Chinese Journal of Applied Chemistry ›› 2023, Vol. 40 ›› Issue (10): 1335-1346.DOI: 10.19894/j.issn.1000-0518.230141
• Review •
Research Progress of in Situ Exsolution of Electrode Surface of Solid Oxide Fuel Cells
Jun-Ling MENG1, Chuan TIAN1,2, Na XU1, Li-Na ZHAO1, Hai-Xia ZHONG2( ), Zhan-Lin XU1(
), Zhan-Lin XU1( )
)
- 1.Key Laboratory of Preparation and Applications of Environmental Friendly Materials,Ministry of Education,College of Chemistry,Jilin Normal University,Siping 136000,China
2.State Key Laboratory of Rare Earth Resource Utilization,Changchun Institute of Applied Chemistry,Chinese Academy of Sciences,Changchun 130022,China
-
Received:2023-05-12Accepted:2023-08-18Published:2023-10-01Online:2023-10-13 -
Contact:Hai-Xia ZHONG,Zhan-Lin XU -
About author:xuzhanlin1964@163.com
hxzhong@ciac.ac.cn
-
Supported by:the National Natural Science Foundation of China(52002369);the Scientific and Technological Developing Project of Jilin Province(20220101097JC);the Research Program on Science and Technology from the Education Department of Jilin Province(JJKH20230495KJ);the Open Funds of the State Key Laboratory of Rare Earth Resource Utilization(RERU2022009);the Key Laboratory for Comprehensive Energy Saving of Cold Regions Architecture of Ministry of Education, Jilin Jianzhu University(JLJZHDK202205)
CLC Number:
Cite this article
Jun-Ling MENG, Chuan TIAN, Na XU, Li-Na ZHAO, Hai-Xia ZHONG, Zhan-Lin XU. Research Progress of in Situ Exsolution of Electrode Surface of Solid Oxide Fuel Cells[J]. Chinese Journal of Applied Chemistry, 2023, 40(10): 1335-1346.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.230141
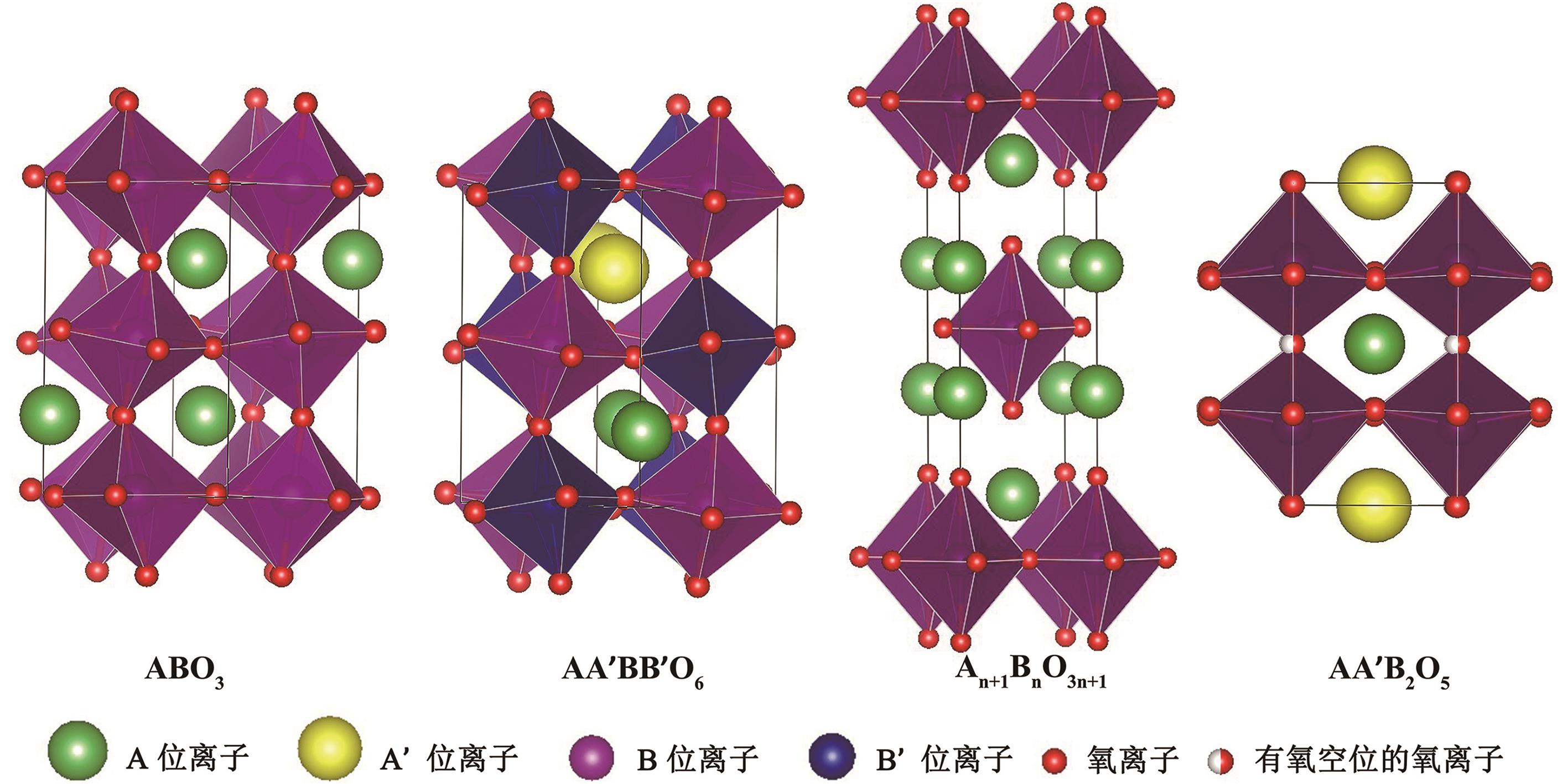
| Structure types | Representative materials | Exsolved nanoparticles |
|---|---|---|
| Single perovskite | La0.3Ca0.7Fe0.7Cr0.3O3?δ[ | Ni-Fe alloy |
| Pr0.4Sr0.6Co0.2Fe0.7Nb0.1O3?δ[ | Co3Fe7 alloy | |
| La0.5Sr0.5Fe0.9Mo0.1?x Ni x O3?δ[ | Fe-Ni alloy | |
| Ni-La0.8Sr0.2FeO3?δ[ | Ni-Fe alloy | |
| SrFe0.85Ti0.1Ni0.05O3?δ[ | NiO | |
| Double perovskite | (PrBa)0.95Fe2?x Ti x O6?δ[ | Fe |
| SrBiFeTiO6?δ[ | Bi and Fe | |
| Sr1.95Fe1.4Co0.1Mo0.5O6?δ[ | Co | |
| Sr2FeMo1?x Co x O6?δ[ | Co-Fe alloy | |
| La0.5Sr1.5Fe1.5Mo0.5O6?δ[ | Fe | |
| Ruddlesden-Popper perovskite | (La0.6Sr1.4)0.95Mn0.9B0.1O4 (B=Co,Ni,Cu)[ | Co, Ni, Cu |
| (LaSr)0.9Fe0.9Cu0.1O4[ | Cu | |
| La1.5Sr1.5Mn1.5Ni0.5O7[ | Ni | |
| (Pr0.4Sr0.6)3(Fe0.85Nb0.15)2O7[ | Co-Fe alloy | |
| La1.2Sr0.8Mn0.4Fe0.6O4[ | Fe | |
| A-site-deficient perovskite | La0.4Sr0.4Ti0.94Ni0.06O3?δ[ | Ni |
| Pr0.4Sr0.5Co x Fe0.9?x Mo0.1O3?δ[ | Co-Fe alloy | |
| (Ba0.2Sr0.8)0.9Ni0.07Fe0.63Mo0.3O3?δ[ | NiFe/NiFeO x core-shell structure | |
| (Pr0.5Sr0.5)0.9Fe0.8Ru0.1Nb0.1O3?δ[ | FeRu/FeO x core-shell structure | |
| La0.6Sr0.2Cr0.85Ni0.15O3[ | Ni |
Table 1 Study on exsolution of SOFC electrode with different structure types of perovskite and representative materials
| Structure types | Representative materials | Exsolved nanoparticles |
|---|---|---|
| Single perovskite | La0.3Ca0.7Fe0.7Cr0.3O3?δ[ | Ni-Fe alloy |
| Pr0.4Sr0.6Co0.2Fe0.7Nb0.1O3?δ[ | Co3Fe7 alloy | |
| La0.5Sr0.5Fe0.9Mo0.1?x Ni x O3?δ[ | Fe-Ni alloy | |
| Ni-La0.8Sr0.2FeO3?δ[ | Ni-Fe alloy | |
| SrFe0.85Ti0.1Ni0.05O3?δ[ | NiO | |
| Double perovskite | (PrBa)0.95Fe2?x Ti x O6?δ[ | Fe |
