0.28 | 0.25 | 0.23 | ||
| k2 | 0.26 | 0.26 | 0.25 | 0.27 | ||
| k3 | 0.25 | 0.25 | 0.22 | 0.30 | ||
| k4 | 0.25 | 0.24 | 0.21 | 0.24 | ||
| k5 | 0.20 | 0.23 | 0.21 | 0.22 | ||
| Range R2 | 0.10 | 0.05 | 0.04 | 0.08 | ||
| Order of factors | A>D>B>C | |||||
| 1 | GALLOWAY J N, TOWNSEND A R, ERISMAN J W, et al. Transformation of the nitrogen cycle: recent trends, questions, and potential solutions[J]. Science, 2008, 320(5878): 889-892. |
| 2 | LICHT S, CUI B, WANG B, et al. Ammonia synthesis by N2 and steam electrolysis in molten hydroxide suspensions of nanoscale Fe2O3[J]. Science, 2015, 45(43): 637-640. |
| 3 | 刘化章. 合成氨工业: 过去、现在和未来——合成氨工业创立100周年回顾、启迪和挑战[J]. 化工进展, 2013, 32(9):1995-2005. |
| LIU H Z. Synthetic ammonia industry: past, present and future—review, enlightenment and challenges of the 100th anniversary of synthetic ammonia industry[J]. Prog Chem Ind, 2013, 32(9): 1995-2005. | |
| 4 | CHEN C, ARYAFAR H, LOVEGROVE K M, et al. Modeling of ammonia synthesis to produce supercritical steam for solar thermochemical energy storage[J]. Sol Energy, 2017, 155: 363-371. |
| 5 | VALERA-MEDINA A, XIAO H, OWEN-JONES M, et al. Ammonia for power[J]. Prog Energ Combus, 2018, 69: 63-102. |
| 6 | YAPICIOGLU A, DINCER I. A review on clean ammonia as a potential fuel for power generators[J]. Renew Sust Energ Rev, 2019, 103: 96-108. |
| 7 | WANG W, PATIL B, HEIJKERS S, et al. Nitrogen fixation by gliding arc plasma: better insight by chemical kinetics modelling[J]. Chem Sus Chem, 2017, 10(10): 2145-2157. |
| 8 | ROBERT S. Catalytic synthesis of ammonia—a “never-ending story”[J]. Angew Chem Int Ed, 2003, 42: 2004-2008. |
| 9 | 韩慧. 高气压非平衡等离子体化学合成氨的研究[D]. 大连: 大连海事大学, 2002. |
| HAN H. Research on chemical synthesis of ammonia by high pressure non-equilibrium plasma[D]. Dalian: Dalian Maritime University, 2002. | |
| 10 | 诸葛绍渊. 常压介质阻挡放电等离子体协同催化合成氨应用基础研究[D]. 杭州: 浙江工业大学, 2015. |
| ZHUGE S Y. Basic research on the application of atmospheric pressure dielectric barrier discharge plasma synergistic catalytic synthesis of ammonia[D]. Hongzhou: Zhejiang University of Technology, 2015. | |
| 11 | TANABE Y, NISHIBAYASHI Y. Developing more sustainable processes for ammonia synthesis[J]. Coord Chem Rev, 2013, 257(17/18): 2551-2564. |
| 12 | WANG Y L, CRAVEN M, YU X T, et al. Plasma-enhanced catalytic synthesis of ammonia over a Ni/Al2O3 catalyst at near-room temperature: insights into the importance of the catalyst surface on the reaction mechanism[J]. ACS Catal, 2019, 9: 10780. |
| 13 | SAADATJOU N, JAFARI A, SAHEBDELFAR S. Ruthenium nano-catalysts for ammonia synthesis: a review[J]. Chem Eng Commun, 2015, 202(4): 420-448. |
| 14 | MARNELLOS G, STOUKIDES M. Ammonia synthesis at atmospheric pressure[J]. Science, 1998, 282(5386): 98-100. |
| 15 | KITANO M, INOUE Y, YAMAZAKI Y, et al. Ammonia synthesis using a stable electride as an electron donor and reversible hydrogen store[J]. Nat Chem, 2012, 4(11): 934-940. |
| 16 | MEHTA P, BARBOUN P, HERRERA F A, et al. Overcoming ammonia synthesis scaling relations with plasma-enabled catalysis[J]. Nat Chem, 2018, 1(4): 269-275. |
