Chinese Journal of Applied Chemistry ›› 2022, Vol. 39 ›› Issue (8): 1190-1201.DOI: 10.19894/j.issn.1000-0518.210507
• Review • Previous Articles Next Articles
Research Progress of Regulation of Driving Forces in Short Peptide Supramolecular Self‑Assembly
Wei-Qiang ZHANG, Chen WANG, Yu-Rong ZHAO, Dong WANG, Ji-Qian WANG( ), Hai XU(
), Hai XU( )
)
- Center for Bioengineering & Biotechnology,College of Chemical Engineering,China University of Petroleum (East China),Qingdao 266580,China
-
Received:2021-10-24Accepted:2022-04-21Published:2022-08-01Online:2022-08-04 -
Contact:Ji-Qian WANG,Hai XU -
About author:xuh@upc.edu.cn
jqwang@upc.edu.cn
-
Supported by:the National Natural Science Foundational of China(22072181)
CLC Number:
Cite this article
Wei-Qiang ZHANG, Chen WANG, Yu-Rong ZHAO, Dong WANG, Ji-Qian WANG, Hai XU. Research Progress of Regulation of Driving Forces in Short Peptide Supramolecular Self‑Assembly[J]. Chinese Journal of Applied Chemistry, 2022, 39(8): 1190-1201.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.210507
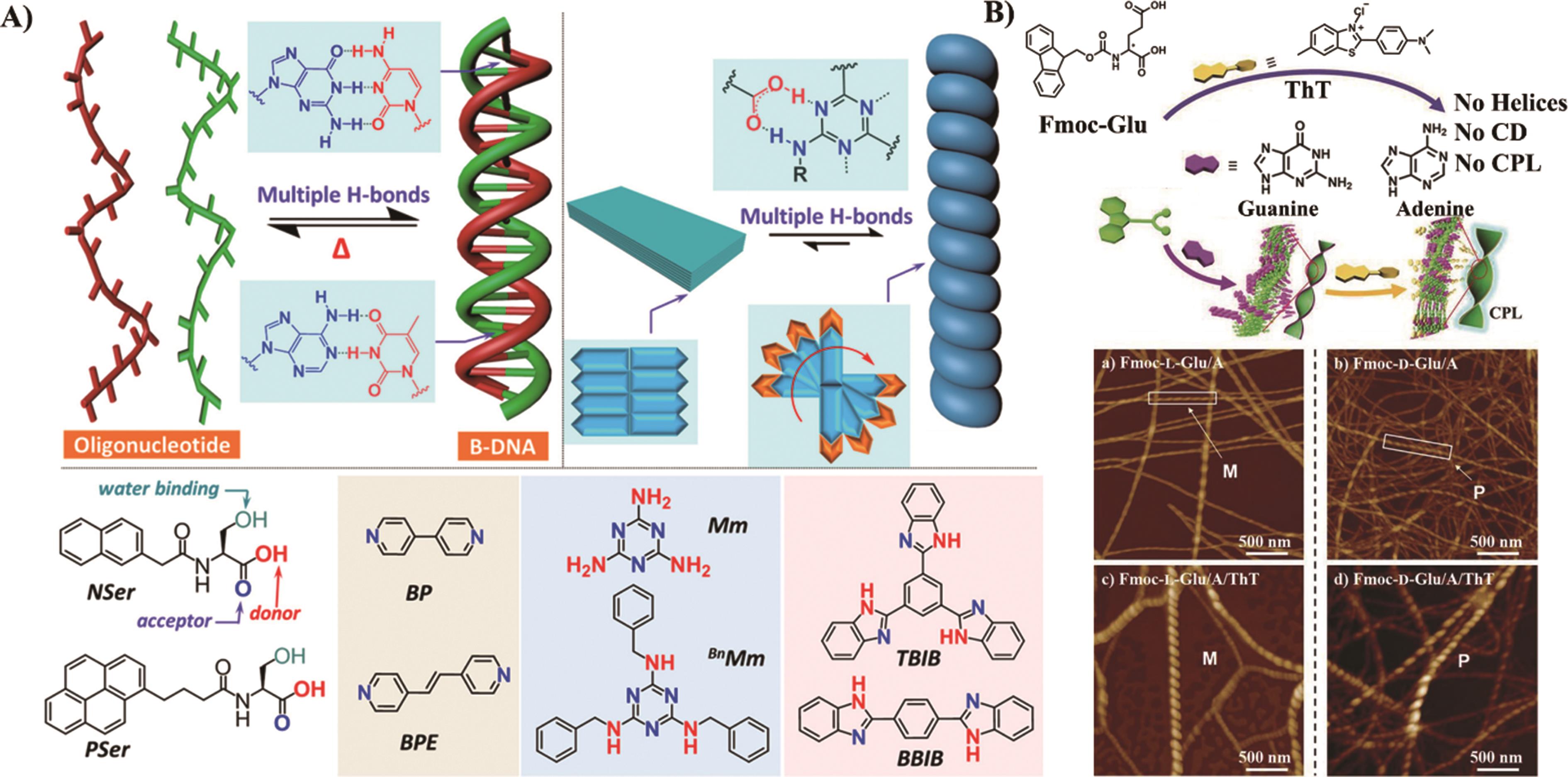
Fig.3 (A) The schematic illustration of aromatic groups chiral stacking by hydrogen bonds and the molecular structure of Nser/Pser, melamine[43]; (B) The co-assembly mechanism and morphology of Fmoc-Glu, Purines (G/A) and ThT[44]
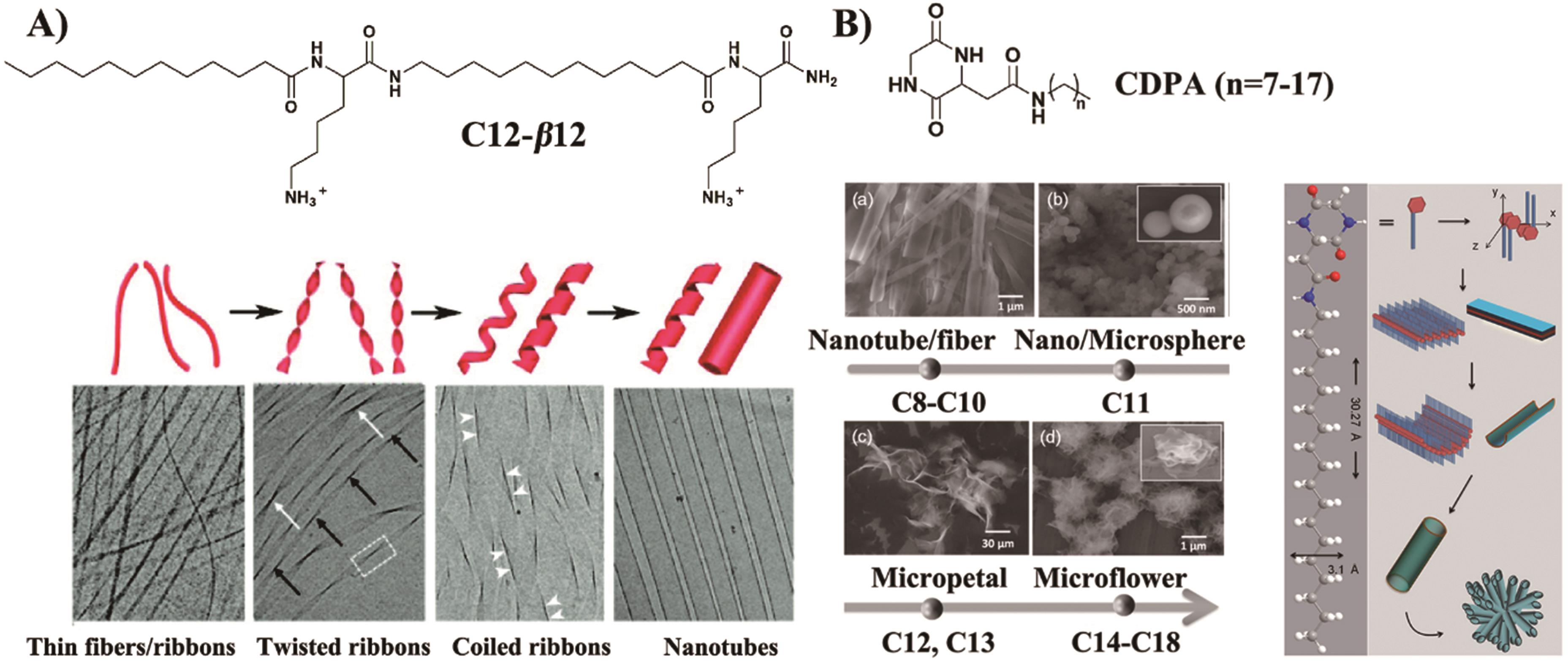
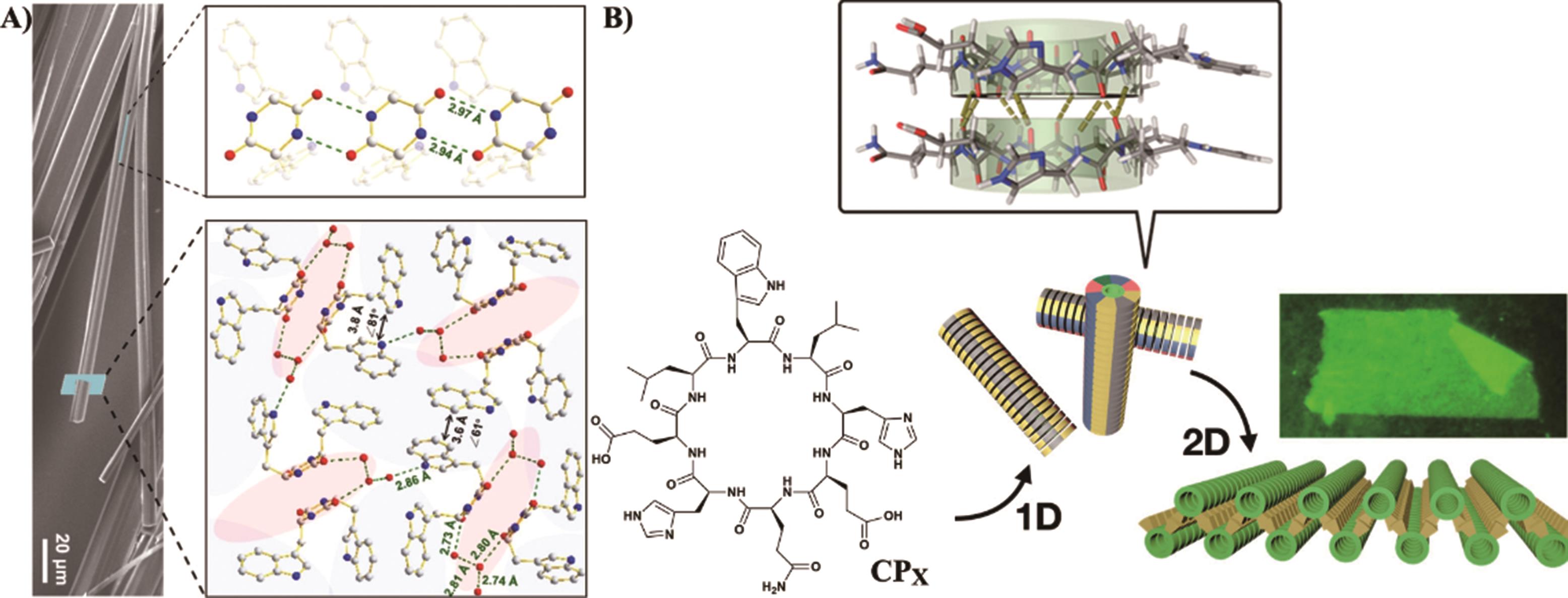
Fig.4 (A) The evolution images of the 1-D supramolecular structures of C12-β12[47]; (B) Morphological evolution of CDPA nano and microarchitectures and schematic diagram of C14-CDPA self-assembly mechanism[48]
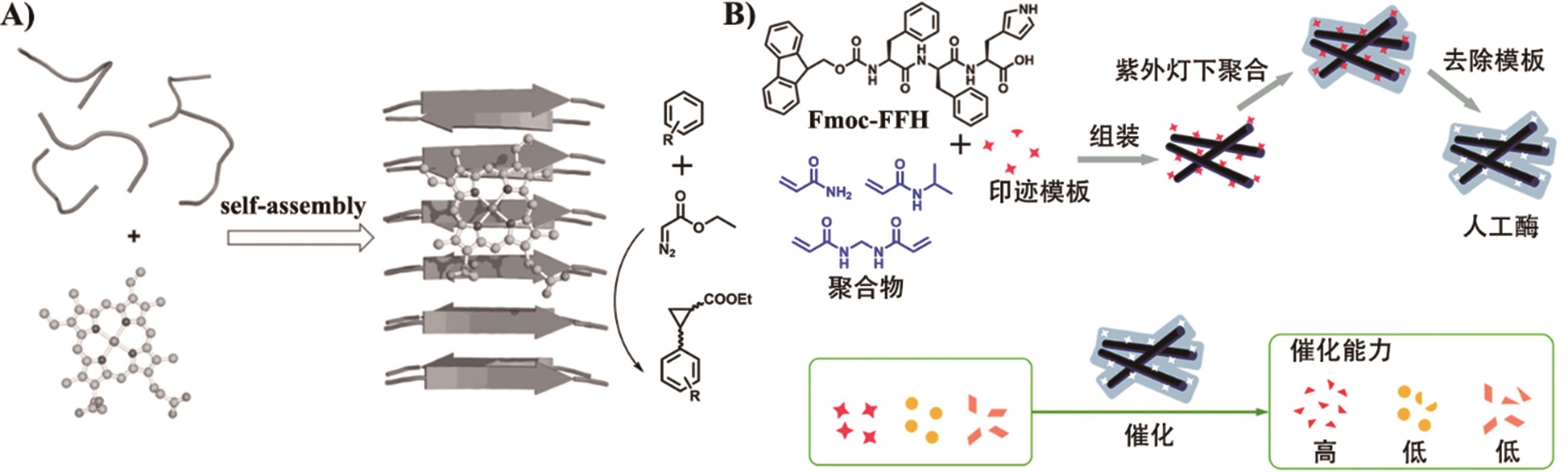
Fig.5 (A) Schematic representation of hemin association with catalytic amyloids to promote cyclopropanation[85]; (B) The preparation and catalytic reaction of a peptide-based artificial hydrolase[87]
| 1 | SONG W L, SONG P, SUN Y J, et al. Self-assembly of multifunctional DNA nanohydrogels with tumor microenvironment-responsive cascade reactions for cooperative cancer therapy[J]. ACS Biomater Sci Eng, 2021, 7(11): 5165-5174. |
| 2 | BISWAS S, DATTA L P, KUMAR DAS T. A bioinspired stimuli-responsive amino acid-based antibacterial drug delivery system in cancer therapy[J]. New J Chem, 2022, 46(15): 7024-7031. |
| 3 | FANG C, ZHAO Q Q, ZHAO F P, et al. All-vacuum deposited perovskite solar cells with glycine modified NiOx hole-transport layers[J]. RSC Adv, 2022, 12(18): 10863-10869. |
| 4 | SURíS-VALLS R, HOGERVORST T P, SCHOENMAKERS S M C, et al. Inhibition of ice recrystallization by nanotube-forming cyclic peptides[J]. Biomacromolecules, 2022, 23(2): 520-529. |