| SrBiFeTiO6?δ[ | Bi and Fe | |
| Sr1.95Fe1.4Co0.1Mo0.5O6?δ[ | Co | |
| Sr2FeMo1?x Co x O6?δ[ | Co-Fe alloy | |
| La0.5Sr1.5Fe1.5Mo0.5O6?δ[ | Fe | |
| Ruddlesden-Popper perovskite | (La0.6Sr1.4)0.95Mn0.9B0.1O4 (B=Co,Ni,Cu)[ | Co, Ni, Cu |
| (LaSr)0.9Fe0.9Cu0.1O4[ | Cu | |
| La1.5Sr1.5Mn1.5Ni0.5O7[ | Ni | |
| (Pr0.4Sr0.6)3(Fe0.85Nb0.15)2O7[ | Co-Fe alloy | |
| La1.2Sr0.8Mn0.4Fe0.6O4[ | Fe | |
| A-site-deficient perovskite | La0.4Sr0.4Ti0.94Ni0.06O3?δ[ | Ni |
| Pr0.4Sr0.5Co x Fe0.9?x Mo0.1O3?δ[ | Co-Fe alloy | |
| (Ba0.2Sr0.8)0.9Ni0.07Fe0.63Mo0.3O3?δ[ | NiFe/NiFeO x core-shell structure | |
| (Pr0.5Sr0.5)0.9Fe0.8Ru0.1Nb0.1O3?δ[ | FeRu/FeO x core-shell structure | |
| La0.6Sr0.2Cr0.85Ni0.15O3[ | Ni |

Fig.3 Schematic diagram of the key role of in situ exsolution in non-stoichiometric perovskite. (A) Schematic diagram of the in situ exsolution of metal particles from the sample; (B) Equation of exsolution from perovskite oxides with A-site defects (equation 1) and without cationic defects (equation 2); (C) Cartesian plot of the non-stoichiometric perovskite oxides. Compared to perfect ABO3, non-stoichiometric perovskites with excess ions in the lattice are located in the first quadrant and non-stoichiometric perovskites with defects in the lattice are located in the third quadrant[27]

Fig.4 Comparison of the in situ exsolution methods by hydrogen reduction and electric reduction. (A) Schematic diagram of the in situ exsolution process; (B) Metal nanoparticles are exsoluted by hydrogen reduction; (C) Metal nanoparticles are exsoluted by electric reduction; (D) The blue curve is the TG data, showing the change of oxygen loss with time after hydrogen reduction, and the orange curve is the change of current density with time after the application of 2 V voltage. SEM micrographs of electrodes surface; (E) Reduction by hydrogen at 900 ℃ for 20 h; (F) The enlarged morphology of figure E; (G) Electrochemical switching under 900 ℃ for 150 s; (H) The enlarged morphology of figure G; (I) Fuel cell testing at 750 ℃ for 100 h after electrochemical reduction; (J) The enlarged morphology of figure I; (K) Various characteristics of the samples shown in E-J, plotted in a parallel coordinate system[28]
| 1 | BOLDRIN P, BRANDON N P. Progress and outlook for solid oxide fuel cells for transportation applications[J]. Nat Catal, 2019, 2: 571-577. |
| 2 | GREEN R D, ELANGOVAN S E, CHEN F. Perspective-solid oxide cell technology for space exploration[J]. J Electrochem Soc, 2022, 169: 054528-054532. |
| 3 | SINGH M, ZAPPA D, COMINI E. Solid oxide fuel cell: decade of progress, future perspectives and challenges[J]. Int J Hydrogen Energy, 2021, 46: 27643-27674. |
| 4 | ZHENG Z, JING J, YU H, et al. Boosting and robust multifunction cathode layer for solid oxide fuel cells[J]. ACS Sustainable Chem Eng, 2022, 10: 6817-6825. |
| 5 | YU X, WANG Z, REN R, et al. In situ self-reconstructed nanoheterostructure catalysts for promoting oxygen reduction[J]. ACS Energy Lett, 2022, 7: 2961-2969. |
| 6 | ZHANG B, ZHANG S, HAN H, et al. Cobalt-free souble perovskite oxide as a promising cathode for solid oxide fuel cells[J]. ACS Appl Mater Interfaces, 2023, 15: 8253-8262. |
| 7 | MENG J L, LIU X J, HAN L, et al. Improved electrochemical performance by doping cathode materials Sr2Fe1.5Mo0.5- xTaxO6- δ (0.0≤x≤0.15) for solid state fuel cell[J]. J Power Sources, 2014, 247: 845-851. |
