Chinese Journal of Applied Chemistry ›› 2024, Vol. 41 ›› Issue (6): 878-889.DOI: 10.19894/j.issn.1000-0518.240021
• Full Papers • Previous Articles Next Articles
Structural Identification and Physicochemical Properties of Nonacetylated Sophorolipids Biosurfactant
Hao YANG1,2, Xiao-Meng MA1,2, Jin-Hua HUANG1,2, Jun-Feng LI3, Sheng-Kang LIANG1,2( )
)
- 1.College of Chemistry and Chemistry Engineering,Ocean University of China,Qingdao 266100,China
2.The Key Laboratory of Marine Chemistry Theory and Technology,Ministry of Education,Ocean University of China,Qingdao 266100,China
3.College of Marine Science and Biological Engineering,Qingdao University of Science and Technology,Qingdao 266045,China
-
Received:2024-01-22Accepted:2024-05-04Published:2024-06-01Online:2024-07-09 -
Contact:Sheng-Kang LIANG -
About author:liangsk@ouc.edu.cn
-
Supported by:the Shandong Province Major Science and Technology Innovation Engineering Project(2021CXGC010705);the Laoshan Laboratory's 14th Five Year Plan Major Project(2022QNLM040002)
CLC Number:
Cite this article
Hao YANG, Xiao-Meng MA, Jin-Hua HUANG, Jun-Feng LI, Sheng-Kang LIANG. Structural Identification and Physicochemical Properties of Nonacetylated Sophorolipids Biosurfactant[J]. Chinese Journal of Applied Chemistry, 2024, 41(6): 878-889.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.240021

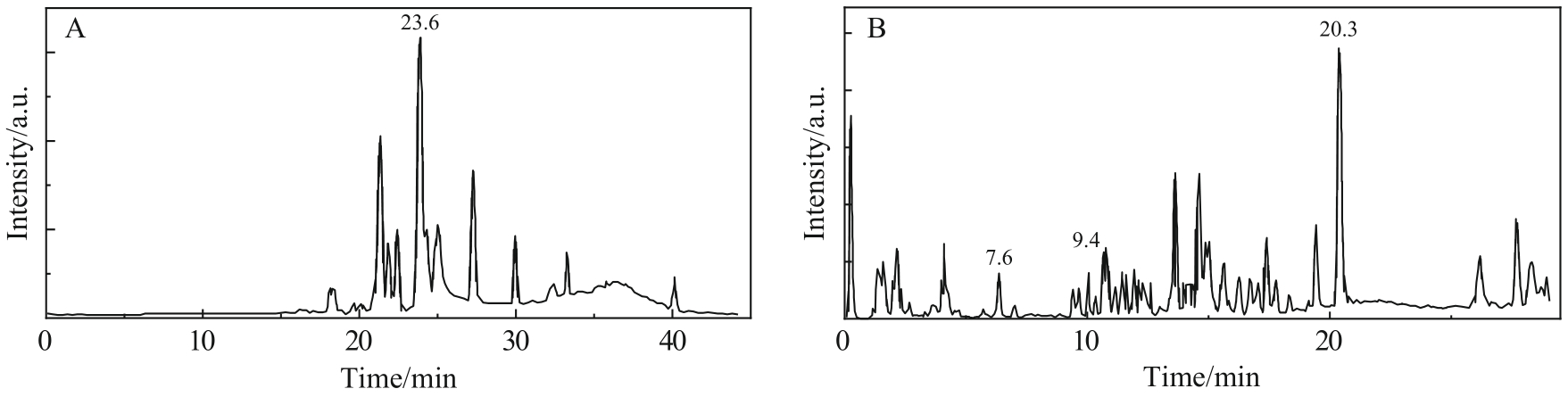
Fig.2 (A) The mass spectrum of components with acetylated SLs retention time of 23.6 min. (B) The mass spectrum of components with nonacetylated SLs retention time of 20.3 min. (C) The mass spectrum of components with nonacetylated SLs retention time of 9.4 min. (D) The mass spectrum of components with nonacetylated SLs retention time of 7.6 min
| Type | Molecular formula | m/z of [M+H]+ | Full name of component | w/% | |||
|---|---|---|---|---|---|---|---|
| Fatty acid groups | L/A/B* | Measured value | Simulation value | ||||
| Nonacetylated | C18∶2 | A | C30H52O13 | 621.342 1 | 621.340 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid | 0.32 |
| Monoacetylated | C16∶0 | L | C30H52O13 | 621.743 5 | 621.340 8 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanoic acid-1′,4"- lactone-6"-monoacetyl ester | 0.11 |
| C18∶0 | L | C32H56O13 | 649.369 7 | 649.372 2 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanoic acid-1′,4"- lactone-6"-monoacetyl ester | 0.18 | |
| C18∶1 | L | C32H54O13 | 647.358 1 | 647.356 5 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1′,4"-lactone-6"- monoacetyl ester | 5.29 | |
| C18∶2 | L | C32H52O13 | 645.341 9 | 645.340 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-1′,4"-lactone-6"-monoacetyl ester | 1.60 | |
