Chinese Journal of Applied Chemistry ›› 2022, Vol. 39 ›› Issue (1): 142-153.DOI: 10.19894/j.issn.1000-0518.210486
• Review • Previous Articles Next Articles
Research Progress on Condensation⁃Induced Invalid of Super⁃hydrophobicity
ZHANG Wen-Jing1,WANG De-Hui1,2( ),DENG Xu2
),DENG Xu2
- 1.Yangtze Delta Region Institute of University of Electronic Science and Technology of China,Huzhou 313000,China
2.Institute of Fundamental and Frontier Sciences,University of Electronic Science and Technology,Chengdu 610054,China
-
Received:2021-09-30Accepted:2021-11-01Published:2022-01-01Online:2022-01-10 -
Contact:De-Hui WANG -
About author:wangdehui@uestc.edu.cn
-
Supported by:the National Natural Science Foundation of China(22072014);the Program for Scientific?Technical Young Talents of Sichuan(2021JDJQ0013);Sichuan Science and Technology Program(2021JDRC0016);the Key Projects in International Science & Technology Cooperation Program of Chengdu, Sichuan(2021?GH02?00105?HZ)
CLC Number:
Cite this article
ZHANG Wen-Jing, WANG De-Hui, DENG Xu . Research Progress on Condensation⁃Induced Invalid of Super⁃hydrophobicity[J]. Chinese Journal of Applied Chemistry, 2022, 39(1): 142-153.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.210486

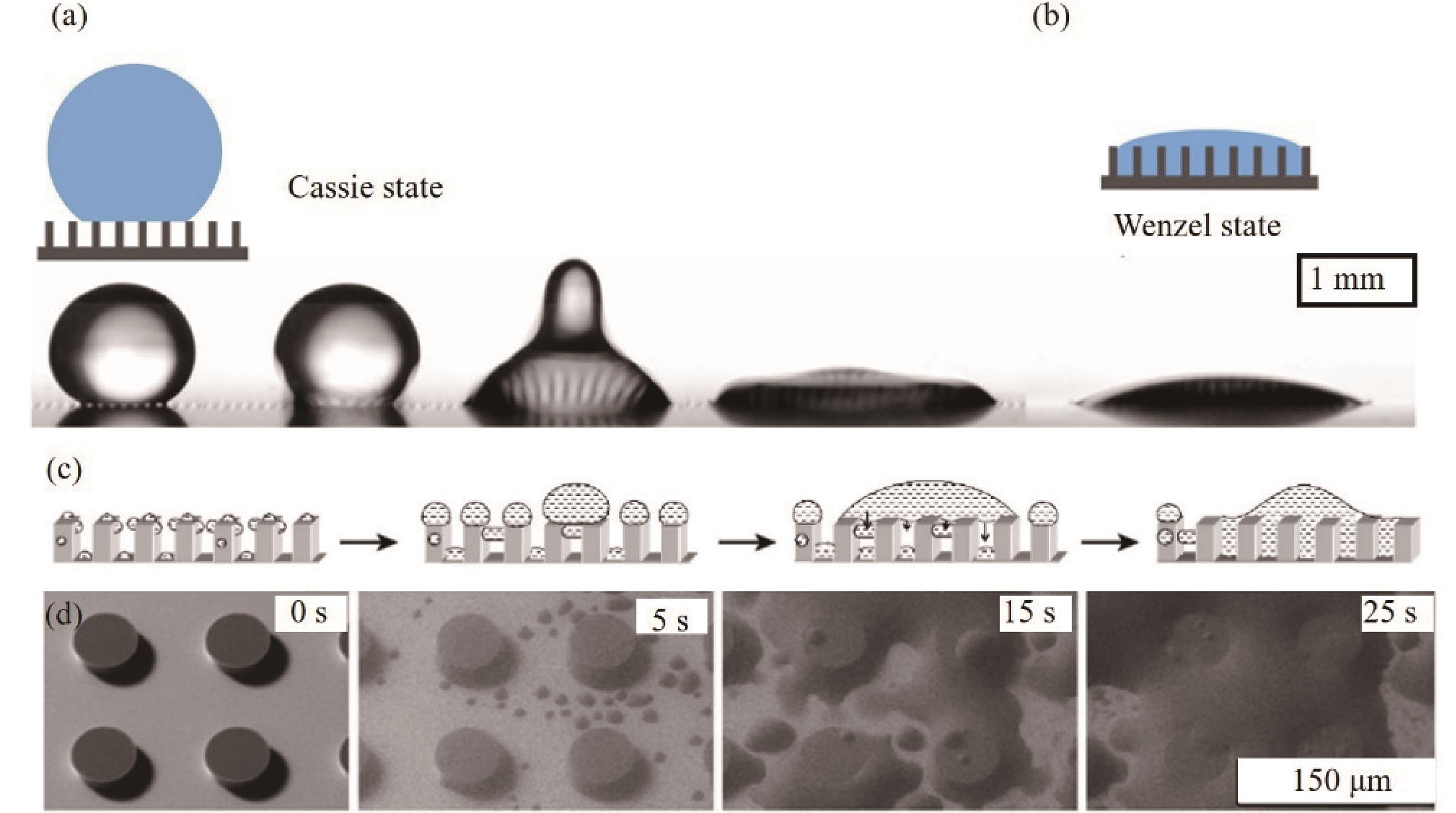
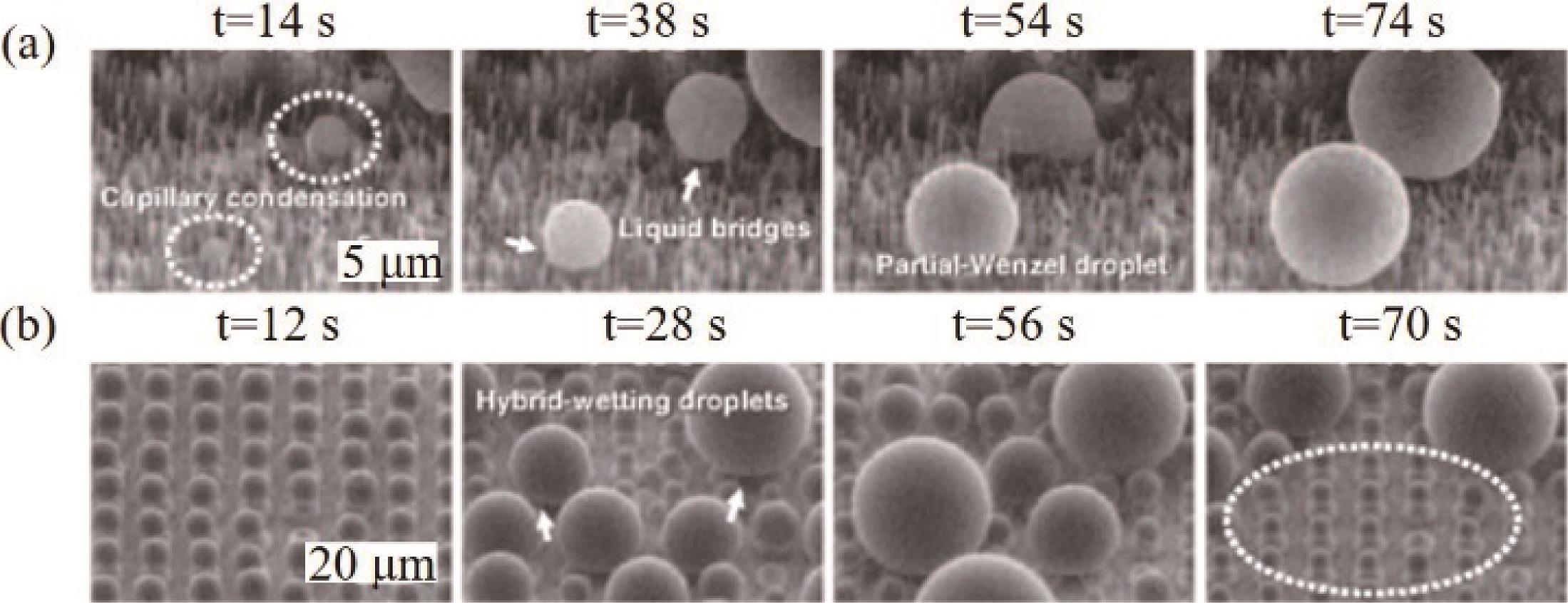
Fig.3 ESEM images of the condensation of water vapor on a superhydrophobic surface comprising of an array of hydrophobic square posts. (a) Dry surface. (b-c) Snapshot images of the condensation phenomenon on the surface[57]
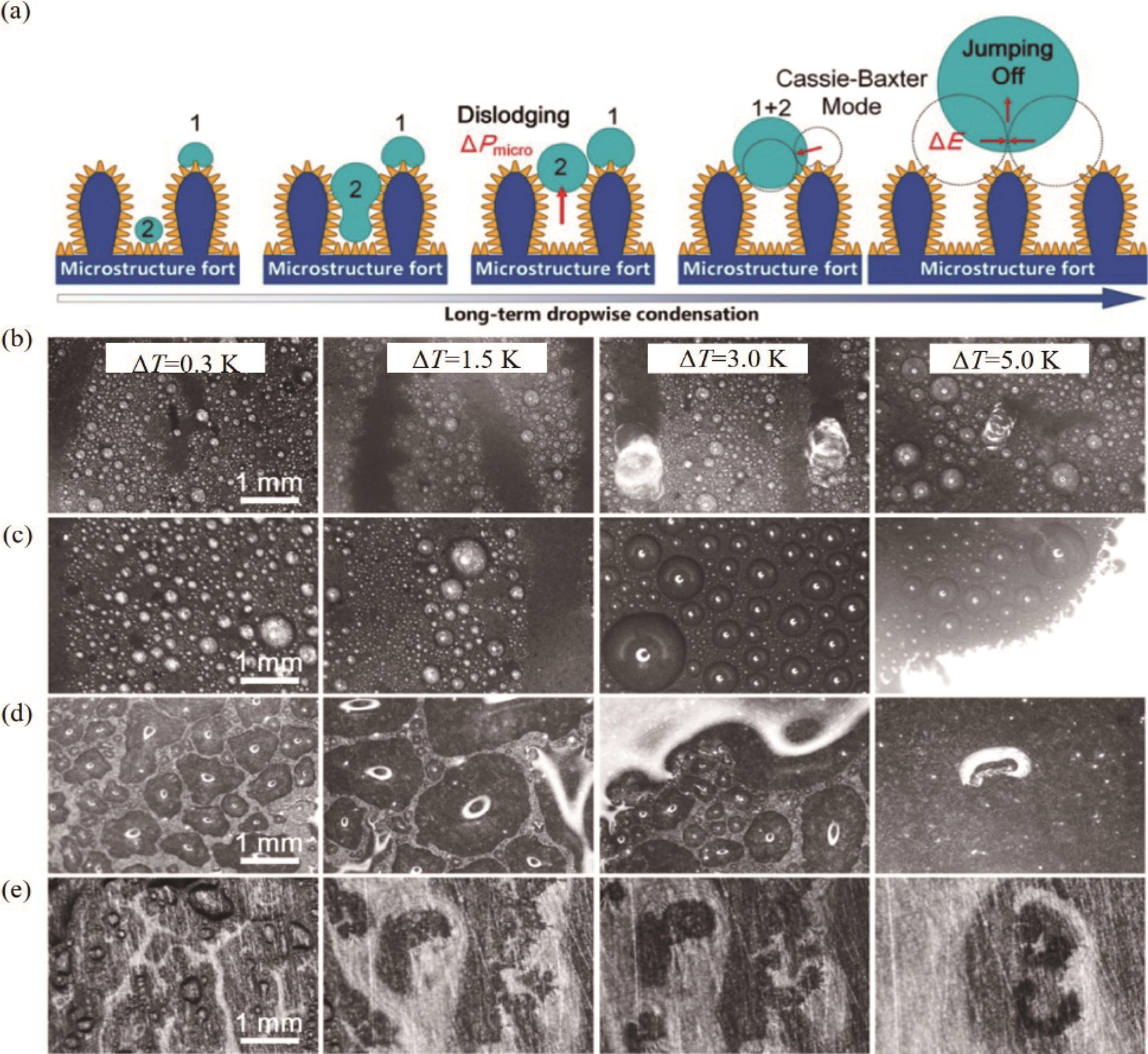
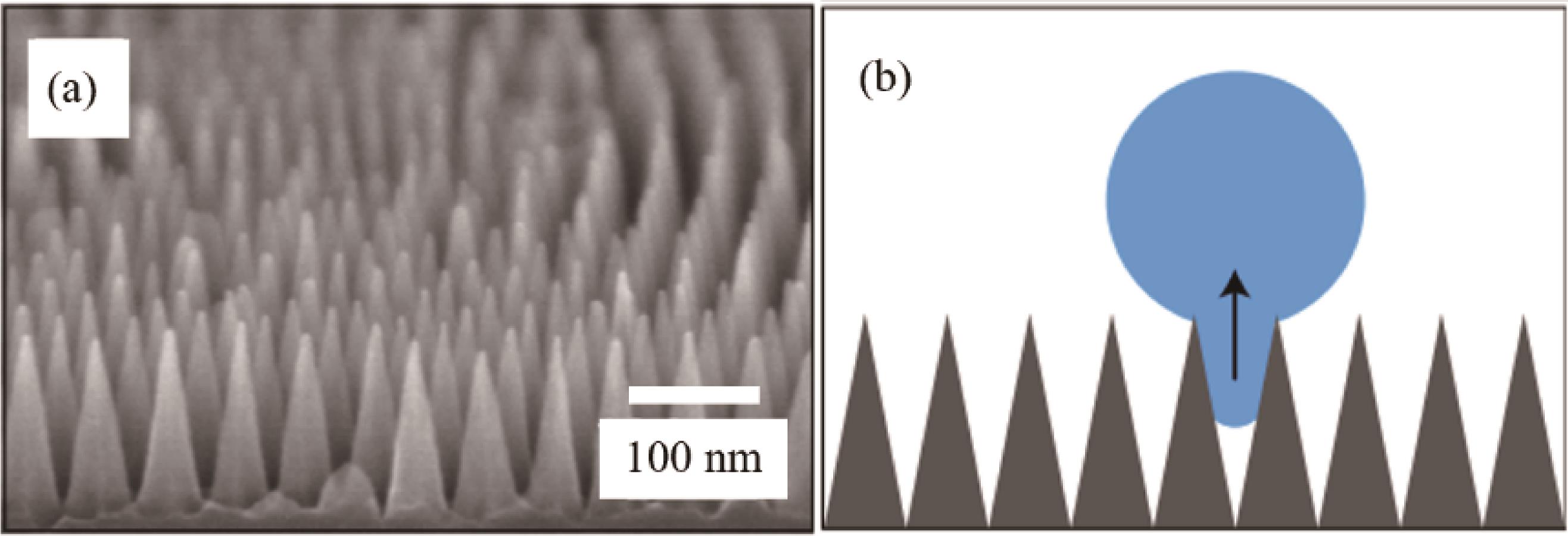
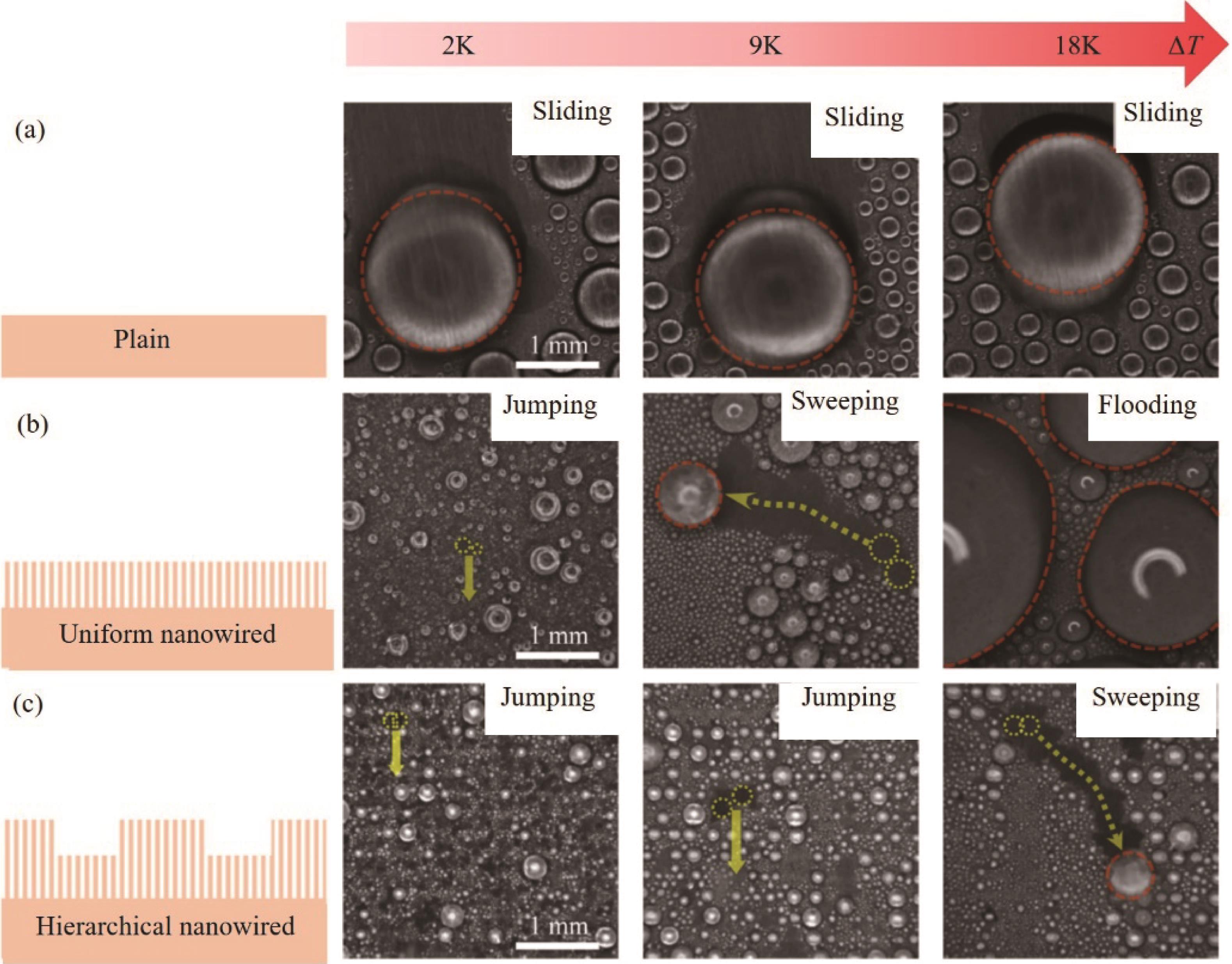
Fig.8 (a) Dropwise condensation and schematics of long-term dropwise condensation on the surface; (b - e) Images of condensated droplets on four types of surfaces at different subcooling, including MNSS, NGSS, MPSS, and Cu[68]
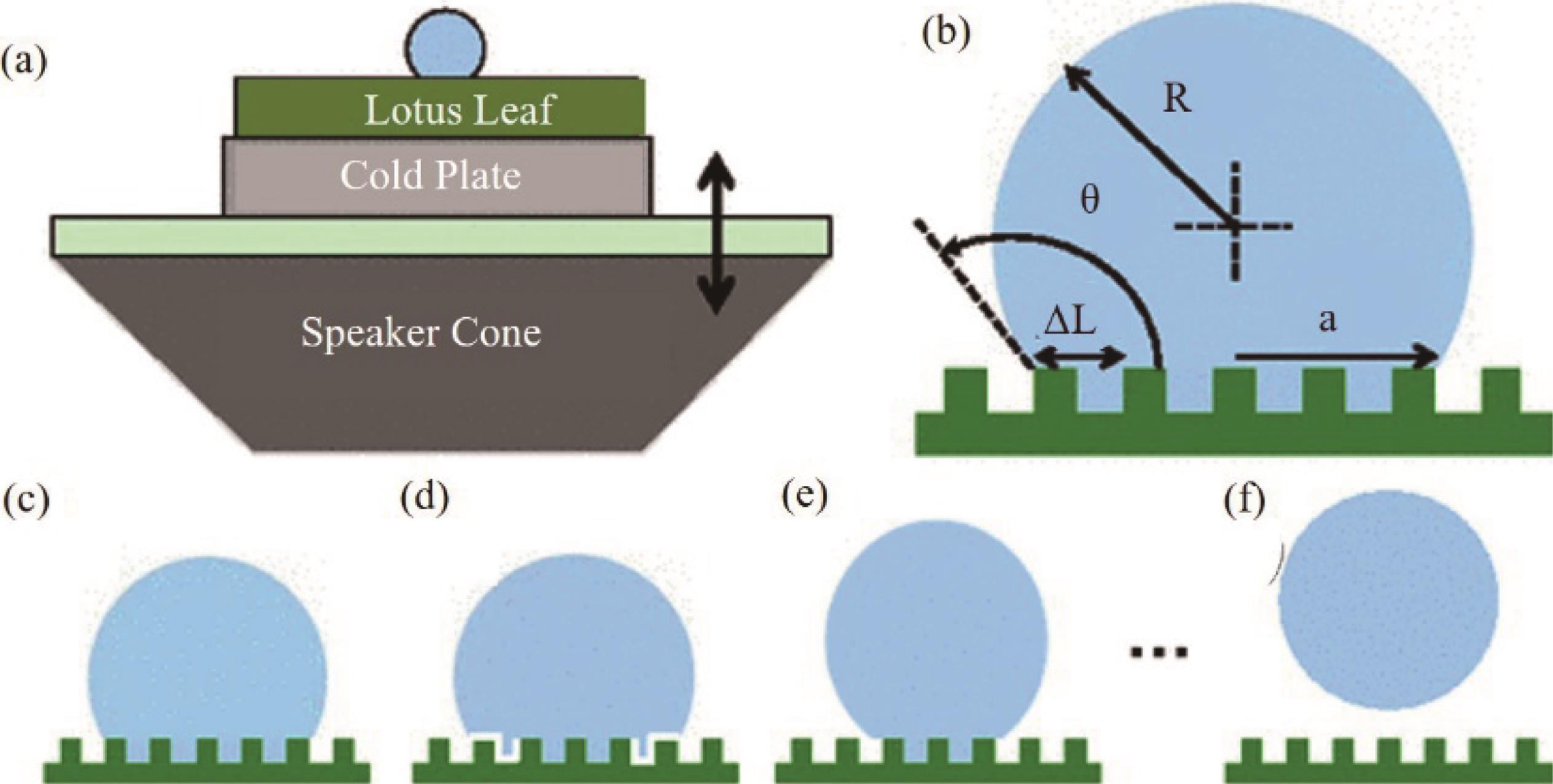
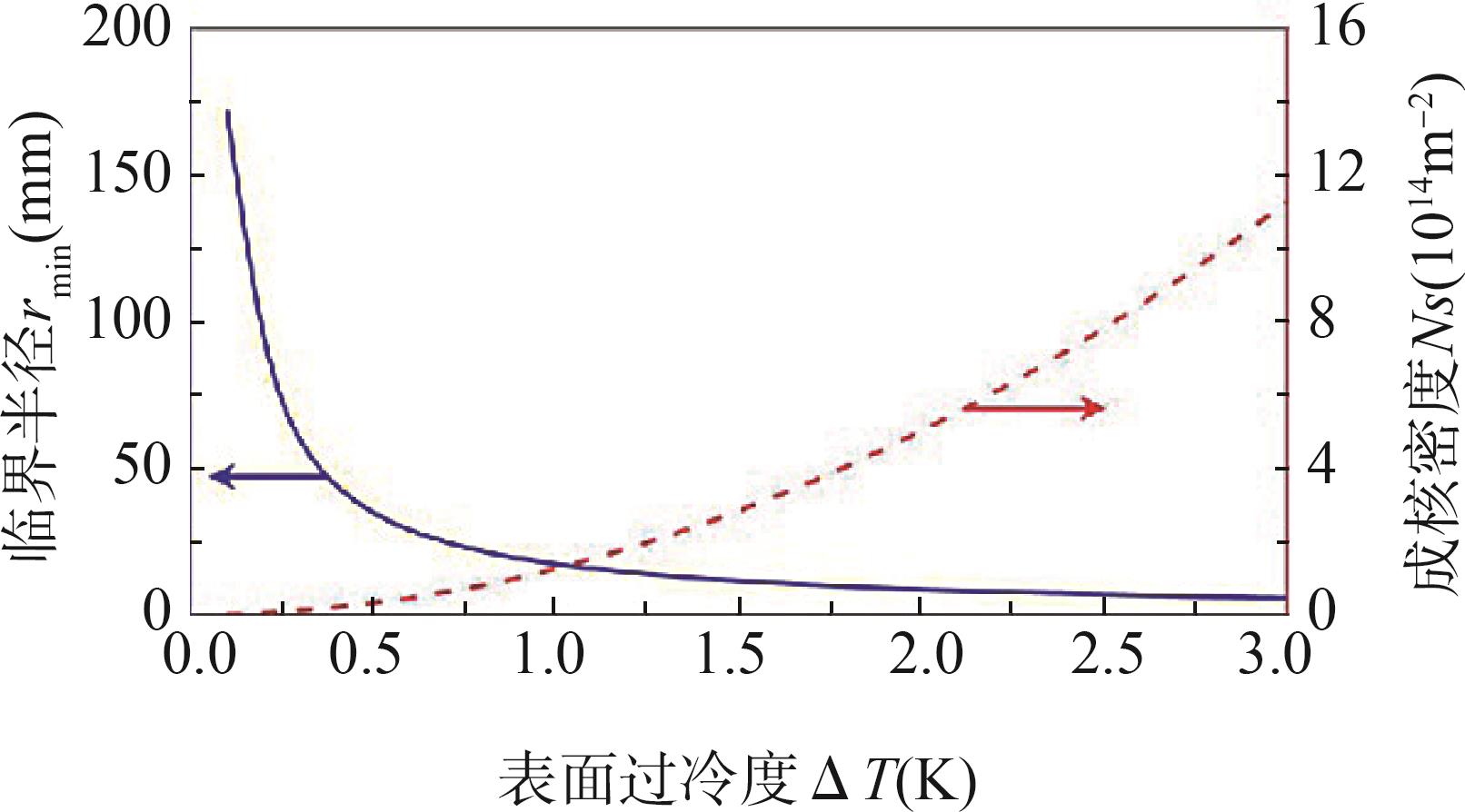
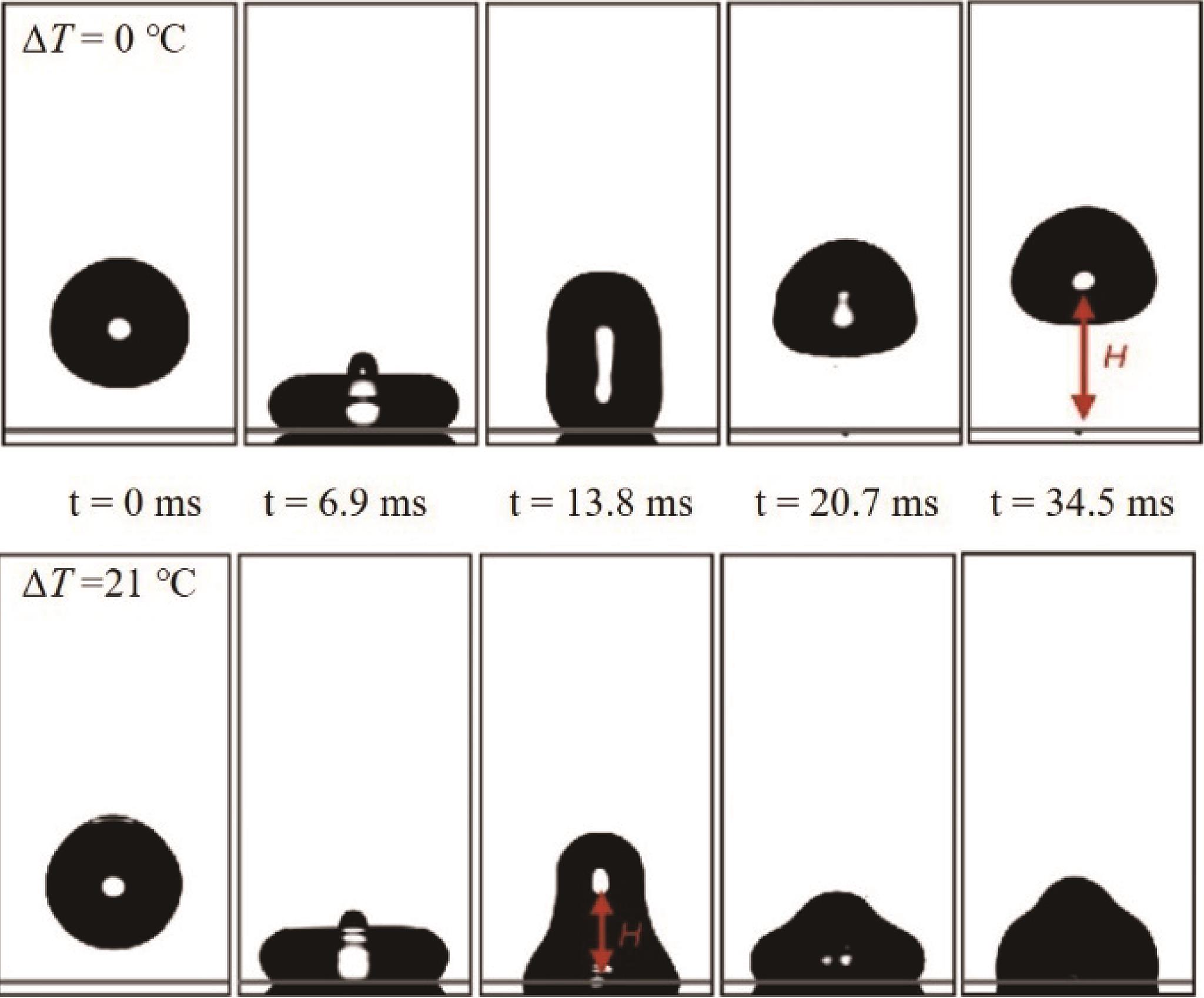
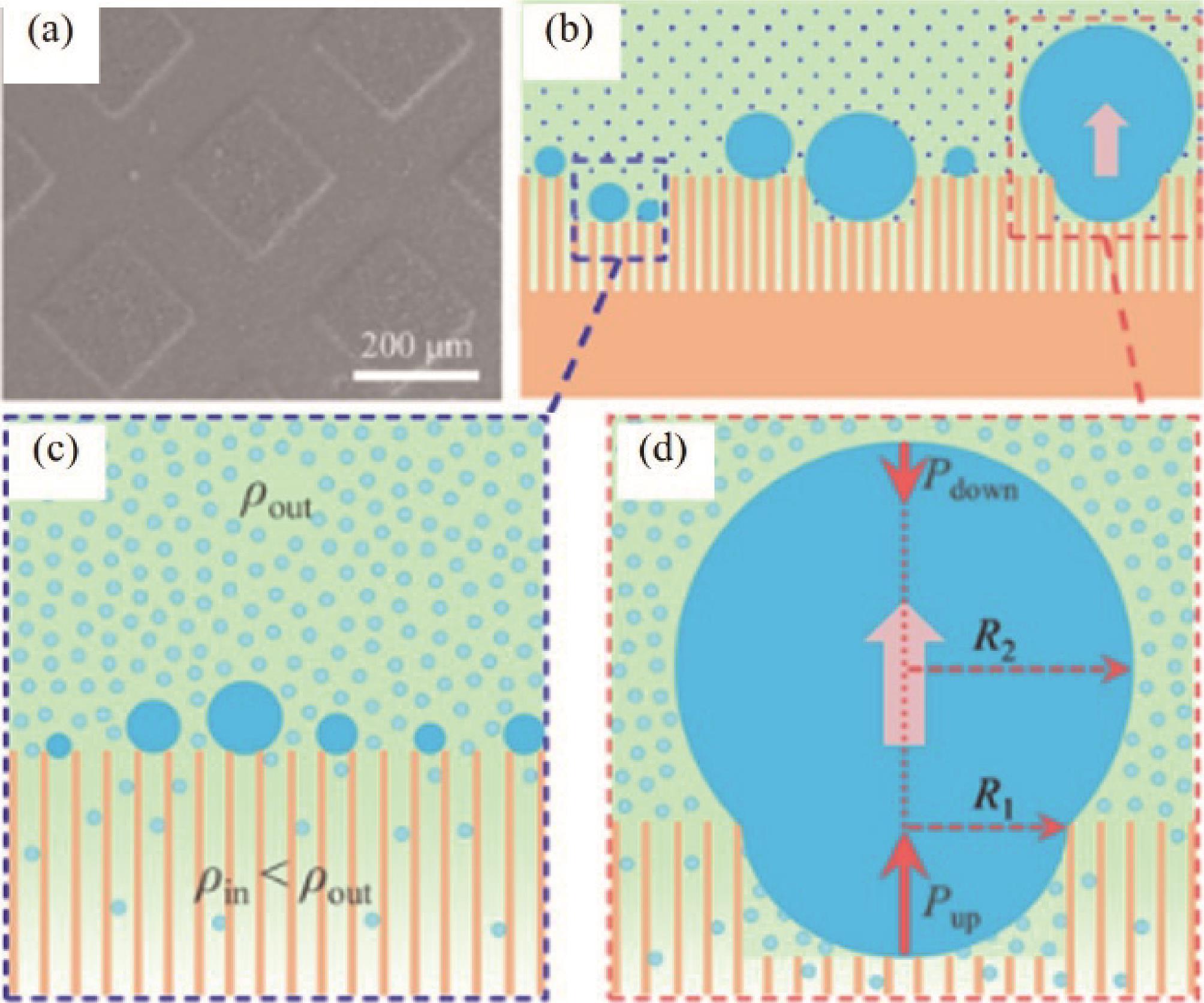
Fig.12 (a) Experimental setup where the cold plate was used only for the condensation experiments; (b) Parameters of the drop used in the model; (c - f) A hypothetical route for the gradual de-wetting during a Wenzel to Cassie transition[70]
