
应用化学 ›› 2024, Vol. 41 ›› Issue (3): 365-376.DOI: 10.19894/j.issn.1000-0518.230262
• 综合评述 • 上一篇
质子交换膜电解池阳极钛基气体扩散层研究进展
顾婷婷1, 张科1, 张心周1, 刘阳1, 孙伟才1, 谭爱东2, 刘建国2( )
)
- 1.(中钛国创(青岛)科技有限公司,青岛 266111 )
2.华北电力大学,北京 100054
-
收稿日期:2023-08-30接受日期:2023-12-08出版日期:2024-03-01发布日期:2024-04-09 -
通讯作者:刘建国 -
基金资助:国家重点研发计划项目(2021YFB4000100)
Research Progress on Anodic Titanium‑Based Gas Diffusion Layer in Proton Exchange Membrane Electrolysis Cell
Ting-Ting GU1, Ke ZHANG1, Xin-Zhou ZHANG1, Yang LIU1, Wei-Cai SUN1, Ai-Dong TAN2, Jian-Guo LIU2( )
)
- 1.(China Titanium Guochuang (Qingdao) Technology Co. ,Ltd. ,Qingdao 266111,China )
2.North China Electric Power University,Beijing 100054,China
-
Received:2023-08-30Accepted:2023-12-08Published:2024-03-01Online:2024-04-09 -
Contact:Jian-Guo LIU -
About author:jianguoliu@ncepu.edu.cn
-
Supported by:the National Key Research and Development Program(2021YFB4000100)
摘要:
气体扩散层在质子交换膜(PEM)水电解池中有着支撑膜组件、供给反应水、移除气体产物以及降低欧姆电阻的重要作用。 PEM水电解池阳极区具有酸性、富氧且高电位的工作环境,对阳极区的气体扩散层具有严苛的要求。 气体扩散层结构特性、导电性与耐腐蚀性是决定其电化学性能的关键。 本文总结了可用于PEM电解池阳极气体扩散层的材料,简述了其结构特性对PEM电解池电化学性能的影响,分析了各种镀层材料在提高气体扩散层的导电性、耐腐蚀性以及电解池阳极氧析出反应(OER)性能方面的作用。 最后,展望了气体扩散层在降低成本和提高电解池性能方面的研究趋势。
中图分类号:
引用本文
顾婷婷, 张科, 张心周, 刘阳, 孙伟才, 谭爱东, 刘建国. 质子交换膜电解池阳极钛基气体扩散层研究进展[J]. 应用化学, 2024, 41(3): 365-376.
Ting-Ting GU, Ke ZHANG, Xin-Zhou ZHANG, Yang LIU, Wei-Cai SUN, Ai-Dong TAN, Jian-Guo LIU. Research Progress on Anodic Titanium‑Based Gas Diffusion Layer in Proton Exchange Membrane Electrolysis Cell[J]. Chinese Journal of Applied Chemistry, 2024, 41(3): 365-376.

图1 PEM电解池的工作原理图和阳极的气、液、固三相界面反应区示意图
Fig.1 Principle diagram of PEM electrolytic cell and schematic diagram of the interface between gas diffusion layer and membrane electrode

图2 (a)多孔钛粉烧结毡、(b)钛纤维烧结毡、(c)多孔钛箔和(d)泡沫钛的结构示意图
Fig.2 Schematic diagram of (a) porous titanium powder sintered felt, (b) titanium fiber sintered felt, (c) porous titanium foil and (d) metal foam
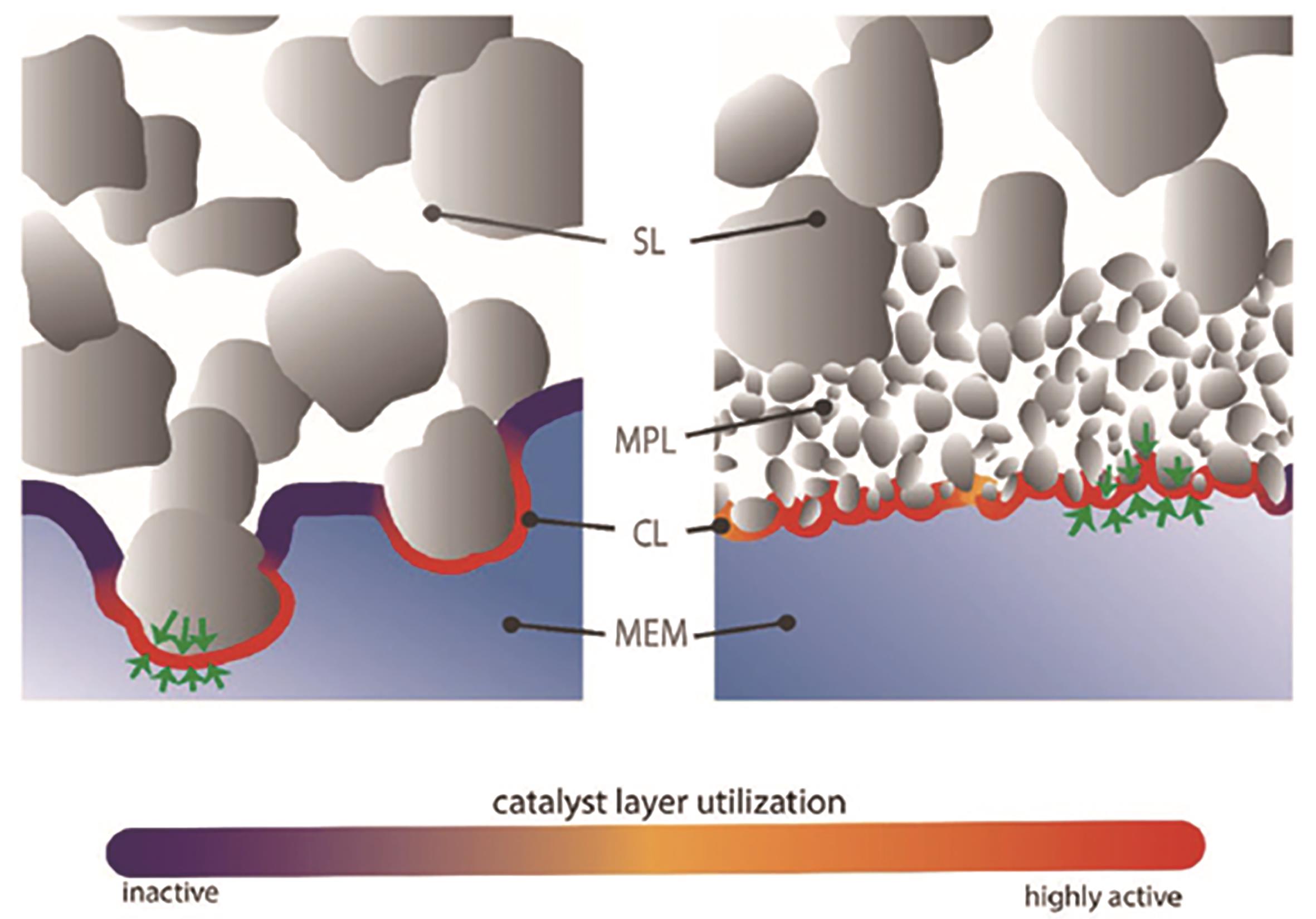
图3 气体扩散层与膜电极界面示意图[34]SL: support layer; MPL: mesoporous layer; CL: catalytic layer; MEM: mass exchange membrane
Fig.3 Schematic diagram of the interface between gas diffusion layer and membrane electrode[34]
| Type | Raw material | Raw material diameter/μm | Porosity ratio/% | Pore diameter/μm | Thickness/μm | Ref. |
|---|---|---|---|---|---|---|
Titanium powder sintered felt | Ordinary titanium powder | / | 10~33 | / | 253~526 | [ |
| 75~100 | 35~40 | 11~25 | 1 200~1 400 | [ | ||
| 380~520 | / | 100 | 1 000 | |||
| 100~200 | 34.3 | 16 | 1 300 | [ | ||
| 330~780 | 32.7 | 60 | 1 200 | |||
| 20 | 50~70 | 10~35 | 200/300 | [ | ||
