
应用化学 ›› 2024, Vol. 41 ›› Issue (1): 3-20.DOI: 10.19894/j.issn.1000-0518.230085
多色比色法在生物传感平台的研究进展
- 天津大学生命科学学院,天津 300072
-
收稿日期:2023-04-03接受日期:2023-08-03出版日期:2024-01-01发布日期:2024-01-30 -
通讯作者:宫晓群 -
基金资助:天津市科技支撑重点项目(20YFZCSY00990)
Research Progress of Multicolor Colorimetric Method in Biosensing Platform
Yi XU, Meng-Yao LIN, Xiao-Qun GONG( )
)
- School of Life Science,Tianjin University,Tianjin 300072,China
-
Received:2023-04-03Accepted:2023-08-03Published:2024-01-01Online:2024-01-30 -
Contact:Xiao-Qun GONG -
About author:gongxiaoqun@tju.edu.cn
-
Supported by:the Scientific and Technical Supporting Key Project of Tianjin, China(20YFZCSY00990)
摘要:
比色法是生物传感中较为常用的一种检测手段,因其具有较高的灵敏度和特异性而受到广泛关注。其中,基于多色探针的多色比色法已成为近年来的研究热点,灵敏度较单色比色法显著提高、且具有更高的分辨率和肉眼辨识度,通过观察颜色变化可进行可视化半定量检测,又可通过计算颜色比率或基于智能手机图像处理软件进行快速定量检测。基于有机小分子荧光显色、纳米材料显色及酶促反应显色3种不同的显色机理,综述了多色荧光小分子探针、多色纳米材料比色探针及多色酶促反应比色探针的检测机理以及它们在实际样本检测中的应用,并进一步探讨了其面临的挑战及未来发展机遇。
中图分类号:
引用本文
徐一, 林梦瑶, 宫晓群. 多色比色法在生物传感平台的研究进展[J]. 应用化学, 2024, 41(1): 3-20.
Yi XU, Meng-Yao LIN, Xiao-Qun GONG. Research Progress of Multicolor Colorimetric Method in Biosensing Platform[J]. Chinese Journal of Applied Chemistry, 2024, 41(1): 3-20.
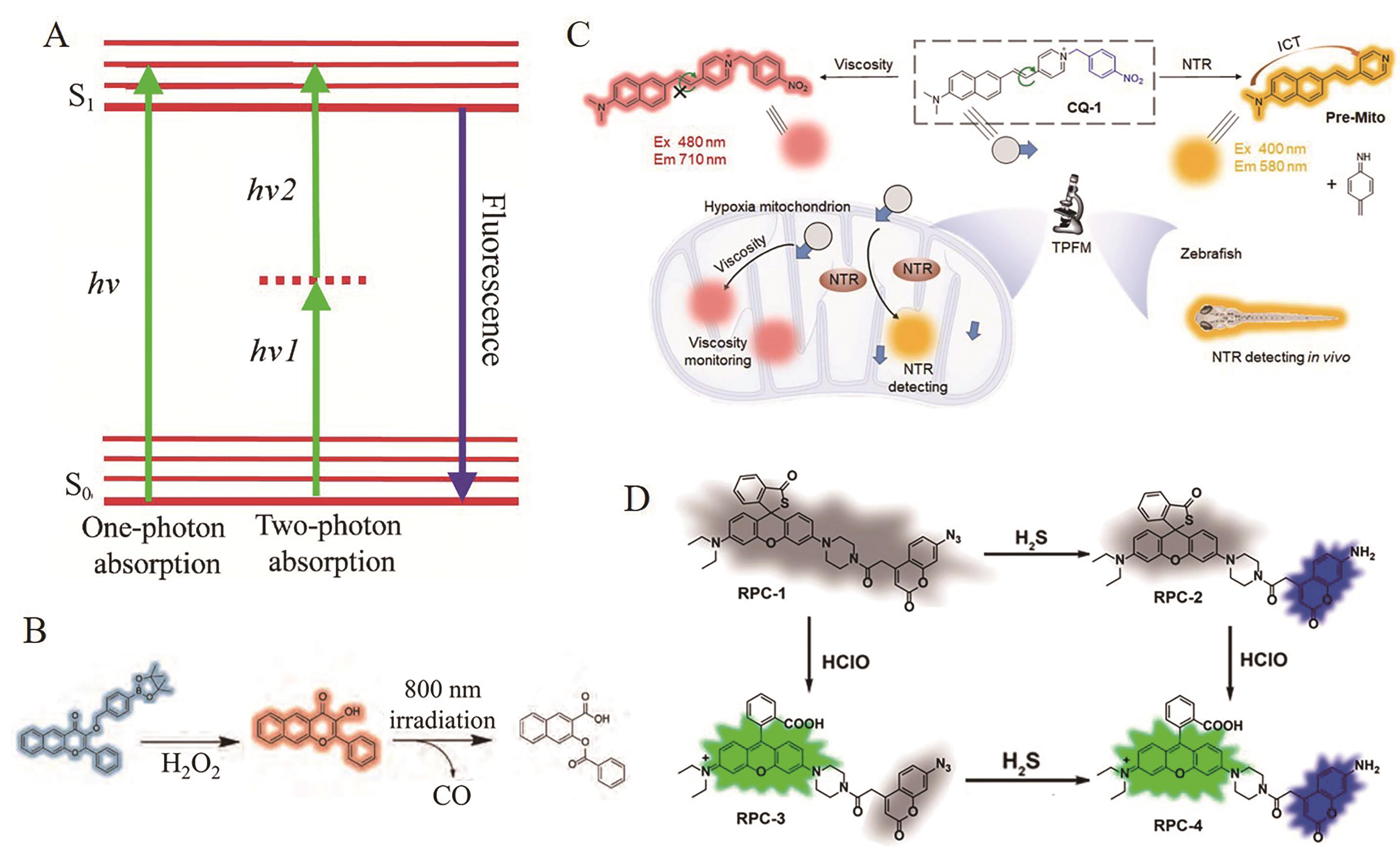
图1 双光子荧光分子探针原理及应用(A) Schematic diagram of the two-photon excitation principle[21]; (B) Schematic diagram of the principle of a flavonol-borate multicolor probe based on the detection of H2O2[26]; (C) Schematic diagram of the CQ-1 dual inspection probe[27]; (D) Schematic diagram of the RPC dual-detection probe principle[28]
Fig.1 Principles and applications of two-photon fluorescent probes
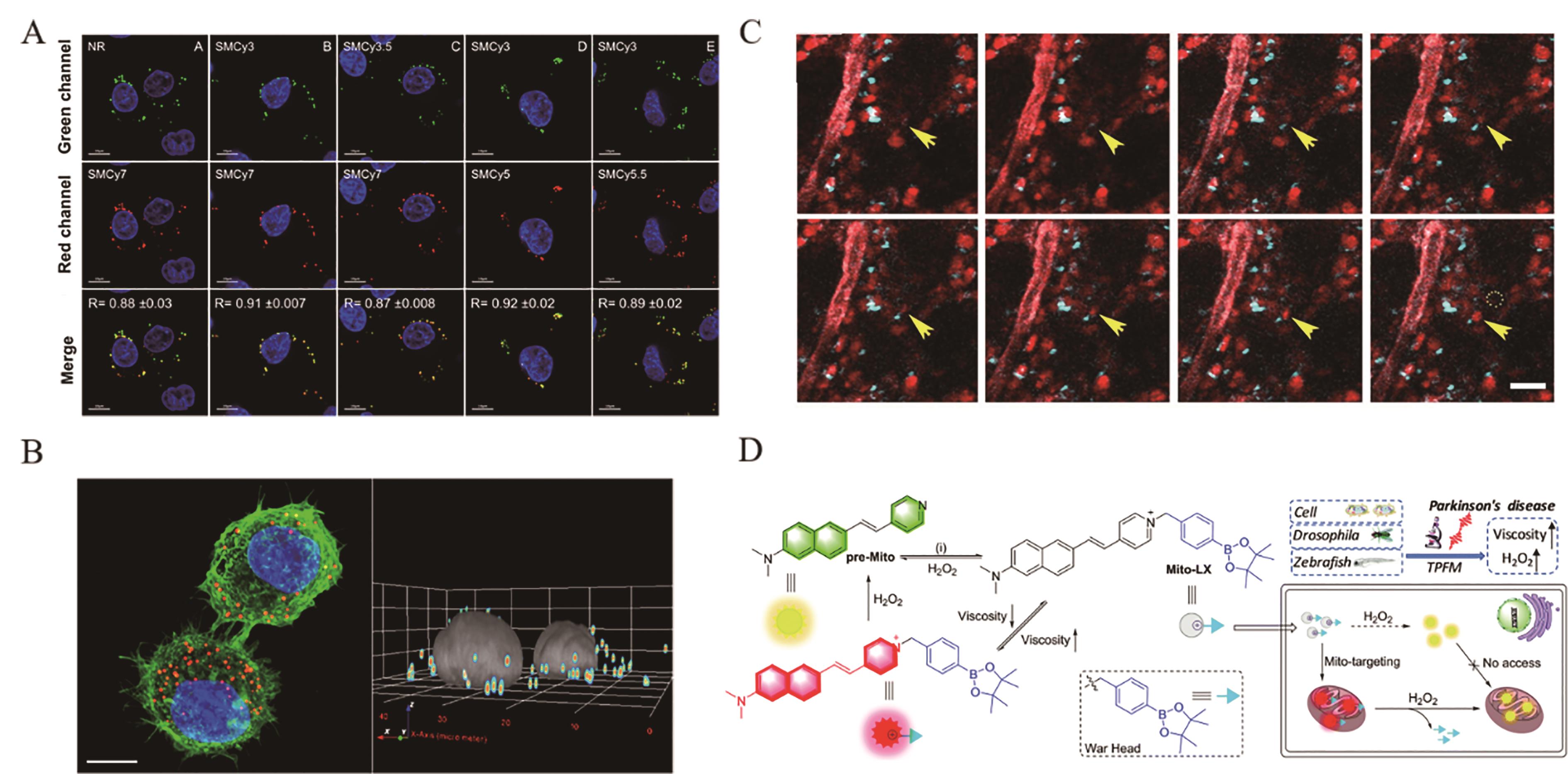
图2 多响应靶向型荧光分子探针应用(A, B) Multicolor fluorescent probes for targeting cellular lipid droplets[29]; (C) Multicolor fluorescence microimaging of NLS-LSS-mKate1 labelled nuclei (red) and GalT-ECFP labelled Golgi apparatus (blue) in tumour cells[30]; (D) Principle of the Mito-LX double detection probe[31]
Fig.2 Applications of multi-response targeted fluorescent probes
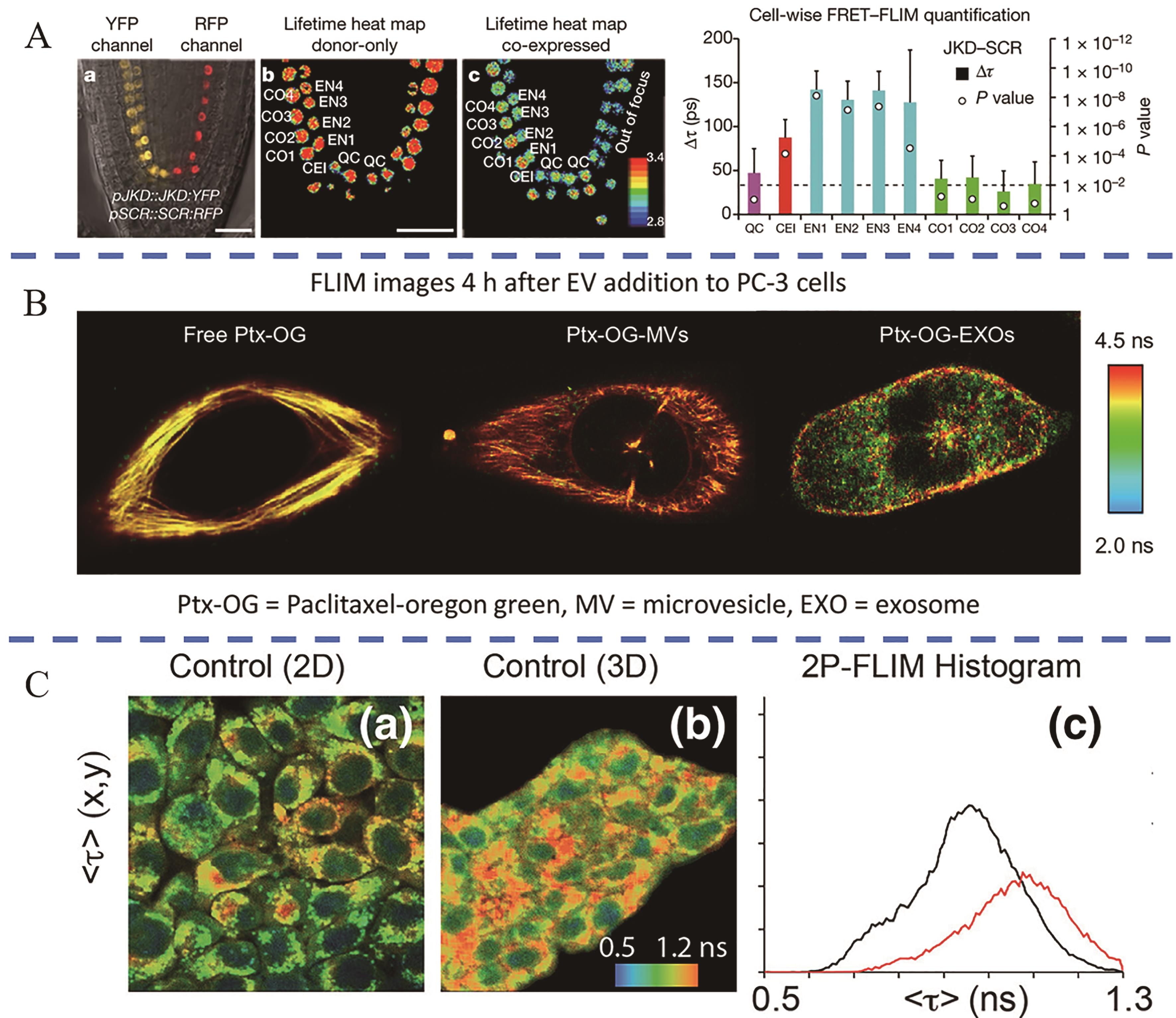
图3 多响应荧光寿命探针应用(A) FRET-FLIM imaging analysis reveals JKD-SCR interactions in endodermal cells[33]; (B) FLIM imaging compares the different cellular uptake mechanisms of OG-PTX[34]; (C) The NADH fluorescence lifetime of 4T1 cells in 2D culture (a) is shorter than that of 3D culture (b), the black and red lines in (c) respectively represent the fluorescence lifetimes of NADH in 2D and 3D culture[36]
Fig.3 Applications of multi-response fluorescent lifetime probes
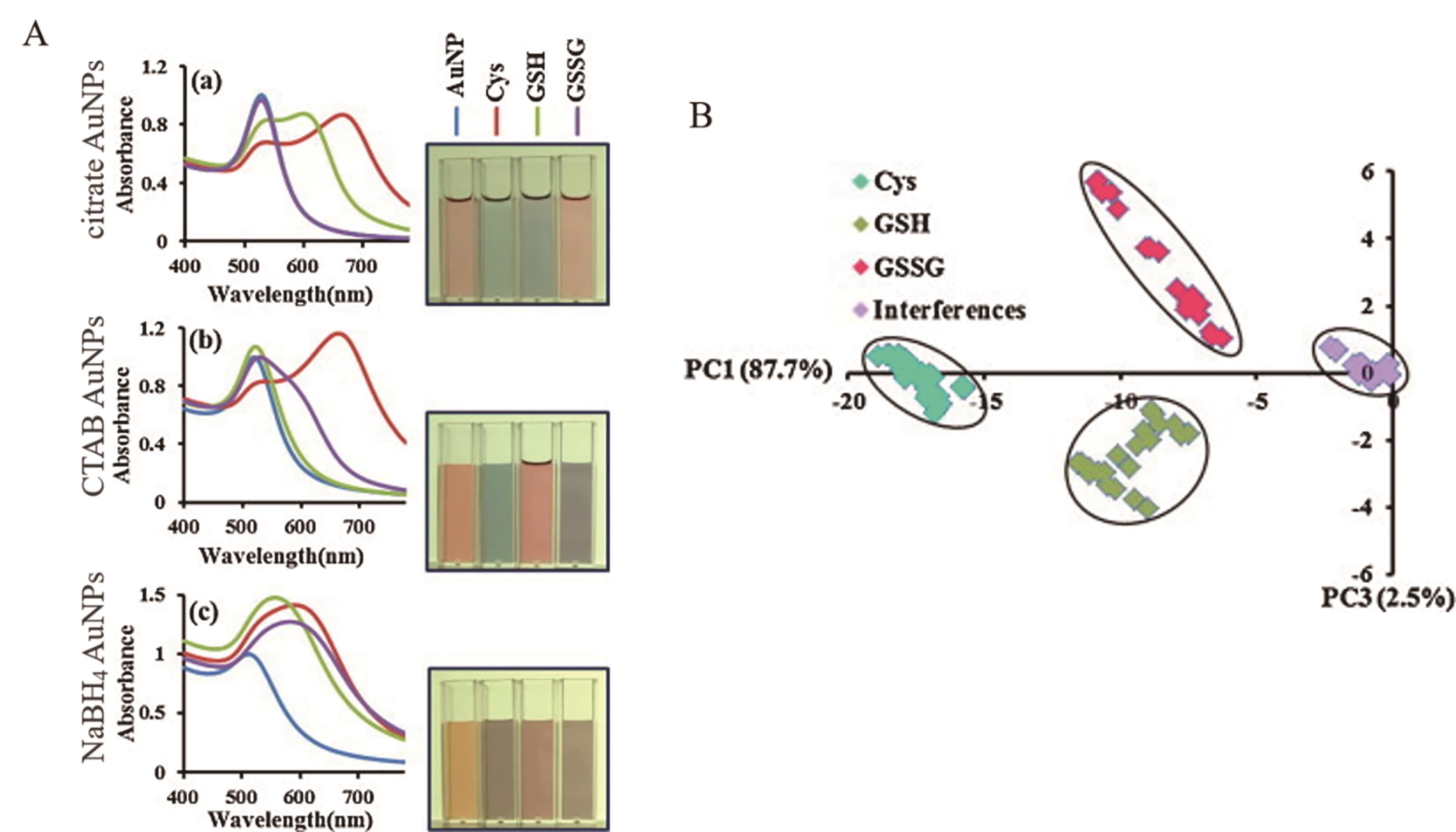
图4 聚集显色多色比色平台应用(A) Aggregation color changes of three functionalized AuNPs induced by Cys, GSH and GSSG and the corresponding UV-Vis spectra[41]; (B) Two-dimensional score plot of a categorised mercaptan multicolor colorimetric platform, demonstrating the platform's good discrimination of various mercaptans[41]
Fig.4 Applications of aggregated chromogenic multicolor platforms
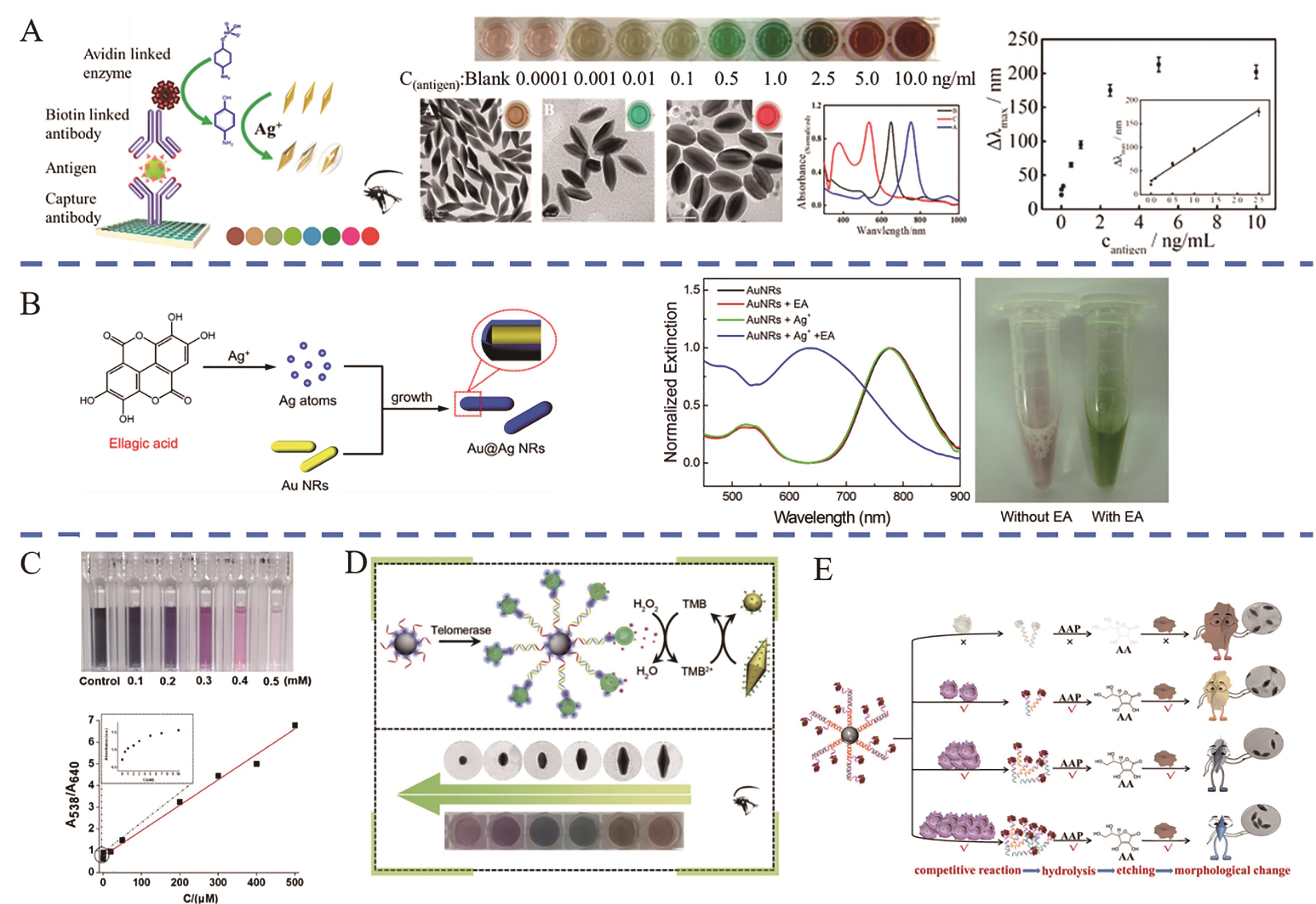
图5 表面生长蚀刻多色比色平台应用(A) Schematic diagram and color change of AuNBPs growth method for detection of H5N1 virus[49]; (B) Schematic diagram and color change of AuNRs growth method for the detection of EA and vitamin C[50]; (C) Color change and concentration-absorbance change curves of AuNRs etching for cyanide detection[55]; (D) Schematic diagram and color change of AuNBPs etching method for the detection of telomerase activity[56]; (E) Schematic diagram and color change of AuNBP@MnO2 NSs etching method for amplification detection of exosomes[57]
Fig.5 Applications of multi-rcolor colorimetric platform for surface growth etching

