应用化学 ›› 2024, Vol. 41 ›› Issue (4): 484-495.DOI: 10.19894/j.issn.1000-0518.230373
• 综合评述 • 上一篇
石榴石Li7La3Zr2O12固态电解质电导率优化策略研究进展
- 吉林大学化学学院,无机合成与制备化学国家重点实验室,长春 130012
-
收稿日期:2023-11-28接受日期:2024-02-11出版日期:2024-04-01发布日期:2024-04-28 -
通讯作者:黄科科 -
基金资助:吉林大学研究生教育教学改革研究项目(2021JGZ08)
Research Progress of Optimizing Conductivity of Garnet-Type Solid Electrolyte Li7La3Zr2O12
Yi ZHANG, Yu-Tong CHEN, Jing-Yu SHI, Ke-Ke HUANG( )
)
- State Key Laboratory of Inorganic Synthesis and Preparative Chemistry,College of Chemistry,Jilin University,Changchun 130012,China
-
Received:2023-11-28Accepted:2024-02-11Published:2024-04-01Online:2024-04-28 -
Contact:Ke-Ke HUANG -
About author:kkhuang@jlu.edu.cn
-
Supported by:the Research Project on Postgraduate Education and Teaching Reform of Jilin University(2021JGZ08)
摘要:
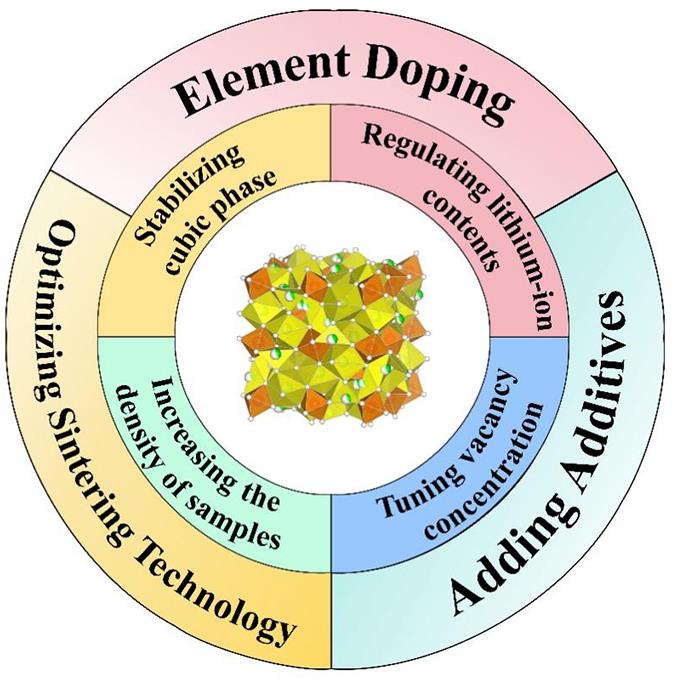
近几年,应用碳材料负极和有机电解液的液态锂离子电池(LIBs)的弊端日益凸显,电解液泄漏和过热燃烧等安全事故频发。 另外,传统的LIBs也无法满足当今社会对高能量密度电池的需求。 由于上述LIBs存在的诸多缺点,市场急需开发兼顾高能量密度与高安全性能的新型电池,现已发现可通过引入固态电解质的途径来实现。 固态锂电池(SSLBs)相较于传统的LIBs,具有较高的能量密度、较宽的工作温度范围和更高的安全性。 其中,固态电解质作为固态电池的重要元件之一,对电池性能的影响至关重要。 石榴石Li7La3Zr2O12凭借其高锂离子电导率(1×10-4~1×10-3 S/cm)、宽电化学窗口(9 V)以及对锂负极的高稳定性等优点,在众多固态电解质中脱颖而出。 本综述就提高石榴石基电解质锂离子电导率的研究予以总结: 首先,介绍了Li7La3Zr2O12晶体结构并分析了结构与电导率之间的关系; 然后,综述了几种提高电导率的方法,包括引入掺杂离子、改进烧结技术和添加烧结助剂等; 最后,提出了石榴石基固态电池的未来发展的挑战,为固态电解质相关研究提供参考。
中图分类号:
引用本文
张轶, 陈宇童, 师靖宇, 黄科科. 石榴石Li7La3Zr2O12固态电解质电导率优化策略研究进展[J]. 应用化学, 2024, 41(4): 484-495.