| Hydrogenated dehydrogenation titanium powder | / | 10~55 | / | 246~515 | [ | |
| / | 54 | / | 513 | [ | ||
| 40 | 28 | 8 | 800 | [ | ||
| Titanium fiber sintered felt | Titanium fiber | / | 53 | / | 1 000 | [ |
| 20 | 75 | 50 | 300 | [ | ||
| 80 | 75 | 180 | 300 | |||
| 20 | 50 | 28 | 300 | |||
| / | 75 | / | 300 | [ | ||
| 20 | 50 | 12.7 | 350/500/1 000 | [ | ||
| Porous titanium foil | Titanium foil | / | / | 1~25 | 50.8 | [ |
表1 各种多孔钛材料的结构参数
Table 1 Structural parameters of various porous titanium materials
| Type | Raw material | Raw material diameter/μm | Porosity ratio/% | Pore diameter/μm | Thickness/μm | Ref. |
|---|---|---|---|---|---|---|
Titanium powder sintered felt | Ordinary titanium powder | / | 10~33 | / | 253~526 | [ |
| 75~100 | 35~40 | 11~25 | 1 200~1 400 | [ | ||
| 380~520 | / | 100 | 1 000 | |||
| 100~200 | 34.3 | 16 | 1 300 | [ | ||
| 330~780 | 32.7 | 60 | 1 200 | |||
| 20 | 50~70 | 10~35 | 200/300 | [ | ||
| Hydrogenated dehydrogenation titanium powder | / | 10~55 | / | 246~515 | [ | |
| / | 54 | / | 513 | [ | ||
| 40 | 28 | 8 | 800 | [ | ||
| Titanium fiber sintered felt | Titanium fiber | / | 53 | / | 1 000 | [ |
| 20 | 75 | 50 | 300 | [ | ||
| 80 | 75 | 180 | 300 | |||
| 20 | 50 | 28 | 300 | |||
| / | 75 | / | 300 | [ | ||
| 20 | 50 | 12.7 | 350/500/1 000 | [ | ||
| Porous titanium foil | Titanium foil | / | / | 1~25 | 50.8 | [ |
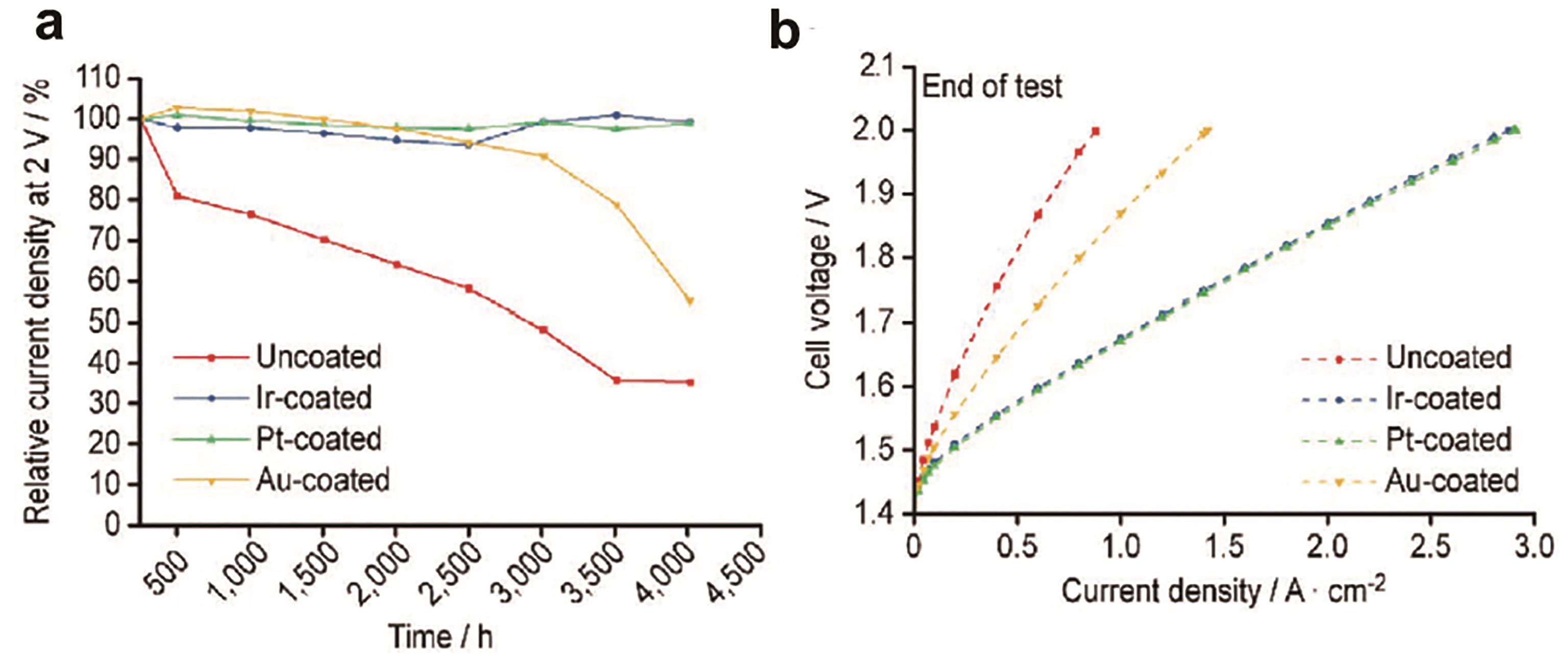
图4 稳定性测试和极化测试结果: (a) Au、Ir和Pt作为镀层材料和未镀层钛毡运行4000 h的电压变化情况; (b) Au、Ir和Pt作为镀层材料的钛毡运行4000 h后的极化曲线[43]
Fig.4 The results of stability test and polarization test: (a) The voltage change of Au, Ir and Pt as coating materials and uncoated titanium felt after 4000 h operation; (b) The polarization curve of titanium felt with Au, Ir and Pt as coating material after 4000 h operation[43]

图5 接触电阻和极化曲线测试结果图: (a)未涂层的气体传输层和涂层Ir质量为0.005、0.013、0.025和0.05 mg/cm2的气体传输层的接触电阻; (b)未涂层的气体传输层和涂层Ir质量为0.005、0.013、0.025和0.05 mg/cm2的气体传输层的极化曲线[47]
Fig.5 Test results of contact resistance and polarization curves: (a) Contact resistance of uncoated gas transport layer and coated gas transport layer with Ir mass of 0.005, 0.013, 0.025 and 0.025 mg/cm2; (b) Polarization curve of uncoated gas transport layer and coated gas transport layer with Ir mass of 0.005, 0.013, 0.025 and 0.05 mg/cm2[47]
| Coating | Contact resistance | Load capacity | Electrolytic performance | Stability | Ref. |
|---|---|---|---|---|---|
| IrO2 | / | 1.0 mg/cm2 | 0.36 A/cm2@1.60 V | 53 h@1.52 A/cm2 | [ |
| / | 0.60 mg/cm2 | 2.72 A/cm2@2.0 V | / | [ | |
| Ir | 11.5 mΩ·cm2@0.5 MPa | 0.10 mg/cm2 | 2.0 A/ cm2@1.86 V | / | [ |
