
Chinese Journal of Applied Chemistry ›› 2024, Vol. 41 ›› Issue (1): 39-59.DOI: 10.19894/j.issn.1000-0518.230264
• Review • Previous Articles Next Articles
Research Progress in Activatable NIR-Ⅱ Small Molecule Fluorescent Probes
Xin-Yu ZHAO, Zuo-Jia QIN, Xiao-Bing ZHANG, Lin YUAN( )
)
- State Key Laboratory of Chemo/Biosensing and Chemometrics,College of Chemistry and Chemical Engineering,Hunan University,Changsha 410082,China
-
Received:2023-09-01Accepted:2023-11-17Published:2024-01-01Online:2024-01-30 -
Contact:Lin YUAN -
About author:lyuan@hnu.edu.cn
-
Supported by:the National Natural Science Foundation of China(22074036)
CLC Number:
Cite this article
Xin-Yu ZHAO, Zuo-Jia QIN, Xiao-Bing ZHANG, Lin YUAN. Research Progress in Activatable NIR-Ⅱ Small Molecule Fluorescent Probes[J]. Chinese Journal of Applied Chemistry, 2024, 41(1): 39-59.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.230264
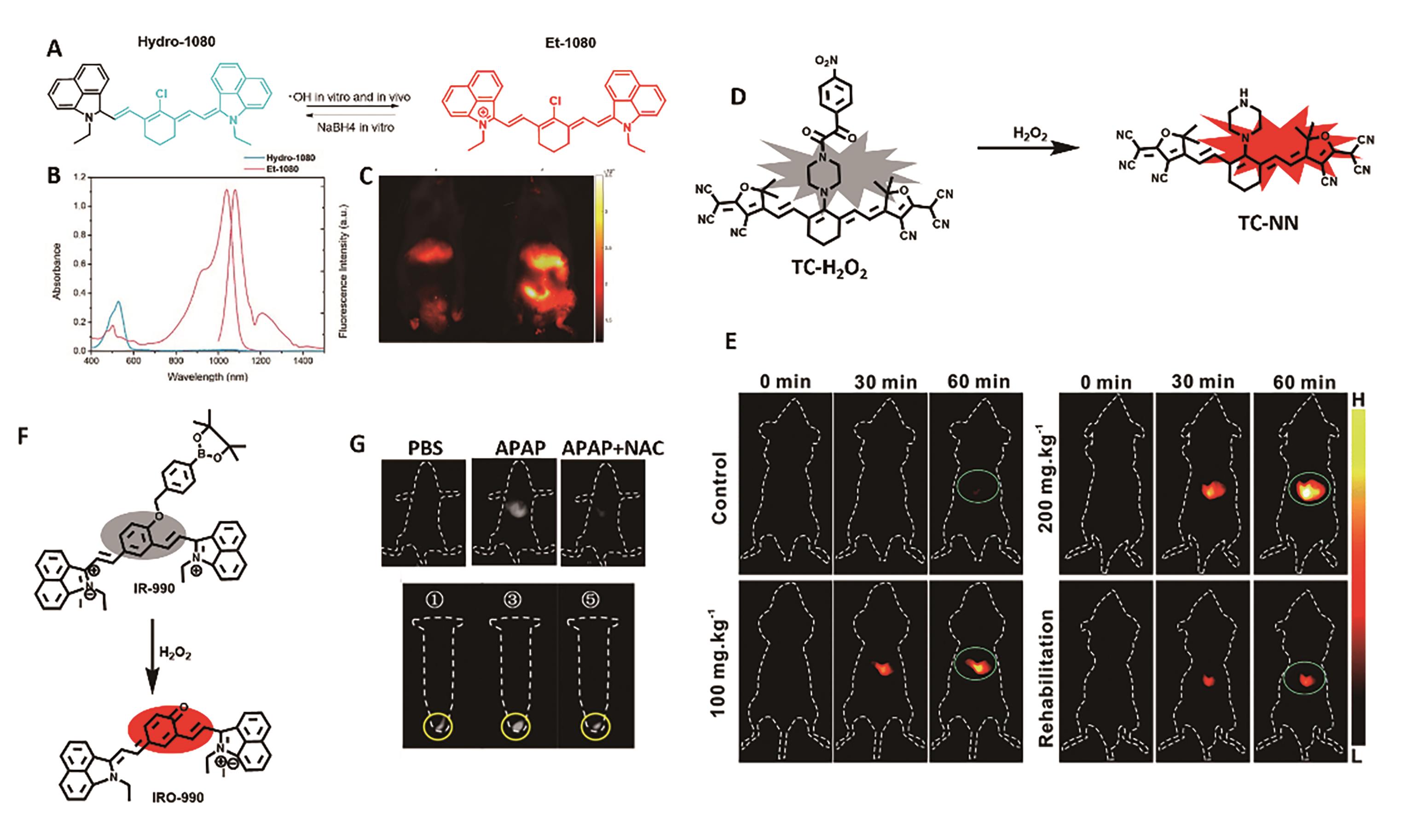
Fig.1 Representative probe for liver inflammation imaging. (A) Structure and response mechanism of Hydro-1080[23]. (B) Absorption and fluorescence spectra of Hydro-1080 and Et-1080 in DMSO[23]. (C) NIR-Ⅱ fluorescence images of mice liver injury[23]. (D) Structure and response mechanism of probe TC-H2O2[24]. (E) MOST and fluorescence imaging of mice liver injury model[24]. (F) Structure and response mechanism of IR-990[26]. (G) NIR-Ⅱ fluorescence imaging of H2O2 in HepG2 cells and APAP-induced mice liver injury models[26]
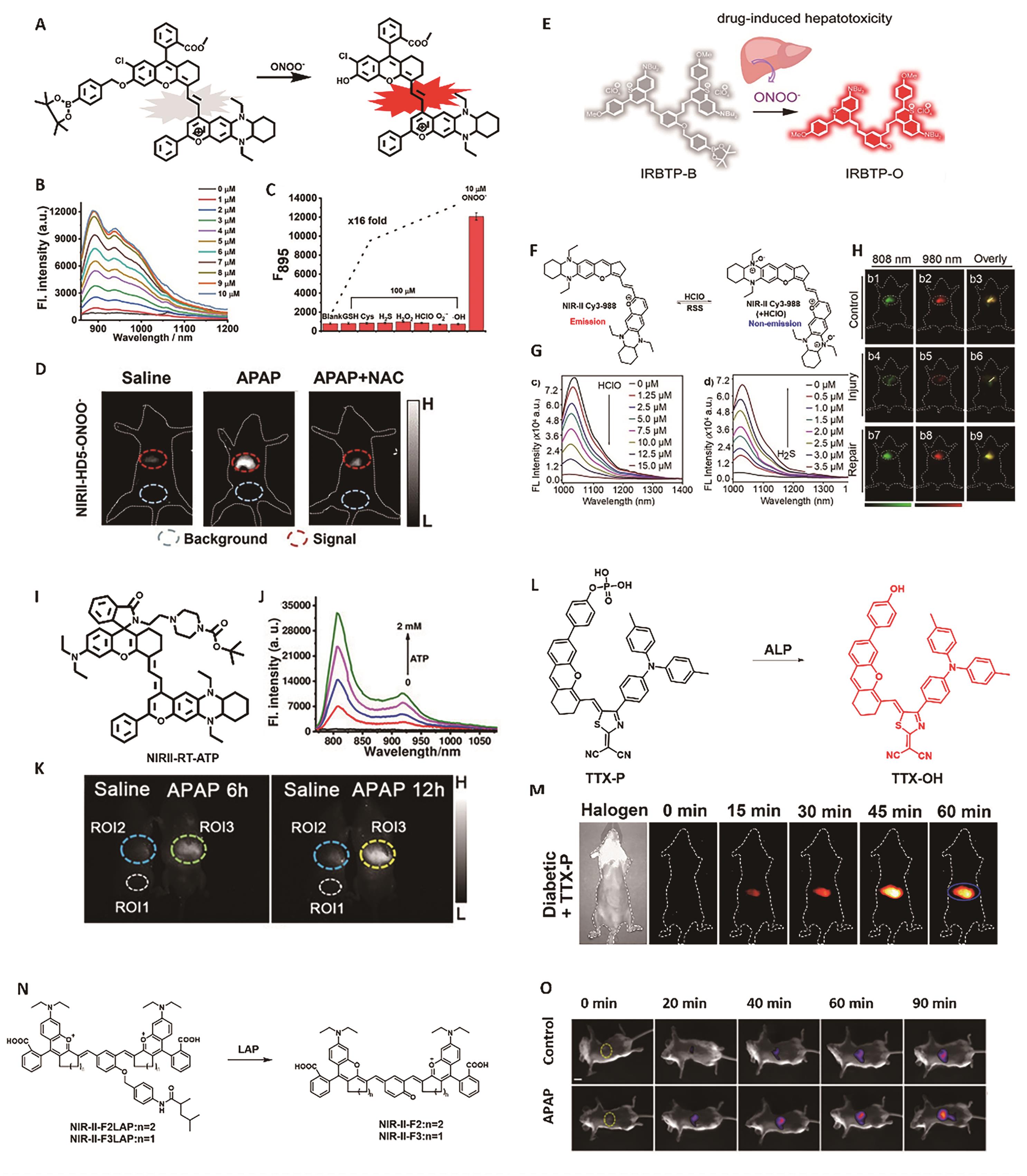
Fig.2 Representative probes for liver inflammation imaging. (A) Structure and response mechanism of NIRII-HD5-ONOO-[30]. (B) NIR-Ⅱ fluorescence spectra of NIRII-HD5-ONOO- (5 μmol/L) after reaction with various concentrations of ONOO-. (C) NIR-Ⅱ fluorescence response of NIRII-HD5-ONOO- (5 μmol/L) toward different analytes[30]. (D) NIR-Ⅱ fluorescence imaging of liver injury after APAP treatment[30]. (E) Structure and response mechanism of IRBTP-B[31]. (F) Response mechanism of probe NIR-Ⅱ Cy3-988[33]. (G) Fluorescence intensity changes of NIR-Ⅱ Cy3-988 (5 μmol/L) after reaction with various concentrations of HClO (0~15 μmol/L) and H2S (0~3.5 μmol/L)[33]. (H) NIR-Ⅱ fluorescence imaging of mice liver injury and repair[33]. (I) Structure of probe NIRII-RT-ATP[34]. (J) NIR-Ⅱ fluorescence spectra of NIRII-RT-ATP (5 μmol/L) after reaction with various concentrations of ATP[34]. (K) Fluorescence imaging of liver injury after APAP treatment[34]. (L) Structure and response mechanism of TTX-P[35]. (M) NIR-Ⅱ fluorescence imaging of liver injury[35]. (N) Structure and response mechanism of NIR-Ⅱ-LAP[36]. (O) Fluorescence imaging of liver injury after APAP treatment[36]

