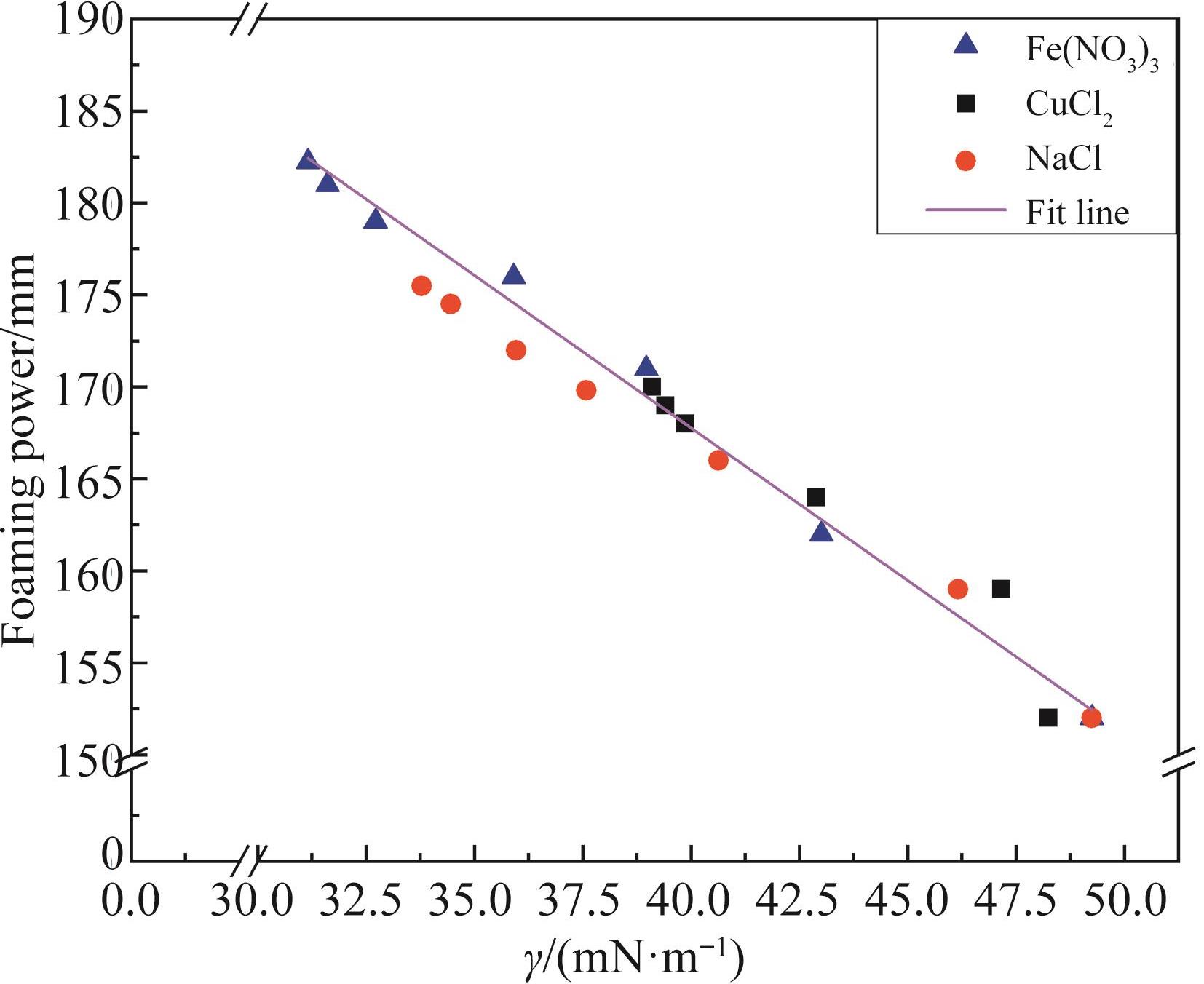
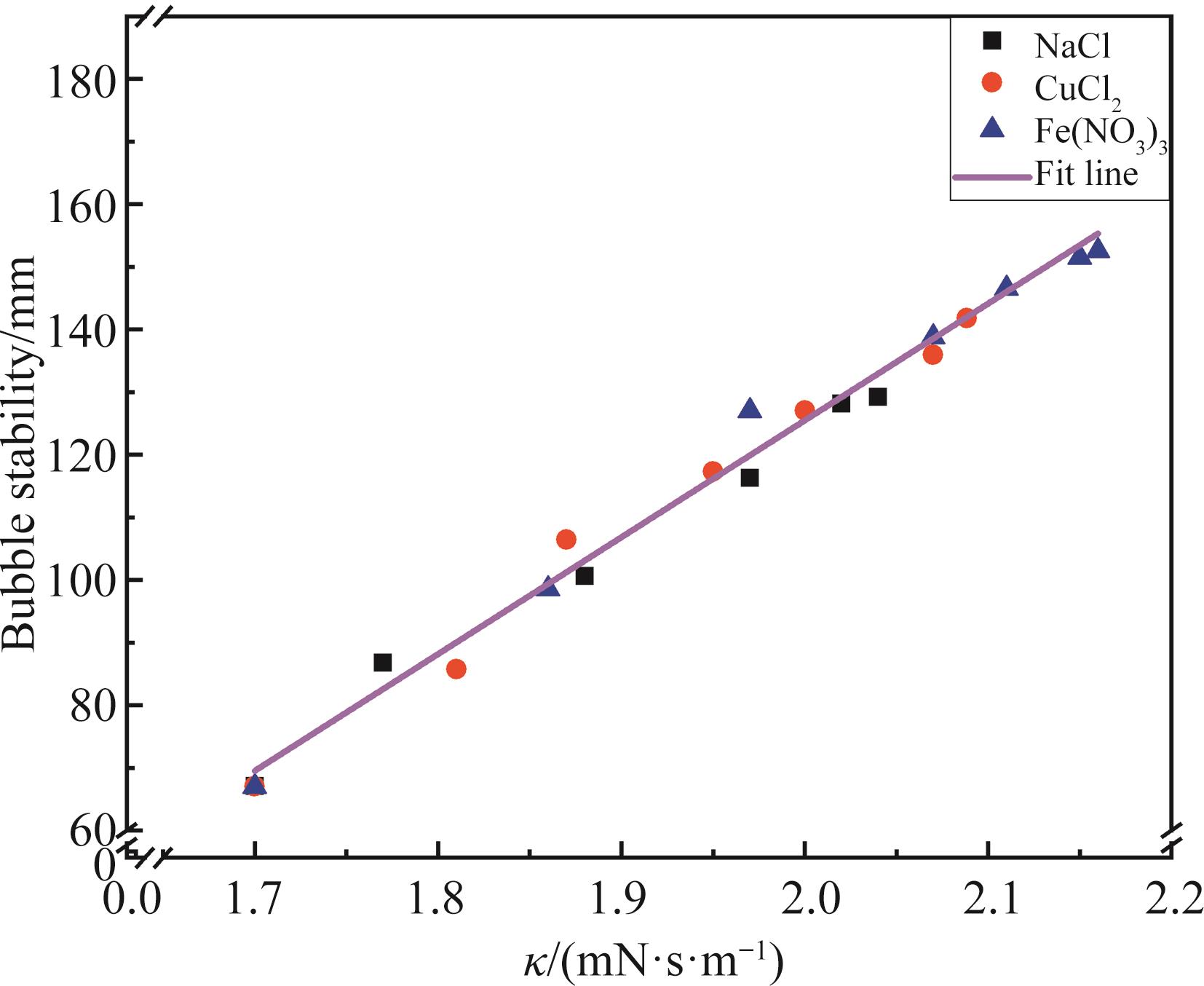
Chinese Journal of Applied Chemistry ›› 2022, Vol. 39 ›› Issue (7): 1108-1118.DOI: 10.19894/j.issn.1000-0518.210289
• Full Papers • Previous Articles Next Articles
Method of Evaluating the Foaming Properties of Surfactant Solutions— Real Sphere Bubble Method
Chen-Si-Fan JIAO1, Shao‑Bo ZHENG1, Peng-Jun XU2, Wei WANG2, Takebe HIROMICHI3, Mukai KUSUHIRO4, Zhong-Da YU1( )
)
- 1.Department of Material Science and Engineering,Shanghai University,State Key Lab of Metallurgy and Preparation of High Quality Special Steel,Shanghai 200072,China
2.Key Laboratory for Ferrous Metallurgy and Resources Utilization of Ministry of Education,Wuhan University of Science and Technology,Wuhan 430081,China
3.Ehime University,Matsuyama 9008267,Japan
4.Kyushu Institute of Technology,Kyushu 8048550,Japan
-
Received:2021-06-15Accepted:2021-12-16Published:2022-07-01Online:2022-07-11 -
Contact:Zhong-Da YU -
About author:yuzhongda66@163.com
-
Supported by:the National Natural Science Foundation of China(51974212);the National Natural Science Foundation of China?China Baowu Steel Group Steel Joint Research Fundation(U1760101)
CLC Number:
Cite this article
Chen-Si-Fan JIAO, Shao‑Bo ZHENG, Peng-Jun XU, Wei WANG, Takebe HIROMICHI, Mukai KUSUHIRO, Zhong-Da YU. Method of Evaluating the Foaming Properties of Surfactant Solutions— Real Sphere Bubble Method[J]. Chinese Journal of Applied Chemistry, 2022, 39(7): 1108-1118.
share this article
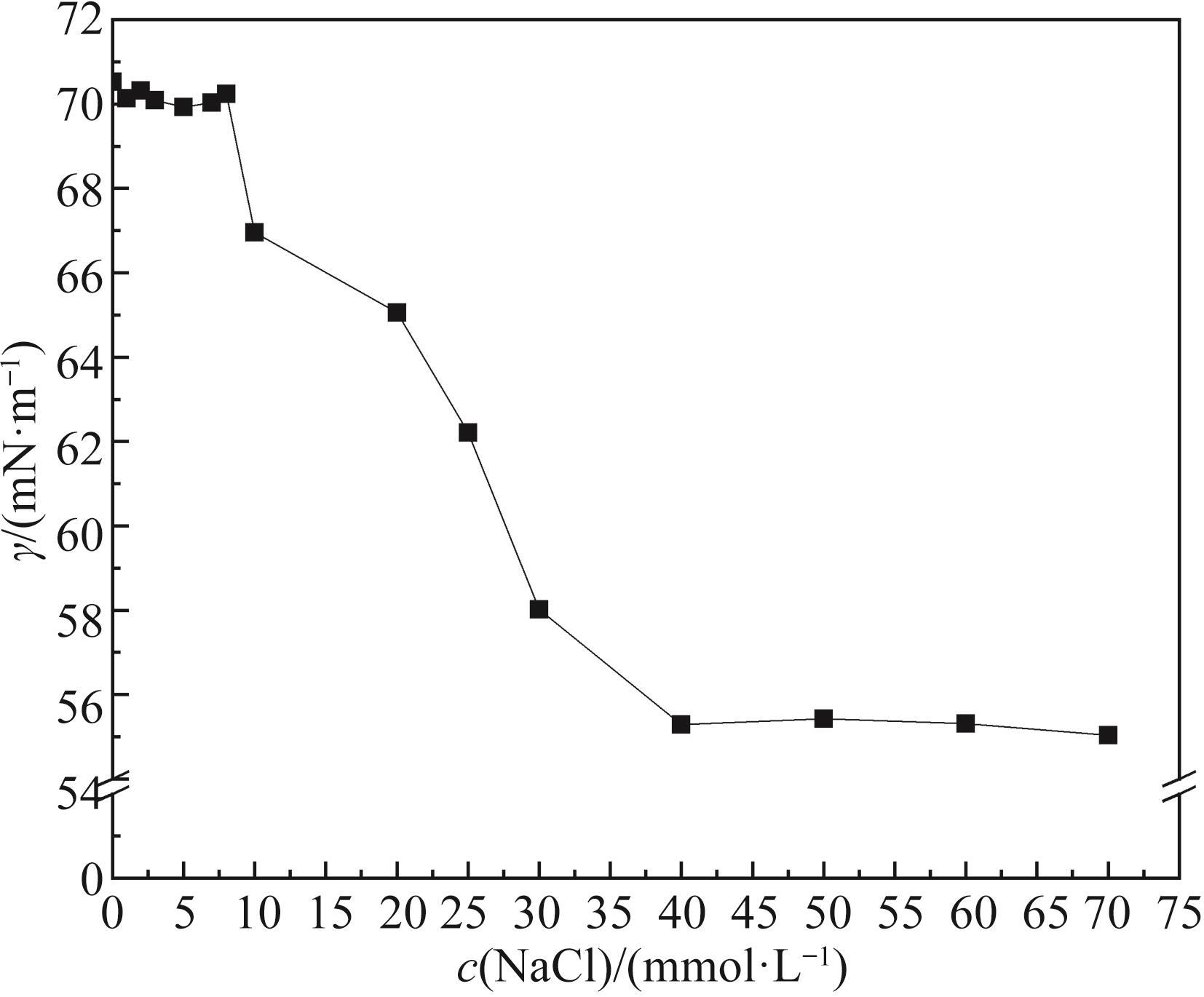
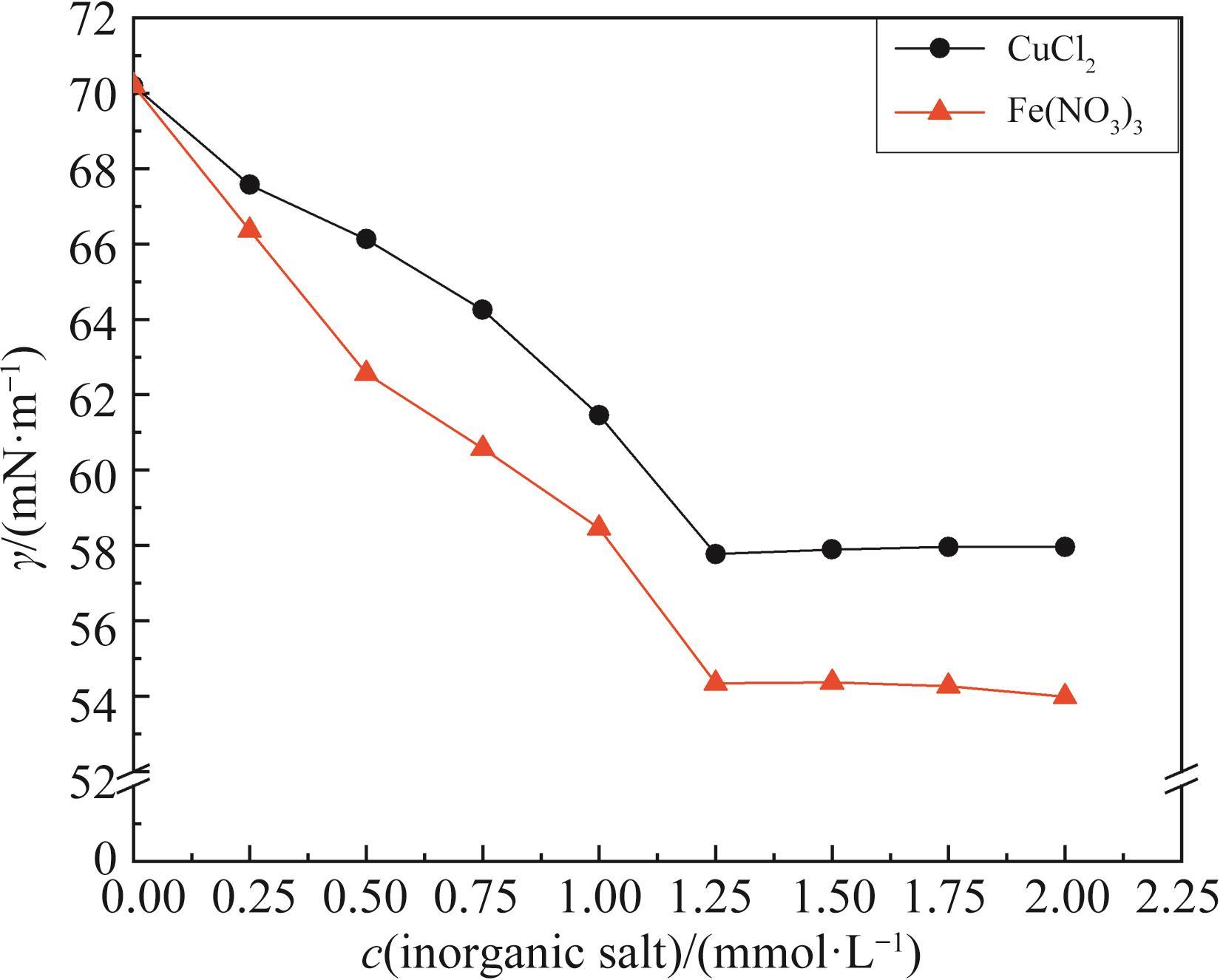
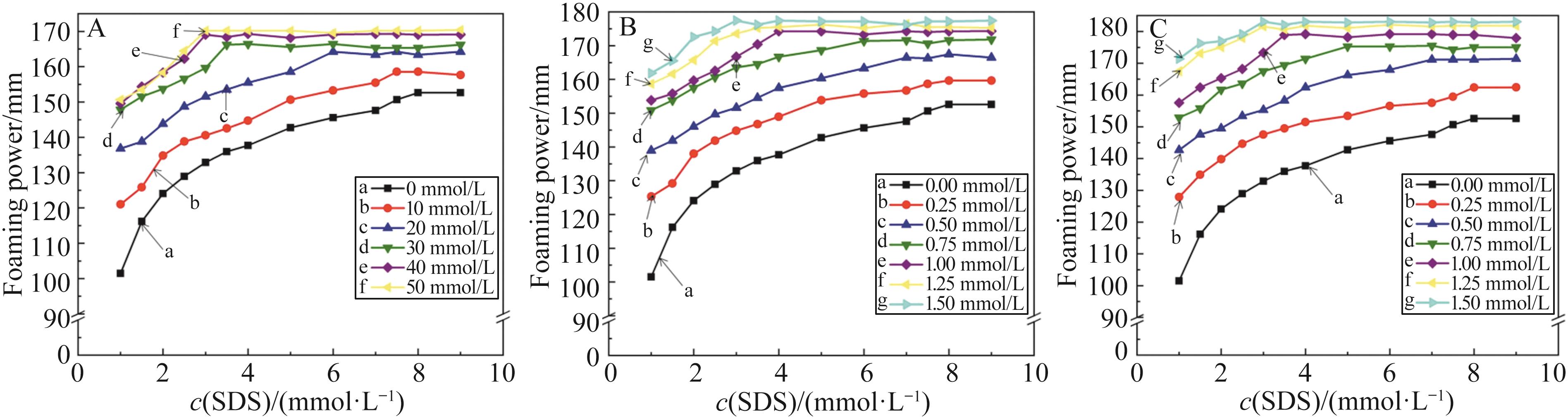
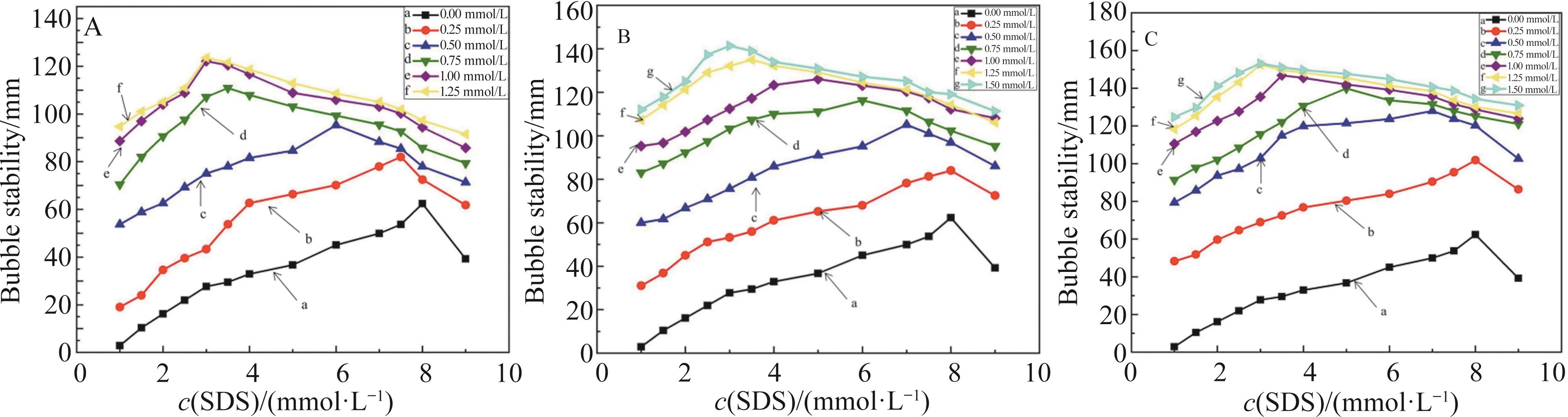
无机盐 Inorganic salts | c(NaCl)/(mmol·L-1) | c(CuCl2)/(mmol·L-1) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 10 | 20 | 30 | 40 | 50 | 0.25 | 0.50 | 0.75 | 1.00 | 1.25 | 1.50 | |
| CMC/(mmol·L-1) | 7.00 | 5.50 | 3.50 | 3.00 | 3.00 | 8.00 | 6.50 | 5.50 | 4.00 | 3.00 | 3.00 |