图6 QDs多色比色平台应用(A) QD immunofluorescence imaging images and fluorescence spectra for cryoplethy smography of hepatocytes[63]; (B) Simultaneous detection of four toxins in individual and mixed fluorescence spectra by four-color QD[65]; (C) Schematic diagram of lateral flow strip for simultaneous detection of four nitrofuran metabolites by four-color QD[66]; (D) Schematic diagram of the principle of portable colorimetric detection of Hb by multicolor SiQDs[67]
Fig.6 Applications of multicolor colorimetric platform based on the QDs

图7 UCNP和MOF多色比色平台应用(A) Multicolor UCNP in vivo orthogonal imaging images[70]; (B) Schematic diagram of the two-color UCNP-LFT and portable reader for ST1 and BNP detection[74]; (C) Schematic diagram of the multi-color CDs-MOF sensing platform for the detection of Ag+ and Cys[75]; (D) Schematic diagram of a multi-color MOF sensing platform for the simultaneous detection of three types of DNA[76]
Fig.7 Applications of multicolor colorimetric platforms based on the UCNP and MOF
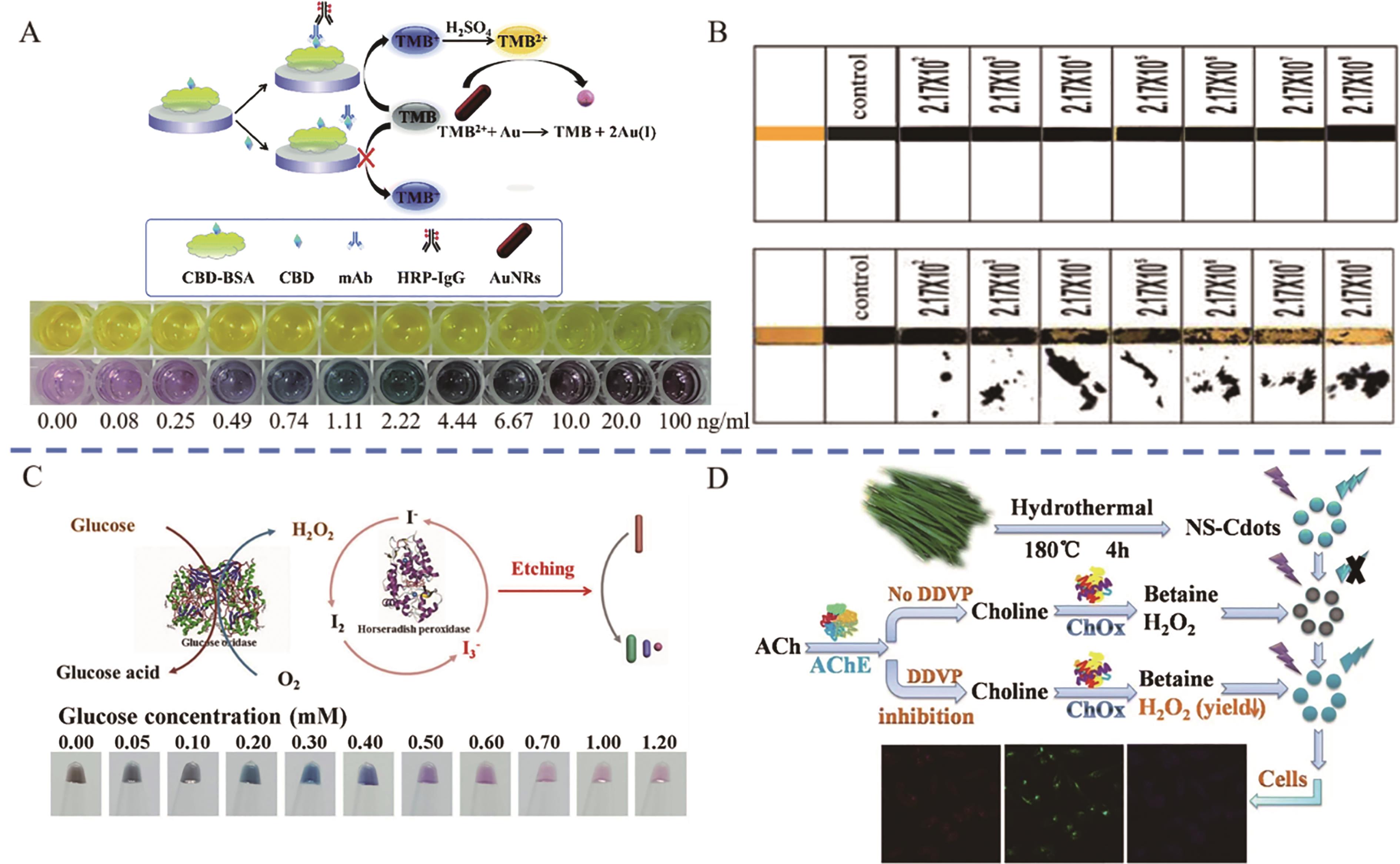
图8 蛋白酶催化的多色比色平台应用(A) Schematic diagram of the principle and color change of the multi-color ELISA for the detection of CBD[79]; (B) Color change of Listeria monocytogenes protease detected by multicolor lateral flow strip[81]?; (C) Schematic diagram of the principle and color change of a multicolor colorimetric platform for the detection of glucose by enzyme cascade amplification[83]?; (D) Schematic diagram of the principle and color change of the multicolor colorimetric platform for the detection of DDVP by enzyme cascade amplification[84]
Fig.8 Applications of protease-catalyzed multicolor colorimetric platforms
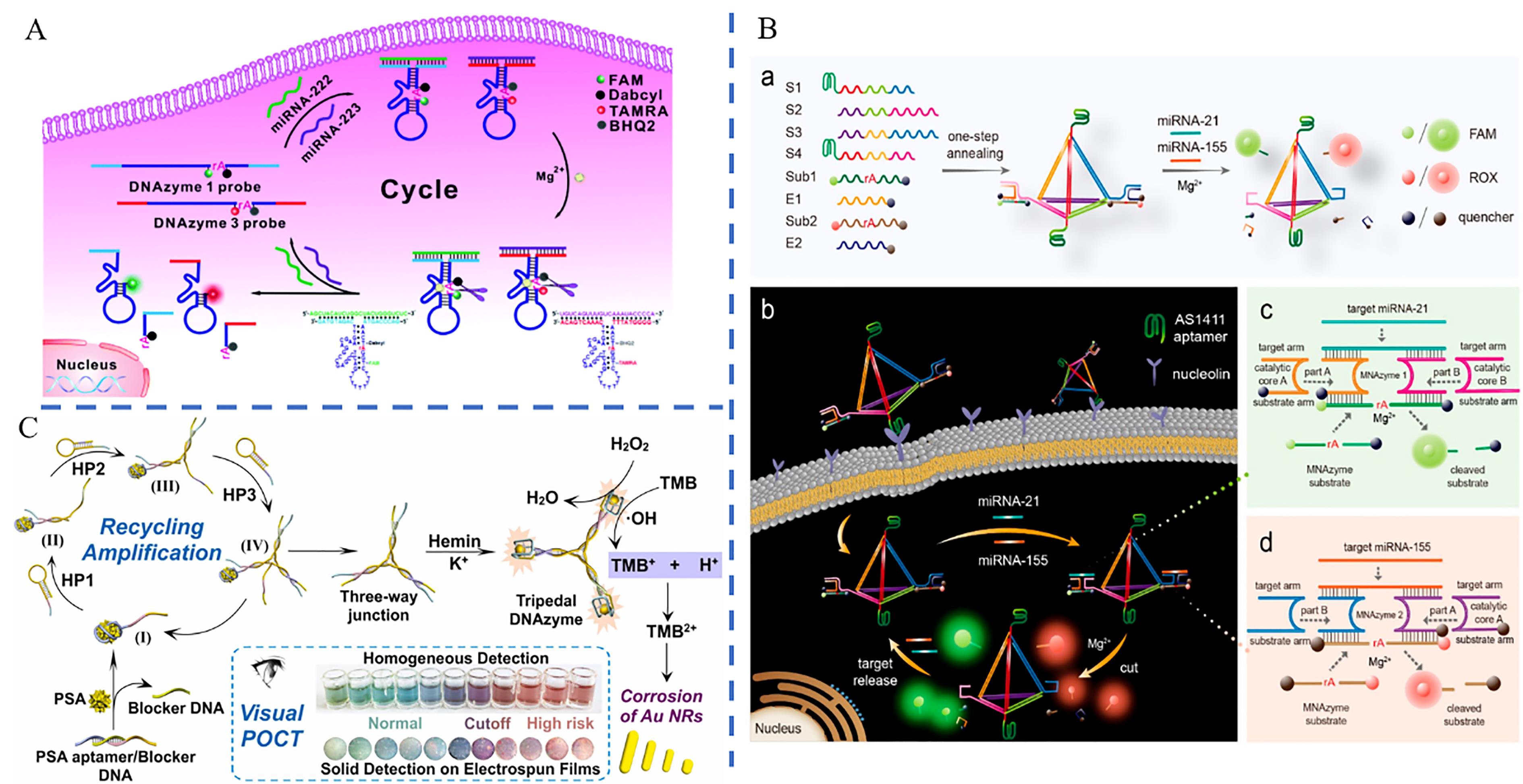
图9 基于DNA酶模拟物的多色比色平台应用(A) Schematic diagram of the principle of DNAzyme probes to detect miRNA in cells[97]; (B) Schematic diagram of the principle of multicolor detection of intracellular miRNA-21 and miRNA-155 by DNA orthotetrahedral probes[99]; (C) Schematic diagram of the principle of multicolor detection of PSA by DNA tridentate probes and film color change[100]
Fig.9 Applications of a multi-color colorimetric platform based on DNA enzyme simulants
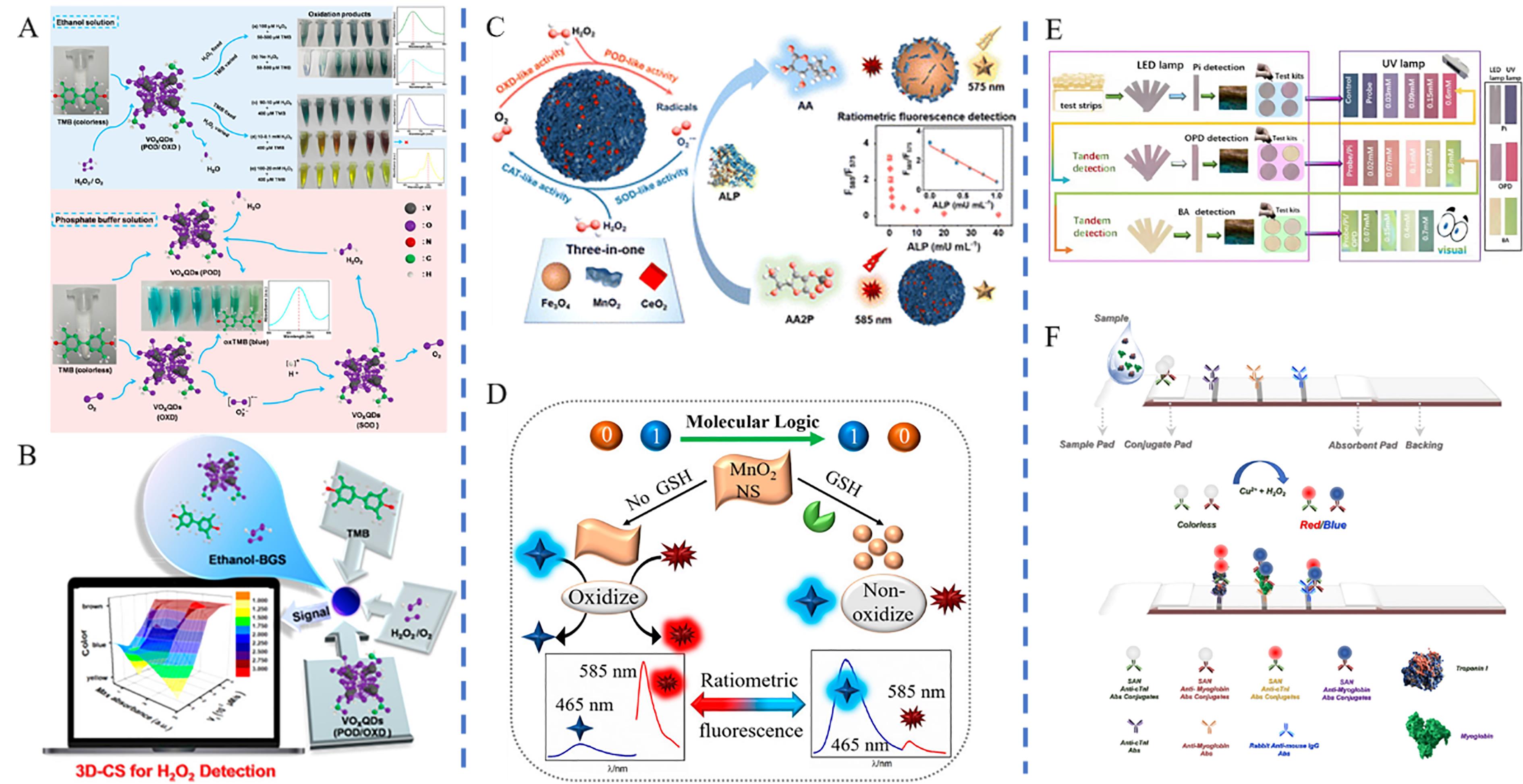
图10 基于纳米酶的多色比色平台应用(A,B) Schematic diagram of the principle of VO2-QD naked eye multicolor detection of H2O2 and color change[104]; (C) Schematic diagram of the principle of multicolor fluorescence detection of ALP by Fef NCs[105]; (D) Schematic diagram of the principle of multicolor fluorescence detection of GSH by MnO2NS[106]; (E) Color changes in colorimetric cards for Pi, OPD and BA detected by MOF mimetic enzyme probes[107]; (F) Schematic diagram of the principle of SAN multicolor colorimetric strips for the detection of cardiac troponin I and myoglobin[108]
Fig.10 Applications of a nano-enzyme based multi-color colorimetric platform
| 1 | YANO T, KAJISA T, ONO M, et al. Ultrasensitive detection of SARS-CoV-2 nucleocapsid protein using large gold nanoparticle-enhanced surface plasmon resonance[J]. Sci Rep, 2022, 12(1): 1060. |
| 2 | WANG B, WANG R, WANG D, et al. Cas12aVDet: a CRISPR/Cas12a-based platform for rapid and visual nucleic acid detection[J]. Anal Chem, 2019, 91(19): 12156-12161. |
| 3 | FENG W, NEWBIGGING A M, TAO J, et al. CRISPR technology incorporating amplification strategies: molecular assays for nucleic acids, proteins, and small molecules[J]. Chem Sci, 2021, 12(13): 4683-4698. |
| 4 | COHEN J D, JAVED A A, THOBURN C, et al. Combined circulating tumor DNA and protein biomarker-based liquid biopsy for the earlier detection of pancreatic cancers[J]. PNAS, 2017, 114(38): 10202-10207. |