| Diacetylated | C16∶0 | L | C32H54O14 | 663.360 4 | 663.351 4 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanoic acid-1′,4"- lactone-6′,6"-diacetyl ester | 12.27 |
| C16∶1 | L | C32H52O14 | 661.338 6 | 661.335 7 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecenoic acid-1′,4"-lactone-6',6"-diacetyl ester | 0.15 | |
| C18∶0 | L | C34H58O14 | 691.384 9 | 691.382 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanoic acid-1′,4"-lactone-6′,6"-diacetyl ester | 16.97 | |
| C18∶1 | L | C34H56O14 | 689.375 4 | 689.367 1 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1′,4"-lactone-6′,6"-diacetyl ester | 42.47 | |
| C18∶2 | L | C34H54O14 | 687.356 3 | 687.351 4 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-1′,4"-lactone-6',6"-diacetyl ester | 18.82 | |
| C16∶2 | A | C32H52O15 | 677.331 9 | 677.330 6 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanodienoic acid-6′,6″-diacetyl ester | 1.29 | |
| C18∶1 | A | C34H58O15 | 707.382 5 | 707.377 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-6′,6″-diacetylester | 0.19 | |
| C18∶2 | A | C34H56O15 | 705.351 6 | 705.362 0 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-6′,6″- diacetyl ester | 0.18 | |
| C22∶1 | A | C38H66O15 | 745.432 9 | 745.429 9 | 21-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]- docosenoic acid -6′,6″- diacetyl ester | 0.16 | |
Table 1 Composition and mass fraction of acetylated SLs
| Type | Molecular formula | m/z of [M+H]+ | Full name of component | w/% | |||
|---|---|---|---|---|---|---|---|
| Fatty acid groups | L/A/B* | Measured value | Simulation value | ||||
| Nonacetylated | C18∶2 | A | C30H52O13 | 621.342 1 | 621.340 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid | 0.32 |
| Monoacetylated | C16∶0 | L | C30H52O13 | 621.743 5 | 621.340 8 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanoic acid-1′,4"- lactone-6"-monoacetyl ester | 0.11 |
| C18∶0 | L | C32H56O13 | 649.369 7 | 649.372 2 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanoic acid-1′,4"- lactone-6"-monoacetyl ester | 0.18 | |
| C18∶1 | L | C32H54O13 | 647.358 1 | 647.356 5 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1′,4"-lactone-6"- monoacetyl ester | 5.29 | |
| C18∶2 | L | C32H52O13 | 645.341 9 | 645.340 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-1′,4"-lactone-6"-monoacetyl ester | 1.60 | |
| Diacetylated | C16∶0 | L | C32H54O14 | 663.360 4 | 663.351 4 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanoic acid-1′,4"- lactone-6′,6"-diacetyl ester | 12.27 |
| C16∶1 | L | C32H52O14 | 661.338 6 | 661.335 7 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecenoic acid-1′,4"-lactone-6',6"-diacetyl ester | 0.15 | |
| C18∶0 | L | C34H58O14 | 691.384 9 | 691.382 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanoic acid-1′,4"-lactone-6′,6"-diacetyl ester | 16.97 | |
