
Chinese Journal of Applied Chemistry ›› 2024, Vol. 41 ›› Issue (2): 217-229.DOI: 10.19894/j.issn.1000-0518.230310
• Review • Previous Articles
Research Prospect of Single-Atom Catalysts for Fenton-Like Water Treatment
Tian-Li SUN1, Guo ZHU1, Hai HE1, Bing-Kun HUANG2, Zhao-Kun XIONG2( ), Bo LAI2(
), Bo LAI2( )
)
- 1.The Second Gas Production Plant of Southwest Branch,Sinopec,Langzhong 637400,China
2.Sino-German Centre for Water and Health Research,College of Architecture and Environment,Sichuan University,Chengdu 610065,China
-
Received:2023-10-10Accepted:2023-12-24Published:2024-02-01Online:2024-03-05 -
Contact:Zhao-Kun XIONG,Bo LAI -
About author:laibo@scu.edu.cn
scuxzk@scu.edu.cn;
-
Supported by:the National Natural Science Foundation of China(52200105);the Natural Science Foundation of Sichuan Province(2023NSFSC0344)
CLC Number:
Cite this article
Tian-Li SUN, Guo ZHU, Hai HE, Bing-Kun HUANG, Zhao-Kun XIONG, Bo LAI. Research Prospect of Single-Atom Catalysts for Fenton-Like Water Treatment[J]. Chinese Journal of Applied Chemistry, 2024, 41(2): 217-229.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.230310

Fig.1 (a) High-angle annular dark field scanning transmission electron microscopy (HAADF-STEM) images and high-resolution HAADF-STEM images (insets) of Pd-nanoparticles@ZIF-8, intermediate Ⅰ, intermediate Ⅱ and Pd single atoms[24]; (b) Schematic illustrations of Pt ALD mechanism on graphene nanosheets[27]
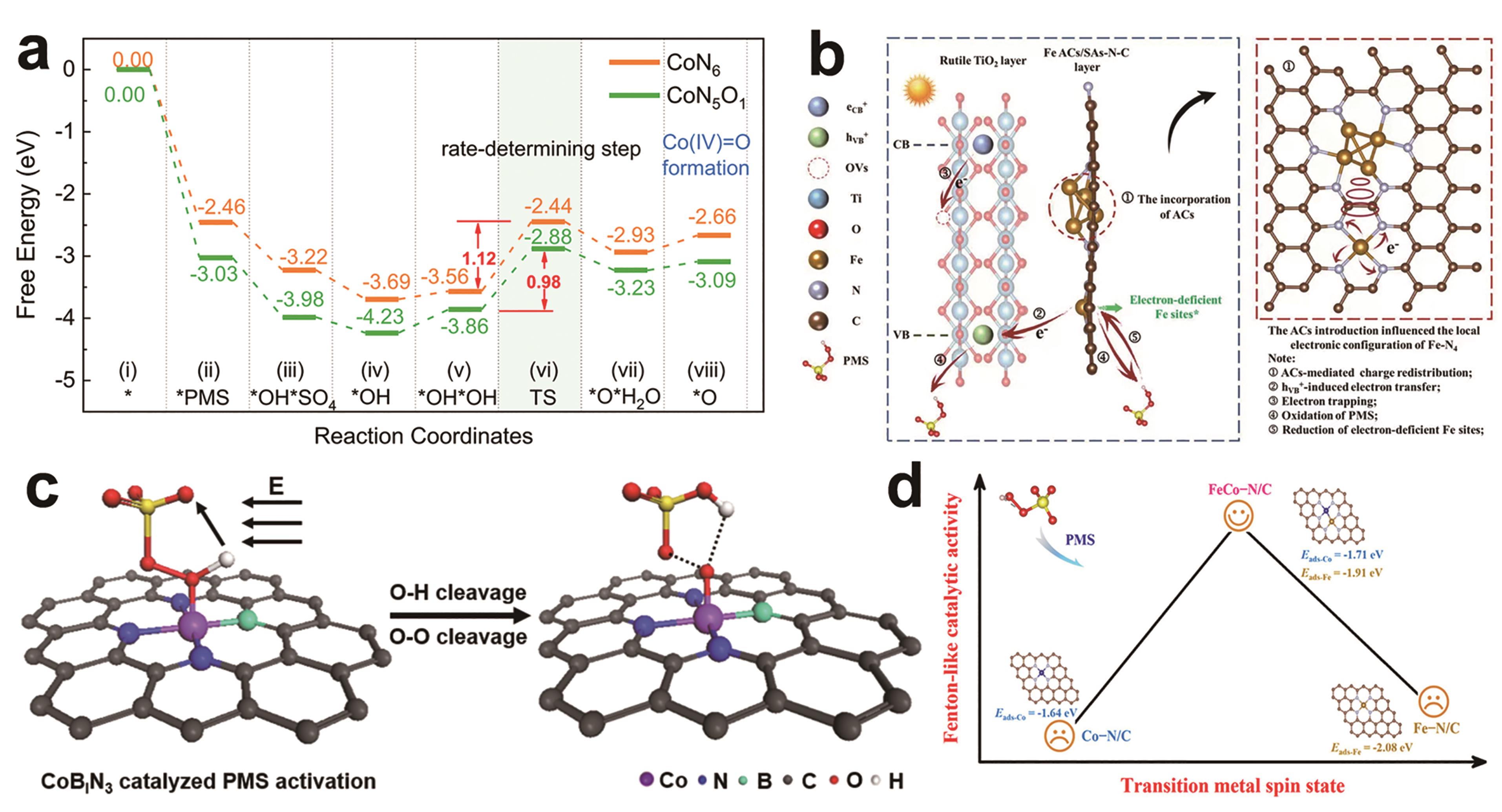
Fig.3 (a) Energy profiles of Co(Ⅳ)O formation for CoN6/PMS and CoN5O1/PMS systems; *O represents the reactive species of Co(?Ⅳ??)?O[36]; (b) The proposed reaction mechanisms of Vis/TiFeAS/PMS system[37]; (c) Schematic illustration of a local electric field-induced coupled electron-proton transfer process promoting the conversion of high-valent metal-oxo species[38]; (d) Relationship between spin states and Fenton-like catalytic activity of transition metals[39]
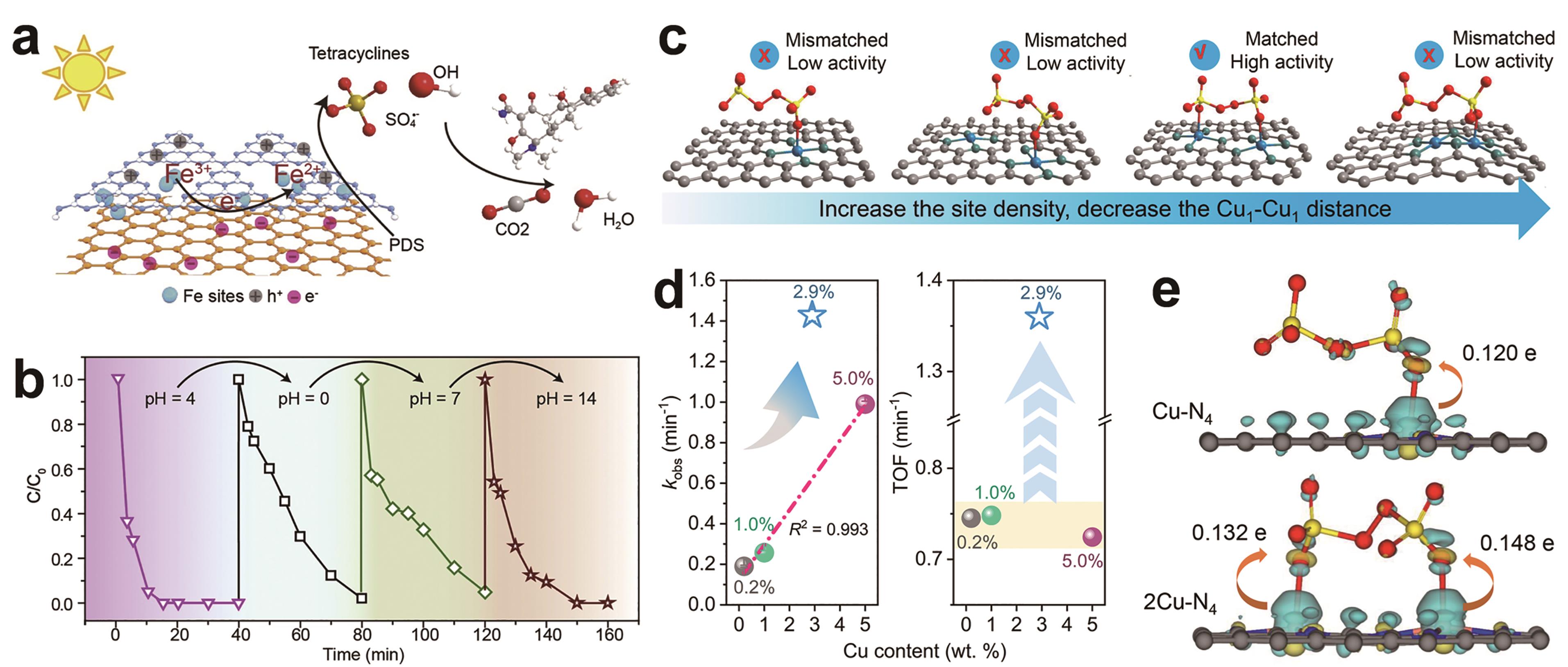
Fig.4 (a) Scheme diagram proposed for the Fenton-like reaction mechanism of C3N4-Fe-rGO/PDS[4]?; (b) The degradation performance during cycles of solution with different pH conditions, with C3N4-Fe-rGO as the catalyst[4]; (c) Depiction of Cu1-Cu1 distance manipulation for PDS adsorption and activation[41]; (d) First-order rate constants and TOFs of BPA removal by Cu1/NG with different Cu site densities activating PDS[41]??; (e) Electron density difference for PDS adsorption on 2Cu-N4 and Cu-N4 and the corresponding charge transfer. Yellow and cyan contours stand for electron accumulation and deletion, respectively[41]