| 4.0 mΩ·cm2@3 MPa | 0.10 mg/cm2 | 2.38 A/cm2@1.90 V | 4 000 h@2.0 V | [ | |
| 3.1 mΩ·cm2@3 MPa | 0.025 mg/cm2 | 1.0 A/cm2@1.70 V | / | [ | |
| Pt | 6.0 mΩ·cm2@3 MPa | 0.16 mg/cm2 | 2.4 A/cm2@1.90 V | 4 000 h@2.0 V | [ |
| / | 200 nm (thickness) | 2.82 A/cm2@2.0 V | 1 100 h@2.0 A/cm2 | [ | |
| Au | 2.0 mΩ·cm2@3 MPa | 0.18 mg/cm2 | 2.35 A/cm2@1.90 V | 2 000 h@2.0 V | [ |
| / | 180 nm (thickness) | 2.0 A/cm2 @1.63 V | 100 h@0.2 A/cm2 | [22] | |
| Ir0.7Ru0.3O2 | 6.5 mΩ·cm2@2 MPa | 1.0 mg/cm2 | 1.84 V@2.0 A/cm2 | / | [ |
| IrO2?RuO2?TaO x | / | 1.0 mg (Ir+Ru)/cm2 | 1.84 V@2.0 A/cm2 | / | [ |
| STN?RuTi | / | STN (thickness)99 nm | / | 225 h@0.10A/cm2 | [ |
| NbN_500_60 | / | 1.0 μm (thickness) | / | / | [ |
表2 文献中各种镀层处理气体扩散层电化学性能
Table 2 Electrochemical energy table of gas diffusion layer treated by various coatings in literature
| Coating | Contact resistance | Load capacity | Electrolytic performance | Stability | Ref. |
|---|---|---|---|---|---|
| IrO2 | / | 1.0 mg/cm2 | 0.36 A/cm2@1.60 V | 53 h@1.52 A/cm2 | [ |
| / | 0.60 mg/cm2 | 2.72 A/cm2@2.0 V | / | [ | |
| Ir | 11.5 mΩ·cm2@0.5 MPa | 0.10 mg/cm2 | 2.0 A/ cm2@1.86 V | / | [ |
| 4.0 mΩ·cm2@3 MPa | 0.10 mg/cm2 | 2.38 A/cm2@1.90 V | 4 000 h@2.0 V | [ | |
| 3.1 mΩ·cm2@3 MPa | 0.025 mg/cm2 | 1.0 A/cm2@1.70 V | / | [ | |
| Pt | 6.0 mΩ·cm2@3 MPa | 0.16 mg/cm2 | 2.4 A/cm2@1.90 V | 4 000 h@2.0 V | [ |
| / | 200 nm (thickness) | 2.82 A/cm2@2.0 V | 1 100 h@2.0 A/cm2 | [ | |
| Au | 2.0 mΩ·cm2@3 MPa | 0.18 mg/cm2 | 2.35 A/cm2@1.90 V | 2 000 h@2.0 V | [ |
| / | 180 nm (thickness) | 2.0 A/cm2 @1.63 V | 100 h@0.2 A/cm2 | [22] | |
| Ir0.7Ru0.3O2 | 6.5 mΩ·cm2@2 MPa | 1.0 mg/cm2 | 1.84 V@2.0 A/cm2 | / | [ |
| IrO2?RuO2?TaO x | / | 1.0 mg (Ir+Ru)/cm2 | 1.84 V@2.0 A/cm2 | / | [ |
| STN?RuTi | / | STN (thickness)99 nm | / | 225 h@0.10A/cm2 | [ |
| NbN_500_60 | / | 1.0 μm (thickness) | / | / | [ |
| Coating material | Electrical conductivity | Corrosion resistance | OER catalytic performance |
|---|---|---|---|
| Ir/IrO2 | ++ | +++ | +++ |
| Pt | ++ | +++ | + |
| Au | +++ | + | - |
| RuO2 | - | - | +++ |
| Other metallic materials | + | + | - |
表3 各种涂层材料性能对比
Table 3 Comparison table of properties of various coating materials
| Coating material | Electrical conductivity | Corrosion resistance | OER catalytic performance |
|---|---|---|---|
| Ir/IrO2 | ++ | +++ | +++ |
| Pt | ++ | +++ | + |
| Au | +++ | + | - |
| RuO2 | - | - | +++ |
| Other metallic materials | + | + | - |
| 1 | NOYAN O F, HASAN M M, PALA N. A global review of the hydrogen energy ECO-system[J]. Energies, 2023, 16(3): 1462-1484. |
