Chinese Journal of Applied Chemistry ›› 2022, Vol. 39 ›› Issue (5): 760-768.DOI: 10.19894/j.issn.1000-0518.210107
• Full Papers • Previous Articles Next Articles
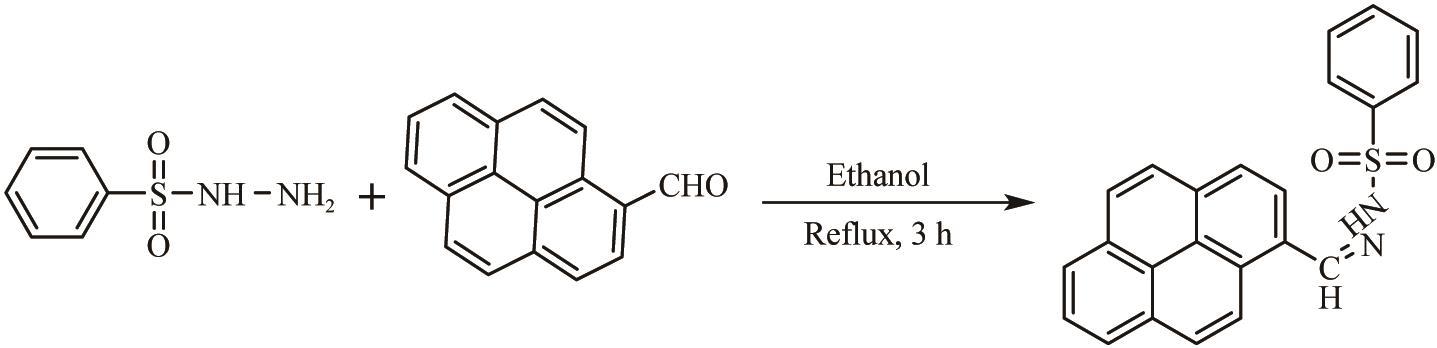
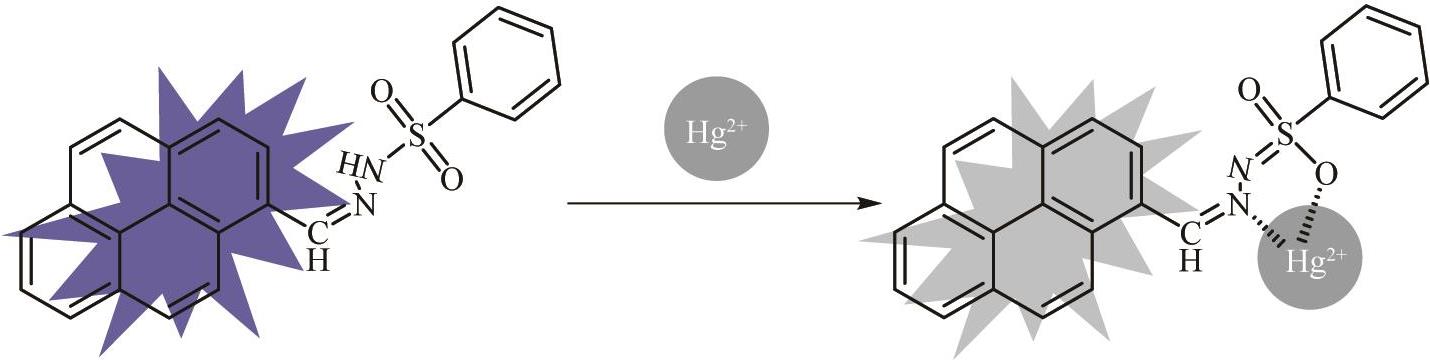
Synthesis of Sulfonylhydrazone Probe with High Selectivity and Rapid Identification of Hg(Ⅱ) Ion and Its Application in Adsorption
Song-Song XUE1, Zheng-Feng XIE1,2( ), Jia-Wei HE1, Tian-Yi ZHANG1, Bao-Ping XIA1, Yu-Qin LI1
), Jia-Wei HE1, Tian-Yi ZHANG1, Bao-Ping XIA1, Yu-Qin LI1
- 1.Oil & Gas Field Applied Chemistry Key Laboratory of Sichuan Province,College of Chemistry and Chemical Engineering,Southwest Petroleum University,Chengdu 610500,China
2.Research Institute of Industrial Hazardous Waste Disposal and Resource Utilization,Southwest Petroleum University,Chengdu 610500,China
-
Received:2021-03-10Accepted:2021-06-26Published:2022-05-01Online:2022-05-24 -
Contact:Zheng-Feng XIE -
About author:xiezhf@swpu.edu.cn
-
Supported by:Sichuan Youth Science and Technology Innovation Research Team Project(2020JDTD0018)
CLC Number:
Cite this article
Song-Song XUE, Zheng-Feng XIE, Jia-Wei HE, Tian-Yi ZHANG, Bao-Ping XIA, Yu-Qin LI. Synthesis of Sulfonylhydrazone Probe with High Selectivity and Rapid Identification of Hg(Ⅱ) Ion and Its Application in Adsorption[J]. Chinese Journal of Applied Chemistry, 2022, 39(5): 760-768.