Fig.5 (a) Photo and SEM image of the filter medium. Scale bar: 100 μm. Inset, magnified SEM image showing the Cu-C3N4 catalyst coated on the surface of a carbon fibre. Scale bar: 5 μm[44]?; (b) Dye removal and Cu concentration in effluent as functions of filtration time[44]?; (c) Charge density differences of FeN5 and FeN4 models. The yellow and skyblue regions represent electron accumulation and electron depletion, respectively[45]; (d) Energy diagram of the reaction process for FeN5 and FeN4 models[45]?; (e) Degradation of select organic pollutants in FeN4/NG+H2O2, FeN5/NG+H2O2, conventional homogeneous Fenton (Fe2++H2O2) and control (H2O2) systems[45]

Fig.6 (a) Comparison between the apparent rate constants of g-C3N4 and FeCN (inset: TOC result)?[49]; (b) Full-scan chromatogram of CH3COO?-TEMPO[49]; (c) Mass spectral analyses of the 18O-labeled or unlabeled PMSO2 generated in the FeCN+PAA system[49]; (d) Degradation of 4-CP by IO4- activated using different activators[50]; (e) Initial solution pH on the degradation of 4-CP in the N-rGO-CoSA IO4- system[50]; (f) Current response after the sequential addition of IO4- and 4-CP at the working electrode coated with the N-rGO-CoSA powder. The inset shows the EIS profiles of the N-rGO-CoSA and N-rGO-800 electrodes in the electrolyte solution[50]; (g) Substrate dependence diagram in Fe5-NC/O3 system[51]; (h) Dynamic processes simulated by AIMD[51]; (i) Relative energy profile calculated by DFT in the interaction of O3 and the Fe-N4 site[51]
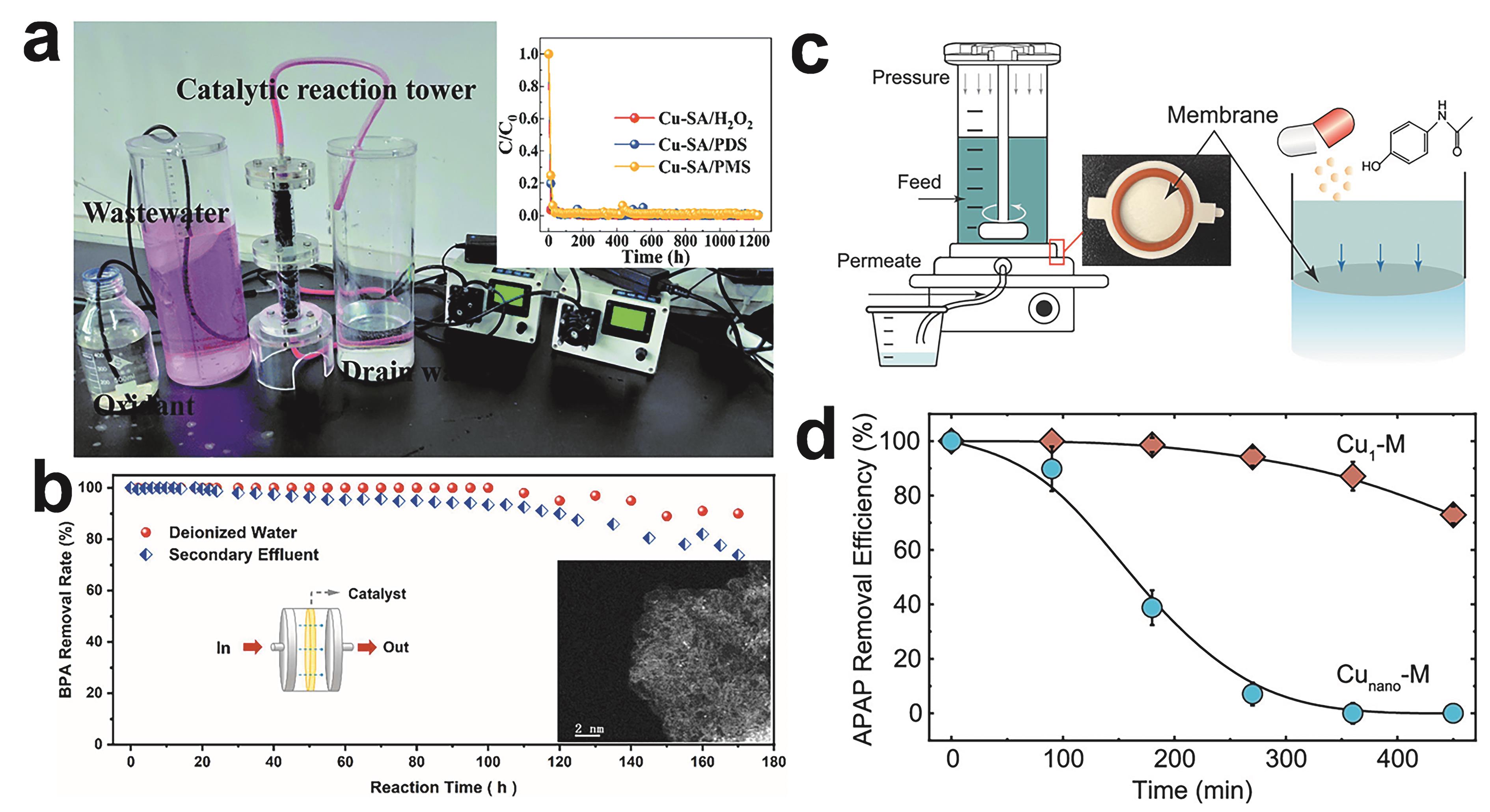
Fig.7 (a) Pilot device and effect of Cu-SA in continuous flow unit[55]; (b) BPA removal in deionized water and secondary effluent of WWTP with Fe-CNW3 coated membrane filter under continuous flow. Inset is the HADDF-STEM image of Fe-CNW3 after the reaction[59]; (c) Schematic and photograph of the catalytic membrane (effective area of 3.1 cm2) are installed and the dead-end filtration cell for micropollutant removal. APAP is selected as a model micropollutant[60]; (c) APAP removal efficiency by the catalytic membranes during filtration[60]
| 1 | HUANG L Z, WEI X, GAO E, et al. Single Fe atoms confined in two-dimensional MoS2 for sulfite activation: a biomimetic approach towards efficient radical generation[J]. Appl Catal B: Environ, 2020, 268: 118459. |
| 2 | HUANG B, XIONG Z, ZHOU P, et al. Ultrafast degradation of contaminants in a trace cobalt(Ⅱ) activated peroxymonosulfate process triggered through borate: indispensable role of intermediate complex[J]. J Hazard Mater, 2022, 424: 127641. |