| 2 | MAIER M, SMITH K, DODWELL J, et al. Mass transport in PEM water electrolysers: a review[J]. Int J Hydrogen Energy, 2022, 47(1): 30-56. |
| 3 | MO J, STEEN S M, RETTERER S T, et al. Mask-patterned wet etching of thin titanium liquid gas diffusion layers for a PEMEC[J]. ECS Transact, 2015, 66(24): 3-10. |
| 4 | LEE J K, LAU G Y, SABHARWAL M, et al. Titanium porous-transport layers for PEM water electrolysis prepared by tape casting[J]. J Power Sources, 2023, 559(1): 232606. |
| 5 | MO J, KANG Z, RETTERER S T, et al. Discovery of true electrochemical reactions for ultrahigh catalyst mass activity in water splitting[J]. Sci Adv, 2016, 2(11): 1600690. |
| 6 | WANG C, WU Y, BODACH A, et al. A novel electrode for value-generating anode reactions in water electrolyzers at industrial current densities[J]. Angew Chem Int Ed Engl, 2023, 62(7): e202215804. |
| 7 | XUE F, SU J, LI P, et al. Application of proton exchange membrane electrolysis of water hydrogen production technology in power plant[J]. IOP Conference Series: Earth Environ Sci, 2021, 631(1): 012079. |
| 8 | DONG S, ZHANG C, YUE Z, et al. Overall design of anode with gradient ordered structure with low Iridium loading for proton exchange membrane water electrolysis[J]. Nano Lett, 2022, 22(23): 9434-9440. |
| 9 | PUTHIYAPURA V K, MAMLOUK M, PASUPATHI S, et al. Physical and electrochemical evaluation of ATO supported IrO2 catalyst for proton exchange membrane water electrolyser[J]. J Power Sources, 2014, 269(10): 451-460. |
| 10 | FENG Q, YUAN X Z, LIU G, et al. A review of proton exchange membrane water electrolysis on degradation mechanisms and mitigation strategies[J]. J Power Sources, 2017, 366(31): 33-55. |
| 11 | LOPATA J, KANG Z, YOUNG J, et al. Effects of the transport/catalyst layer interface and catalyst loading on mass and charge transport phenomena in polymer electrolyte membrane water electrolysis devices[J]. J Electrochem Soc, 2020, 167(6): 064507. |
| 12 | 徐滨, 王锐, 苏伟, 等. 质子交换膜电解水技术关键材料的研究进展与展望[J]. 储能科学与技术, 2022, 11(11): 3510-3520. |
| XU B, WANG R, SU W, et al. Research progress and prospect of key materials of proton exchange membrane water electrolysis[J]. Energy Storage Sci Technol, 2022, 11(11): 3510-3520. | |
| 13 | ARBABI F, KALANTARIAN A, ABOUATALLAH R, et al. Feasibility study of using microfluidic platforms for visualizing bubble flows in electrolyzer gas diffusion layers[J]. J Power Sources, 2014, 258(15): 142-149. |
| 14 | 阳柳. 质子交换膜燃料电池气体扩散层及流场板结构的模拟研究[D]. 北京: 北京交通大学, 2020. |
| YANG L. Simulation study on the structure of gas diffusion layer and flow field plate in Proton exchange membrane fuel cell[D]. Beijing: Beijing Jiaotong University, 2020. | |