Fig.3 Representative probes for imaging kidney inflammation. (A) Structure and response mechanism of BOD-Ⅱ-NAG[39]. (B) NIR-Ⅱ fluorescence images of living mice after injection of BOD-Ⅱ-NAG-NP (16 μmol/kg) at different time[39]. (C) The fluorescence intensity in different groups after intravenous injection of BOD-Ⅱ-NAG-NP[39]. (D) Changes in KIM-1, NGAL, NAG, Cyst C, sCr and BUN in living mice after different treatment[39]. (E) Structure and design strategy of HP-N dyes and HP-H2O2[40] (F) NIR-Ⅱ fluorescence spectra of HP-H2O2 after reaction with various concentrations of H2O2(10 μmol/L)[40]. (G) Fluorescence intensities of the probe HP-H2O2 at different time[40]. (H) NIR-Ⅱ fluorescence images of control mice and AKI mice[40]

Fig.4 Representative probe for imaging cystitis. (A) Structure and photophysical properties of Chrodol[42]. (B) Response mechanism of dual-activatable probe PN910[42]. (C) Absorption and fluorescence spectra of PN910 and Chrodol-3[42]. (D) Fluorescence intensity upon addition of H2O2 (left) and ONOO- (right)[42]. (E) Structure and response mechanism of BTPE-NO2@F127 probe[43]. (F) MOST images (left) and NIR-Ⅱ fluorescence images (right) of the control and the interstitial cystitis model[43]
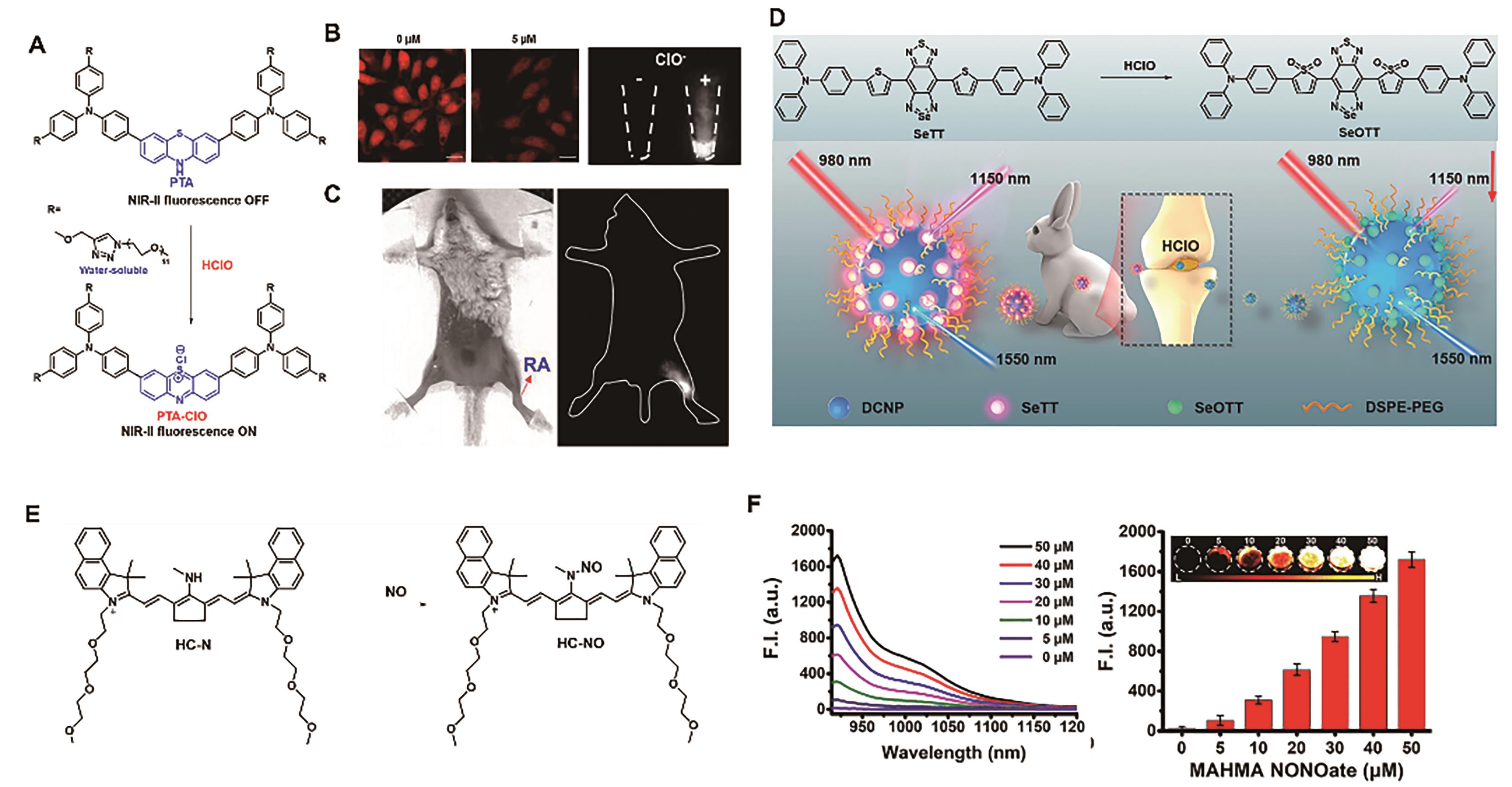
Fig.5 Representative probes for imaging joint inflammation. (A) Structure and response mechanism of probe PTA[46]. (B) Fluorescence images of exogenous and endogenous HClO in HeLa cell[46]. (C) Fluorescence imaging of HClO in RA mouse model[46]. (D) Structure and response mechanism of DNCP@SeTT[47]. (E) Structure and response mechanism of probe HC-N[48]. (F) NIR-Ⅱ fluorescence spectra (left) and fluorescence intensity at 923 nm (right) of HC-N (5 μmol/L) after reaction with various concentrations of NO[48]
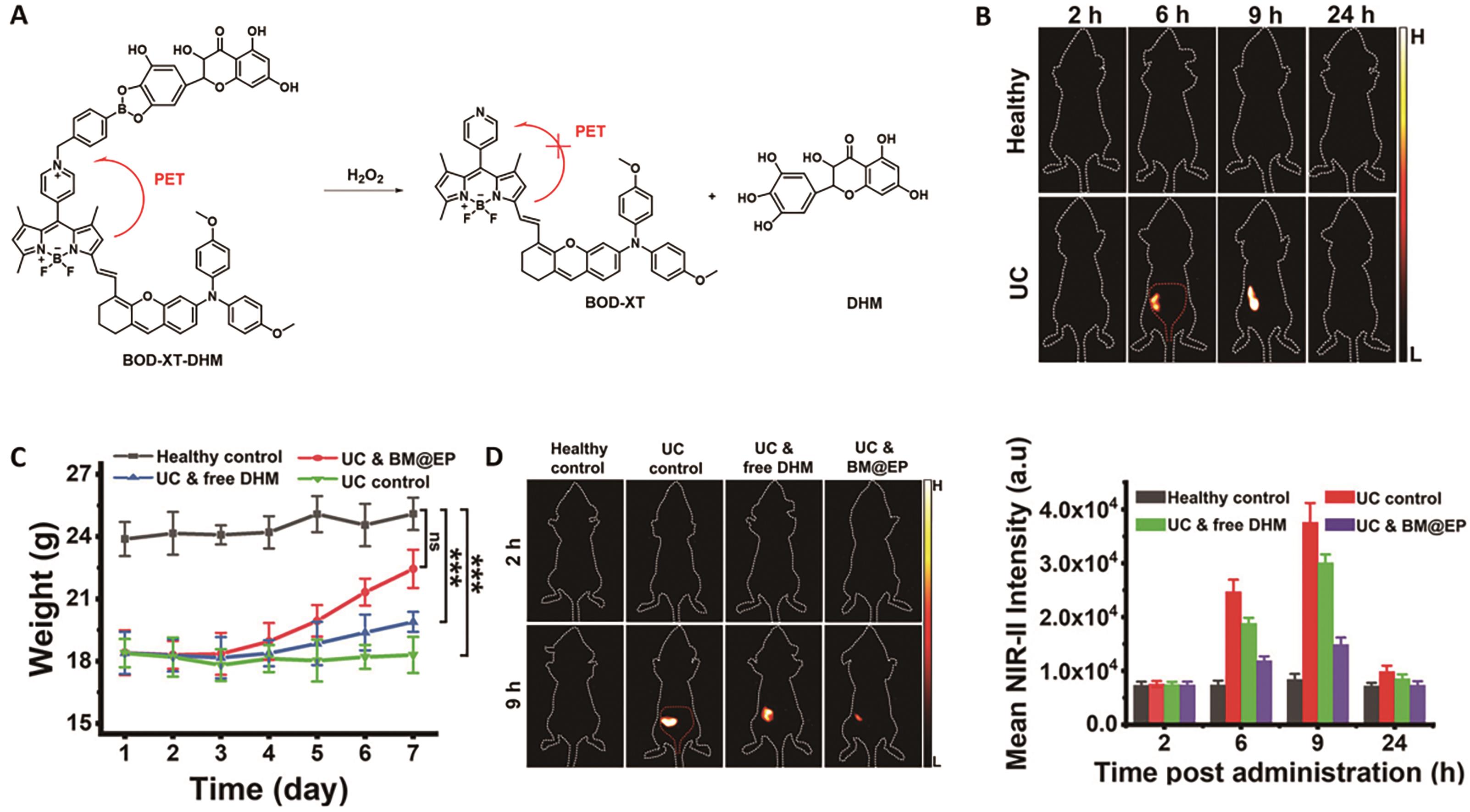
Fig.6 Representative probe for imaging ulcerative colitis[50]. (A) Structure and response mechanism of probe BOD-XT-DHM. (B) NIR-Ⅱ fluorescent images of mice after oral administration of BM@EP (50 mg/kg). (C) Changes in body mass of different groups' mice. (D) NIR-Ⅱ fluorescence images of mice (left) and average fluorescent intensities at mice′s colon region (right) after oral dosing of BM@EP (43.6 mg/kg)
| Liver injury | ?OH | Hydro-1080 | |||
| H2O2 | TC-H2O2 | ||||
| IR-990 | 0.59 μ | ||||
| BHC-Lut | |||||
| NIRII-HD5-ONOO- | |||||
| IRBTP-B | 55.9 n | ||||
| HClO | NIR Ⅱ Cy3-988 | ||||
| NIRII-RT-ATP | |||||
| TTX-P | |||||
| NIR-Ⅱ-F2LAP | 0.063 U/L | ||||
| Kidney injury | NAG | BOD-II-NAG-NP | |||
| HP-H2O2 | |||||
| Cystitis | ROS/RNS and base | PN910 | |||
| H2O2 | BTPE-NO2 | ||||
| Joint inflammation | HClO | ||||
| HC-N | |||||
| Colitis | pH/ROS | BM@EP |
Table 1 NIR-Ⅱ activatable probes for inflammation models
| Liver injury | ?OH | Hydro-1080 | |||
| H2O2 | TC-H2O2 | ||||
| IR-990 | 0.59 μ | ||||
| BHC-Lut | |||||
| NIRII-HD5-ONOO- | |||||
| IRBTP-B | 55.9 n | ||||
| HClO | NIR Ⅱ Cy3-988 | ||||
| NIRII-RT-ATP | |||||
| TTX-P | |||||
| NIR-Ⅱ-F2LAP | 0.063 U/L | ||||
| Kidney injury | NAG | BOD-II-NAG-NP | |||
| HP-H2O2 | |||||
| Cystitis | ROS/RNS and base | PN910 | |||