Yi ZHANG, Yu-Tong CHEN, Jing-Yu SHI, Ke-Ke HUANG. Research Progress of Optimizing Conductivity of Garnet-Type Solid Electrolyte Li7La3Zr2O12[J]. Chinese Journal of Applied Chemistry, 2024, 41(4): 484-495.
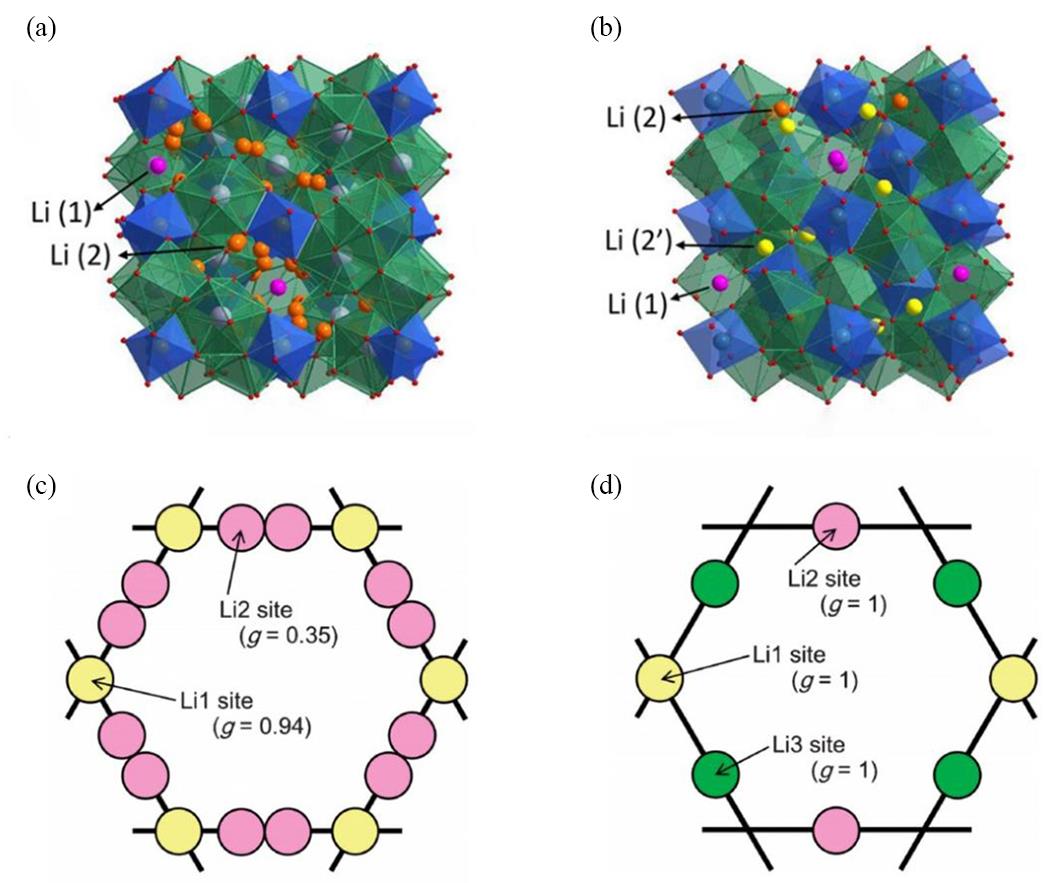
图1 (a)立方相和(b)四方相Li7La3Zr2O12结构[33]; (c)立方相和(d)四方相中锂排列的环状结构[31]
Fig.1 The structure of (a) cubic and (b) tetragonal Li7La3Zr2O12[33]; The loop structures constructed by Li atomic arrangement in (c) cubic and (d) tetragonal phase[31]

图3 (a) Li6.4A0.2La3Zr2O12(A=Al、Fe、Ga)的Nyquist图[42]; (b) LLZO中Zr位点掺杂离子半径和离子电导率总结[61]; (c)高密度和低密度LLZO中锂成核和传播机制示意图[62]
Fig.3 (a) Nyquist plots of Li6.4A0.2La3Zr2O12 (A=Al, Fe, Ga)[42]; (b) Summary of dopant ion radius and ion conductivity of Zr site in LLZO[61]; (c) Schematic illustration of Li nucleation and propagation mechanisms in high-density and low-density LLZO[62]
| 1 | LIU Y, TAO X, WANG Y, et al. Self-assembled monolayers direct a LiF-rich interphase toward long-life lithium metal batteries[J]. Science, 2022, 375(6582): 739-745. |
| 2 | YIN Y C, YANG J T, LUO J D, et al. A LaCl3-based lithium superionic conductor compatible with lithium metal[J]. Nature, 2023, 616(7955): 77-83. |
| 3 | KWAK W J, ROS Y, SHARON D, et al. Lithium-oxygen batteries and related systems: potential, status, and future[J]. Chem Rev, 2020, 120(14): 6626-6683. |
| 4 | CHENG X B, LIU H, YUAN H, et al. A perspective on sustainable energy materials for lithium batteries[J]. Sus Mat, 2021, 1(1): 38-50. |
| 5 | LI Y, SONG S, KIM H, et al. A lithium superionic conductor for millimeter-thick battery electrode[J]. Science, 2023, 381(6653): 50-53. |
| 6 | KALNAUS S, DUDNEY N J, WESTOVER A S, et al. Solid-state batteries: the critical role of mechanics[J]. Science, 2023, 381(6664): eabg5998. |
| 7 | MANTHIRAM A, YU X, WANG S. Lithium battery chemistries enabled by solid-state electrolytes[J]. Nat Rev Mater, 2017, 2(4): 1-16. |
| 8 | ABOUALI S, YIM C H, MERATI A, et al. Garnet-based solid-state Li batteries: from materials design to battery architecture[J]. ACS Energy Lett, 2021, 6(5): 1920-1941. |
| 9 | YANG X, ADAIR K R, GAO X, et al. Recent advances and perspectives on thin electrolytes for high-energy-density solid-state lithium batteries[J]. Energy Environ Sci, 2021, 14(2): 643-671. |
| 10 | MISHRA A K, CHALIYAWALA H A, PATEL R, et al. Inorganic solid state electrolytes: insights on current and future scope[J]. J Electrochem Soc, 2021, 168(8): 080536. |
| 11 | HE F, HU Z, TANG W, et al. Vertically heterostructured solid electrolytes for lithium metal batteries[J]. Adv Funct Mater, 2022, 32(25): 2201465. |