| 5 | FREI A P, BAVA F A, ZUNDER E R, et al. Highly multiplexed simultaneous detection of RNAs and proteins in single cells[J]. Nat Methods, 2016, 13(3): 269-275. |
| 6 | LIU W B, LIU L, KOU G M, et al. Evaluation of nucleocapsid and spike protein-based enzyme-linked immunosorbent assays for detecting antibodies against SARS-CoV-2[J]. J Clin Microbiol, 2020, 58(6): e00461-20. |
| 7 | LI H, LEI Y, LI S, et al. MicroRNA-20a-5p inhibits the autophagy and cisplatin resistance in ovarian cancer via regulating DNMT3B-mediated DNA methylation of RBP1[J]. Reprod Toxicol, 2022, 109: 93-100. |
| 8 | QI H, HU Z, YANG Z, et al. Capacitive aptasensor coupled with microfluidic enrichment for real-time detection of trace SARS-CoV-2 nucleocapsid protein[J]. Anal Chem, 2022, 94(6): 2812-2819. |
| 9 | LASSERRE P, BALANSETHUPATHY B, VEZZA V J, et al. SARS-CoV-2 aptasensors based on electrochemical impedance spectroscopy and low-cost gold electrode substrates[J]. Anal Chem, 2022, 94(4): 2126-2133. |
| 10 | RODA A, CAVALERA S, DI NARDO F, et al. Dual lateral flow optical/chemiluminescence immunosensors for the rapid detection of salivary and serum IgA in patients with COVID-19 disease[J]. Biosens Bioelectron, 2021, 172: 112765. |
| 11 | LI W, ZHANG X, HU X, et al. A smartphone-integrated ratiometric fluorescence sensor for visual detection of cadmium ions[J]. J Hazard, 2021, 408: 124872. |
| 12 | TANG Y, HU Y, ZHOU P, et al. Colorimetric detection of kanamycin residue in foods based on the aptamer-enhanced peroxidase-mimicking activity of layered WS2 nanosheets[J]. J Agric Food Chem, 2021, 69(9): 2884-2893. |
| 13 | HAN H, WANG C, YANG X, et al. Rapid field determination of SARS-CoV-2 by a colorimetric and fluorescent dual-functional lateral flow immunoassay biosensor[J]. Sens Actuators B: Chem, 2022, 351: 130897. |
| 14 | MA X, HE S, QIU B, et al. Noble metal nanoparticle-based multicolor immunoassays: an approach toward visual quantification of the analytes with the naked eye[J]. ACS Sens, 2019, 4(4): 782-791. |
| 15 | LIU Y, ZHANG Z, YU J, et al. A concentration-dependent multicolor conversion strategy for ultrasensitive colorimetric immunoassay with the naked eye[J]. Anal Chim Acta, 2017, 963: 129-135. |
| 16 | ZHANG H, WEI X, CHAN-PARK M B, et al. Colorimetric sensors based on multifunctional polymers for highly sensitive detection of food spoilage[J]. Food Sci Technol, 2022, 2(4): 703-711. |
| 17 | LUO L, LUO S Z, JIA B Z, et al. A high-resolution colorimetric immunoassay for tyramine detection based on enzyme-enabled growth of gold nanostar coupled with smartphone readout[J]. Food Chem, 2022, 396: 133729. |
| 18 | CHAO S, KREJCI E, BERNARD V, et al. A selective and sensitive near-infrared fluorescent probe for acetylcholinesterase imaging[J]. Chem Commun, 2016, 52(77): 11599-11602. |
| 19 | CHEN J, LIAO D, WANG Y, et al. Real-time fluorometric assay for acetylcholinesterase activity and inhibitor screening through the pyrene probe monomer-excimer transition[J]. Org Lett, 2013, 15(9): 2132-2135. |
| 20 | HUANG B, BATES M, ZHUANG X. Super-resolution fluorescence microscopy[J]. Annu Rev Biochem, 2009, 78(1): 993-1016. |
| 21 | HE G S, TAN L S, ZHENG Q, et al. Multiphoton absorbing materials: molecular designs, characterizations, and applications[J]. Chem Rev, 2008, 108(4): 1245-1330. |
| 22 | LIM C S, MASANTA G, KIM H J, et al. Ratiometric detection of mitochondrial thiols with a two-photon fluorescent probe[J]. J Am Chem Soc, 2011, 133(29): 11132-11135. |
| 23 | REN M, DONG D, XU Q, et al. A biotin-guided two-photon fluorescent probe for detection of hydrogen peroxide in cancer cells ferroptosis process[J]. Talanta, 2021, 234: 122684. |