| H2O2 | BTPE-NO2 | ||||
| Joint inflammation | HClO | ||||
| HC-N | |||||
| Colitis | pH/ROS | BM@EP |

Fig.7 Representative probe for liver cancer imaging[52]. (A) Structure and response mechanism of probe BH-NO2@BSA. (B) 3D MOST imaging of mouse liver tumors (middle) and NIR-Ⅱ fluorescence image-guided resection of liver tumors (right)
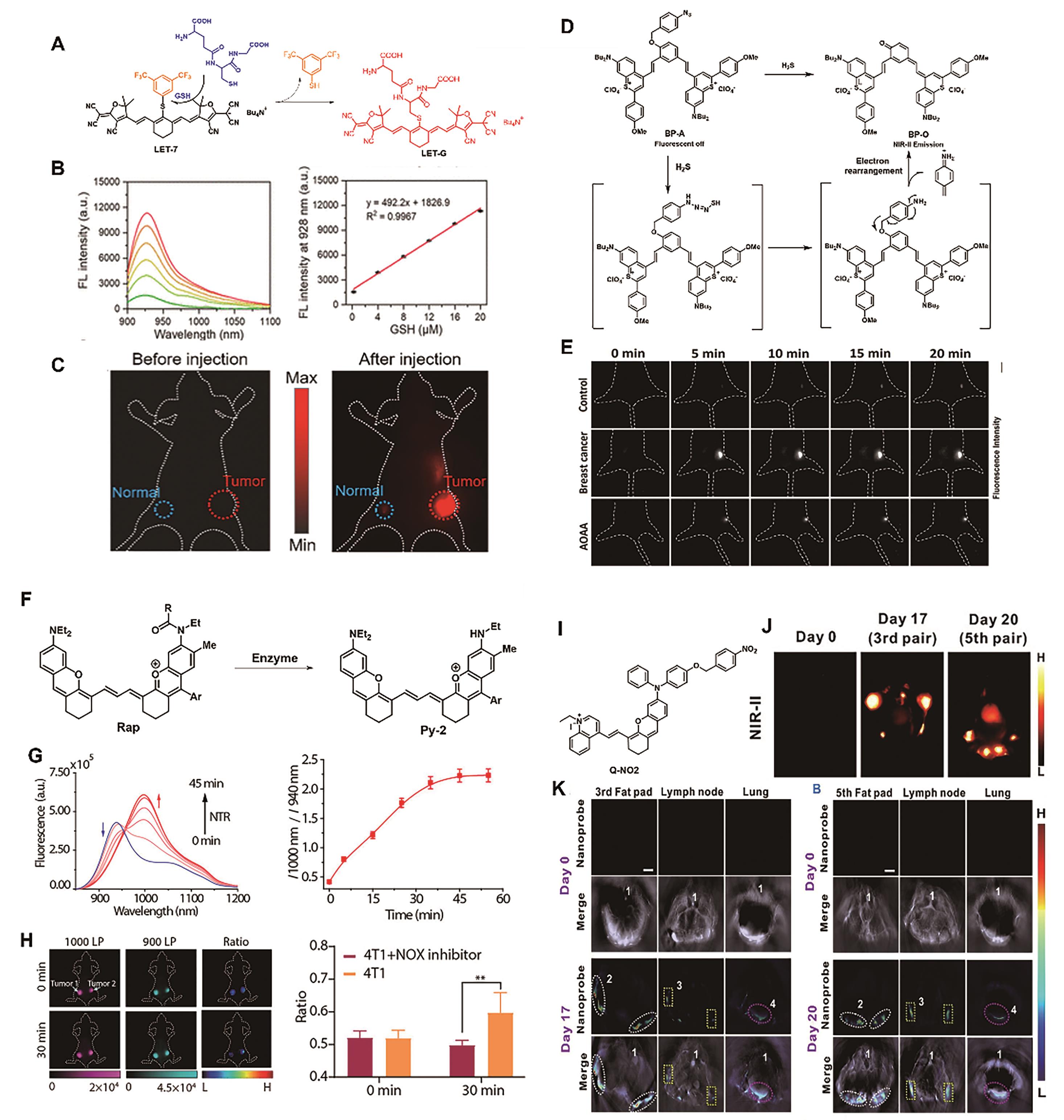
Fig.8 Representative probes for breast cancer imaging. (A) Structure and response mechanism of probe LET-7[53]. (B) NIR-Ⅱ fluorescence spectra of probe LET-7 (2 μmol/L) after reaction with various concentrations of GSH (left) and corresponding linear relationships (right) [53]. (C) NIR-Ⅱ fluorescence images of mice[53]. (D) Structure and response mechanism of probe BP-A[55]. (E) Different groups′ mice′s NIR-Ⅱ fluorescence images after intratumoral injection of BP-A (200 μmol/L)[55]. (F) Design strategy of the ratiometric probe Rap-N[57]. (G) Time dependence of fluorescence spectra (left) and I1000 nm/I940 nm ratio of Rap-N (right) incubation with NTR (10 μg/mL)[57]. (H) Ratiometric fluorescence images (left) and signal intensity (right) of mice bearing breast tumors (the left tumor injected with the NTR inhibitor dicoumarol)[57]. (I) Structure of the molecular probe Q-NO2 [59]. (J)NIR-Ⅱ images and (K) MOST images of regional and distant breast cancer metastases in mice[59]

Fig.9 Representative probes for lung cancer imaging. (A) Structure and response mechanism of enzyme-activated probes[61]. (B) Time dependence of fluorescence spectra of Rap-N (20 mg/mL) incubation with NTR[61]. (C) Different groups' mice's NIR-Ⅱ fluorescence images after injection of the probe NTR-InD (30 nmol)[61]. (D) Structure and response mechanism of probe RHC-NO2[62]. (E) NIR-Ⅱ fluorescence spectra of probe RHC-NO2 (10 μmol/L) after reaction with various concentrations of NTR[62]. (F) NIR-Ⅱ images of lung tumor-bearing mice after tail vein injection of RHC-NO2 (500 μmol/L)[62]
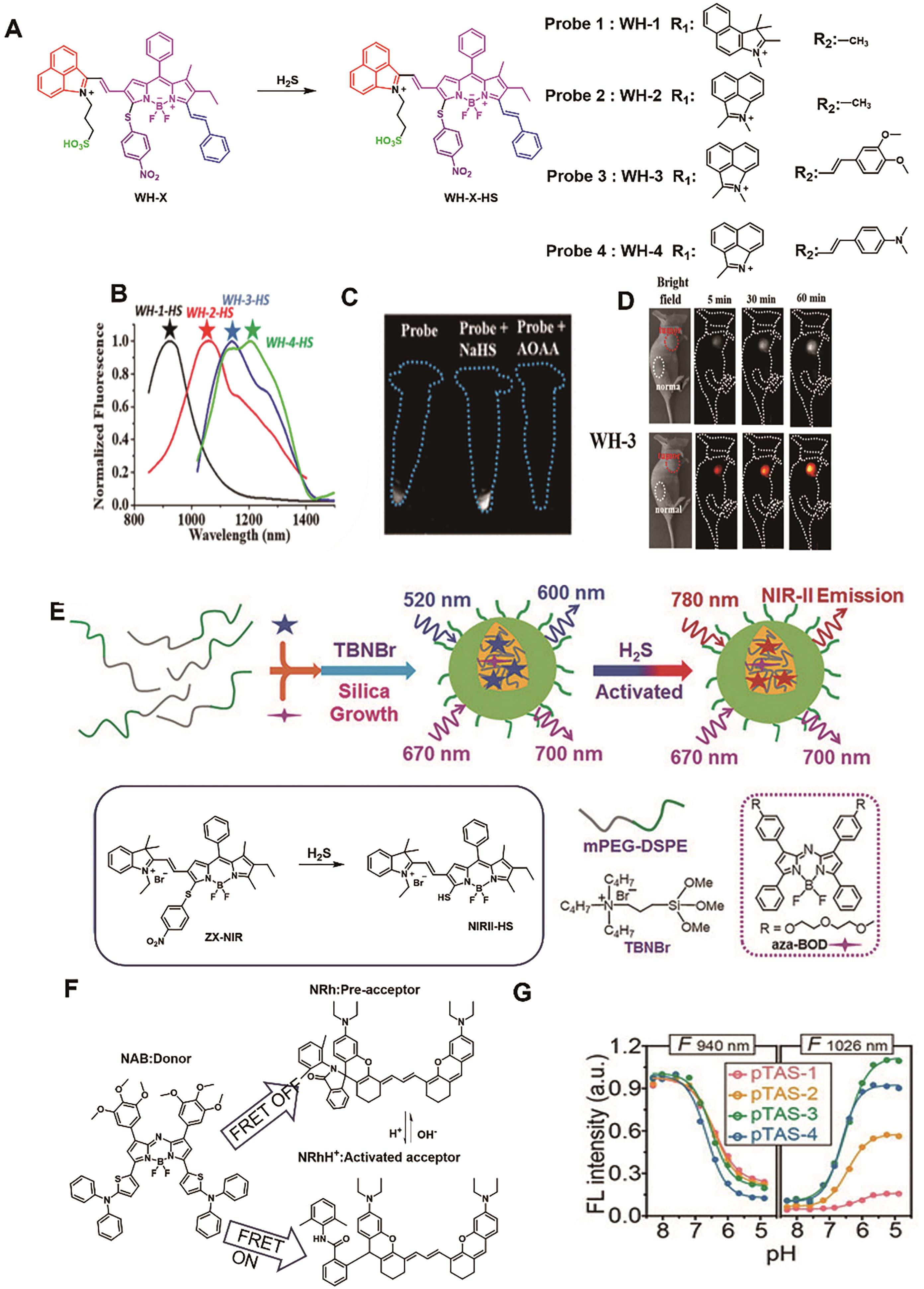
Fig.10 Representative probes for colon cancer imaging. (A) Structure and response mechanism of WH-X probes[64]. (B) Fluorescence spectra of WH-X probe[64]. (C) Fluorescence images of different treated cells with probe WH-3 [64]. (D) NIR-Ⅱ fluorescence images of mice. after injection of probe WH-3 (20 μL, 1 mmol/L)[64]. (E) Structure and response mechanism of NIRII@Si[65]. (F) The structure of FRET donor NAB and acceptor NRh[67]. (G) Fluorescence intensity at 940 and 1026 nm of probe pTAS as a function of pH[67]

Fig.11 Representative probes for imaging ovarian cancer. (A) Structure and response mechanism of BOD-M-βGal[70]. (B) NIR-Ⅱ fluorescence images of mice after injection of probe WH-3 (30 nmol)[70]. (C) Structure of Flavchromenes[71]. (D) Response mechanism of Flavchrom-4 probe[71]. (E) Absorption and fluorescence spectra of Flavchrom-4 after incubation with β-Gal[71]. (F) MOST images (left) and NIR-Ⅱ fluorescence images (right) of the ovarian cancer model after injection of Flavchrom-4[71]