| Γ/(mN·m-1) | 47.29 | 45.12 | 39.25 | 39.26 | 39.09 | 46.16 | 44.42 | 43.16 | 37.63 | 33.94 | 33.52 |
| c(Fe(NO3)3)/(mmol·L-1) | |||||||||||
| 0.25 | 0.50 | 0.75 | 1.00 | 1.25 | 1.50 | ||||||
| 7.00 | 6.00 | 4.50 | 3.50 | 3.00 | 3.00 | ||||||
| 47.03 | 44.28 | 41.99 | 36.34 | 31.27 | 31.25 | ||||||
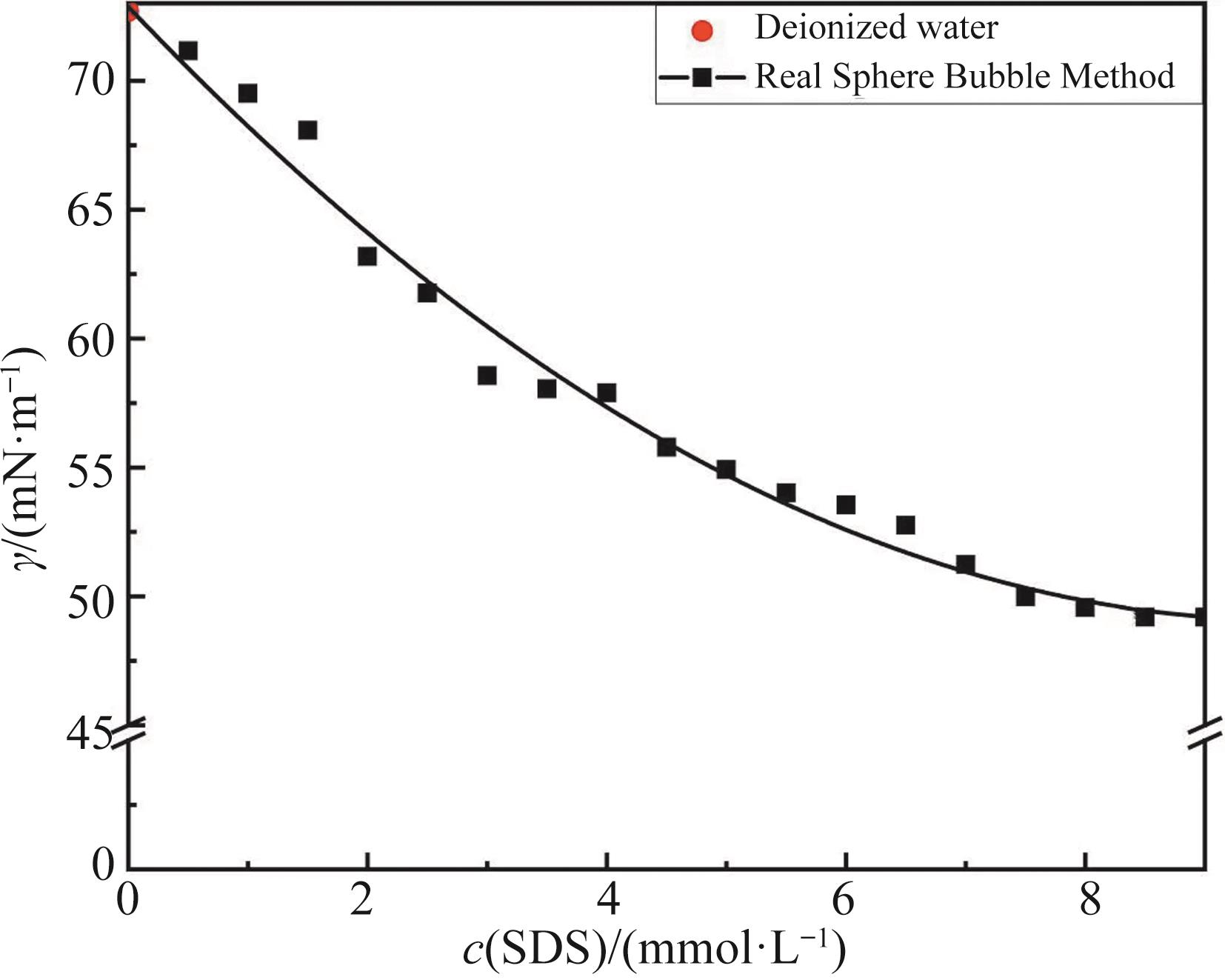
Table 1 CMC value and surface tension corresponding to the CMC value of SDS surfactant solution under the influence of inorganic salts
无机盐 Inorganic salts | c(NaCl)/(mmol·L-1) | c(CuCl2)/(mmol·L-1) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 10 | 20 | 30 | 40 | 50 | 0.25 | 0.50 | 0.75 | 1.00 | 1.25 | 1.50 | |
| CMC/(mmol·L-1) | 7.00 | 5.50 | 3.50 | 3.00 | 3.00 | 8.00 | 6.50 | 5.50 | 4.00 | 3.00 | 3.00 |
| Γ/(mN·m-1) | 47.29 | 45.12 | 39.25 | 39.26 | 39.09 | 46.16 | 44.42 | 43.16 | 37.63 | 33.94 | 33.52 |
| c(Fe(NO3)3)/(mmol·L-1) | |||||||||||
| 0.25 | 0.50 | 0.75 | 1.00 | 1.25 | 1.50 | ||||||
| 7.00 | 6.00 | 4.50 | 3.50 | 3.00 | 3.00 | ||||||
| 47.03 | 44.28 | 41.99 | 36.34 | 31.27 | 31.25 | ||||||
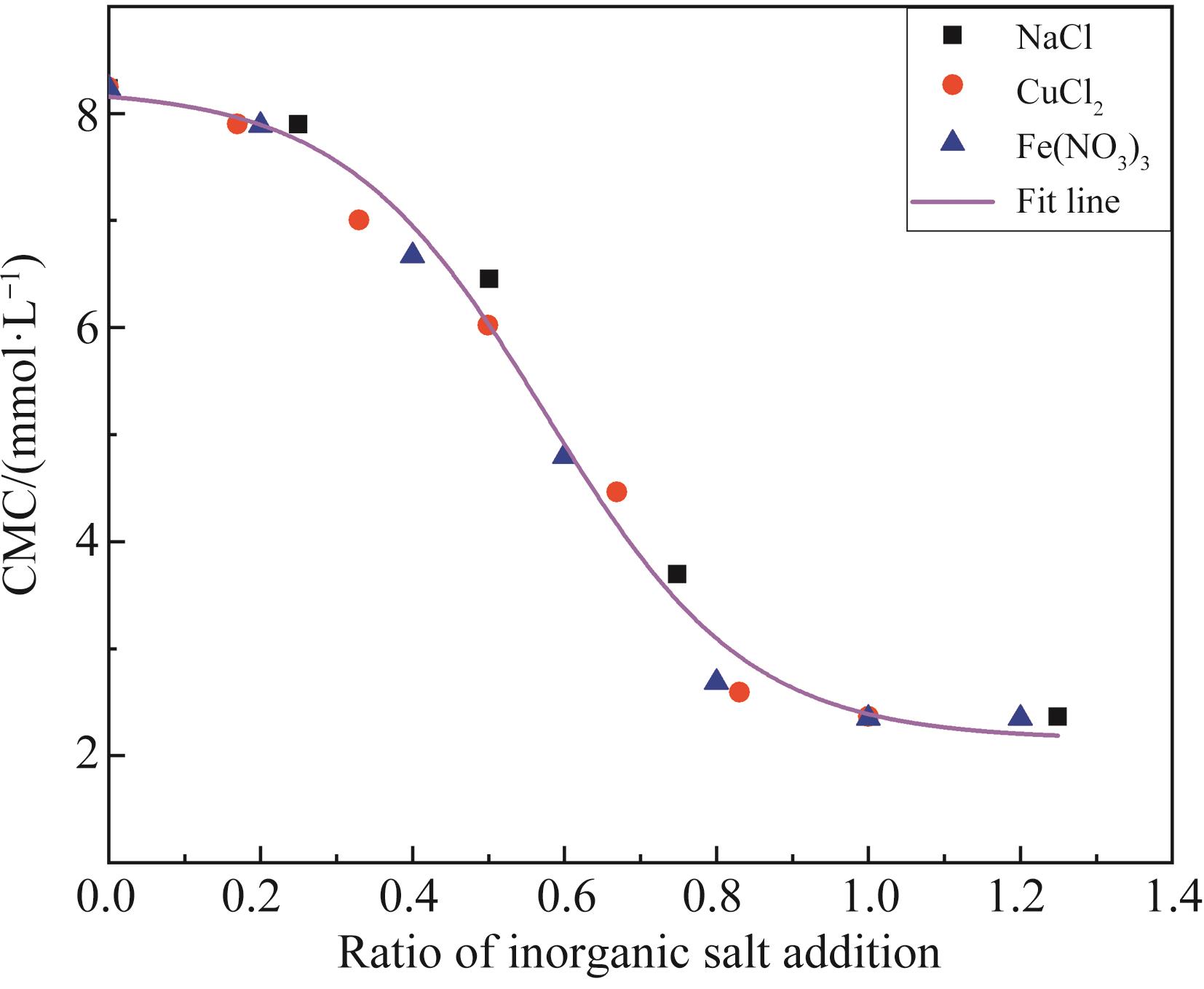
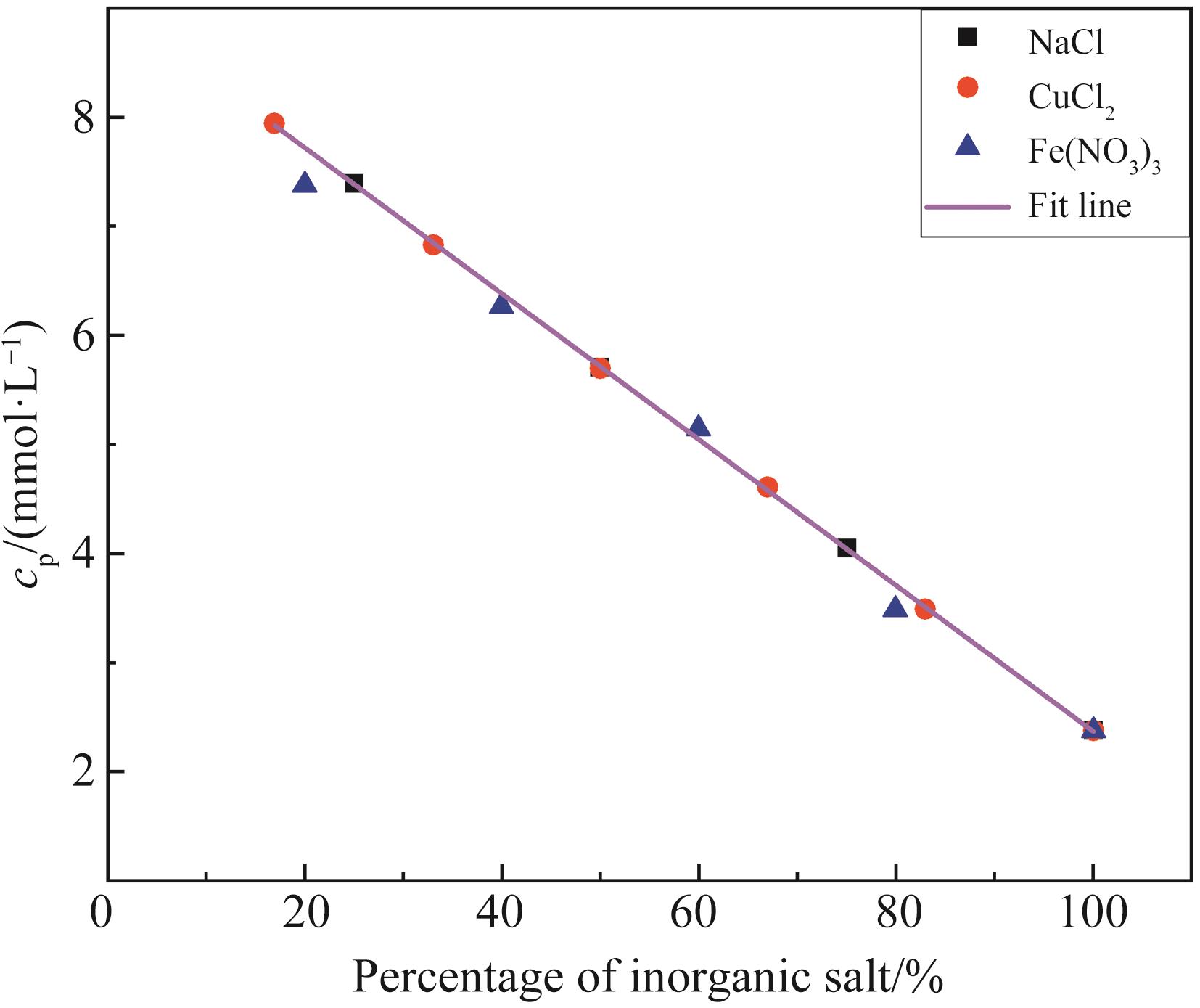
无机盐添加量比值 Ratio of inorganic salt addition | 0.17 | 0.20 | 0.25 | 0.33 | 0.40 | 0.50 | 0.60 | 0.67 | 0.75 | 0.80 | 0.83 | 1.00 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CMC/(mmol·L-1) | 7.97 | 7.89 | 7.70 | 7.26 | 6.69 | 5.65 | 4.61 | 4.04 | 3.59 | 3.41 | 3.32 | 3.01 |
Table 2 CMC values of different amounts of inorganic salts
无机盐添加量比值 Ratio of inorganic salt addition | 0.17 | 0.20 | 0.25 | 0.33 | 0.40 | 0.50 | 0.60 | 0.67 | 0.75 | 0.80 | 0.83 | 1.00 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CMC/(mmol·L-1) | 7.97 | 7.89 | 7.70 | 7.26 | 6.69 | 5.65 | 4.61 | 4.04 | 3.59 | 3.41 | 3.32 | 3.01 |
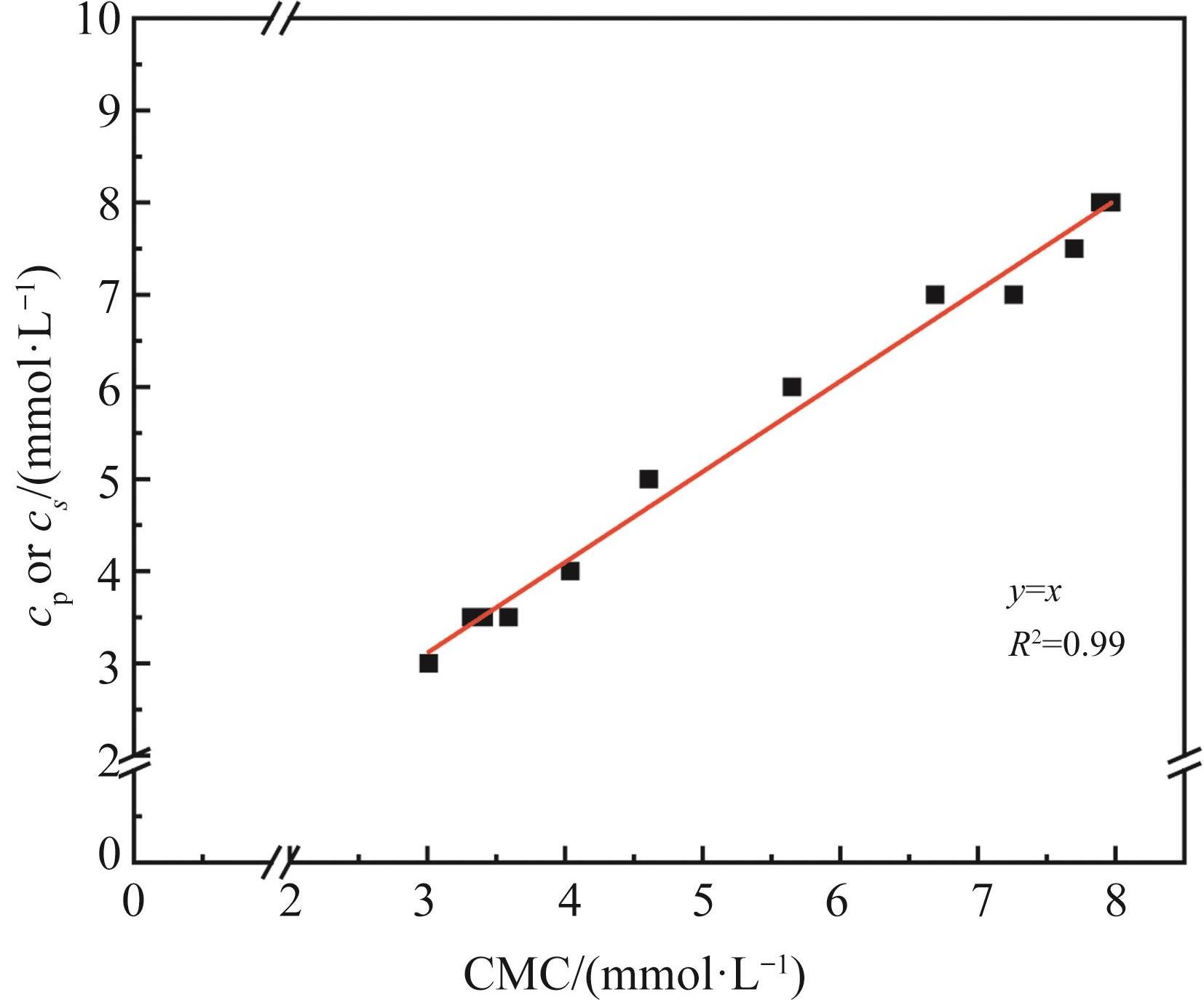