| 15 | ATHANASAKI G, JAYAKUMAR A, KANNAN A M. Gas diffusion layers for PEM fuel cells: materials, properties and manufacturing-a review[J]. Int J Hydrog Energy, 2023, 48(6): 2294-2313. |
| 16 | DOAN T L, LEE H E, SHAH S S H, et al. A review of the porous transport layer in polymer electrolyte membrane water electrolysis[J]. Int J Energy Res, 2021, 45(10): 14207-14220. |
| 17 | 刘春轩, 王骏斌, 高平平, 等. PEMFC钛金属双极板表面改性研究进展[J]. 稀有金属与硬质合金, 2019, 47(6): 6-9. |
| LIU C X, WANG J B, GAO P P, et al. Research progress on surface modification of PEMFC titanium metal bipolar plate[J]. Rare Metal Cemented Carbides, 2019, 47(6): 6-9. | |
| 18 | 张萍俊. 质子交换膜水电解池的性能优化及动态响应的研究[D]. 大连: 大连交通大学, 2019. |
| ZHANG J P. Performance optimization and dynammic response of proton exchange membrane water electolysis cell[D]. Dalian: Dalian Jiaotong University, 2019. | |
| 19 | BORGARDT E, PANCHENKO O, HACKEMÜLLER F J, et al. Mechanical characterization and durability of sintered porous transport layers for polymer electrolyte membrane electrolysis[J]. J Power Sources, 2018, 374(15): 84-91. |
| 20 | BAUTKINOVA T, UTSCH N, BYSTRON T, et al. Introducing titanium hydride on porous transport layer for more energy efficient water electrolysis with proton exchange membrane[J]. J Power Sources, 2023, 565(1): 232913. |
| 21 | PANCHENKO O, BORGARDT E, ZWAYGARDT W, et al. In‑situ two-phase flow investigation of different porous transport layer for a polymer electrolyte membrane (PEM) electrolyzer with neutron spectroscopy[J]. J Power Sources, 2018, 390: 108-115. [22] KANG Z, MO J, YANG G, et al. Thin film surface modifications of thin/tunable liquid/gas diffusion layers for high-efficiency proton exchange membrane electrolyzer cells[J]. Appl Energy, 2017, 206(15): 983-990. |
| 23 | MO J, DEHOFF R R, PETER W H, et al. Additive manufacturing of liquid/gas diffusion layers for low-cost and high-efficiency hydrogen production[J]. Int J Hydrog Energy, 2016, 41(4): 3128-3135. |
| 24 | ARISETTY S, PRASAD A K, ADVANI S G. Metal foams as flow field and gas diffusion layer in direct methanol fuel cells[J]. J Power Sources, 2007, 165(1): 49-57. |
| 25 | BAROUTAJI A, CARTON J G, STOKES J, et al. Application of open pore cellular foam for air breathing PEM fuel cell[J]. Int J Hydrog Energy, 2017, 42(40): 25630-25638. |
| 26 | MI B, WANG Q, QI T, et al. Performance and structure of Ti-doped amorphous carbon/CrN/Ti multilayer coating deposited on 316L stainless steel for use as bipolar plate in proton exchange membrane fuel cell[J]. J Alloys Compd, 2023, 943(15): 169080. |