| 5 | WEERAKKODY J S, EL KAZZY M, JACQUIER E, et al. Surfactant-like peptide self-assembled into hybrid nanostructures for electronic nose applications[J]. ACS Nano, 2022, 16(3): 4444-4457. |
| 6 | CHEN T T, LYU Y F, TAN M S, et al. Fabrication of supramolecular antibacterial nanofibers with membrane-disruptive mechanism[J]. J Med Chem, 2021, 64(22): 16480-16496. |
| 7 | ENGELBERG Y, RAGONIS-BACHAR P, LANDAU M. Rare by natural selection: disulfide-bonded supramolecular antimicrobial peptides[J]. Biomacromolecules, 2022, 23(3): 926-936. |
| 8 | ABBAS M, SUSAPTO H H, HAUSER C A E. Synthesis and organization of gold-peptide nanoparticles for catalytic activities[J]. ACS Omega, 2022, 7(2): 2082-2090. |
| 9 | WU Y T, LI Q, CAO J J, et al. Pillararene-peptide nanogels and their biomimetic mineralization hybrids for heterogeneous catalysis[J]. ACS Appl Nano Mater, 2021, 4(10): 11126-11133. |
| 10 | GONG Z Y, ZHOU B L, LIU X Y, et al. Enzyme-induced transformable peptide nanocarriers with enhanced drug permeability and retention to improve tumor nanotherapy efficacy[J]. ACS Appl Mater Interfaces, 2021, 13(47): 55913-55927. |
| 11 | KULKARNI K, MINEHAN R L, GAMOT T, et al. Esterase-mediated sustained release of peptide-based therapeutics from a self-assembled injectable hydrogel[J]. ACS Appl Mater Interfaces, 2021, 13(49): 58279-58290. |
| 12 | LI B, CRIADO-GONZALEZ M, ADAM A, et al. Peptide hydrogels assembled from enzyme-adsorbed mesoporous silica nanostructures for thermoresponsive doxorubicin release[J]. ACS Appl Nano Mater, 2022, 5(1): 120-125. |
| 13 | MA Z Y, LIU X H, NIE J L, et al. Nano-antimicrobial peptides based on constitutional isomerism-dictated self-assembly[J]. Biomacromolecules, 2022, 23(3): 1302-1313. |
| 14 | DE MELLO L R, CARRASCOSA V, REBELATO E, et al. Nanostructure formation and cell spheroid morphogenesis of a peptide supramolecular hydrogel[J]. Langmuir, 2022, 38(11): 3434-3445. |
| 15 | WU B H, ZHAO S, YANG X J, et al. Biomimetic heterodimerization of tetrapeptides to generate liquid crystalline hydrogel in atwo-component system[J]. ACS Nano, 2022, 16(3): 4126-4138. |
| 16 | GUO K X, LIU W, WANG Y, et al. Handedness inversion of the self-assemblies of lipotetrapeptides regulated by the shift of the methyl group[J]. New J Chem, 2022, 46(15): 6971-6976. |
| 17 | MASON M L, LIN T, LINVILLE J J, et al. Co-assembly of a multicomponent network of nanofiber-wrapped nanotubes[J]. Nanoscale, 2022, 14(12): 4531-4537. |
| 18 | SIVAGNANAM S, DAS K, BASAK M, et al. Self-assembled dipeptide based fluorescent nanoparticles as a platform for developing cellular imaging probes and targeted drug delivery chaperones[J]. Nanoscale Adv, 2022, 4(6): 1694-1706. |
