Chinese Journal of Applied Chemistry ›› 2023, Vol. 40 ›› Issue (7): 938-950.DOI: 10.19894/j.issn.1000-0518.230009
• Review • Previous Articles Next Articles
Research Progress of Activated Persulfate by MOFs-Based Catalyst in Wastewater Treatment
Zhen-Chun TANG, Xin-Quan ZHOU, Pei-Pei WANG, Juan MIAO, Ning ZHANG, Rui-Chang ZHANG, Xue-Feng WEI( )
)
- School of Chemistry and Chemical Engineering,Henan University of Science and Technology,Luoyang 471000,China
-
Received:2023-01-15Accepted:2023-05-23Published:2023-07-01Online:2023-07-19 -
Contact:Xue-Feng WEI -
About author:xfwei@haust.edu.cn
-
Supported by:the Intellectual Introduction Project of Henan Province(HNGD2022058);the Key Scientific Research Project of Colleges and Universities of Henan Province(23A610010)
CLC Number:
Cite this article
Zhen-Chun TANG, Xin-Quan ZHOU, Pei-Pei WANG, Juan MIAO, Ning ZHANG, Rui-Chang ZHANG, Xue-Feng WEI. Research Progress of Activated Persulfate by MOFs-Based Catalyst in Wastewater Treatment[J]. Chinese Journal of Applied Chemistry, 2023, 40(7): 938-950.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.230009
| MOFs-based catalyst | Strengths | Precursor | Catalyst | Contaminant | Ref. |
|---|---|---|---|---|---|
| MILs catalysts | Porosity, large surface area and unsaturated metal coordination | MIL-88A(Fe) | MIL-88A(Fe)/MoS2 | Bisphenol A | [ |
| MIL-88B(Fe) | MGA | Norfloxacin | [ | ||
| MIL-53(Fe) | MIL-53(Fe)/BiOCl | Rhodamine B | [ | ||
| MIL-101(Fe) | CuS@MIL-101(Fe) | Coumarin | [ | ||
| MIL-53(Fe) | Mn-MIL-53(Fe) | Tetracycline | [ | ||
| MIL-101(Fe) | g-C3N4/MIL-101(Fe) | Bisphenol A | [ | ||
| ZIFs catalysts | Good thermal and chemical stability | ZIF-8 | Fe3O4-MnO2-ZIF-8 | Bisphenol A | [ |
| ZIF-67 | ZIF-67 | Rhodamine B | [ | ||
| ZIF-9 | ZIF-9 | Rhodamine B | [ | ||
| ZIF-12 | ZIF-12 | Rhodamine B | [ | ||
| n-BTCs catalysts | Contains Lewis acid active sites | Co-BTC | Co-BTC(A) Co-BTC(B) | Dibutyl phthalate | [ |
| HKUST-1 | HKUST-1 | Rhodamine B | [ |
Table 1 Summary of MOFs-based catalyst in the field of persulfate activation
| MOFs-based catalyst | Strengths | Precursor | Catalyst | Contaminant | Ref. |
|---|---|---|---|---|---|
| MILs catalysts | Porosity, large surface area and unsaturated metal coordination | MIL-88A(Fe) | MIL-88A(Fe)/MoS2 | Bisphenol A | [ |
| MIL-88B(Fe) | MGA | Norfloxacin | [ | ||
| MIL-53(Fe) | MIL-53(Fe)/BiOCl | Rhodamine B | [ | ||
| MIL-101(Fe) | CuS@MIL-101(Fe) | Coumarin | [ | ||
| MIL-53(Fe) | Mn-MIL-53(Fe) | Tetracycline | [ | ||
| MIL-101(Fe) | g-C3N4/MIL-101(Fe) | Bisphenol A | [ | ||
| ZIFs catalysts | Good thermal and chemical stability | ZIF-8 | Fe3O4-MnO2-ZIF-8 | Bisphenol A | [ |
| ZIF-67 | ZIF-67 | Rhodamine B | [ | ||
| ZIF-9 | ZIF-9 | Rhodamine B | [ | ||
| ZIF-12 | ZIF-12 | Rhodamine B | [ | ||
| n-BTCs catalysts | Contains Lewis acid active sites | Co-BTC | Co-BTC(A) Co-BTC(B) | Dibutyl phthalate | [ |
| HKUST-1 | HKUST-1 | Rhodamine B | [ |
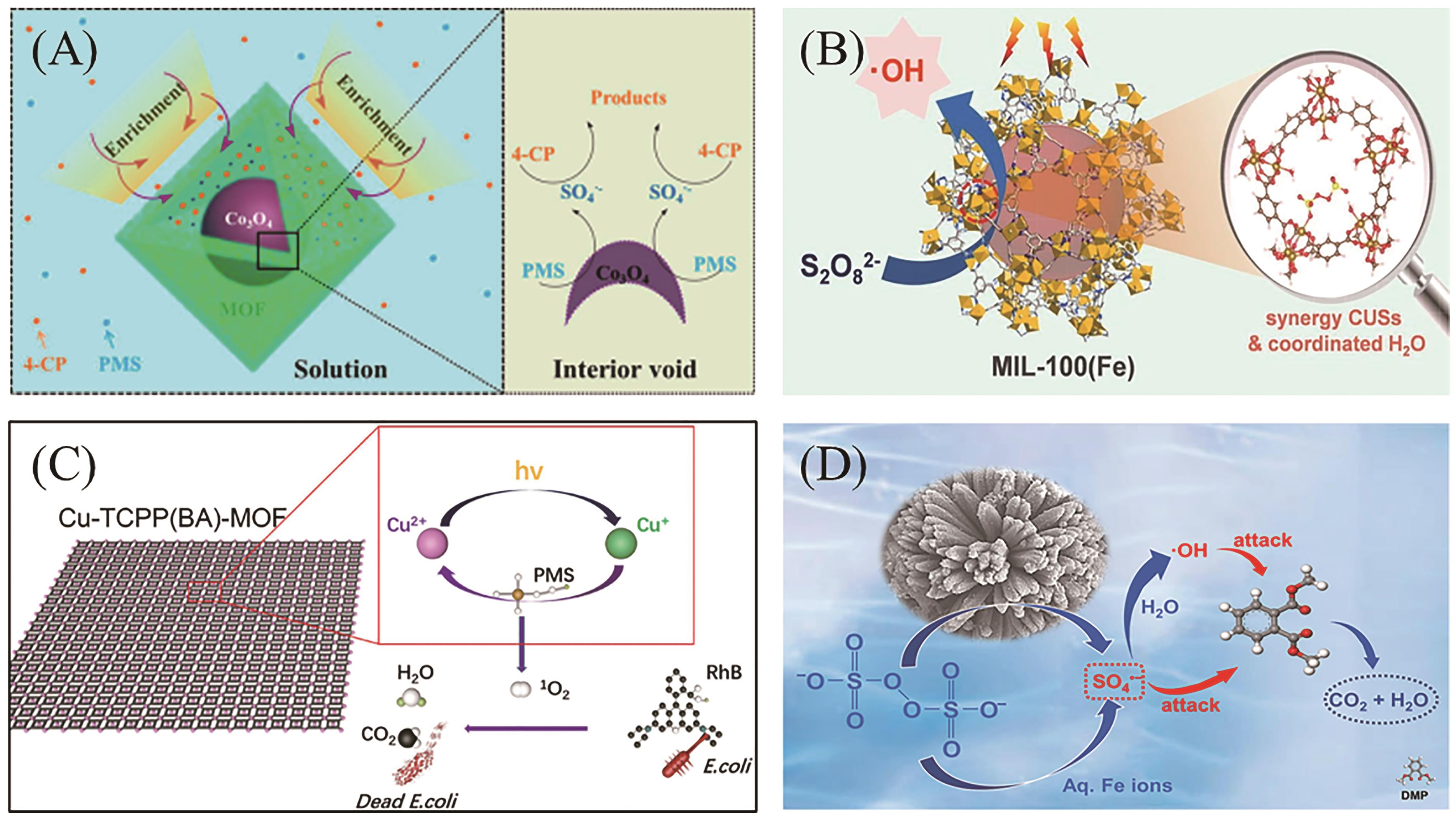
Fig.3 Oxidation mechanism diagram of activated persulfate from several typical MOFs-based catalyst. (A) Possible mechanism diagram of SO4?- participating in the degradation of 4-chlorophenol in Co3O4@ MOFs nanoreactor[56]; (B) The ·OH production on MIL-100(Fe)[57]; (C) Schematic diagram of 1O2 participating in degradation in Cu-TCPP(BA)-MOF/PMS/Vis system[44]; (D) Fe-MOF-74@SiO2 diagram of possible mechanism of activation of PS to produce SO4?- and ·OH[58]