| 1 | HENSEL R, NEINHUIS C, WERNER C. The springtail cuticle as a blueprint for omniphobic surfaces[J]. Chem Soc Rev, 2016, 45(2): 323-341. |
| 2 | KOCH K, BHUSHAN B, BARTHLOTT W. Multifunctional surface structures of plants: an inspiration for biomimetics[J]. Prog Mater Sci, 2009, 54(2): 137-178. |
| 3 | FENG L, LI S, LI Y, et al. Super-hydrophobic surfaces: from natural to artificial[J]. Adv Mater, 2002, 14(24): 1857-1860. |
| 4 | LIU K, JIANG L. Bio-inspired self-cleaning surfaces[J]. Annu Rev Mater Res, 2012, 42(1): 231-263. |
| 5 | LIU M, WANG S, JIANG L. Nature-inspired superwettability systems[J]. Nat Rev Mater, 2017, 2(7): 17036(1-17). |
| 6 | LIU M, WANG S, WEI Z, et al. Bioinspired design of a superoleophobic and low adhesive water/solid interface[J]. Adv Mater, 2009, 21(6): 665-669. |
| 7 | ZHAO Y, YU C, LAN H, et al. Improved interfacial floatability of superhydrophobic/superhydrophilic Janus sheet inspired by lotus leaf[J]. Adv Funct Mater, 2017, 27(27): 1701466. |
| 8 | WANG S, LIU K, YAO X, et al. Bioinspired surfaces with superwettability: new insight on theory, design, and applications[J]. Chem Rev, 2015, 115(16): 8230-8293. |
| 9 | BARTHLOTT W, NEINHUIS C. Purity of the sacred lotus, or escape from contamination in biological surfaces[J]. Planta, 1997, 202(1): 1-8. |
| 10 | NEINHUIS C B W. Characterization and distribution of water-repellent, self-cleaning plant surfaces[J]. Annu Botany, 1997, 79(6): 667-677. |
| 11 | QUÉRÉ D. Wetting and roughness[J]. Annu Rev Mater Res, 2008, 38(1): 71-99. |
| 12 | GEYER F, D'ACUNZI M, AGHILI A, et al. When and how self-cleaning of superhydrophobic surfaces works[J]. Sci Adv, 2020, 6(3): 9727. |
| 13 | LIANG Z, ZHOU Z, ZHAO L, et al. Fabrication of transparent, durable and self-cleaning superhydrophobic coatings for solar cells[J]. New J Chem, 2020, 44(34): 14481-14489. |
| 14 | CAO M, GUO D, YU C, et al. Water-repellent properties of superhydrophobic and lubricant-infused “slippery” surfaces: a brief study on the functions and applications[J]. ACS Appl Mater Interfaces, 2016, 8(6): 3615-3623. |
