
应用化学 ›› 2024, Vol. 41 ›› Issue (2): 279-296.DOI: 10.19894/j.issn.1000-0518.230317
活性炭强化泡沫钛基空气扩散电极电过臭氧氧化及降解布洛芬效能
刘思蓓1, 梅竣乔1, 谢谨裕2, 刘轶君1, 邓凤霞1( ), 邱珊1(
), 邱珊1( )
)
- 1.哈尔滨工业大学环境学院,哈尔滨 150000
2.成都市排水有限责任公司,成都 610023
-
收稿日期:2023-10-12接受日期:2023-12-29出版日期:2024-02-01发布日期:2024-03-05 -
通讯作者:邓凤霞,邱珊 -
基金资助:国家自然科学基金(52070056);城市水资源与环境国家重点实验室(哈尔滨工业大学(2021TS26);2023TS17)、污泥安全处置与资源化回收国家工程研究中心(2021B005);教育部“春晖计划”合作科研项目(202200482)
Activated Carbon Boosted Performance of a Titanium-Based Air Diffusion Electrode in Electro-Peroxone Oxidation and Ibuprofen Degradation
Si-Bei LIU1, Jun-Qiao MEI1, Jin-Yu XIE2, Yi-Jun LIU1, Feng-Xia DENG1( ), Shan QIU1(
), Shan QIU1( )
)
- 1.School of Environment,Harbin Institute of Technology,Harbin 150000,China
2.Chengdu Drainage Co. ,Ltd,Chengdu 610023,China
-
Received:2023-10-12Accepted:2023-12-29Published:2024-02-01Online:2024-03-05 -
Contact:Feng-Xia DENG,Shan QIU -
About author:qiushan@hit.edu.cn;
dengfx@hit.edu.cn
-
Supported by:the National Natural Science Foundation of China(52070056);the State Key Laboratory of Urban Water Resource and Environment (Harbin Institute of Technology(2021TS26);2023TS17), the National Engineering Research Center for Safe Sludge Disposal and Resource Recovery(2021B005);the Ministry of Education's “Chunhui Project” Cooperative Scientific Research Project(202200482)
摘要:
为了提高传统污水处理技术对布洛芬这一新型污染物的去除效率,本研究构建了一种基于活性炭强化的泡沫钛空气扩散电极(AC@Ti-F GDE)的电过臭氧处理体系。 该体系通过增强传统电过臭氧体系的氧化效能,用于布洛芬的去除。 结果表明,相对于传统的电极(Ti-F),微孔泡沫钛空气扩散电极(Ti-F GDE)体系中的HO?相对含量提升了155.7%。 在投加活性炭后,AC@Ti-F GDE体系中的HO?相对含量在Ti-F GDE体系的基础上进一步提升了35.4%。 这种显著提升的HO?产量得益于AC@Ti-F GDE体系中H2O2的增加,从而进一步强化了电过臭氧的氧化效能。 通过精确调控活性炭的投加密度和颗粒粒径,确定了AC@Ti-F GDE电过臭氧体系中最佳的活性炭投加密度为0.8 mg/cm3,最佳颗粒粒径为850 μm。 在此基础上,将AC@Ti-F GDE电过臭氧体系应用于布洛芬污染物的处理,通过对比不同因素下该体系降解布洛芬的程度,得到最佳运行条件: pH值为7.00,电流为150 mA,O3,gas浓度为52.2 mg/L,并采用液相质谱检测布洛芬降解过程中中间产物,得出了该体系的降解矿化机理: 布洛芬首先通过羟基化、去甲基化和脱羧等反应逐步氧化支链,随后攻击苯环并开环生成脂肪酸,最终被进一步矿化生成CO2和H2O。 AC@Ti-F GDE电过臭氧新体系,强化了传统电过臭氧体系氧化效能,为后续电过臭氧处理布洛芬提供新思路。
中图分类号:
引用本文
刘思蓓, 梅竣乔, 谢谨裕, 刘轶君, 邓凤霞, 邱珊. 活性炭强化泡沫钛基空气扩散电极电过臭氧氧化及降解布洛芬效能[J]. 应用化学, 2024, 41(2): 279-296.