| Catalyst | Precursor | Contaminant | Oxidant | Removal rate/% | ROS | Ref. |
|---|---|---|---|---|---|---|
| M/Z2 | ZIF-67 MIL-101(Fe) | 2-Chlorophenol (0.78 mmol/L) | PMS(0.98 mmol/L) | 90 | SO | [ |
| MSMIL | MIL-88A(Fe) | Bisphenol A (0.11 mmol/L) | PMS(1.63 mmol/L) | 98.2 | SO | [ |
| CuS@MIL-101(Fe) | MIL-101(Fe) | Coumarin (0.03 mmol/L) | PMS(0.5 mmol/L) | 100 | SO | [ |
| Fe3O4-MnO2-ZIF-8 | ZIF-8 | Bisphenol A (0.26 mmol/L) | PMS(0.98 mmol/L) | 100 | SO | [ |
| ZIF-67/PAN | ZIF-67 | Acid yellow 17 (0.91 mmol/L) | PMS(1.63 mmol/L) | 95.1 | SO | [ |
| Co-BTC(A) | Co-BTC | Dibutyl phthalate (0.018 mmol/L) | PMS(1.08 mmol/L) | 100 | SO | [ |
| Cu-MOF-74/PVDF | MOF-74 | Rhodamine B (0.04 mmol/L) | PMS(1.5 mmol/L) | 97.1 | SO | [ |
Table 2 MOFs-based catalysts and their activation of persulfates produce main reactive oxygen species
| Catalyst | Precursor | Contaminant | Oxidant | Removal rate/% | ROS | Ref. |
|---|---|---|---|---|---|---|
| M/Z2 | ZIF-67 MIL-101(Fe) | 2-Chlorophenol (0.78 mmol/L) | PMS(0.98 mmol/L) | 90 | SO | [ |
| MSMIL | MIL-88A(Fe) | Bisphenol A (0.11 mmol/L) | PMS(1.63 mmol/L) | 98.2 | SO | [ |
| CuS@MIL-101(Fe) | MIL-101(Fe) | Coumarin (0.03 mmol/L) | PMS(0.5 mmol/L) | 100 | SO | [ |
| Fe3O4-MnO2-ZIF-8 | ZIF-8 | Bisphenol A (0.26 mmol/L) | PMS(0.98 mmol/L) | 100 | SO | [ |
| ZIF-67/PAN | ZIF-67 | Acid yellow 17 (0.91 mmol/L) | PMS(1.63 mmol/L) | 95.1 | SO | [ |
| Co-BTC(A) | Co-BTC | Dibutyl phthalate (0.018 mmol/L) | PMS(1.08 mmol/L) | 100 | SO | [ |
| Cu-MOF-74/PVDF | MOF-74 | Rhodamine B (0.04 mmol/L) | PMS(1.5 mmol/L) | 97.1 | SO | [ |
| Modification method | Catalyst | Precursor | Contaminant | Oxidant | Time/min | Removal rate/% | Ref. |
|---|---|---|---|---|---|---|---|
| Ion doping | Mn-MIL-53(Fe) (200 mg/L) | MIL-53(Fe) | Tetracycline (30 mg/L) | PMS (0.98 mmol/L) | 60 | 93.2 | [ |
| Combined functional material | CoFe2O4/ZIF-8 (50 mg/L) | ZIF-8 | Methylene blue (20 mg/L) | PMS (0.98 mmol/L) | 60 | 97.9 | [ |
| Ion doping | FeCu-MOF (600 mg/L) | Fe-MOF | Methylene blue (64 mg/L) | PMS (6 mmol/L) | 30 | 100 | [ |
| Functional group modification | NH2-MIL-88B(Fe) (600 mg/L) | MIL-88B(Fe) | Acid orangeⅡ(20 mg/L) | PMS (1.3 mmol/L) | 25 | 97 | [ |
| Construction of core-shell structure | α-Fe2O3/ZIF-67 (100 mg/L) | ZIF-67 | Ciprofloxacin (20 mg/L) | PMS (0.65 mmol/L) | 30 | 100 | [ |
| Combined functional material | Mn3O4/ZIF-8 (400 mg/L) | ZIF-8 | Rhodamine B (10 mg/L) | PMS (0.98 mmol/L) | 60 | 98 | [ |
Table 3 Comparison of common modification methods and activation efficiency of PS in MOFs-based catalyst
| Modification method | Catalyst | Precursor | Contaminant | Oxidant | Time/min | Removal rate/% | Ref. |
|---|---|---|---|---|---|---|---|
| Ion doping | Mn-MIL-53(Fe) (200 mg/L) | MIL-53(Fe) | Tetracycline (30 mg/L) | PMS (0.98 mmol/L) | 60 | 93.2 | [ |
| Combined functional material | CoFe2O4/ZIF-8 (50 mg/L) | ZIF-8 | Methylene blue (20 mg/L) | PMS (0.98 mmol/L) | 60 | 97.9 | [ |
| Ion doping | FeCu-MOF (600 mg/L) | Fe-MOF | Methylene blue (64 mg/L) | PMS (6 mmol/L) | 30 | 100 | [ |
| Functional group modification | NH2-MIL-88B(Fe) (600 mg/L) | MIL-88B(Fe) | Acid orangeⅡ(20 mg/L) | PMS (1.3 mmol/L) | 25 | 97 | [ |