| 8 | MENG J L, YUAN N, LIU X J, et al. Assessment of LaM0.25Mn0.75O3- δ (M=Fe, Co, Ni, Cu) as promising cathode materials for intermediate-temperature solid oxide fuel cell[J]. Electrochim Acta, 2015, 169: 264-275. |
| 9 | MENG J L, LIU X J, YAO C G, et al. Investigations on structures, thermal expansion and electrochemical properties of La0.75Sr0.25Cu0.5- xCoxMn0.5O3- δ (x=0, 0.25, and 0.5) as potential cathodes for intermediate temperature solid oxide fuel cells[J]. Electrochim Acta, 2015, 186: 262-270. |
| 10 | MENG J L, YUAN N, LIU X J, et al. Synergistic effects of intrinsic cation disorder and electron-deficient substitution on ion and electron conductivity in La1- xSrxCo0.5Mn0.5O3- δ (x=0, 0.5, and 0.75)[J]. Inorg Chem, 2015, 54: 2820-2829. |
| 11 | MENG J L, LIU X J, YAO C G, et al. SrFe0.8Mo0.2O3- δ: cathode materialfor intermediate temperature solid oxide fuel cells[J]. Solid State Ionics, 2014, 260: 43-48. |
| 12 | MENG J L, LIU X J, YAO C G, et al. Bi-doped La2ZnMnO6- δ and relevant Bi-deficient compound as potential cathodes for intermediate temperature solid oxide fuel cells[J]. Solid State Ionics, 2015, 279: 32-38. |
| 13 | SASE M, HERMES F, YSHIRO K, et al. Enhancement of oxygen surface exchange at the hereo-interface of (La, Sr)CoO3/(La, Sr)2CoO4 with PLD-layered films[J]. J Electrochem Soc, 2008, 155: B793-B797. |
| 14 | YOON J, CHO S, KIM J H, et al. Vertically aligned nanocomposite thin films as a cathode/electrolyte interface layer for thin-film solid oxide fuel cells[J]. Adv Funct Mater, 2009, 19: 3868-3873. |
| 15 | CRUMLIN E J, MUTORO E, AHN S J, et al. Oxygen reduction kinetics enhancement on a heterostructured oxide surface for solid oxide fuel cells[J]. J Phys Chem Lett, 2010, 1: 3149-3155. |
| 16 | LYNCH M E, YANG L, QIN W, et al. Enhancement of La0.6Sr0.4Co0.2Fe0.8O3- δ durability and surface electrocatalytic activity by La0.85Sr0.15MnO3± δ investigated using a new test electrode platform[J]. Energy Environ Sci, 2011, 4: 2249-2258. |
| 17 | MA W, KIM J J, TSVETKOV N, et al. Vertically aligned nanocomposite La0.8Sr0.2CoO3/(La0.5Sr0.5)2CoO4 cathodes-electronic structure, surface chemistry and oxygen reduction kinetics[J]. J Mater Chem A, 2015, 3: 207-219. |
| 18 | CHOI H J, BAE K, GRIESHAMMER S, et al. Surface turning of solid oxide fuel cell cathode by atomic layer deposition[J]. Adv Energy Mater, 2018, 8: 1802506-1802514. |
| 19 | JIANG S P. Nanoscale and nano-structured electrodes of solid oxide fuel cells by infiltration: advances and challenges[J]. Int J Hydrogen Energy, 2012, 37: 449-470. |
| 20 | DING D, LI X, LAI S Y, et al. Enhancing SOFC cathode performance by surface modification through infiltration[J]. Energy Environ Sci, 2014, 7: 552-575. |
| 21 | WANG H C, ZHANG W W, GUAN K, et al. Enhancing activity and durability of A-site-deficient (La0.6Sr0.4)0.95Co0.2Fe0.8O3- δ cathode by surface modification with PrO2- δ nanoparticles[J]. ACS Sustainable Chem Eng, 2020, 8: 3367-3380. |
| 22 | LEE S, ZHANG W, KHATKHATAY F, et al. Strain tuning and strong enhancement of ionic conductivity in SrZrO3-RE2O3 (RE=Sm, Eu, Gd, Dy and Er) nanocomposite films[J]. Adv Funct Mater, 2015, 25: 4328-4333. |