| BH-NO2 | |||||
| LET-7 | |||||
| BP-A | |||||
| Rap-N | |||||
| NP-Q-NO2 | |||||
| NTR-InD | |||||
| RHC-NO2 | |||||
| Colorectal cancer | WH-3 | ||||
| NIR-Ⅱ@Si | 780/900 | 37 n | |||
| pTAS | |||||
| Ovarian cancers | βGal | BOD-M-βGal | |||
| Flavchrom-4 | 0.037 U/m |
Table 2 NIR-Ⅱ activatable probes for cancer models
| BH-NO2 | |||||
| LET-7 | |||||
| BP-A | |||||
| Rap-N | |||||
| NP-Q-NO2 | |||||
| NTR-InD | |||||
| RHC-NO2 | |||||
| Colorectal cancer | WH-3 | ||||
| NIR-Ⅱ@Si | 780/900 | 37 n | |||
| pTAS | |||||
| Ovarian cancers | βGal | BOD-M-βGal | |||
| Flavchrom-4 | 0.037 U/m |
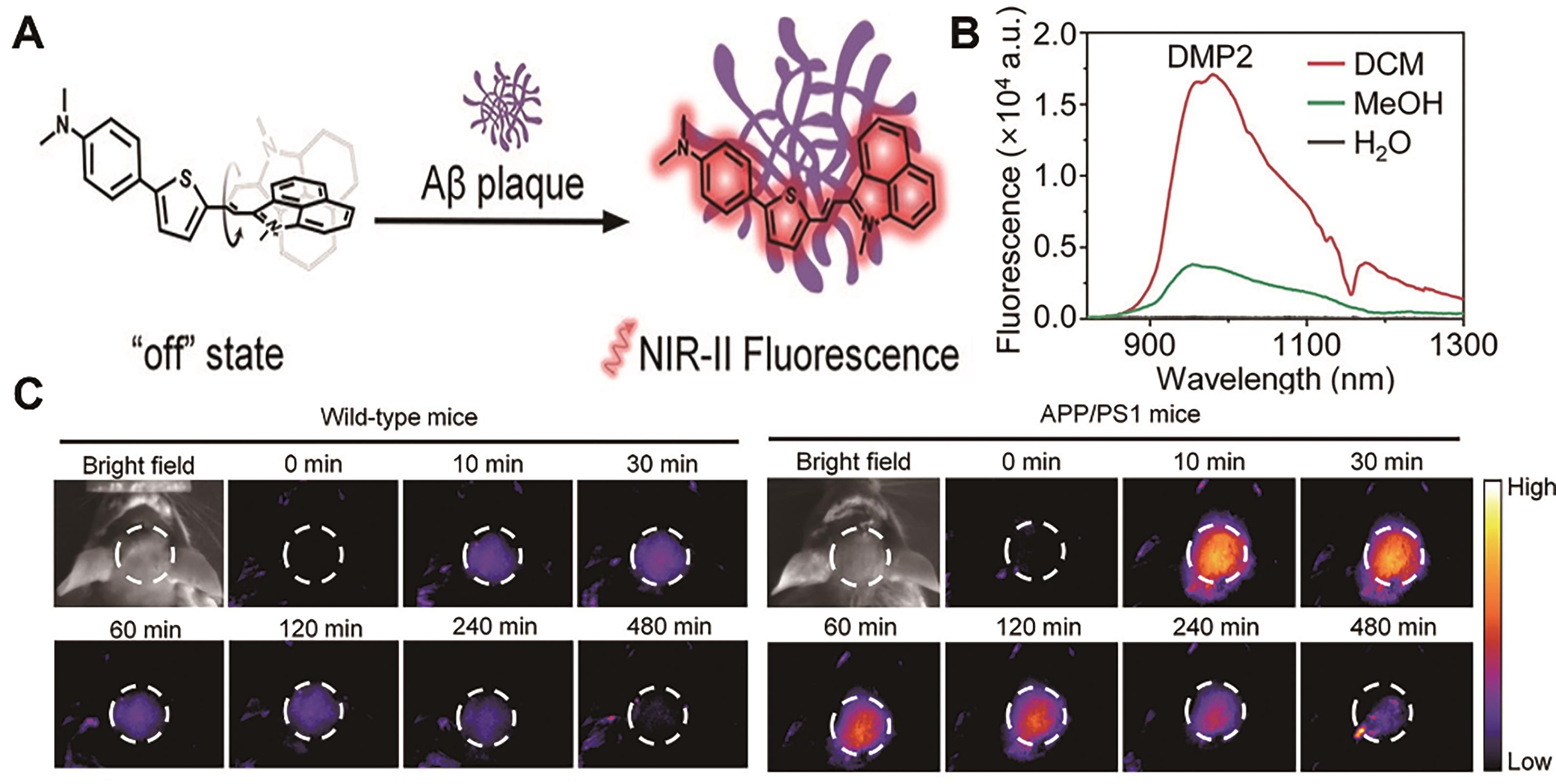
Fig.12 Representative probes for imaging Alzheimer′s disease[73]. (A) Structure and response mechanism of DMP2. (B) Fluorescence spectra of DMP2 in different solvents. (C) NIR-Ⅱ fluorescence imaging of the brain of healthy mice (left) and AD-model mice (right) after injection of probe DMP2 (2 mg/kg)
| 1 | HONG G, ANTARIS A L, DAI H. Near-infrared fluorophores for biomedical imaging[J]. Nat Biomed Eng, 2017, 1(1): 0010. |
| 2 | ANTARIS A L, CHEN H, CHENG K, et al. A small-molecule dye for NIR-Ⅱ imaging[J]. Nat Mater, 2015, 15(2): 235-242. |
| 3 | DIAO S, HONG G, ANTARIS A L, et al. Biological imaging without autofluorescence in the second near-infrared region[J]. Nano Res, 2015, 8(9): 3027-3034. |
| 4 | LI C, CHEN G, ZHANG Y, et al. Advanced fluorescence imaging technology in the near-infrared-Ⅱ window for biomedical applications[J]. J Am Chem Soc, 2020, 142(35): 14789-14804. |
| 5 | LEI Z, ZHANG F. Molecular Engineering of NIR-Ⅱ fluorophores for improved biomedical detection[J]. Angew Chem Int Ed, 2021, 60(30): 16294-16308. |
| 6 | YI H J, GHOSH D, HAM M H, et al. M13 Phage-functionalized single-walled carbon nanotubes as nanoprobes for second near-infrared window fluorescence imaging of targeted tumors[J]. Nano Lett, 2012, 12(3): 1176-1183. |
| 7 | LI Y, ZHANG P, TANG W, et al. Bright, magnetic NIR-Ⅱ quantum dot probe for sensitive dual-modality imaging and intensive combination therapy of cancer[J]. ACS Nano, 2022, 16(5): 8076-8094. |
| 8 | SONG Y, LU M, XIE Y, et al. Deep learning fluorescence imaging of visible to NIR-Ⅱ based on modulated multimode emissions lanthanide nanocrystals[J]. Adv Funct Mater, 2022, 32(45): 2206802. |
| 9 | TANG Y, LI Y, LU X, et al. Bio-erasable intermolecular donor-acceptor interaction of organic semiconducting nanoprobes for activatable NIR-Ⅱ fluorescence imaging[J]. Adv Funct Mater, 2019, 29(10): 1807376. |
| 10 | LIU X, YU B, SHEN Y, et al. Design of NIR-Ⅱ high performance organic small molecule fluorescent probes and summary of their biomedical application[J]. Coord Chem Rev, 2022, 468: 214609. |
| 11 | LI C, GUAN X, ZHANG X, et al. NIR-Ⅱ bioimaging of small molecule fluorophores: from basic research to clinical applications[J]. Biosens Bioelectron, 2022, 216: 114620. |
