
Chinese Journal of Applied Chemistry ›› 2024, Vol. 41 ›› Issue (3): 309-327.DOI: 10.19894/j.issn.1000-0518.230308
• Rare Earth •
Applications of Rare Earth-Based Nanoparticles in Brain Tumors
Na YIN1, Ying-Hui WANG1( ), Hong-Jie ZHANG1,2(
), Hong-Jie ZHANG1,2( )
)
- 1.State Key Laboratory of Rare Earth Resource Utilization,Changchun Institute of Applied Chemistry,Chinese Academy of Sciences,Changchun 130022,China
2.Department of Chemistry,Tsinghua University,Beijing 100084,China
-
Received:2023-10-08Accepted:2024-01-09Published:2024-03-01Online:2024-04-09 -
Contact:Ying-Hui WANG,Hong-Jie ZHANG -
Supported by:the National Natural Science Foundation of China(52022094)
CLC Number:
Cite this article
Na YIN, Ying-Hui WANG, Hong-Jie ZHANG. Applications of Rare Earth-Based Nanoparticles in Brain Tumors[J]. Chinese Journal of Applied Chemistry, 2024, 41(3): 309-327.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.230308

Fig.1 Simplified energy state diagrams, typical adsorption and emission wavelength of trivalent lathanide ions (Yb3+, Er3+, Tm3+, Nd3+, Ho3+, Pr3+ and Sm3+)
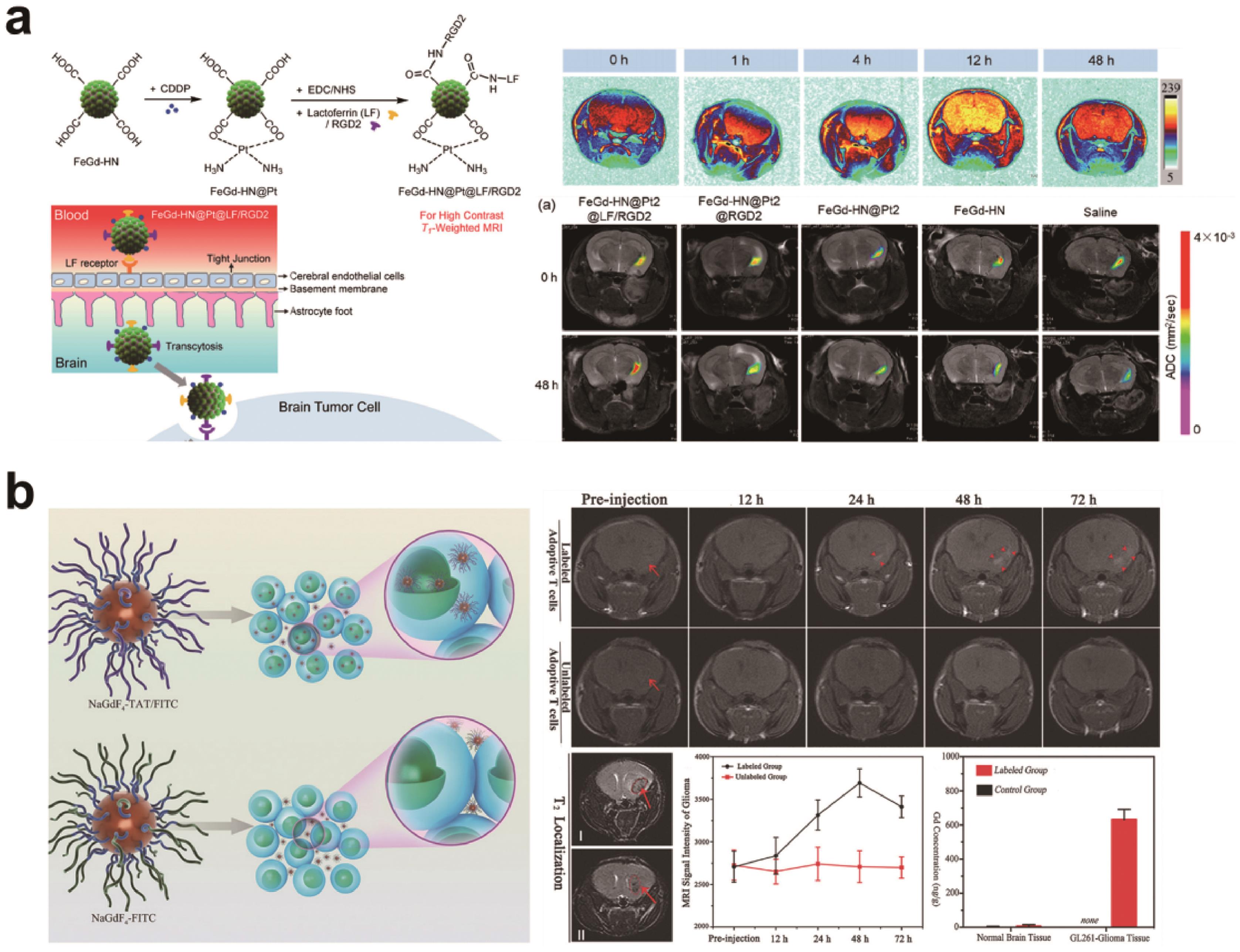
Fig.2 (a) Schematic illustration for synthesis of the Fe3O4/Gd2O3 hybrid nanoparticles and the results for T1-weighted imaging effect[36]; (b) Schematic illustration for cell-penetrating NaGdF4-TAT/FITC and non-cell-penetrating NaGdF4-FITC labeling of adoptive T cell and T1-weighted magnetic resonance imaging effects of both on adoptive T cell in orthotopic glioma[39]
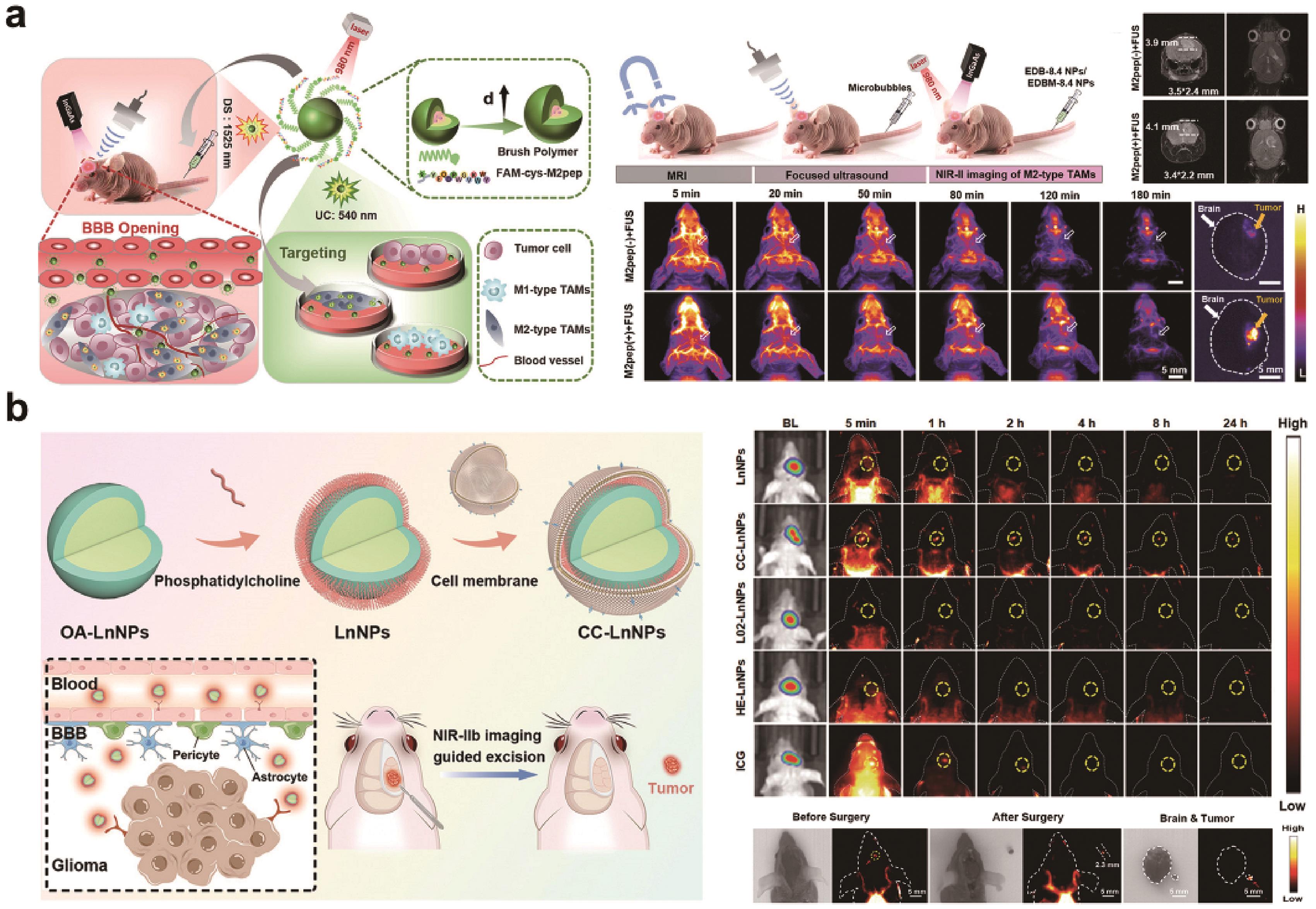
Fig.3 (a) Schematic illustration of NIR-Ⅱb EDBM-8.4 nanoprobes for targeted imaging of M2-type TAMs both in vitro and in orthotopic GBM. And T2-weighted MRI and NIR-Ⅱ fluorescence imaging of orthotopic GBM-bearing mice post intravenous injection of EDB-8.4 NPs and EDBM-8.4 NPs, with or without FUS[45]; (b) Schematic illustration of the preparation processes of brain tumor cell membrane-coated RENPs and its application in brain tumor imaging and surgical navigation. and the results in vivo fluorescence imaging and surgery of mice[46]
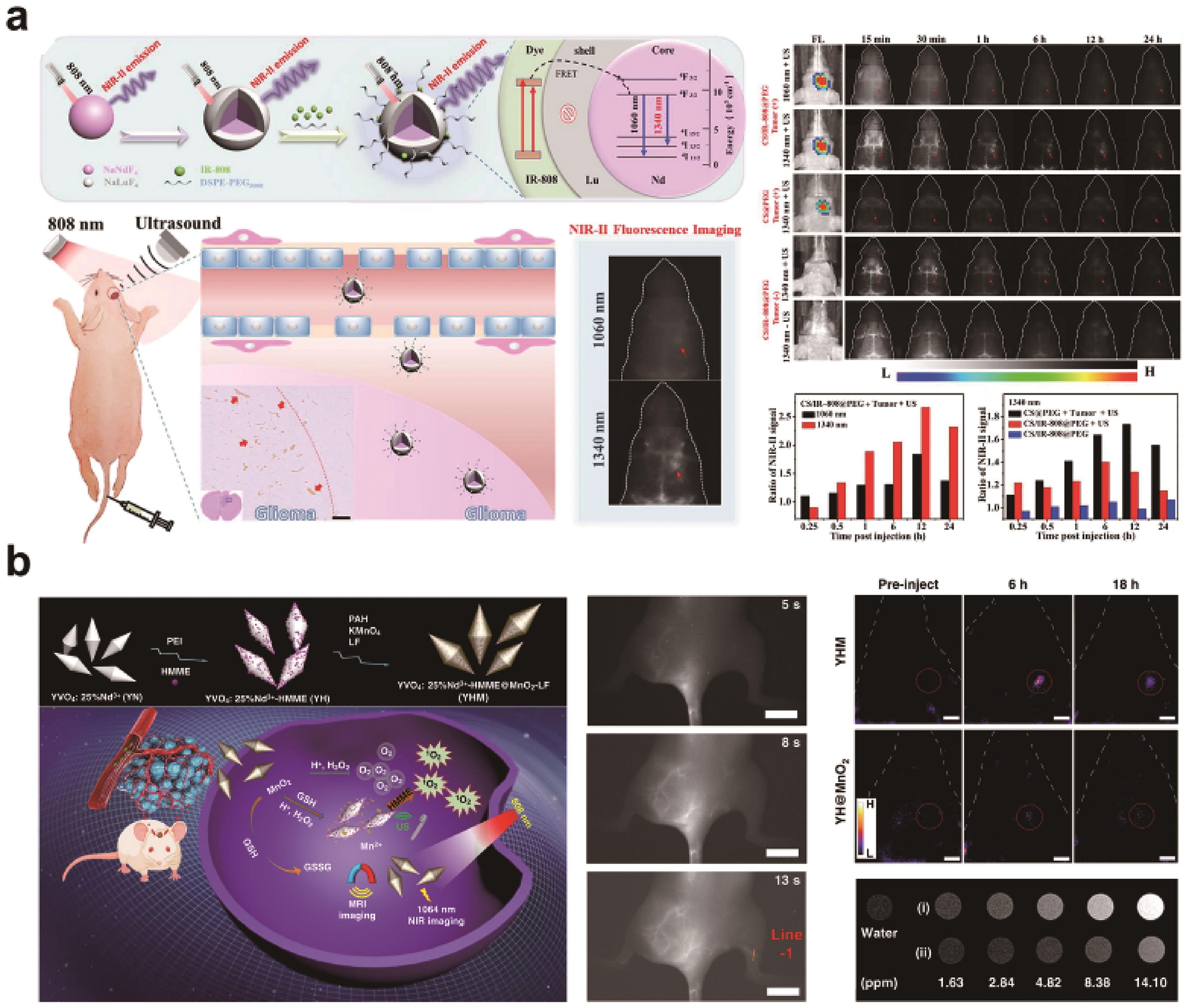
Fig.4 (a) Schematic illustration of the design of the NaNdF4@NaLuF4/IR-808@DSPE-PEG5000 RENPs and their applications in NIR-Ⅱ fluorescence imaging of orthotopic glioblastoma under ultrasound-mediated BBB opening and in vivo fluorescence imaging of orthotopic glioblastomas at 1340 nm emission under ultrasound[52]; (b) Schematic diagram of the construction of YHM and main mechanism of multimodal bioimaging and therapy, and its NIR-Ⅱ imaging in vascular and orthotopic gliomas[53]
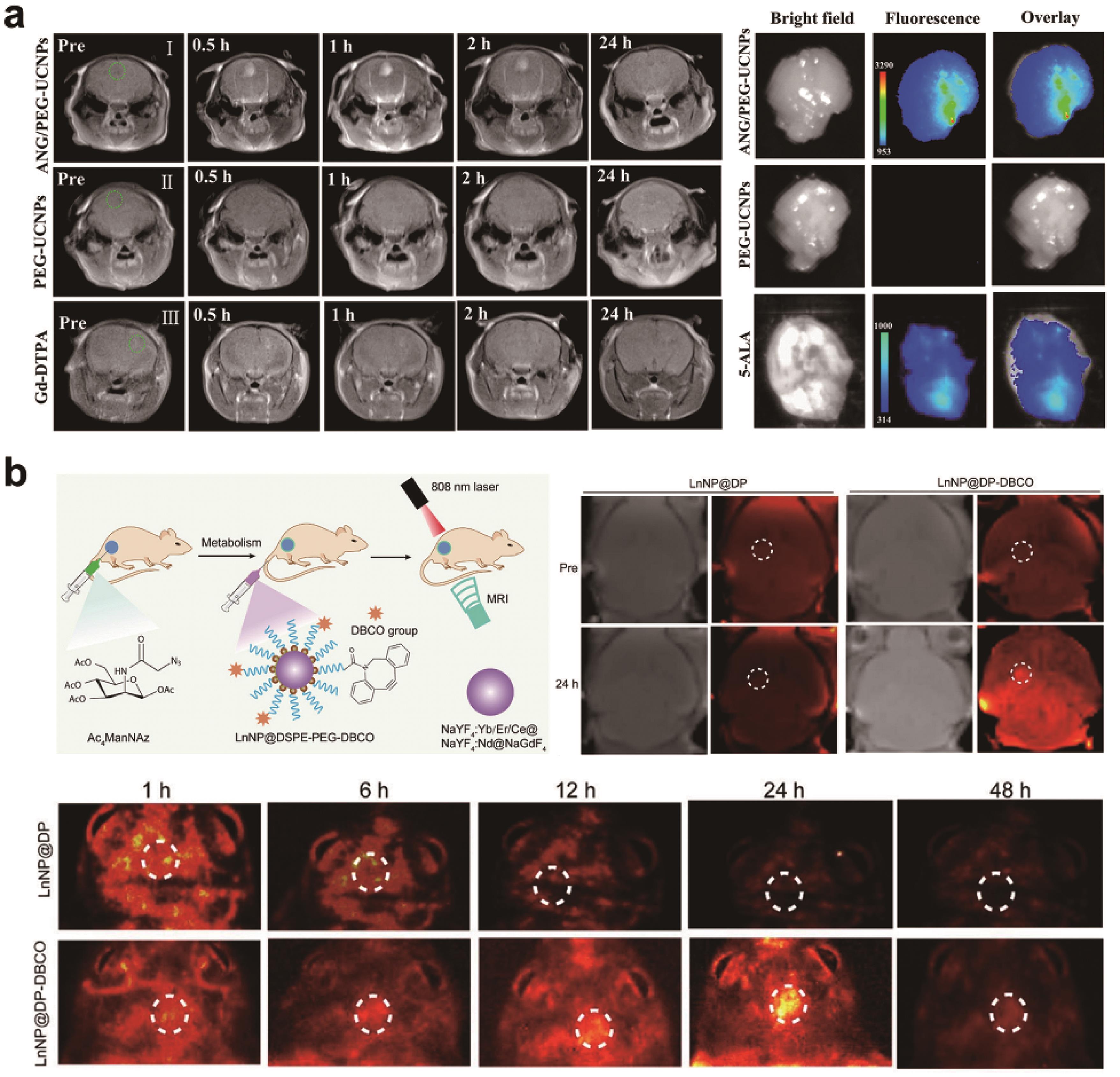
Fig.5 (a) In vivo T1-weighted MRI in glioblastoma-bearing mice before and at various time points after the intravenous injections of ANG/PEG-UCNPs, and ex vivo fluorescent images of glioblastoma-bearing brain[63]?; (b) Schematic illustration of biofunctionalization of multilayer nanoparticles (LnNP@DSPE-PEG-DBCO) for dual-modal tumor imaging, and the results of T1-weighted MRI images and NIR-Ⅱb fluorescence imaging in glioblastoma-bearing mice at different time points[65]
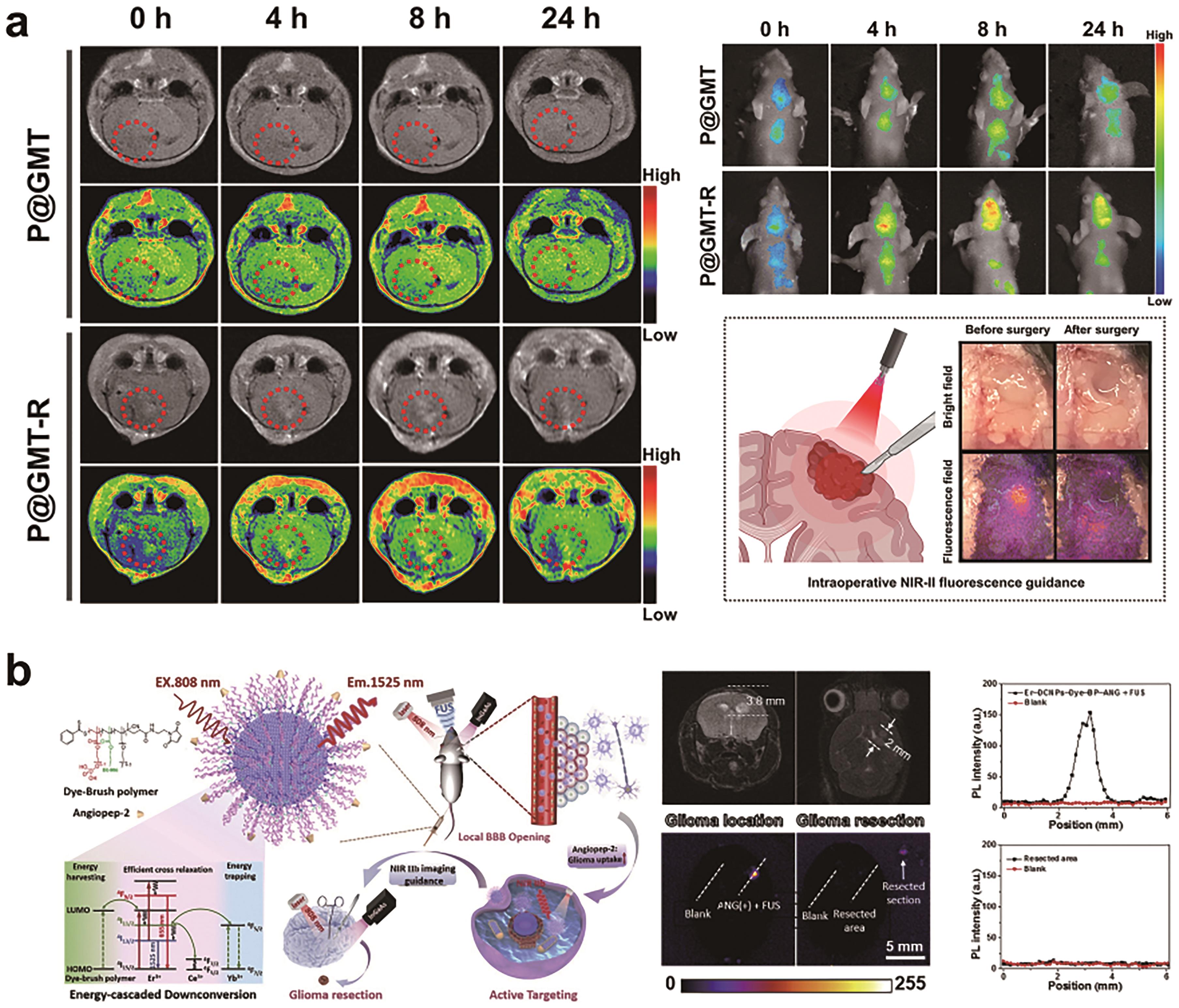