| C18∶1 | L | C34H56O14 | 689.375 4 | 689.367 1 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1′,4"-lactone-6′,6"-diacetyl ester | 42.47 | |
| C18∶2 | L | C34H54O14 | 687.356 3 | 687.351 4 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-1′,4"-lactone-6',6"-diacetyl ester | 18.82 | |
| C16∶2 | A | C32H52O15 | 677.331 9 | 677.330 6 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanodienoic acid-6′,6″-diacetyl ester | 1.29 | |
| C18∶1 | A | C34H58O15 | 707.382 5 | 707.377 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-6′,6″-diacetylester | 0.19 | |
| C18∶2 | A | C34H56O15 | 705.351 6 | 705.362 0 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-6′,6″- diacetyl ester | 0.18 | |
| C22∶1 | A | C38H66O15 | 745.432 9 | 745.429 9 | 21-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]- docosenoic acid -6′,6″- diacetyl ester | 0.16 | |
| Type | Molecular formula | m/z of [M+H]+ | Full name of component | w/% | |||
|---|---|---|---|---|---|---|---|
| Fatty acid groups | L/A/B* | Measured value | Simulation value | ||||
| Nonacetylated | C16∶2 | L | C28H46O12 | 575.310 4 | 575.298 8 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-hexadecanodienoic acid-1′,4″-lactone | 8.73 |
| C18∶1 | L | C30H52O12 | 605.354 5 | 605.345 9 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-oleic acid-1′,4″-lactone | 16.50 | |
| C18∶3 | L | C30H48O12 | 637.365 2 | 637.335 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]- octadecanotrienoic acid-1′,4″-lactone | 1.76 | |
| C16∶0 | A | C28H52O13 | 597.509 0 | 597.340 8 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-hexadecenoic acid | 16.96 | |
| C16∶0 | A | C28H52O13 | 597.347 9 | 597.340 8 | 16-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-hexadecenoic acid | 1.74 | |
| C18∶0 | A | C30H56O13 | 625.386 3 | 625.372 2 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-octadecanoic acid | 6.37 | |
| C18∶1 | A | C30H54O13 | 623.364 2 | 623.356 5 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-oleic acid | 10.18 | |
| C18∶2 | A | C30H52O13 | 621.356 7 | 621.340 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-octadecanodienoic acid | 1.38 | |
| C20∶1 | A | C32H56O12 | 651.395 8 | 651.387 9 | 19-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-eicosenoic acid | 3.59 | |
| C24∶0 | A | C36H74O12 | 709.496 9 | 709.466 4 | 23-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-lignoceric acid | 1.49 | |
| C16∶0 | B | C40H72O23 | 921.450 2 | 921.446 4 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanoic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 6.60 | |
| C16∶1 | B | C40H70O23 | 901.435 5 | 919.430 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]- hexadecenoic acid -1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 10.16 | |
