Chinese Journal of Applied Chemistry ›› 2024, Vol. 41 ›› Issue (4): 568-576.DOI: 10.19894/j.issn.1000-0518.230291
• Full Papers • Previous Articles Next Articles
Synthesis and Properties of Mg‑Doped Ni‑Rich Ternary Cathode Material LiNi0.90Co0.05Mn0.05O2
Sheng CHEN2, Zu-Fei HU2, Hong-Mei CAO3, Zhen-Hua ZHAO1, Yu-Dong ZHANG1( )
)
- 1.School of Materials Science and Engineering,Jiangsu University of Science and Technology,Zhenjiang 212100,China
2.Hunan Jiuri New Materials Co. ,Ltd. ,Huaihua 418200,China
3.School of Energy and Power,Jiangsu University of Science and Technology,Zhenjiang 212100,China
-
Received:2023-09-23Accepted:2024-02-02Published:2024-04-01Online:2024-04-28 -
Contact:Yu-Dong ZHANG -
About author:yudongzhang@just.edu.cn
-
Supported by:Hunan Science and Technology Association “Xiaohe” Science and Technology Talent Lifting Project(2023TJ?X92)
CLC Number:
Cite this article
Sheng CHEN, Zu-Fei HU, Hong-Mei CAO, Zhen-Hua ZHAO, Yu-Dong ZHANG. Synthesis and Properties of Mg‑Doped Ni‑Rich Ternary Cathode Material LiNi0.90Co0.05Mn0.05O2[J]. Chinese Journal of Applied Chemistry, 2024, 41(4): 568-576.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.230291
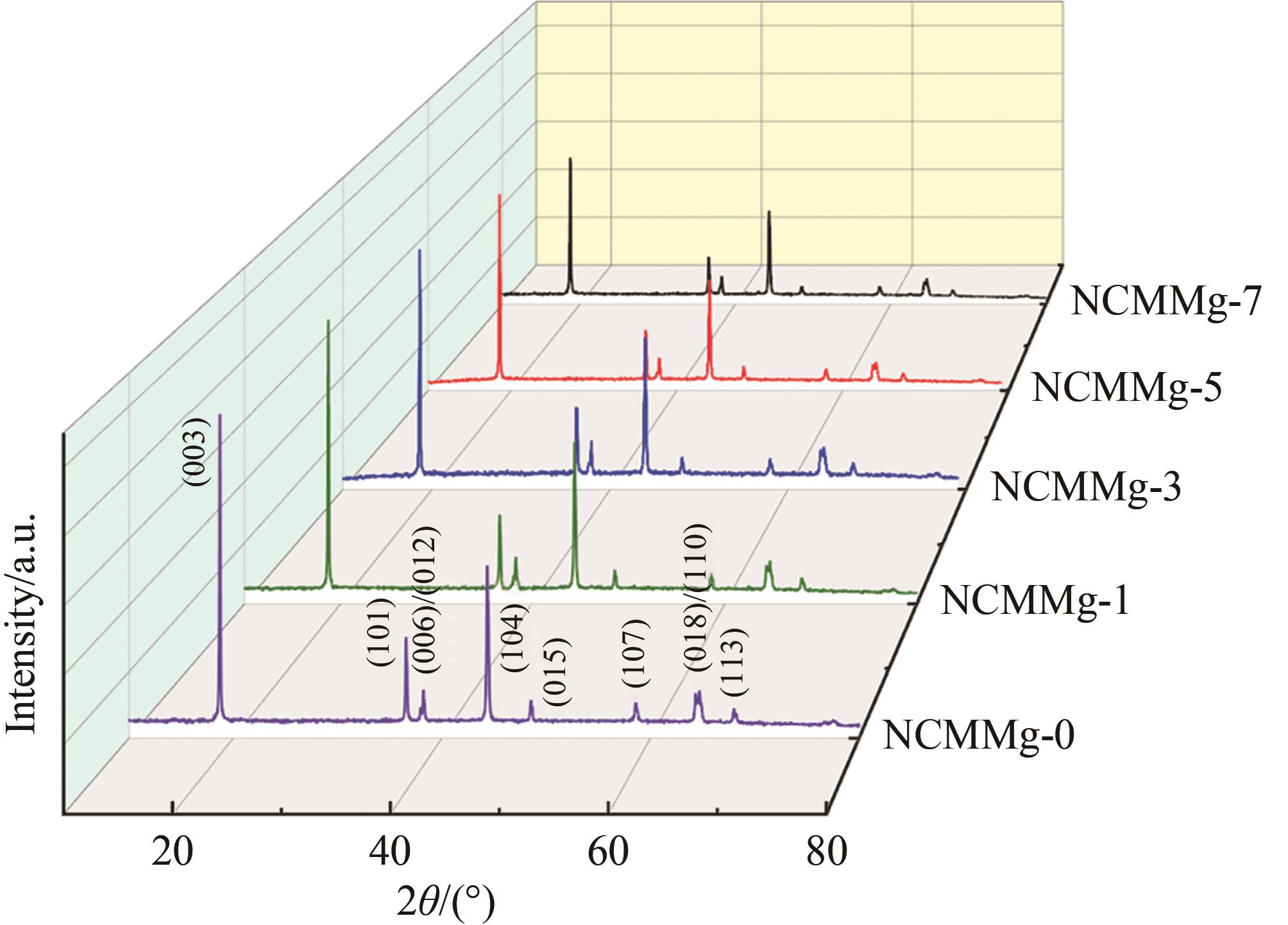
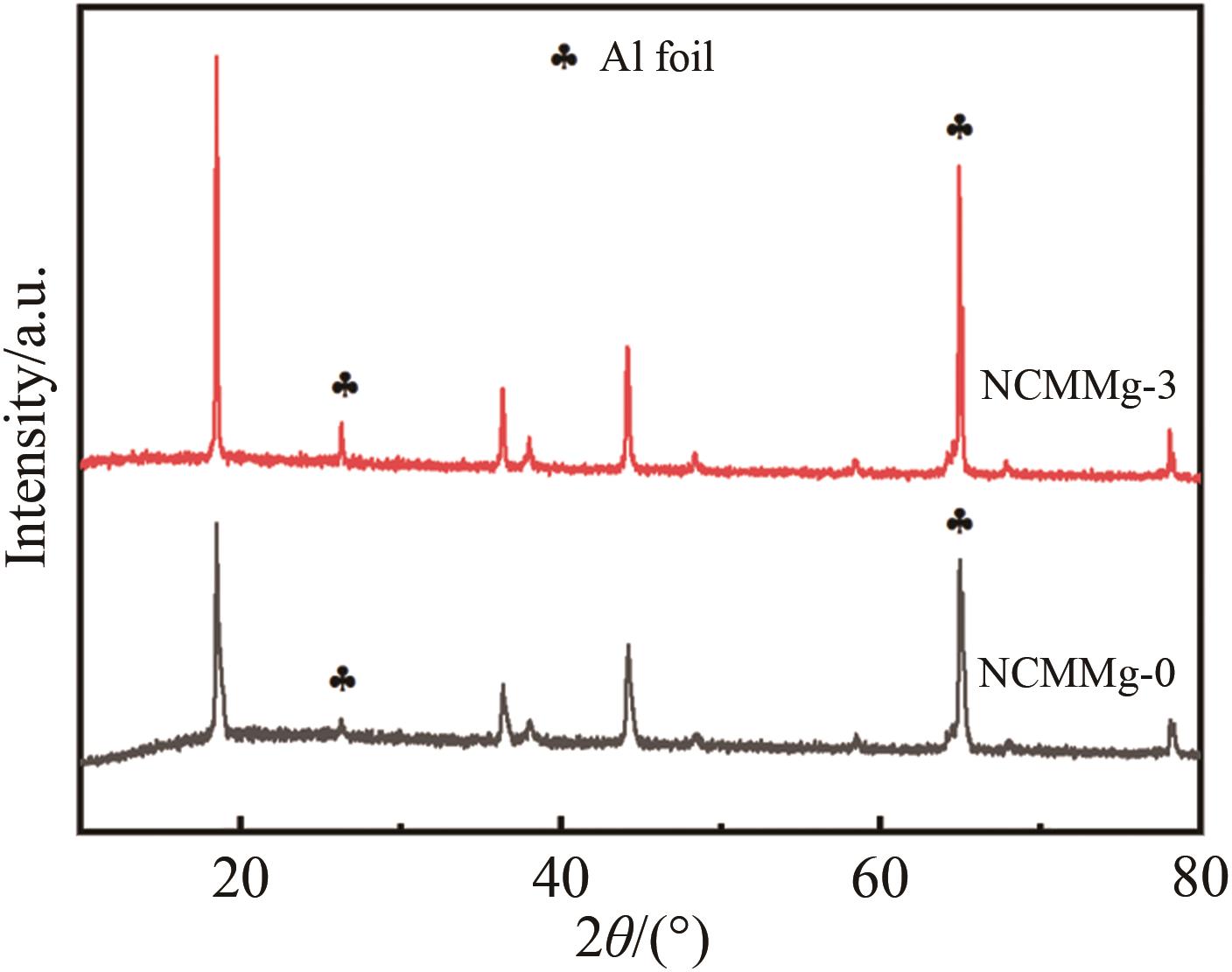
| Sample | a/nm | c/nm | V/nm3 | c/a | I(003)/I(104) |