| 12 | BACHMAN J C, MUY S, GRIMAUD A, et al. Inorganic solid-state electrolytes for lithium batteries: mechanisms and properties governing ion conduction[J]. Chem Rev, 2016, 116(1): 140-162. |
| 13 | WANG C, KIM J T, WANG C, et al. Progress and prospects of inorganic solid-state electrolyte-based all-solid-state pouch cells[J]. Adv Mater, 2023, 35(19): 2209074. |
| 14 | WU Y, WANG S, LI H, et al. Progress in thermal stability of all-solid-state-Li-ion-batteries[J]. InfoMat, 2021, 3(8): 827-853. |
| 15 | WU J, LIU S, HAN F, et al. Lithium/sulfide all-solid-state batteries using sulfide electrolytes[J]. Adv Mater, 2021, 33(6): 2000751. |
| 16 | KIM K J, BALAISH M, WADAGUCHI M, et al. Solid-state Li-metal batteries: challenges and horizons of oxide and sulfide solid electrolytes and their interfaces[J]. Adv Energy Mater, 2021, 11(1): 2002689. |
| 17 | WANG C, LIANG J, KIM J T, et al. Prospects of halide-based all-solid-state batteries: from material design to practical application[J]. Sci Adv, 2022, 8(36): eadc9516. |
| 18 | ABOUALI S, YIM C H, MERATI A, et al. Garnet-based solid-state Li batteries: from materials design to battery architecture[J]. ACS Energy Lett, 2021, 6(5): 1920-1941. |
| 19 | WU J F, PANG W K, PETERSON V K, et al. Garnet-type fast Li-ion conductors with high ionic conductivities for all-solid-state batteries[J]. ACS Appl Mater Interfaces, 2017, 9(14): 12461-12468. |
| 20 | LU J, LI Y. Perovskite-type Li-ion solid electrolytes: a review[J]. J Mater Sci: Mater Electron, 2021, 32(8): 9736-9754. |
| 21 | SUN C, ALONSO J A, BIAN J. Recent advances in perovskite-type oxides for energy conversion and storage applications[J]. Adv Energy Mater, 2021, 11(2): 2000459. |
| 22 | JIAN Z, HU Y S, JI X, et al. Nasicon-structured materials for energy storage[J]. Adv Mater, 2017, 29(20): 1601925. |
| 23 | HOU M, LIANG F, CHEN K, et al. Challenges and perspectives of NASICON-type solid electrolytes for all-solid-state lithium batteries[J]. Nanotechnol, 2020, 31(13): 132003. |
| 24 | MURUGAN R, THANGADURAI V, WEPPNER W. Fast lithium ion conduction in garnet-type Li7La3Zr2O12[J]. Angew Chem Int Ed, 2007, 46(41): 7778-7781. |
| 25 | BACHMAN J C, MUY S, GRIMAUD A, et al. Inorganic solid-state electrolytes for lithium batteries: mechanisms and properties governing ion conduction[J]. Chem Rev, 2016, 116(1): 140-162. |
| 26 | LUO W, GONG Y, ZHU Y, et al. Reducing interfacial resistance between garnet‐structured solid-state electrolyte and Li-metal anode by a germanium layer[J]. Adv Mater, 2017, 29(22): 1606042. |
| 27 | PENG H, WU Q, XIAO L. Low temperature synthesis of Li5La3Nb2O12 with cubic garnet-type structure by sol-gel process[J]. J Sol-Gel Sci Technol, 2013, 66(1): 175-179. |