| Construction of core-shell structure | α-Fe2O3/ZIF-67 (100 mg/L) | ZIF-67 | Ciprofloxacin (20 mg/L) | PMS (0.65 mmol/L) | 30 | 100 | [ |
| Combined functional material | Mn3O4/ZIF-8 (400 mg/L) | ZIF-8 | Rhodamine B (10 mg/L) | PMS (0.98 mmol/L) | 60 | 98 | [ |
| 1 | JAWAD A, ZHAN K, WANG H B, et al. Tuning of persulfate activation from a free radical to a nonradical pathway through the incorporation of non-redox magnesium oxide[J]. Environ Sci Technol, 2020, 54(4): 2476-2488. |
| 2 | 李小娟, 廖凤珍, 叶兰妹, 等. 金属有机骨架及其衍生材料活化过硫酸盐在水处理中的应用进展[J]. 化工进展, 2019, 38(10): 4712-4721. |
| LI X J, LIAO F Z, YE L M, et al. Progress in the applications of metal-organic frameworks and derivatives activate persulfate in water treatment[J]. Chem Ind Eng Prog, 2019, 38(10): 4712-4721. | |
| 3 | LI J L, ZHU W H, GAO Y, et al. The catalyst derived from the sulfurized Co-doped metal-organic framework (MOF) for peroxymonosulfate (PMS) activation and its application to pollutant removal[J]. Sep Purif Technol, 2022, 285: 120362. |
| 4 | KHAN A, ZHANG K K, TARAQQI A K A, et al. Degradation of antibiotics in aqueous media using manganese nanocatalyst-activated peroxymonosulfate[J]. J Colloid Interface Sci, 2021, 599: 805-818. |
| 5 | HUANG Y L, TIAN X K, NIE Y L, et al. Enhanced peroxymonosulfate activation for phenol degradation over MnO2 at pH 3.5~9.0 via Cu(Ⅱ) substitution[J]. J Hazard Mater, 2018, 360: 303-310. |
| 6 | WANG J L, WANG S Z. Activation of persulfate (PS) and peroxymonosulfate (PMS) and application for the degradation of emerging contaminants[J]. Chem Eng J, 2018, 334: 1502-1517. |
| 7 | 张楠, 陈蕾. 超声活化过硫酸盐降解废水中有机污染物的研究进展[J]. 应用化工, 2021, 50(10): 2805-2808, 2813. |
| ZHANG N, CHEN L. Research progress of ultrasonic activated persulfate for degradation of organic pollutants in wastewater[J]. Appl Chem Ind, 2021, 50(10): 2805-2808, 2813. | |
| 8 | 姜思佳, 张碧涵,单潇清, 等. 热活化过硫酸钠氧化降解水中2,4-二氯苯酚的研究[J]. 现代化工, 2019, 39(4): 94-98. |
| JIANG S J, ZHANG B H, SHAN X Q, et al. Degradation of 2,4-dichlorophenol in aqueous solution by thermal activated sodium persulfate[J]. Mod Chem Ind, 2019, 39(4): 94-98. | |
| 9 | 张丹, 陈博凯, 晁聪, 等. 紫外光活化过硫酸盐脱色处理甲基橙染料的研究[J]. 工业水处理, 2022, 42(5): 117-124. |
| ZHANG D, CHENG B K, CHAO C, et al. Decolorization treatment methyl orange dye by ultraviolet activated persulfate process[J]. Ind Water Treat, 2022, 42(5): 117-124. | |
| 10 | 朱睿, 谭烨, 李春全, 等. 基于过渡金属活化的过硫酸盐高级氧化技术研究进展[J]. 化工矿物与加工, 2022, 51(1): 49-55. |
| ZHU R, TAN Y, LI C Q, et al. Research progress of advanced persulfate oxidation technology based on activation by transition metals[J]. Ind Miner Process, 2022, 51(1): 49-55. | |
| 11 | LI Y X, YANG Z X, WANG Y L, et al. A mesoporous cationic thorium-organic framework that rapidly traps anionic persistent organic pollutants[J]. Nat Commun, 2017, 8(1): 1354. |
| 12 | LI M H, LIU Y B, LI F, et al. Defect-rich hierarchical porous UiO-66(Zr) for tunable phosphate removal[J]. Environ Sci Technol, 2021, 55(19): 13209-13218. |
| 13 | PEI L, ZHAO X D, LIU B S, et al. Rationally tailoring pore and surface properties of metal-organic frameworks for boosting adsorption of Dy3+[J]. ACS Appl Mater Interfaces, 2021, 13(39): 46763-46771. |
| 14 | LI H T, FU M, WANG S Q, et al. Stable Zr-based metal-organic framework nanoporous membrane for efficient desalination of hypersaline water[J]. Environ Sci Technol, 2021, 55(21): 14917-14927. |