share this article
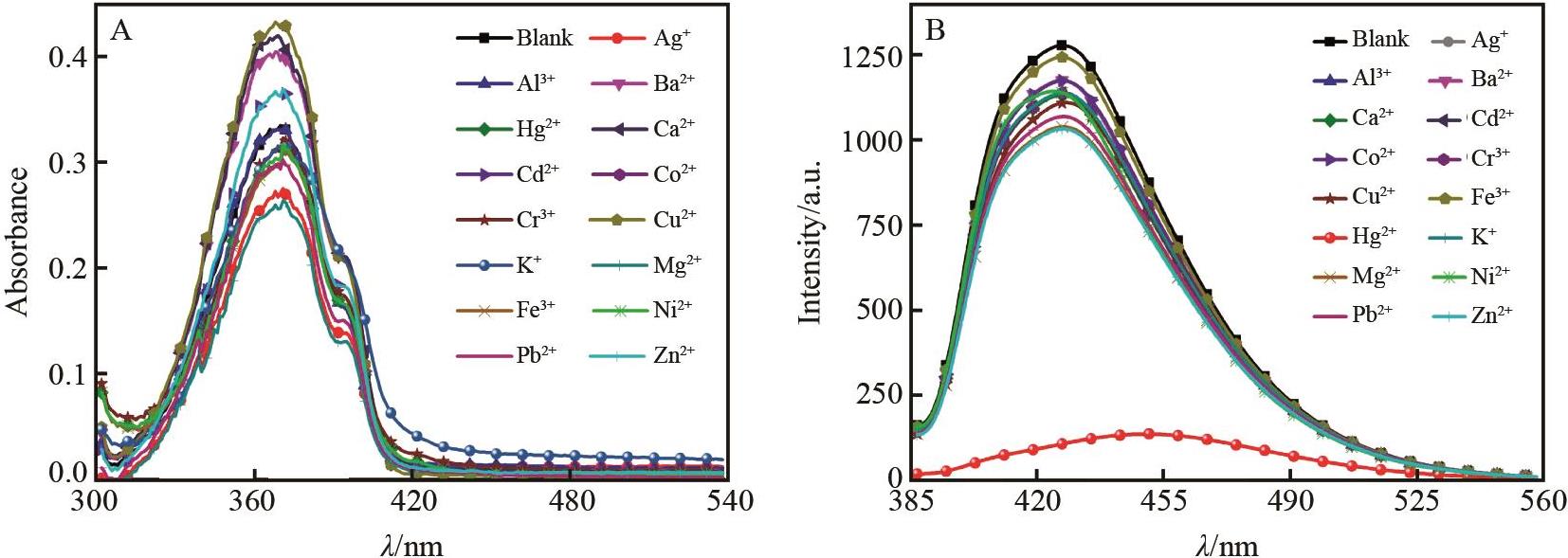
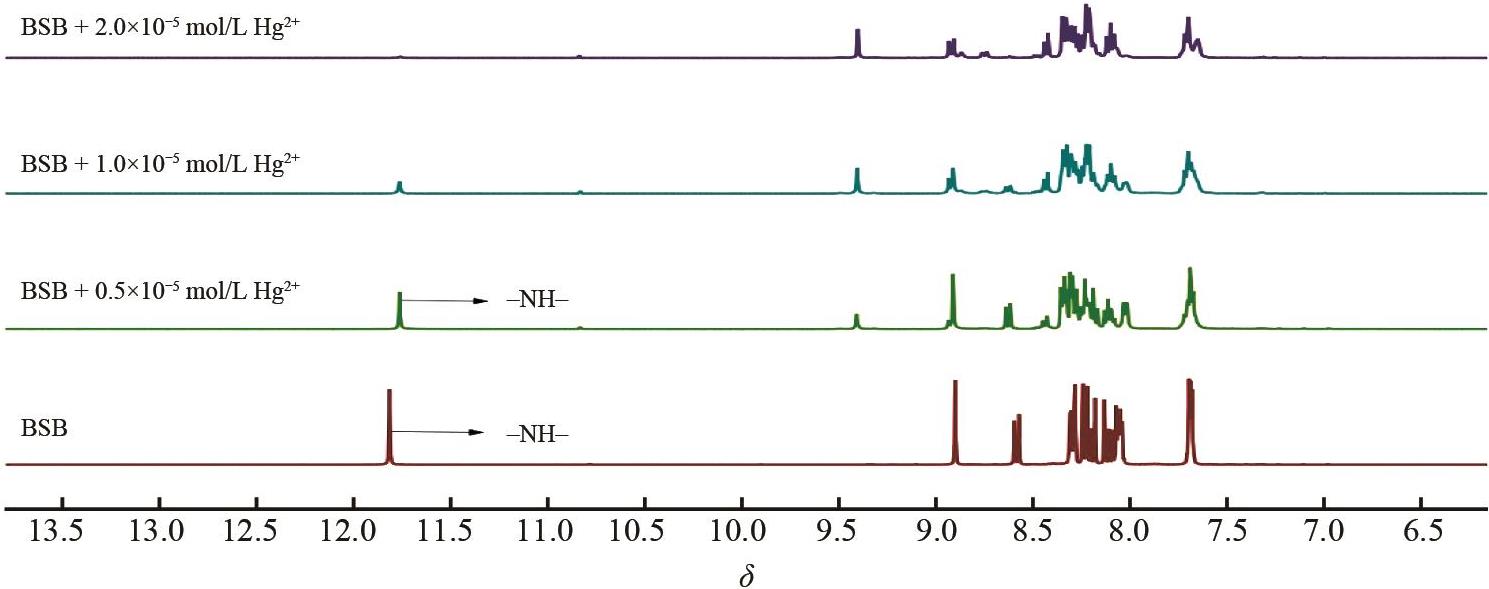
Fig.1 (A) Various cations (1.0×10-4 mol/L) are added to BSB (1.0×10-5 mol/L) DMSO/H2O (volume ratio 1∶1,pH=7) and (B) fluorescence emission spectra (excitation wavelength 387 nm, slit width 5 nm × 5 nm)
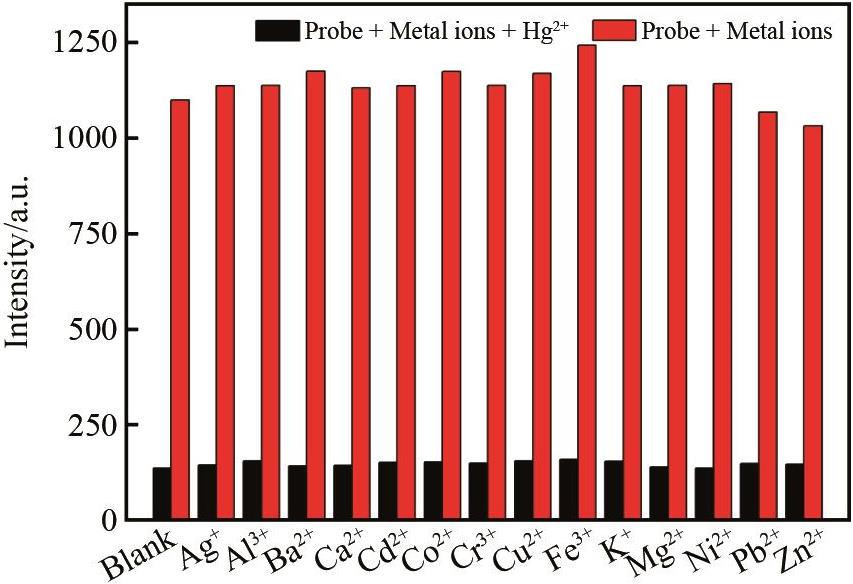
Fig.2 Competitive diagram of Hg2+ (1.0×10-4 mol/L) in the presence of various cations (1.0×10-4 mol/L) in DMSO/H2O (V∶V = 1∶1,pH = 7) solution of BSB (excitation wavelength 387 nm, slit width 5 nm × 5 nm)
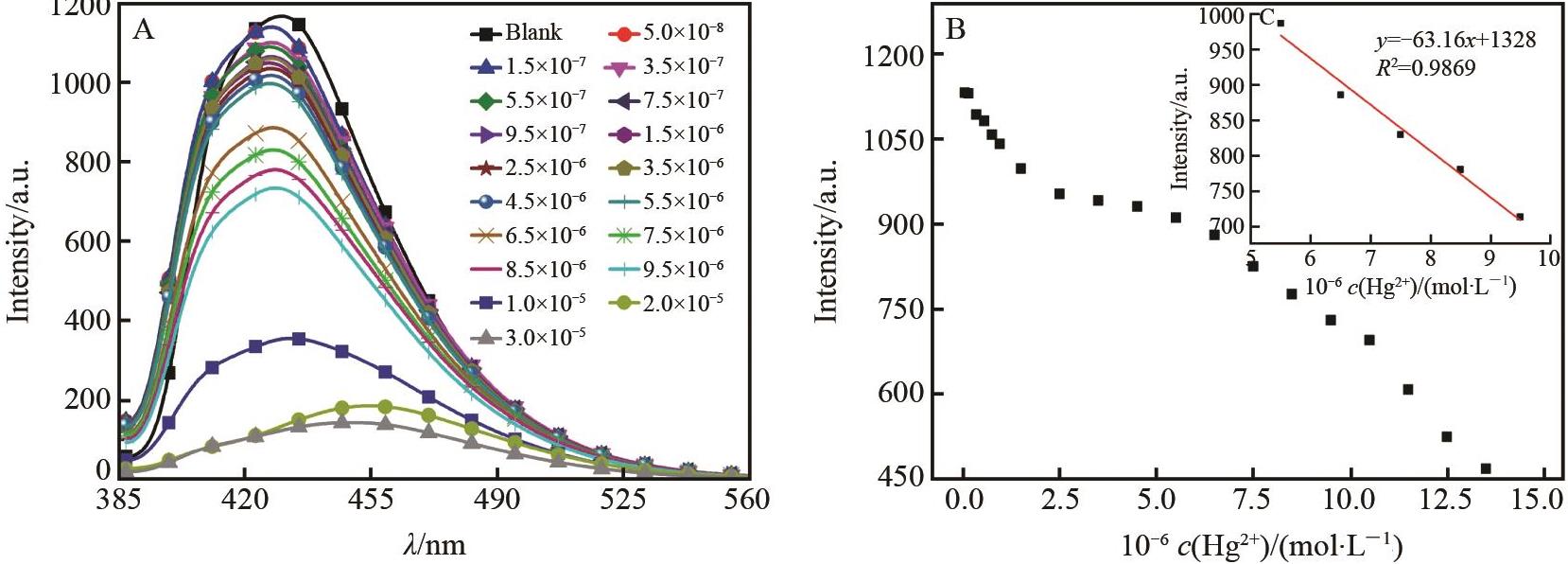
Fig.3 (A) Fluorescence spectra of BSB (1×10-5 mol/L) in DMSO/H2O (volume ratio1∶1,pH=7) for Hg2+ of different concentrations and (b) fluorescence dot graphs of BSB (1×10-5 mol/L) for Hg2+ of different concentrations
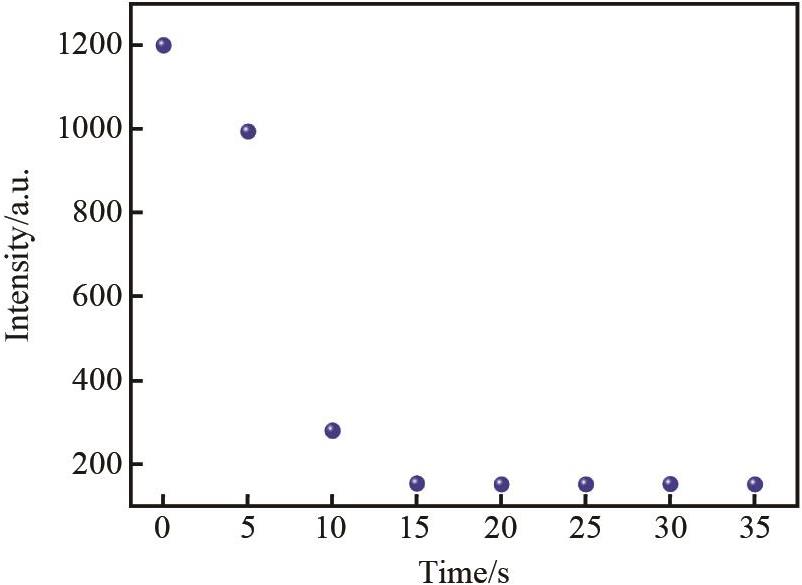
Fig.4 Time response of BSB (1.0×10-5 mol/L) to Hg2+ (1.0×10-4 mol/L) in DMSO/H2O (volume ratio 1∶1,pH=7)solution (excitation wavelength 387 nm, slit width 5 nm × 5 nm)
样品 Sample | 加入 Added/(mol·L-1) | 测定 Found/(mol·L-1) | 回收率 Recovery/% |
|---|---|---|---|
自来水 Tape water | 1.0×10-6 | 0.97×10-6 | 97 |
| 5.0×10-6 | 5.13×10-6 | 102.6 | |
| 1.0×10-5 | 1.12×10-5 | 112 | |
| 5.0×10-5 | 4.89×10-5 | 97.8 | |
矿泉水 Mineral water | 1.0×10-6 | 1.02×10-6 | 102 |
| 5.0×10-6 | 5.17×10-6 | 103.4 | |
| 1.0×10-5 | 0.98×10-5 | 98 | |
| 5.0×10-5 | 4.82×10-5 | 96.4 | |
河水 River water | 1.0×10-6 | 1.26×10-6 | 126 |
| 5.0×10-6 | 5.62×10-6 | 112.4 | |
| 1.0×10-5 | 0.93×10-5 | 93 | |
| 5.0×10-5 | 5.21×10-5 | 104.2 |
Table 1 Determination of Hg2+ in different water samples
样品 Sample | 加入 Added/(mol·L-1) | 测定 Found/(mol·L-1) | 回收率 Recovery/% |
|---|---|---|---|
自来水 Tape water | 1.0×10-6 | 0.97×10-6 | 97 |
| 5.0×10-6 | 5.13×10-6 | 102.6 | |
| 1.0×10-5 | 1.12×10-5 | 112 | |
| 5.0×10-5 | 4.89×10-5 | 97.8 | |
矿泉水 Mineral water | 1.0×10-6 | 1.02×10-6 | 102 |