| 17 | HONG J, PRAWER S, MURPHY A B. Plasma catalysis as an alternative route for ammonia production: status, mechanisms, and prospects for progress[J]. ACS Sustain Chem Eng, 2018, 6, 15-31. |
| 18 | 郑硕. 介质阻挡放电等离子体协同催化合成氨的实验研究[D]. 北京: 北京交通大学, 2019. |
| ZHENG S. Experimental study on synergistic catalytic synthesis of ammonia by dielectric barrier discharge plasma[D]. Beijing: Beijing Jiaotong University, 2019. | |
| 19 | PATIL B S, CHERKASOV N, LANG J, et al. Low temperature plasma-catalytic NOx synthesis in a packed DBD reactor: effect of support materials and supported active metal oxides[J]. Appl Catal B: Environ, 2016, 194: 123-133. |
| 20 | BAI M, ZHANG Z, BAI X, et al. Plasma synthesis of ammonia with a microgap dielectric barrier discharge at ambient pressure[J]. IEEE T Plasma Sci, 2003, 31(6): 1285-1291. |
| 21 | SHAH J, WU T, LUCERO J, et al. Non-thermal plasma synthesis of ammonia over Ni-MOF-74[J]. ACS Sustain Chem Eng, 2019, 7: 377-383. |
| 22 | JAVISHK S, WANG W, ANNEMIE B, et al. Ammonia synthesis by radio frequency plasma catalysis: revealing the underlying mechanisms[J]. ACS Appl Energy Mater, 2018, 1: 4824-4839. |
| 23 | UYAMA H, NAKAMURA T, TANAKA S, et al. Catalytic effect of iron wires on the syntheses of ammonia and hydrazine in a radio-frequency discharge[J]. Plasma Chem Plasma P, 1993, 13(1): 117-131. |
| 24 | YIN K S, VENUGOPALAN M. Plasma chemical synthesis. I. effect of electrode material on the synthesis of ammonia[J]. Plasma Chem Plasma P, 1983, 3(3): 343-350. |
| 25 | SUGIYAMA K, AKAZAWA K, OSHIMA M, et al. Ammonia synthesis by means of plasma over MgO catalyst[J]. Plasma Chem Plasma P, 1986, 6(2): 179-193. |
| 26 | UYAMA H, MATSUMOT O. Synthesis of ammonia in high-frequency discharges. II. synthesis of ammonia in a microwave discharge under various conditions[J]. Plasma Chem Plasma P, 1989, 9(3): 13-24. |
| 27 | NAKAJIMA J, SEKIGUCHI H. Synthesis of ammonia using microwave discharge at atmospheric pressure[J]. Thin Solid Films, 2008, 516(13): 4446-4451. |
| 28 | 陈杰, 梁华, 魏彪, 等. 参数化纳秒脉冲电源激励下表面介质阻挡放电特性[J]. 高电压技术, 2019, 45(10): 3365-3374. |
| CHEN J, LIANG H, WEI B, et al. Discharge characteristics of surface dielectric barrier discharge driven by parameterized nanosecond pulsed power supply[J]. High Voltage Eng, 2019, 45(10): 3365-3374. | |
| 29 | 康少芬, 张帅, 陈晓晓, 等. 纳秒脉冲介质阻挡放电等离子体氮还原合成氨的研究[J]. 高电压技术, 2021, 47(1): 368-375. |
| KANG S F, ZHANG S, CHEN X X, et al. Research on nitrogen reduction of synthetic ammonia by nanosecond pulsed dielectric barrier discharge plasma[J]. High Voltage Eng, 2021, 47(1): 368-375. | |
| 30 | LITTLE J, TAKASHIMA K, NISHIHARA M, et al. Separation control with nanosecond-pulse-driven dielectric barrier discharge plasma actuators[J]. AIAA J, 2012, 50(2): 350-365. |
| 31 | MIZUSHIMA T, MATSUMOTO K, OHKITA H, et al. Catalytic effects of metal-loaded membrane-like alumina tubes on ammonia synthesis in atmospheric pressure plasma by dielectric barrier discharge[J]. Plasma Chem Plasma P, 2007, 27(1): 1-11. |
| 32 | PATIL B S, KAATHOVEN A S R V, PEETERS F J J, et al. Deciphering the synergy between plasma and catalyst support for ammonia synthesis in a packed dielectric barrier discharge reactor[J]. J Phys D Appl Phys, 2020, 53(14): 144003. |