| 19 | DONG Q Q, WANG M Y, WANG A H, et al. Self-assembly of Fmoc-amino acids in capillary confined space forming a parallel ordered fiber network for application in vascularization[J]. Biomater Sci, 2022, 10(6): 1470-1475. |
| 20 | MACPHERSON D S, MCPHEE S A, ZEGLIS B M, et al. The impact of tyrosine iodination on the aggregation and cleavage kinetics of MMP-9-responsive peptide sequences[J]. ACS Biomater Sci Eng, 2022, 8(2): 579-587. |
| 21 | ZHAO Y R, DENG L, YANG W, et al. Tuning one-dimensional nanostructures of bola-like peptide amphiphiles by varying the hydrophilic amino acids[J]. Chem Eur J, 2016, 22(32): 11394-11404. |
| 22 | ZHENG C, LIN S W, CHEN Y L, et al. Bola-type Ala-Ala dipeptides: odd-even effect in molecular packing structures[J]. Langmuir, 2019, 35(35): 11406-11413. |
| 23 | TAO K, TANG Y M, RENCUS-LAZAR S, et al. Bioinspired supramolecular packing enables high thermo-sustainability[J]. Angew Chem Int Ed, 2020, 59(43): 19037-19041. |
| 24 | ZIGANSHIN M A, SAFIULLINA A S, GERASIMOV A V, et al. Thermally induced self-assembly and cyclization of L-Leucyl-L-Leucine in solid state[J]. J Phys Chem B, 2017, 121(36): 8603-8610. |
| 25 | INSUA I, MONTENEGRO J. 1D to 2D self assembly of cyclic peptides[J]. J Am Chem Soc, 2020, 142(1): 300-307. |
| 26 | DRAPER E R, DIETRICH B, MCAULAY K, et al. Using small-angle scattering and contrast matching to understand molecular packing in low molecular weight gels[J]. Matter, 2020, 2(3): 764-778. |
| 27 | WYCHOWANIEC J K, PATEL R, LEACH J, et al. Aromatic stacking facilitated self-assembly of ultrashort ionic complementary peptide sequence: β-sheet nanofibers with remarkable gelation and interfacial properties[J]. Biomacromolecules, 2020, 21(7): 2670-2680. |
| 28 | MéNARD-MOYON C, VENKATESH V, KRISHNA K V, et al. Self-assembly of tyrosine into controlled supramolecular nanostructures[J]. Chem Eur J, 2015, 21(33): 11681-11686. |
| 29 | BERA S, XUE B, REHAK P, et al. Self-assembly of aromatic amino acid enantiomers into supramolecular materials of high rigidity[J]. ACS Nano, 2020, 14(2): 1694-1706. |
| 30 | ZHANG J K, LIU S N, LI H, et al. Tryptophan-based self-assembling peptides with bacterial flocculation and antimicrobial properties[J]. Langmuir, 2020, 36(38): 11316-11323. |
| 31 | HOSOKAWA T, TAKAHASHI Y, MATSUSHIMA T, et al. Synthesis, structures, and properties of hexapole helicenes: assembling six [5]helicene substructures into highly twisted aromatic systems[J]. J Am Chem Soc, 2017, 139(51): 18512-18521. |
| 32 | SCHUSTER N J, SÁNCHEZ R H, BUKHARINA D, et al. A helicene nanoribbon with greatly amplified chirality[J]. J Am Chem Soc, 2018, 140(20): 6235-6239. |
| 33 | NAKAKUKI Y, HIROSE T, MATSUDA K. Synthesis of a helical analogue of kekulene: a flexible π-expanded helicene with large helical diameter acting as a soft molecular spring[J]. J Am Chem Soc, 2018, 140(45): 15461-15469. |
