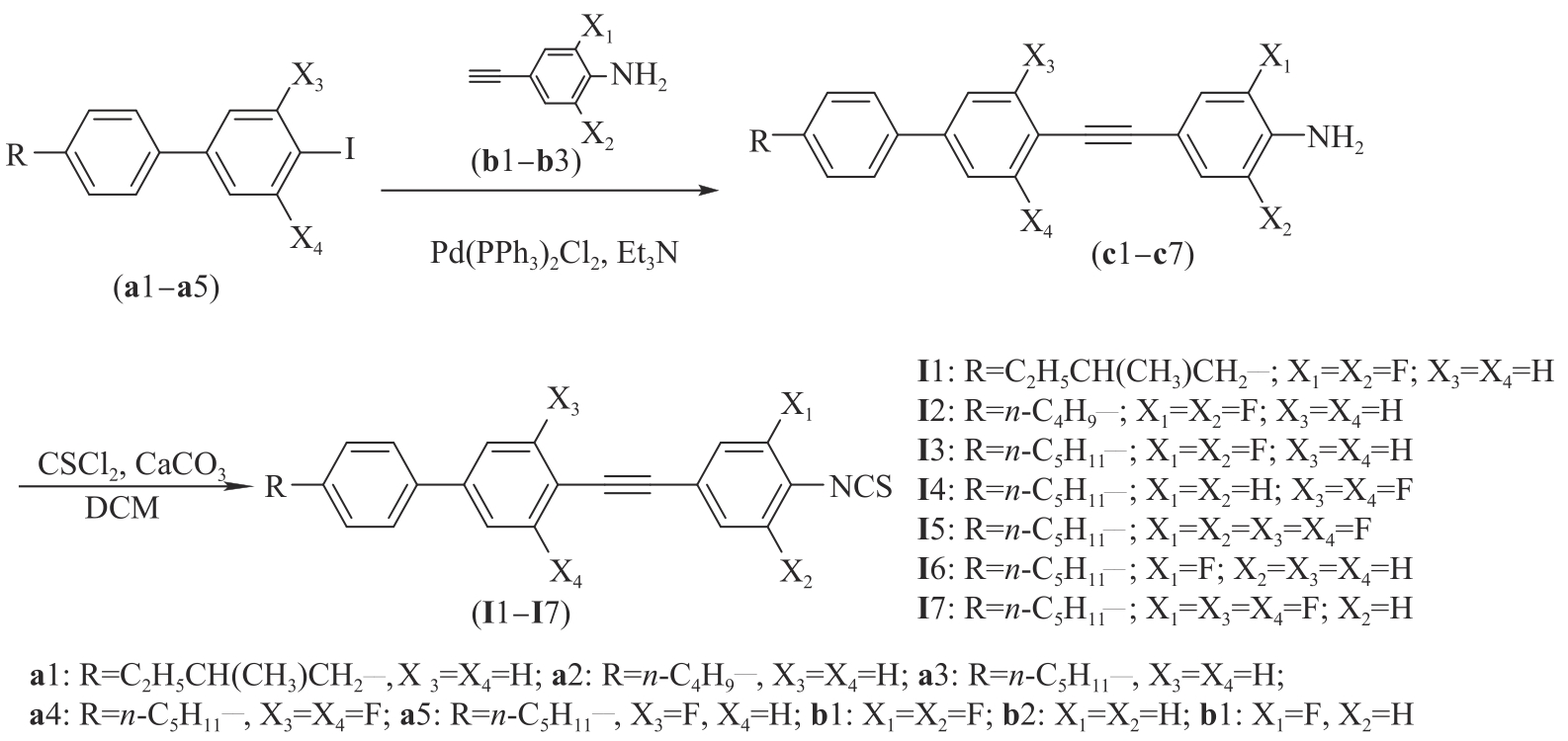
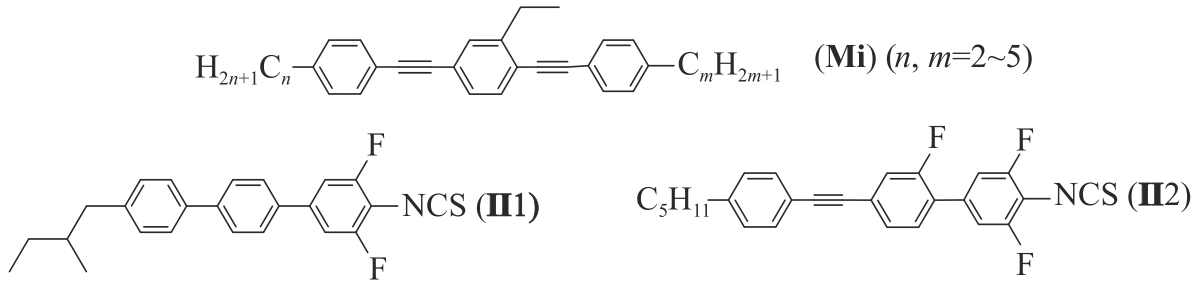
Chinese Journal of Applied Chemistry ›› 2023, Vol. 40 ›› Issue (12): 1682-1692.DOI: 10.19894/j.issn.1000-0518.230178
• Full Papers • Previous Articles Next Articles
Synthesis and Microwave Properties of Fluorinated Triphenylacetylene Isothiocyano Liquid Crystals
Shi-Wei LI1, Xu HU1, Chen-Xiao LUO1, Zhi-He LIU1, Zhi-Yong ZHANG1( ), Xiang-Ru WANG2
), Xiang-Ru WANG2
- 1.Department of Chemistry and Envionmental Engineering,Wuhan Polytechnic University,Wuhan 430023,China
2.School of Electronic Science and Engineering,University of Electronic Science and Technology,Chengdu 611730,China
-
Received:2023-06-17Accepted:2023-11-21Published:2023-12-01Online:2024-01-03 -
Contact:Zhi-Yong ZHANG -
About author:zzy6211@126.com
-
Supported by:the the Major Pre?research Project Fund of Equipment Development Department of Central Military(61409230701);the Key-Area Research and Development Program of Guangdong Province(2019B010158001)
CLC Number:
Cite this article
Shi-Wei LI, Xu HU, Chen-Xiao LUO, Zhi-He LIU, Zhi-Yong ZHANG, Xiang-Ru WANG. Synthesis and Microwave Properties of Fluorinated Triphenylacetylene Isothiocyano Liquid Crystals[J]. Chinese Journal of Applied Chemistry, 2023, 40(12): 1682-1692.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.230178
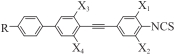 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Comp. | R― | X1 | X2 | X3 | X4 | Phase transition temperature/℃(ΔH, kJ/mol) | ΔTS/℃ | ΔTN/℃ | Δn* |
| Ⅰ1 | C2H5CH(CH3)CH2— | F | F | H | H | Cr 45.8(22.33) SmA 54.0(17.15) N 171.3(0.99) Iso | 8.2 | 117.3 | 0.463 |
| Ⅰ2 | n-C4H9— | F | F | H | H | Cr 67.2(22.58) SmA 104.9(17.94) N 210.3 (0.10) Iso | 37.7 | 105.4 | 0.467 |
| Ⅰ3 | n-C5H11— | F | F | H | H | Cr 55.0(22.43) Sm 119.0(18.23) N 208.5(0.84) Iso | 64.0 | 89.5 | 0.465 |
| Ⅰ4 | n-C5H11— | H | H | F | F | Cr 96.6(24.61) N 218.8(1.01) Iso | — | 122.2 | 0.450 |
| Ⅰ5 | n-C5H11— | F | F | F | F | Cr 108.5(22.75) N 202.9(0.87) Iso | — | 94.4 | 0.433 |
| Ⅰ6 | n-C5H11— | F | H | H | H | Cr 140.8(29.36) N 241.3(0.96) Iso | — | 100.5 | 0.471 |