| 15 | CHI F, LIU D, WU H, et al. Mechanically robust and self-cleaning antireflection coatings from nanoscale binding of hydrophobic silica nanoparticles[J]. Solar Energy Mater Solar Cells, 2019, 200: 109939. |
| 16 | CAO Y, SALVINI A, CAMAITI M. One-step fabrication of robust and durable superamphiphobic, self-cleaning surface for outdoor and in situ application on building substrates[J]. J Colloid Interface Sci, 2021, 591: 239-252. |
| 17 | ZHU J, WU J. One-step fabrication of an anti-corrosion superhydrophobic surface on stainless steel[J]. Mater Res Express, 2020, 7(7): 076404. |
| 18 | HE S, SHI J, HUANG J, et al. Rational designed structured superhydrophobic iron oxide surface towards sustainable anti-corrosion and self-cleaning[J]. Chem Eng J, 2021, 416: 127768. |
| 19 | XIN G, WU C, LIU W, et al. Anti-corrosion superhydrophobic surfaces of Al alloy based on micro-protrusion array structure fabricated by laser direct writing[J]. J Alloys Compd, 2021, 881: 160649. |
| 20 | CHEN J, LIU Y, GUO D, et al. Under-water unidirectional air penetration via a Janus mesh[J]. Chem Commun (Camb), 2015, 51(59): 11872-11875. |
| 21 | LEE C, KIM C J. Underwater restoration and retention of gases on superhydrophobic surfaces for drag reduction[J]. Phys Rev Lett, 2011, 106(1): 014502. |
| 22 | XUE Z, CAO Y, LIU N, et al. Special wettable materials for oil/water separation[J]. J Mater Chem A, 2014, 2(8): 2445-2460. |
| 23 | WANG B, LIANG W, GUO Z, et al. Biomimetic super-lyophobic and super-lyophilic materials applied for oil/water separation: a new strategy beyond nature[J]. Chem Soc Rev, 2015, 44(1): 336-361. |
| 24 | MA Q, CHENG H, FANE A G, et al. Recent development of advanced materials with special wettability for selective oil/water separation [J]. Small, 2016, 12(16): 2186-2202. |
| 25 | TANG X, ZHU P, TIAN Y, et al. Mechano-regulated surface for manipulating liquid droplets[J]. Nat Commun, 2017, 8: 14831. |
| 26 | SUN Q, WANG D, LI Y, et al. Surface charge printing for programmed droplet transport[J]. Nat Mater, 2019, 18(9): 936-941. |
| 27 | DAI H, GAO C, SUN J, et al. Controllable high-speed electrostatic manipulation of water droplets on a superhydrophobic surface[J]. Adv Mater, 2019, 31(43): 1905449. |