| 34 | XING Q G, ZHANG J X, XIE Y Y, et al. Aromatic motifs dictate nanohelix handedness of tripeptides[J]. ACS Nano, 2018, 12(12): 12305-12314. |
| 35 | XIE Y Y, WANG Y F, QI W, et al. Reconfigurable chiral self-assembly of peptides through control of terminal charges[J]. Small, 2017, 13(30): 1700999. |
| 36 | MASON M L, LALISSE R F, FINNEGAN T J, et al. pH-controlled chiral packing and self-assembly of a coumarin tetrapeptide[J]. Langmuir, 2019, 35(38): 12460-12468. |
| 37 | BISWAS S, DAS A K. Tuning the handedness: role of chiral component in peptide-appended bolaamphiphile-based coassembled hydrogels[J]. Langmuir, 2019, 35(6): 2383-2391. |
| 38 | LIU Y Q, CHEN C F, WANG T Y, et al. Supramolecular chirality of the two-component supramolecular copolymer gels: who determines the handedness?[J]. Langmuir, 2016, 32(1): 322-328. |
| 39 | WU S S, YIN Z Z, WU D T, et al. Chiral enantioselective assemblies induced from achiral porphyrin by L- and D-lysine[J]. Langmuir, 2019, 35(51): 16761-16769. |
| 40 | XING P Y, LI P Z, CHEN H Z, et al. Understanding pathway complexity of organic micro/nanofiber growth in hydrogen-bonded coassembly of aromatic amino acids[J]. ACS Nano, 2017, 11(4): 4206-4216. |
| 41 | WU A L, GUO Y X, LI X B, et al. Co-assembled supramolecular gel of dipeptide and pyridine derivatives with controlled chirality[J]. Angew Chem Int Ed, 2021, 60(4): 2099-2103. |
| 42 | LIANG J C, HAO A Y, XING P Y, et al. Inverse evolution of helicity from the molecular to the macroscopic level based on N-terminal aromatic amino acids[J]. ACS Nano, 2021, 15(3): 5322-5332. |
| 43 | XING P Y, LI Y X, XUE S X, et al. Occurrence of chiral nanostructures induced by multiple hydrogen bonds[J]. J Am Chem Soc, 2019, 141(25): 9946-9954. |
| 44 | DENG M, ZHANG L, JIANG Y Q, et al. Role of achiral nucleobases in multicomponent chiral self-assembly: purine-triggered helix and chirality transfer[J]. Angew Chem Int Ed, 2016, 55(48): 15062-15066. |
| 45 | CUI H G, MURAOKA T, CHEETHAM A G, et al. Self-assembly of giant peptide nanobelts[J]. Nano Lett, 2009, 9(3): 945-951. |
| 46 | CUI H G, CHEETHAM A G, PASHUCK E T, et al. Amino acid sequence in constitutionally isomeric tetrapeptide amphiphiles dictates architecture of one-dimensional nanostructures[J]. J Am Chem Soc, 2014, 136(35): 12461-12468. |
| 47 | ZISERMAN L, LEE H Y, RAGHAVAN S R, et al. Unraveling the mechanism of nanotube formation by chiral self-assembly of amphiphiles[J]. J Am Chem Soc, 2011, 133(8): 2511-2517. |
| 48 | PANDURANGAN K, ROY B, RAJASEKHAR K, et al. Molecular architectonics of cyclic dipeptide amphiphiles and their application in drug delivery[J]. ACS Appl Bio Mater, 2020, 3(5): 3413-3422. |