| 24 | KIM H M, CHO B R. Two-photon probes for intracellular free metal ions, acidic vesicles, and lipid rafts in live tissues[J]. Acc Chem Res, 2009, 42(7): 863-872. |
| 25 | LI J, ZHANG Y, ZHANG H, et al. Nucleoside-based ultrasensitive fluorescent probe for the dual-mode imaging of microviscosity in living cells[J]. Anal Chem, 2016, 88(10): 5554-5560. |
| 26 | LI Y, SHU Y, LIANG M, et al. A two-photon H2O2-activated CO photoreleaser[J]. Angew Chem Int Ed, 2018, 57(38): 12415-12419. |
| 27 | DU W, WANG J, FANG H, et al. Mitochondria-specific two-photon fluorogenic probe for simultaneously visualizing nitroreductase and viscosity in cancer cells[J]. Sens Actuators B: Chem, 2022, 370: 132456. |
| 28 | JIAO X, XIAO Y, LI Y, et al. Evaluating drug-induced liver injury and its remission via discrimination and imaging of HClO and H2S with a two-photon fluorescent probe[J]. Anal Chem, 2018, 90(12): 7510-7516. |
| 29 | COLLOT M, FAM T K, ASHOKKUMAR P, et al. Ultrabright and fluorogenic probes for multicolor imaging and tracking of lipid droplets in cells and tissues[J]. J Am Chem Soc, 2018, 140(16): 5401-5411. |
| 30 | PIATKEVICH K D, HULIT J, SUBACH O M, et al. Monomeric red fluorescent proteins with a large Stokes shift[J]. PNAS, 2010, 107(12): 5369-5374. |
| 31 | LI H, XIN C, ZHANG G, et al. A mitochondria-targeted two-photon fluorogenic probe for the dual-imaging of viscosity and H2O2 levels in Parkinson′s disease models[J]. J Mater Chem B, 2019, 7(27): 4243-4251. |
| 32 | LIN D, LUO T, LIU L, et al. Implementation of FLIM and SIFT for improved intraoperative delineation of glioblastoma margin[J]. Chin Opt Lett, 2017, 15(9): 090006. |
| 33 | LONG Y, STAHL Y, WEIDTKAMP-PETERS S, et al. In vivo FRET-FLIM reveals cell-type-specific protein interactions in Arabidopsis roots[J]. Nature, 2017, 548(7665): 97-102. |
| 34 | SAARI H, LISITSYNA E, RAUTANIEMI K, et al. FLIM reveals alternative EV-mediated cellular up-take pathways of paclitaxel[J]. J Control Release, 2018, 284: 133-143. |
| 35 | RANAWAT H, PAL S, MAZUMDER N. Recent trends in two-photon auto-fluorescence lifetime imaging (2P-FLIM) and its biomedical applications[J]. Biomed Eng Lett, 2019, 9(3): 293-310. |
| 36 | CONG A, PIMENTA R M L, LEE H B, et al. Two-photon fluorescence lifetime imaging of intrinsic NADH in three-dimensional tumor models[J]. Cytometry A, 2019, 95(1): 80-92. |
| 37 | RUCK A, HAUSER C, MOSCH S, et al. Spectrally resolved fluorescence lifetime imaging to investigate cell metabolism in malignant and nonmalignant oral mucosa cells[J]. J Biomed Opt, 2014, 19(9): 096005. |
| 38 | CHEN M, LUO R, LI S, et al. Paper-based strip for ultrasensitive detection of OSCC-associated salivary microRNA via CRISPR/Cas12a coupling with IS-Primer amplification reaction[J]. Anal Chem, 2020, 92(19): 13336-13342. |
| 39 | SABELA M, BALME S, BECHELANY M, et al. A review of gold and silver nanoparticle-based colorimetric sensing assays[J]. Adv Eng Mater, 2017, 19(12): 1700270. |
| 40 | HAJIZADEH S, FARHADI K, FOROUGH M, et al. Silver nanoparticles in the presence of Ca2+ as a selective and sensitive probe for the colorimetric detection of cysteine[J]. Anal Methods, 2012, 4(6): 1747-1752. |
| 41 | GHASEMI F, HORMOZI-NEZHAD M R, MAHMOUDI M. A colorimetric sensor array for detection and discrimination of biothiols based on aggregation of gold nanoparticles[J]. Anal Chim Acta, 2015, 882: 58-67. |
| 42 | SONG G, XU C, LI B. Visual chiral recognition of mandelic acid enantiomers with l-tartaric acid-capped gold nanoparticles as colorimetric probes[J]. Sens Actuators B: Chem, 2015, 215: 504-509. |