Si-Bei LIU, Jun-Qiao MEI, Jin-Yu XIE, Yi-Jun LIU, Feng-Xia DENG, Shan QIU. Activated Carbon Boosted Performance of a Titanium-Based Air Diffusion Electrode in Electro-Peroxone Oxidation and Ibuprofen Degradation[J]. Chinese Journal of Applied Chemistry, 2024, 41(2): 279-296.
| Region | Sampling date | ρ/(mg·L-1) | Ref. |
|---|---|---|---|
| Taihu basin (China) | December 2020-May 2021 | 2.6 | [ |
| Pearl river basin (China) | January 2017 | 1.4 | [ |
| Llobregat river basin (Spain) | - | 1.4 | [ |
| Umgeni river basin (South Africa) | January 2015-July 2016 | 17.6 | [ |
表1 不同污水处理厂进水中布洛芬检出最高质量浓度
Table 1 The highest mass concentration of ibuprofen detected in influent of different sewage treatment plants
| Region | Sampling date | ρ/(mg·L-1) | Ref. |
|---|---|---|---|
| Taihu basin (China) | December 2020-May 2021 | 2.6 | [ |
| Pearl river basin (China) | January 2017 | 1.4 | [ |
| Llobregat river basin (Spain) | - | 1.4 | [ |
| Umgeni river basin (South Africa) | January 2015-July 2016 | 17.6 | [ |

图2 不同体系HO?相对含量(A)、 O3,liquid浓度(B) 和H2O2的积累量 (C)。反应在电流为50 mA,活性炭粒径为850 μm,投加密度为0.8 mg/cm3,Na2SO4溶液浓度为50 mmol/L,pH值为7,O3,gas质量浓度为16.5 mg/L,曝气流量为50 mL/min,反应时间为15 min的条件下进行
Fig.2 Relative content of HO? (A), concentration of O3,liquid (B) and accumulation of H2O2 (C) in different systems. The reaction was carried out under the conditions of current of 50 mA, activated carbon particle size of 850 μm, dosage of 0.8 mg/cm3, Na2SO4 solution concentration of 50 mmol/L, pH of 7, O3,gas mass concentration of 16.5 mg/L, aeration flow rate of 50 mL/min and reaction time of 15 min

图3 活性炭投加密度为0、0.8、8.3和12.4 mg/cm3对HO?相对含量影响No current application; d(GAC)=850 μm; c(Na2SO4)=50 mmol/L; pH=7; Reration flow rate=50 mL/min;O3,gas=15 mg/L; reaction time=15 min
Fig.3 Effect of activated carbon dosage density of 0, 0.8,8.3 and 12.4 mg/cm3 on the relative content of HO?.

图5 不同活性炭投加密度(A)及电流(B)对H2O2的积累量影响
Fig.5 Effect of different activated carbon dosage density (A) and current (B) on the accumulation of H2O2 Reaction current:50 mA; 100 mA. Particlesize: 850 μm; c(Na2SO4)=50 mmol/L; pH=7; v(oxygen aeration)=50 mL/min; Anode: BDD, reaction time≥15 min

图6 不同粒径活性炭对H2O2积累量(A)和H2O2分解(B)的影响及其吸附等温线(C)和孔径分布图(D)
Fig.6 Effects of different particle sizes activated carbon on the accumulation (A) and decomposition (B) of H2O2, as well as their adsorption isotherms (C) and pore size distribution (D)

图7 不同粒径活性炭的RRDE图(A)及电子转移数(B)
Fig.7 Rotating ring-disk electrode (RRDE) diagram (A) and electron transfer number (B) of activated carbon with different particle sizes

图9 不同粒径活性炭的C1s分峰拟合图(A-C)、化学键占比(D)、N1s峰(E)及C—N占比与电子转移数相关性拟合(F)
Fig.9 C1s peak fitting diagrams (A-C), chemical bond proportion (D), N1s peak (E) and correlation fitting between C—N proportion and electron transfer number (F) of activated carbon with different particle sizes