|---|---|---|---|---|---|
| NCMMg?0 | 0.287 705 | 1.420 487 | 0.101 827 | 4.937 3 | 1.885 |
| NCMMg?1 | 0.287 832 | 1.420 928 | 0.101 949 | 4.936 6 | 1.745 |
| NCMMg?3 | 0.287 965 | 1.421 538 | 0.102 087 | 4.936 5 | 1.771 |
| NCMMg?5 | 0.287 921 | 1.421 426 | 0.102 047 | 4.936 8 | 1.667 |
| NCMMg?7 | 0.288 038 | 1.421 963 | 0.102 169 | 4.936 7 | 1.567 |
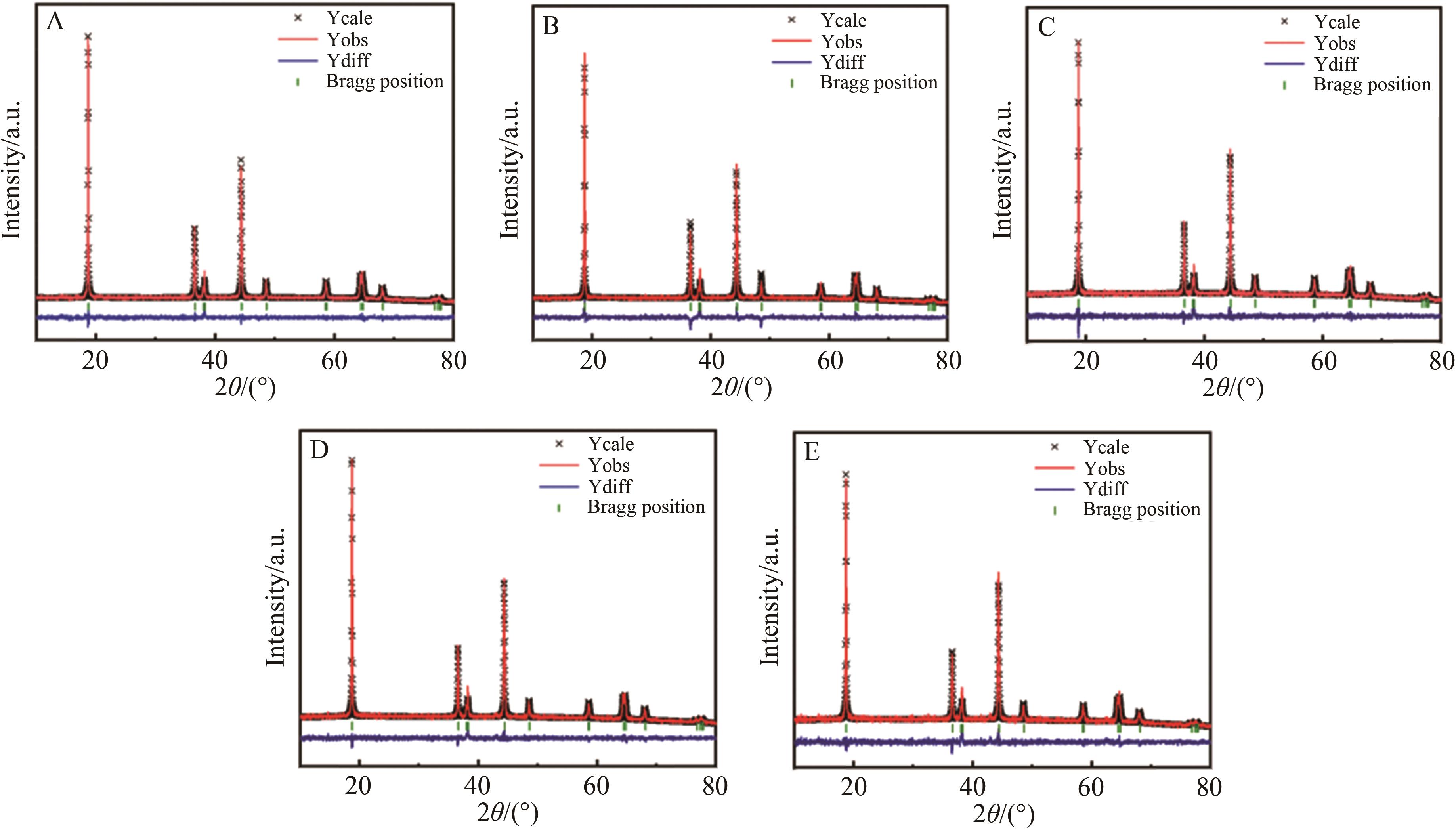
Table 1 The cell parameters of each cathode material obtained by refinement
| Sample | a/nm | c/nm | V/nm3 | c/a | I(003)/I(104) |
|---|---|---|---|---|---|
| NCMMg?0 | 0.287 705 | 1.420 487 | 0.101 827 | 4.937 3 | 1.885 |
| NCMMg?1 | 0.287 832 | 1.420 928 | 0.101 949 | 4.936 6 | 1.745 |
| NCMMg?3 | 0.287 965 | 1.421 538 | 0.102 087 | 4.936 5 | 1.771 |
| NCMMg?5 | 0.287 921 | 1.421 426 | 0.102 047 | 4.936 8 | 1.667 |
| NCMMg?7 | 0.288 038 | 1.421 963 | 0.102 169 | 4.936 7 | 1.567 |
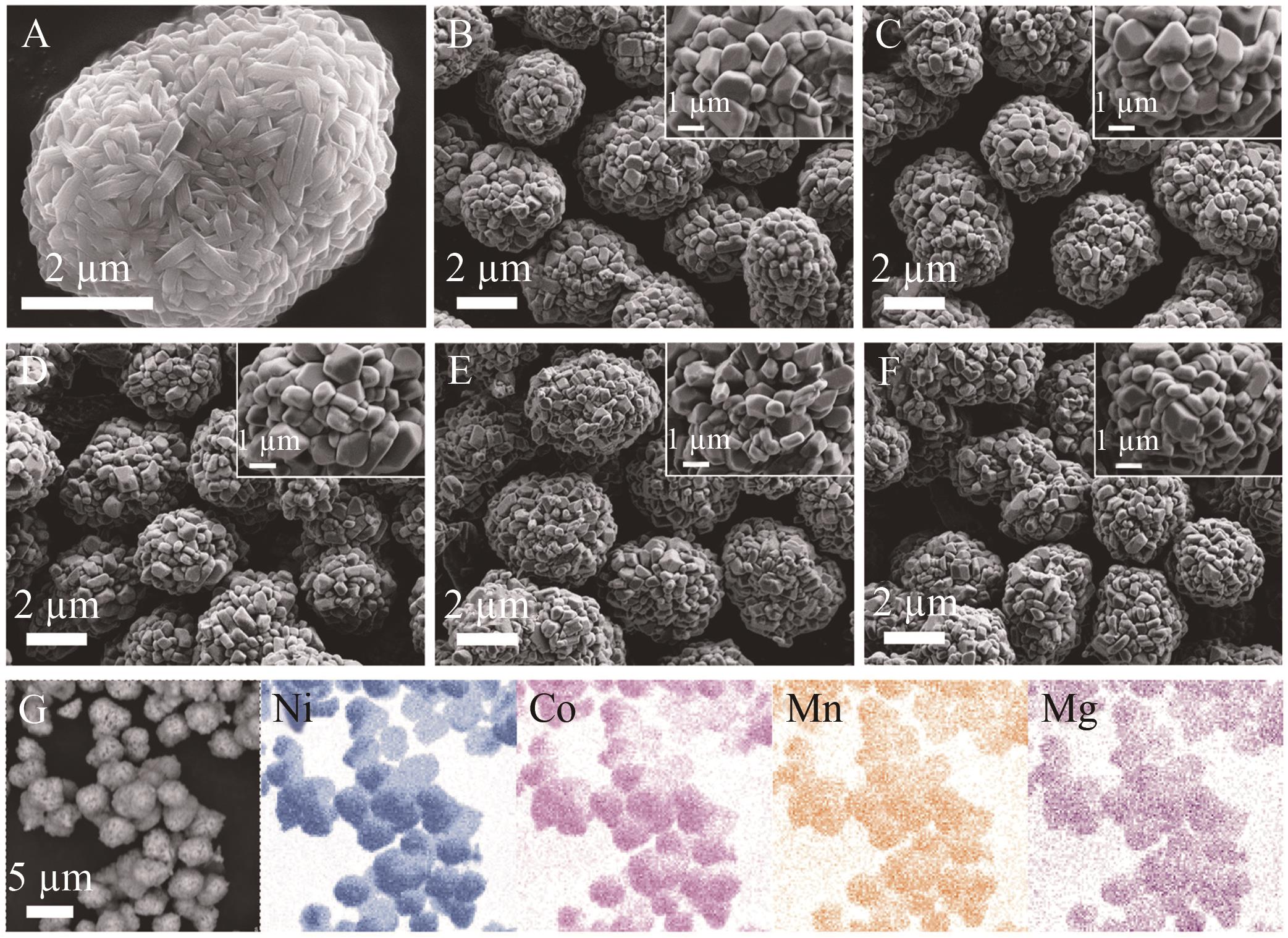
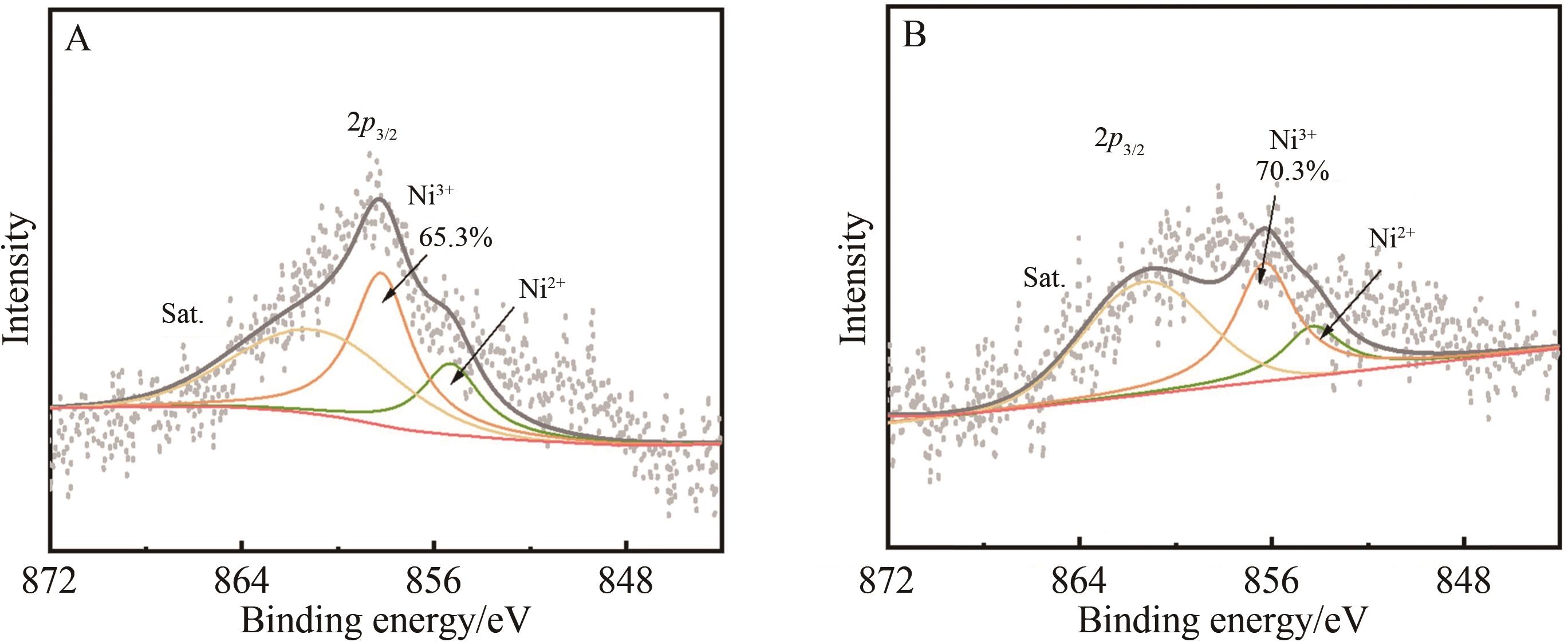
Fig.3 SEM images of (A) Ni0.90Co0.05Mn0.05(OH)2 precursor and cathode materials (B) NCAMMg-0, (C) NCAMMg-1, (D) NCAMMg-3, (E) NCAMMg-5, (F) NCAMMg-7, and (G) EDS spectra of NCAMMg-3
| Sample | x(Ni)/% | x(Co)/% | x(Mn)/% | x(Mg)/% |
|---|---|---|---|---|
| NCMMg?0 | 90.26 | 4.75 | 4.99 | 0 |
| NCMMg?1 | 89.53 | 4.54 | 4.87 | 1.06 |