| 28 | TU Y, WANG R, ZHANG Y, et al. Progress and expectation of atmospheric water harvesting[J]. Joule, 2018, 2(8): 1452-1475. |
| 29 | LI J, ZHU J, GAO X. Bio-inspired high-performance antireflection and antifogging polymer films[J]. Small, 2014, 10(13): 2578-2582. |
| 30 | MOUTERDE T, LEHOUCQ G, XAVIER S, et al. Antifogging abilities of model nanotextures[J]. Nat Mater, 2017, 16(6): 658-663. |
| 31 | HOU W, SHEN Y, TAO J, et al. Anti-icing performance of the superhydrophobic surface with micro-cubic array structures fabricated by plasma etching[J]. Colloids Surf A, 2020, 586: 124180. |
| 32 | QI Y, YANG Z, CHEN T, et al. Fabrication of superhydrophobic surface with desirable anti-icing performance based on micro/nano-structures and organosilane groups[J]. Appl Surf Sci, 2020, 501: 144165. |
| 33 | ZHU T, CHENG Y, HUANG J, et al. A transparent superhydrophobic coating with mechanochemical robustness for anti-icing, photocatalysis and self-cleaning[J]. Chem Eng J, 2020, 399: 125746. |
| 34 | SHEN Y, XIE X, XIE Y, et al. Statistically understanding the roles of nanostructure features in interfacial ice nucleation for enhancing icing delay performance[J]. Phys Chem Chem Phys, 2019, 21(36): 19785-19794. |
| 35 | BARTHWAL S, LIM S H. Rapid fabrication of a dual-scale micro-nanostructured superhydrophobic aluminum surface with delayed condensation and ice formation properties[J]. Soft Matter, 2019, 15(39): 7945-7955. |
| 36 | WANG L, GONG Q, ZHAN S, et al. Robust anti-icing performance of a flexible superhydrophobic surface[J]. Adv Mater, 2016, 28(35): 7729-7735. |
| 37 | HOU Y, YU M, SHANG Y, et al. Suppressing ice nucleation of supercooled condensate with biphilic topography[J]. Phys Rev Lett, 2018, 120(7): 075902. |
| 38 | YANG S, WU C, ZHAO G, et al. Condensation frosting and passive anti-frosting[J]. Cell Rep Phys Sci, 2021, 2(7): 100474. |
| 39 | WEN R, XU S, ZHAO D, et al. Hierarchical superhydrophobic surfaces with micropatterned nanowire arrays for high-efficiency jumping droplet condensation[J]. ACS Appl Mater Interfaces, 2017, 9(51): 44911-44921. |