| 28 | O'CALLAGHAN M P, LYNHAM D R, CUSSEN E J, et al. Structure and ionic-transport properties of lithium-containing garnets Li3Ln3Te2O12 (Ln=Y, Pr, Nd, Sm-Lu)[J]. Chem Mater, 2006, 18(19): 4681-4689. |
| 29 | O'CALLAGHAN M P, POWELL A S, TITMAN J J, et al. Switching on fast lithium ion conductivity in garnets: the structure and transport properties of Li3+ xNd3Te2- xSbxO12[J]. Chem Mater, 2008, 20(6): 2360-2369. |
| 30 | RAMAKUMAR S, DEVIANNAPOORANI C, DHIVYA L, et al. Lithium garnets: synthesis, structure, Li+ conductivity, Li+ dynamics and applications[J]. Prog Mater Sci, 2017, 88: 325-411. |
| 31 | AWAKA J, TAKASHIMA A, KATAOKA K, et al. Crystal structure of fast lithium-ion-conducting cubic Li7La3Zr2O12[J]. Chem Lett, 2011, 40(1): 60-62. |
| 32 | AWAKA J, KIJIMA N, HAYAKAWA H, et al. Synthesis and structure analysis of tetragonal Li7La3Zr2O12 with the garnet-related type structure[J]. J Solid State Chem, 2009, 182(8): 2046-2052. |
| 33 | WANG C, FU K, KAMMAMPATA S P, et al. Garnet-type solid-state electrolytes: materials, interfaces, and batteries[J]. Chem Rev, 2020, 120(10): 4257-4300. |
| 34 | ADAMS S, RAO R. Ion transport and phase transition in Li7- xLa3(Zr2 xMx)O12 (M=Ta5+, Nb5+, x=0, 0.25)[J]. J Mater Chem, 2012, 22(4): 1426-1434. |
| 35 | XIE H, ALONSO J A, LI Y, et al. Lithium distribution in aluminum-free cubic Li7La3Zr2O12[J]. Chem Mater, 2011, 23(16): 3587-3589. |
| 36 | THOMPSON T, SHARAFI A, JOHANNES M D, et al. A tale of two sites: on defining the carrier concentration in garnet-based ionic conductors for advanced Li batteries[J]. Adv Energy Mater, 2015, 5(11): 1500096. |
| 37 | QIN Z, XIE Y, MENG X, et al. High cycling stability enabled by Li vacancy regulation in Ta-doped garnet-type solid-state electrolyte[J]. J Eur Ceram Soc, 2023, 43(5): 2023-2032. |
| 38 | YANG H, WU N. Ionic conductivity and ion transport mechanisms of solid-state lithium-ion battery electrolytes: a review[J]. Energy Sci Eng, 2022, 10(5): 1643-1671. |
| 39 | RAMZY A, THANGADURAI V. Tailor-made development of fast Li ion conducting garnet-like solid electrolytes[J]. ACS Appl Mater Interfaces, 2010, 2(2): 385-390. |
| 40 | LI Y, HAN J T, WANG C A, et al. Optimizing Li+ conductivity in a garnet framework[J]. J Mater Chem, 2012, 22(30): 15357-15361. |
| 41 | BUCHELI W, DURAN T, JIMENEZ R, et al. On the influence of the vacancy distribution on the structure and ionic conductivity of A-site-deficient LixSrxLa2/3- xTiO3 perovskites[J]. Inorg Chem, 2012, 51(10): 5831-5838. |