| Ⅰ7 | n-C5H11— | F | H | F | F | Cr 96.1(21.66) N 218.8(1.34) Iso | — | 127.7 | 0.445 |
| Ⅱ1 | Cr 89(48.18) N 161(0.92) Iso | — | 72 | 0.396 | |||||
| Ⅱ2 | Cr 49.7(26.9) N 186.6(2.9) Iso | — | 137.2 | 0.424 | |||||
Table 1 Phase transition temperature and optical anisotropy of target compounds
 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Comp. | R― | X1 | X2 | X3 | X4 | Phase transition temperature/℃(ΔH, kJ/mol) | ΔTS/℃ | ΔTN/℃ | Δn* |
| Ⅰ1 | C2H5CH(CH3)CH2— | F | F | H | H | Cr 45.8(22.33) SmA 54.0(17.15) N 171.3(0.99) Iso | 8.2 | 117.3 | 0.463 |
| Ⅰ2 | n-C4H9— | F | F | H | H | Cr 67.2(22.58) SmA 104.9(17.94) N 210.3 (0.10) Iso | 37.7 | 105.4 | 0.467 |
| Ⅰ3 | n-C5H11— | F | F | H | H | Cr 55.0(22.43) Sm 119.0(18.23) N 208.5(0.84) Iso | 64.0 | 89.5 | 0.465 |
| Ⅰ4 | n-C5H11— | H | H | F | F | Cr 96.6(24.61) N 218.8(1.01) Iso | — | 122.2 | 0.450 |
| Ⅰ5 | n-C5H11— | F | F | F | F | Cr 108.5(22.75) N 202.9(0.87) Iso | — | 94.4 | 0.433 |
| Ⅰ6 | n-C5H11— | F | H | H | H | Cr 140.8(29.36) N 241.3(0.96) Iso | — | 100.5 | 0.471 |
| Ⅰ7 | n-C5H11— | F | H | F | F | Cr 96.1(21.66) N 218.8(1.34) Iso | — | 127.7 | 0.445 |
| Ⅱ1 | Cr 89(48.18) N 161(0.92) Iso | — | 72 | 0.396 | |||||
| Ⅱ2 | Cr 49.7(26.9) N 186.6(2.9) Iso | — | 137.2 | 0.424 | |||||

Fig.3 Dielectric properties of target compounds Ⅰ1-Ⅰ7 in 11~33 GHz at 20 ℃. (A) Dielectric anisotropy curves; (B) Vertical dielectric loss curves; (C) Phase tunability curves; (D) Quality factor curves
| Compound | εr∥ | εr⊥ | Δεr | tanδεr⊥ | tanδεr∥ | τ | η |
|---|---|---|---|---|---|---|---|
| M-Ⅰ1 | 3.679 | 2.610 | 1.069 | 0.007 51 | 0.003 81 | 0.291 1 | 38.76 |
| M-Ⅰ2 | 3.572 | 2.510 | 1.062 | 0.007 31 | 0.003 09 | 0.300 0 | 41.07 |
| M-Ⅰ3 | 3.548 | 2.484 | 1.064 | 0.007 42 | 0.002 90 | 0.301 5 | 40.68 |
| M-Ⅰ4 | 3.601 | 2.66 | 0.941 | 0.007 12 | 0.003 21 | 0.260 9 | 36.65 |
| M-Ⅰ5 | 3.707 | 2.671 | 1.036 | 0.008 85 | 0.004 30 | 0.279 4 | 31.58 |
| M-Ⅰ6 | 3.705 | 2.680 | 1.025 | 0.007 89 | 0.003 75 | 0.276 5 | 35.04 |
| M-Ⅰ7 | 3.658 | 2.661 | 0.997 | 0.008 15 | 0.003 76 | 0.272 5 | 33.44 |