| 23 | LEE S, ZHANG W, KHATKHATAY F, et al. Ionic conductivity increased by two orders of magnitude in macrometer-thick vertical yttria-stabilized ZrO2 nanocomposite films[J]. Nano Lett, 2015, 11: 7362-7369. |
| 24 | 魏振业, 孟君玲, 王浩聪, 等. 同型异质表面修饰提高La2NiO4+ δ阴极的电催化活性[J]. 应用化学, 2020, 37(8): 939-951. |
| WEI Z Y, MENG J L, WANG H C, et al. Improving the electrocatalytic activity of La2NiO4+ δ cathode by surface modification with conformal hereojunction[J]. Chin J Appl Chem, 2020, 37(8): 939-951. | |
| 25 | JI Q Z, MENG J L, WANG H C, et al. Ruddlesden-popper-based lanthanum cuprate thin film cathodes for solid oxide fuel cells: effects of doping and structural transformation on the oxygen reduction reaction[J]. Int J Hydrogen Energy, 2021, 46: 27173-27182. |
| 26 | 纪荃增, 孟君玲, 王浩聪, 等. La2CuO4 阴极薄膜的相转变及其对电化学性能的影响[J]. 应用化学, 2021, 38(8): 976-985. |
| JI Q Z, MENG J L, WANG H C, et al. Phase transformation of La2CuO4 cathode film and its influence on electrochemical properties[J]. Chin J Appl Chem, 2021, 38(8): 976-985. | |
| 27 | NEAGU D, TSEKOURAS G, MILLER D N, et al. In situ growth of nanoparticles through control of non-stoichiometry[J]. Nat Chem, 2013, 5: 916-923. |
| 28 | MYUNG J, NEAGU D, MILLER D N, et al. Switching on electrocatalytic activity in solid oxide cells[J]. Nature, 2016, 537: 528-531. |
| 29 | QIU P, YANG X, ZOU L, et al. LaCrO3-coated La0.6Sr0.4Co0.2Fe0.8O3- δ core-shell structured cathode with enhanced Cr tolerance for intermediate-temperature solid oxide fuel cells[J]. ACS Appl Mater Interfaces, 2020, 12: 29133-29142. |
| 30 | CHEN Y, CHEN Y, DING D, et al. A robust and active hybrid catalyst for facile oxygen reduction in solid oxide fuel cells[J]. Energy Environ Sci, 2017, 10: 964-971. |
| 31 | TANAKA H, UENISHI M, TANIGUCHI M, et al. The intelligent catalyst having the self-regenerative function of Pd, Rh, Pt for automotive emissions control[J]. Catal Today, 2006, 117: 321-328. |
| 32 | MADSEN B D, KOBSIRIPHAT W, WANG Y, et al. Nucleation of nanometer-scale electrocatalyst particles in solid oxide fuel cell anodes[J]. J Power Sources, 2007, 166: 64-67. |
| 33 | MADSEN B D, KOBSIRIPHAT W, WANG Y, et al. SOFC anode performance enhancement through precipitation of nanoscale catalysts[J]. ECS Trans, 2007, 7: 1339-1348. |
| 34 | KOBSIRIPHAT W, MADSEN B D, WANG Y, et al. La0.8Sr0.2Cr1- xRuxO3- δ-Gd0.1Ce0.9O1.95 solid oxide fuel cell anodes: Ru precipitation and electrochemical performance[J]. Solid State Ionics, 2009, 180: 257-264. |
| 35 | BIERSCHENK D M, POTTER-NELSON E, HOEL C, et al. Pd-substituted (La,Sr)CrO3- δ-Ce0.9Gd0.1O2- δ solid oxide fuel cell anodes exhibiting regenerative behavior[J]. J Power Sources, 2011, 196: 3089-3094. |
| 36 | KOBSIRIPHAT W, MADSEN B D, WANG Y, et al. Nickel- and ruthenium-doped lanthanum chromite anodes: effects of nanoscale metal precipitation on solid oxide fuel cell performance[J]. J Electrochem Soc, 2010, 157: B279-B284. |
| 37 | SUN Y F, ZHANG Y Q, CHEN J, et al. New opportunity for in situ exsolution of metallic nanoparticles on perovskite parent[J]. Nano Lett, 2016, 16: 5303-5309. |
| 38 | YANG C, LI J, LIN Y, et al. In situ fabrication of CoFe alloy nanoparticles structured (Pr0.4Sr0.6)3(Fe0.85Nb0.15)2O7 ceramic anode for direct hydrocarbon solid oxide fuel cells[J]. Nano Energy, 2015, 11: 704-710. |