| 12 | SHOU K, QU C, SUN Y, et al. Multifunctional biomedical imaging in physiological and pathological conditions using a NIR-Ⅱ probe[J]. Adv Funct Mater, 2017, 27(23): 1700995. |
| 13 | TANG Y, PEI F, LU X, et al. Recent advances on activatable NIR-Ⅱ fluorescence probes for biomedical imaging[J]. Adv Opt Mater, 2019, 7(21): 1900917. |
| 14 | HUANG J, PU K. Activatable molecular probes for second near-infrared fluorescence, chemiluminescence, and photoacoustic imaging[J]. Angew Chem Int Ed, 2020, 59(29): 11717-11731. |
| 15 | LI D, PAN J, XU S, et al. Activatable second near-infrared fluorescent probes: a new accurate diagnosis strategy for diseases[J]. Biosensors, 2021, 11(11): 436. |
| 16 | ZHAO M, LI B, ZHANG H, et al. Activatable fluorescence sensors for in vivo bio-detection in the second near-infrared window[J]. Chem Sci, 2021, 12(10): 3448-3459. |
| 17 | ZHANG X, AN L, TIAN Q, et al. Tumor microenvironment-activated NIR-> reagents for tumor imaging and therapy[J]. J Mater Chem B, 2020, 8(22): 4738-4747. |
| 18 | NETEA M G, BALKWILL F, CHONCHOL M, et al. A guiding map for inflammation[J]. Nat Immunol, 2017, 18(8): 826-831. |
| 19 | KOTAS MAYA E, MEDZHITOV R. Homeostasis, inflammation, and disease susceptibility[J]. Cell, 2015, 160(5): 816-827. |
| 20 | TU Z, ZHONG Y, HU H, et al. Design of therapeutic biomaterials to control inflammation[J]. Nat Rev Mater, 2022, 7(7): 557-574. |
| 21 | METUSHI I, UETRECHT J, PHILLIPS E. Mechanism of isoniazid-induced hepatotoxicity: then and now[J]. Br J Clin Pharmacol, 2016, 81(6): 1030-1036. |
| 22 | YAMAMOTO H, WATANABE T, MIZUNO H, et al. In vivo evidence for accelerated generation of hydroxyl radicals in liver of Long-Evans Cinnamon (LEC) rats with acute hepatitis[J]. Free Radic Biol Med, 2001, 30(5): 547-554. |
| 23 | FENG W, ZHANG Y, LI Z, et al. Lighting up NIR-Ⅱ fluorescence in vivo: an activable probe for noninvasive hydroxyl radical imaging[J]. Anal Chem, 2019, 91(24): 15757-15762. |
| 24 | CHEN L, CHEN J, FANG Y, et al. A turn-on probe for detecting antituberculotic drug-induced liver injury in mice via NIR-Ⅱ fluorescence/optoacoustic imaging[J]. Chem Commun, 2021, 57(63): 7842-7845. |
| 25 | OU Y F, REN T B, YUAN L, et al. Molecular design of NIR-Ⅱ polymethine fluorophores for bioimaging and biosensing[J]. Chem Biomed Imaging, 2023, 1(3): 220-233. |
| 26 | TIAN Y, LIU S, CAO W, et al. H2O2-activated NIR-Ⅱ fluorescent probe with a large stokes shift for high-contrast imaging in drug-induced liver injury mice[J]. Anal Chem, 2022, 94(32): 11321-11328. |
| 27 | FERYSIUK K, WÓJCIAK K M. Reduction of nitrite in meat products through the application of various plant-based ingredients[J]. Antioxidants, 2020, 9(8): 711. |
| 28 | GEORGE J, TSUCHISHIMA M, TSUTSUMI M. Molecular mechanisms in the pathogenesis of N-nitrosodimethylamine induced hepatic fibrosis[J]. Cell Death Dis, 2019, 10(1): 18. |