Fig.6 (a) Gd2O3∶Nd3+-based nanomaterials for MRI imaging as well as NIR-Ⅱ fluorescence imaging of orthotopic gliomas and fluorescence-guided surgical resection[76]; (b) Schematic illustration of the Er-DCNPs-Dye-BP-ANG design, emission mechanism and the application of biomedical imaging and the assessment about NIR-Ⅱb fluorescence imaging to guide surgical resection of orthotopic gliomas[50]
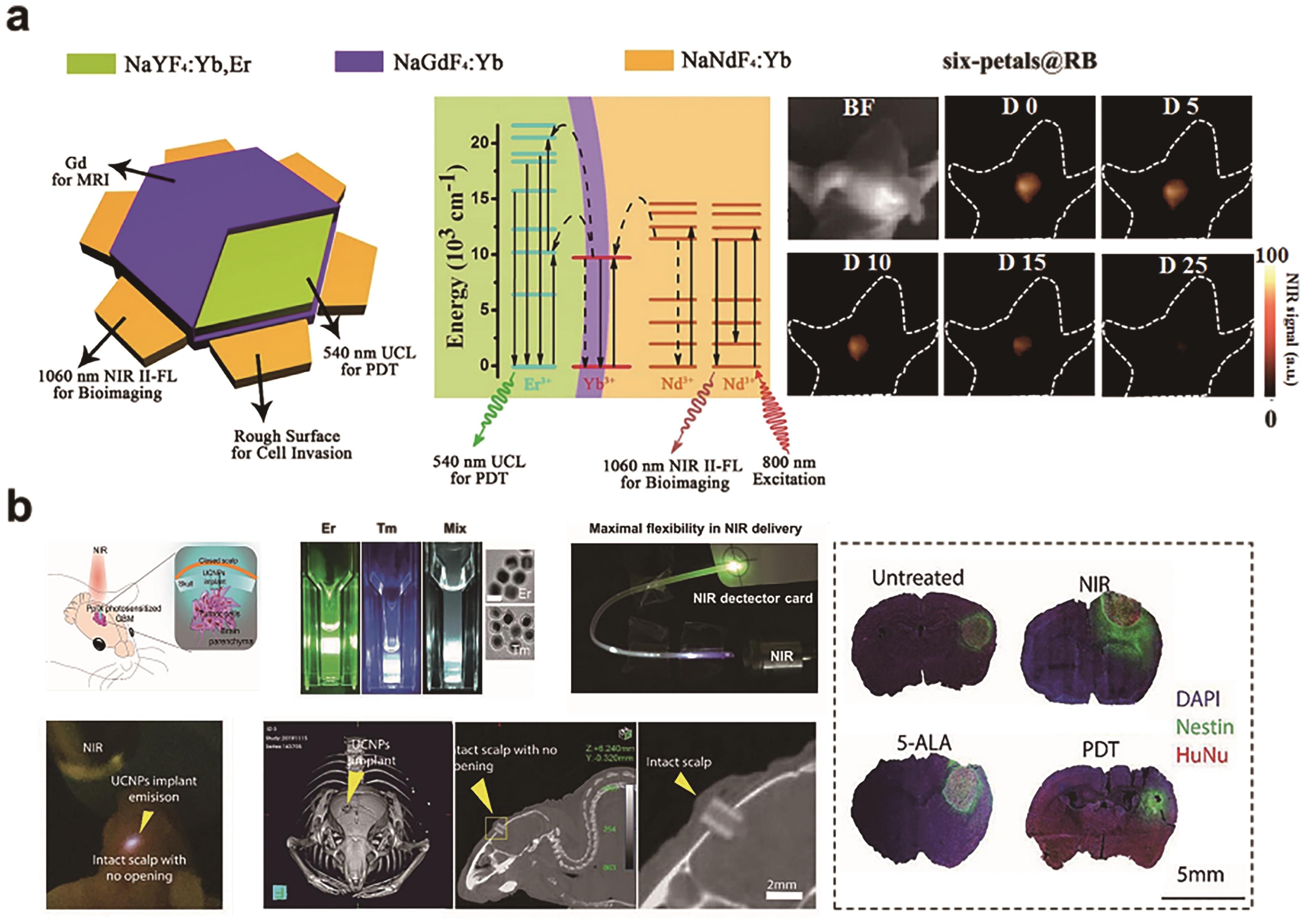
Fig.8 (a) The structure scheme and energy-transfer mechanisms of the six-petals nanoplates. And NIR-?Ⅱ FL bioimaging guided glioblastoma-bearing mice PDT of six-petals@RB[90]?; (b) The concept of UCNPs implant for wireless NIR-PDT in a mouse GBM model, and the results of microCT scan of UCNPs implant and effectiveness of PDT treatment[24]
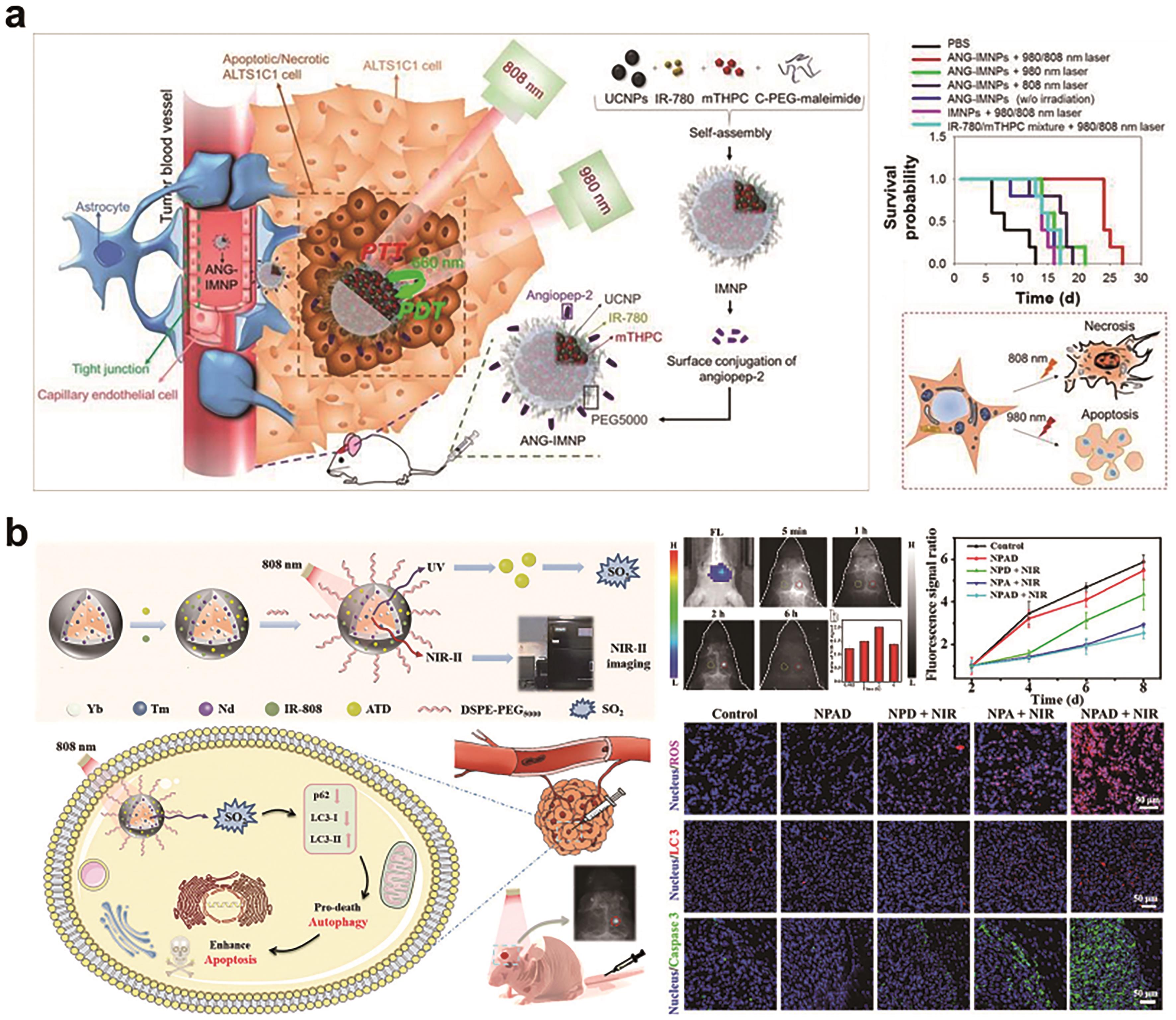
Fig.9 (a) Illustration of active BBB penetration and the photothermal/photodynamic therapeutic design of ANG-IMNPs in an orthotopic glioblastoma tumor model, and the assessment of ANG-IMNPs treatment effects[92]?; (b) Illustration of the core-shell NaYF4∶?Yb/Tm@NaYF4∶?Nd@ATD/Dye@DSPE-PEG5000 NPs in the NIR-Ⅱ fluorescence imaging of orthotopic glioblastoma and photo-controlled release of SO2 for therapy, and the assessment of NIR-Ⅱ fluorescence imaging and gas therapy[100]
| 1 | SUNG H, FERLAY J, SIEGEL R L, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71(3): 209-249. |
| 2 | DELGADO-LOPEZ P D, CORRALES-GARCIA E M. Survival in glioblastoma: a review on the impact of treatment modalities[J]. Clin Transl Oncol, 2016, 18(11): 1062-1071. |
| 3 | REIFENBERGER G, WIRSCHING H G, KNOBBE-THOMSEN C B, et al. Advances in the molecular genetics of gliomas-implications for classification and therapy[J]. Nat Rev Clin Oncol, 2017, 14(7): 434-452. |
| 4 | LIN S J, XU H S, ZHANG A K, et al. Prognosis analysis and validation of m6A signature and tumor immune microenvironment in glioma[J]. Front Oncol, 2020, 10: 541401. |
| 5 | SUN C J, HU R Y, LI Z C, et al. An engineered abcb4 expression model reveals the central role of NF-κB in the regulation of drug resistance in zebrafish[J]. Drug Dev Res, 2022, 83(4): 927-939. |
| 6 | WANG Y H, ZHANG F C, XIONG N X, et al. Remodelling and treatment of the blood-brain barrier in glioma[J]. Cancer Manag Res, 2021, 13: 4217-4232. |
| 7 | CAO Y, ZHANG S, LV Z, et al. An intelligent nanoplatform for orthotopic glioblastoma therapy by nonferrous ferroptosis[J]. Adv Funct Mater, 2022, 32(51): 2209227. |
| 8 | YIN N, WANG Y, LIU Y, et al. A cholesterol metabolic regulated hydrogen-bonded organic framework (HOF)-based biotuner for antibody non-dependent immunotherapy tailored for glioblastoma[J]. Adv Mater, 2023: 2303567. |
| 9 | CHARLES N A, HOLLAND E C, GILBERTSON R, et al. The brain tumor microenvironment[J]. Glia, 2012, 60(3): 502-514. |
| 10 | HEGI M E, DISERENS A C, GODARD S, et al. Clinical trial substantiates the predictive value of O-6-methylguanine-DNA methyltransferase promoter methylation in glioblastoma patients treated with temozolomide[J]. Clin Cancer Res, 2004, 10(6): 1871-1874. |