无机盐添加量 Percentage of inorganic salt/% | 17 | 20 | 25 | 33 | 40 | 50 | 60 | 67 | 75 | 80 | 83 | 100 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| cS=cP/(mmol·L-1) | 8.00 | 8.00 | 7.50 | 7.00 | 7.00 | 6.00 | 5.00 | 4.00 | 3.50 | 3.50 | 3.50 | 3.00 |
Table 3 cS and cP of different inorganic salt addition
无机盐添加量 Percentage of inorganic salt/% | 17 | 20 | 25 | 33 | 40 | 50 | 60 | 67 | 75 | 80 | 83 | 100 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| cS=cP/(mmol·L-1) | 8.00 | 8.00 | 7.50 | 7.00 | 7.00 | 6.00 | 5.00 | 4.00 | 3.50 | 3.50 | 3.50 | 3.00 |
| 1 | 王春雨, 赵辉, 代正华, 等. 表面活性剂对亲水性颗粒悬浮液流变性的影响[J]. 应用化学, 2021, 38(4): 398-406. |
| WANG C Y, ZHAO H, DAI Z H, et al. Effect of surfactant on the rheological properties of hydrophilic particle suspension[J]. Chinese J Appl Chem, 2021, 38(4): 398-406. | |
| 2 | 程锦, 陈章洋, 张峪铭, 等. 多元表面活性剂复配的分子热力学模型研究[J]. 化工学报, 2020, 71(10): 4590-4600. |
| CHENG J, CHEN Z Y, ZHANG Y M, et al. Molecular thermodynamic model for compounding of multiple surfactants[J]. CIESC J, 2020, 71(10): 4590-4600. | |
| 3 | 娄朋晓, 王玉洁, 白光月, 等. 表面活性剂分子间弱相互作用的直接能量表征—高灵敏等温滴定量热法[J]. 物理化学学报, 2013, 29(7): 1401-1407. |
| LOU P X, WANG Y J, BAI G Y, et al. Direct evidence for the energy of weak interactions between surfactant molecules using high sensitivity isothermal titration calorimetry[J]. Acta Phys Chim Sin, 2013, 29(7): 1401-1407. | |
| 4 | 林喆, 匡亚莉, 张海阳. 射流发泡与小球藻的批次气浮采收[J]. 中国矿业大学学报, 2012, 41(5): 839-843. |
| LING Z, KUANG Y L, ZHANG H Y. Generating of jet bubble and harvesting of chlorella biomass by air flotation[J]. CUMT J, 2012, 41(5): 839-843. | |
| 5 | MAJEED T, SØLLING T, KAMAL M. Foam stability: the interplay between salt-, surfactant- and critical micelle concentration[J]. J Pet Sci Eng, 2020, 187: 1-8. |
| 6 | 朱文会, 张金彦, 吴学渊. 滴体积法测定表面张力求临界胶团浓度及热力学函数[J]. 精细化工, 1988(6): 24-26. |
| ZHU W H, ZHANG J Y, WU X Y. Determination of critical micelle concentration and thermodynamic function of surface tension by drop volume method[J]. Fine Chem, 1988(6): 24-26. | |