| No. | Molecular formula | Relative molecular mass | Chemical formula |
|---|---|---|---|
| A | C13H18O2 | 206 |  |
| B | C13H19O4 | 238 |  |
| C1, C2 | C13H18O3 | 221 |  |
| D | C11H14O3 | 193 |  |
| E | C12H16O | 175 |  |
| F | C10H12O4 | 196 |  |
| G | C11H14O2 | 177 |  |
| H | C10H11O2 | 164 |  |
| I | C12H16 | 160 |  |
| J | C9H8O2 | 147 |  |
| K1, K2 | C9H10O | 133 | 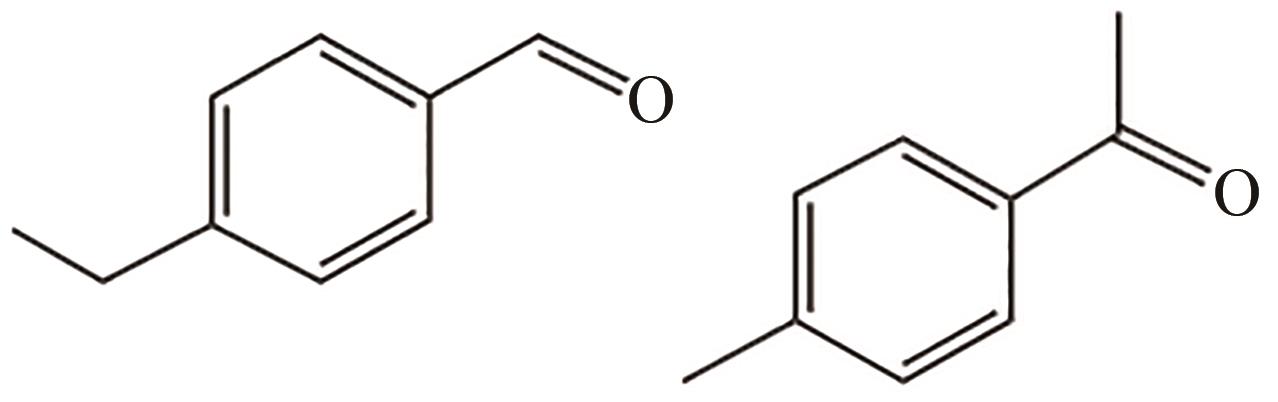 |
| L | C6H10O3 | 129 |  |
| M | C5H10O3 | 117 |  |
| N1, N2 | C4H4O4 | 116 |  |
| O | C3H4O3 | 87 |  |
| P | C2H4O2 | 59 |  |
| Q | C2H2O4 | 90 |  |
表2 LC-MS对布洛芬降解中间产物检测结果
Table 2 Determination of ibuprofen degradation intermediates by LC-MS
| No. | Molecular formula | Relative molecular mass | Chemical formula |
|---|---|---|---|
| A | C13H18O2 | 206 |  |
| B | C13H19O4 | 238 |  |
| C1, C2 | C13H18O3 | 221 |  |
| D | C11H14O3 | 193 |  |
| E | C12H16O | 175 |  |
| F | C10H12O4 | 196 |  |
| G | C11H14O2 | 177 |  |
| H | C10H11O2 | 164 |  |
| I | C12H16 | 160 |  |
| J | C9H8O2 | 147 |  |
| K1, K2 | C9H10O | 133 |  |
| L | C6H10O3 | 129 |  |
| M | C5H10O3 | 117 |  |
| N1, N2 | C4H4O4 | 116 |  |
| O | C3H4O3 | 87 |  |
| P | C2H4O2 | 59 |  |
| Q | C2H2O4 | 90 |  |

图15 电流-时间模式运行7 h情况(A);反应前(B)及电解10次后(C)电极SEM图
Fig.15 Current time mode operation for 7 h (A); SEM images of electrode before reaction (B) and after 10 electrolysis cycles (C)
| 1 | 唐忠辉, 谢莉. 浅谈新污染物的特征、防治进展及建议[J]. 广东化工, 2022, 49(23): 203-204, 207. |
| TANG Z H, XIE L. Characteristics, prevention and treatment progress and suggestions of emerging pollutants[J]. Guangdong Chem Ind, 2022, 49(23): 203-204, 207. | |
| 2 | 宫晨皓, 任晓晶, 何灿, 等. 电催化强化对布洛芬的去除效果及机制[J]. 环境工程学报, 2022, 16(7): 2156-2164. |
| GONG C H, REN X J, HE C, et al. Removal effect and mechanism of ibuprofen by enhanced electrocatalysis[J]. Chin J Environ Eng, 2022, 16(7): 2156-2164. | |
| 3 | 崔欣欣, 林志荣, 王会姣, 等. 基于穿透电极的Electro-peroxone技术降解布洛芬[J]. 中国环境科学, 2019, 39(4): 1619-1626. |
| CUI X X, LIN Z R, WANG H J, et al. Effective degradation of ibuprofen by flow-through electro-peroxone process[J]. China Environ Sci, 2019, 39(4): 1619-1626. | |
| 4 | YAO W K, REHMAN S W U, WANG H J, et al. Pilot-scale evaluation of micropollutant abatements by conventional ozonation, UV/O3, and an electro-peroxone process[J]. Water Res, 2018, 138: 106-107. |
| 5 | LI Y K, SHEN W H, FU S J, et al. Inhibition of bromate formation during drinking water treatment by adapting ozonation to electro-peroxone process[J]. Chem Eng J, 2015, 264: 322-328. |
| 6 | WANG Y J, YU G, DENG S B, et al. The electro-peroxone process for the abatement of emerging contaminants: mechanisms, recent advances, and prospects[J]. Chemosphere, 2018, 208: 640-654. |
| 7 | GHANBARI F, ZIRRAHI F, LIN K Y A, et al. Enhanced electro-peroxone using ultrasound irradiation for the degradation of organic compounds: a comparative study[J]. J Environ Chem Eng, 2020, 8(5): 104167. |