| M-Ⅱ1 | 3.364 | 2.368 | 0.966 | 0.007 66 | 0.003 35 | 0.291 6 | 38.07 |
| M-Ⅱ2 | 3.803 | 2.746 | 1.057 | 0.008 21 | 0.003 71 | 0.277 5 | 33.80 |
| M | 3.634 | 2.639 | 0.955 | 0.008 54 | 0.004 20 | 0.267 5 | 31.33 |
Table 2 The dielectric properties of compounds at 18 GHz(20 ℃)
| Compound | εr∥ | εr⊥ | Δεr | tanδεr⊥ | tanδεr∥ | τ | η |
|---|---|---|---|---|---|---|---|
| M-Ⅰ1 | 3.679 | 2.610 | 1.069 | 0.007 51 | 0.003 81 | 0.291 1 | 38.76 |
| M-Ⅰ2 | 3.572 | 2.510 | 1.062 | 0.007 31 | 0.003 09 | 0.300 0 | 41.07 |
| M-Ⅰ3 | 3.548 | 2.484 | 1.064 | 0.007 42 | 0.002 90 | 0.301 5 | 40.68 |
| M-Ⅰ4 | 3.601 | 2.66 | 0.941 | 0.007 12 | 0.003 21 | 0.260 9 | 36.65 |
| M-Ⅰ5 | 3.707 | 2.671 | 1.036 | 0.008 85 | 0.004 30 | 0.279 4 | 31.58 |
| M-Ⅰ6 | 3.705 | 2.680 | 1.025 | 0.007 89 | 0.003 75 | 0.276 5 | 35.04 |
| M-Ⅰ7 | 3.658 | 2.661 | 0.997 | 0.008 15 | 0.003 76 | 0.272 5 | 33.44 |
| M-Ⅱ1 | 3.364 | 2.368 | 0.966 | 0.007 66 | 0.003 35 | 0.291 6 | 38.07 |
| M-Ⅱ2 | 3.803 | 2.746 | 1.057 | 0.008 21 | 0.003 71 | 0.277 5 | 33.80 |
| M | 3.634 | 2.639 | 0.955 | 0.008 54 | 0.004 20 | 0.267 5 | 31.33 |
| Compound | Structure | μ(x)/D | μ(y)/D | μ(z)/D | μ/D | Δα |
|---|---|---|---|---|---|---|
| Ⅰ1 | 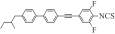 | 6.112 1 | -0.266 3 | 0.215 4 | 6.121 7 | 639.991 6 |
| Ⅰ2 | 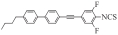 | 6.222 1 | -0.058 9 | 0.181 4 | 6.225 1 | 640.221 8 |
| Ⅰ3 |  | 6.217 1 | -0.093 0 | 0.223 8 | 6.221 8 | 648.888 5 |
| Ⅰ4 |  | -5.992 5 | -0.087 6 | 0.224 0 | 5.997 3 | 653.539 4 |
| Ⅰ5 | 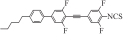 | 7.304 2 | -0.165 9 | 0.273 1 | 7.311 2 | 649.639 0 |
| Ⅰ6 | 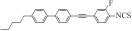 | 5.463 3 | 1.021 8 | 0.252 6 | 5.563 8 | 645.658 8 |
| Ⅰ7 |  | 6.547 1 | 0.927 3 | 0.273 8 | 6.618 1 | 647.337 5 |
| Ⅱ1 | 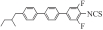 | 6.087 6 | -0.269 5 | 0.229 2 | 6.012 6 | 649.548 8 |
| Ⅱ2 |  | -6.818 3 | 0.738 7 | 0.694 9 | 6.893 3 | 635.347 0 |
Table 3 Calculation of polarizability Δα, dipole moment μ, and the component μ(x), μ(y) and μ(z) of target compounds
| Compound | Structure | μ(x)/D | μ(y)/D | μ(z)/D | μ/D | Δα |