| 49 | LEE N R, BOWERMAN C J, NILSSON B L. Effects of varied sequence pattern on the self-assembly of amphipathic peptides[J]. Biomacromolecules, 2013, 14(9): 3267-3277. |
| 50 | ZHAO Y R, WANG J Q, DENG L, et al. Tuning the self-assembly of short peptides via sequence variations[J]. Langmuir, 2013, 29(44): 13457-13464. |
| 51 | ZHAO Y R, DENG L, WANG J Q, et al. Solvent controlled structural transition of KI4K self-assemblies: from nanotubes to nanofibrils[J]. Langmuir, 2015, 31(47): 12975-12983. |
| 52 | WANG Y, ZHENG X J, LIU J, et al. Tumor-targeted anti-VEGF RNAi capable of sequentially responding to intracellular microenvironments for potent systemic tumor suppression[J]. ACS Appl Bio Mater, 2020, 3(12): 9145-9155. |
| 53 | XU F Y, LIU J, TIAN J, et al. Supramolecular self-assemblies with nanoscale RGD clusters promote cell growth and intracellular drug delivery[J]. ACS Appl Mater Interfaces, 2016, 8(44): 29906-29914. |
| 54 | MOYER T J, CUI H G, STUPP S I. Tuning nanostructure dimensions with supramolecular twisting[J]. J Phys Chem B, 2013, 117(16): 4604-4610. |
| 55 | KAUR H, SHARMA P, PATEL N, et al. Accessing highly tunable nanostructured hydrogels in a short ionic complementary peptide sequence via pH trigger[J]. Langmuir, 2020, 36(41): 12107-12120. |
| 56 | CLOVER T M, O’NEILL C L, APPAVU R, et al. Self-assembly of block heterochiral peptides into helical tapes[J]. J Am Chem Soc, 2020, 142(47): 19809-19813. |
| 57 | MARCHESAN S, EASTON C D, STYAN K E, et al. Chirality effects at each amino acid position on tripeptide self-assembly into hydrogel biomaterials[J]. Nanoscale, 2014, 6(10): 5172-5180. |
| 58 | FU Y T, LI B Z, HUANG Z B, et al. Terminal is important for the helicity of the self-assemblies of dipeptides derived from alanine[J]. Langmuir, 2013, 29(20): 6013-6017. |
| 59 | LI Y, LI B Z, FU Y T, et al. Solvent-induced handedness inversion of dipeptide sodium salts derived from alanine[J]. Langmuir, 2013, 29(31): 9721-9726. |
| 60 | WANG M, ZHOU P, WANG J Q, et al. Left or right: how does amino acid chirality affect the handedness of nanostructures self-assembled from short amphiphilic peptides?[J]. J Am Chem Soc, 2017, 139(11): 4185-4194. |
| 61 | SATO K, JI W, ÁLVAREZ Z, et al. Chiral recognition of lipid bilayer membranes by supramolecular assemblies of peptide amphiphiles[J]. ACS Biomater Sci Eng, 2019, 5(6): 2786-2792. |
| 62 | LIN S W, QIN J M, LI Y, et al. Chirality-driven parallel and antiparallel β-sheet secondary structures of Phe-Ala lipodipeptides[J]. Langmuir, 2017, 33(33): 8246-8252. |