| 8 | 周鹏, 展巨宏, 赵尔卓, 等. 电催化臭氧-BAF工艺深度处理印染废水中试研究[J]. 工业水处理, 2022, 42(11): 100-106. |
| ZHOU P, ZHAN J H, ZHAO E Z, et al. Pilot study on advanced treatment of printing and dyeing wastewater by electro-peroxone-BAF process[J]. Ind Water Treat, 2022, 42(11): 100-106. | |
| 9 | GUO Y, ZHAO E Z, WANG J, et al. Comparison of emerging contaminant abatement by conventional ozonation, catalytic ozonation, O3/H2O2 and electro-peroxone processes[J]. J Hazard Mater, 2020, 389: 8. |
| 10 | LI X, WANG Y J, YUAN S, et al. Degradation of the anti-inflammatory drug ibuprofen by electro-peroxone process[J]. Water Res, 2014, 63: 81-93. |
| 11 | HUBER M M, CANONICA S, PARK G Y, et al. Oxidation of pharmaceuticals during ozonation and advanced oxidation processes[J]. Environ Sci Technol, 2003, 37(5): 1016-1024. |
| 12 | LIU Q X, FENG X, CHEN N, et al. Occurrence and risk assessment of typical PPCPs and biodegradation pathway of ribavirin in wastewater treatment plants[J]. Environ Sci Ecotechnol, 2022, 11: 100184. |
| 13 | 陈文文, 李蕊, 李晓明, 等. 河岸过滤系统中新污染物的生物降解机制研究进展[J]. 环境科学学报, 2023, 43(7): 1-9. |
| CHEN W W, LI R, LI X M, et al. Microbial degradation mechanism of emerging contaminants in the riverbank filtration system: a review[J]. Acta Sci Circumstantiae, 2023, 43(7): 1-9. | |
| 14 | 黄文平, 鲍轶凡, 胡霞林, 等. 黄浦江上游水源地中31种内分泌干扰物的分布特征以及生态风险评价[J]. 环境化学, 2020, 39(6): 1488-1495. |
| HUANG W P, BAO Y F, HU X L, et al. Occurrence and ecological risk assessment of 31 endocrine disrupting chemicals in the water source of upstream Huangpu river[J]. Environ Chem, 2020, 39(6): 1488-1495. | |
| 15 | BOLEDA M R, GALCERAN M T, VENTURA F. Validation and uncertainty estimation of a multiresidue method for pharmaceuticals in surface and treated waters by liquid chromatography-tandem mass spectrometry[J]. J Chromatogr A, 2013, 1286: 146-158. |
| 16 | GUMBI B P, MOODLEY B, BIRUNGI G, et al. Detection and quantification of acidic drug residues in South African surface water using gas chromatography-mass spectrometry[J]. Chemosphere, 2017, 168: 1042-1050. |
| 17 | JUNGLEE S, URBAN L, SALLANON H, et al. Optimized assay for hydrogen peroxide determination in plant tissue using potassium iodide[J]. Am J Anal Chem, 2014, 5: 7. |
| 18 | DENG F X, LI S X, ZHOU M H, et al. A biochar modified nickel-foam cathode with iron-foam catalyst in electro-Fenton for sulfamerazine degradation[J]. Appl Catal B: Environ, 2019, 256: 14. |
| 19 | 邱珊, 高伟杰, 邓凤霞, 等. 3D打印气体扩散电极产H2O2及其对焦化废水的处理研究[J]. 中国环境科学, 2018, 38: 4075-4084. |
| QIU S, GAO W J, DENG F X, et al. Enhancement of H2O2 accumulationat gas diffusion electrodes (GDEs) optimized by 3D-printed technique and its utilization in electro-Fenton for coking wastewater treatment[J]. China Environ Sci, 2018, 38(11): 4075-4084. | |
| 20 | 王业耀, 王占生. 靛红钾法测定水中的臭氧浓度[J]. 中国给水排水, 2003, 19: 95-97. |