|---|---|---|---|---|---|---|
| Ⅰ1 |  | 6.112 1 | -0.266 3 | 0.215 4 | 6.121 7 | 639.991 6 |
| Ⅰ2 |  | 6.222 1 | -0.058 9 | 0.181 4 | 6.225 1 | 640.221 8 |
| Ⅰ3 |  | 6.217 1 | -0.093 0 | 0.223 8 | 6.221 8 | 648.888 5 |
| Ⅰ4 |  | -5.992 5 | -0.087 6 | 0.224 0 | 5.997 3 | 653.539 4 |
| Ⅰ5 |  | 7.304 2 | -0.165 9 | 0.273 1 | 7.311 2 | 649.639 0 |
| Ⅰ6 |  | 5.463 3 | 1.021 8 | 0.252 6 | 5.563 8 | 645.658 8 |
| Ⅰ7 |  | 6.547 1 | 0.927 3 | 0.273 8 | 6.618 1 | 647.337 5 |
| Ⅱ1 |  | 6.087 6 | -0.269 5 | 0.229 2 | 6.012 6 | 649.548 8 |
| Ⅱ2 |  | -6.818 3 | 0.738 7 | 0.694 9 | 6.893 3 | 635.347 0 |
| 1 | BULJA S, MIRSHEKAR-SYAHKAL D. Meander line millimetre-wave liquid crystal based phase shifter[J]. Electron Lett, 2010, 46(11): 769-771. |
| 2 | TAY F, LEE L H, WANG L. Production scheduling of a MEMS manufacturing system with a wafer bonding process[J]. J Manuf Syst, 2002, 21(4): 287-301. |
| 3 | LIM K C, MARGERUM D J, LACKNER A M, et al. Microwave phase modulation with liquid crystals, EP, 0472403 Ⅰ3[P]. 1992-08-26. |
| 4 | WELL C, LUESSEM G, JAKOBY R J I. Tunable inverted-microstrip phase shifter device using nematic liquid crystals[C]. 2002 IEEE MTT-S International Microwave Symposium Digest, Seattle, WA, USA, 2002: 367-371. |
| 5 | HIRD M, TOYNE K J, GOODBY J W, et al. Synthesis, mesomorphic behaviour and optical anisotropy of some novel materials for nematic mixtures of high birefringence[J]. J Mate Chem, 2004, 14(11): 1731-1743. |
| 6 | DABROWSKI R, KULA P, HERMAN J. High birefringence liquid crystals[J]. Crystals, 2013, 3(3): 443-482. |
| 7 | 彭增辉, 姚丽双, 穆全全, 等. 氟代苯基二苯乙炔基异硫氰酸酯的合成与液晶性质研究[J]. 液晶与显示, 2013, 28(4): 479-483. |
| PENG Z H, YAO L S, MU Q Q, et al. Synthesis and liquid crystal properties of fluorophenyl diphenylethynyl isothiocyanate[J]. Chin J Liq Cryst Disp, 2013, 28 (4): 479-483. | |
| 8 | KOWERDZIEJ R, OLIFIERCZUK M, PARKA J. Thermally induced tunability of a terahertz metamaterial by using a specially designed nematic liquid crystal mixture[J]. Opt Express, 2018, 26(3): 2443-2452. |