| NCMMg?3 | 87.51 | 4.53 | 4.93 | 3.03 |
| NCMMg?5 | 86.20 | 4.46 | 4.50 | 4.84 |
| NCMMg?7 | 84.24 | 4.51 | 4.64 | 6.61 |
Table 2 The proportion of Ni, Co, Mn and Mg elements in each sample obtained by ICP test
| Sample | x(Ni)/% | x(Co)/% | x(Mn)/% | x(Mg)/% |
|---|---|---|---|---|
| NCMMg?0 | 90.26 | 4.75 | 4.99 | 0 |
| NCMMg?1 | 89.53 | 4.54 | 4.87 | 1.06 |
| NCMMg?3 | 87.51 | 4.53 | 4.93 | 3.03 |
| NCMMg?5 | 86.20 | 4.46 | 4.50 | 4.84 |
| NCMMg?7 | 84.24 | 4.51 | 4.64 | 6.61 |
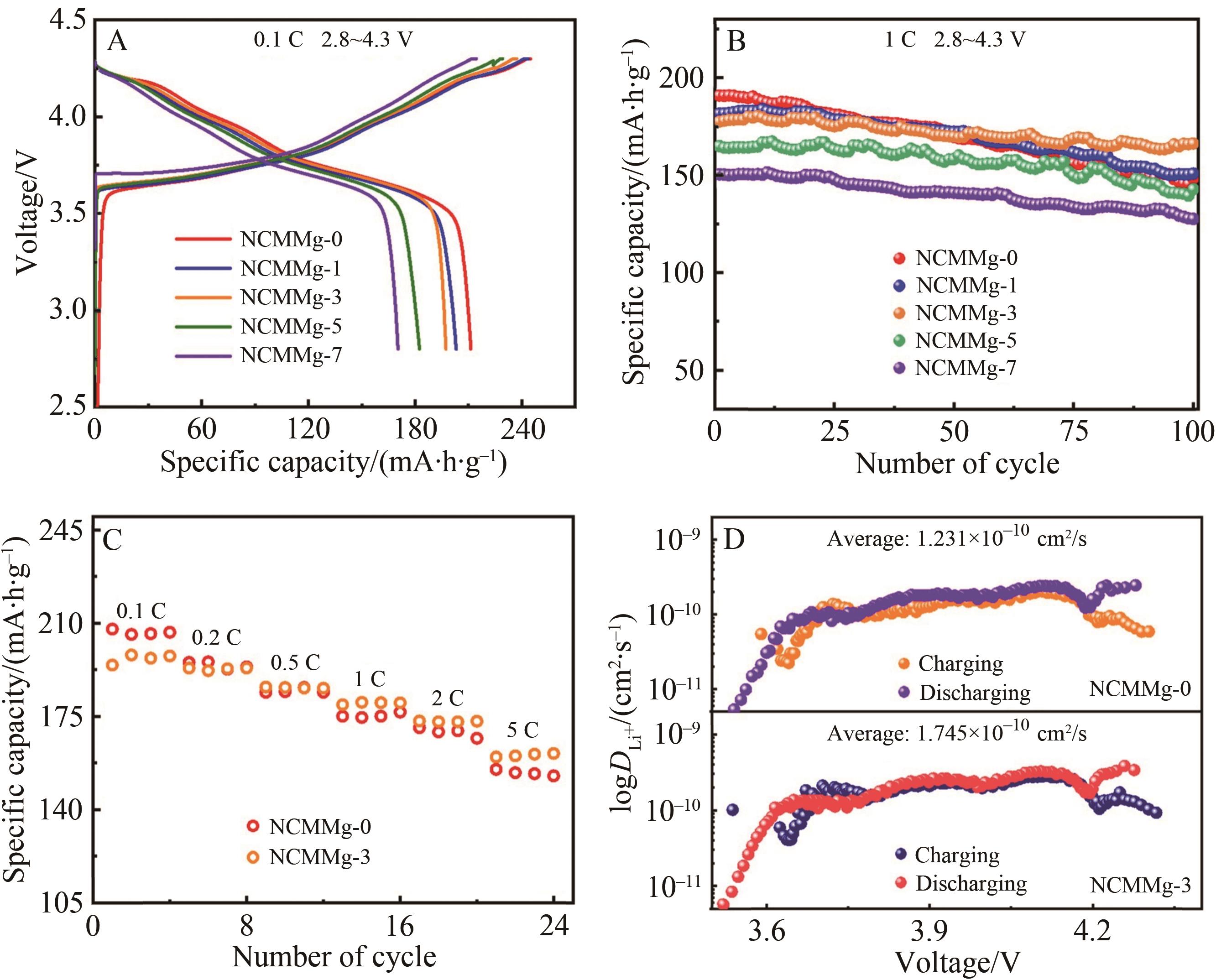
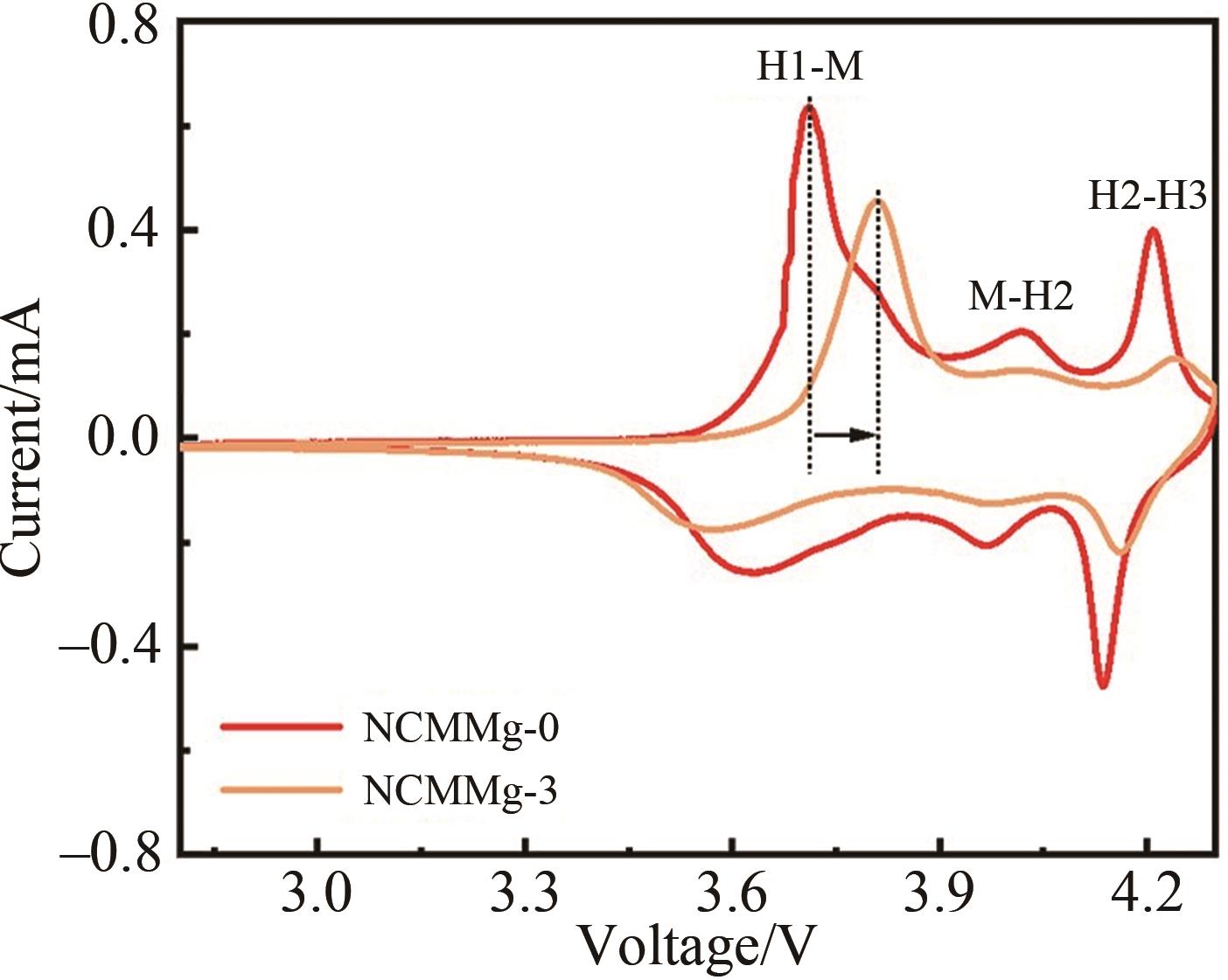
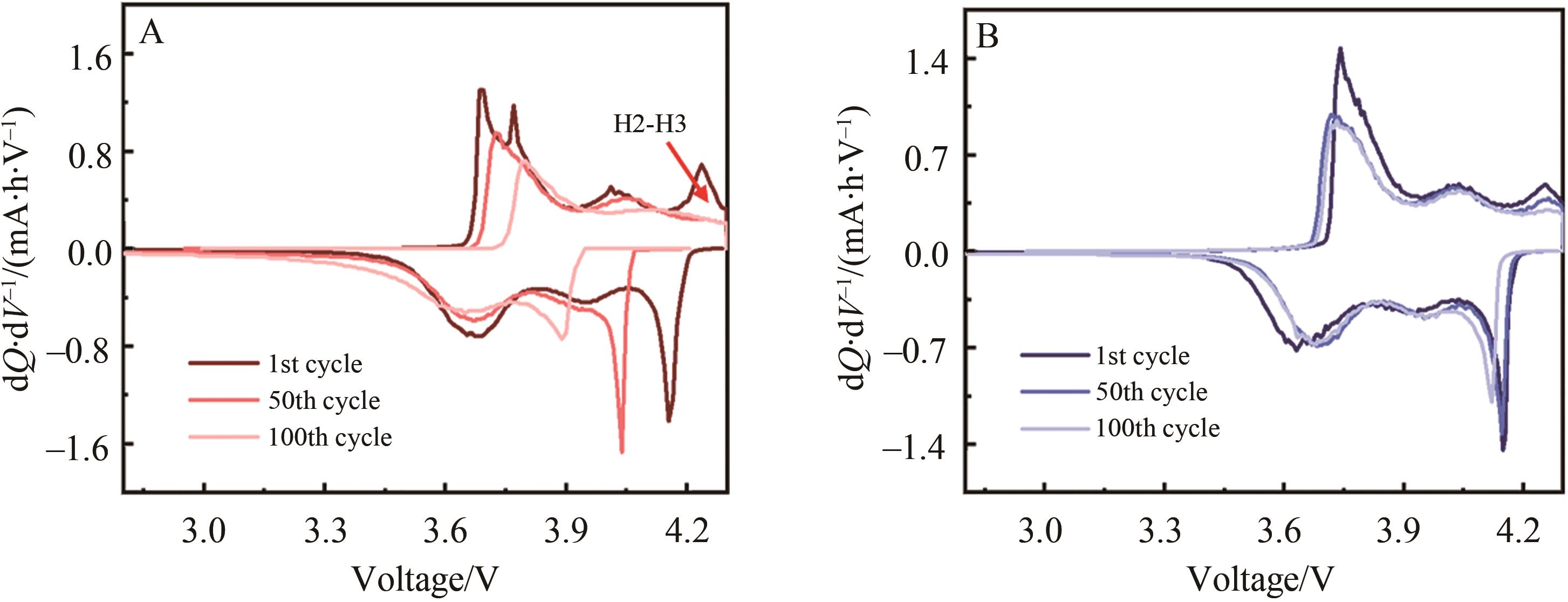
Fig.4 (A) The initial charge and discharge curves of NCMMg-0, NCMMg-1, NCMMg-3, NCMMg-5 and NCMMg-7 samples at 2.8~4.3 V and 0.1 C; (B) Cycle performance of NCMMg-0 and NCMMg-3 at 1 C; (C) Rate performance of NCMMg-0 and NCMMg-3; (D) Lithium ion diffusion coefficient curve of NCMMg-0 and NCMMg-3 sample during charge and discharge
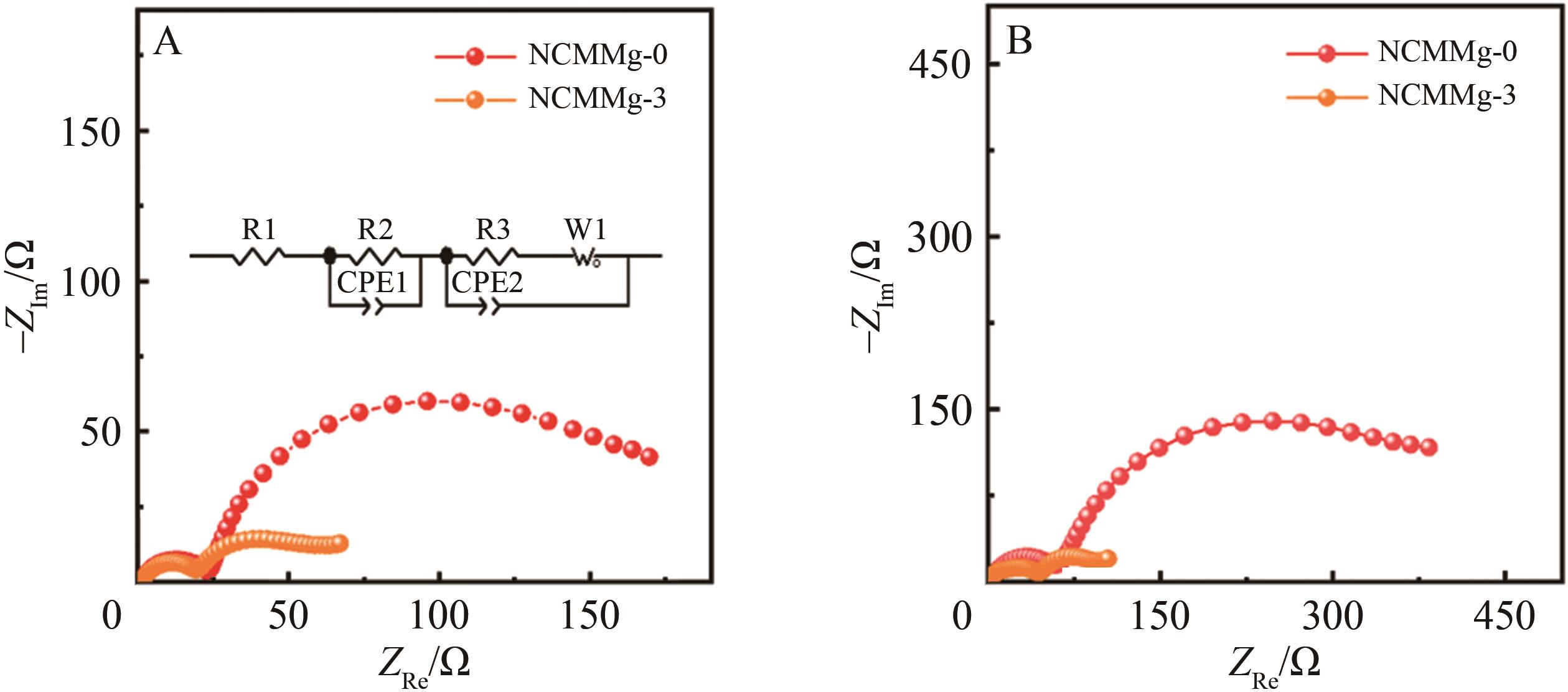
| Sample | Rsf after 10 cycles/Ω | Rct after 10 cycles/Ω | Rsf after 100 cycles/Ω | Rct after 100 cycles/Ω |
|---|---|---|---|---|
| NCMMg?0 | 21.8 | 153.9 | 61.5 | 374.7 |
| NCMMg?3 | 18.1 | 43.8 | 52.4 | 85.4 |
Table 3 Fitting data for EIS
| Sample | Rsf after 10 cycles/Ω | Rct after 10 cycles/Ω | Rsf after 100 cycles/Ω | Rct after 100 cycles/Ω |