| WANG Y Y, WANG Z S. Determination of ozone concentration in water by isatin potassium method[J]. China Water Wastewater, 2003, 19: 95-97. | |
| 21 | 柴铖. 改性生物炭催化臭氧降解水中布洛芬的特性与机理研究[D]. 西安: 西安建筑科技大学, 2023. |
| CHAI C. Characteristics and mechanism of ibuprofen degradation in water catalyzed by ozone with modified biochar[D]. Xi'an: Xi′an University of Architecture and Technology, 2023. | |
| 22 | SANCHEZ-POLO M, VON GUNTEN U, RIVERA-UTRILLA J. Efficiency of activated carbon to transform ozone into center dot OH radicals: influence of operational parameters[J]. Water Res, 2005, 39(14): 3189-3198. |
| 23 | 李根, 朱雷, 陈天翼, 等. 粒径与前驱体对活性炭催化臭氧氧化的影响[J]. 环境科学学报, 2018, 38(4): 1494-1500. |
| LI G, ZHU L, CHEN T Y, et al. The influence of particle size and precursor material on activated carbo catalytic ozonation[J]. Acta Sci Circumstantiae, 2018, 38(4): 1494-1500. | |
| 24 | COUGNAUD A, FAUR C, LE CLOIREC P. Removal of pesticides from aqueous solution: quantitative relationship between activated carbon characteristics and adsorption properties[J]. Environ Technol, 2005, 26(8): 857-866. |
| 25 | SKIBINSKI B, GOTZE C, WORCH E, et al. Pore diffusion limits removal of monochloramine in treatment of swimming pool water using granular activated carbon[J]. Water Res, 2018, 132: 270-281. |
| 26 | TEJEDOR-SANZ S, QUEJIGO J R, BERNA A, et al. The planktonic relationship between fluid-like electrodes and bacteria: wiring in motion[J]. ChemSusChem, 2017, 10(4): 693-700. |
| 27 | RIBEIRO R S, SILVA A M T, FIGUEIREDO J L, et al. The influence of structure and surface chemistry of carbon materials on the decomposition of hydrogen peroxide[J]. Carbon, 2013, 62: 97-108. |
| 28 | HUANG H H, LU M C, CHEN J N, et al. Catalytic decomposition of hydrogen peroxide and 4-chlorophenol in the presence of modified activated carbons[J]. Chemosphere, 2003, 51(9): 935-943. |
| 29 | NABAE Y, PARK J, HAYAKAWA T, et al. Highly selective two electron reduction of oxygen catalyzed by N-doped mesoporous carbon: 247th national spring meeting of the American-Chemical-Society (ACS)[C]. Dallas: American Chemical Society, 2014, 247: 1. |
| 30 | LI Y, SHEN J M, LI J S, et al. A protic salt-derived porous carbon for efficient capacitive deionization: balance between porous structure and chemical composition[J]. Carbon, 2017, 116: 21-32. |
| 31 | RISH S K, TAHMASEBI A, WANG R, et al. Formation mechanism of nano graphitic structures during microwave catalytic graphitization of activated carbon[J]. Diamond Relat Mater, 2021, 120: 14. |
| 32 | QIU T, YANG J G, BAI X J. Insight into the change in carbon structure and thermodynamics during anthracite transformation into graphite[J]. Int J Miner Metall Mater, 2020, 27(2): 162-172. |
| 33 | ZHOU H H, YU Q, PENG Q L, et al. Catalytic graphitization of carbon fibers with electrodeposited Ni-B alloy coating[J]. Mater Chem Phys, 2008, 110(2/3): 434-439. |