| 11 | OZDEMIR-KAYNAK E, QUTUB A A, YESIL-CELIKTAS O. Advances in glioblastoma multiforme treatment: new models for nanoparticle therapy[J]. Front Physiol, 2018, 9: 170. |
| 12 | GANZ J C. Low grade gliomas[J]. Prog Brain Res, 2022, 268(1): 271-277. |
| 13 | LIU Y, ZHEN W, JIN L, et al. All-in-one theranostic nanoagent with enhanced reactive oxygen species generation and modulating tumor microenvironment ability for effective tumor eradication[J]. ACS Nano, 2018, 12(5): 4886-4893. |
| 14 | LIU Y W, ZHAO K L, REN Y B, et al. Highly plasticized lanthanide luminescence for information storage and encryption applications[J]. Adv Sci, 2022, 9(7): 2105108. |
| 15 | WAN S K, CONG W, SHAO B Q, et al. A library of thermotropic liquid crystals of inorganic nanoparticles and extraordinary performances based on their collective ordering[J]. Nano Today, 2021, 38: 101115. |
| 16 | QU Z B, SHEN J L, LI Q, et al. Near-IR emissive rare-earth nanoparticles for guided surgery[J]. Theranostics, 2020, 10(6): 2631-2644. |
| 17 | ZHAO M N, VAN STRATEN D, BROEKMAN M L D, et al. Nanocarrier-based drug combination therapy for glioblastoma[J]. Theranostics, 2020, 10(3): 1355-1372. |
| 18 | LIU Y L, AI K L, LIU J H, et al. A high-performance ytterbium-based nanoparticulate contrast agent for in vivo X-ray computed tomography imaging[J]. Angew Chem Int Ed, 2012, 51(6): 1437-1442. |
| 19 | VISWANATHAN S, KOVACS Z, GREEN K N, et al. Alternatives to gadolinium-based metal chelates for magnetic resonance imaging[J]. Chem Rev, 2010, 110(5): 2960-3018. |
| 20 | CAO Y, XU L J, KUANG Y, et al. Gadolinium-based nanoscale MRI contrast agents for tumor imaging[J]. J Mater Chem B, 2017, 5(19): 3431-3461. |
| 21 | ZHONG Y, DAI H. A mini-review on rare-earth down-conversion nanoparticles for NIR-Ⅱ imaging of biological systems[J]. Nano Res, 2020, 13(5): 1281-1294. |
| 22 | ESCUDERO A, BECERRO A I, CARRILLO-CARRION C, et al. Rare earth based nanostructured materials: synthesis, functionalization, properties and bioimaging and biosensing applications[J]. Nanophotonics, 2017, 6(5): 881-921. |
| 23 | LE DUC G, MILADI I, ALRIC C, et al. Toward an image-guided microbeam radiation therapy using gadolinium-based nanoparticles[J]. ACS Nano, 2011, 5(12): 9566-9574. |
| 24 | TEH D B L, BANSAL A, CHAI C, et al. A flexi-PEGDA upconversion implant for wireless brain photodynamic therapy[J]. Adv Mater, 2020, 32(29): 2001459. |
| 25 | GAI S L, LI C X, YANG P P, et al. Recent progress in rare earth micro/nanocrystals: soft chemical synthesis, luminescent properties, and biomedical applications [J]. Chem Rev, 2014, 114(4): 2343-2389. |
| 26 | AVRAM D, COJOCARU B, URDA A, et al. Pure and almost pure NIR emission of Tm and Tm,Yb-CeO2 under UV, X-ray and NIR up-conversion excitation: key roles of level selective antenna sensitization and charge-compensation[J]. Phys Chem Chem Phys, 2015, 17(46): 30988-30992. |
| 27 | NOREK M, PETERS J A. MRI contrast agents based on dysprosium or holmium[J]. Prog Nucl Magn Reson Spectrosc, 2011, 59(1): 64-82. |
| 28 | FRIES P H, BELORIZKY E. Electronic relaxation of paramagnetic metal ions and NMR relaxivity in solution: critical analysis of various approaches and application to a Gd(Ⅲ)-based contrast agent[J]. J Chem Phys, 2005, 123(12): 124510. |