| 29 | ZENG C, OUYANG J, SUN L, et al. An activatable probe for detection and therapy of food-additive-related hepatic injury via NIR-Ⅱ fluorescence/optoacoustic imaging and biomarker-triggered drug release[J]. Anal Chim Acta, 2022, 1208: 339831. |
| 30 | QIN Z, REN T B, ZHOU H, et al. NIRII-HDs: a versatile platform for developing activatable NIR-Ⅱ fluorogenic probes for reliable in vivo analyte sensing[J]. Angew Chem Int Ed, 2022, 61(19): e202201541. |
| 31 | LI D, WANG S, LEI Z, et al. Peroxynitrite activatable NIR-Ⅱ fluorescent molecular probe for drug-induced hepatotoxicity monitoring[J]. Anal Chem, 2019, 91(7): 4771-4779. |
| 32 | GRIFFITHS H R, GAO D, PARARASA C. Redox regulation in metabolic programming and inflammation[J]. Redox Biol, 2017, 12: 50-57. |
| 33 | HE L, HE L H, XU S, et al. Engineering of reversible NIR-Ⅱ redox-responsive fluorescent probes for imaging of inflammation in vivo[J]. Angew Chem Int Ed, 2022, 61(46): e202211409. |
| 34 | REN T B, WANG Z Y, XIANG Z, et al. A General strategy for development of activatable NIR-Ⅱ fluorescent probes for in vivo high-contrast bioimaging[J]. Angew Chem Int Ed, 2020, 60(2): 800-805. |
| 35 | CHEN Z, ZHANG Z, ZENG F, et al. Visualizing detection of diabetic liver injury by a biomarker-activatable probe via NIR-Ⅱ fluorescence imaging[J]. Chem Biomed Imaging, 2023: https://doi.org/10.1021/cbmi.3c00030. |
| 36 | ZHANG X, SHEN S, LIU D, et al. Combination of changeable π-conjugation and hydrophilic groups for developing water-soluble small-molecule NIR-Ⅱ fluorogenic probes[J]. Chem Sci, 2023, 14(11): 2928-2934. |
| 37 | DARMON M, OSTERMANN M, CERDA J, et al. Diagnostic work-up and specific causes of acute kidney injury[J]. Intensive Care Med, 2017, 43(6): 829-840. |
| 38 | VAIDYA V S, OZER J S, DIETERLE F, et al. Kidney injury molecule-1 outperforms traditional biomarkers of kidney injury in preclinical biomarker qualification studies[J]. Nat Biotechnol, 2010, 28(5): 478-485. |
| 39 | TAN J, YIN K, OUYANG Z, et al. Real-time monitoring renal impairment due to drug-induced AKI and diabetes-caused CKD using an NAG-activatable NIR-Ⅱ nanoprobe[J]. Anal Chem, 2021, 93(48): 16158-16165. |
| 40 | OUYANG J, SUN L, ZENG F, et al. Rational design of stable heptamethine cyanines and development of a biomarker-activatable probe for detecting acute lung/kidney injuries via NIR-Ⅱ fluorescence imaging[J]. Analyst, 2022, 147(3): 410-416. |
| 41 | TYAGI P, MOON C H, JANICKI J, et al. Recent advances in imaging and understanding interstitial cystitis[J]. F1000Research, 2018, 7: 1771. |
| 42 | ZHANG X, CHEN Y, HE H, et al. ROS/RNS and base dual activatable merocyanine-based NIR-Ⅱ fluorescent molecular probe for in vivo biosensing[J]. Angew Chem Int Ed, 2021, 60(50): 26337-26341. |