| 42 | XIANG X, LIU Y, CHEN F, et al. Crystal structure and lithium ionic transport behavior of Li site doped Li7La3Zr2O12[J]. J Eur Ceram Soc, 2020, 40(8): 3065-3071. |
| 43 | GEIGER C A, ALEKSEEV E, LAZIC B, et al. Crystal chemistry and stability of “Li7La3Zr2O12” garnet: a fast lithium-ion conductor[J]. Inorg Chem, 2011, 50(3): 1089-1097. |
| 44 | XU B, DUAN H, XIA W, et al. Multistep sintering to synthesize fast lithium garnets[J]. J Power Sources, 2016, 302: 291-297. |
| 45 | ZHUANG L, HUANG X, LU Y, et al. Phase transformation and grain-boundary segregation in Al-doped Li7La3Zr2O12 ceramics[J]. Ceram Int, 2021, 47(16): 22768-22775. |
| 46 | MURUGAN R, RAMAKUMAR S, JANANI N. High conductive yttrium doped Li7La3Zr2O12 cubic lithium garnet[J]. Electrochem Commun, 2011, 13(12): 1373-1375. |
| 47 | QIN S, ZHU X, JIANG Y, et al. Growth of self-textured Ga3+-substituted Li7La3Zr2O12 ceramics by solid state reaction and their significant enhancement in ionic conductivity[J]. Appl Phys Lett, 2018, 112(11): 113901. |
| 48 | WAGNER R, REDHAMMER G J, RETTENWANDER D, et al. Fast Li-ion-conducting garnet-related Li7-3 xFexLa3Zr2O12 with uncommon I 4 ¯ 3d Structure[J]. Chem Mater, 2016, 28(16): 5943-5951. |
| 49 | BERNUY-LOPEZ C, MANALASTAS J R W, LOPEZ DEL AMO J M, et al. Atmosphere controlled processing of Ga-substituted garnets for high Li-ion conductivity ceramics[J]. Chem Mater, 2014, 26(12): 3610-3617. |
| 50 | RETTENWANDERE D, GEIGER C A, TRIBUS M, et al. A synthesis and crystal chemical study of the fast ion conductor Li7-3 xGaxLa3Zr2O12 with x=0.08 to 0.84[J]. Inorg Chem, 2014, 53(12): 6264-6269. |
| 51 | THANGADURAI V, WEPPNER W. Li6ALa2Nb2O12 (A=Ca, Sr, Ba): a new class of fast lithium ion conductors with garnet-like structure[J]. J Am Ceram Soc, 2005, 88(2): 411-418. |
| 52 | DUMON A, HUANG M, SHEN Y, et al. High Li ion conductivity in strontium doped Li7La3Zr2O12 garnet[J]. Solid State Ionics, 2013, 243: 36-41. |
| 53 | RANGASAMY E, WOLFENSTINE J, ALLEN J, et al. The effect of 24c-site (A) cation substitution on the tetragonal-cubic phase transition in Li7- xLa3- xAxZr2O12 garnet-based ceramic electrolyte[J]. J Power Sources, 2013, 230: 261-266. |
| 54 | LI Y T, WANG C A, XIE H, et al. High lithium ion conduction in garnet-type Li6La3ZrTaO12[J]. Electrochem Commun, 2011, 13(12): 1289-1292. |
| 55 | ZHU Y, CONNELL J G, TEPAVCEVIC S, et al. Dopant-dependent stability of garnet solid electrolyte interfaces with lithium metal[J]. Adv Energy Mater, 2019, 9(12): 1803440. |
| 56 | SHAO C, YU Z, LIU H, et al. Enhanced ionic conductivity of titanium doped Li7La3Zr2O12 solid electrolyte[J]. Electrochim Acta, 2017, 225: 345-349. |