| 29 | CHEN X F, SONG J B, CHEN X Y, et al. X-ray-activated nanosystems for theranostic applications[J]. Chem Soc Rev, 2019, 48(11): 3073-3101. |
| 30 | LUSIC H, GRINSTAFF M W. X-ray-computed tomography contrast agents[J]. Chem Rev, 2013, 113(3): 1641-1666. |
| 31 | MCDONALD M A, WATKIN K L. Small particulate gadolinium oxide and gadolinium oxide albumin microspheres as multimodal contrast and therapeutic agents[J]. Invest Radiol, 2003, 38(6): 305-310. |
| 32 | PARK J Y, BAEK M J, CHOI E S, et al. Paramagnetic ultrasmall gadolinium oxide nanoparticles as advanced T1 MRI contrast agent: account for large longitudinal relaxivity, optimal particle diameter, and in vivo T1 MR images[J]. ACS Nano, 2009, 3(11): 3663-3669. |
| 33 | FAUCHER L, TREMBLAY M, LAGUEUX J, et al. Rapid synthesis of pegylated ultrasmall gadolinium oxide nanoparticles for cell labeling and tracking with MRI[J]. ACS Appl Mater Interfaces, 2012, 4(9): 4506-4515. |
| 34 | GU W, SONG G, LI S, et al. Chlorotoxin-conjugated, PEGylated Gd2O3 nanoparticles as a glioma-specific magnetic resonance imaging contrast agent[J]. RSC Adv, 2014, 4(91): 50254-50260. |
| 35 | LI W, QIU J H, LI X L, et al. BBB pathophysiology-independent delivery of siRNA in traumatic brain injury[J]. Sci Adv, 2021, 7(1): eabd6889. |
| 36 | SHEN Z, LIU T, LI Y, et al. Fenton-reaction-acceleratable magnetic nanoparticles for ferroptosis therapy of orthotopic brain tumors[J]. ACS Nano, 2018, 12(11): 11355-11365. |
| 37 | SHAO C, LI S, GU W, et al. Multifunctional gadolinium-doped manganese carbonate nanoparticles for targeted MR/fluorescence imaging of tiny brain gliomas[J]. Anal Chem, 2015, 87(12): 6251-6257. |
| 38 | LI T, MURPHY S, KISELEV B, et al. A new interleukin-13 amino-coated gadolinium metallofullerene nanoparticle for targeted MRI detection of glioblastoma tumor cells[J]. J Am Chem Soc, 2015, 137(24): 7881-7888. |
| 39 | ZHANG H, WU Y, WANG J, et al. In vivo MR imaging of glioma recruitment of adoptive T-cells labeled with NaGdF4-TAT nanoprobes[J]. Small, 2018, 14(3): 1702951. |
| 40 | KENRY, DUAN Y, LIU B. Recent advances of optical imaging in the second near-infrared window[J]. Adv Mater, 2018, 30(47): 1802394. |
| 41 | CHEN S Y, MIAO H, JIANG X Y, et al. Starlike polymer brush-based ultrasmall nanoparticles with simultaneously improved NIR-Ⅱ fluorescence and blood circulation for efficient orthotopic glioblastoma imaging[J]. Biomaterials, 2021, 275: 120916. |
| 42 | ZHONG Y, MA Z, ZHU S, et al. Boosting the down-shifting luminescence of rare-earth nanocrystals for biological imaging beyond 1500 nm[J]. Nat Commun, 2017, 8: 737. |
| 43 | LV Z J, JIN L H, GAO W H, et al. Novel YOF-based theranostic agents with a cascade effect for NIR-Ⅱ fluorescence imaging and synergistic starvation/photodynamic therapy of orthotopic gliomas[J]. ACS Appl Mater Interfaces, 2022, 14(27): 30523-30532. |
| 44 | FAN Y, ZHANG F. A new generation of NIR-Ⅱ probes: lanthanide-based nanocrystals for bioimaging and biosensing[J]. Adv Opt Mater, 2019, 7(7): 1801417. |
