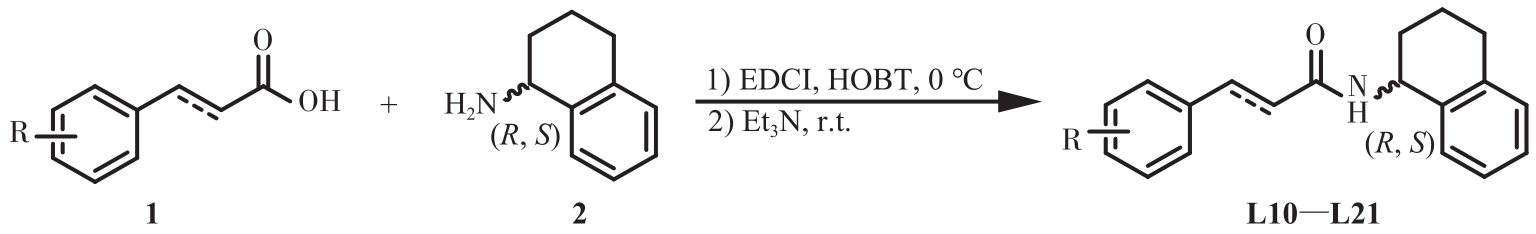应用化学 ›› 2023, Vol. 40 ›› Issue (2): 261-267.DOI: 10.19894/j.issn.1000-0518.220182
肉桂酰胺类化合物的结构优化、合成及其抗肝癌活性
杨弘力1, 王倩琪2, 汪欢1, 颜晗1, 林芃晖2, 宋金秋1, 丁洋1, 李善花2, 李福男1( )
)
- 1.厦门大学药学院,厦门 361102
2.厦门大学医学院,厦门 361102
Structure Optimization, Synthesis and Anti-hepatocarcinoma Activity of Cinnamamide Compounds
Hong-Li YANG1, Qian-Qi WANG2, Huan WANG1, Han YAN1, Peng-Hui LIN2, Jin-Qiu SONG1, Yang DING1, Shan-Hua LI2, Fu-Nan LI1( )
)
- 1.School of Pharmaceutical Sciences,Xiamen University,Xiamen 361102,China
2.School of Medicine,Xiamen University,Xiamen 361102,China
摘要:
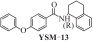