| 15 | PU M J, YE D Q, WAN J Q, et al. Zinc-based metal-organic framework nanofibers membrane ZIF-65/PAN as efficient peroxymonosulfate activator to degrade aqueous ciprofloxacin[J].Sep Purif Technol, 2022, 299: 121716. |
| 16 | LI Y, WANG F, REN X Y. et al. Peroxymonosulfate activation for effective atrazine degradation over a 3D cobalt-MOF: performance and mechanism[J]. J Environ Chem Eng, 2023, 11(1): 109116. |
| 17 | YAO Y Y, WANG C H, YAN X, et al. Rational regulation of Co-N-C coordination for high-efficiency generation of 1O2 toward nearly 100% selective degradation of organic pollutants[J]. Environ Sci Technol, 2022, 56(12): 8833-8843. |
| 18 | HUANG D L, ZHANG G X, YI J, et al. Progress and challenges of metal-organic frameworks-based materials for SR-AOPs applications in water treatment[J]. Chemosphere, 2021, 263: 127672. |
| 19 | WANG L Y, LUO D, YANG J P, et al. Metal-organic frameworks-derived catalysts for contaminant degradation in persulfate-based advanced oxidation processes[J]. J Clean Prod, 2022, 375: 134118. |
| 20 | WANG C H, KIM J H, MALGRAS V, et al. Metal-organic frameworks and their derived materials: emerging catalysts for a sulfate radicals-based advanced oxidation process in water purification[J]. Small, 2019, 15(16): 1900744. |
| 21 | RIOU D, SEREE C, FEREY G, Composite microporous compounds (MIL-n): Ⅱ. hydrothermal synthesis and ab initio resolution by X-ray powder diffraction of MIL-5: a vanadodiphosphonate with a three-dimensional neutral framework[J]. J Solid State Chem, 1998, 141(1): 89-93. |
| 22 | JIANG J J, ZHANG F J, WANG Y R. Review of different series of MOF/g-C3N4 composites for photocatalytic hydrogen production and CO2 reduction[J]. New J Chem, 2023, 47: 1599-1609. |
| 23 | GU A T, WANG P, CHEN K W, et al. Core-shell bimetallic Fe-Co MOFs to activated peroxymonosulfate for efficient degradation of 2-chlorophenol[J]. Sep Purif Technol, 2022, 298: 121461. |
| 24 | ROY D, NEOGI S, DE S. Mechanistic investigation of photocatalytic degradation of Bisphenol-A using MIL-88A(Fe)/MoS2 Z-scheme heterojunction composite assisted peroxymonosulfate activation[J]. Chem Eng J, 2022, 428: 131028. |
| 25 | JING Y, JIA M Y, XU Z Y, et al. Facile synthesis of recyclable 3D gelatin aerogel decorated with MIL-88B(Fe) for activation peroxydisulfate degradation of norfloxacin[J]. J Hazard Mater, 2022, 424: 127503. |
| 26 | MIAO S C, ZHA Z X, LI Y, et al. Visible-light-driven MIL-53(Fe)/BiOCl composite assisted by persulfate: photocatalytic performance and mechanism[J]. J Photochem Photobiol A, 2019, 380: 111862. |
| 27 | ZHOU C, ZHU L, DENG L, et al. Efficient activation of peroxymonosulfate on CuS@MIL-101(Fe) spheres featured with abundant sulfur vacancies for coumarin degradation: performance and mechanisms[J]. Sep Purif Technol, 2021, 276: 119404. |
| 28 | XU W T, MA L, KE F, et al. Metal-organic frameworks MIL-88A hexagonal microrods as a new photocatalyst for efficient decolorization of methylene blue dye[J]. Dalt Trans, 2014, 43(9): 3792-3798. |
| 29 | YANG Q, MA Y, CHEN F. Recent advances in photo-activated sulfate radical-advanced oxidation process (SR-AOP) for refractory organic pollutants removal in water[J]. Chem Eng J, 2019, 378: 122149. |
| 30 | WANG C, LIU X, DEMIR N K, et al. Applications of water stable metal organic frameworks[J]. Chem Soc Rev, 2016, 45(18): 5107-5134. |