| 5.0×10-6 | 5.17×10-6 | 103.4 | |
| 1.0×10-5 | 0.98×10-5 | 98 | |
| 5.0×10-5 | 4.82×10-5 | 96.4 | |
河水 River water | 1.0×10-6 | 1.26×10-6 | 126 |
| 5.0×10-6 | 5.62×10-6 | 112.4 | |
| 1.0×10-5 | 0.93×10-5 | 93 | |
| 5.0×10-5 | 5.21×10-5 | 104.2 |
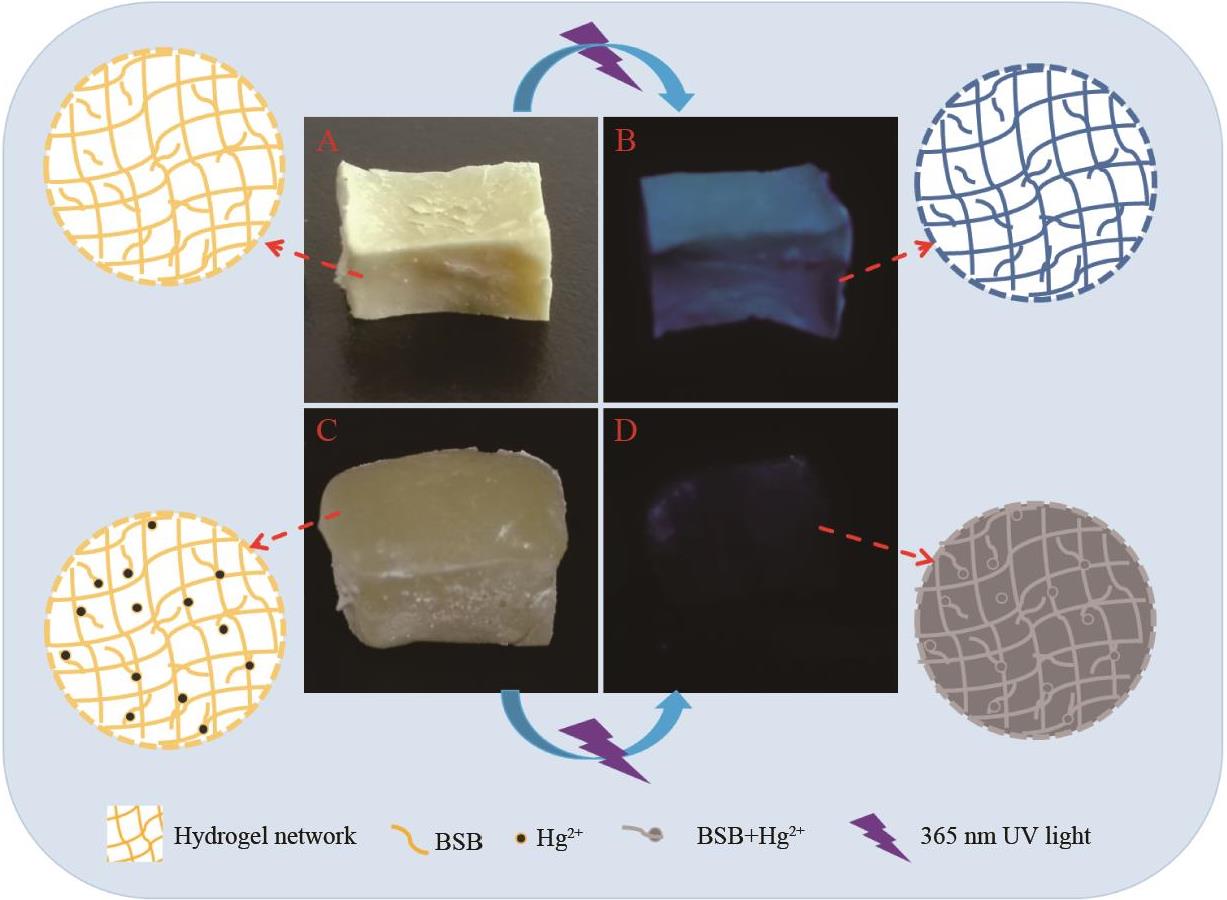
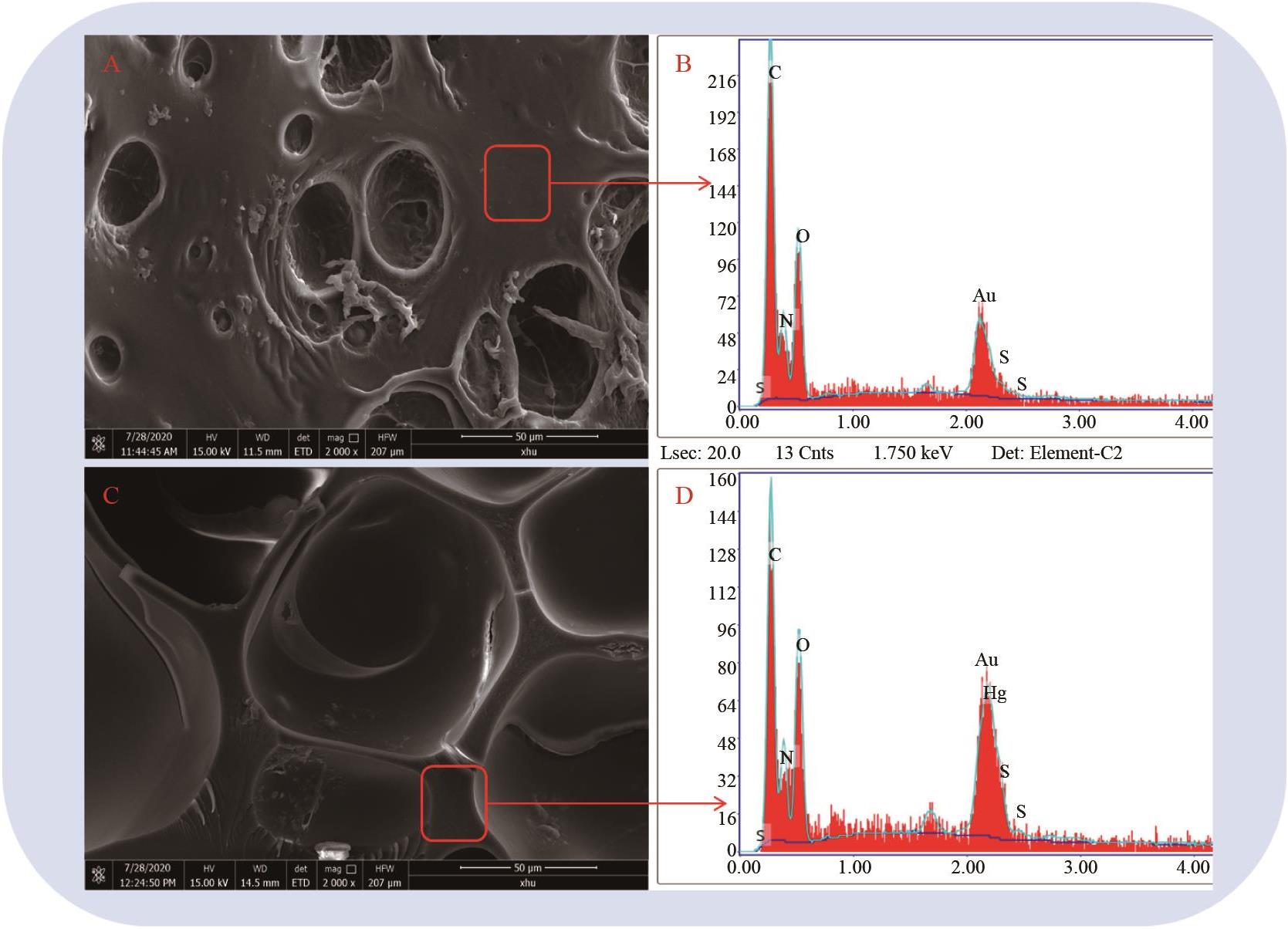
Fig.7 (A) and (B) are the pictures of PAM?BSB under natural light and ultraviolet light before adsorption respectively; (C) and (D) are the pictures of PAM-BSB under natural light and ultraviolet light after adsorption, respectively
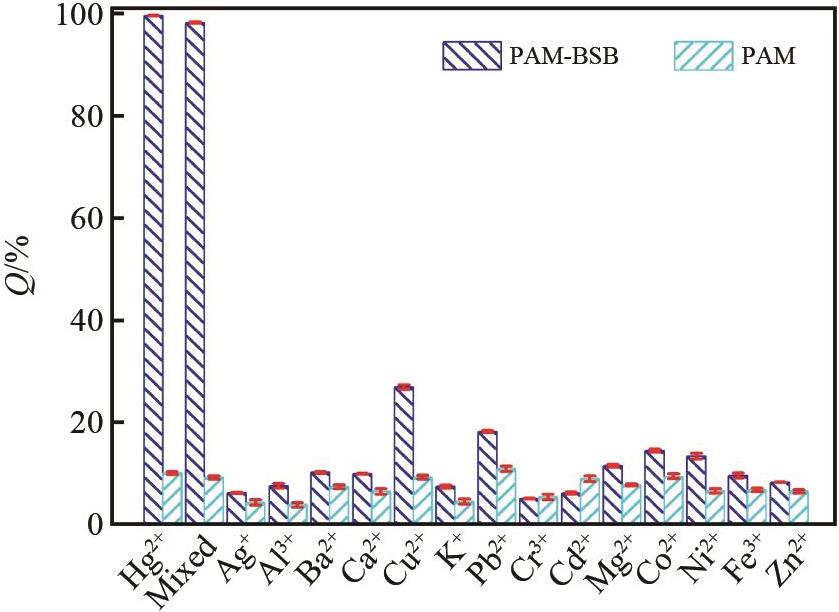
Fig.8 The adsorption performance of PAM and PAM?BSB on different metal ions (0.1 mol/L) (Mixed: shows the adsorption performance of PAM and PAM-BSB on mercury ions when other ions coexist)
| 1 | CARTER K P, YOUNG A M, PALMER A E. Fluorescent sensors for measuring metal ions in living systems[J]. Chem Rev, 2014, 114(8): 4564-4601. |
| 2 | PARAMANIK B, BAIN D, PATRA A. Making and breaking of DNA-metal base pairs: Hg2+ and Au nanocluster based off/on probe[J]. Indian J Chem A, 2016, 120(30): 17127-17135. |
| 3 | MAHATO P, SAHA S, SURESH E, et al. Ratiometric detection of Cr3+ and Hg2+ by a naphthalimide-rhodamine based fluorescent probe[J]. Inorg Chem, 2012, 51: 1769-1777. |
| 4 | ZHAO Y, LI Y, LONG Y, et al. Highly selective fluorescence turn-on determination of fluoride ions via chromogenic aggregation of a silyloxy-functionalized salicylaldehyde azine[J]. Tetrahedron Lett, 2017, 58(13): 1351-1355. |
| 5 | WANG Y, LIN L, LIU J, et al. Ferric ion induced enhancement of ultraviolet vapour generation coupled with atomic fluorescence spectrometry for the determination of ultratrace inorganic arsenic in surface water[J]. The Analyst, 2016, 141(4): 1530-1536. |