| 45 | ZHU H, REN F, WANG T, et al. Targeted immunoimaging of tumor-associated macrophages in orthotopic glioblastoma by the NIR-Ⅱb nanoprobes[J]. Small, 2022, 18(30): 2202201. |
| 46 | WANG Z J, ZHANG M, CHI S Y, et al. Brain tumor cell membrane-coated lanthanide-doped nanoparticles for NIR-Ⅱb luminescence imaging and surgical navigation of glioma[J]. Adv Healthc Mater, 2022, 11(16): 2200521. |
| 47 | ZHANG X B, CHEN W W, XIE X Y, et al. Boosting luminance energy transfer efficiency in upconversion nanoparticles with an energy-concentrating zone[J]. Angew Chem Int Ed, 2019, 58(35): 12117-12122. |
| 48 | XUE B, WANG D, TU L P, et al. Ultrastrong absorption meets ultraweak absorption: unraveling the energy-dissipative routes for dye-sensitized upconversion luminescence[J]. J Phys Chem Lett, 2018, 9(16): 4625-4631 . |
| 49 | ZOU W Q, VISSER C, MADURO J A, et al. Broadband dye-sensitized upconversion of near-infrared light[J]. Nat Photonics, 2012, 6(8): 560-564. |
| 50 | REN F, LIU H, ZHANG H, et al. Engineering NIR-Ⅱb fluorescence of Er-based lanthanide nanoparticles for through-skull targeted imaging and imaging-guided surgery of orthotopic glioma[J]. Nano Today, 2020, 34: 100905. |
| 51 | PENG J J, XU W, TEOH C L, et al. High-efficiency in vitro and in vivo detection of Zn2+-dye-assembled upconversion nanoparticles[J]. J Am Chem Soc, 2015, 137(6): 2336-2342. |
| 52 | LIU Z, REN F, ZHANG H, et al. Boosting often overlooked long wavelength emissions of rare-earth nanoparticles for NIR-Ⅱ fluorescence imaging of orthotopic glioblastoma[J]. Biomaterials, 2019, 219: 119364. |
| 53 | LV Z, JIN L, CAO Y, et al. A nanotheranostic agent based on Nd3+-doped YVO4 with blood-brain-barrier permeability for NIR-Ⅱ fluorescence imaging/magnetic resonance imaging and boosted sonodynamic therapy of orthotopic glioma[J]. Light Sci Appl, 2022, 11(1): 116. |
| 54 | PAN D, ROESSL E, SCHLOMKA J P, et al. Computed tomography in color: nanoK-enhanced spectral CT molecular imaging[J]. Angew Chem Int Ed, 2010, 49(50): 9635-9639. |
| 55 | JIN X, FANG F, LIU J, et al. An ultrasmall and metabolizable PEGylated NaGdF4∶Dy nanoprobe for high-performance T1/T2-weighted MR and CT multimodal imaging[J]. Nanoscale, 2015, 7(38): 15680-15688. |
| 56 | HU Y, ZHOU Y Q, ZHAO N N, et al. Multifunctional pDNA-conjugated polycationic Au nanorod-coated Fe3O4 hierarchical nanocomposites for trimodal imaging and combined photothermal/gene therapy[J]. Small, 2016, 12(18): 2459-2468. |
| 57 | LIU Z, LI Z, LIU J, et al. Long-circulating Er3+-doped Yb2O3 up-conversion nanoparticle as an in vivo X-Ray CT imaging contrast agent[J]. Biomaterials, 2012, 33(28): 6748-6757. |
| 58 | NI D L, BU W B, ZHANG S J, et al. Single Ho3+-doped upconversion nanoparticles for high-performance T2-weighted brain tumor diagnosis and MR/UCL/CT multimodal imaging[J]. Adv Funct Mater, 2014, 24(42): 6613-6620. |
| 59 | LEE D E, KOO H, SUN I C, et al. Multifunctional nanoparticles for multimodal imaging and theragnosis[J]. Chem Soc Rev, 2012, 41(7): 2656-2672. |
| 60 | XUE D, LIU Y, JIN L, et al. Novel multifunctional theranostic nanoagents based on Ho3+ for CT/MRI dual-modality imaging-guided photothermal therapy[J]. Sci China Chem, 2021, 64(4): 558-564. |