| 34 | WU G, JOHNSTON C M, MACK N H, et al. Synthesis-structure-performance correlation for polyaniline-Me-C non-precious metal cathode catalysts for oxygen reduction in fuel cells[J]. J Mater Chem, 2011, 21(30): 11392-11405. |
| 35 | PAN F P, DENG W, JUSTINIANO C, et al. Identification of champion transition metals centers in metal and nitrogen-codoped carbon catalysts for CO2 reduction[J]. Appl Catal B: Environ, 2018, 226: 463-472. |
| 36 | YANG Q, HUANG H, LI K L, et al. Ibuprofen removal from drinking water by electro-peroxone in carbon cloth filter[J]. Chem Eng J, 2021, 415: 127618. |
| 37 | ZHANG J Y, XIA C A, WANG H F, et al. Recent advances in electrocatalytic oxygen reduction for on-site hydrogen peroxide synthesis in acidic media[J]. J Energy Chem, 2022, 67: 432-450. |
| 38 | ASGARI G, SEID-MOHAMMADI A, RAHMANI A, et al. Diuron degradation using three-dimensional electro-peroxone (3D/E-peroxone) process in the presence of TiO2/GAC: application for real wastewater and optimization using RSM-CCD and ANN-GA approaches[J]. Chemosphere, 2021, 266: 129179. |
| 39 | YAO W K, QU Q Y, VON GUNTEN U, et al. Comparison of methylisoborneol and geosmin abatement in surface water by conventional ozonation and an electro-peroxone process[J]. Water Res, 2017, 108: 373-382. |
| 40 | GUO Y, LONG J F, HUANG J, et al. Can the commonly used quenching method really evaluate the role of reactive oxygen species in pollutant abatement during catalytic ozonation?[J]. Water Res, 2022, 215: 118275. |
| 41 | GUO Y, ZHANG Y X, YU G, et al. Revisiting the role of reactive oxygen species for pollutant abatement during catalytic ozonation: the probe approach versus the scavenger approach[J]. Appl Catal B: Environ, 2021, 280: 119418. |
| 42 | SRINIVASAN R, NAMBI I M. An electro-peroxone-based multi-pronged strategy for the treatment of ibuprofen and an emerging pharmaceutical wastewater using a novel graphene-coated nickel foam electrode[J]. Chem Eng J, 2022, 450: 137618. |
| 43 | GUO M C, ZHOU L X, LI Y, et al. Unique nanosheet-nanowire structured CoMnFe layered triple hydroxide arrays as self-supporting electrodes for a high-efficiency oxygen evolution reaction[J]. J Mater Chem A, 2019, 7(21): 13130-13141. |
| [1] | 喻昌木, 张荣, 卢小鸾, 杨敏, 彭黔荣. 具有过氧化物酶活性的Imm-Fe3+-IL的制备及用于比色法测定H2O2和葡萄糖[J]. 应用化学, 2020, 37(10): 1211-1220. |
| [2] | 杨磊, 张赟星, 汪小琴. 布洛芬插层类水滑石/淀粉凝胶复合物的制备及缓释行为[J]. 应用化学, 2018, 35(7): 781-787. |
| [3] | 金超, 符华林, 张伟, 张艳丽, 曹航, 周涛, 卢朝成, 刘梦娇, 周建瑜. 布洛芬-扑热息痛孪药的合成[J]. 应用化学, 2014, 31(07): 782-787. |
| [4] | 李洋, 宁志刚, 谭颖, 孙淑苗, 王丕新. 淀粉基荧光微球的制备及其布洛芬包载和释药性能[J]. 应用化学, 2011, 28(10): 1114-1121. |
| [5] | 苏小笛, 刘昭林, 朱敏, 沈含熙. 增效试剂对过氧化物模拟酶催化显色体系的增效作用——Fe-TPPS4-4-AAP-苯酚衍生物-H2O2体系[J]. 应用化学, 1997, 0(3): 33-36. |
| [6] | 钱逸泰, 陈乾旺, 陈祖耀, 周贵恩, 范成高. H2O2氧化-水热处理联用法制备高纯超微BaTiO3粉末[J]. 应用化学, 1993, 0(3): 32-34. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||