| 3 | CHENG Y, HE S, LU S, et al. Iron single atoms on graphene as nonprecious metal catalysts for high-temperature polymer electrolyte membrane fuel cells[J]. Adv Sci, 2019, 6(10): 1802066. |
| 4 | ZUO S, JIN X, WANG X, et al. Sandwich structure stabilized atomic Fe catalyst for highly efficient Fenton-like reaction at all pH values[J]. Appl Catal B: Environ, 2021, 282: 119551. |
| 5 | QIAO B, WANG A, YANG X, et al. Single-atom catalysis of CO oxidation using Pt1/FeOx[J]. Nat Chem, 2011, 3(8): 634-641. |
| 6 | 曹蓉, 夏杰桢, 廖漫华, 等. 单原子催化剂在电化学合成氨中的理论研究进展[J]. 应用化学, 2023, 40(1): 9-23. |
| CAO R, XIA J Z, LIAO M H, et al. Theoretical research progress of single atom catalysts in electrochemical synthesis of ammonia[J]. Chin J Appl Chem, 2023, 40(1): 9-23. | |
| 7 | 张超. 单原子催化剂电催化还原二氧化碳研究进展[J]. 应用化学, 2022, 39(6): 871-887. |
| ZHANG C. Research prospect of single atom catalysts towards electrocatalytic reduction of carbon dioxide[J]. Chin J Appl Chem, 2022, 39(6): 871-887. | |
| 8 | 王欣, 张冬, 杜菲. 单原子催化剂在锂硫电池中的研究进展[J]. 应用化学, 2022, 39(4): 513-527. |
| WANG X, ZHANG D, DU F. Recent progress of single⁃atom catalytic materials for lithium sulfur batteries[J]. Chin J Appl Chem, 2022, 39(4): 513-527. | |
| 9 | WEON S, HUANG D, RIGBY K, et al. Environmental materials beyond and below the nanoscale: single-atom catalysts[J]. ACS EST Eng, 2020, 1(2): 157-172. |
| 10 | WU X, KIM J. Outlook on single atom catalysts for persulfate-based advanced oxidation[J]. ACS EST Eng, 2022, 2(10): 1776-1796. |
| 11 | HUANG B, WU Z, ZHOU H, et al. Recent advances in single-atom catalysts for advanced oxidation processes in water purification[J]. J Hazard Mater, 2021, 412: 125253. |
| 12 | LI W H, YANG J, WANG D, et al. Striding the threshold of an atom era of organic synthesis by single-atom catalysis[J]. Chem, 2022, 8(1): 119-140. |
| 13 | YAN H, SU C, HE J, et al. Single-atom catalysts and their applications in organic chemistry[J]. J Mater Chem A, 2018, 6(19): 8793-8814. |
| 14 | GIANNAKAKIS G, MITCHELL S, PÉREZ-RAMíREZ J. Single-atom heterogeneous catalysts for sustainable organic synthesis[J]. Trends Chem, 2022, 4(4): 264-276. |
| 15 | LIU Y, YAO M, HAN W, et al. Construction of a single-atom nanozyme for enhanced chemodynamic therapy and chemotherapy[J]. Chem Eur J, 2021, 27(53): 13418-13425. |
| 16 | LU X, GAO S, LIN H, et al. Bioinspired copper single-atom catalysts for tumor parallel catalytic therapy[J]. Adv Mater, 2020, 32(36): 2002246. |