| 39 | XIAO G L, WANG S W, LIN Y, et al. Ni-doped Sr2Fe1.5Mo0.5O6- δ as anode materials for solid oxide fuel cells[J]. J Electrochem Soc, 2014, 161: F305-F310. |
| 40 | WANG Y, LIU T, LI M, et al. Exsolved Fe-Ni nano-particles from Sr2Fe1.3Ni0.2Mo0.5O6 perovskite oxide as a cathode for solid oxide steam electrolysis cells[J]. J Mater Chem A, 2016, 4: 14163-14169. |
| 41 | DU Z H, ZHAO H L, YI S, et al. High-performance anode material Sr2FeMo0.65Ni0.35O6- δ with in situ exsolved nanoparticle catalyst[J]. ACS Nano, 2016, 10: 8660-8669. |
| 42 | ZHOU J, SHIN T H, NI C S, et al. In situ growth of nanoparticles in layered perovskite La0.8Sr1.2Fe0.9Co0.1O4- δ as an active and stable electrode for symmetrical solid oxide fuel cells[J]. Chem Mater, 2016, 28: 2981-2993. |
| 43 | ANSARI H M, BASS A S, AHMAD N, et al. Unraveling the evolution of exsolved Fe-Ni alloy nanoparticles in Ni-doped La0.3Ca0.7Fe0.7Cr0.3O3- δ and their role in enhancing CO2-CO electrocatalys[J]. J Mater Chem A, 2022, 10: 2280-2294. |
| 44 | LI P, DONG R Z, WANG Y C, et al. Improving the performance of Pr0.4Sr0.6Co0.2Fe0.7Nb0.1O3- δ-based single-component fuel cell and reversible single-component cells by manufacturing A-site deficiency[J]. Renew Energy, 2021, 177: 387-396. |
| 45 | XU J S, WU M, SONG Z Y, et al. In situ grown of LaSr(Fe,Mo)O4 cermic anodes with exsolved Fe-Ni nanoparticles for SOFCs: electrochemical performance and stability in H2, CO, and syngas[J]. J Eur Ceram Soc, 2021, 41: 4537-4551. |
| 46 | YAO X L, ASGHAR M I, ZHAO Y C, et al. Coking resistant Ni-La0.8Sr0.2FeO3 composite anode improves the stability of syngas-fueled SOFC[J]. Int J Hydrogen Energy, 2021, 46: 9809-9817. |
| 47 | YANG G, ZHOU W, LIU M, et al. Enhancing Electrode performance by exsolved nanoparticles: a superior cobalt-free perovskite electrocatalyst for solid oxide fuel cells[J]. ACS Appl Mater Interfaces, 2016, 8: 35308-35314. |
| 48 | QIAO J S, CHEN X J, AI C Y, et al. Fe-based layered double perovskite anode with in situ exsolved nanoparticles for direct carbon solid oxide fuel cells[J]. Ind Eng Chem Res, 2023, 62: 445-454. |
| 49 | SUN N, JIN F J, LIU X L, et al. In situ coexsolution of metal nanoparticle-decorated double perovskite as anode materials for solid oxide fuel cells[J]. ACS Appl Energy Mater, 2021, 4: 7992-8002. |
| 50 | XU C M, SUN W, REN R Z, et al. A highly active and carbon-tolerant anode decorated with in situ grown cobalt nano-catalyst for intermediate-temperature solid oxide fuel cells[J]. Appl Catal B-Environ, 2021, 282: 119553. |
| 51 | LI H D, SONG Y F, XU M G, et al. Exsolved alloy nanoparticles decorated Ruddlesden-Popper perovskite as sulfur-tolerant anodes for solid oxide fuel cells[J]. Energy Fuels, 2020, 34: 11449-11457. |
| 52 | QI H, XIA F, YANG T, et al. In situ exsolved nanoparticles on La0.5Sr1.5Fe1.5Mo0.5O6- δ anode enhance the hydrogen oxidation reaction in SOFCs[J]. J Electrochem Soc, 2020, 167: 024510-024521. |
| 53 | WANG J K, ZHOU J, YANG J M, et al. Nanoscale architecture of (La0.6Sr1.4)0.95Mn0.9B0.1O4 (B=Co, Ni, Cu) Ruddlesden-Popper oxides as efficient and durable catalysts for symmetrical solid oxide fuel cells[J]. Renew Energ, 2020, 157: 840-850. |
| 54 | FU L, ZHOU J, YANG J M, et al. Exsolution of Cu nanoparticles in (LaSr)0.9Fe0.9Cu0.1O4 Ruddlesden-Popper oxide as symmetrical electrode for solid oxide cells[J]. Appl Surf Sci, 2020, 511: 145525. |