| 7 | 倪良, 胡莹海, 吴春笃, 等.电导法研究环己烷/水/CTAB乳状液的稳定性[J]. 江苏大学学报(自然科学版), 2005, 26(6): 550-552. |
| NI L, HU Y H, WU C D, et al. Study on the stability of cyclohexane/water/CTAB emulsion by conductivity method[J]. J Jiangsu Univ (Nat Sci Ed), 2005, 26(6): 550-552. | |
| 8 | 巩孝栋, 魏西莲. 吸附伏安法测定阳离子表面活性剂的临界胶束浓度[J]. 山东理工大学学报(自然科学版), 2003, 31(4): 123-125. |
| GONG X D, WEI X L. Determination of critical micelle concentration of cationic surfactants by adsorptive voltammetry[J]. J Shandong Univ Technol (Nat Sci Ed), 2003, 31(4): 123-125. | |
| 9 | 宗李燕, 蔡琨, 刘雪锋, 等. 超滤法测定表面活性剂临界胶束浓度[J]. 无锡轻工大学学报, 2001, 20(5): 510-514. |
| ZONG L Y, CAI K, LIU X F, et al. Determination of critical micelle concentration of surfactants by ultrafiltration[J]. J Wuxi Univ Light Ind, 2001, 20(5): 510-514. | |
| 10 | 卢惠娟, 陈冲, 郭宏涛, 等.无探针紫外光谱法测定CTAB的第二临界胶束浓度[J]. 化学学报, 2006, 64(24): 2437-2441. |
| LU H J, CHEN C, GUO H T, et al. Determination of the second critical micelle concentration of CTAB by UV spectra without probe[J]. Acta Chim Sin, 2006, 64(24): 2437-2441. | |
| 11 | 王美荣, 金志琳. 光散射技术及其在化学与化工领域中的应用[J]. 山东化工, 2005(1): 19-23. |
| WANG M R, JIN Z L. Light scattering technology and its application in chemistry and chemical industry[J]. Shandong Chem Ind, 2005(1): 19-23. | |
| 12 | 严峰. 荧光法测定N-(α-烷苯氧基)十四酰基牛磺酸钠的临界胶束浓度[J]. 影像科学与光化学, 2007, 25(2): 115-122. |
| YAN F. Determination of critical micelle concentration of N-(α-alkanephenoxy) tetranoyl taurine by fluorescence method[J]. Photogra Sci Photochem, 2007, 25(2): 115-122. | |
| 13 | 陆振荣. 十二烷基磷酸钠盐的制备及其在水溶液中CMC的测定[J]. 苏州大学学报(自然科学版), 1991(3): 329-333. |
| LU Z R. Preparation of sodium dodecyl phosphate and determination of CMC in aqueous solution[J]. J Soochow Univ (Nat Sci Ed), 1991(3): 329-333. | |
| 14 | 赵国玺, 朱步瑶. 表面活性剂作用原理[M]. 北京: 中国轻工业出版社, 2003. |
| ZHAO G X, ZHU B Y. Principle of surfactant action[M]. Beijing: China Light Industry Press, 2003. | |