| 43 | CAO R, LI B, ZHANG Y, et al. Naked-eye sensitive detection of nuclease activity using positively-charged gold nanoparticles as colorimetric probes[J]. Chem Commun, 2011, 47(45): 12301-12303. |
| 44 | HE S, LIN X, LIANG H, et al. Colorimetric detection of Cr(Ⅵ) using silver nanoparticles functionalized with PVP[J]. Anal Methods, 2019, 11(45): 5819-5825. |
| 45 | GAO Y, HU Z, WU J, et al. Size-tunable Au@Ag nanoparticles for colorimetric and SERS dual-mode sensing of palmatine in traditional Chinese medicine[J]. J Pharm Anal, 2019, 174: 123-133. |
| 46 | GHOSH S K, PAL T. Interparticle coupling effect on the surface plasmon resonance of gold nanoparticles: from theory to applications[J]. Chem Rev, 2007, 107(11): 4797-4862. |
| 47 | GUO C, IRUDAYARAJ J. fluorescent Ag clusters via a protein-directed approach as a Hg(Ⅱ) ion sensor[J]. Anal Chem, 2011, 83(8): 2883-2889. |
| 48 | WANG Y, ZHANG P, MAO X, et al. Seed-mediated growth of bimetallic nanoparticles as an effective strategy for sensitive detection of vitamin C[J]. Sens Actuators B: Chem, 2016, 231: 95-101. |
| 49 | XU S, OUYANG W, XIE P, et al. Highly uniform gold nanobipyramids for ultrasensitive colorimetric detection of influenza virus[J]. Anal Chem, 2017, 89(3): 1617-1623. |
| 50 | WANG Y, ZENG Y, FU W, et al. Seed-mediated growth of Au@Ag core-shell nanorods for the detection of ellagic acid in whitening cosmetics[J]. Anal Chim Acta, 2018, 1002: 97-104. |
| 51 | HUANG C C, HUNG Y L, SHIANG Y C, et al. Photoassisted synthesis of luminescent mannose-Au nanodots for the detection of thyroglobulin in serum[J]. Chem Asian J, 2010, 5(2): 334-341. |
| 52 | HUANG C C, YANG Z, LEE K H, et al. Synthesis of highly fluorescent gold nanoparticles for sensing mercury(Ⅱ)[J]. Angew Chem Int Ed, 2007, 119(36): 6948-6952. |
| 53 | XIA Y, XIA X, PENG H C. Shape-controlled synthesis of colloidal metal nanocrystals: thermodynamic versus kinetic products[J]. J Am Chem Soc, 2015, 137(25): 7947-7966. |
| 54 | NIU W, ZHANG L, XU G. Seed-mediated growth of noble metal nanocrystals: crystal growth and shape control[J]. Nanoscale, 2013, 5(8): 3172-3181. |
| 55 | LEE S, NAM Y S, CHOI S H, et al. Highly sensitive photometric determination of cyanide based on selective etching of gold nanorods[J]. Mikrochim Acta, 2016, 183(11): 3035-3041. |
| 56 | WANG D, ZHANG Y, ZHAO X, et al. Plasmonic colorimetric biosensor for visual detection of telomerase activity based on horseradish peroxidase-encapsulated liposomes and etching of Au nanobipyramids[J]. Sens Actuators B: Chem, 2019, 296: 126646. |
| 57 | ZHANG Y, JIAO J, WEI Y, et al. Plasmonic colorimetric biosensor for sensitive exosome detection via enzyme-induced etching of gold nanobipyramid@MnO2 nanosheet nanostructures[J]. Anal Chem, 2020, 92(22): 15244-15252. |
| 58 | HOUGH K P, CHANDA D, DUNCAN S R, et al. Exosomes in immunoregulation of chronic lung diseases[J]. Allergy, 2017, 72(4): 534-544. |
| 59 | LIU Y, XU E, XU C, et al. Colorimetric method for PARP-1 detection based on preventing AuNRs from etching by molybdate[J]. Sens Actuators B: Chem, 2020, 325: 128806. |
| 60 | LI N, SU X, LU Y. Nanomaterial-based biosensors using dual transducing elements for solution phase detection[J]. Analyst, 2015, 140(9): 2916-2943. |
| 61 | MEDINTZ I L, MATTOUSSI H. Quantum dot-based resonance energy transfer and its growing application in biology[J]. Phys Chem Chem Phys, 2009, 11(1): 17-45. |
| 62 | ALGAR W R, SUSUMU K, DELEHANTY J B, et al. Semiconductor quantum dots in bioanalysis: crossing the valley of death[J]. Anal Chem, 2011, 83(23): 8826-8837. |