| 9 | LIU H, LIU M, GAO S, et al. Synthesis and microwave dielectric properties of polyphenylene liquid crystal compounds with lateral substitution by methyl and fluorine[J]. Liq Cryst, 2021,48(11): 1581-1592. |
| 10 | 李诗妍, 王婵, 关金涛, 等. 侧位含氟苯乙炔类液晶化合物的微波介电性能[J]. 液晶与显示, 2021, 36(7): 7-8. |
| LI S Y, WANG C, GUAN J T, et al. Microwave dielectric properties of lateral fluorophenylacetylene liquid crystal compounds[J]. Chin J Liq Cryst Disp, 2021, 36 (7): 7-8. | |
| 11 | 刘曼曼, 刘豪洁, 鄢道仁, 等. 新型弯曲形双偶氮苯类液晶合成及光致异构性能研究[J]. 有机化学, 2020, 40(1): 125-132. |
| LIU M, LIU H, YAN D, et al. Synthesis and photo-induced isomerization performance for the novel bent diazobenzene liquid crystals[J]. Chin J Org Chem, 2020, 40(1): 125-132. | |
| 12 | 刘胜, 申烦, 黄江涛, 等. 异硫氰基含氟联苯乙炔液晶合成研究[J]. 液晶与显示, 2015, 30(6): 895-903. |
| LIU S, SHEN F, HUANG J T, et al. Study on liquid crystal synthesis of isothiocyano fluorobiphenyl acetylene[J]. Chin J Liq Cryst Disp, 2015, 30(6): 895-903. | |
| 13 | 黄江涛. 高双折射率含氟液晶的合成与性能研究[D]. 武汉: 武汉轻工大学, 2013. |
| HUANG J T. Synthesis and properties of fluorinated liquid crystals with high birefringence[D]. Wuhan: Wuhan Polytechnic University, 2013. | |
| 14 | 张智勇, 李诗研, 向陆军, 等. 一种高介低耗的液晶组合物及其应用: 中国, 110699091A[P]. 2019-10-31. |
| ZHANG Z Y, LI S Y, XIANG L J, et al. Liquid crystal composition with high dielectric and low consumption and its application: China, CN 110699091A[P]. 2019-10-31. | |
| 15 | 卢金应. DL-2-甲基丁基多联苯类液晶材料合成及性能研究[D]. 武汉: 武汉轻工大学, 2020. |
| LU J Y. Synthesis and properties of achiral 2-methylbutyl polyphenyl liquid crystal materials[D]. Wuhan: Wuhan Polytechnic University, 2020. | |
| 16 | LI J, LI J, HU M, et al. The effect of locations of triple bond at terphenyl skeleton on the properties of isothiocyanate liquid crystals[J]. Liq Cryst, 2017, 44(9): 1374-1383. |
| 17 | LI S, CAO Q, WANG C, et al. Studies on the dielectric properties of laterally fluorine-substituted multiphenylacetylene-type liquid crystal compounds at microwave region[J]. J Mol Liq 2021, 337(9): 116600. |
| 18 | KARABEY O H J F. Microwave material properties of nanoparticle-doped nematic liquid crystals[J]. Frequenz, 2015, 69(3/4): 105-109. |