| 27 | SCHULER T, SCHMIDT T J, BÜCHI F N. Polymer electrolyte water electrolysis: correlating performance and porous transport layer structure: part II. electrochemical performance analysis[J]. J Electrochem Soc, 2019, 166(10): 555-565. |
| 28 | YUAN S, ZHAO C, CAI X, et al. Bubble evolution and transport in PEM water electrolysis: mechanism, impact, and management[J]. Prog Energy Combust Sci, 2023, 96(1): 101075. |
| 29 | HWANG C M, ISHIDA M, ITO H, et al. Influence of properties of gas diffusion layers on the performance of polymer electrolyte-based unitized reversible fuel cells[J]. Int J Hydrog Energy, 2011, 36(2): 1740-1753. |
| 30 | ITO H, MAEDA T, NAKANO A, et al. Influence of different gas diffusion layers on the water management of polymer electrolyte unitized reversible fuel cell[J]. ECS Trans, 2010, 33(1): 945-954. |
| 31 | GRIGORIEV S A, MILLET P, VOLOBUEV S A, et al. Optimization of porous current collectors for PEM water electrolysers[J]. Int J Hydrogen Energy, 2009, 34(11): 4968-4973. |
| 32 | JUDE O M, FRANCESCO L, PAUL R S, et al. Effect of microstructure of porous transport layer on performance in polymer electrolyte membrane water electrolyser[J]. Energy Procedia, 2018, 151: 111-119. |
| 33 | LEE J K, LEE C H, BAZYLAK A. Pore network modelling to enhance liquid water transport through porous transport layers for polymer electrolyte membrane electrolyzers[J]. J Power Sources, 2019, 437: 226910. |
| 34 | SCHULER T, CICCONE J M, KRENTSCHER B, et al. Hierarchically structured porous transport layers for polymer electrolyte water electrolysis[J]. Adv Energy Mater, 2019, 10(2): 1903216. |
| 35 | 唐英伦, 杨小涛, 赵军, 等. 叠层扩散层对质子交换膜电解池性能影响的实验研究[J]. 工程热物理学报, 2023, 44(2): 304-310. |
| TANG Y L, YANG X T, ZHAO J, et al. Experimental study on the effects of stacked diffusion layer on the performance of proton-exchange membrane electrolysis cells[J]. J Eng Thermophys, 2023, 44(2): 304-310. | |
| 36 | MO J, STEEN S, HAN B, et al. Investigation of titanium felt transport parameters for energy storage and hydrogen/oxygen production[M]. 13th International Energy Conversion Engineering Conference, 2015. |
| 37 | LI H, FUJIGAYA T, NAKAJIMA H, et al. Optimum structural properties for an anode current collector used in a polymer electrolyte membrane water electrolyzer operated at the boiling point of water[J]. J Power Sources, 2016, 332(15): 16-23. |
| 38 | OMRANI R, SHABANI B. Review of gas diffusion layer for proton exchange membrane-based technologies with a focus on unitised regenerative fuel cells[J]. Int J Hydrogen Energy, 2019, 44(7): 3834-3860. |