| 63 | HE Y F, CHENG K, ZHONG Z T, et al. Simultaneous labeling and multicolor fluorescence imaging of multiple immune cells on liver frozen section by polychromatic quantum dots below freezing points[J]. J Colloid Interface Sci, 2023, 636: 42-54. |
| 64 | HE Y, WANG Y, MAO G, et al. Ratiometric fluorescent nanoprobes based on carbon dots and multicolor CdTe quantum dots for multiplexed determination of heavy metal ions[J]. Anal Chim Acta, 2022, 1191: 339251. |
| 65 | GOLDMAN E R, CLAPP A R, ANDERSON G P, et al. Multiplexed toxin analysis using four colors of quantum dot fluororeagents[J]. Anal Chem, 2004, 76(3): 684-688. |
| 66 | LIU X, CHENG Y, GUAN B, et al. Quantum dot nanobeads as multicolor labels for simultaneous multiplex immunochromatographic detection of four nitrofuran metabolites in aquatic products[J]. Molecules, 2022, 27(23): 8324. |
| 67 | CHEN P, WU J, FEI H, et al. Acoustofluidics-manipulated triple-emission fluorescent nanoprobe aggregates with multicolor-variation for colorimetric quantitative assay[J]. Chem Eng J, 2022, 441: 135976. |
| 68 | ZHOU J, LIU Z, LI F. Upconversion nanophosphors for small-animal imaging[J]. Chem Soc Rev, 2012, 41(3): 1323-1349. |
| 69 | SUN L D, WANG Y F, YAN C H. Paradigms and challenges for bioapplication of rare earth upconversion luminescent nanoparticles: small size and tunable emission/excitation spectra[J]. Acc Chem Res, 2014, 47(4): 1001-1009. |
| 70 | ZHANG M, TIAN K, WANG S, et al. Color variation of the up-conversion luminescence in Er3+-Yb3+ co-doped lead germanate glasses and microsphere integrated devices[J]. J Light Technol, 2020, 38(16): 4397-4401. |
| 71 | MA B L, ZHAI X, DU G, et al. Orthogonal shortwave infrared emission based on rare earth nanoparticles for interference-free logical codes and bio-imaging[J]. Chem Sci, 2019, 10(11): 3281-3288. |
| 72 | WANG L, HARUNA S A, AHMAD W, et al. Tunable multiplexed fluorescence biosensing platform for simultaneous and selective detection of paraquat and carbendazim pesticides[J]. Food Chem, 2022, 388: 132950. |
| 73 | CHEN C, CAO J, WANG X, et al. A novel dual-flux immunochromatographic test strip based on luminescence resonance energy transfer for simultaneous detection of ochratoxin A and deoxynivalenol[J]. Mikrochim Acta, 2022, 189(12): 466. |
| 74 | YOU M, LIN M, GONG Y, et al. Household fluorescent lateral flow strip platform for sensitive and quantitative prognosis of heart failure using dual-color upconversion nanoparticles[J]. ACS Nano, 2017, 11(6): 6261-6270. |
| 75 | AMEEIV S S M, MOHAMMED N M S, UMER K M. Visual monitoring of silver ions and cysteine using bi-ligand Eu-based metal organic framework as a reference signal: color tonality[J]. Microchem, 2022, 181: 107721. |
| 76 | WU S, LI C, SHI H, et al. Design of metal-organic framework-based nanoprobes for multicolor detection of DNA targets with improved sensitivity[J]. Anal Chem, 2018, 90(16): 9929-9935. |
| 77 | VRIEZEMA D M, GARCIA P M L, SANCHO OLTRA N, et al. Positional assembly of enzymes in polymersome nanoreactors for cascade reactions[J]. Angew Chem Int Ed, 2007, 46(39): 7378-7382. |
| 78 | WU Y, GUO W, PENG W, et al. Enhanced fluorescence ELISA based on HAT triggering fluorescence “turn-on” with enzyme-antibody dual labeled AuNP probes for ultrasensitive detection of AFP and HBsAg[J]. ACS Appl Mater, 2017, 9(11): 9369-9377. |
| 79 | LIU H, WANG Y, FU R, et al. A multicolor enzyme-linked immunoassay method for visual readout of carbendazim[J]. Anal Methods, 2021, 13(37): 4256-4265. |