| 61 | BOTTRILL M, NICHOLAS L K, LONG N J. Lanthanides in magnetic resonance imaging[J]. Chem Soc Rev, 2006, 35(6): 557-571. |
| 62 | JIN J, XU Z, ZHANG Y, et al. Upconversion nanoparticles conjugated with Gd3+-DOTA and RGD for targeted dual-modality imaging of brain tumor xenografts[J]. Adv Healthc Mater, 2013, 2(11): 1501-1512. |
| 63 | NI D, ZHANG J, BU W, et al. Dual-targeting upconversion nanoprobes across the blood-brain barrier for magnetic resonance/fluorescence imaging of intracranial glioblastoma[J]. ACS Nano, 2014, 8(2): 1231-1242. |
| 64 | LIU Y, LI L, GUO Q, et al. Novel Cs-based upconversion nanoparticles as dual-modal CT and UCL imaging agents for chemo-photothermal synergistic therapy[J]. Theranostics, 2016, 6(10): 1491-1505. |
| 65 | LUO Z, HU D, GAO D, et al. High-specificity in vivo tumor imaging using bioorthogonal NIR-Ⅱb nanoparticles[J]. Adv Mater, 2021, 33(49): 2102950. |
| 66 | DE BOER E, HARLAAR N J, TARUTTIS A, et al. Optical innovations in surgery[J]. Br J Surg, 2015, 102(2): E56-E72. |
| 67 | DEBIE P, HERNOT S. Emerging fluorescent molecular tracers to guide intra-operative surgical decision-making[J]. Front Pharmacol, 2019, 10: 510. |
| 68 | YANG L L, SAJJA H K, CAO Z H, et al. uPAR-targeted optical imaging contrasts as theranostic agents for tumor margin detection[J]. Theranostics, 2014, 4(1): 106-118. |
| 69 | KUHNT D, BECKER A, GANSLANDT O, et al. Correlation of the extent of tumor volume resection and patient survival in surgery of glioblastoma multiforme with high-field intraoperative MRI guidance[J]. Neuro-Oncology, 2011, 13(12): 1339-1348. |
| 70 | CHI C, DU Y, YE J, et al. Intraoperative imaging-guided cancer surgery: from current fluorescence molecular imaging methods to future multi-modality imaging technology[J]. Theranostics, 2014, 4(11): 1072-1084. |
| 71 | GU K, XU Y, LI H, et al. Real-time tracking and in vivo visualization of β-galactosidase activity in colorectal tumor with a ratiometric near-infrared fluorescent probe[J]. J Am Chem Soc, 2016, 138(16): 5334-5340. |
| 72 | WANG C, FAN W, ZHANG Z, et al. Advanced nanotechnology leading the way to multimodal imaging-guided precision surgical therapy[J]. Adv Mater, 2019, 31(49): 1904329. |
| 73 | VAHRMEIJER A L, HUTTEMAN M, VAN DER VORST J R, et al. Image-guided cancer surgery using near-infrared fluorescence[J]. Nat Rev Clin Oncol, 2013, 10(9): 507-518. |
| 74 | HONG G, ANTARIS A L, DAI H. Near-infrared fluorophores for biomedical imaging[J]. Nat Biomed Eng, 2017, 1(1): 0010. |
| 75 | HE S, SONG J, QU J, et al. Crucial breakthrough of second near-infrared biological window fluorophores: design and synthesis toward multimodal imaging and theranostics[J]. Chem Soc Rev, 2018, 47(12): 4258-4278. |
| 76 | YIN N, WANG Y, HUANG Y, et al. A biodegradable nanocapsule for through-skull NIR-Ⅱ fluorescence imaging/magnetic resonance imaging and selectively enhanced radio-chemotherapy for orthotopic glioma[J]. Nano Today, 2022, 46: 101619. |
| 77 | ZHANG H, WANG T T, QIU W B, et al. Monitoring the opening and recovery of the blood-brain barrier with noninvasive molecular imaging by biodegradable ultrasmall Cu2- xSe nanoparticles[J]. Nano Lett, 2018, 18(8): 4985-4992. |