| C18∶1 | B | C42H74O23 | 947.469 3 | 947.462 1 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 2.74 | |
| C18∶2 | B | C42H72O23 | 945.485 5 | 945.446 4 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 2.36 | |
| Monoacetylated | C18∶1 | A | C32H56O14 | 669.365 0 | 665.367 1 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-6″-monoacetyl ester | 8.27 |
| C18∶1 | B | C44H76O23 | 989.478 5 | 989.472 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester-6″-monoacetyl ester | 0.36 | |
| C26∶0 | B | C52H94O23 | 1103.664 2 | 1103.614 0 | 25-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexacosanoic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester-6″-monoacetyl ester | 0.81 | |
Table 2 Composition and mass fraction of nonacetylated SLs
| Type | Molecular formula | m/z of [M+H]+ | Full name of component | w/% | |||
|---|---|---|---|---|---|---|---|
| Fatty acid groups | L/A/B* | Measured value | Simulation value | ||||
| Nonacetylated | C16∶2 | L | C28H46O12 | 575.310 4 | 575.298 8 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-hexadecanodienoic acid-1′,4″-lactone | 8.73 |
| C18∶1 | L | C30H52O12 | 605.354 5 | 605.345 9 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-oleic acid-1′,4″-lactone | 16.50 | |
| C18∶3 | L | C30H48O12 | 637.365 2 | 637.335 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]- octadecanotrienoic acid-1′,4″-lactone | 1.76 | |
| C16∶0 | A | C28H52O13 | 597.509 0 | 597.340 8 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-hexadecenoic acid | 16.96 | |
| C16∶0 | A | C28H52O13 | 597.347 9 | 597.340 8 | 16-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-hexadecenoic acid | 1.74 | |
| C18∶0 | A | C30H56O13 | 625.386 3 | 625.372 2 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-octadecanoic acid | 6.37 | |
| C18∶1 | A | C30H54O13 | 623.364 2 | 623.356 5 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-oleic acid | 10.18 | |
| C18∶2 | A | C30H52O13 | 621.356 7 | 621.340 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-octadecanodienoic acid | 1.38 | |
| C20∶1 | A | C32H56O12 | 651.395 8 | 651.387 9 | 19-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-eicosenoic acid | 3.59 | |
| C24∶0 | A | C36H74O12 | 709.496 9 | 709.466 4 | 23-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-lignoceric acid | 1.49 | |
| C16∶0 | B | C40H72O23 | 921.450 2 | 921.446 4 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanoic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 6.60 | |
| C16∶1 | B | C40H70O23 | 901.435 5 | 919.430 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]- hexadecenoic acid -1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 10.16 | |