| 80 | MA L, ABUGALYON Y, LI X. Multicolorimetric ELISA biosensors on a paper/polymer hybrid analytical device for visual point-of-care detection of infection diseases[J]. Anal Bioanal Chem, 2021, 413(18): 4655-4663. |
| 81 | ALHOGAIL S, SUAIFAN G A R Y, ZOUROB M. Rapid colorimetric sensing platform for the detection of Listeria monocytogenes foodborne pathogen[J]. Biosens Bioelectron, 2016, 86: 1061-1066. |
| 82 | LU S, HU T, WANG S, et al. Ultra-sensitive colorimetric assay system based on the hybridization chain reaction-triggered enzyme cascade amplification[J]. ACS Appl Mater Interfaces, 2017, 9(1): 167-175. |
| 83 | HE S, LIAN H, CAO X, et al. Cascaded enzymatic reaction-mediated multicolor pixelated quantitative system integrated microfluidic wearable analytical device (McPiQ-μWAD) for non-invasive and sensitive glucose diagnostics[J]. Sens Actuators B: Chem, 2022, 369: 132345. |
| 84 | HU Y, LI J, LI X. Leek-derived codoped carbon dots as efficient fluorescent probes for dichlorvos sensitive detection and cell multicolor imaging[J]. Anal Bioanal Chem, 2019, 411(29): 7879-7887. |
| 85 | CHEN J, MA Q, LI M, et al. Glucose-oxidase like catalytic mechanism of noble metal nanozymes[J]. Nat Commun, 2021, 12(1): 3375. |
| 86 | XI Z, WEI K, WANG Q, et al. Nickel-platinum nanoparticles as peroxidase mimics with a record high catalytic efficiency[J]. J Am Chem Soc, 2021, 143(7): 2660-2664. |
| 87 | LI W, CHEN B, ZHANG H, et al. BSA-stabilized Pt nanozyme for peroxidase mimetics and its application on colorimetric detection of mercury(Ⅱ) ions[J]. Biosens Bioelectron, 2015, 66: 251-258. |
| 88 | PENG H, LI X F, ZHANG H, et al. A microRNA-initiated DNAzyme motor operating in living cells[J]. Nat Commun, 2017, 8(1): 14378. |
| 89 | MASUD M K, YADAV S, ISLAM M N, et al. Gold-loaded nanoporous ferric oxide nanocubes with peroxidase-mimicking activity for electrocatalytic and colorimetric detection of autoantibody[J]. Anal Chem, 2017, 89(20): 11005-11013. |
| 90 | TIAN Z, YAO T, QU C, et al. Photolyase-like catalytic behavior of CeO2[J]. Nano Lett, 2019, 19(11): 8270-8277. |
| 91 | HUANG Y, REN J, QU X. Nanozymes: classification, catalytic mechanisms, activity regulation, and applications[J]. Chem Rev, 2019, 119(6): 4357-4412. |
| 92 | ZHANG Z, BRAGG L M, SERVOS M R, et al. Gold nanoparticles as dehydrogenase mimicking nanozymes for estradiol degradation[J]. Chin Chem Lett, 2019, 30(9): 1655-1658. |
| 93 | ZHANG J, WU S, MA L, et al. Graphene oxide as a photocatalytic nuclease mimicking nanozyme for DNA cleavage[J]. Nano Res, 2020, 13(2): 455-460. |
| 94 | LI W, PENG W, ZHANG Y, et al. A lateral flow strip biosensor platform based on cascade nucleic acid amplification technology for ultrasensitive detection of OSCC-associated salivary microRNA[J]. Anal Chim Acta, 2022, 1221: 340112. |
| 95 | GONG L, ZHAO Z, LV Y F, et al. DNAzyme-based biosensors and nanodevices[J]. Chem Commun, 2015, 51(6): 979-995. |
| 96 | LU Y. Metalloprotein and metallo-DNA/RNAzyme design: current approaches, success measures, and future challenges[J]. Inorg Chem, 2006, 45(25): 9930-9940. |
| 97 | WAN Y H, ZHOU Y J, XIAO K J, et al. Target-assisted self-cleavage DNAzyme probes for multicolor simultaneous imaging of tumor-related microRNAs with signal amplification[J]. Chem Commun, 2019, 55(22): 3278-3281. |
| 98 | SANG P, LU G, YU D, et al. Simultaneous determination of antibiotics, mycotoxins, and hormones in milk by an 8-17 DNAzyme-based enzyme-linked immunosorbent assay[J]. J Agric Food Chem, 2022, 70(39): 12681-12688. |
| 99 | ZHU D, WEI Y, SUN T, et al. Encoding DNA frameworks for amplified multiplexed imaging of intracellular microRNAs[J]. Anal Chem, 2021, 93(4): 2226-2234. |