| 63 | LI S, YANG Y L, LIN X, et al. A novel particulate delivery system based on antigen-Zn2+ coordination interactions enhances stability and cellular immune response of inactivated foot and mouth disease virus[J]. Mol Pharmaceutics, 2020, 17(8): 2952-2963. |
| 64 | WANG M, ZHENG Z J, LIU C H, et al. Investigating the calcium binding characteristics of black bean protein hydrolysate[J]. Food Funct, 2020, 11(10): 8724-8734. |
| 65 | LI J, ZHAO Y R, ZHOU P, et al. Ordered nanofibers fabricated from hierarchical self-assembling processes of designed α-helical peptides[J]. Small, 2020, 16(45): 2003945. |
| 66 | PERINELLI M, TEGONI M, FREISINGER E. Different behavior of the histidine residue toward cadmium and zinc in a cadmium-specific metallothionein from an aquatic fungus[J]. Inorg Chem, 2020, 59(23): 16988-16997. |
| 67 | MA Q M, ZHANG M, XU X H, et al. Multiresponsive supramolecular luminescent hydrogels based on a nucleoside/lanthanide complex[J]. ACS Appl Mater Interfaces, 2019, 11(50): 47404-47412. |
| 68 | ZAITHUN B S, EMILIA A, MOHAMED T M I, et al. Histidine-based copper tetrapeptides as enantioselective catalysts for aldol reactions[J]. RSC Adv, 2018, 8(59): 34004-34011. |
| 69 | BEN-SHUSHAN S, MILLER Y. Molecular mechanisms and aspects on the role of neuropeptide Y as a Zn2+ and Cu2+ chelator[J]. Inorg Chem, 2021, 60(1): 484-493. |
| 70 | WANG Y, WEI Y Y, GAO P C, et al. Preparation of Fe3O4@PMAA@Ni microspheres towards the efficient and selective enrichment of histidine-rich proteins[J]. ACS Appl Mater Interfaces, 2021, 13(9): 11166-11176. |
| 71 | SHARMA B, SINGH A, SARMA T K, et al. Chirality control of multi-stimuli responsive and self-healing supramolecular metallo-hydrogels[J]. New J Chem, 2018, 42(8): 6427-6432. |
| 72 | ZENG L, SONG M M, GU J, et al. A highly stretchable, tough, fast self-healing hydrogel based on peptide-metal ion coordination[J]. Biomimetics, 2019, 4(2): 36. |
| 73 | SUN L M, LIU D C, FU D J, et al. Fluorescent peptide nanoparticles to detect amyloid-beta aggregation in cerebrospinal fluid and serum for Alzheimer′s disease diagnosis and progression monitoring[J]. Chem Eng J, 2021, 405(2021): 126733. |
| 74 | RAMIREZ-CALDERON G, SUSAPTO H H, HAUSER C A E. Delivery of endothelial cell-laden microgel elicits angiogenesis in self-assembling ultrashort peptide hydrogels in vitro[J]. ACS Appl Mater Interfaces, 2021, 13(25): 29281-29292. |
| 75 | XU P P, WANG X Y, LI T W, et al. Bioinspired microenvironment responsive nanoprodrug as an efficient hydrophobic drug self-delivery system for cancer therapy[J]. ACS Appl Mater Interfaces, 2021, 13(29): 33926-33936. |
| 76 | NGUYEN P K, GAO W, PATEL S D, et al. Self-assembly of a dentinogenic peptide hydrogel[J]. ACS Omega, 2018, 3(6): 5980-5987. |