| 6 | YANG L, SU Y, GENG Y, et al. A red-emitting fluorescent probe for the detection of Hg2+ in aqueous medium, living cells and organisms with a large Stokes shift[J]. Org Biomol Chem, 2018, 16(27): 5036-5042. |
| 7 | 胡明明, 樊江莉, 李宏林, 等.荧光探针可视化检测水溶液中铜离子[J].分析化学, 2011, 39(8): 1195-1200. |
| HU M M, FAN J L, LI H L, et al.A sensitive probe for determination of Cu2+ with optical detection in aqueous solution[J]. Chinese J Anal Chem, 2011, 39(8): 1195-1200. | |
| 8 | 戚少龙, 杜建时, 李容杭, 等. 水溶性荧光探针的合成及对亚硫酸氢根和pH值的双重检测[J]. 分析化学, 2020, 48(3): 347-354. |
| QI S L, DU J S, LI R H, et al. Synthesis of water-soluble fluorescent probe for dual detection of HSO3 and pH value[J]. Chinese J Anal Chem, 2020, 48(3): 347-354. | |
| 9 | XU J, WANG Z, LIU C, et al. A highly selective colorimetric and long-wavelength fluorescent probe for the detection of Hg2+[J]. Luminescence, 2018, 33(6): 1122-1127. |
| 10 | 李 迪, 孙建强, 许丙嵩, 等. 基于查尔酮衍生物的铜离子探针的设计合成[J]. 应用化学, 2020, 37(11): 1268-1275. |
| LI D, SUN J Q, XU B S, et al. Design and synthesis of chalcone-based derivative for sensing Cu2+ ion[J]. Chinese J Appl Chem, 2020, 37(11): 1268-1275. | |
| 11 | 陈丽娟, 刘仁勇, 赵 丹, 等. 聚乙烯亚胺修饰的碳点荧光探针用于检测汞离子[J]. 分析化学, 2020, 48(8): 1067-1074. |
| CHEN L J, LIU R Y, ZHAO D, et al. A novel fluorescent probe for mercury ion detection based on polyethyleneimine modified carbon dots [J]. Chinese J Anal Chem, 2020, 48(8): 1067-1074. | |
| 12 | HUO F J, ZHANG Y Q, KANG J, et al. A novel alkyne compound as a Pd(Ⅱ) fluorescent probe in aqueous medium and its bioimaging[J]. Sens Actuators B: Chem, 2017, 243: 429-434. |
| 13 | ZHANG H F, YIN C X, LIU T, et al. Selective “off-on” detection of magnesium (Ⅱ) ions using a naphthalimide-derived fluorescent probe[J]. Dyes Pigm, 2017, 146: 344-351. |
| 14 | ZHU Z F, DING H C, WANG Y S, et al. A ratiometric and colorimetric fluorescent probe for the detection of mercury ion based on rhodamine and quinoline‑benzothiazole conjugated dyad[J]. J Photochem Photobiol A, 2020, 400: 112657-112666. |