| C18∶1 | B | C42H74O23 | 947.469 3 | 947.462 1 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 2.74 | |
| C18∶2 | B | C42H72O23 | 945.485 5 | 945.446 4 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 2.36 | |
| Monoacetylated | C18∶1 | A | C32H56O14 | 669.365 0 | 665.367 1 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-6″-monoacetyl ester | 8.27 |
| C18∶1 | B | C44H76O23 | 989.478 5 | 989.472 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester-6″-monoacetyl ester | 0.36 | |
| C26∶0 | B | C52H94O23 | 1103.664 2 | 1103.614 0 | 25-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexacosanoic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester-6″-monoacetyl ester | 0.81 | |
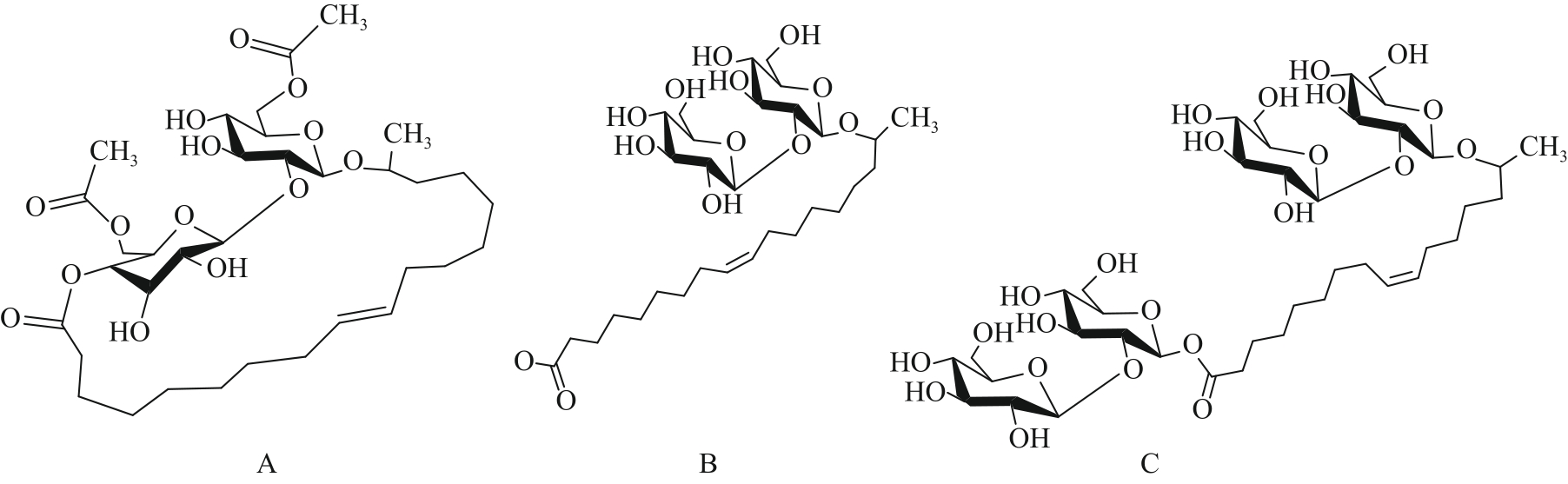
Fig.3 Three representative structural formulas of sophorolipids: (A) Diacetylated lactone type SLs; (B) Nonacetylated acid type SLs; (C) Nonacetylated bola type SLs
| Acetylated SLs | Nonacetylated SLs | |
|---|---|---|
| Solubility/(g·L-1) | 33.8 | 485.8 |
| Cloud point/℃ | 36 | Not detected |
| Acid resistance | Conditional acid resistance | Non acid resistance |
| Alkali resistance | Non alkali resistance | Non alkali resistance |
| Hard water resistance/grade | 5 | 5 |
Table 3 Solubility and acid and alkali resistance of acetylated SLs and nonacetylated SLs
| Acetylated SLs | Nonacetylated SLs | |
|---|---|---|
| Solubility/(g·L-1) | 33.8 | 485.8 |
| Cloud point/℃ | 36 | Not detected |
| Acid resistance | Conditional acid resistance | Non acid resistance |
| Alkali resistance | Non alkali resistance | Non alkali resistance |
| Hard water resistance/grade | 5 | 5 |
| Sophorolipid | CMC/(mg·L-1) | γCMC/(mN·m-1) | Гmax/(μmol·m-2) | Amin/nm2 |
|---|---|---|---|---|
| Acetylated-SLs | 30.3 | 36.2 | 2.55 | 0.653 |
| Nonacetylated SLs | 152.5 | 41.0 | 3.01 | 0.551 |
Table 4 The adsorption parameters of acetylated SLs and nonacetylated SLs
| Sophorolipid | CMC/(mg·L-1) | γCMC/(mN·m-1) | Гmax/(μmol·m-2) | Amin/nm2 |
|---|---|---|---|---|
| Acetylated-SLs | 30.3 | 36.2 | 2.55 | 0.653 |
| Nonacetylated SLs | 152.5 | 41.0 | 3.01 | 0.551 |

Fig.6 The foam performance of acetylated SLs and nonacetylated SLs (A) Foam volume of 30 s and 5 min in pure water, (B) foam properties at different salinities
| 1 | PAL S, CHATTERJEE N, DAS A K, et al. Sophorolipids: a comprehensive review on properties and applications[J]. Adv Colloid Interface Sci, 2023, 313: 102856. |
| 2 | MADANKAR C S, BORDE P K. Review on sophorolipids-a promising microbial bio-surfactant[J]. Tenside Surfact Det, 2023, 60(2): 95-105. |
| 3 | CHO W Y, NG J F, YAP W H, et al. Bio-based antimicrobial formulating agents for applications in food and health[J]. Molecules, 2022, 27(17): 5556. |