| 55 | VECINO-MANTILLA S, QUINTERO E, FONSECA C, et al. Catalytic steam reforming of natural gas over a new Ni exsolved Ruddlesden-Popper manganite in SOFC anode conditions[J]. ChemCatChem, 2020, 12: 1453-1466. |
| 56 | YANG C, LI J, LIN Y, et al. In situ fabrication of CoFe alloy nanoparticles structured (Pr0.4Sr0.6)3(Fe0.85Nb0.15)2O7 ceramic anode for direct hydrocarbon solid oxide fuel cells[J]. Nano Energy, 2015, 11: 704-710. |
| 57 | CHUNG Y S, KIM, T, SHIN T H, et al. In situ preparation of a La1.2Sr0.8Mn0.4Fe0.6O4 Ruddlesden-Popper phase with exsolved Fe nanoparticles as an anode for SOFCs[J]. J Mater Chem A, 2017, 5: 6437-6446. |
| 58 | LEE J J, KIM K, KIM K J, et al. In-situ exsolution of Ni nanoparticles to achieve an active and stable solid oxide fuel cell anode catalyst on A-site deficient La0.4Sr0.4Ti0.94Ni0.06O3- δ[J]. J Ind Eng Chem, 2021, 103: 264-274. |
| 59 | LI P, HAN Y, XU M, et al. Electrochemical performance of Pr0.4Sr0.5CoxFe0.9- xMo0.1O3- δ oxides in a reversible SOFC/SOEC system[J]. Energy Fuels, 2022, 36: 15165-15176. |
| 60 | YANG H Y, LA M, WANG Z M, et al. In situ exsolved NiFe/(NiFe)Ox core-shell-structured nanocatalysts on perovskite anode with enhanced coking resistance[J]. ACS Sustainable Chem Eng, 2022, 10: 12510-12519. |
| 61 | QIN M X, XIAO Y, YANG H Y, et al. Ru/Nb co-doped perovskite anode: achieving good resistance in hydrocarbon fuels via core-shell nanocatalysts exsolution[J]. Appl Catal B-Environ, 2021, 299: 120613. |
| 62 | XI X, WANG X W, FAN Y, et al. Efficient bifunctional electrocatalysts for solid oxide cells based on the structural evolution of perovskites with abundant defects and exsolved CoFe nanoparticles[J]. J Power Sources, 2021, 482: 228981. |
| 63 | SUN Y F, LI J H, CUI L, et al. A-site-deficiency facilitated in situ growth of bimetallic Ni-Fe nano-alloys: a novel coking-tolerant fuel cell anode catalyst[J]. Nanoscale, 2015, 7: 11173-11181. |
| 64 | HU H, LI M, MIN H, et al. Enhancing the catalytic activity and coking tolerance of the perovskite anode for solid oxide fuel cells through in situ exsolution of Co-Fe nanoparticles[J]. ACS Catal, 2022, 12: 828-836. |
| 65 | KWON O, SENGODAN S, KIM K, et al. Exsolution trends and co-segregation aspects of self-grown catalyst nanoparticles in perovskites[J]. Nat Commun, 2017, 8: 15967. |
| 66 | LU Y, HUANG Y W, XU Z H, et al. Quantifying electrochemical driving force for exsolution in perovskite oxide by designing graded oxygen chemical potential[J]. ACS Nano, 2023, 17: 14005-14013. |
| 67 | WANG J Y, YANG J, OPITZ A K, et al. Tuning point defects by elastic stain modulates nanoparticle exsolution on perovskite oxides[J]. Chem Mater, 2021, 33: 5021-5034. |
| 68 | WANG J Y, KALAEV D, YANG J, et al. Fast surface oxygen release kinetics accelerate nanoparticle exsolution in perovskite oxides[J]. J Am Chem Soc, 2023, 145: 1714-1727. |
| 69 | LI P, YANG T Q, SHAO Y N, et al. Evaluating the effect of B-site cation doping on the properties of Pr0.4Sr0.5Fe0.9Mo0.1O3 for reversible single-component cells[J]. Ind Eng Chem Res, 2022, 61: 5030-5041. |
| 70 | ZHANG W W, WANG H C, GUAN K, et al. Enhanced anode performance and coking resistance by in situ exsolved multiples-twinned Co-Fe nanoparticles for solid oxide fuel cells[J]. ACS Appl Mater Interfaces, 2020, 12: 461-473. |