| 77 | CHAUHAN N, SINGH Y. Self-assembled Fmoc-Arg-Phe-Phe peptide gels with highly potent bactericidal activities[J]. ACS Biomater Sci Eng, 2020, 6(10): 5507-5518. |
| 78 | GONTSARIK M, YAGHMUR A, REN Q, et al. From structure to function: pH-switchable antimicrobial nano-self-assemblies[J]. ACS Appl Mater Interfaces, 2019, 11(3): 2821-2829. |
| 79 | REN P, LI J T, ZHAO L Y, et al. Dipeptide self-assembled hydrogels with shear-thinning and instantaneous self-healing properties determined by peptide sequences[J]. ACS Appl Mate Interfaces, 2020, 12(19): 21433-21440. |
| 80 | CHU B, HE J M, LIU L L, et al. Proangiogenic peptide nanofiber hydrogels for wound healing[J]. ACS Biomater Sci Eng, 2021, 7(3): 1100-1110. |
| 81 | YAN J, WANG Y, LI X, et al. A bionic nano-band-aid constructed by the three-stage self-assembly of peptides for rapid liver hemostasis[J]. Nano Lett, 2021, 21(17): 7166-7174. |
| 82 | SHA X, LI P, FENG Y H, et al. Self-assembled peptide nanofibrils designed to release membrane-lysing antimicrobial peptides[J]. ACS Appl Bio Mater, 2020, 3(6): 3648-3655. |
| 83 | GULSEREN G, KHALILY M A, TEKINAY A B, et al. Catalytic supramolecular self-assembled peptide nanostructures for ester hydrolysis[J]. J Mater Chem B, 2016, 4(26): 4605-4611. |
| 84 | WANG Z G, WANG H, LIU Q, et al. Designed self-assembly of peptides with G-quadruplex/hemin DNAzyme into nanofibrils possessing enzyme-mimicking active sites and catalytic functions[J]. ACS Catal, 2018, 8(8): 7016-7024. |
| 85 | ZOZULIA O, KORENDOVYCH I V. Semi-rationally designed short peptides self-assemble and bind hemin to promote cyclopropanation[J]. Angew Chem Int Ed, 2020, 59(21): 8108-8112. |
| 86 | YANG X J, ZHANG L W, LIANG Y Y, et al. Self-assembled bio-organometallic nanocatalysts for highly enantioselective direct aldol reactions[J]. Langmuir, 2020, 36(46): 13735-13742. |
| 87 | ZHU M J, WANG M F, QI W, et al. Constructing peptide-based artificial hydrolases with customized selectivity[J]. J Mater Chem B, 2019, 7(24): 3804-3810. |
| [1] | Jin-Jian LIU, Yi-Wei LU. Multiple-Responsive Coordination Polymers Based on a Carboxybenzyl Viologen Ligand [J]. Chinese Journal of Applied Chemistry, 2023, 40(3): 397-403. |
| [2] | Jin-Jian LIU, Na LIU, Feng-Yi YANG. Synthesis and Photochromic Properties of Two Isostructural Viologen Coordination Polymers [J]. Chinese Journal of Applied Chemistry, 2023, 40(2): 245-251. |
| [3] | Ren MU, Shun NA, Natsagdorj NARANTSOGT, Chong-Jiu LI. Effect of Dimethyl Sulfoxide on the Retention Time of Acetic Acid in Gas Chromatography and Its Mechanism [J]. Chinese Journal of Applied Chemistry, 2022, 39(5): 852-854. |
| [4] | Hui DU, Chen-Yang YAO, Hao PENG, Bo JIANG, Shun-Xiang LI, Jun-Lie YAO, Fang ZHENG, Fang YANG, Ai-Guo WU. Applications of Transition Metal⁃doped Iron⁃based Nanoparticles in Biomedicine [J]. Chinese Journal of Applied Chemistry, 2022, 39(3): 391-406. |