|---|---|---|---|---|
| NCMMg?0 | 21.8 | 153.9 | 61.5 | 374.7 |
| NCMMg?3 | 18.1 | 43.8 | 52.4 | 85.4 |

Fig.10 (A) Low resolution TEM image and (B) high resolution TEM image of NCMMg-0 sample particles after cycling, high resolution TEM image of (C) fresh NCMMg-3 and (D) cycled NCMMg-3
| 1 | MANTHIRAM A. A reflection on lithium-ion battery cathode chemistry[J]. Nat Commun, 2020, 11(1): 1550. |
| 2 | JUNG C H, SHIM H, EUM D, et al. Challenges and recent progress in LiNixCoyMn1- x- yO2 (NCM) cathodes for lithium ion batteries[J]. J Korean Ceram Soc, 2021, 58(1): 1-27. |
| 3 | LU Y, ZHANG Z D, ZHANG Q, et al. Recent advances in Ni-rich layered oxide particle materials for lithium-ion batteries[J]. Particuology, 2020, 53: 1-11. |
| 4 | QIAN H M, REN H Q, ZHANG Y, et al. Surface doping vs. bulk doping of cathode materials for lithium-ion batteries: a review[J]. Electrochem Energy Rev, 2022, 5(4): 2. |
| 5 | CUI Z Z, LI X, BAI X Y, et al. A comprehensive review of foreign-ion doping and recent achievements for nickel-rich cathode materials[J]. Energy Storage Mater, 2023, 57: 14-43. |
| 6 | 王恩通, 杨林芳. 高比容量锂离子电池正极材料LiNi0.6Co0.2Mn0.2O2的制备及性能[J]. 应用化学, 2022, 39(8): 1209-1215. |