| 57 | LIU X, LI Y, YANG T, et al. High lithium ionic conductivity in the garnet-type oxide Li7-2 xLa3Zr2- xMoxO12 (x=0~0.3) ceramics by sol-gel method[J]. J Am Ceram Soc, 2017, 100(4): 1527-1533. |
| 58 | RAMAKUMAR S, SATYANARAYANA L, MANORAMA S V, et al. Structure and Li+ dynamics of Sb-doped Li7La3Zr2O12 fast lithium ion conductors[J]. Phys Chem Chem Phys, 2013, 15(27): 11327-11338. |
| 59 | DEVIANNAPOORANI C, DHIVYA L, RAMAKUMAR S, et al. Lithium ion transport properties of high conductive tellurium substituted Li7La3Zr2O12 cubic lithium garnets[J]. Power Sources, 2013, 240: 18-25. |
| 60 | DHIVYA L, JANANI N, PALANIVEL B, et al. Li+ transport properties of W substituted Li7La3Zr2O12 cubic lithium garnets[J]. AIP Adv, 2013, 3(8): 082115. |
| 61 | CAO S, SONG S, XIANG X, et al. Modeling, preparation, and elemental doping of Li7La3Zr2O12 garnet-type solid electrolytes: a review[J]. J Korean Ceram Soc, 2019, 56(2): 111-129. |
| 62 | MA X, XU Y. Efficient anion fluoride-doping strategy to enhance the performance in garnet-type solid electrolyte Li7La3Zr2O12[J]. ACS Appl Mater Interfaces, 2022, 14(2): 2939-2948. |
| 63 | DONG B, YEANDEL S R, GODDARD P, et al. Combined experimental and computational study of Ce-doped La3Zr2Li7O12 garnet solid-state electrolyte[J]. Chem Mater, 2019, 32(1): 215-223. |
| 64 | WANG M, SAKAMOTO J. Dramatic reduction in the densification temperature of garnet-type solid electrolytes[J]. Ionics, 2018, 24(7): 1861-1868. |
| 65 | KUMAZAKI S, IRIYAMA Y, KIM K H, et al. High lithium ion conductive Li7La3Zr2O12 by inclusion of both Al and Si[J]. Electrochem Commun, 2011, 13(5): 509-512. |
| 66 | SHIN D O, OH K, KIM K M, et al. Synergistic multi-doping effects on the Li7La3Zr2O12 solid electrolyte for fast lithium ion conduction[J]. Sci Rep, 2015, 5: 18053. |
| 67 | MEESALA Y, LIAO Y K, JENA A, et al. An efficient multi-doping strategy to enhance Li-ion conductivity in the garnet-type solid electrolyte Li7La3Zr2O12[J]. J Mater Chem A, 2019, 7(14): 8589-8601. |
| 68 | XIANG H, XXING Y, DAI F Z,et al. High-entropy ceramics: present status, challenges, and a look forward[J]. J Adv Ceram, 2021, 10(3): 385-441. |
| 69 | FU Z, FERGUSON J. Processing and characterization of an Li7La3Zr0.5Nb0.5Ta0.5Hf0.5O12 high-entropy Li-garnet electrolyte[J]. J Am Ceram Soc, 2022, 105(10): 6175-6183. |
| 70 | AFYON S, KRUMEICH F, RUPP J L M. A shortcut to garnet-type fast Li-ion conductors for all-solid state batteries[J]. J Mater Chem A, 2015, 3(36): 18636-18648. |
| 71 | SHAO C, LIU H, YU Z, et al. Structure and ionic conductivity of cubic Li7La3Zr2O12 solid electrolyte prepared by chemical co-precipitation method[J]. Solid State Ionics, 2016, 287: 13-16. |