| 15 | REENA V R, SMRITILEKHA B, PRASENJIT M, et al. Mechanochemical synthesis of a fluorescein-based sensor for the selective detection and removal of Hg2+ ions in industrial effluents[J]. ACS Omega, 2020, 5(10): 4982-4990. |
| 16 | PAVEL A P, YURI V F, OLGA A F. Selective fluorometric sensing of Hg2+ in aqueous solution by the inhibition of PET from dithia-15-crown-5 ether receptor conjugated to 4-amino-1,8-naphthalimide fluorophore[J]. J Photochem Photobiol A, 2018,364: 124-129. |
| 17 | ZHANG H F, LIU T, YIN C X, et al. A novel ratiometric fluorescent probe based on 1,8-naphthalimide for the detection of Ho3+ and its bioimaging[J]. Spectrochim Acta A, 2017, 174: 230-235. |
| 18 | ZHANG H F, YIN C X, LIU T, et al. “Turn-on” fluorescent probe detection of Ca2+ ions and applications to bioimaging[J]. Spectrochim Acta A, 2017, 180: 211-216. |
| 19 | PANG X, GAO L, FENG H, et al. A peptide-based multifunctional fluorescent probe for Cu2+, Hg2+ and biothiols[J]. New J Chem, 2018, 42(12): 15770-15777. |
| 20 | CUI T, YU S, CHEN Z, et al. Rational design of fluorescent probe for Hg2+ by changing the chemical bond type[J]. RSC Adv, 2018, 8: 12276-12281. |
| 21 | HUANG X, LU Z, WANG Z, et al. A colorimetric and turn-on fluorescent chemosensor for selectively sensing Hg2+ and its resultant complex for fast detection of I- over S2-[J]. Dyes Pigm, 2016, 128: 33-40. |
| 22 | GE Y, XING X, LIU A, et al. A novel imidazo[1,5-a]pyridine-rhodamine FRET system as an efficient ratiometric fluorescent probe for Hg2+ in living cells[J]. Dyes Pigm, 2017, 146: 136-142. |
| 23 | ZHANG L, SHI Y, SHENG Z, et al. Bioluminescence imaging of selenocysteine in vivo with a highly sensitive probe[J]. ACS Sens, 2019, 4(12): 1234-1242. |