| 19 | 兰庚, 曹秋豪, 欧阳慧琦,等. 含氟三联苯类异硫氰酸酯类液晶合成与微波性能研究[J]. 液晶与显示, 2021, 36(9): 9-11. |
| LAN G, CAO Q H, OUYANG H Q, et al. Synthesis and microwave properties of fluorinated triphenyl isothiocyanate liquid crystals[J]. Chin J Liq Cryst Disp, 2021, 36 (9): 9-11. | |
| 20 | LU J Y, QU Y, YAN D R, et al. Synthesis, characterisation, and effects of molecular structure on phase behaviour of 4-chloro-1,3-diazobenzene bent-core liquid crystals with high photosensitivity[J]. Liq Cryst,2022, 49(4): 442-455. |
| 21 | 李娟利, 安忠维. 侧向单氟取代双烷基环己基联苯类液晶的合成及其相行为[J]. 应用化学, 2007, 24(2): 200-205. |
| LI J L, AN Z W. Synthesis and phase behavior of lateral monofluoro substituted dialkyl cyclohexyl biphenyl liquid crystals[J]. Chin J Appl Chem, 2007, 24(2): 200-205. | |
| 22 | 宋宽广, 李建, 高潮, 等. 三氟丙炔基类液晶的合成及性能[J]. 应用化学, 2017, 34(6): 676-684. |
| SONG K G, LI J, GAO C, et al. Synthesis and properties of trifluoropropynyl liquid crystals[J]. Chin J Appl Chem, 2017, 34(6): 676-684. |
| [1] | Guo-Qing CAI, Jing-Ru DONG, Jun-Ming MO. Green Synthesis and Antibacterial Activity of N‑Benzyl Sulfoximines [J]. Chinese Journal of Applied Chemistry, 2023, 40(12): 1693-1699. |
| [2] | Lei LI, Li LI, Jun-Jie MA, Ju-Biao FU, Chun-Sheng CAI, Mei-Ling CHEN, Xue-Yan QIN, Zhong-Yi YUAN, Yi-Wang CHEN. Synthesis and Properties of Gallium Naphthalene Phthalocyanine Imide Electron Acceptor Materials [J]. Chinese Journal of Applied Chemistry, 2023, 40(11): 1531-1538. |
| [3] | Min-Xiao XU, Liang WANG, Cheng-Fang CHEN. Rongalite Promoted Arylation of Benzoquinone [J]. Chinese Journal of Applied Chemistry, 2023, 40(3): 413-419. |
| [4] | REN Li-Lei, XIAO Li-Wei, LIU Guang-Xian, XU Ming-Ming. Synthesis of Cyclopenta[d]pyrimidinones and Hexahydroquinazolinones Catalyzed by Activated Carbon Supported Sulfuric Acid [J]. Chinese Journal of Applied Chemistry, 2021, 38(1): 44-50. |
| [5] | ZHANG Tianyi, NIAN Wenxia, JIN Ying . Organocatalytic Asymmetric Michael Addition of Anthrone with β-Nitroolefins [J]. Chinese Journal of Applied Chemistry, 2015, 32(4): 422-427. |