| 72 | POLIZOS G, SHARMA J, JAFTA C J, et al. Nanostructured ligament and fiber Al-doped Li7La3Zr2O12 scaffolds to mediate cathode-electrolyte interface chemistry[J]. J Power Sources, 2021, 513: 230551. |
| 73 | DJENADIC R, BOTROS M, BENEL C, et al. Nebulized spray pyrolysis of Al-doped Li7La3Zr2O12 solid electrolyte for battery applications[J]. Solid State Ionics, 2014, 263: 49-56. |
| 74 | RANGASAMY E, WOLFENSTINE J, SAKAMOTO J. The role of Al and Li concentration on the formation of cubic garnet solid electrolyte of nominal composition Li7La3Zr2O12[J].Solid State Ionics, 2012, 206: 28-32. |
| 75 | WANG M, SAKAMOTO J. Correlating the interface resistance and surface adhesion of the Li metal-solid electrolyte interface[J]. J Power Sources, 2018, 377: 7-11. |
| 76 | KAMMAMPATA S P, BASAPPA R H, ITO T, et al. Microstructural and electrochemical properties of alkaline earth metal-doped Li garnet-type solid electrolytes prepared by solid-state sintering and spark plasma sintering methods[J]. ACS Appl Energy Mater, 2019, 2(3): 1765-1773. |
| 77 | CUI C, YE Q, ZENG C, et al. One-step fabrication of garnet solid electrolyte with integrated lithiophilic surface[J]. Energy Stor Mater, 2022, 45: 814-820. |
| 78 | ZHANG Y, CHEN F, TU R, et al. Effect of lithium ion concentration on the microstructure evolution and its association with the ionic conductivity of cubic garnet-type nominal Li7Al0.25La3Zr2O12 solid electrolytes[J]. Solid State Ionics, 2016, 284: 53-60. |
| 79 | LI H Y, HUANG B, HUANG Z, et al. Enhanced mechanical strength and ionic conductivity of LLZO solid electrolytes by oscillatory pressure sintering[J]. Ceram Int, 2019, 45(14): 18115-18118. |
| 80 | LI C, LIU Y, HE J, et al. Ga-substituted Li7La3Zr2O12: an investigation based on grain coarsening in garnet-type lithium ion conductors[J]. J Alloys Compd, 2017, 695: 3744-3752. |
| 81 | ROSERO-NAVARRO N C, YAMASHITA T, MIURA A, et al. Effect of sintering additives on relative density and Li-ion conductivity of Nb-doped Li7La3Zr2O12 solid electrolyte[J]. J Am Ceram Soc, 2017, 100(1): 276-285. |
| 82 | ZHANG K, XU T, ZHAO H, et al. Unveiling the roles of alumina as a sintering aid in Li-garnet solid electrolyte[J]. Int J Energy Res, 2020, 44(11): 9177-9184. |
| 83 | LI Y, CAO Y, GUO X. Influence of lithium oxide additives on densification and ionic conductivity of garnet-type Li6.75La3Zr1.75Ta0.25O12 solid electrolytes[J]. Solid State Ionics, 2013, 253: 76-80. |
| 84 | LI C, ISHII A, ROY L, et al. Solid-state reactive sintering of dense and highly conductive Ta-doped Li7La3Zr2O12 using CuO as a sintering aid[J]. J Mater Sci, 2020, 55: 16470-16481. |