| 40 | MILJKOVIC N, ENRIGHT R, WANG E. Effect of droplet morphology on growth dynamics and heat transfer during condensation on superhydrophobic nanostructured surfaces[J]. ACS Nano, 2012, 6(2): 1776-1785. |
| 41 | LAN Z, CHEN Y, HU S, et al. Droplet regulation and dropwise condensation heat transfer enhancement on hydrophobic-superhydrophobic hybrid surfaces[J]. Heat Transfer Eng, 2017, 39(17-18): 1540-1551. |
| 42 | WEN R, XU S, MA X, et al. Three-dimensional superhydrophobic nanowire networks for enhancing condensation heat transfer[J]. Joule, 2018, 2(2): 269-279. |
| 43 | LIU Y, CHOI C H. Condensation-induced wetting state and contact angle hysteresis on superhydrophobic lotus leaves[J]. Colloid Polym Sci, 2012, 291(2): 437-445. |
| 44 | SONG Z, LU M, CHEN X. Investigation of dropwise condensation heat transfer on laser-ablated superhydrophobic/hydrophilic hybrid copper surfaces[J]. ACS Omega, 2020, 5(37): 23588-23595. |
| 45 | ZHU Y, TSO C Y, HO T C, et al. Heat transfer enhancement on tube surfaces with biphilic nanomorphology[J]. Appl Therm Eng, 2020, 180: 115778. |
| 46 | ENSIKAT H J, DITSCHE-KURU P, NEINHUIS C, et al. Superhydrophobicity in perfection: the outstanding properties of the lotus leaf[J]. Beilstein J Nanotechnol, 2011, 2: 152-161. |
| 47 | R. D. NARHE D A B. Growth dynamics of water drops on a square-pattern rough hydrophobic surface[J]. Langmuir, 2007, 23: 6486-6489. |
| 48 | WILKE K L, PRESTON D J, LU Z, et al. Toward condensation-resistant omniphobic surfaces[J]. ACS Nano, 2018, 12(11): 11013-11021. |
| 49 | LIU T Q, SUN W, LI X Q, et al. Growth modes of condensates on nanotextured surfaces and mechanism of partially wetted droplet formation[J]. Acta Phys-Chim Sin, 2013, 29(8): 1762-1770. |
| 50 | FENG J, PANG Y, QIN Z, et al. Why condensate drops can spontaneously move away on some superhydrophobic surfaces but not on others[J]. ACS Appl Mater Interfaces, 2012, 4(12): 6618-6625. |
| 51 | CHENG Y T, RODAK D E. Is the lotus leaf superhydrophobic?[J]. Appl Phys Lett, 2005, 86(14): 144101. |
| 52 | SEO D, LEE C, NAM Y. Influence of geometric patterns of microstructured superhydrophobic surfaces on water-harvesting performance via dewing[J]. Langmuir, 2014, 30(51): 15468-15476. |