| [6] | TANG Nana,LIU Leifang . Research Progress in Suzuki Coupling Reaction of Potassium Organotrifluoroborates [J]. Chinese Journal of Applied Chemistry, 2015, 32(1): 10-18. |
| [7] | YAO Yuanyong1,2, WU Lanyan1, SHU Hua1, TANG Bangcheng1*, XUE Wei2*. Synthesis of α-Aminophosphonates by Bronsted Acid Catalysis [J]. Chinese Journal of Applied Chemistry, 2014, 31(12): 1428-1433. |
| [8] | ZHOU Weijie, SUN Xiaoqiang, XI Haitao*. Synthesis and Fluorescence Properties of Hyaluronic Acid Derivatives Containing Fluorinated Coumarin [J]. Chinese Journal of Applied Chemistry, 2014, 31(08): 916-921. |
| [9] | YIN Zhigang*, GUO Haiyan, QIAN Hengyu, ZHANG Tongyan, QIN Jinwan, ZHANG Yingjun. Synthesis and Application of a Special Colorant Used in Preparing Yellow Polyurethane Elastic Membrane [J]. Chinese Journal of Applied Chemistry, 2014, 31(08): 901-910. |
| [10] | ZENG Wei, CHEN Fuxue*. Recent Advances in Radical Trifluoromethylation Reactions [J]. Chinese Journal of Applied Chemistry, 2014, 31(06): 627-641. |
| [11] | XIAO Liwei*, KONG jie, ZHOU Qiuxiang, HU Yueli. One Pot Synthesis of β-Amino Ketone Derivatives Catalyzed by Polystyrene-supported Ferric Chloride [J]. Chinese Journal of Applied Chemistry, 2014, 31(06): 715-720. |
| [12] | KE Fang1*, WU Wen2, LI Peng1, LIN Chen1. Microwave-assisted Synthesis of Benzothiazole Derivatives in Aqueous Solution [J]. Chinese Journal of Applied Chemistry, 2014, 31(06): 721-725. |
| [13] | FENG Cuilan*, XU Haiyun, LIU Lantao, WANG Jing, LIU Ying. Catalytic Synthesis of Diaryl Ethers with Magnetically Recoverable Fe3O4 Nanoparticles Under Solvent-free and Ligand-free Conditions [J]. Chinese Journal of Applied Chemistry, 2014, 31(05): 536-540. |
| [14] | ZHAO Xuejing, ZHU Jie, LU Ming*. Improvement of the Oxidation Process in the Synthesis of 2,6-Diamino-3,5-dinitropyrazine-1-oxide [J]. Chinese Journal of Applied Chemistry, 2014, 31(01): 41-49. |
| [15] | DUAN Zhifang, LI Chongbi*, WU Xufang, CHEN Gang. Solid Phase Grinding Synthesis and Plant Growth Activities of Arylureido-acylthiourea Derivatives [J]. Chinese Journal of Applied Chemistry, 2013, 30(12): 1429-1433. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||