| 85 | LIN J, WU L, HUANG Z, et al. La2Zr2O7 and MgO co-doped composite Li-garnet solid electrolyte[J]. J Energy Chem, 2020, 40: 132-136. |
| 86 | LI Q, LIU H, GAO C, et al. Investigation the electrochemical properties of LiCl-LiBr-LiF-doped Li7La3Zr2O12 electrolyte for lithium thermal batteries[J]. Ionics, 2020, 26: 3875-3882. |
| 87 | TIAN Y, DING F, ZHONG H, et al. Li6.75La3Zr1.75Ta0.25O12@amorphous Li3OCl composite electrolyte for solid state lithium-metal batteries[J]. Energy Stor Mater, 2018, 14: 49-57. |
| 88 | TADANAGA K, TAKANO R, ICHINOSE T, et al. Low temperature synthesis of highly ion conductive Li7La3Zr2O12-Li3BO3 composites[J]. Electrochem Commun, 2013, 33: 51-54. |
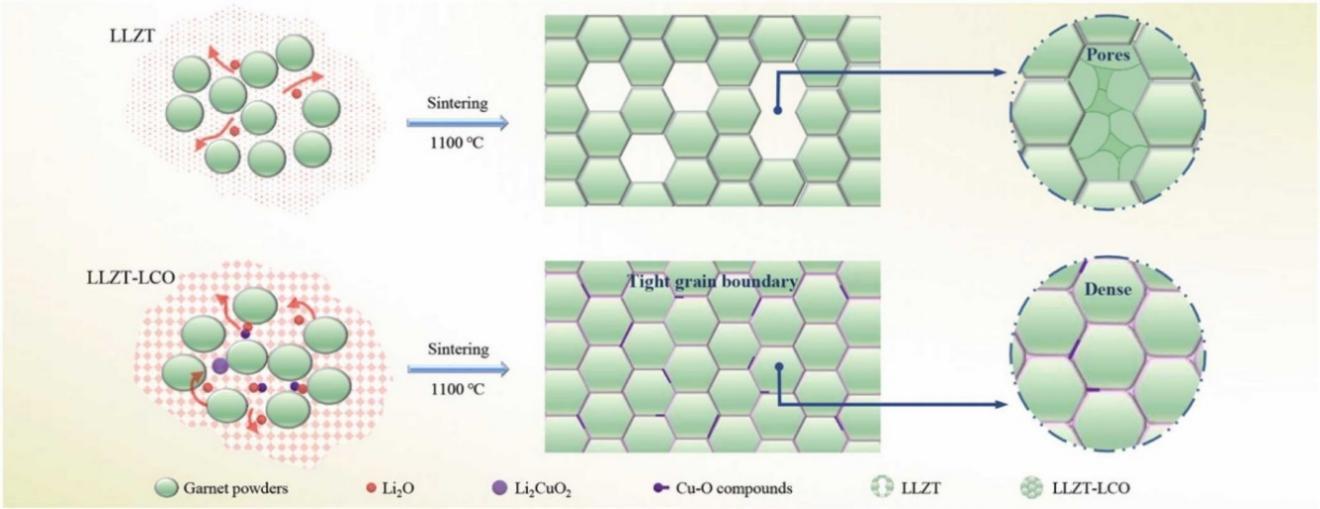
| 89 | ZHENG C, SU J, SONG Z, et al. Sintering promotion and electrochemical performance of garnet-type electrolyte with Li2CuO2 additive[J]. J Alloys Compd, 2023, 933: 167810. |
| 90 | KANAI K, OZAWA S, KOZAWA T, et al. Low temperature synthesis of Ga-doped Li7La3Zr2O12 garnet-type solid electrolyte by mechanical method[J]. Adv Powder Technol, 2021, 32(10): 3860-3868. |
| [1] | 陈兵帅, 卓海涛, 黄书, 陈少军. 高性能硅基负极聚合物粘结剂的研究进展[J]. 应用化学, 2023, 40(5): 625-639. |
| [2] | 胡方正, 高兴, 刘雷, 袁天恒, 曹宁, 李凯, 王亚涛, 李建华, 连慧琴, 汪晓东, 崔秀国. 锂离子电池黑磷负极的储能优势及其优化的研究进展[J]. 应用化学, 2023, 40(4): 571-582. |
| [3] | 王恩通, 杨林芳. 高比容量锂离子电池正极材料LiNi0.6Co0.2Mn0.2O2的制备及性能[J]. 应用化学, 2022, 39(8): 1209-1215. |
| [4] | 宋林虎, 李世友, 王洁, 张晶晶, 张宁霜, 赵冬妮, 徐菲. 锂离子电池电解液除酸除水添加剂的研究进展[J]. 应用化学, 2022, 39(5): 697-706. |
| [5] | 张琦, 张乾, 师晓梦, 孔娅淇, 高可心, 杜亚平. 稀土溴化物固态电解质材料在全固态电池中的应用研究进展[J]. 应用化学, 2022, 39(4): 585-598. |
| [6] | 王玉乐, 杨柯利, 高艳芳. 碳化钼改性二氧化硅的制备及其电化学性能[J]. 应用化学, 2022, 39(11): 1716-1725. |
| [7] | 赵莹, 邵奕嘉, 李罗钱, 任建伟, 廖世军. 富锂正极材料的衰减机理及循环稳定性提升的研究进展[J]. 应用化学, 2022, 39(02): 205-222. |
| [8] | 王旭尧, 方应军, 张灵志. 氰基功能化聚乙烯亚胺交联固态聚合物电解质的制备及理化性能[J]. 应用化学, 2021, 38(8): 946-953. |
| [9] | 陶承业, 杨占旭, 李玥. 红磷/石墨复合材料的制备及电化学性能[J]. 应用化学, 2020, 37(9): 1056-1061. |
| [10] | 刘学, 马华, 徐恒, 计海聪, 王栋. 无纺布基聚吡咯柔性电极的储锂性能[J]. 应用化学, 2020, 37(5): 555-561. |
| [11] | 王金莹, 曲江英, 李杰兰, 汤占磊, 臧云浩, 王涛, 顾建峰, 周钢, 高峰. 二次包覆法制备煤沥青基硅/碳复合物及其锂离子电池性能[J]. 应用化学, 2020, 37(5): 562-569. |
| [12] | 胡晨,金翼,朱少青,徐晔,水江澜. 磷酸铁锂电池低温性能的改性方法简述[J]. 应用化学, 2020, 37(4): 380-386. |
| [13] | 崔华敏, 掌学谦, 胡攀登, 闫冰, 黄苇苇, 郭文锋. 杯[4]醌/N掺杂的无定形碳纳米纤维复合材料储锂性能[J]. 应用化学, 2020, 37(2): 198-204. |
| [14] | 惠康龙, 傅继澎, 高湉, 唐明学. 金属硫化物在可充电电池中的研究进展[J]. 应用化学, 2020, 37(12): 1384-1402. |
| [15] | 刘丽鑫, 董建红, 张光辉, 朱陆益, 王新强, 许东, CHOW Yuktakb. 静电纺聚偏氟乙烯@硅藻土锂离子电池隔膜的制备及性能[J]. 应用化学, 2020, 37(12): 1441-1446. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||