| 78 | DELANEY G, JACOB S, FEATHERSTONE C, et al. The role of radiotherapy in cancer treatment-estimating optimal utilization from a review of evidence-based clinical guidelines[J]. Cancer, 2005, 104(6): 1129-1137. |
| 79 | LIU Y, ZHEN W, WANG Y, et al. Na2S2O8 nanoparticles trigger antitumor immunotherapy through reactive oxygen species storm and surge of tumor osmolarity[J]. J Am Chem Soc, 2020, 142(52): 21751-21757. |
| 80 | MISAWA M, TAKAHASHI J. Generation of reactive oxygen species induced by gold nanoparticles under X-ray and UV irradiations[J]. Nanomedicine, 2011, 7(5): 604-614. |
| 81 | KOBAYASHI K, USAMI N, PORCEL E, et al. Enhancement of radiation effect by heavy elements[J]. Mutat Res Rev Mutat Res, 2010, 704(1/2/3): 123-131. |
| 82 | MOWAT P, MIGNOT A, RIMA W, et al. In vitro radiosensitizing effects of ultrasmall gadolinium based particles on tumour cells[J]. J Nanosci Nanotechnol, 2011, 11(9): 7833-7839. |
| 83 | LE DUC G, ROUX S, PARUTA-TUAREZ A, et al. Advantages of gadolinium based ultrasmall nanoparticles vs molecular gadolinium chelates for radiotherapy guided by MRI for glioma treatment[J]. Cancer Nanotechnol, 2014, 5(1): 4. |
| 84 | BRIGGS A, CORDE S, OKTARIA S, et al. Cerium oxide nanoparticles: influence of the high-Z component revealed on radioresistant 9L cell survival under X-ray irradiation[J]. Nanomedicine, 2013, 9(7): 1098-1105. |
| 85 | BULIN A L, BROEKGAARDEN M, CHAPUT F, et al. Radiation dose-enhancement is a potent radiotherapeutic effect of rare-earth composite nanoscintillators in preclinical models of glioblastoma[J]. Adv Sci, 2020, 7(20): 2001675. |
| 86 | YANG Z, FAN W, ZOU J, et al. Precision cancer theranostic platform by in situ polymerization in perylene diimide-hybridized hollow mesoporous organosilica nanoparticles[J]. J Am Chem Soc, 2019, 141(37): 14687-14698. |
| 87 | SUN W, LUO L, FENG Y, et al. Aggregation-induced emission gold clustoluminogens for enhanced low-dose X-ray-induced photodynamic therapy[J]. Angew Chem Int Ed, 2020, 59(25): 9914-9921. |
| 88 | LV Z, CAO Y, XUE D, et al. A multiphoton transition activated iron based metal organic framework for synergistic therapy of photodynamic therapy/chemodynamic therapy/chemotherapy for orthotopic gliomas[J]. J Mater Chem B, 2023, 11(5): 1100-1107. |
| 89 | PARK Y, KIM H M, KIM J H, et al. Theranostic probe based on lanthanide-doped nanoparticles for simultaneous in vivo dual-modal imaging and photodynamic therapy[J]. Adv Mater, 2012, 24(42): 5755-5761. |
| 90 | WANG P, WANG C, LU L, et al. Kinetics-mediate fabrication of multi-model bioimaging lanthanide nanoplates with controllable surface roughness for blood brain barrier transportation[J]. Biomaterials, 2017, 141: 223-232. |
| 91 | ZHANG M, WANG W, MOHAMMADNIAEI M, et al. Upregulating aggregation-induced-emission nanoparticles with blood-tumor-barrier permeability for precise photothermal eradication of brain tumors and induction of local immune responses[J]. Adv Mater, 2021, 33(22): 2008802. |