| 4 | DEVELTER D W G, LAURYSSEN L M L. Properties and industrial applications of sophorolipids[J]. Eur J Lipid Sci Technol, 2010, 112(6): 628-638. |
| 5 | ASMER H J, LANG S, WAGNER F, et al. Microbial production, structure elucidation and bioconversion of sophorose lipids[J]. J Am Oil Chem Soc, 1988, 65(9): 1460-1466. |
| 6 | DENGLE PULATE V, BHAGWAT S, PRABHUNE A. Microbial oxidation of medium chain fatty alcohol in the synthesis of sophorolipids by candida bombicola and its physicochemical characterization[J]. J Surfactants Deterg, 2013, 16(2): 173-181. |
| 7 | KONISHI M, FUKUOKA T, MORITA T, et al. Production of new types of sophorolipids by by candida batistae[J]. J Oleo Sci, 2008, 57(6): 359-369. |
| 8 | VAN BOGAERT, SAERENS K, DE MUYNCK C, et al. Microbial production and application of sophorolipids[J]. Appl Microbiol Biot, 2007, 76(1): 23-34. |
| 9 | SEN A R, KARIM S A, SANTRA A. Effect of defaunation on carcass and meat characteristics of finisher lambs[J]. Indian J Anim Sci, 2000, 70(6): 659-661. |
| 10 | OTTO R T, DANIEL H J, PEKIN G, et al. Production of sophorolipids from whey[J]. Appl Microbiol Biot, 1999, 52(4): 495-501. |
| 11 | LI S, QIAN X, XU L, et al. Biological tailoring of novel sophorolipid molecules and their derivatives[J]. Biofuel Bioprod Bior, 2021, 15(6): 1938-1949. |
| 12 | SHAO L, SONG X, MA X,et al. Bioactivities of sophorolipid with different structures against human esophageal cancer cells[J]. J Surg Res, 2012, 173(2): 286-291. |
| 13 | RAU U, HECKMANN R, WRAY V, et al. Enzymatic conversion of a sophorolipid into a glucose lipid[J]. Biotechnol Lett, 1999, 21(11): 973-977. |
| 14 | BORSANYIOVA M, PATIL A, MUKHERJI R, et al. Biological activity of sophorolipids and their possible use as antiviral agents[J]. Folia Microbiol, 2016, 61(1): 85-89. |
| 15 | KASTURE M, SINGH S, PATEL P, et al. Multiutility sophorolipids as nanoparticle capping agents: synthesis of stable and water dispersible Co nanoparticles[J]. Langmuir, 2007, 23(23): 11409-11412. |
| 16 | LYDON H L, BACCILE N, CALLAGHAN B, et al. Adjuvant antibiotic activity of acidic sophorolipids with potential for facilitating wound healing[J]. Antimicrob Agents Chemother, 2017, 61(5): E02547. |
| 17 | SAERENS K M J, SAEY L, SOETAERT W. One-step production of unacetylated sophorolipids by an acetyltransferase negative Candida bombicola[J]. Biotechnol Bioeng, 2011, 108(12): 2923-2931. |
| 18 | VAN B, BUYST D, MARTINS J C, et al. Synthesis of bolaform biosurfactants by an engineered starmerella bombicola yeast[J]. Biotechnol Bioeng, 2016, 113(12): 2644-2651. |
| 19 | 马孝萌. 去乙酰化槐糖脂基因工程菌株的构建及发酵条件优化[D]. 青岛: 中国海洋大学, 2023. |
| MA X M. Construction of genetically engineered strains producing deacetylated sophorolipids and its optimization of fermentation conditions[D]. Qingdao: Ocean University of China, 2023. | |