| 39 | BYSTRON T, VESELY M, PAIDAR M, et al. Enhancing PEM water electrolysis efficiency by reducing the extent of Ti gas diffusion layer passivation[J]. J Appl Electrochem, 2018, 48(6): 713-723. |
| 40 | TAN A, ZHANG Y, SHI X, et al. The poisoning effects of Ti-ion from porous transport layers on the membrane electrode assembly of proton exchange membrane water electrolyzers[J]. Chem Eng J, 2023, 471(1): 144624-144631. |
| 41 | 欧阳韬, 杨扬, 张亮, 等. 酸处理钛毡对质子交换膜电解槽性能的影响[J]. 中国电机工程学报, 2023: 1-12. |
| OU-YANG T, YANG Y, ZHANG L, et al. Effect of acid-treated titanium felt on the performance of proton exchange electrolyzer[J]. Proc CSEE, 2023: 1-12. | |
| 42 | LIU C, CARMO M, BENDER G, et al. Performance enhancement of PEM electrolyzers through iridium-coated titanium porous transport layers[J]. Electrochem Commun, 2018, 97(1): 96-100. |
| 43 | LIU C, SHVIRO M, GAGO A S, et al. Exploring the interface of skin-layered titanium fibers for electrochemical water splitting[J]. Adv Energy Mater, 2021, 11(8): 2002926. |
| 44 | LIU C, SHVIRO M, BENDER G, et al. Degradation effects at the porous transport layer/catalyst layer interface in polymer electrolyte membrane water electrolyzer[J]. J Electrochem Soc, 2023, 170(3): 034508. |
| 45 | KULKARNI D, HUYNH A, SATJARUTANUN P, et al. Elucidating effects of catalyst loadings and porous transport layer morphologies on operation of proton exchange membrane water electrolyzers[J]. Appl Catal B, 2022, 308(5): 121213. |
| 46 | DOAN T L, LEE H E, KIM M, et al. Influence of IrO2/TiO2 coated titanium porous transport layer on the performance of PEM water electrolysis [J]. J Power Sources, 2022, 533(15): 231370. |
| 47 | LIU C, WIPPERMANN K, RASINSKI M, et al. Constructing a multifunctional interface between membrane and porous transport layer for water electrolyzers[J]. ACS Appl Mater Interfaces, 2021, 13(14): 16182-16196. |
| 48 | ROST U, PODLESCHNY P, SCHUMACHER M, et al. Long-term stable electrodes based on platinum electrocatalysts supported on titanium sintered felt for the use in PEM fuel cells[J]. IOP Conference Series: Mater Sci Eng, 2018, 416(1): 012013. |
| 49 | BERNT M, SCHRAMM C, SCHRÖTER J, et al. Effect of the IrOx conductivity on the anode electrode/porous transport layer interfacial resistance in PEM water electrolyzers[J]. J Electrochem Soc, 2021, 168(8): 084513. |
| 50 | RAKOUSKY C, KEELEY G P, WIPPERMANN K, et al. The stability challenge on the pathway to high-current-density polymer electrolyte membrane water electrolyzers[J]. Electrochim Acta, 2018, 278(10): 324-331. |