| 31 | YU J, CAO J, YANG Z H, et al. One-step synthesis of Mn-doped MIL-53(Fe) for synergistically enhanced generation of sulfate radicals towards tetracycline degradation[J]. J Colloid Interface Sci, 2020, 580: 470-479. |
| 32 | PU M J, GUAN Z Y, MA Y W, et al. Synthesis of iron-based metal-organic framework MIL-53 as an efficient catalyst to activate persulfate for the degradation of orange G in aqueous solution[J]. Appl Catal A, 2018, 549: 82-92. |
| 33 | BANERJEE R, PHAN A, WANG B C, et al. High-throughput synthesis of zeolitic imidazolate frameworks and application to CO2 capture[J]. Science, 2008, 319(5865): 939-943. |
| 34 | PARK K S, ZHENG N, ADRIEN PCÔTÉ, et al, et al. Exceptional chemical and thermal stability of zeolitic imidazolate frameworks[J]. Proc Natl Acad Sci, 2006, 103(27):10186-10191. |
| 35 | SAIUNKHE R R, YONG C, TANG J, et al. A high-performance supercapacitor cell based on ZIF-8-derived nanoporous carbon using an organic electrolyte[J]. Chem Commun, 2016, 52(26): 4764-4767. |
| 36 | LIU J, ZHAO Z, SHAO P, et al. Activation of peroxymonosulfate with magnetic Fe3O4-MnO2 core-shell nanocomposites for 4-chlorophenol degradation[J]. Chem Eng J, 2015, 262: 854-861. |
| 37 | LI X M, YAN X L, HU X Y, et al. Yolk-shell ZIFs@SiO2 and its derived carbon composite as robust catalyst for peroxymonosulfate activation[J]. J Environ Manage, 2020, 262: 110299. |
| 38 | WANG C, WANG H, LUO R, et al. Metal-organic framework one-dimensional fibers as efficient catalysts for activating peroxymonosulfate[J]. Chem Eng J, 2017, 330: 262-271. |
| 39 | CONG J, LEI F, ZHAO T, et al. Two Co-zeolite imidazolate frameworks with different topologies for degradation of organic dyes via peroxymonosulfate activation[J]. J Solid State Chem, 2017, 256: 10-13. |
| 40 | LI H X, WAN J, MA Y, et al. Degradation of refractory dibutyl phthalate by peroxymonosulfate activated with novel catalysts cobalt metal-organic frameworks: mechanism, performance, and stability[J]. J Hazard Mater, 2016, 318: 154-163. |
| 41 | CHUI S S Y, LO S M F, CHARMANT J P H, et al. A chemically functionalizable nanoporous material [Cu3(TMA)2(H2O)3]n[J]. Science, 1999, 283(5405): 1148. |
| 42 | TIAN T, ZENG Z, VULPE D, et al. A sol-gel monolithic metal-organic framework with enhanced methane uptake[J]. Nat Mater, 2018, 17(2): 174-179. |
| 43 | ZHANG J Y, SU C L, XIE X J, et al. Enhanced visible light photocatalytic degradation of dyes in aqueous solution activated by HKUST-1: performance and mechanism[J]. RSC Adv, 2020, 10(61): 37028-37034. |
| 44 | ZHAO Y W, WANG J E, PEI R J. Micron-sized ultrathin metal-organic framework sheet[J]. J Am Chem Soc, 2020, 142: 10331-10336. |
| 45 | HOU S Y, ZHANG H Z, WANG P, et al. Multiwall carbon nanotube decorated hemin/Mn-MOF towards BPA degradation through PMS activation[J]. J Environ Chem Eng, 2022, 10(5): 108426. |
| 46 | LI H X, YANG Z X, LU S. Nano-porous bimetallic CuCo-MOF-74 with coordinatively unsaturated metal sites for peroxymonosulfate activation to eliminate organic pollutants: performance and mechanism[J]. Chemosphere, 2021, 273: 129643. |
| 47 | ZHENG H A, ZHOU Y, WANG D R, et al. Surface-functionalized PVDF membranes by facile synthetic Cu-MOF-74 for enhanced contaminant degradation and antifouling performance[J]. Colloids Surf A, 2022, 651: 129640. |