| 53 | YOUNG T. An essay on the cohesion of fliuds[J]. Philos Trans R S London, 1805, 95: 65-87. |
| 54 | WENZEL R N. Resistance of solid surfaces to wetting by water[J]. Ind Eng Chem, 1936, 28(8): 988-994. |
| 55 | CASSIE A, BAXTER S. Wettability of porous surfaces[J]. Trans Faraday Soc, 1944, 40: 546-51. |
| 56 | WEN R, LAN Z, PENG B, et al. Wetting transition of condensed droplets on nanostructured superhydrophobic surfaces: coordination of surface properties and condensing conditions[J]. ACS Appl Mater Interfaces, 2017, 9(15): 13770-13777. |
| 57 | VARANASI K K, HSU M, BHATE N, et al. Spatial control in the heterogeneous nucleation of water[J]. Appl Phys Lett, 2009, 95(9): 094101. |
| 58 | BROWN R A, ORR F M Jr, SCRIVEN L E. Static drop on an inclined plate analysis by the finite element method[J]. J Collods Interface Sci, 1980, 73(1): 76-87. |
| 59 | EXTRAND C W, GENT A N. Retention of liquid drops by solid surfaces[J]. J Collods Interface Sci, 1990, 138(2): 431-442. |
| 60 | MOUTERDE T, LECOINTRE P, LEHOUCQ G, et al. Two recipes for repelling hot water[J]. Nat Commun, 2019, 10(1): 1410. |
| 61 | PAXSON A T, YAGUE J L, GLEASON K K, et al. Stable dropwise condensation for enhancing heat transfer via the initiated chemical vapor deposition (iCVD) of grafted polymer films[J]. Adv Mater, 2014, 26(3): 418-423. |
| 62 | CHO H J, PRESTON D J, ZHU Y, et al. Nanoengineered materials for liquid-vapour phase-change heat transfer[J]. Nat Rev Mater, 2016, 2(2): 16092. |
| 63 | SHANG Y, HOU Y, YU M, et al. Modeling and optimization of condensation heat transfer at biphilic interface[J]. Int J Heat Mass Transfer, 2018, 122: 117-127. |
| 64 | MILJKOVIC N, ENRIGHT R, NAM Y, et al. Jumping-droplet-enhanced condensation on scalable superhydrophobic nanostructured surfaces[J]. Nano Lett, 2013, 13(1): 179-187. |
| 65 | CHEN X, WU J, MA R, et al. Nanograssed micropyramidal architectures for continuous dropwise condensation[J]. Adv Funct Mater, 2011, 21(24): 4617-4623. |
| 66 | WANG S, LIU M, FENG Y, et al. Bioinspired hierarchical copper oxide surfaces for rapid dropwise condensation[J]. J Mater Chem A, 2017, 5(40): 21422-21428. |