| 15 | 张锐, 王瑞和, 邱正松, 等. 利用光散射原理评价泡沫钻井液的稳定性[J]. 石油学报, 2005, 26(1): 108-111. |
| ZHANG R, WANG R H, QIU Z S, et al. Using light scattering principle to evaluate the stability of foam drilling fluids[J]. Acta Petro Sin, 2005, 26(1): 108-111. | |
| 16 | PHIANMONGKHOL A, VARLEY J. A multi point conductivity measurement system for characterisation of protein foams[J]. Colloids Surf B, 1999, 12(3/6): 247-259. |
| 17 | VIKINGSTAD A, AARRA M, SKAUGE A. Effect of surfactant structure on foam-oil interactions: comparing fluorinated surfactant and alpha olefin sulfonate in static foam tests[J]. Collloids Surf A, 2005, 279(1/3): 105-112. |
| 18 | XU Y Q, MITSUTOSHI N, SOSAKU I, et al. Effects of surfactant and electrolyte concentrations on bubble formation and stabilization[J]. J Colloid Interface Sci, 2008, 332(1): 108-114 . |
| 19 | BEHERA M R, VARADE S R, GHOSH P, et al. Foaming in micellar solutions: effects of surfactant, salt, and oil concentrations[J]. Ind Eng Chem Res, 2014, 53(48): 18497-18507. |
| 20 | YEKEEN N, MANAN M, IDRIS A, et al. Influence of surfactant and electrolyte concentrations on surfactant adsorption and foaming characteristics[J]. J Pet Sci Eng, 2017, 149(20): 612-622. |
| 21 | 余仲达. 表面张力和表面面积粘性测定装置: 中国, CN201410327619.3[P]. 2014-11-05. |
| YU Z D. Surface tension and surface area viscosity measuring device: China, CN201410327619.3[P]. 2014-11-05. | |
| 22 | 郭杰, 余仲达, 郑少波, 等. 一种新的表面张力测定方法-真球气泡法[J]. 上海大学学报(自然科学版), 2020, 26(2): 244-254. |
| GUO J, YU Z D, ZHENG S B, et al. A new method for measuring surface tension-real sphere bubble method[J]. J Shanghai Univ (Nat Sci Ed), 2020, 26(2): 244-254. | |
| 23 | 许鹏军, 余仲达, 郑少波, 等. 一种测定表面扩展粘度的新方法—真球气泡法[J]. 中国矿业大学学报(自然科学版), 2020, 49(5): 998-1005. |
| XU P J, YU Z D, ZHENG S B. A new method for measuring surface dilatational viscosity-real sphere bubble method[J]. J China Univ Min Technol, 2020, 49(5): 998-1005. | |
| 24 | 周凤山. 钻井液用泡沫剂性能评价方法[J]. 钻井液与完井液, 1990, 7(3): 1-5, 28. |