| 92 | TSAI Y C, VIJAYARAGHAVAN P, CHIANG W H, et al. Targeted delivery of functionalized upconversion nanoparticles for externally triggered photothermal/photodynamic therapies of brain glioblastoma[J]. Theranostics, 2018, 8(5): 1435-1448. |
| 93 | YIN N, WANG Y, HUANG Y, et al. Modulating nanozyme-based nanomachines via microenvironmental feedback for differential photothermal therapy of orthotopic gliomas[J]. Adv Sci, 2023, 10(3): 2204937. |
| 94 | FAN W, BU W, ZHANG Z, et al. X-ray radiation-controlled NO-release for on-demand depth-independent hypoxic radiosensitization[J]. Angew Chem Int Ed, 2015, 54(47): 14026-14030. |
| 95 | QIANGLAN L, TONG L, MIN X, et al. SO2 prodrug doped nanorattles with extra-high drug payload for ldquocollusion inside and outsiderdquo photothermal/pH triggered-gas therapy[J]. Biomaterials, 2020, 257: 120236. |
| 96 | SUN R, LIU X C, LI G Z, et al. Photoactivated H2 nanogenerator for enhanced chemotherapy of bladder cancer[J]. ACS Nano, 2020, 14(7): 8135-8148. |
| 97 | CHUNG M F, LIU H Y, LIN K J, et al. A pH-responsive carrier system that generates NO bubbles to trigger drug release and reverse P-glycoprotein-mediated multidrug resistance[J]. Angew Chem Int Ed, 2015, 54(34): 9890- 9893. |
| 98 | DONG H, DU S R, ZHENG X Y, et al. Lanthanide nanoparticles: from design toward bioimaging and therapy[J]. Chem Rev, 2015, 115(19): 10725-10815. |
| 99 | NACZYNSKI D J, TAN M C, ZEVON M, et al. Rare-earth-doped biological composites as in vivo shortwave infrared reporters[J]. Nat Commun, 2013, 4: 2199. |
| 100 | LIU Z, YUN B, HAN Y, et al. Dye-sensitized rare earth nanoparticles with up/down conversion luminescence for on-demand gas therapy of glioblastoma guided by NIR-Ⅱ fluorescence imaging[J]. Adv Healthc Mater., 2022, 11(3): 2102042. |
| 101 | YUN B F, GU Z P, LIU Z, et al. Reducing chemo-/radioresistance to boost the therapeutic efficacy against temozolomide-resistant glioblastoma[J]. ACS Appl Mater Interfaces, 2022, 14(34): 38617-38630. |
| [1] | Xin-Yu ZHAO, Zuo-Jia QIN, Xiao-Bing ZHANG, Lin YUAN. Research Progress in Activatable NIR-Ⅱ Small Molecule Fluorescent Probes [J]. Chinese Journal of Applied Chemistry, 2024, 41(1): 39-59. |
| [2] | Hui DU, Chen-Yang YAO, Hao PENG, Bo JIANG, Shun-Xiang LI, Jun-Lie YAO, Fang ZHENG, Fang YANG, Ai-Guo WU. Applications of Transition Metal⁃doped Iron⁃based Nanoparticles in Biomedicine [J]. Chinese Journal of Applied Chemistry, 2022, 39(3): 391-406. |
| [3] | HUANG Zike, LIU Chao, FU Qiangqiang, LI Jin, ZOU Jianmei, XIE Sitao, QIU Liping. Aptamer-based Fluorescence Probe for Bioanalysis and Bioimaging [J]. Chinese Journal of Applied Chemistry, 2018, 35(1): 28-39. |
| [4] | Jia ZHOU, Yun NI, Chengwu ZHANG, Xinghan QIU, Yanfei ZHAO, Lei BAI, Gaobin ZHANG, Lin LI. Design and Synthesis of Pyrimidine Based Two-Photon Fluorescence Probe and Its Application in Bioimaging [J]. Chinese Journal of Applied Chemistry, 2017, 34(12): 1450-1456. |
| [5] | Jia ZHOU, Yun NI, Chengwu ZHANG, Xinghan QIU, Yanfei ZHAO, Lei BAI, Gaobin ZHANG, Lin LI. Design and Synthesis of Pyrimidine Based Two-Photon Fluorescence Probe and Its Application in Bioimaging [J]. Chinese Journal of Applied Chemistry, 2017, 34(12): 0-0. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