| 100 | LI D Y, LI Z Y, HAN L, et al. Target-induced tripedal G-quadruplex DNAzyme for multicolor visual point-of-care testing of biomarkers using Au nanorods-decorated electrospun nanofibrous films[J]. Sens Actuators B: Chem, 2022, 371: 132510. |
| 101 | CHEN Q, LI S, LIU Y, et al. Size-controllable Fe-N/C single-atom nanozyme with exceptional oxidase-like activity for sensitive detection of alkaline phosphatase[J]. Sens Actuators B: Chem, 2020, 305: 127511. |
| 102 | LUO D, HUANG X, LIU B, et al. Facile colorimetric nanozyme sheet for the rapid detection of glyphosate in agricultural products based on inhibiting peroxidase-like catalytic activity of porous Co3O4 nanoplates[J]. J Agric Food Chem, 2021, 69(11): 3537-3547. |
| 103 | LIU D, JU C, HAN C, et al. Nanozyme chemiluminescence paper test for rapid and sensitive detection of SARS-CoV-2 antigen[J]. Biosens Bioelectron, 2021, 173: 112817. |
| 104 | HUANG L, NIU Y, LI R, et al. VOx quantum dots with multienzyme-mimic activities and the application in constructing a three-dimensional (3D) coordinate system for accurate discrimination of the hydrogen peroxide over a broad concentration range[J]. Anal Chem, 2019, 91(9): 5753-5761. |
| 105 | LI X, CAI M, SHEN Z, et al. “Three-in-one” nanocomposites as multifunctional nanozymes for ultrasensitive ratiometric fluorescence detection of alkaline phosphatase[J]. J Mater Chem B, 2022, 10(33): 6328-6337. |
| 106 | FAN D, SHANG C, GU W, et al. Introducing ratiometric fluorescence to MnO2 nanosheet-based biosensing: a simple, label-free ratiometric fluorescent sensor programmed by cascade logic circuit for ultrasensitive GSH detection[J]. ACS Appl Mater Interfaces, 2017, 9(31): 25870-25877. |
| 107 | ZHANG Y, XU X, ZHANG L. Capsulation of red emission chromophore into the CoZn ZIF as nanozymes for on-site visual cascade detection of phosphate ions, o-phenylenediamine, and benzaldehyde[J]. Sci Total Environ, 2023, 856: 159091. |
| 108 | SONG Y, CAI X, OSTERMEYER G, et al. Self-assembling allochroic nanocatalyst for improving nanozyme-based immunochromatographic assays[J]. ACS Sens, 2021, 6(1): 220-228. |
| [1] | 舒杨, 杨曼, 李志豪, 王建华. 基于金纳米粒子聚集与杂交链式扩增的microRNA传感[J]. 应用化学, 2024, 41(1): 109-117. |
| [2] | 张月霞, 范小鹏, 曹宇娟, 杨欣彤, 李忠平, 杨振华, 董川. 热解法制备油溶性碳量子点用于土霉素的检测[J]. 应用化学, 2023, 40(4): 509-517. |
| [3] | 周叶红, 张旭艺, 芦冬涛, 徐会文, 刘洋, 董川. 基于氮化碳量子点/罗丹明B系统检测汞离子的比率荧光探针[J]. 应用化学, 2023, 40(11): 1550-1557. |
| [4] | 张鼎, 杨微微, 缪嵩松, 苏怡. 含氮多孔碳限域金纳米粒子用于电化学检测液相二氧化氯[J]. 应用化学, 2023, 40(11): 1572-1580. |
| [5] | 王文栋, 李在均. 钌-石墨烯量子点人工酶合成及用于胡萝卜中辛硫磷的光度检测[J]. 应用化学, 2022, 39(8): 1285-1293. |
| [6] | 唐连波, 付大友, 陈琦, 奉阳润, 熊桠林, 王竹青. 碳量子点增强气液相化学发光检测二氧化碳[J]. 应用化学, 2022, 39(8): 1294-1302. |
| [7] | 郭峤志, 杨振华, 张月霞, 孟雅婷, 曹宇娟, 孙宣森, 张琪琦, 双少敏, 董川. 基于柠檬酸的石墨烯量子点的制备及其应用[J]. 应用化学, 2022, 39(6): 888-899. |
| [8] | 董国华, 郝丽娟, 张文治, 柴东凤, 赵明, 郎坤. 碳量子点纳米材料在铅卤钙钛矿太阳能电池中的应用研究进展[J]. 应用化学, 2022, 39(5): 707-722. |
| [9] | 徐一鑫, 王爽, 全静, 高婉婷, 宋天群, 杨梅. 二硫化钼量子点/还原氧化石墨烯复合材料的制备及其光催化降解有机染料、四环素和Cr(VI)[J]. 应用化学, 2022, 39(5): 769-778. |
| [10] | 吕金枝, 张鑫浩. 胆碱氧化酶功能化室温磷光量子点的制备及其对氯化胆碱的定量检测[J]. 应用化学, 2022, 39(5): 828-836. |
| [11] | 彭红珍, 张瑜, 郭琳洁, 宋伟, 李晴暖, 孟祥英. 一步原位还原法制备WS2@Au量子点复合物及其传感应用[J]. 应用化学, 2022, 39(3): 480-488. |
| [12] | 张萍萍, 杨高岭, 康果果, 石建兵, 钟海政. 量子点光刻技术及其显示应用[J]. 应用化学, 2021, 38(9): 1175-1188. |
| [13] | 薄纯辉, 姜维佳, 王玉高, 石利红, 董川. 煤基碳量子点合成研究进展[J]. 应用化学, 2021, 38(7): 767-788. |
| [14] | 温广明, 焦婷, 杜孝艳, 李忠平. 硫氮共掺杂碳量子点的制备及应用[J]. 应用化学, 2021, 38(6): 722-730. |
| [15] | 赵越, 孟祥芹, 阎锡蕴, 范克龙. 纳米酶:一种新型的生物安全材料[J]. 应用化学, 2021, 38(5): 524-545. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||