| ZHOU F S. Performance evaluation method of foaming agent for drilling fluid[J]. Drill Complet Fluid, 1990, 7(3): 1-5, 28. | |
| 25 | 王寿武, 李泉, 于红梅, 等. 氯化钠对表面活性剂粘度效应的研究[J]. 盐科学与化工, 2003, 32(6): 8-10. |
| WANG S W, LI Q, YU H M, et al. Study on the effect of sodium chloride on the viscosity of surfactants[J]. Salt Sci Chem Ind, 2003, 32(6): 8-10. | |
| 26 | 刘樟, 王毅琳. 多价金属离子对阴离子磺酸盐Gemini表面活性剂的表界面行为的影响[C]// 中国化学会第十五届胶体与界面化学会议论文集(第一分会), 武汉, 2015. |
| LIU Z, WANG Y L. The effect of multivalent metal ions on the surface and interface behavior of anionic sulfonate gemini surfactants [C]//the 15th Colloid and Interface Chemistry Conference of the Chinese Chemical Society (the first session), Wuhan, 2015. | |
| 27 | 耿林. 表面活性剂的临界胶束浓度与分子能量项和分子结构的关系研究[D]. 无锡: 江南大学, 2012. |
| GENG L. Study on the relationship between the critical micellar concentration of surfactants and the molecular energy term and molecular structure[D]. Wuxi: Jiangnan University, 2012. | |
| 28 | ROSEN M. Surfactants and interfacial phenomena[M]. New York: John Wiley & Sons, 1978: 123-126. |
| 29 | 舒梦, 陈萍华, 蒋华麟, 等. 十二烷基硫酸钠的临界胶束浓度的测定及影响分析[J]. 化工时刊, 2014, 28(3): 1-3. |
| SHU M, CHEN P H, JIANG H L, et al. Determination of sodium dodecyl sulfate critical micelle concentration and impact analysis[J]. Chem Ind Time. 2014, 28(3): 1-3. | |
| 30 | 李修芝. 季铵盐Gemini表面活性剂与SDS的相互作用—相行为、胶团化及流变特性[D]. 福州: 福州大学, 2003. |
| LI X Z. Interaction between Gemini surfactant and SDS: phase behavior, micellization and rheological properties[D]. Fuzhou: Fuzhou University, 2003. | |
| 31 | FENFLER, ELEANOR J. Catalysis in micellar and macromolecular systems[M]. Academic Press, 1975. |
| 32 | FRANK H, EVANS M. Free volume and entropy in condensed systemsⅢ. entropy in binary liquid mixtures; partial molal entropy in dilute solutions; structure and thermodynamics in squeous electrolytes[J]. J Chem Phys, 1945, 13(11): 507. |
| 33 | 邓丽君, 李国胜, 曹亦俊, 等. 浮选起泡剂对气泡兼并行为的影响研究[J]. 中国矿业大学学报, 2017(2): 410-414. |