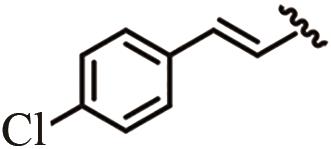
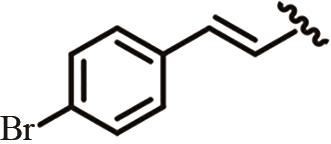
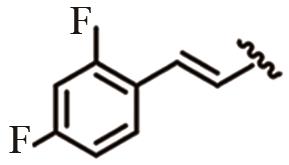
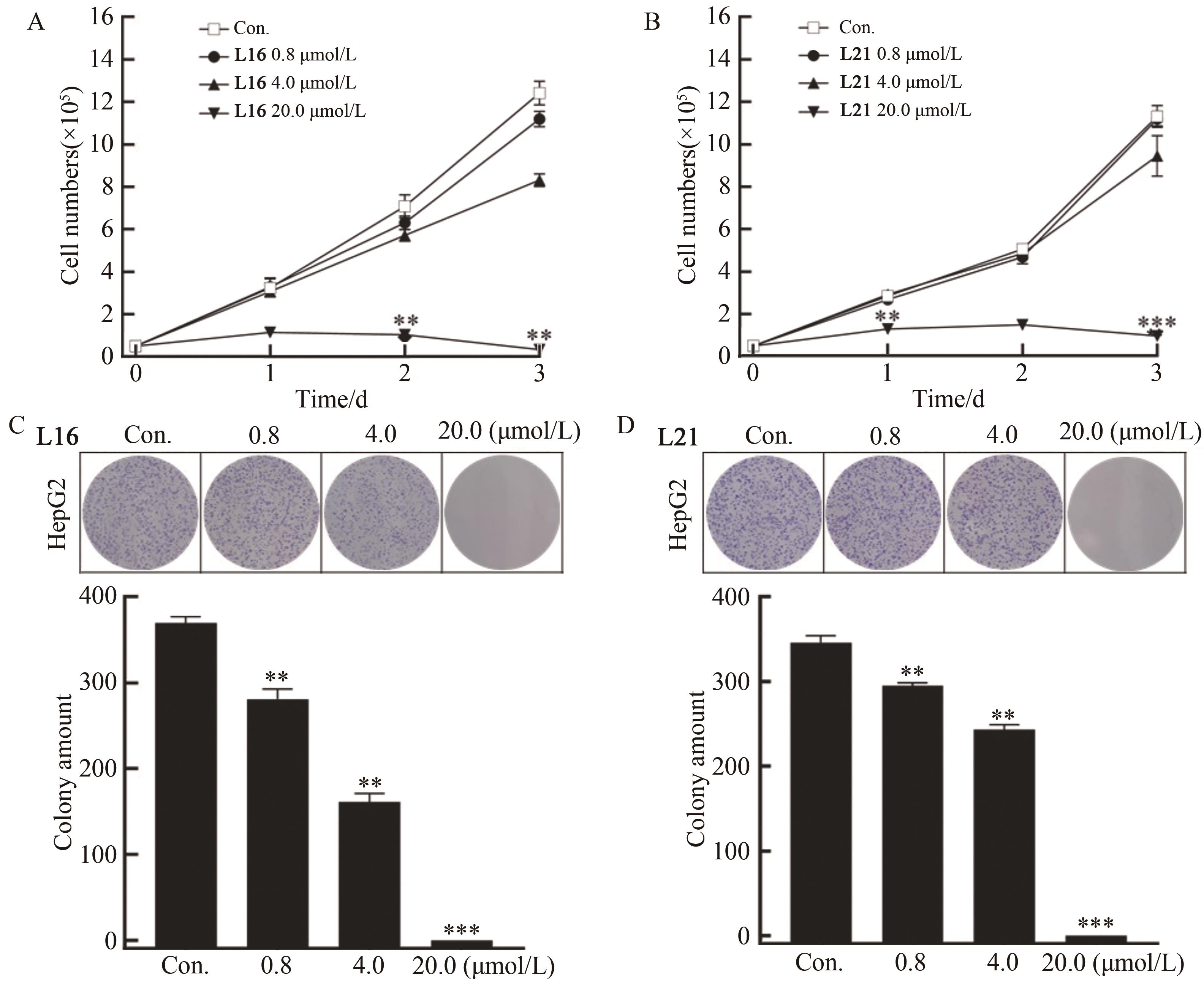
为开发治疗肝癌的新型分子靶向药物,以前期发现的具有抗肝癌活性的手性酰胺类化合物YSM-13为起始化合物,设计合成了12个N-四氢萘肉桂酰胺类化合物L10—L21。目标化合物的结构通过核磁共振氢谱、碳谱和质谱技术进行表征,进一步通过细胞毒性实验、细胞增殖实验和克隆形成实验,评价了目标化合物对人源肝细胞癌细胞系(HepG2)细胞活性的抑制作用。结果表明,(S)-构型的目标化合物抗肝癌活性明显高于(R)-构型,无环外双键的目标化合物几乎没有抗肝癌活性。此外,卤素原子氟、氯、溴对位取代或二氟间位取代目标化合物的苯环,可以使其抗肝癌活性得到明显提升,其中,氯对位取代的目标化合物(S,E)-3-(4-氯苯基)-N-(1,2,3,4-四氢萘-1-基)丙烯酰胺(L16)的抗肝癌活性最优,L16给药组的HepG2细胞生存率仅为对照组的26.36%,细胞增殖实验和克隆形成实验结果进一步表明L16可以显著抑制肝癌细胞的体外恶性增殖能力。综上所述,化合物L16作为一种潜在的抗肝癌小分子药物,有望从生物学机制方面进一步研究其抗肝癌活性。
中图分类号: