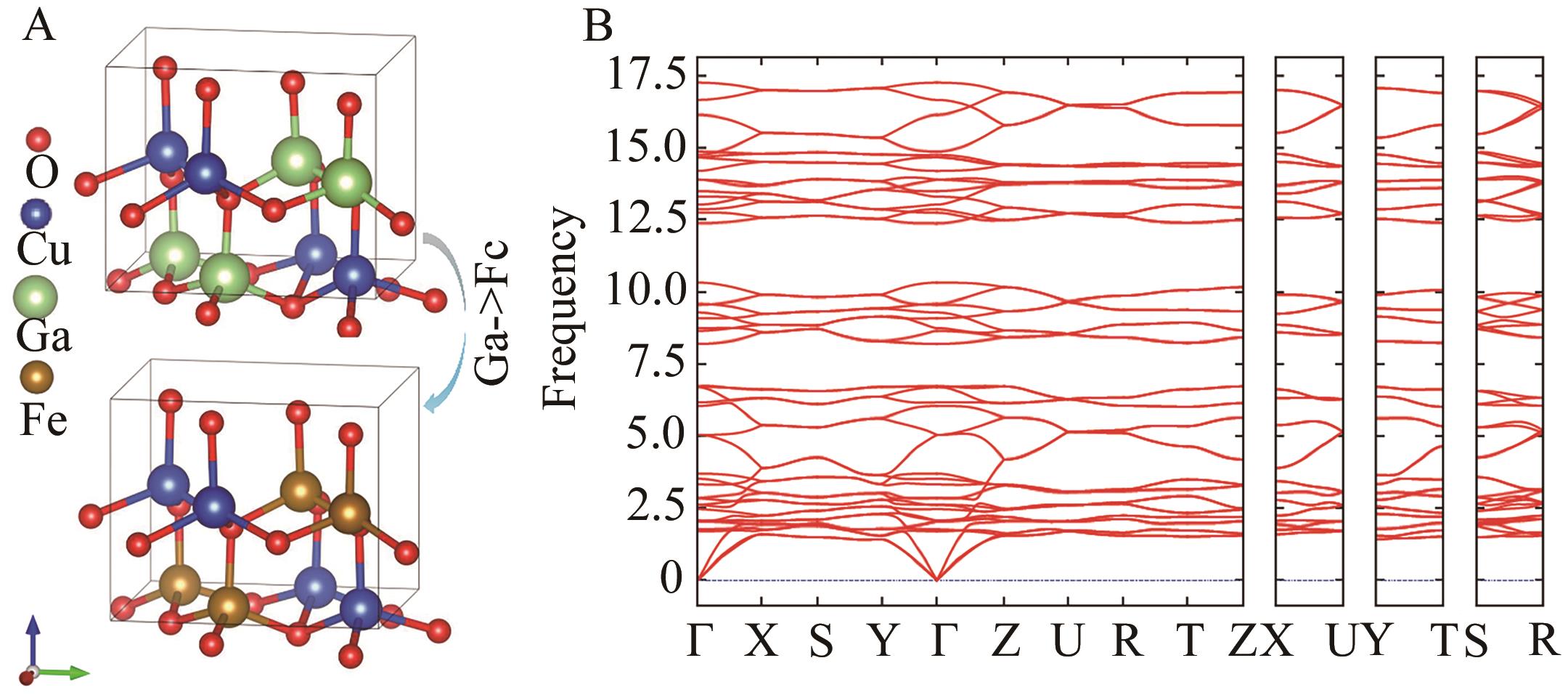
Chinese Journal of Applied Chemistry ›› 2024, Vol. 41 ›› Issue (7): 1010-1023.DOI: 10.19894/j.issn.1000-0518.240031
• Full Papers • Previous Articles Next Articles
The Properties of Narrow Bandgap β-CuFeO2 Ferroelectric Photocatalysts and Surface Oxygen Evolution Reaction Characteristics
Dong-Hao LYU1,2, Lan-Lan XU1, Xiao-Juan LIU1,2( )
)
- 1.State Key Laboratory of Rare Earth Resources Utilization,Changchun Institute of Applied Chemistry,Chinese Academy of Sciences,Changchun 130022,China
2.University of Science and Technology of China,Hefei 230026,China
-
Received:2024-01-30Accepted:2024-05-07Published:2024-07-01Online:2024-08-03 -
Contact:Xiao-Juan LIU -
About author:lxjuan@ciac.ac.cn
-
Supported by:the National Natural Science Foundation of China(U2130114);the Natural Science Foundation of Jilin Province(YDZJ202201ZYS378)
CLC Number:
Cite this article
Dong-Hao LYU, Lan-Lan XU, Xiao-Juan LIU. The Properties of Narrow Bandgap β-CuFeO2 Ferroelectric Photocatalysts and Surface Oxygen Evolution Reaction Characteristics[J]. Chinese Journal of Applied Chemistry, 2024, 41(7): 1010-1023.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.240031
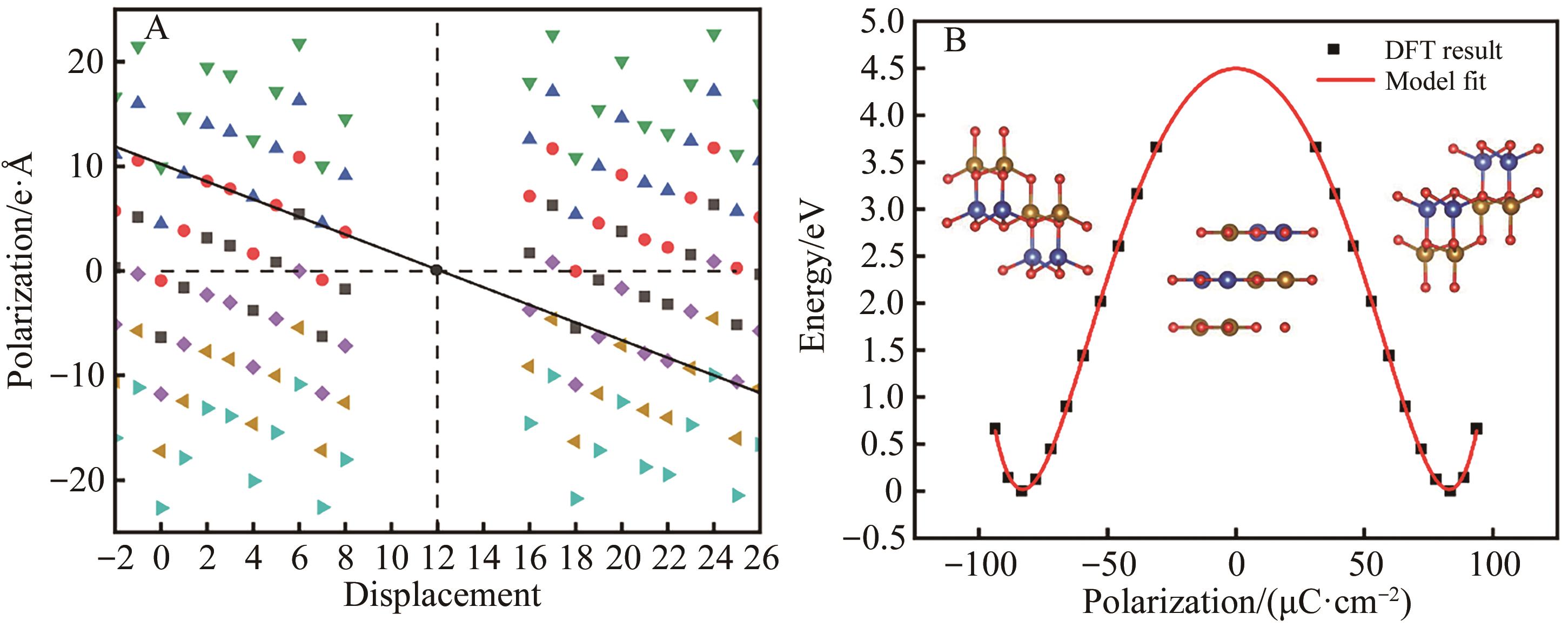
Fig.2 (A) The relationship between phase migration and polarization intensity of β-CuFeO2 and (B) the relationship between polarization and energy of β-CuFeO2
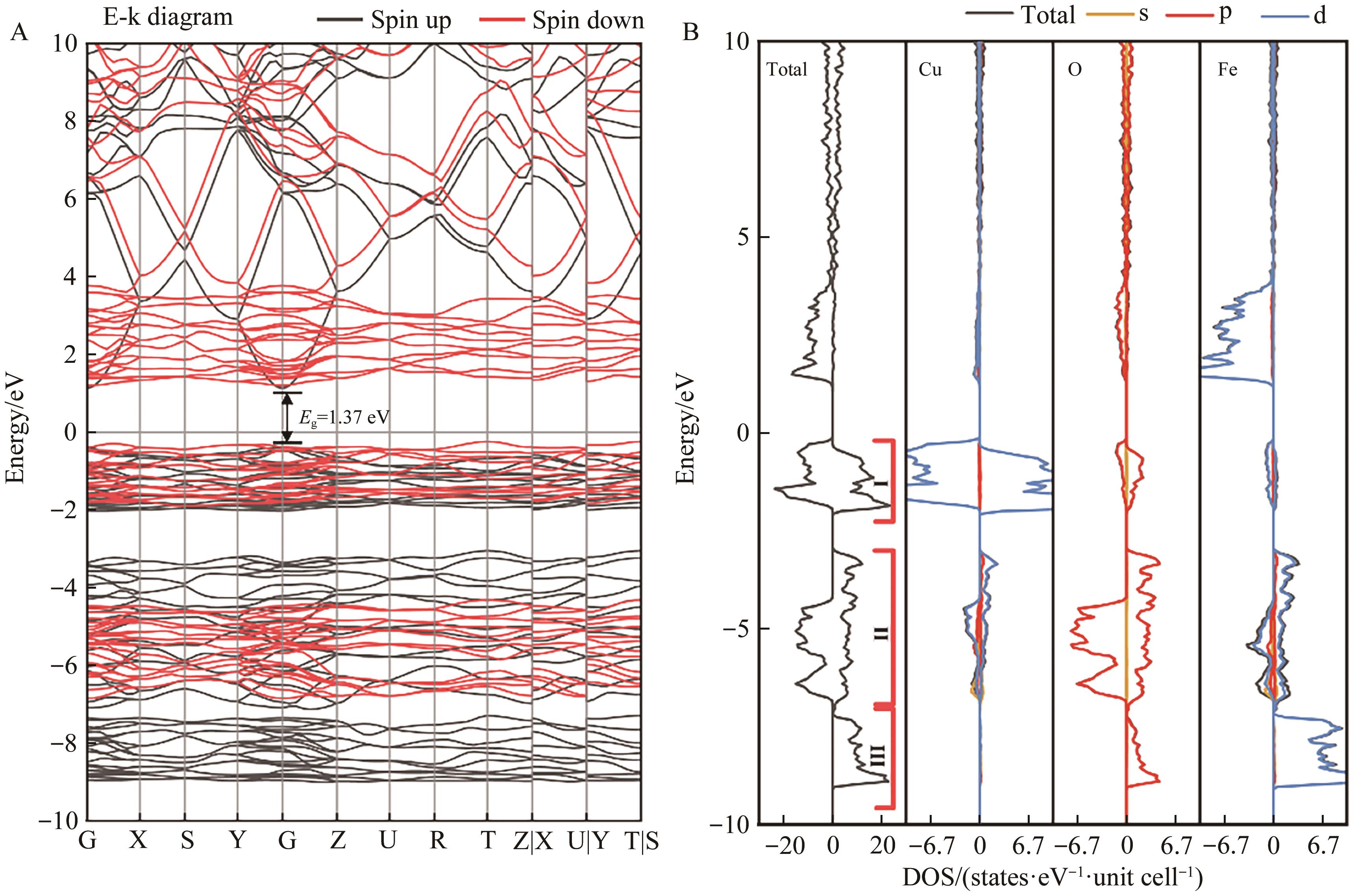
Fig.3 (A) Energy band structure of the bulk phase β-CuFeO2 and (B) total density of states of β-CuFeO2 and the partial wave state densityof each element
| Method | Gap/eV | VBM/eV | CBM/eV | |
|---|---|---|---|---|
| Hse06 | 1.37 | -0.242 | 1.125 | |
| GGA+U | UFe=5 eV, UCu=8 eV | 0.76 | -0.235 | 0.521 |
| UFe=6 eV, UCu=8 eV | 0.89 | -0.247 | 0.639 | |
| UFe=7 eV, UCu=8 eV | 0.92 | -0.247 | 0.668 | |
| UFe=8 eV, UCu=8 eV | 0.91 | -0.238 | 0.671 | |
| UFe=9 eV, UCu=8 eV | 0.90 | -0.223 | 0.675 | |
| UFe=7 eV, UCu=6 eV | 0.76 | -0.238 | 0.517 | |
| UFe=7 eV, UCu=7 eV | 0.83 | -0.221 | 0.614 | |
| UFe=7 eV, UCu=9 eV | 1.00 | -0.226 | 0.773 | |
Table 1 Band gap and band edge position under different U values of β-CuFeO2
| Method | Gap/eV | VBM/eV | CBM/eV | |
|---|---|---|---|---|
| Hse06 | 1.37 | -0.242 | 1.125 | |
| GGA+U | UFe=5 eV, UCu=8 eV | 0.76 | -0.235 | 0.521 |
| UFe=6 eV, UCu=8 eV | 0.89 | -0.247 | 0.639 | |
| UFe=7 eV, UCu=8 eV | 0.92 | -0.247 | 0.668 | |
| UFe=8 eV, UCu=8 eV | 0.91 | -0.238 | 0.671 | |
| UFe=9 eV, UCu=8 eV | 0.90 | -0.223 | 0.675 | |
| UFe=7 eV, UCu=6 eV | 0.76 | -0.238 | 0.517 | |
| UFe=7 eV, UCu=7 eV | 0.83 | -0.221 | 0.614 | |
| UFe=7 eV, UCu=9 eV | 1.00 | -0.226 | 0.773 | |
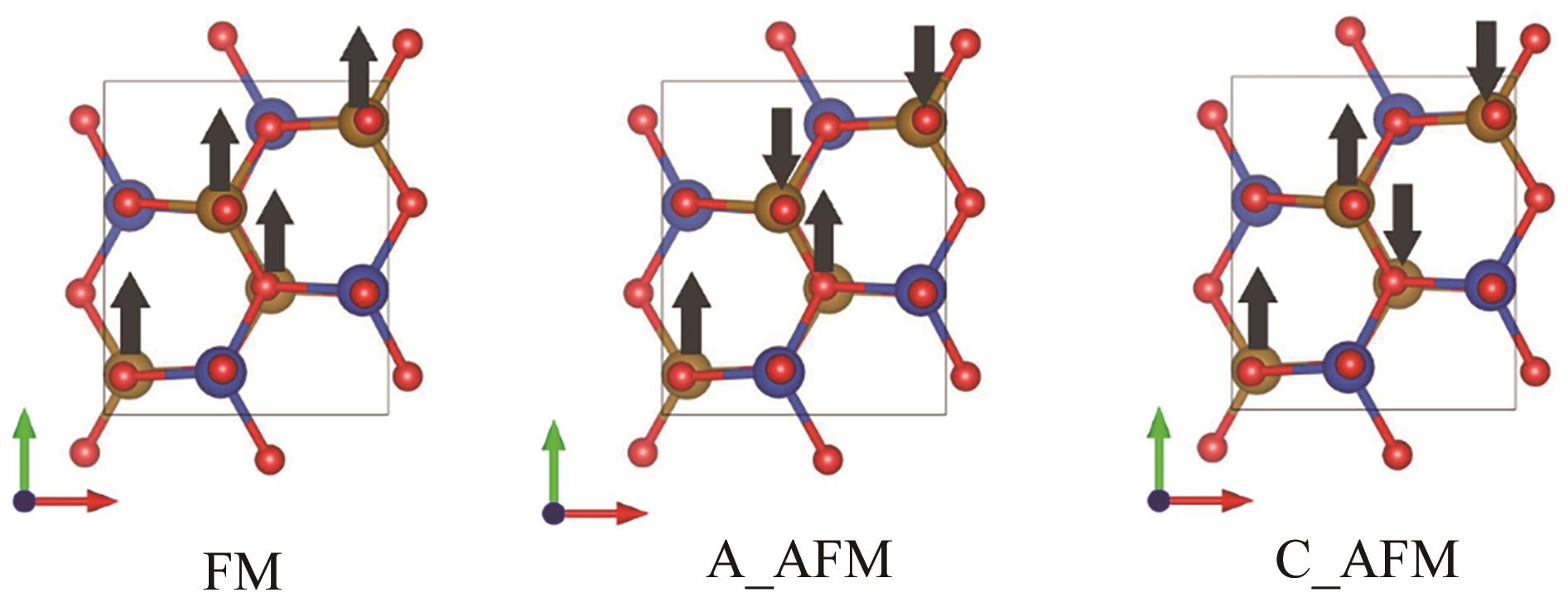
| Magnetic structure | Lattice parameters/? | Energy/eV | Mean magnetic moment (Fe)/μB |
|---|---|---|---|
| FM | a=5.376, b=6.245, c=5.384 | -107.446 | 3.74 |
| A_AFM | a=5.375, b=6.189, c=5.394 | -108.426 | 3.54 |
| C_AFM | a=5.353, b=6.164, c=5.380 | -109.375 | 3.40 |
Table 2 Structural parameters, energies, and magnetic moments in different magnetic configurations
| Magnetic structure | Lattice parameters/? | Energy/eV | Mean magnetic moment (Fe)/μB |
|---|---|---|---|
| FM | a=5.376, b=6.245, c=5.384 | -107.446 | 3.74 |
| A_AFM | a=5.375, b=6.189, c=5.394 | -108.426 | 3.54 |
| C_AFM | a=5.353, b=6.164, c=5.380 | -109.375 | 3.40 |
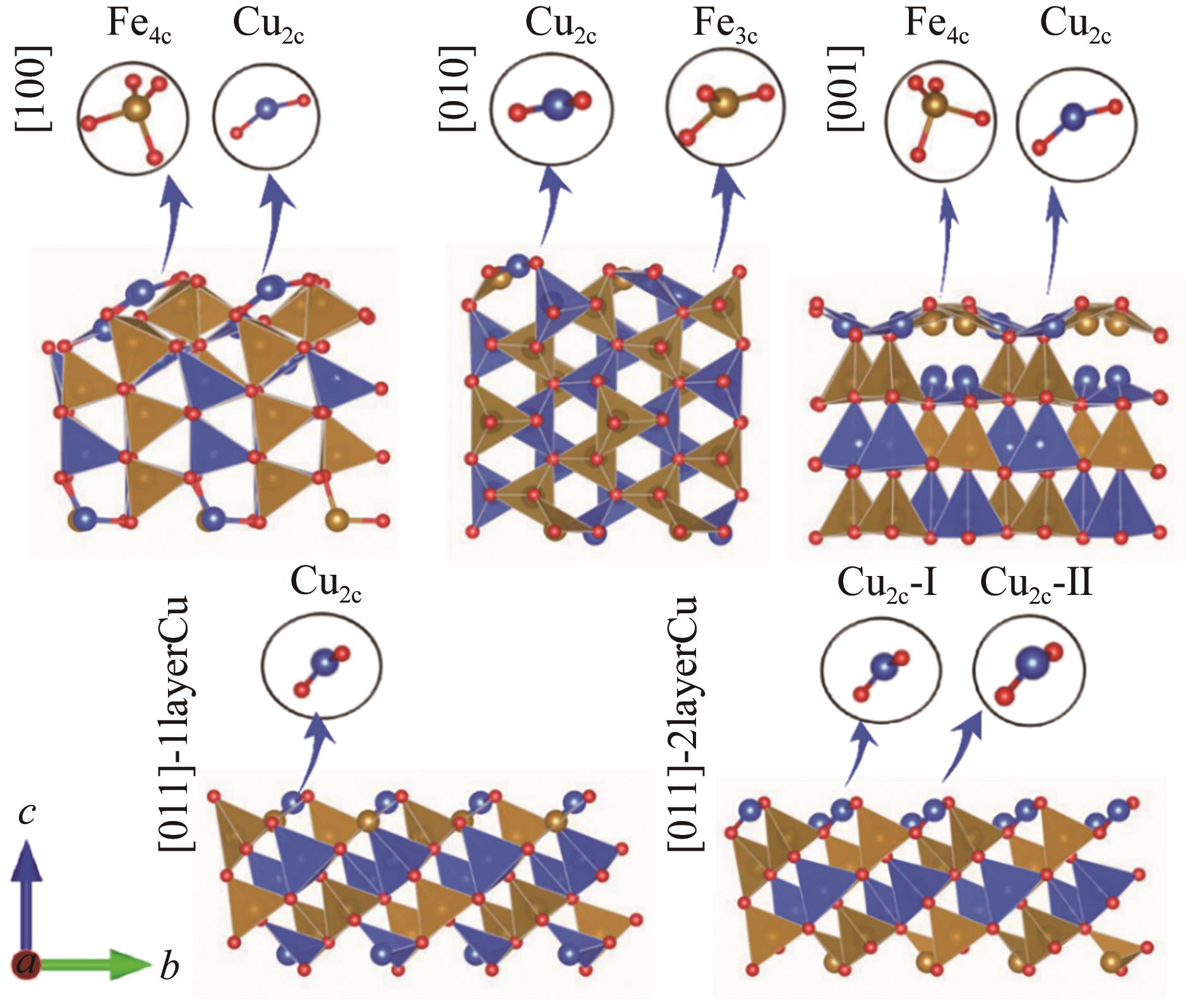
| Surfaces | Coordination (unrelax) | ||||
|---|---|---|---|---|---|
| Cu | Fe | ||||
| [100] | 5.19 | -1.24 | 3.95 | 2 | 2 |
| [010] | 4.89 | -1.52 | 3.37 | 3 | 3 |
| [001] | 4.92 | -2.71 | 2.20 | 3 | 3 |
| [011]-2layerCu | 3.84 | -0.94 | 2.90 | 3,2 | 4 |
| [011]-1layerCu | 3.84 | -0.86 | 2.97 | 2 | 3 |
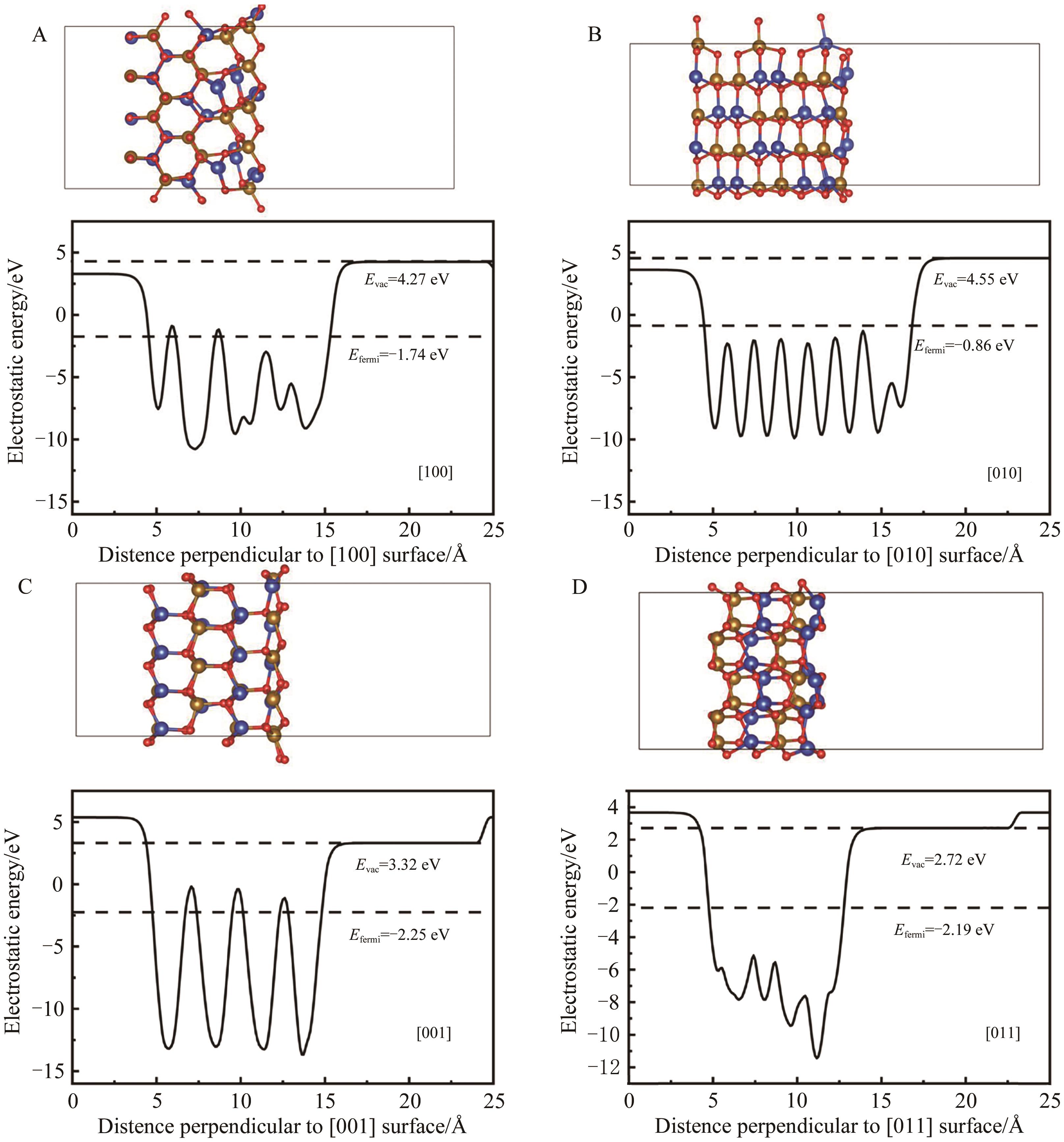
Table 3 Surface energy of differently oriented surfaces of β-CuFeO2 and the Cu, Fe atomic coordination number of the surface without structural optimization
| Surfaces | Coordination (unrelax) | ||||
|---|---|---|---|---|---|
| Cu | Fe | ||||
| [100] | 5.19 | -1.24 | 3.95 | 2 | 2 |
| [010] | 4.89 | -1.52 | 3.37 | 3 | 3 |
| [001] | 4.92 | -2.71 | 2.20 | 3 | 3 |
| [011]-2layerCu | 3.84 | -0.94 | 2.90 | 3,2 | 4 |
| [011]-1layerCu | 3.84 | -0.86 | 2.97 | 2 | 3 |
| Surface | Coordination | Surface | Coordination | ||
|---|---|---|---|---|---|
| [100] | 4Fe2c->4c | 0.722 | [010] | 4Fe3c->3c | -0.032 |
| 4Cu2c->2c | 0.112 | 2Cu3c->2c | -0.381 | ||
| 4O2c->2c | -0.126 8 | 2Cu3c->3c | 0.215 | ||
| 4O2c->3c | -0.192 | 8O3c->3c | -0.314 | ||
| [001] | 8Fe3c->3c | 0.005 | 8Cu2c->2c | 0.469 | |
| 8Cu3c->3c | 0.316 | [011]-2layerCu | 8O4c->4c | -0.221 | |
| 8O4c->4c | -0.264 | 8O3c->3c | -0.550 | ||
| 8O4c->3c | -1.219 | 8Cu2c->2c | 0.555 | ||
| [011]-1layerCu | 8O4c->4c | -1.300 | |||
| 8O3c->3c | -0.517 |
Table 4 Relaxation displacements of surface atoms on differently oriented surfaces of β-CuFeO2
| Surface | Coordination | Surface | Coordination | ||
|---|---|---|---|---|---|
| [100] | 4Fe2c->4c | 0.722 | [010] | 4Fe3c->3c | -0.032 |
| 4Cu2c->2c | 0.112 | 2Cu3c->2c | -0.381 | ||
| 4O2c->2c | -0.126 8 | 2Cu3c->3c | 0.215 | ||
| 4O2c->3c | -0.192 | 8O3c->3c | -0.314 | ||
| [001] | 8Fe3c->3c | 0.005 | 8Cu2c->2c | 0.469 | |
| 8Cu3c->3c | 0.316 | [011]-2layerCu | 8O4c->4c | -0.221 | |
| 8O4c->4c | -0.264 | 8O3c->3c | -0.550 | ||
| 8O4c->3c | -1.219 | 8Cu2c->2c | 0.555 | ||
| [011]-1layerCu | 8O4c->4c | -1.300 | |||
| 8O3c->3c | -0.517 |
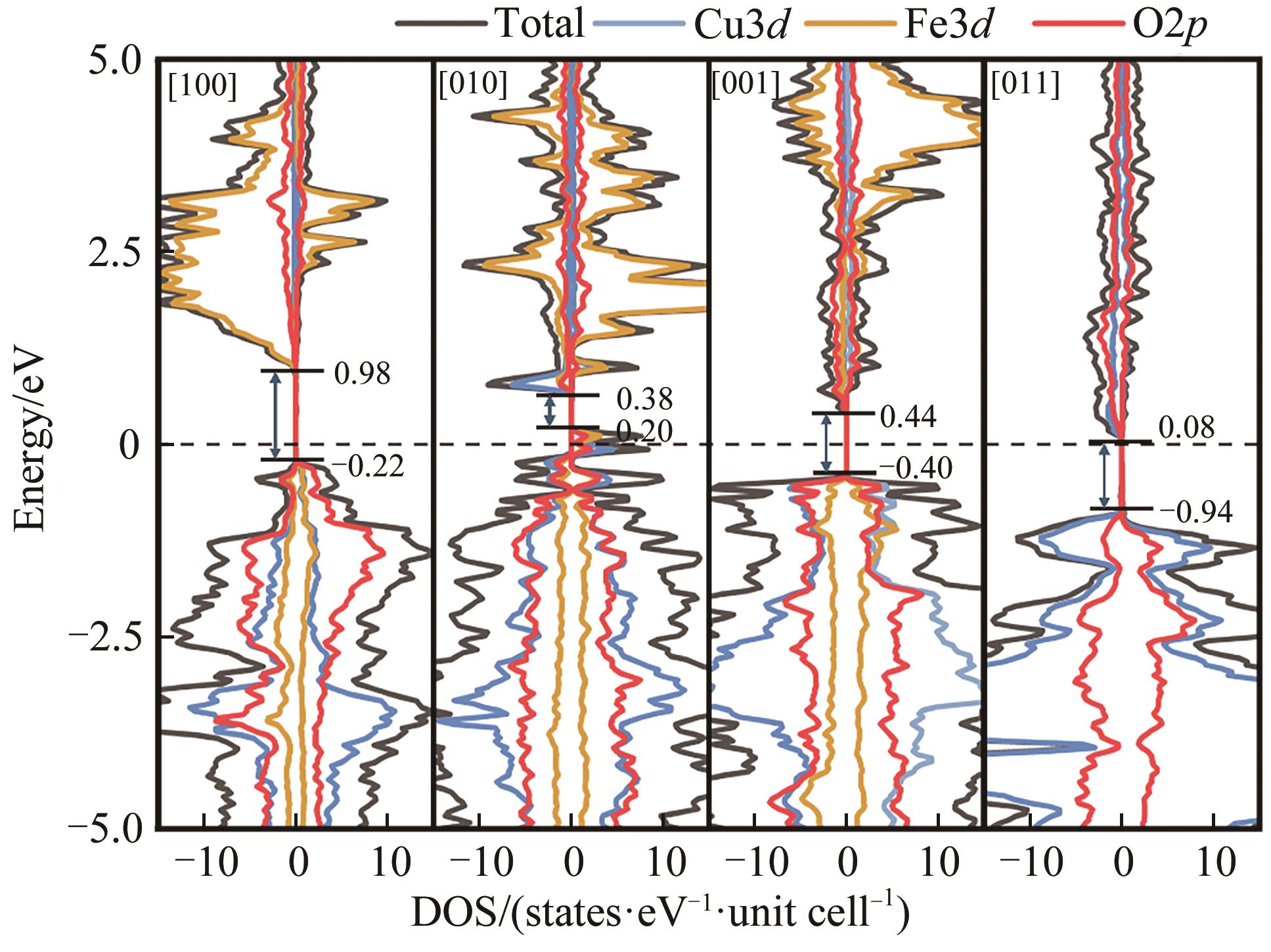
| Surface | lCu—O/? | lH—O/? | θ/(°) | |||
|---|---|---|---|---|---|---|
| [100] | 1.891 | 0.974 | 1.755 | 1.004 | 1.016 | 99.589 |
| [010] | 2.063 | -0.039 | 1.834 | 0.997 | 0.992 | 104.666 |
| [001] | 2.340 | 0.182 | 1.906 | 0.973 | 0.988 | 107.153 |
| [011] | 1.902 | 1.079 | 1.504 | 0.973 | 1.047 | 109.004 |
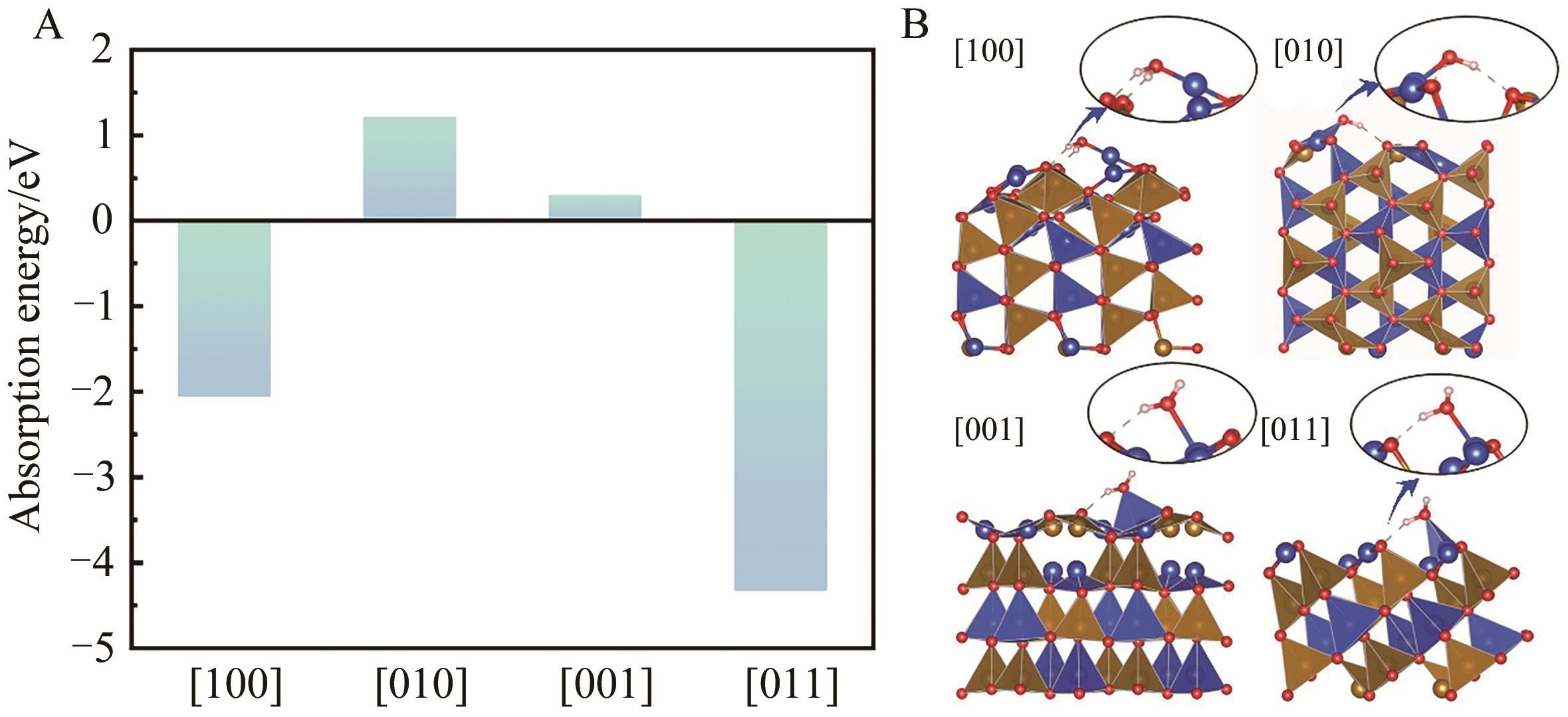
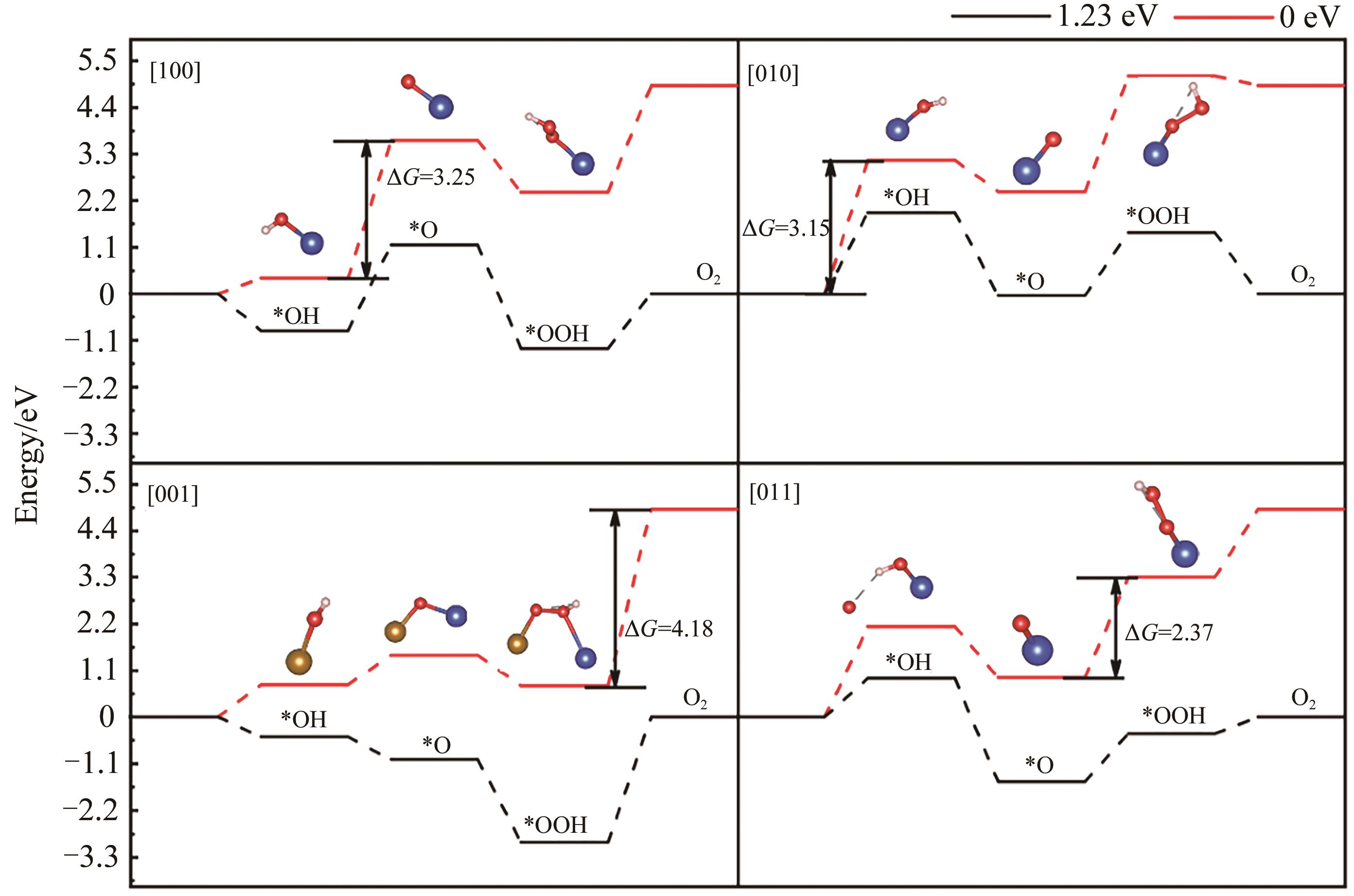
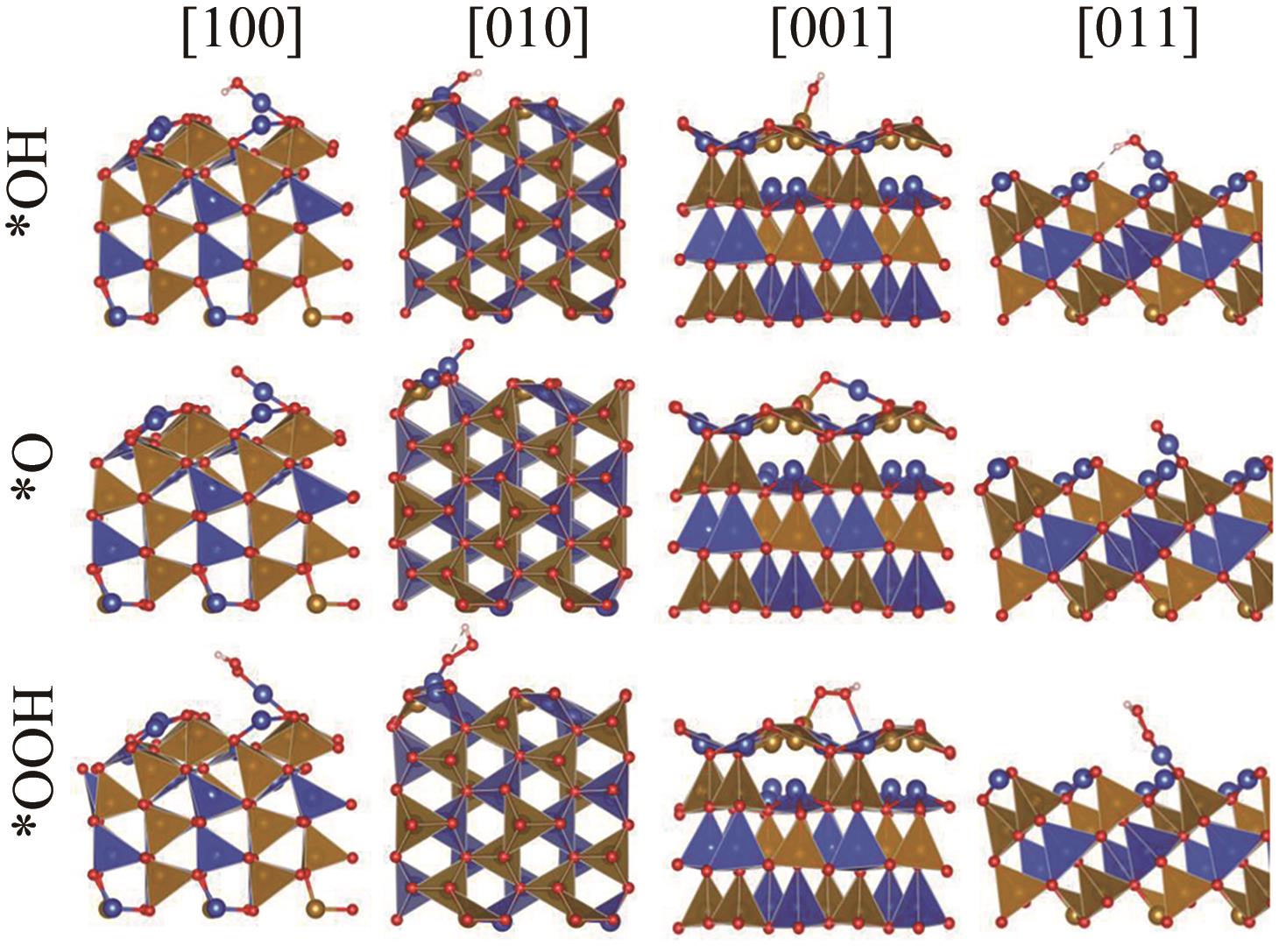
Table 5 Relaxation displacements of surface atoms on differently oriented surfaces of β-CuFeO2
| Surface | lCu—O/? | lH—O/? | θ/(°) | |||
|---|---|---|---|---|---|---|
| [100] | 1.891 | 0.974 | 1.755 | 1.004 | 1.016 | 99.589 |
| [010] | 2.063 | -0.039 | 1.834 | 0.997 | 0.992 | 104.666 |
| [001] | 2.340 | 0.182 | 1.906 | 0.973 | 0.988 | 107.153 |
| [011] | 1.902 | 1.079 | 1.504 | 0.973 | 1.047 | 109.004 |
| 1 | YAN S C, LI Z S, ZOU Z G. Photodegradation performance of g-C3N4 fabricated by directly heating melamine[J]. Langmuir, 2009, 25(17): 10397-10401. |
| 2 | HOUAS A, LACHHEB H, KSIBI M, et al. Photocatalytic degradation pathway of methylene blue in water[J]. Appl Catal B: Environ, 2001, 31(2): 145-157. |
| 3 | TURCHI C S, OLLIS D F. Photocatalytic degradation of organic-water contaminants-mechanisms involving hydroxyl radical attack[J]. J Catal, 1990, 122(1): 178-192. |
| 4 | ZOU Z G, YE J H, SAYAMA K, et al. Direct splitting of water under visible light irradiation with an oxide semiconductor photocatalyst[J]. Nature, 2001, 414(6864): 625-627. |
| 5 | XIANG Q, YU J, JARONIEC M. Synergetic effect of MoS2 and graphene as cocatalysts for enhanced photocatalytic H2 production activity of TiO2 nanoparticles[J]. J Am Chem Soc, 2012, 134(15): 6575-6578. |
| 6 | ZONG X, YAN H, WU G, et al. Enhancement of photocatalytic H2 evolution on CdS by loading MOS2 as cocatalyst under visible light irradiation[J]. J Am Chem Soc, 2008, 130(23): 7176-7177 |
| 7 | YAN S C, LI Z S, ZOU Z G. Photodegradation of rhodamine B and methyl orange over boron-doped g-C3N4 under visible light irradiation[J]. Langmuir, 2010, 26(6): 3894-3901. |
| 8 | PENG C, XU Z, LUO G, et al. Highly exposed single interlayered Cu edges enable high rate CO2 to CH4 electrosynthesis[J]. Adv Energy Mater, 2022, 12(15): 2200195.. |
| 9 | HAGFELDT A, GRAETZEL M. Light-induced redox reactions in nanocrystalline systems[J]. Chem Rev, 1995, 95(1): 49-68. |
| 10 | FANG Y, HOU Y, FU X, et al. Semiconducting polymers for oxygen evolution reaction under light illumination[J]. Chem Rev, 2022, 122(3): 4204-4256. |
| 11 | TEE S Y, WIN K Y, TEO W S, et al. Recent progress in energy-driven water splitting[J]. Adv Sci, 2017, 4(5): 1600337. |
| 12 | TAKANABE K. Photocatalytic water splitting: quantitative approaches toward photocatalyst by design[J]. ACS Catal, 2017, 7(11): 8006-8022. |
| 13 | WANG Q, DOMEN K. Particulate photocatalysts for light-driven water splitting: mechanisms, challenges, and design strategies[J]. Chem Rev, 2020, 120(2): 919-985. |
| 14 | LIN B, CHATURVEDI A, DI J, et al. Ferroelectric-field accelerated charge transfer in 2D CuInP2S6 heterostructure for enhanced photocatalytic H2 evolution[J]. Nano Energy, 2020, 76: 104972 . |
| 15 | HUANG H, TU S, DU X, et al. Ferroelectric spontaneous polarization steering charge carriers migration for promoting photocatalysis and molecular oxygen activation[J]. J Colloid Interface Sci, 2018, 509: 113-122. |
| 16 | HUANG X, LEI R, YUAN J, et al. Insight into the piezo-photo coupling effect of PbTiO3/CdS composites for piezo-photocatalytic hydrogen production[J]. Appl Catal B: Environ, 2021, 282: 119586. |
| 17 | CUI Y, BRISCOE J, DUNN S. Effect of ferroelectricity on solar-light-driven photocatalytic activity of BaTiO3-influence on the carrier separation and stern layer formation[J]. Chem Mat, 2013, 25(21): 4215-4223. |
| 18 | WAN G, YIN L, CHEN X, et al. Photocatalytic overall water splitting over PbTiO3 modulated by oxygen vacancy and ferroelectric polarization[J]. J Am Chem Soc, 2022, 144(44): 20342-20350. |
| 19 | WANG R, XU L, LIU Q, et al. Search for simple β-AIMIIIO2-type intrinsic ferroelectric semiconductors with simultaneous robust built-in electric field and full-spectrum absorption for superior photocatalysts[J]. J Mater Chem A, 2023, 11(10): 5233-5244. |
| 20 | YAO M C, WU X J, XU L L, et al. β-CuGaO2: a ferroelectric semiconductor with narrow band gap as degradation catalyst for wastewater environmental remediation[J]. Rare Met, 2021, 41(3): 972-981. |
| 21 | OUYANG S, LI Z, OUYANG Z, et al. Correlation of crystal structures, electronic structures, and photocatalytic properties in a series of Ag-based oxides: AgAlO2, AgCrO2, and Ag2CrO4[J]. J Phys Chem C, 2008, 112(8): 3134-3141. |
| 22 | WANG R, AN H, ZHANG H, et al. High active radicals induced from peroxymonosulfate by mixed crystal types of CuFeO2 as catalysts in the water[J]. Appl Surf Sci, 2019, 484: 1118-1127. |
| 23 | LIU Q L, ZHAO Z Y, ZHAO R D, et al. Fundamental properties of delafossite CuFeO2 as photocatalyst for solar energy conversion[J]. J Alloy Compd, 2020, 819: 153032. |
| 24 | JIANG T, XU C, ZHANG Y, et al. Wet chemical epitaxial growth of a cactus-like CuFeO2/ZnO heterojunction for improved photocatalysis[J]. Dalton Trans, 2020, 49(28): 9574-9578. |
| 25 | TU L W, CHANG K S. Hydrothermal fabrication and photocatalytic study of delafossite (CuFeO2) thin films on fluorine-doped tin oxide substrates[J]. Mater Chem Phys, 2021, 267: 124620. |
| 26 | WANG S, LIAO W, SU H, et al. Review on the application of semiconductor heterostructures in photocatalytic hydrogen evolution: state of the art and outlook[J]. Energy Fuels, 2023, 37(3): 1633-1656. |
| 27 | KRESSE G, FURTHMüLLER J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set[J]. Phys Rev B, 1996, 54(16): 11169-11186. |
| 28 | KRESSE G, FURTHMÜLLER J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set[J]. Comput Mater Sci, 1996, 6(1): 15-50. |
| 29 | PERDEW J P, BURKE K, ERNZERHOF M. Generalized gradient approximation made simple[J]. Phys Rev Lett, 1996, 77(18): 3865-3868. |
| 30 | AMRUTE A P, ŁODZIANA Z, MONDELLI C, et al. Solid-state chemistry of cuprous delafossites: synthesis and stability aspects[J]. Chem Mater, 2013, 25(21): 4423-4435. |
| 31 | TOGO A. First-principles phonon calculations with Phonopy and Phono3py[J]. J Phys Soc Jpn, 2022, 92(1): 012001. |
| 32 | KRUKAU A V, VYDROV O A, IZMAYLOV A F, et al. Influence of the exchange screening parameter on the performance of screened hybrid functionals[J]. J Chem Phys, 2006, 125(22): 224106. |
| 33 | WANG V, XU N, LIU J C, et al. VASPKIT: a user-friendly interface facilitating high-throughput computing and analysis using VASP code[J]. Comput Phys Commun, 2019, 267: 108033. |
| 34 | NøRSKOV J K, ROSSMEISL J, LOGADOTTIR A, et al. Origin of the overpotential for oxygen reduction at a fuel-cell cathode[J]. J Phys Chem B, 2004, 108(46): 17886-17892. |
| 35 | VALDÉS Á, QU Z W, KROES G J, et al. Oxidation and photo-oxidation of water on TiO2 surface[J]. J Phys Chem C, 2008, 112(26): 9872-9879. |
| 36 | OMATA T, NAGATANI H, SUZUKI I, et al. Wurtzite CuGaO2: a new direct and narrow band gap oxide semiconductor applicable as a solar cell absorber[J]. J Am Chem Soc, 2014, 136(9): 3378-3381. |
| 37 | HENKELMAN G, UBERUAGA B P, JÓNSSON H. A climbing image nudged elastic band method for finding saddle points and minimum energy paths[J]. J Chem Phys, 2000, 113(22): 9901-9904. |
| 38 | MA X Y, LYU H Y, HAO K R, et al. Large family of two-dimensional ferroelectric metals discovered via machine learning[J]. Sci Bull, 2021, 66(3): 233-242. |
| 39 | MAOUHOUBI A, OUZAROUAL L, TOUAL Y, et al. Structural, electronic and magnetic properties of the CuFeO2 multiferroic compound[J]. Arab J Chem, 2024, 17(1): 105437. |
| 40 | YOON S H, KANG U, PARK H, et al. Computational density functional theory study on the selective conversion of CO2 to formate on homogeneously and heterogeneously mixed CuFeO2 and CuO surfaces[J]. Catal Today, 2019, 335: 345-353. |
| 41 | RIBEIRO R A P, ANDRÉS J, LONGO E, et al. Magnetism and multiferroic properties at MnTiO3 surfaces: a DFT study[J]. Appl Surf Sci, 2018, 452: 463-472. |
| 42 | LI Q, RELLAN-PINEIRO M, ALMORA-BARRIOS N, et al. Shape control in concave metal nanoparticles by etching[J]. Nanoscale, 2017, 9(35): 13089-13094. |
| 43 | JU L, SHANG J, TANG X, et al. Tunable photocatalytic water splitting by the ferroelectric switch in a 2D AgBiP2Se6 monolayer[J]. J Am Chem Soc, 2020, 142(3): 1492-1500. |
| 44 | BERGERHOFF G, HUNDT R, SIEVERS R, et al. The inorganic crystal structure data base[J]. J Chem Inf Comput Sci, 1983, 23(2): 66-69. |
| [1] | Yin-Nan QIAN, Chuan SHI, Wei ZHANG, Zhao-Yan LUO. Research Progress of Noble Metal Electrocatalysts for Oxygen Evolution Reaction in Acidic Environment [J]. Chinese Journal of Applied Chemistry, 2023, 40(8): 1126-1139. |
| [2] | Zhi-Peng DU, Yang ZHOU, San-Gen ZHAO. Synthesis, Crystal Structure and Birefringence Properties of Silver Cluster Compound Ag3B6O10I [J]. Chinese Journal of Applied Chemistry, 2023, 40(2): 229-235. |
| [3] | Wei-Min DU, Xin LIU, Lin ZHU, Jia-Min FU, Wen-Shan GUO, Xiao-Qing YANG, Pei-Shuo SHUANG. Facile Synthesis and High⁃Efficiency Electrocatalytic Oxygen Evolution Performance of Ternary Nickel⁃Based Chalcogenide Nanorod Arrays [J]. Chinese Journal of Applied Chemistry, 2022, 39(8): 1252-1261. |
| [4] | Wei-Jin CAO, Lu BAI, Lan-Lan WU, Jing-De LI, Shu-Yan SONG. Multi⁃Shell Hollow Nickel⁃Cobalt Bimetallic Phosphide Nanospheres for Highly Efficient Oxygen Evolution Reaction [J]. Chinese Journal of Applied Chemistry, 2022, 39(4): 666-672. |
| [5] | Xue WANG, Yi-Bo WANG, Xian WANG, Jian-Bing ZHU, Jun-Jie GE, Chang-Peng LIU, Wei XING. Research Progress of Mechanism of Acidic Oxygen Evolution Reaction and Development of Ir⁃based Catalysts [J]. Chinese Journal of Applied Chemistry, 2022, 39(4): 616-628. |
| [6] | Dan WANG, Xian-Biao HOU, Xing-Kun WANG, Zhi-Cheng LIU, Huan-Lei WANG, Ming-Hua HUANG. Research Progress of Carbon‑Encapsulated Iron‑Based Nanoparticles Electrocatalysts for Zinc‑Air Batteries [J]. Chinese Journal of Applied Chemistry, 2022, 39(10): 1488-1500. |
| [7] | DA Zulin, ZHAO Yong, SHI Weidong. Facile Preparation of Bi4V2O11/Reduced Graphene Oxide Heterojunction Photocatalysts for the Degradation of Antibiotic Pollutants [J]. Chinese Journal of Applied Chemistry, 2018, 35(8): 946-955. |
| [8] | CHEN Si,SUN Lizhen,SHU Xinxin,ZHANG Jintao. Graphene-based Catalysts for Efficient Electrocatalytic Applications [J]. Chinese Journal of Applied Chemistry, 2018, 35(3): 272-285. |
| [9] | SONG Hongwei, HUANG Hui, TAN Ning, CHENG Buming, GUO Zhongcheng. Electrochemical Behavior of Al/Pb-0.2%Ag Anode in Fluoride Ion and Sulfuric Acid [J]. Chinese Journal of Applied Chemistry, 2016, 33(12): 1455-1461. |
| [10] | SUN Tingting1,2, LI Wenliang1, XIE Zhigang1*, JING Xiabin1. Preparation and Application of Heterogeneous Photocatalysts Based on New Cross-Linked Polymers [J]. Chinese Journal of Applied Chemistry, 2014, 31(05): 548-552. |
| [11] | WANG Lipin, WANG Senlin*, DUAN Qianhua. Ni/NiFe2O4 Composite Electrode Prepared by Electro-deposition and Its Electro-catalytic Performance Towards Oxygen Evolution Reaction [J]. Chinese Journal of Applied Chemistry, 2013, 30(06): 690-697. |
| [12] | ZHANG Yongchun1, CHEN Buming1, GUO Zhongcheng1,2*, LIU Jianhua1. Corrosion Resistance of Electrodeposited Al/Pb-Ag Anode During Zinc Electrowinning [J]. Chinese Journal of Applied Chemistry, 2013, 30(04): 458-463. |
| [13] | ZHANG Jin-Yan1, XIAO Xiao-Ming1*, JIANG Yan1, TAN Nian-Yuan2. Synthesis of Chromium (Ⅲ) Complex of 2,6-Di(2’-imidazoline-2’-yl) Pyridine and Its Interaction with DNA [J]. Chinese Journal of Applied Chemistry, 2010, 27(09): 1032-1037. |
| [14] | LI Lan-Lan, CHENG Fang-Yi, TAO Zhan-Liang, CHEN Jun*. Progress of First-principles Calculations on Hydrogen Storage Materials [J]. Chinese Journal of Applied Chemistry, 2010, 27(09): 998-1003. |
| [15] | WANG Feng-Wu, ZENG Xiu-Lin*, FANG Wen-Yan, LI Fen. Thermal Decomposition Characteristics of Nitrates [J]. Chinese Journal of Applied Chemistry, 2010, 27(03): 308-312. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||