| WANG E T, YANG L F. Preparation and properties of LiNi0.6Co0.2Mn0.2O2 cathode material for high specific capacity lithium ion battery[J]. Chin J Appl Chem, 2022, 39(8): 1209-1215. | |
| 7 | ZHAO W G, ZOU L F, JIA H P, et al. Optimized Al doping improves both interphase stability and bulk structural integrity of Ni-rich NMC cathode materials[J]. ACS Appl Energy Mater, 2020, 3(4): 3369-3377. |
| 8 | GOMEZ-MARTIN A, REISSIG F, FRANKENSTEIN L, et al. Magnesium substitution in Ni-rich NMC layered cathodes for high-energy lithium ion batteries[J]. Adv Energy Mater, 2022, 12(8): 2103045. |
| 9 | LI H, ZHOU P F, LIU F M, et al. Stabilizing nickel-rich layered oxide cathodes by magnesium doping for rechargeable lithium-ion batteries[J]. Chem Sci, 2019, 10(5): 1374-1379. |
| 10 | DO S J, SANTHOSHKUMAR P, KANG S H, et al. Al-doped Li[Ni0.78Co0.1Mn0.1Al0.02]O2 for high performance of lithium ion batteries[J]. Ceram Int, 2019, 45(6): 6972-6977. |
| 11 | GAO H, CAI J Y, XU G L, et al. Surface modification for suppressing interfacial parasitic reactions of a nickel-rich lithium-ion cathode[J]. Chem Mater, 2019, 31(8): 2723-2730. |
| 12 | ZHANG Y D, LI H, LIU J X, et al. LiNi0.90Co0.07Mg0.03O2 cathode materials with Mg-concentration gradient for rechargeable lithium-ion batteries[J]. J Mater Chem A, 2019, 7(36): 20958-20964. |