| 43 | CHEN J, CHEN L, WU Y, et al. A H2O2-activatable nanoprobe for diagnosing interstitial cystitis and liver ischemia-reperfusion injury via multispectral optoacoustic tomography and NIR-Ⅱ fluorescent imaging[J]. Nat Commun, 2021, 12(1): 6870. |
| 44 | WU W, YANG Y, YANG Y, et al. Molecular engineering of an organic NIR-Ⅱ fluorophore with aggregation-induced emission characteristics for in vivo imaging[J]. Small, 2019, 15(20): 1805549. |
| 45 | HUNTER D J, BIERMA-ZEINSTRA S. Osteoarthritis[J]. Lancet, 2019, 393(10182): 1745-1759. |
| 46 | WU P, ZHU Y, CHEN L, et al. A Fast-responsive OFF-ON near-infrared-Ⅱ fluorescent probe for in vivo detection of hypochlorous acid in rheumatoid arthritis[J]. Anal Chem, 2021, 93(38): 13014-13021. |
| 47 | GE X, LOU Y, SU L, et al. Single wavelength laser excitation ratiometric NIR-Ⅱ fluorescent probe for molecule imaging in vivo[J]. Anal Chem, 2020, 92(8): 6111-6120. |
| 48 | SHE Z, CHEN J, SUN L, et al. An NO-responsive probe for detecting acute inflammation using NIR-Ⅱ fluorescence/optoacoustic imaging[J]. Chem Commun, 2022, 58(94): 13123-13126. |
| 49 | KOBAYASHI T, SIEGMUND B, LE BERRE C, et al. Ulcerative colitis[J]. Nat Rev Dis Prim, 2020, 6(1): 74. |
| 50 | ZENG Z, OUYANG J, SUN L, et al. A Biomarker-responsive nanosystem with colon‐targeted delivery for ulcerative colitis's detection and treatment with optoacoustic/NIR-Ⅱ fluorescence imaging[J]. Adv Healthc Mater, 2022, 11(22): 2201544. |
| 51 | LLOVET J M, KELLEY R K, VILLANUEVA A, et al. Hepatocellular carcinoma[J]. Nat Rev Dis Prim, 2021, 7(1): 6. |
| 52 | ZENG Z, OUYANG J, SUN L, et al. Activatable nanocomposite probe for preoperative location and intraoperative navigation for orthotopic hepatic tumor resection via MSOT and aggregation-induced Near-IR-Ⅰ/Ⅱ fluorescence imaging[J]. Anal Chem, 2020, 92(13): 9257-9264. |
| 53 | PAN Y, LEI S, ZHANG J, et al. Activatable NIR-Ⅱ fluorescence probe for highly sensitive and selective visualization of glutathione in vivo[J]. Anal Chem, 2021, 93(51): 17103-17109. |
| 54 | WALLACE J L, WANG R. Hydrogen sulfide-based therapeutics: exploiting a unique but ubiquitous gasotransmitter[J]. Nat Rev Drug Discov, 2015, 14(5): 329-345. |
| 55 | YANG L, JIANG L, XU F, et al. Hydrogen sulfide activatable NIR-Ⅱ fluorescent probe for highly specific imaging of breast cancer[J]. Sens Actuators B: Chem, 2023, 379: 133251. |
| 56 | XU S, WANG Q, ZHANG Q, et al. Real time detection of ESKAPE pathogens by a nitroreductase-triggered fluorescence turn-on probe[J]. Chem Commun, 2017, 53(81): 11177-11180. |
| 57 | LAN Q, YU P, YAN K, et al. Polymethine molecular platform for ratiometric fluorescent probes in the second near-infrared window[J]. J Am Chem Soc, 2022, 144(46): 21010-21015. |
| 58 | SCHNEBLE E J, GRAHAM L J, SHUPE M P, et al. Future directions for the early detection of recurrent breast cancer[J]. J Cancer, 2014, 5(4): 291-300. |
| 59 | OUYANG J, SUN L, ZENG Z, et al. Nanoaggregate probe for breast cancer metastasis through multispectral optoacoustic tomography and aggregation-induced NIR-Ⅰ/Ⅱ fluorescence imaging[J]. Angew Chem Int Ed, 2019, 59(25): 10111-10121. |