| 33 | XIE Q, ZHUGE S, SONG X, et al. Non-thermal atmospheric plasma synthesis of ammonia in a DBD reactor packed with various catalysts[J]. J Phys D Appl Phys, 2020, 53(6): 064002. |
| 34 | KIM H H, TERAMOTO Y, OGATA A, et al. Atmospheric-pressure nonthermal plasma synthesis of ammonia over ruthenium catalysts[J]. Plasma Process Polym, 2017, 14(6): 1600157-1600179. |
| 35 | PENG P, CHENG Y, HATZENBLLER R, et al. Ru-based multifunctional mesoporous catalyst for low-pressure and non-thermal plasma synthesis of ammonia[J]. Int J Hydrogen Energ, 2017, 42(30): 19056-19066. |
| 36 | WAGNER H E, BRANDENBURG R, KOZLOV K V, et al. The barrier discharge[J]. Vacuum, 2003, 71(3): 417-436. |
| 37 | TU X, GALLON H J, WHITEHEAD J C. Transition behavior of packed-bed dielectric barrier discharge in argon[J]. IEEE T Plasma Sci, 2011, 39(11): 2172-2173. |
| 38 | HONG J, PANCHESHNYI S, TAM E, et al. Corrigendum: kinetic modelling of NH3 production in N2-H2 non-equilibrium atmospheric-pressure plasma catalysis[J]. J Phys D Appl Phys, 2018, 51(10): 1450-1459. |
| 39 | GAO M X, ZHANG Y, WANG H Y, et al. Mode transition of filaments in packed-bed dielectric barrier discharges[J]. Catalysts, 2018, 8(6): 248. |
| 40 | GÓMEZ-RAMÍREZ A, COTRINO J, LAMBERT R M, et al. Efficient synthesis of ammonia from N2 and H2 alone in a ferroelectric packed-bed DBD reactor[J]. Plasma Sources Sci Technol, 2015, 24(6): 065011. |
| 41 | ZHANG C, LIU Z P, HU P. Stepwise addition reactions in ammonia synthesis: a first principles study[J]. J Chem Phys, 2001, 115: 609-611. |
| 42 | LIN B, WEI K, NI J, et al. KOH activation of thermally modified carbon as a support of Ru catalysts for ammonia synthesis[J]. ChemCatChem, 2013, 5(7): 1941-1947. |
| [1] | 曹蓉, 夏杰桢, 廖漫华, 赵路超, 赵晨, 吴琪. 单原子催化剂在电化学合成氨中的理论研究进展[J]. 应用化学, 2023, 40(1): 9-23. |
| [2] | 罗策, 颜燕, 李剑, 刘婷, 白焕焕, 卢凡, 冯婧. 甲烷动态反应-电感耦合等离子体质谱标准加入测定高纯钛中痕量铁[J]. 应用化学, 2021, 38(7): 874-880. |
| [3] | 刘洋, 张海宝, 陈强. 低温等离子体合成氨研究进展[J]. 应用化学, 2021, 38(6): 622-636. |
| [4] | 张春华, 赵晓波, 李跃军, 孙大伟. (BiO)2CO3-Bi-TiO2复合纳米纤维制备及其光催化降解抗生素[J]. 应用化学, 2021, 38(1): 99-106. |
| [5] | 毕一飘, 宫雪, 杨发, 阮明波, 宋平, 徐维林. 多价MnOx/C电催化剂用于高效氮还原反应[J]. 应用化学, 2020, 37(9): 1048-1055. |
| [6] | 李跃军, 曹铁平, 孙大伟, 赵艳辉, 柏本昂. 三元Ho3+-TiO2/Bi等离子体复合纤维制备及其可见光催化产氢性能[J]. 应用化学, 2020, 37(5): 570-578. |
| [7] | 苏风梅, 张达, 梁风. 低温等离子体制备与改性纳米催化材料的研究进展[J]. 应用化学, 2019, 36(8): 882-891. |
| [8] | 申书昌, 彭程, 王荻. 蒙脱土表面键合配位体固相萃取填料的制备及其对重金属离子的吸附性能[J]. 应用化学, 2019, 36(6): 717-725. |
| [9] | 郑义智, 王海水. 银/二氧化硅核壳纳米颗粒的制备及其壳层厚度对其光学性质的影响[J]. 应用化学, 2014, 31(03): 323-327. |
| [10] | 戴燕, 吕春绪, 李斌栋, 谭卫红. 微波辅助氢氧化钾催化合成2-(4-羟基苯基)六氟异丙醇[J]. 应用化学, 2013, 30(12): 1438-1442. |
| [11] | 任杰, 廖瑞瑞, 杨武, 李岩, 高锦章. 辉光放电电解等离子体处理制备铁基表面超疏水材料[J]. 应用化学, 2013, 30(02): 208-213. |
| [12] | 胡爽慧, 吕一军, 闫文娟, 王保伟. 滑动弧电极放电等离子体作用甲烷制C2烃的工艺[J]. 应用化学, 2013, 30(01): 61-66. |
| [13] | 冷飞, 李英, 甄淑君, 李原芳. 基于金纳米粒子的等离子体共振吸收测定阿莫西林[J]. 应用化学, 2012, 29(11): 1329-1334. |
| [14] | 任杰, 陶丽红, 高锦章, 李岩, 刘宏伟, 李亚萍. 辉光放电电解等离子体引发合成聚丙烯酸钠/腐植酸复合高吸水性树脂[J]. 应用化学, 2012, 29(04): 376-382. |
| [15] | 李靖云, 王燚, 许佳丽, 李原芳. 金纳米颗粒等离子体共振吸收光谱检测头孢唑啉[J]. 应用化学, 2012, 29(04): 455-461. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||