| 17 | LIANG J, JOHANNESSEN B, WU Z, et al. Regulating the coordination environment of mesopore-confined single atoms from metalloprotein-MOFs for highly efficient biocatalysis[J]. Adv Mater, 2022, 34(44): 2205674. |
| 18 | JIAO L, YAN H, WU Y, et al. When nanozymes meet single-atom catalysis[J]. Angew Chem Int Ed, 2020, 59(7): 2565-2576. |
| 19 | DOHERTY F, WANG H, YANG M, et al. Nanocluster and single-atom catalysts for thermocatalytic conversion of CO and CO2[J]. Catal Sci Technol, 2020, 10(17): 5772-5791. |
| 20 | LIU L, CORMA A. Metal catalysts for heterogeneous catalysis: from single atoms to nanoclusters and nanoparticles[J]. Chem Rev, 2018, 118(10): 4981-5079. |
| 21 | ZHANG H, TIAN W, DUAN X, et al. Catalysis of a single transition metal site for water oxidation: from mononuclear molecules to single atoms[J]. Adv Mater, 2020, 32(18): 1904037. |
| 22 | REN W, CHENG C, SHAO P, et al. Origins of electron-transfer regime in persulfate-based nonradical oxidation processes[J]. Environ Sci Technol, 2022, 56(1): 78-97. |
| 23 | SHANG Y, XU X, GAO B, et al. Single-atom catalysis in advanced oxidation processes for environmental remediation[J]. Chem Soc Rev, 2021, 50(8): 5281-322. |
| 24 | WEI S, LI A, LIU J C, et al. Direct observation of noble metal nanoparticles transforming to thermally stable single atoms[J]. Nat Nanotechnol, 2018, 13(9): 856-861. |
| 25 | YIN P, YAO T, WU Y, et al. Single cobalt atoms with precise N-coordination as superior oxygen reduction reaction catalysts[J]. Angew Chem Int Ed, 2016, 55(36): 10800. |
| 26 | HAN A, WANG B, KUMAR A, et al. Recent advances for MOF-derived carbon-supported single-atom catalysts[J]. Small Methods, 2019, 3(9): 1800471. |
| 27 | SUN S, ZHANG G, GAUQUELIN N, et al. Single-atom catalysis using Pt/graphene achieved through atomic layer deposition[J]. Sci Rep, 2013, 3(1): 1775. |
| 28 | DUAN S, WANG R, LIU J. Stability investigation of a high number density Pt1/Fe2O3 single-atom catalyst under different gas environments by HAADF-STEM[J]. Nanotechnology, 2018, 29(20): 204002. |
| 29 | XIONG Y, SUN W, XIN P, et al. Gram-scale synthesis of high-loading single-atomic-site Fe catalysts for effective epoxidation of styrene[J]. Adv Mater, 2020: e2000896. |
| 30 | HUANG B, WU Z, WANG X, et al. Coupled surface-confinement effect and pore engineering in a single-Fe-atom catalyst for ultrafast Fenton-like reaction with high-valent iron-oxo complex oxidation[J]. Environ Sci Technol, 2023, 57(41): 15667-15679. |