| 24 | MIN K S, MANIVANNAN R, SON Y A. Rhodamine-fluorene based dual channel probe for the detection of Hg2+ ions and its application in digital printing[J]. Sens Actuators B: Chem, 2018, 261: 545-552. |
| 25 | ZHANG Y, LENG J. Theoretical studies on two-photon fluorescent Hg2+ probes based on the coumarin-rhodamine system[J]. Sensors-Basel, 2017, 17(7): 346-258. |
| 26 | JESUS S, IXONE E, BINGEN G M, et al. Phase-selective staining of model and cell membranes, lipid droplets and lipoproteins with fluorescent solvatochromic pyrene probes[J]. BBA-Biomembranes, 2021, 1863(1): 183470-183479. |
| 27 | YUE Y Y, XIE Z F, CHU Y C, et al. A novel fluorescent probe based on spiro[chromeno[2,3-c]pyrazole-4,1-[2]benzofuran]-3-one for detecting copper(Ⅱ) ions in aqueous solution[J]. Russ J Org Chem, 2019, 55(6): 866-873. |
| 28 | CHU Y C, XIE Z F, YUE Y Y, et al. New fast, highly selective probe with both aggregation-induced emission enhancement and intramolecular charge-transfer characteristics for homocysteine detection[J]. ACS Omega, 2019, 4(3): 5367-5373. |
| 29 | REN X D, XIE Z F, WU H, et al. Multiple-responsive organogels with self-colorimetric chemo sensing responsiveness towards Hg2+ ions[J]. Colloid Surf A, 2019, 583: 124003-124008. |
| 30 | 薛松松, 解正峰, 褚义成, 等. 高选择性快速检测Hg2+的磺酰腙型探针的合成及在吸附和HeLa细胞中的应用[J]. 有机化学, 2021, 41(3): 1138-1145. |
| XUE S S, XIE Z F, CHU Y C, et al. Synthesis of sulfonylhydrazone type probe with high selectivity for rapid detection of mercury and its application in adsorption and HeLa cells[J]. Chinese J Org Chem, 2021, 41(3): 1138-1145. | |
| 31 | XIE Z F, KONG X J, FENG L, et al. A novel highly selective probe with both aggregation-induced emission enhancement and intramolecular charge transfer characteristics for CN- detection[J]. Sens Actuators B: Chem, 2018, 257: 154-165. |
| 32 | LI Y Q, SHI W, MA J C, et al. A novel optical probe for Hg2+ in aqueous media based on mono-thiosemicarbazone Schiff base[J]. Spectrochim Acta A, 2017, 338: 1-7. |