| 71 | ZHU Y, ZHOU W, RAN R, et al. Promotion of oxygen reduction by exsolved silver nanoparticles on a perovskite scaffold for low-temperature solid oxide fuel cells[J]. Nano Lett, 2015, 16: 512-518. |
| 72 | ZHANG X, ZHANG L F, MENG J L, et al. Enhanced electrochemical property of La0.6Sr0.4Co0.8Fe0.2O3 as cathode for solid oxide fuel cell by efficient in situ polarization-exsolution treatment [J]. Electrochim Acta, 2017, 258: 1096-1105. |
| 73 | LEE J G, MYUNG J H, NADEN A B, et al. Replacement of Ca by Ni in a perovskite titanate to yield a novel perovskite exsolution architecture for oxygen-evolution reactions[J]. Adv Energy Mater, 2020, 10: 1903693. |
| 74 | FU L, ZHOU J, ZHOU L K, et al. Facile fabrication of exsolved nanoparticle-decorated hollow ferrite fibers as active electrocatalyst for oxygen evolution reaction[J]. Chem Eng J, 2021, 418: 129422. |
| 75 | ZHU Y, ZHOU W, RAN R, et al. Promotion of oxygen reduction by exsolved silver nanoparticles on a perovskite scaffold for low-temperature solid oxide fuel cells[J]. Nano Lett, 2016, 16: 512-518. |
| 76 | ARRIVÉ C, DELAHAYE T, JOUBERT O, et al. Exsolution of nickel nanoparticles at the surface of a conducting titanate as potential hydrogen electrode material for solid oxide electrochemical cells[J]. J Power Sources, 2013, 223: 341-348. |
| 77 | LI J W, WEI B, CAO Z Q, et al. Niobium doped lanthanum strontium ferrite as a redox-stable and sulfur-tolerant anode for solid oxide fuel cells[J]. ChemSusChem, 2018, 11: 254-263. |
| 78 | DU Z, ZHAO H, YI S, et al. High-performance anode material Sr2FeMo0.65Ni0.35O6- δ with in situ exsolved nanoparticle catalyst[J]. ACS Nano, 2016, 10: 8660-8669. |
| 79 | JIANG Y L, GENG Z B, SUN Y, et al. Highly efficient B-site exsolution assisted by Co doping in lanthanum ferrite toward high-performance electrocatalysts for oxygen evolution and oxygen reduction[J]. ACS Sustainable Chem Eng, 2020, 8: 302-310. |
| 80 | ZHU T L, TROIANI H E, MOGNI L V, et al. Ni-substituted Sr(Ti,Fe)O3 SOFC anodes: achieving high performance via metal alloy nanoparticle exsolution[J]. Joule, 2018, 2: 478-496. |
| 81 | LIANG Y, CUI Y, CHAO Y, et al. Exsolution of CoFe(Ru) nanoparticles in Ru-doped (La0.8Sr0.2)0.9Co0.1Fe0.8Ru0.1O3- δ for efficient oxygen evolution reaction[J]. Nano Res, 2022, 15: 6977-6986. |
| 82 | MA J J, GENG Z B, JIANG Y L, et al. Exsolution manipulated local surface cobalt/iron alloying and dealloying conversion in La0.95Fe0.8Co0.2O3 perovskite for oxygen evolution reaction[J]. J Alloys Compd, 2021, 854: 157154. |
| 83 | GUI L Q, PAN G H, MA X, et al. In-situ exsolution of CoNi alloy nanoparticles on LiFe0.8Co0.1Ni0.1O2 parent: new opportunity for boosting oxygen evolution and reduction reaction[J]. Appl Surf Sci, 2021, 543: 148817. |
| 84 | CAO X J, KE L, ZHAO K, et al. Surface decomposition of doped PrBaMn2O5+δ induced by in situ nanoparticle exsolution: quantitative characterization and catalytic effect in methane dry reforming reaction[J]. Chem Mater, 2022, 34: 10484-10494. |
| [1] | Xue-Jian SHI, Wan-Qiang LIU, Chun-Li WANG, Yong CHENG, Li-Min WANG. Research Progress of Sb-based Anode Materials for Potassium Ion Batteries [J]. Chinese Journal of Applied Chemistry, 2023, 40(2): 210-228. |