| 20 | 宋丹丹, 梁生康, 王江涛. 槐糖脂生物表面活性剂的结构特征及理化性质初探[J]. 环境化学, 2011, 30(8): 1474-1479. |
| SONG D D, LIANG S K, WANG J T. Structural characteristics and physical-chemical properties of sophorolipid biosurfactants[J]. Environ Chem, 2011, 30(8): 1474-1479. | |
| 21 | 孙旭璐, 陈慧, 郑伟. 改性大豆磷脂的HLB值检测方法优化研究[J]. 粮食与油脂, 2015, 28(4): 47-48. |
| SUN X L, CHEN H, ZHENG W. Study on the optimal method for detecting of modified soybean phospholipids HLB value[J]. Cereals Oils, 2015, 28(4): 47-48. | |
| 22 | 周家华, 崔英德. 表面活性剂HLB值的分析测定与计算Ⅰ∶ HLB值的分析测定[J]. 精细石油化工, 2001(2): 11-14. |
| ZHOU J H, CUI Y D. Measurement and calculation of HLB value of surfactants: the measurement of HLB value[J]. Spec Petrochem, 2001(2): 11-14. | |
| 23 | DAVID J T, ROBERT L R. Commercial synthesis of monoalkly phosphates[J]. J Surfactants Deterg, 2002, 5(2): 169-172. |
| 24 | 李双阳, 王佳颖, 郭高强, 等. BOLA型表面活性剂的研究进展[J]. 广州化工, 2015, 43(5): 18-20. |
| LI S Y, WANG J Y, GUO G Q, et al. Progress in research work with respect to bolaform surfactants[J]. Guangzhou Chem Ind, 2015, 43(5): 18-20. | |
| 25 | 周家华, 崔英德, 吴雅红. 表面活性剂HLB值的分析测定与计算 Ⅱ.HLB值的计算[J]. 精细石油化工, 2001(4): 38-41. |
| ZHOU J H, CUI Y D, WU Y H. Measurement and calculation of HLB value of surfactants: the calculation of HLB value[J]. Spec Petrochem, 2001(4): 38-41. | |
| 26 | KRAYNIK A M. The structure of random foam[J]. Adv Eng Mater, 2006, 8(9): 900-906. |
| 27 | KUMAR V, PAL N, JANGIR A K, et al. Dynamic interfacial properties and tuning aqueous foamability stabilized by cationic surfactants in terms of their structural hydrophobicity, free drainage and bubble extent[J]. Colloid Surface A, 2020, 588: 124362. |
| 28 | WANG C, FANG H, GONG Q,et al. Roles of catanionic surfactant mixtures on the stability of foams in the presence of oil[J]. Energ Fuel, 2016, 30(8): 6355-6364. |
| 29 | 赵修太, 王泉, 王增宝, 等. 高温高盐油藏常用起泡剂研究进展[J]. 材料导报, 2016, 30(5): 75-80. |
| ZHAO X T, WANG Q, WANG Z B, et al. Commonly used foaming agents in high temperature and high salinity reservoir[J]. Mater Rep, 2016, 30(5): 75-80. | |
| 30 | SUN L, SUN X H, ZHANG Y C, et al. Stability of high-salinity-enhanced foam: surface behavior and thin-film drainage[J]. Petrol Sci, 2023, 20(4): 2343-2353. |
| 31 | 刘晓敏. 驱油用泡沫体系研究[D]. 无锡: 江南大学, 2023. |
| LIU X M. Study on foam systems for enhanced oil recovery[D]. Wuxi: Jiangnan University, 2023. | |
| 32 | 胡贝贝, 袁悦, 周小平, 等. 基于丝氨酸的Bola型表面活性剂的合成及理化性质[J]. 沈阳药科大学学报, 2016, 33(6): 419-425. |
| HU B B, YUAN Y, ZHOU X P, et al. Synthesis of a novel Bola amphiphile based on serine and investigation of its physicochemical properties[J]. J Shenyang Pharm Univ, 2016, 33(6): 419-425. | |