| 48 | WEN Q, LI D, LI H M, et al. Synergetic effect of photocatalysis and peroxymonosulfate activated by Co/Mn-MOF-74@ g-C3N4 Z-scheme photocatalyst for removal of tetracycline hydrochloride[J]. Sep Purif Technol, 2023, 313: 123518. |
| 49 | GONG Y, YANG B, ZHANG H, et al. A g-C3N4/MIL-101(Fe) heterostructure composite for highly efficient BPA degradation with persulfate under visible light irradiation[J]. J Mater Chem A, 2018, 6(46): 23703-23711. |
| 50 | WANG S, LIAO Y, FARHA O K, et al. Metal-organic framework nanoparticles[J]. Chem Mater, 2018, 30(37): 1800202. |
| 51 | JIAO L, SEOW J, SKINNER W S, et al. Metal-organic frameworks: structures and functional applications[J]. Mater Today, 2019, 27: 43-68. |
| 52 | SONG G, WANG Z, WANG L, et al. Preparation of MOF(Fe) and its catalytic activity for oxygen reduction reaction in an alkaline electrolyte[J]. Chin J Catal, 2014, 35(2): 185-195. |
| 53 | 张付昭, 许文莉, 李良军, 等. 一种新型阴离子型金属有机框架微孔材料的制备及其超声法快速合成研究[J]. 当代化工研究, 2022, 20: 55-57. |
| ZHANG F Z, XU W L, LI L J, et al. Fabrication of anionic-pillared metal organic frameworks and the study of rapidsynthesis through ultrasonic method[J]. Mod Chem Res, 2022, 20: 55-57. | |
| 54 | YAO B, LUA S K, LIM H S, et al. Rapid ultrasound-assisted synthesis of controllable Zn/Co-based zeolitic imidazolate framework nanoparticles for heterogeneous catalysis[J]. Microporous Mesoporous Mater, 2020, 314: 110777. |
| 55 | XIONG Z K, JIANG Y N, WU Z L, et al. Synthesis strategies and emerging mechanisms of metal-organic frameworks for sulfate radical-based advanced oxidation process: a review[J]. Chem Eng J, 2021, 421(2): 127863. |
| 56 | TAO Z, ZHANG X, WANG S, et al. Spatial confinement of Co3O4 catalyst in hollow metal-organic framework as nanoreactor for improved degradation of organic pollutant[J]. Environ Sci Technol, 2015, 49(4): 2350-2357. |
| 57 | WANG D K, ZENG H, CHEN S Q, et al. Selective regulation of peroxydisulfate-to-hydroxyl radical for efficient in-situ chemical oxidation over Fe-based metal-organic frameworks under visible light[J]. J Catal, 2022, 406: 1-8. |
| 58 | DING S, WAN J Q, MA Y W, et al. Water stable SiO2-coated Fe-MOF-74 for aqueous dimethyl phthalate degradation in PS activated medium[J]. J Hazard Mater, 2021, 411: 125194. |
| 59 | CHEN X, OH W D, LIM T T. Graphene- and CNTs-based carbocatalysts in persulfates activation: material design and catalytic mechanisms[J]. Chem Eng J, 2018, 354: 941-976. |
| 60 | ANDREW LIN K Y, CHANG H A, HSU C J. Iron-based metal organic framework, MIL-88A, as a heterogeneous persulfate catalyst for decolorization of rhodamine B in water[J]. RSC Adv, 2015, 5: 32520-32530. |
| 61 | LI H C, SHAN C, PAN B C. Fe(Ⅲ)-doped g-C3N4 mediated peroxymonosulfate activation for selective degradation of phenolic compounds via high-valent iron-oxo species[J]. Environ Sci Technol, 2018, 52(4): 2197-2205. |
| 62 | ZHANG K, SUN D D, MA C, et al. Activation of peroxymonosulfate by CoFe2O4 loaded on metal-organic framework for the degradation of organic dye[J]. Chemosphere, 2020, 241: 125021. |
| 63 | 曾慧. 铁基金属有机框架(Fe-MOFs)光催化活化过硫酸盐及降解有机污染物的研究[D]. 南昌: 南昌航空大学, 2021. |
| ZENG H. Study on photocatalytic activation of persulfate and degradation of organic pollutants by iron-based metal-organic framework (Fe-MOFs)[D]. Nanchang: Nanchang Hangkong University, 2021. | |