| 31 | LI X, CAO C S, HUNG S F, et al. Identification of the electronic and structural dynamics of catalytic centers in single-Fe-atom material[J]. Chem, 2020, 6(12): 3440-3454. |
| 32 | HUANG B, REN X, ZHAO J, et al. Modulating electronic structure engineering of atomically dispersed cobalt catalyst in Fenton-like reaction for efficient degradation of organic pollutants[J]. Environ Sci Technol, 2023, 57(37): 14071-14081. |
| 33 | LAI L, YAN J, LI J, et al. Co/Al2O3-EPM as peroxymonosulfate activator for sulfamethoxazole removal: performance, biotoxicity, degradation pathways and mechanism[J]. Chem Eng J, 2018, 343: 676-688. |
| 34 | XIONG Z, JIANG Y, WU Z, et al. Synthesis strategies and emerging mechanisms of metal-organic frameworks for sulfate radical-based advanced oxidation process: a review[J]. Chem Eng J, 2021, 421: 127863. |
| 35 | LI X, HUANG X, XI S, et al. Single cobalt atoms anchored on porous N-doped graphene with dual reaction sites for efficient Fenton-like catalysis[J]. J Am Chem Soc, 2018, 140(39): 12469-12475. |
| 36 | WU Q Y, YANG Z W, WANG Z W, et al. Oxygen doping of cobalt-single-atom coordination enhances peroxymonosulfate activation and high-valent cobalt-oxo species formation[J]. Proc Natl Acad Sci USA, 2023, 120(16): e2219923120. |
| 37 | MO F, SONG C, ZHOU Q, et al. The optimized Fenton-like activity of Fe single-atom sites by Fe atomic clusters-mediated electronic configuration modulation[J]. Proc Natl Acad Sci USA, 2023, 120(15): e2300281120. |
| 38 | SONG J, HOU N, LIU X, et al. Asymmetrically coordinated CoB1N3 moieties for selective generation of high-valent co-oxo species via coupled electron-proton transfer in Fenton-like reactions[J]. Adv Mater, 2023: e2209552. |
| 39 | ZHAO Z, HU M, NIE T, et al. Improved electronic structure from spin-state reconstruction of a heteronuclear Fe-Co diatomic pair to boost the Fenton-like reaction[J]. Environ Sci Technol, 2023, 57(11): 4556-4567. |
| 40 | JIANG N, XU H, WANG L, et al. Nonradical oxidation of pollutants with single-atom-Fe(Ⅲ)-activated persulfate: Fe(V) being the possible intermediate oxidant[J]. Environ Sci Technol, 2020, 54(21): 14057-14065. |
| 41 | WANG B, CHENG C, JIN M, et al. A site distance effect induced by reactant molecule matchup in single-atom catalysts for Fenton-like reactions[J]. Angew Chem Int Ed Engl, 2022, 61(33): e202207268. |
| 42 | FU H, WEI J, CHEN G, et al. Axial coordination tuning Fe single-atom catalysts for boosting H2O2 activation[J]. Appl Catal B: Environ, 2023, 321: 122012. |