| DENG L J, LI G S, CAO Y J, et al. Study on the effect of flotation frother on bubble merger behavior[J]. J China Univ Min Technol, 2017(2): 410-414. |
| [1] | WANG Chun-Yu, ZHAO Hui, DAI Zheng-Hua, LI Wei-Feng, LIU Hai-Feng. Effect of Surfactant on the Rheological Properties of Hydrophilic Particle Suspension [J]. Chinese Journal of Applied Chemistry, 2021, 38(4): 398-406. |
| [2] | LIU Jiajia, XIE Yicheng, XU Hujun. Synthesis and Properties of a Cationic Gemini Asphalt Emulsifier [J]. Chinese Journal of Applied Chemistry, 2018, 35(5): 552-558. |
| [3] | ZHANG Qian, SUN Yongqiang, ZHI Lifei, ZHANG Yong, SUN Jinyuan, WU Huaping, DI SERIO Martino. Dynamic Surface Tensions of Castor Oil Acid Methyl Ester Ethoxylate Aqueous Solutions [J]. Chinese Journal of Applied Chemistry, 2015, 32(6): 689-694. |
| [4] | CHENG Qi1, LIU Xinmei1*, DAI Caili2. Synthesis and Surface Activity of N-(2-Hydroxylpropyl) Perfluorooctane Amide Foaming Agent [J]. Chinese Journal of Applied Chemistry, 2013, 30(11): 1276-1280. |
| [5] | DING Wei*, LI Jinhong, YU Tao, XING Xinxin, ZHANG Wei, YANG Liu. Surface/Interface Performance of New Sulfobetaine/Polyacrylamide System [J]. Chinese Journal of Applied Chemistry, 2013, 30(11): 1270-1275. |
| [6] | ZHAO Qinqin, SHI Hongxin*, WU Hongke, CHEN Lijun, XIANG Juping. Synthesis and Performance of Polyethylene Glycol p-Perfluorous Nonenyloxyphenyl Sulflonate Surfactant [J]. Chinese Journal of Applied Chemistry, 2012, 29(08): 906-909. |
| [7] | DING Wei1*, REN Ya′ni1, LIU Hongbin1, SHI Peng1, YU Tao1, QU Guangmiao1, CHENG Jiecheng2, WU Junzheng2. Effect of Relative Molecular Mass on the Adsorption of Alkyl Aryl Sulfonate Composite Systems at the Solid-Liquid Interface [J]. Chinese Journal of Applied Chemistry, 2011, 28(08): 963-968. |