| 64 | WANG J M, WAN J Q, MA Y W, et al. Metal-organic frameworks MIL-88A with suitable synthesis conditions and optimal dosage for effective catalytic degradation of orange G through persulfate activation[J]. RSC Adv, 2016, 6(113): 112502-112511. |
| 65 | HASHEMZADEH B, ALAMGHOLILOO H, NOROOZI PESYAN N, et al. Degradation of ciprofloxacin using hematite/MOF nanocomposite as a heterogeneous fenton-like catalyst: a comparison of composite and core-shell structures[J]. Chemosphere, 2021, 281: 130970. |
| 66 | AHN Y Y, YUN E T, SEO J W, et al. Activation of peroxymonosulfate by surface-loaded noble metal nanoparticles for oxidative degradation of organic compounds[J]. Environ Sci Technol, 2016, 50(18): 10187-10197. |
| 67 | ZHANG N, FENG X B, RAO D W, et al. Lattice oxygen activation enabled by high-valence metal sites for enhanced water oxidation[J]. Nat Commun, 2020, 11: 4066. |
| 68 | DU N J, LIU Y, LI Q J, et al. Peroxydisulfate activation by atomically-dispersed Fe-Nx on N-doped carbon: mechanism of singlet oxygen evolution for nonradical degradation of aqueous contaminants[J]. Chem Eng J, 2021, 413: 127545. |
| 69 | 何菁菁. MOFs衍生Fe-N共掺杂多孔碳活化过一硫酸盐降解有机污染物的性能与机理[D]. 杭州: 浙江大学, 2021. |
| HE J J. Activation of peroxymonosulfate by MOFs derived Fe-N co-doped porous carbon for degradation of organic pollutants[D]. Hangzhou: Zhejiang University, 2021. | |
| 70 | 曲云鹏, 张丙兴, 石金彪, 等. 钛基金属-有机框架材料的改性及其催化性能研究[J]. 化工学报, 2020, 71(1): 283-289. |
| QU Y P, ZHANG B X, SHI J B, et al. Study on modification of titanium-based metal-organic framework and catalytic performance[J]. CIESC J, 2020, 71(1): 283-289. | |
| 71 | LI H X, CHEN X, LI N, et al. Synthesis of bimetallic FeCu-MOF and its performance as catalyst of peroxymonosulfate for degradation of methylene blue[J]. Materials, 2022, 15: 7252. |
| 72 | 陈吉冲. 氨基修饰的金属有机框架材料NH2-MIL88B(Fe)活化过硫酸氢盐降解酸性橙Ⅱ[D]. 武汉: 武汉纺织大学, 2020. |
| CHEN J C. Activation of peroxymonosulfate by amino-modified metal organic frameworks NH2-MIL88B(Fe) for the degradation of acid orange Ⅱ[D]. Wuhan: Wuhan Textile University, 2020. | |
| 73 | LIU C, WANG Y P, ZHANG Y T, et al. Enhancement of Fe@porous carbon to be an efficient mediator for peroxymonosulfate activation for oxidation of organic contaminants: incorporation NH2-group into structure of its MOF precursor[J]. Chem Eng J, 2018, 354: 835-848. |
| 74 | FLEMING M S, MANDAL T K, WALT D R. Nanosphere-microsphere assembly: methods for core-shell materials preparation[J]. Chem Mater, 2001, 13(6): 2210-2216. |
| 75 | LIU X R, LIU Y, QIN H H, et al. Selective removal of phenolic compounds by peroxydisulfate activation: inherent role of hydrophobicity and interface ROS[J]. Environ Sci Technol, 2022, 56(4): 2665-2676. |
| 76 | HU L X, DENG G H, LU W C, et al. Peroxymonosulfate activation by Mn3O4/metal-organic framework for degradation of refractory aqueous organic pollutant rhodamine B[J]. Chin J Catal, 2017, 38(8): 1360-1372. |
| 77 | KHOJASTEGI A, MOKHTARE A, MOSLEH I, et al. Catalytic activation of peroxymonosulfate using MnO2@quasi-MOF for singlet oxygen mediated degradation of organic pollutants in water[J]. Appl Catal A, 2022, 646: 118883. |
| [1] | Jian-Hua ZHENG, Zhen-Ping GUAN, Guo-Yu LIU, Xiang-Yu CAO, Shun-Ji ZHENG, Mei-Yan GUAN. Enhanced Photodegradation Performance of Flower-like MnCo2O4 with Persulfate Under Visible Light [J]. Chinese Journal of Applied Chemistry, 2023, 40(7): 1034-1043. |