| 43 | ZHANG C, BAI L, CHEN M, et al. Modulating the site density of Mo single atoms to catch adventitious O atoms for efficient H2O2 oxidation with light[J]. Adv Mater, 2023, 35(5): e2208704. |
| 44 | XU J, ZHENG X, FENG Z, et al. Organic wastewater treatment by a single-atom catalyst and electrolytically produced H2O2[J]. Nat Sustainable, 2021, 4: 233-241. |
| 45 | FU H, WEI J, CHEN G, et al. Axial coordination tuning Fe single-atom catalysts for boosting H2O2 activation[J]. Appl Catal B, 2023, 321: 122012. |
| 46 | AN S, ZHANG G, WANG T, et al. High-density ultra-small clusters and single-atom Fe sites embedded in graphitic carbon nitride (g-C3N4) for highly efficient catalytic advanced oxidation processes[J]. ACS Nano, 2018, 12(9): 9441-9450. |
| 47 | ZHOU P, ZHANG J, XIONG Z, et al. C60 Fullerol promoted Fe(Ⅲ)/H2O2 Fenton oxidation: role of photosensitive Fe(Ⅲ)-Fullerol complex[J]. Appl Catal B: Environ, 2020, 265: 118264. |
| 48 | SHENG B, DENG C, LI Y, et al. In situ hydroxylation of a single-atom iron catalyst for preferential 1O2 production from H2O2[J]. ACS Catal, 2022, 12(23): 14679-14688. |
| 49 | CHEN F, LIU L L, WU J H, et al. Single-atom iron anchored tubular g-C3N4 catalysts for ultrafast Fenton-like reaction: roles of high-valency iron-oxo species and organic radicals[J]. Adv Mater, 2022, 34(31): 2202891. |
| 50 | LONG Y, DAI J, ZHAO S, et al. Atomically dispersed cobalt sites on graphene as efficient periodate activators for selective organic pollutant degradation[J]. Environ Sci Technol, 2021, 55: 5357-5370. |
| 51 | REN T, YIN M, CHEN S, et al. Single-atom Fe-N4 sites for catalytic ozonation to selectively induce a nonradical pathway toward wastewater purification[J]. Environ Sci Technol, 2023, 57(9): 3623-3633. |
| 52 | GUO Z, XIE Y, XIAO J, et al. Single-atom Mn-N4 site-catalyzed peroxone reaction for the efficient production of hydroxyl radicals in an acidic solution[J]. J Am Chem Soc, 2019, 141(30): 12005-12010. |
| 53 | LAN S, JING B, YU C, et al. Protrudent iron single-atom accelerated interfacial piezoelectric polarization for self-powered water motion triggered Fenton-like reaction[J]. Small, 2022, 18(2): 2105279. |
| 54 | WANG Z, ALMATRAFI E, WANG H, et al. Cobalt single atoms anchored on oxygen-doped tubular carbon nitride for efficient peroxymonosulfate activation: simultaneous coordination structure and morphology modulation[J]. Angew Chem Int Ed, 2022, 61(29): e202202338. |