| 33 | VAN RENTERGHEM L, ROELANTS S, BACCILE N, et al. From lab to market: an integrated bioprocess design approach for new-to-nature biosurfactants produced by Starmerella bombicola[J]. Biotechnol Bioeng, 2018, 115(5): 1195-1206. |
| [1] | Chen-Si-Fan JIAO, Shao‑Bo ZHENG, Peng-Jun XU, Wei WANG, Takebe HIROMICHI, Mukai KUSUHIRO, Zhong-Da YU. Method of Evaluating the Foaming Properties of Surfactant Solutions— Real Sphere Bubble Method [J]. Chinese Journal of Applied Chemistry, 2022, 39(7): 1108-1118. |
| [2] | WANG Chun-Yu, ZHAO Hui, DAI Zheng-Hua, LI Wei-Feng, LIU Hai-Feng. Effect of Surfactant on the Rheological Properties of Hydrophilic Particle Suspension [J]. Chinese Journal of Applied Chemistry, 2021, 38(4): 398-406. |
| [3] | WEN Fushan, KONG Xiangshuang, WANG Wei, HU Songqing, XIA Zheng, ZHANG Jun*. Synthesis and Surface Properties of a Novel Cationic Trisiloxane Surfacant [J]. Chinese Journal of Applied Chemistry, 2014, 31(10): 1216-1221. |
| [4] | LI Haichao2, LIU Jinyan*, ZHANG Yingchun. Interaction of Sodium Dodecyl Benzene Sulfonate and TritonX-100 Studied by Intrinsic Fluorescence [J]. Chinese Journal of Applied Chemistry, 2011, 28(01): 113-117. |
| [5] |
ZHANG Jian, QIU Yu, YU Dao-Yong*.
Critical Micelle Concentration Determination of Sodium Dodecyl Benzene Sulfonate by Synchronous Fluorescence Spectrometry [J]. Chinese Journal of Applied Chemistry, 2009, 26(12): 1480-1483. |
| [6] | CHEN Hai-Min, SHI Hong-Xin, XIANG Ju-Ping, LIU Qiu-Ping, ZHU Chun-Feng. Synthesis and Surface Activity of Fluorocarbon Surfactant Containing Sulfuric ester [J]. Chinese Journal of Applied Chemistry, 2009, 26(09): 1036-1039. |
| [7] | HU Ying-Mo. Synthesis of a series of Novel Cationic Fluoro-Surfactants [J]. Chinese Journal of Applied Chemistry, 2009, 26(09): 1104-1107. |
| [8] | Liu Daojun, Ma Jiming, Cheng Humin, Zhao Zhenguo. Aggregation Behaviour of Mixed Surfactants in Apolar Solvent [J]. Chinese Journal of Applied Chemistry, 1997, 0(6): 51-53. |
| [9] | Wu Jinchuan, Zhuang Yan, He Zhimin, Tu Yu'en. Surface Active Properties of Disodium Polyoxyalkylene Fatty Acid Ethanolamide Sulfosuccinates [J]. Chinese Journal of Applied Chemistry, 1997, 0(6): 93-95. |
| [10] | Zhang Congyun, Fang Tianru, Qiu Lingwei, Wang Fusong, Liu Peiyan. Water-Soluble and Amphiphilic Polymers 18. Amphiphilic Monomers of 2-Acrylamidoalkanesulfonic Acids [J]. Chinese Journal of Applied Chemistry, 1996, 0(1): 1-5. |
| [11] | Du Shangjian, Lu Yan, Yu Shuyuan, Wang Tao, Di Pingping, Cui Wenge. UV Specttophotometric Determination of the Degree of Deacetylation of Chitin [J]. Chinese Journal of Applied Chemistry, 1994, 0(2): 108-109. |
| [12] | Wangwei, Bo Shuqin, Qin Wen. THE SPECIFIC REFRACTIVE INDEX INCREMENT OF CHITOSAN [J]. Chinese Journal of Applied Chemistry, 1991, 0(2): 56-59. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||