| [2] | Xing-Quan XIONG, Hui ZHANG, Li-Zhu GAO. Progress in Chemical Modification and Application of Lignin [J]. Chinese Journal of Applied Chemistry, 2023, 40(6): 806-819. |
| [3] | Hai-Xiang XIU, Wan-Qiang LIU, Dong-Ming YIN, Yong CHENG, Chun-Li WANG, Li-Min WANG. Research Progress of AB2 Laves Phase Hydrogen Storage Alloys [J]. Chinese Journal of Applied Chemistry, 2023, 40(5): 640-652. |
| [4] | Xue-Bo LEI, Hui-Jing LIU, He-Yu DING, Guo-Dong SHEN, Run-Jun SUN. Research Progress on Photocatalysts for Degradation of Organic Pollutants in Printing and Dyeing Wastewater [J]. Chinese Journal of Applied Chemistry, 2023, 40(5): 681-696. |
| [5] | Wen-Jun SHI, Zhong-Hui SUN, Zhong-Qian SONG, XU-Jia NAN, Dong-Xue HAN, Li NIU. Research Progress of Layered Transition Metal Oxides Cathode Materials for Sodium-ion Batteries [J]. Chinese Journal of Applied Chemistry, 2023, 40(4): 583-596. |
| [6] | Jin LIN, Fang-Zhu WANG, Ling-Ling LYU. Preparation of Pseudo-boehamite from Industrial Materials and Its Application in Selective Hydrogenation of Isophorone [J]. Chinese Journal of Applied Chemistry, 2023, 40(1): 79-90. |
| [7] | Dan ZHANG, Run-Mei SHANG, Zhen-Tao ZHAO, Jun-Hua LI, Jin-Juan XING. Selective Oxidation of Methanol to Dimethoxymethane over V/Ce⁃Al2O3 Catalysts [J]. Chinese Journal of Applied Chemistry, 2022, 39(9): 1429-1436. |
| [8] | Bing-Gang CHEN, San-Rong LIU, Zi-Jiang JIANG, Xi-Fei YU. Preparation and Properties Characterization of Hydrophilic Polysiloxane and Polyvinyl Alcohol Composite as Skin Barrier Material [J]. Chinese Journal of Applied Chemistry, 2022, 39(8): 1224-1236. |
| [9] | Dong-Dong LI, Li QIN, Lu-Hua TANG, Wen-Hui GAO. Preparation and Application of Basic OrangeⅡImprinted Sensor [J]. Chinese Journal of Applied Chemistry, 2022, 39(7): 1052-1064. |
| [10] | Feng LI, Shi-Yu LU, Yu ZHANG, Li-Jun GUO, Xue ZHAI, Cui-Qin LI. Catalytic Properties of Silylated⁃Salicylaldimine Transition Metal Complexes Functionalized Nano⁃silica Catalysts in Ethylene Oligomerization [J]. Chinese Journal of Applied Chemistry, 2022, 39(6): 949-959. |
| [11] | Zhu-Ying ZHANG, De-Yan DU, Jia-Hua ZHOU, Dong-Jian SHI, Ming-Qing CHEN. Preparation and Properties of Mussel-Inspired Modified Gelatin-Based Hemostatic Sponge [J]. Chinese Journal of Applied Chemistry, 2022, 39(02): 247-257. |
| [12] | NIU Zhan-Ning, TANG Hao-Qing, ZHENG Chao, TIAN Tian, ZHENG Li-Yun. Study on [RESA]Br@-COOH@Fe3O4 with Density Functional Theory [J]. Chinese Journal of Applied Chemistry, 2021, 38(7): 825-835. |
| [13] | JIAO Zi-Xue, GAO Cheng-Zhe, SUN Shuo, WU Zhen-Xu, ZHANG Pei-Biao. Preparation, Characterization and Performance of Gelatin Modified Polyetheretherketone Microcarriers [J]. Chinese Journal of Applied Chemistry, 2021, 38(4): 0-0. |
| [14] | JIAO Zi-Xue, GAO Cheng-Zhe, SUN Shuo, WU Zhen-Xu, ZHANG Pei-Biao. Preparation, Characterization and Performance of Gelatin Modified Polyetheretherketone Microcarriers [J]. Chinese Journal of Applied Chemistry, 2021, 38(4): 390-397. |
| [15] | ZHANG Ming-Shan, LI Teng-Ya, LEI Yu, WU Yan, HE Shu-Hai. Determination of Two Sulfonamides in Environmental Water Based on Dispersivesolid-Phase Extraction with MIL-101(Cr) Metal-Organic Framework [J]. Chinese Journal of Applied Chemistry, 2021, 38(2): 236-244. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||