| 13 | LV Y T, CHENG X, QIANG W J, et al. Improved electrochemical performances of Ni-rich LiNi0.83Co0.12Mn0.5O2 by Mg-doping[J]. J Power Sources, 2020, 450(29): 227718. |
| 14 | NAM G W, PARK N Y, PARK K J, et al. Capacity fading of Ni-rich NCA cathodes: effect of microcracking extent[J]. ACS Energy Lett, 2019, 4(12): 2995-3001. |
| 15 | JAMIL S, FASEHULLAH M, JABAR B, et al. Significantly fastened redox kinetics in single crystal layered oxide cathode by gradient doping[J]. Nano Energy, 2022, 94: 106961. |
| [1] | Wen-Jun SHI, Zhong-Hui SUN, Zhong-Qian SONG, XU-Jia NAN, Dong-Xue HAN, Li NIU. Research Progress of Layered Transition Metal Oxides Cathode Materials for Sodium-ion Batteries [J]. Chinese Journal of Applied Chemistry, 2023, 40(4): 583-596. |
| [2] | Xue-Jian SHI, Wan-Qiang LIU, Chun-Li WANG, Yong CHENG, Li-Min WANG. Research Progress of Sb-based Anode Materials for Potassium Ion Batteries [J]. Chinese Journal of Applied Chemistry, 2023, 40(2): 210-228. |
| [3] | Jun-Ling MENG, Chuan TIAN, Na XU, Li-Na ZHAO, Hai-Xia ZHONG, Zhan-Lin XU. Research Progress of in Situ Exsolution of Electrode Surface of Solid Oxide Fuel Cells [J]. Chinese Journal of Applied Chemistry, 2023, 40(10): 1335-1346. |
| [4] | Yu MENG, Qing ZHANG, Wen-Hao PENG, Xiao-Fei ZHU, De-Feng ZHOU. Preparation and Electrochemical Performance of Pr0.8Sr0.2Fe0.7Ni0.3O3-δ ⁃Pr1.2Sr0.8Ni0.6Fe0.4O4+δ Composite Cathode [J]. Chinese Journal of Applied Chemistry, 2022, 39(5): 797-808. |