| [1] | Ning YUAN, Jie MA, Jin-Ling ZHANG, Jian-Sheng ZHANG. Preparation of PCN-6(M) Bimetallic Organic Framework Materials by Steam-assisted Method and Their CO2 and CH4 Adsorption Performance [J]. Chinese Journal of Applied Chemistry, 2023, 40(6): 896-903. |
| [2] | Chen-Li HAO, Qing-Wei DING, Shi-Chang JIA, Yang-Bo MAO, Song-Bai WANG, Jun MA. Adsorption of Methylene Blue on Titanate Nanotubes Modified with Lipoic Acid [J]. Chinese Journal of Applied Chemistry, 2023, 40(5): 749-757. |
| [3] | Peng BU, Hong-Liang LI. Preparation and Upconversion Luminescence Study of Rare Earth Yb3+/Tm3+ Doped NaGd(MO4)2 Phosphors [J]. Chinese Journal of Applied Chemistry, 2023, 40(3): 374-379. |
| [4] | Jia-Mei GENG, Su-Fang MA, Wen LIU, Hai-Peng DIAO, Zhi-Fang WU, Si-Jin LI. Liver-Targeted Fluorescent Probes for Specific Detection of ONOO- in HepG2 Cells [J]. Chinese Journal of Applied Chemistry, 2023, 40(3): 441-448. |
| [5] | Qin ZHANG, Wen-Bin LIU, Li-Jiao FAN, Yu-Ming XIE, Guo-Lin HUANG. Research Progress in the Preparation of Functionalized Mesoporous Silica and Its Application in Adsorption and Separation of Uranium from Water [J]. Chinese Journal of Applied Chemistry, 2023, 40(2): 169-187. |
| [6] | En-Tong WANG, Lin-Fang YANG. Preparation and Properties of LiNi0.6Co0.2Mn0.2O2 Cathode Material for High Specific Capacity Lithium Ion Battery [J]. Chinese Journal of Applied Chemistry, 2022, 39(8): 1209-1215. |
| [7] | Yue-Hua ZHAO, Da-Peng WANG. Coadsorption Kinetics of Amino‑Functionalized Graphene Oxide and Fatty Acids at the Water/Oil Interface [J]. Chinese Journal of Applied Chemistry, 2022, 39(8): 1274-1284. |
| [8] | Qiao-Zhi GUO, Zhen-Hua YANG, Yue-Xia ZHANG, Ya-Ting MENG, Yu-Juan CAO, Xuan-Sen SUN, Qi-Qi ZHANG, Shao-Min SHUANG, Chuan DONG. Synthesis and Applications of Graphene Quantum Dots Derived from Citric Acid [J]. Chinese Journal of Applied Chemistry, 2022, 39(6): 888-899. |