| [8] | YU Tao*, LIU Hongbin, DING Wei, SHI Peng, WEI Jijun, MAO Cheng, REN Ya′ni, QU Guangmiao. Relationship Between Relative Molecular Mass and Distribution of Alkyl Aryl Sulfonate Composite Systems and Surface Properties [J]. Chinese Journal of Applied Chemistry, 2011, 28(07): 831-835. |
| [9] | LI Haichao2, LIU Jinyan*, ZHANG Yingchun. Interaction of Sodium Dodecyl Benzene Sulfonate and TritonX-100 Studied by Intrinsic Fluorescence [J]. Chinese Journal of Applied Chemistry, 2011, 28(01): 113-117. |
| [10] | HU Ying-Mo. Synthesis and Characterization of Polymerizable Nonionic Surfactants [J]. Chinese Journal of Applied Chemistry, 2010, 27(04): 409-412. |
| [11] | XIE Dan-Hua, ZHOU Tian-Hua, ZHAO Jian-Xi*. Effects of NaBr on the Adsorption and Aggregation of Heterogemini Surfactant CmOhpNC8 in Aqueous Solution [J]. Chinese Journal of Applied Chemistry, 2010, 27(03): 318-322. |
| [12] | ZHANG Lei*, LIU Na, KANG Ping-Li, LI Qi, ZHOU Xin-Yu. Nanometer TiO2 Microcolumn Preconcentration of Trace Selenium in Environment Samples [J]. Chinese Journal of Applied Chemistry, 2010, 27(03): 323-327. |
| [13] | HUANG Lin1, WU Yu-Cheng1*, SUN Ai-Hua2, LI Yong2, CUI Bing2. Humidity-sensitive Properties Based on Salts-doped MEBA-co-KH570 Copolymer [J]. Chinese Journal of Applied Chemistry, 2010, 27(01): 43-47. |
| [14] |
ZHANG Jian, QIU Yu, YU Dao-Yong*.
Critical Micelle Concentration Determination of Sodium Dodecyl Benzene Sulfonate by Synchronous Fluorescence Spectrometry [J]. Chinese Journal of Applied Chemistry, 2009, 26(12): 1480-1483. |
| [15] | DING Wei, SU Ya-Bin, ZHANG Chun-Hui, GAO Xiao-Yu, LI An-Jun, YANG Yang, XU Tao, CHENG Jie-Cheng. Study on the Effecting Factors of Surface Properties of pentadecyl m-xylene sodium sulfonates Solution [J]. Chinese Journal of Applied Chemistry, 2009, 26(09): 1023-1026. |
| Viewed | ||||||||||||||||||||||||||||||||||||||||||||||||||
|
Full text 720
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
Abstract 672
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||