| 51 | 范芷萱, 俞红梅, 姜广, 等. PEM水电解池低成本阳极钛纤维毡扩散层研究[J]. 电源技术, 2019, 44(7): 933-937. |
| FAN Z X, YU H M, JIANG G, et al. A low-cost Ti felt anode gas diffusion layer for PEM water electrolysis[J]. Chin J Power Sources, 2019, 44(7): 933-937. | |
| 52 | FAN Z X, YU H M, JIANG G, et al. Low precious metal loading porous transport layer coating and anode catalyst layer for proton exchange membrane water electrolysis[J]. Int J Hydrogen Energy, 2022, 47(44): 18963-18971. |
| 53 | 卫飞彬. 高电密下高耐蚀性钛基阳极的制备及其电催化性能研究[D]. 北京: 北京化工大学, 2022. |
| WEI F B. Study on preparation of high corrosion resistance Ti-based anode and its electrocatalytic performance in acid[D]. Beijing: Beijing University of Chemical Technology, 2022. | |
| 54 | DAUDT N F, SCHNEIDER A D, ARNEMANN E R, et al. Fabrication of NbN-coated porous titanium sheets for PEM electrolyzers[J]. J Mater Eng Perform, 2020, 29(8): 5174-5183. |
| 55 | 黄敏, 刘高阳, 王新东. PEM水电解用多层钛网析氧阳极制备及性能研究[J]. 有色金属科学与工程, 2016, 7(3): 1-5. |
| HUANG M, LIU G Y, WANG X D. Synthesis and performance study of multilayer titanium mesh oxygen evolution anode in the polymer exchange membrane water electrolysis[J]. Nonferrous Met Sci Eng, 2016, 7(3): 1-5. |
| [1] | 程羽, 何灵均, 林楚园, 林慧, 肖富玉, 赖文斌, 钱庆荣, 黄晓霞, 陈庆华, 曾令兴. 水系锌离子电池宽温域性能的电解质改性策略研究进展[J]. 应用化学, 2024, 41(3): 349-364. |
| [2] | 刘思蓓, 梅竣乔, 谢谨裕, 刘轶君, 邓凤霞, 邱珊. 活性炭强化泡沫钛基空气扩散电极电过臭氧氧化及降解布洛芬效能[J]. 应用化学, 2024, 41(2): 279-296. |
| [3] | 张鼎, 杨微微, 缪嵩松, 苏怡. 含氮多孔碳限域金纳米粒子用于电化学检测液相二氧化氯[J]. 应用化学, 2023, 40(11): 1572-1580. |
| [4] | 葛君杰. 《氢能源与燃料电池》专辑序言[J]. 应用化学, 2023, 40(8): 1061-1062. |
| [5] | 罗二桂, 唐涛, 王艺, 张俊明, 常宇虹, 胡天军, 贾建峰. 两电子氧还原制备过氧化氢:贵金属催化剂的几何与电子结构调控的研究进展[J]. 应用化学, 2023, 40(8): 1063-1076. |
| [6] | 董以宁, 李赫, 宫雪, 韩策, 宋平, 徐维林. 非Pt基催化剂在质子交换膜燃料电池阴极氧还原反应中的研究进展[J]. 应用化学, 2023, 40(8): 1077-1093. |
| [7] | 谭翠盈, 丁威超, 马婷婷, 肖瑶, 刘健. 超亲水/超疏气电解水催化剂的研究进展[J]. 应用化学, 2023, 40(8): 1109-1125. |
| [8] | 尹春, 李家欣, 冯立纲. 浅析尿素电解制氢及尿素燃料电池研究进展[J]. 应用化学, 2023, 40(8): 1158-1174. |
| [9] | 郑锐雪, 孟庆磊, 张丽, 刘长鹏, 邢巍, 肖梅玲. 分级孔结构的Fe-N-C催化剂用于高效电催化氧还原[J]. 应用化学, 2023, 40(8): 1187-1194. |
| [10] | 惠连成, 庄鉴行, 肖顺, 李美平, 靳梦圆, 吕青. 镍氮掺杂石墨炔用作高效氧还原电催化剂[J]. 应用化学, 2023, 40(8): 1205-1213. |
| [11] | 徐晓芳, 陈强, 张海宝. 滑动弧等离子体固氮研究进展[J]. 应用化学, 2023, 40(7): 923-937. |
| [12] | 朱凤, 彭小连, 张文彬. 质子给受体对电催化反应影响的研究进展[J]. 应用化学, 2023, 40(5): 666-680. |
| [13] | 胡方正, 高兴, 刘雷, 袁天恒, 曹宁, 李凯, 王亚涛, 李建华, 连慧琴, 汪晓东, 崔秀国. 锂离子电池黑磷负极的储能优势及其优化的研究进展[J]. 应用化学, 2023, 40(4): 571-582. |
| [14] | 师文君, 孙中辉, 宋忠乾, 许佳楠, 韩冬雪, 牛利. 钠离子电池层状过渡金属氧化物正极材料研究进展[J]. 应用化学, 2023, 40(4): 583-596. |
| [15] | 石雪建, 刘万强, 王春丽, 程勇, 王立民. 钾离子电池用Sb基负极材料研究进展[J]. 应用化学, 2023, 40(2): 210-228. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||