| 67 | ZHAO G, ZOU G, WANG W, et al. Competing effects between condensation and self-removal of water droplets determine antifrosting performance of superhydrophobic surfaces[J]. ACS Appl Mater Interfaces, 2020, 12(6): 7805-7814. |
| 68 | TANG Y, YANG X, LI Y, et al. Robust micro-nanostructured superhydrophobic surfaces for long-term dropwise condensation[J]. Nano Lett, 2021, DOI: 10.1021/acs.nanolett.1c01584. |
| 69 | HOU Y, SHANG Y, YU M, et al. Tunable water harvesting surfaces consisting of biphilic nanoscale topography[J]. ACS Nano, 2018, 12(11): 11022-11030. |
| 70 | BOREYKO J B, CHEN C H. Restoring superhydrophobicity of lotus leaves with vibration-induced dewetting[J]. Phys Rev Lett, 2009, 103(17): 174502. |
| 71 | ZHANG L, SHI J, XU B, et al. Experimental study on distribution characteristics of condensate droplets under ultrasonic vibration[J]. Microgravity Sci Technol, 2018, 30(6): 737-746. |
| 72 | MORADI M, CHINI S F, RAHIMIAN M H. Vibration-enhanced condensation heat transfer on superhydrophobic surfaces: an experimental study[J]. AIP Adv, 2020, 10(9): 095123(1-6). |
| [1] | DONG Yilin, ZHANG Hao, CHEN Jinlong. Preparation of Cosmetic-Grade Sodium Polyaspartate [J]. Chinese Journal of Applied Chemistry, 2020, 37(8): 883-888. |
| [2] | CHEN Shaorui, SHEN Fengjuan. Synthesis of 4-Methyl-4-aza-21E-benzylidene-5-pregnene-3,20-dione [J]. Chinese Journal of Applied Chemistry, 2017, 34(6): 671-675. |
| [3] | HE Qiangfang, WU Yinghong, CAI Zhijian, XIE Wang. Synthesis of Fluorescent Cross-linked Stabilized Polymeric Micelles Based on Salicylidene Schiff Base/Zn2+ Complexes and Sensor for Cu2+ Detection [J]. Chinese Journal of Applied Chemistry, 2016, 33(6): 701-709. |
| [4] | XU Zhaohui,LIU Deyong,TU Yuanhong. Efficient Solvent-free Synthesis of Spiro Heterobicyclic Derivatives with Boric Acid as Catalyst [J]. Chinese Journal of Applied Chemistry, 2015, 32(9): 999-1004. |
| [5] | XU Zhaohui, LIN Chunhua, LIAO Weilin. Efficient One-Pot Synthesis of 5-[(Indol-3-yl)-methyl]-2,2-dimethyl-1,3-dioxane-4,6-dione Derivatives [J]. Chinese Journal of Applied Chemistry, 2015, 32(3): 278-283. |