| 55 | ZUO S, GUAN Z, YANG F, et al. Reactive oxygen species regulation and synergistic effect for effective water purification through Fenton-like catalysis on single-atom Cu-N sites[J]. J Mater Chem A, 2022, 10(19): 10503-10513. |
| 56 | LIANG X, WANG D, ZHAO Z, et al. Coordination number dependent catalytic activity of single-atom cobalt catalysts for Fenton-like reaction[J]. Adv Funct Mater, 2022, 32: 2203001. |
| 57 | CHU C, YANG J, ZHOU X, et al. Cobalt single atoms on tetrapyridomacrocyclic support for efficient peroxymonosulfate activation[J]. Environ Sci Technol, 2021, 55(2): 1242-1250. |
| 58 | WU X, RIGBY K, HUANG D, et al. Single-atom cobalt incorporated in a 2D graphene oxide membrane for catalytic pollutant degradation[J]. Environ Sci Technol, 2022, 56(2): 1341-1351. |
| 59 | ZHANG Y, CHEN X, LIANG C, et al. Reconstructing the coordination environment of single atomic Fe-catalysts for boosting the Fenton-like degradation activities[J]. Appl Catal B: Environ, 2022, 315: 121536. |
| 60 | MA W, SUN M, HUANG D, et al. Catalytic membrane with copper single-atom catalysts for effective hydrogen peroxide activation and pollutant destruction[J]. Environ Sci Technol, 2022: 8733-8745. |
| 61 | YANG M, HOU Z, ZHANG X, et al. Unveiling the origins of selective oxidation in single-atom catalysis via Co-N4-C intensified radical and nonradical pathways[J]. Environ Sci Technol, 2022, 56(16): 11635-11645. |
| [1] | Shu-Min CHEN, Zi-Quan LYU, Xuan ZOU, Shui-Qing GUI, Xue-Mei LU. Research Progress of Functional Masks Amid the Normalization of the COVID-19 Pandemic [J]. Chinese Journal of Applied Chemistry, 2023, 40(11): 1504-1517. |
| [2] | Ya-Zhou YU, Xian-Liang ZHAO, Ning-Ning TAN, Yang XIAO, Yong-Jun ZHANG. Polyallylamine Modified Sulfonic Acid Catalyst Catalyse Synthesis of n-Butyl Benzoate [J]. Chinese Journal of Applied Chemistry, 2023, 40(10): 1430-1436. |
| [3] | Jin-Hui LIANG, Le-Cheng LIANG, Zhi-Ming CUI. Research Progress on Intermetallic Compound Electrocatalysts Applied in the Interconversion Between Hydrogen and Electric Power [J]. Chinese Journal of Applied Chemistry, 2023, 40(8): 1140-1157. |
| [4] | Wei WANG, Jia-Yuan LI. Research Progress of Cobalt Phosphide Heterojunction Catalysts for Electrolytic Hydrogen Evolution Reaction [J]. Chinese Journal of Applied Chemistry, 2023, 40(8): 1175-1186. |
| [5] | Jia-Xin LIU, Jia-He FAN, Shu-Hui LI, Liang MA. Synthesis of Rh@Pt/C Concave Cubic Core-Shell Catalyst and Its Ethanol Electro-Oxidation Performance [J]. Chinese Journal of Applied Chemistry, 2023, 40(8): 1195-1204. |