| [9] | You-San ZHOU, Yi-Cheng ZHANG, Si-Yu WU, Fei ZHA, De-Jun CHEN, Yue CHANG. Catalytic Performance of Cu‑Doped Calcium Aluminum Layered Double Hydrotalcites for the Oxidation of Cumene to Cumene Hydrogen Peroxide [J]. Chinese Journal of Applied Chemistry, 2022, 39(6): 941-948. |
| [10] | Cong-Jun CAO, Han-Xiao MA, Cheng-Min HOU, Xiao-Jian DING, Biao GUAN. Adsorption of Cu(Ⅱ) from Solution by Modified Magnetic Ethyl Cellulose [J]. Chinese Journal of Applied Chemistry, 2022, 39(6): 969-979. |
| [11] | Jing ZHOU, Yu-Xuan CHEN, Jun-Ming MA, Xiao-Fei ZHU, De-Feng ZHOU. Effect of MgO and Fe2O3 Incorporation on the Microstructure and Electrochemical Performance of GDCSi System [J]. Chinese Journal of Applied Chemistry, 2022, 39(5): 809-818. |
| [12] | Hui DU, Chen-Yang YAO, Hao PENG, Bo JIANG, Shun-Xiang LI, Jun-Lie YAO, Fang ZHENG, Fang YANG, Ai-Guo WU. Applications of Transition Metal⁃doped Iron⁃based Nanoparticles in Biomedicine [J]. Chinese Journal of Applied Chemistry, 2022, 39(3): 391-406. |
| [13] | Rui HUANG, Chang-Qing YE, Ya-Jun LI, Mong-Feng CHIOU, Da-Liang LI, Hong-Li BAO. Progress of Mitochondria⁃Targeted Near⁃Infrared HClO/ClO- Fluorescent Probes [J]. Chinese Journal of Applied Chemistry, 2022, 39(3): 407-424. |
| [14] | Cheng-Lu ZHANG, Yi-Ming WANG, Zhi-Xuan REN, Lu LI, Yu-Qing LI, Fu-Lu SONG. Fluorescent Probe for Rapid Detection of H2S with Benzimidazole Naphthalimide as the Core [J]. Chinese Journal of Applied Chemistry, 2022, 39(3): 489-497. |
| [15] | Si-Wei YU, Liang-Peng WANG, Ri-Zhe JIN, Chuan-Qing KANG. Recognition of ClO- and Cellular Imaging with Xanthene-based Fluorescent Probes [J]. Chinese Journal of Applied Chemistry, 2022, 39(12): 1903-1911. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||