| [5] | WANG Xu-Yao, FANG Ying-Jun, ZHANG Ling-Zhi. Synthesis and Characterization of Cross-linked Solid Polymer Electrolyte Based on N-Cyanoethylated Polyethylenimine for Lithium-Ion Batteries [J]. Chinese Journal of Applied Chemistry, 2021, 38(8): 946-953. |
| [6] | TAO Chengye, YANG Zhanxu, LI Yue. Preparation and Electrochemical Properties of Red Phosphorus/Graphite Composite [J]. Chinese Journal of Applied Chemistry, 2020, 37(9): 1056-1061. |
| [7] | WANG Chunli,SUN Lianshan,ZHONG Ming,WANG Limin,CHENG Yong. Research Progress of Transition Metal and Compounds for Lithium-Sulfur Batteries [J]. Chinese Journal of Applied Chemistry, 2020, 37(4): 387-404. |
| [8] | CUI Huamin, ZHANG Xueqian, HU Pandeng, YAN Bing, HUANG Weiwei, GUO Wenfeng. Calix[4]quinone/N-Doped Amorphous Carbon Nanofibers Composites for Lithium-Ion Batteries [J]. Chinese Journal of Applied Chemistry, 2020, 37(2): 198-204. |
| [9] | HUI Kanglong, FU Jipeng, GAO Tian, TANG Mingxue. Research Progress of Metal Sulfides in Rechargeable Batteries [J]. Chinese Journal of Applied Chemistry, 2020, 37(12): 1384-1402. |