| [6] | Yan-Bin WU, Li-Zhen LI, Jun-Hua LI, Zhi-Feng XU. Preparation and Properties of Surface Ion Imprinted Material Based on Molybdenum Disulfide Nanoflowers [J]. Chinese Journal of Applied Chemistry, 2023, 40(7): 1024-1033. |
| [7] | Yi-Cheng ZHANG, Fei ZHA, Xiao-Hua TANG, Yue CHANG, Hai-Feng TIAN, Xiao-Jun GUO. Research Progress of Heterogeneous Catalytic Preparation of Organic Peroxides [J]. Chinese Journal of Applied Chemistry, 2023, 40(6): 769-788. |
| [8] | Yi-Chen YU, Yu-Chen ZHANG, Yao-Yuan ZHANG, Qin WU, Da-Xin SHI, Kang-Cheng CHEN, Han-Sheng LI. Research Progress of Bulk Metal Oxides for Non-oxidative Propane Dehydrogenation [J]. Chinese Journal of Applied Chemistry, 2023, 40(6): 789-805. |
| [9] | Yu-Wen YANG, Jing-Yao QI, Lin LI, Guo-Ning CHU, Sai WANG, Yu ZHANG, Shuang ZHANG. Selective Oxidation of 5-Hydroxymethylfurfural to 2,5-Furandicarboxylic Acid over Ru Supported on Magnetic NiFe2O4 [J]. Chinese Journal of Applied Chemistry, 2023, 40(6): 879-887. |
| [10] | Fang-Xie SHEN, Xiang WAN, Wei-Chao WANG. Volatile Organic Compounds Degradation at Room Temperature on Mn-mullite YMn2O5 Catalyst [J]. Chinese Journal of Applied Chemistry, 2023, 40(6): 888-895. |
| [11] | Fan WU, He-Yuan TIAN, Peng LIU, Li-Wei SUN, Yi-Bo ZHANG, Xiang-Guang YANG. Spinel Mangane-Based Catalysts with High Oxygen Vacancy Used for NH3-SCR Reaction at Low Temperature [J]. Chinese Journal of Applied Chemistry, 2023, 40(5): 697-707. |
| [12] | Hui-Hui LI, Kai-Sheng YAO, Ya-Nan ZHAO, Li-Na FAN, Yu-Lin TIAN, Wei-Wei LU. Ionic Liquid-Modulated Synthesis of Pt-Pd Bimetallic Nanomaterials and Their Catalytic Performance for Ammonia Borane Hydrolysis to Generate Hydrogen [J]. Chinese Journal of Applied Chemistry, 2023, 40(4): 597-609. |
| [13] | Bing LI, Jun-Hui LIU, Ya-Kun SONG, Xiang LI, Xu-Ming GUO, Jian XIONG. Recent Advances in Application of Metal-Organic Frameworks for Hydrogen Generation by Catalytic Hydrolysis of Ammonia Borane [J]. Chinese Journal of Applied Chemistry, 2023, 40(3): 329-340. |
| [14] | Jin LIN, Fang-Zhu WANG, Ling-Ling LYU. Preparation of Pseudo-boehamite from Industrial Materials and Its Application in Selective Hydrogenation of Isophorone [J]. Chinese Journal of Applied Chemistry, 2023, 40(1): 79-90. |
| [15] | Rong CAO, Jie-Zhen XIA, Man-Hua LIAO, Lu-Chao ZHAO, Chen ZHAO, Qi WU. Theoretical Research Progress of Single Atom Catalysts in Electrochemical Synthesis of Ammonia [J]. Chinese Journal of Applied Chemistry, 2023, 40(1): 9-23. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