| [5] | Dong-Qing CAI, Jing ZHANG, Qing-Chuan WU, Jing-Hong YE, Lin-Ying WANG, Dong-Fang WANG. Preparation and Application of Attapulgite Based Nano⁃controlled Chlorpyrifos [J]. Chinese Journal of Applied Chemistry, 2022, 39(11): 1774-1782. |
| [6] | Xiao-Ming XIE, Jia-Qi ZHANG. Hydrogen Bond Interaction Driven Procyanidine Assembly into Underwater Adhesive with Antibacterial Activity [J]. Chinese Journal of Applied Chemistry, 2022, 39(10): 1533-1542. |
| [7] | ZHAO Chang-Li, QIN Ming-Gao, DOU Xiao-Qiu, FENG Chuan-Liang. High Mechanical Stability and Osteogenesis of Chiral Supramolecular Hydrogel Induced by Inorganic Nanoparticles [J]. Chinese Journal of Applied Chemistry, 2022, 39(1): 177-187. |
| [8] | GUO Shao-Bo, LIANG Yan-Li, LIU Zhi-Feng, ZHANG Qiang, MA Jian-Qi. Synergistic Antibacterial Activity of Tetracycline Combined with Silver Nanoparticles [J]. Chinese Journal of Applied Chemistry, 2021, 38(11): 1462-1468. |
| [9] | Meng-Wei LI, Hong-Yan WANG, Ying-Ying WANG, Xiao-Ying KANG, Mei-Fang LIU, Yu-Lan CHEN. Supramolecular Self⁃Assembly of Azobenzene⁃4,4'⁃dicarboxylic Acid Derivatives in Water [J]. Chinese Journal of Applied Chemistry, 2021, 38(10): 1362-1370. |
| [10] | CHENG Yinjie, ZHANG Lu, HU Renjie, WANG Ying, JIN Xuexin, QIU Shuo, ZHANG Haoyan, JIANG Dawei. Preparation and Characterization of Hydrogen-Bonded Carbon Nanotubes Reinforced Self-healing Composites [J]. Chinese Journal of Applied Chemistry, 2020, 37(10): 1147-1155. |
| [11] | Shuai HAO, Dongmei JIANG, Lisheng LIU, Xiaoteng ZHANG, Junchang CHEN, Liangshu XIA. Research Progress on Hydrazine Hydrate Reduction of Metal [J]. Chinese Journal of Applied Chemistry, 2018, 35(7): 756-766. |
| [12] | GUO Zhaonan, ZHANG Bin, SUN Xuejiao, LYU Chengwei, YU Shijun. Synthesis and Properties of Novel Bis-8-Hydroxyquinoline Ligands and Zn(Ⅱ), Al(Ⅲ) Coordination Polymers [J]. Chinese Journal of Applied Chemistry, 2018, 35(5): 544-551. |
| [13] | YAO Huiying, YANG Tao, HUANG Xing, ZHU Jia, LI Qing, XU Wei, CHI Lifeng. Coordination Complexes Based on MX4 Structure as Catalyst for Hydrogen Evolution Reaction [J]. Chinese Journal of Applied Chemistry, 2018, 35(3): 328-341. |
| [14] | LIU Chuantao, XIAO Ting, WANG Fang, CHEN Xiaoqiang. Synthesis of Binol-based Fluorescence Probe and Recognition of Arginine [J]. Chinese Journal of Applied Chemistry, 2018, 35(1): 40-45. |
| [15] | LIU Chuantao, XIAO Ting, WANG Fang, CHEN Xiaoqiang. Synthesis of Binol-based Fluorescence Probe and Recognition of Arginine [J]. Chinese Journal of Applied Chemistry, 2018, 35(1): 0-0. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||