| [10] | YANG Tao, LIU Wenfeng, MA Mengyue, DONG Hongyu, YANG Shuting. Fade Mechanism of Ternary Lithium Ion Power Battery [J]. Chinese Journal of Applied Chemistry, 2020, 37(10): 1181-1186. |
| [11] | YAN Bing, XIONG Wenxu, ZHENG Shibing, ZHANG Xueqian, HUANG Weiwei. Single-Walled Carbon Nanotubes Enhanced Electrochemical Performance of High-Capacity Organic Cathode Composites Calix[4]quinone/Mesporous Carbon CMK-3 for Li-Ion Batteries [J]. Chinese Journal of Applied Chemistry, 2019, 36(5): 554-563. |
| [12] | HUANG Jimei, MENG Ruijin, YANG Jinhu. Preparation and Lithium Ion Storage Performance of Sulfur-Doped Titanium Dioxide/Titanium Carbide Composite [J]. Chinese Journal of Applied Chemistry, 2018, 35(8): 925-931. |
| [13] | Jimei HUANG, Ruijin MENG, Jinhu YANG. Preparation and Lithium Ion Storage Performance of Sulfur-Doped Titanium Dioxide/Titanium Carbide Composite [J]. Chinese Journal of Applied Chemistry, 2018, 35(8): 0-0. |
| [14] | Zhaomin WANG, Zheng YI, Ming ZHONG, Yong CHENG, Limin WANG. Research Progress of Antimony-Based Anode Materials for Lithium Ion Batteries [J]. Chinese Journal of Applied Chemistry, 2018, 35(7): 745-755. |
| [15] | ZHANG He,ZHANG Mengshi,LIAO Shijun. Recent Progress in the Lithium-Rich Ternary Layered Cathode Materials [J]. Chinese Journal of Applied Chemistry, 2018, 35(11): 1277-1288. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||