| [2] | Ting WANG, Qi WEI, Qiang FU, Wei LI, Shi-Wei WANG. Review of Perovskite Photovoltaic Cell Encapsulation Material and Technology [J]. Chinese Journal of Applied Chemistry, 2022, 39(9): 1321-1344. |
| [3] | Yu MENG, Qing ZHANG, Wen-Hao PENG, Xiao-Fei ZHU, De-Feng ZHOU. Preparation and Electrochemical Performance of Pr0.8Sr0.2Fe0.7Ni0.3O3-δ ⁃Pr1.2Sr0.8Ni0.6Fe0.4O4+δ Composite Cathode [J]. Chinese Journal of Applied Chemistry, 2022, 39(5): 797-808. |
| [4] | Guo-Hua DONG, Li-Juan HAO, Wen-Zhi ZHANG, Dong-Feng CHAI, Ming ZHAO, Kun LANG. Recent Progress on the Application of Carbon Quantum Dots Nano⁃materials in Lead Halogen Perovskite Solar Photoelectric Devices [J]. Chinese Journal of Applied Chemistry, 2022, 39(5): 707-722. |
| [5] | ZHU Meng-Meng, BAI Jue-Yao, CHEN Ren-Jie, LI Hui-Li. Synthesis and Properties of B-Site Doped All-Inorganic Perovskite Quantum Dots [J]. Chinese Journal of Applied Chemistry, 2021, 38(12): 1541-1555. |
| [6] | FAN Liming,WANG Shimao,YE Yuqi,DING Bowen,SHAN Xueyan,MENG Gang,FANG Xiaodong. Recent Advances in Rational Synthesis and Applications of Halide Perovskite Micro/Nano-Arrays [J]. Chinese Journal of Applied Chemistry, 2020, 37(4): 367-379. |
| [7] | WANG Chunli,SUN Lianshan,ZHONG Ming,WANG Limin,CHENG Yong. Research Progress of Transition Metal and Compounds for Lithium-Sulfur Batteries [J]. Chinese Journal of Applied Chemistry, 2020, 37(4): 387-404. |
| [8] | HUI Kanglong, FU Jipeng, GAO Tian, TANG Mingxue. Research Progress of Metal Sulfides in Rechargeable Batteries [J]. Chinese Journal of Applied Chemistry, 2020, 37(12): 1384-1402. |
| [9] | HUANG Guobin, LUO Dengfeng, ZHANG Maosheng. Preparation of CsPbX3(X=Cl,Br,I) Perovskite Quantum Dots with Multicolor and High Luminescence Efficiency and Its Application in Light Emitting Diode Devices [J]. Chinese Journal of Applied Chemistry, 2019, 36(8): 932-938. |
| [10] | HUANG Guobin, LUO Dengfeng, ZHANG Maosheng. Preparation of CsPbX3(X=Cl,Br,I) Perovskite Quantum Dots with Multicolor and High Luminescence Efficiency and Its Application in Light Emitting Diode Devices [J]. Chinese Journal of Applied Chemistry, 2019, 36(8): 0-. |
| [11] | CHEN Haining. Recent Progress in Carbon-based Perovskite Solar Cells [J]. Chinese Journal of Applied Chemistry, 2018, 35(8): 916-924. |
| [12] | Zhaomin WANG, Zheng YI, Ming ZHONG, Yong CHENG, Limin WANG. Research Progress of Antimony-Based Anode Materials for Lithium Ion Batteries [J]. Chinese Journal of Applied Chemistry, 2018, 35(7): 745-755. |
| [13] | MENG Jingjia,ZHANG Feng,REN Yandong,LIU Lige,LIU Yonghao,ZHONG Haizheng. Halide Perovskite Based Two-Dimensional Nanocrystals:Synthesis and Luminescence Properties [J]. Chinese Journal of Applied Chemistry, 2018, 35(3): 342-350. |
| [14] | ZHAO Liping, WANG Zhen, DONG Xiandui. Effect of Ionic Liquid on the Performance of Perovskite Solar Cells [J]. Chinese Journal of Applied Chemistry, 2018, 35(2): 216-223. |
| [15] | CHEN Lihui,WU Qiuhan,PAN Pei,SONG Zixuan,WANG Feng,DING Yu. Spinel Lithium Manganese Oxide Octahedral Nanoparticles with Excellent Electrochemical Performance as Cathode Materials for Lithium-Ion Batteries [J]. Chinese Journal of Applied Chemistry, 2018, 35(11): 1384-1390. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||