| [6] | LYU Chengwei, LIU Yanhang, WANG Jiajing, ZHOU Xiaoxia. Potassium Dihydrogen Phosphate Catalyzed Yonemitsu Condensation for Synthesis of 5-[(Indol-3-yl)-arylmethyl]-2,2-dimethyl-1,3-dioxane- 4,6-dione Derivatives [J]. Chinese Journal of Applied Chemistry, 2015, 32(12): 1371-1378. |
| [7] | ZHANG Baohua, SHI Lanxiang*. Asymmetric Henry Condensation Catalyzed by Ionic Liquid-supported Proline [J]. Chinese Journal of Applied Chemistry, 2014, 31(07): 812-816. |
| [8] | CHEN Xiong, LIU Jinyun, LI Yabai, LIU Jianfu, YANG Huawu*. Synthesis of Mintlactone via Cross-Aldol Condensation Between 3-Methylcyclohexanone and Ethyl Pyruvate Using Ferric Chloride Hexahydrate as Catalyst [J]. Chinese Journal of Applied Chemistry, 2013, 30(09): 1005-1009. |
| [9] | HE Jinmei*, QU Mengnan, HE Zhongsen. One Pot Continuous Reaction of Benzoin Condensation and Allylic Nucleophilic Substitution Using N-Heterocyclic Carbenes and Nickel(Ⅱ) Chloride as Catalysts [J]. Chinese Journal of Applied Chemistry, 2013, 30(07): 852-854. |
| [10] | XIA Jingjing *, ZHANG Kehua. Synthesis of N-Substituted Acridinedione Derivatives in Refluxing Aqueous Phase [J]. Chinese Journal of Applied Chemistry, 2013, 30(02): 242-244. |
| [11] | REN Jie*, LIAO Ruirui, YANG Wu, LI Yan, GAO Jinzhang. Fabrication of Superhydrophobic Iron-surface by Using Glow Discharge Electrolysis Plasma [J]. Chinese Journal of Applied Chemistry, 2013, 30(02): 208-213. |
| [12] | LIU Changhui, WEN Ruiming*, YE Xiaoqin, YI Xianwen, FANG Lei. Synthesis and Characterization of Ionone-containing Chalcones [J]. Chinese Journal of Applied Chemistry, 2012, 29(07): 780-783. |
| [13] | WANG Yu, LU Ming*. Modified Synthesis of Four Kinds of Azabicyclo Compounds [J]. Chinese Journal of Applied Chemistry, 2012, 29(04): 397-401. |
| [14] | TANG Huaijun1,2*, ZHANG Meifang2, ZHANG Zhiguo2, SHI Qing2, ZHANG Keli2*. Synthesis and Characterization of Two β-Diketones with Carbazole Group [J]. Chinese Journal of Applied Chemistry, 2011, 28(11): 1239-1243. |
| [15] | LI Hongfei*, ZHANG Liangze, WU Dingming, MU Weining, ZHOU Juntao. Synthesis of 6-Chlorin-8-alkyl-9-aryl-purine Derivatives [J]. Chinese Journal of Applied Chemistry, 2011, 28(07): 747-752. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||