| 60 | THAI A A, SOLOMON B J, SEQUIST L V, et al. Lung cancer[J]. Lancet, 2021, 398(10299): 535-554. |
| 61 | WANG R, CHEN J, GAO J, et al. A molecular design strategy toward enzyme-activated probes with near-infrared Ⅰ and Ⅱ fluorescence for targeted cancer imaging[J]. Chem Sci, 2019, 10(30): 7222-7227. |
| 62 | ZHANG X, LI X, SHI W, et al. Sensitive imaging of tumors using a nitroreductase-activated fluorescence probe in the NIR-Ⅱ window[J]. Chem Commun, 2021, 57(66): 8174-8177. |
| 63 | SUO Y, WU F, XU P, et al. NIR-Ⅱ fluorescence endoscopy for targeted imaging of colorectal cancer[J]. Adv Healthc Mater, 2019, 8(23): 1900974. |
| 64 | DOU K, FENG W, FAN C, et al. Flexible designing strategy to construct activatable NIR-Ⅱ fluorescent probes with emission maxima beyond 1200 nm[J]. Anal Chem, 2021, 93(8): 4006-4014. |
| 65 | XU G, YAN Q, LV X, et al. Imaging of colorectal cancers using activatable nanoprobes with second near-infrared window emission[J]. Angew Chem Int Ed, 2018, 57(14): 3626-3630. |
| 66 | XUAN W, XIA Y, LI T, et al. Molecular self-assembly of bioorthogonal aptamer-prodrug conjugate micelles for hydrogen peroxide and pH-independent cancer chemodynamic therapy[J]. J Am Chem Soc, 2019, 142(2): 937-944. |
| 67 | ZHAO M, WANG J, LEI Z, et al. NIR-Ⅱ pH sensor with a FRET adjustable transition point for in situ dynamic tumor microenvironment visualization[J]. Angew Chem Int Ed, 2021, 60(10): 5091-5095. |
| 68 | JESSMON P, BOULANGER T, ZHOU W, et al. Epidemiology and treatment patterns of epithelial ovarian cancer[J]. Expert Rev Anticancer Ther, 2017, 17(5): 427-437. |
| 69 | KIM E J, KUMAR R, SHARMA A, et al. In vivo imaging of β-galactosidase stimulated activity in hepatocellular carcinoma using ligand-targeted fluorescent probe[J]. Biomaterials, 2017, 122: 83-90. |
| 70 | CHEN J A, PAN H, WANG Z, et al. Imaging of ovarian cancers using enzyme activatable probes with second near-infrared window emission[J]. Chem Commun, 2020, 56(18): 2731-2734. |
| 71 | ZHANG L, ZHANG Y, CHI W, et al. “Crossbreeding” small-molecular weight NIR-Ⅱ flavchromenes endows activatable multiplexed in vivo imaging[J]. ACS Mater Lett, 2022, 4(8): 1493-1502. |
| 72 | SCHELTENS P, DE STROOPER B, KIVIPELTO M, et al. Alzheimer's disease[J]. Lancet, 2021, 397(10284): 1577-1590. |
| 73 | MIAO J, MIAO M, JIANG Y, et al. An activatable NIR-Ⅱ fluorescent reporter for in vivo imaging of amyloid-β plaques[J]. Angew Chem Int Ed, 2023, 62(7): e202216351. |
| [1] | Hui DU, Chen-Yang YAO, Hao PENG, Bo JIANG, Shun-Xiang LI, Jun-Lie YAO, Fang ZHENG, Fang YANG, Ai-Guo WU. Applications of Transition Metal⁃doped Iron⁃based Nanoparticles in Biomedicine [J]. Chinese Journal of Applied Chemistry, 2022, 39(3): 391-406. |
| [2] | HUANG Zike, LIU Chao, FU Qiangqiang, LI Jin, ZOU Jianmei, XIE Sitao, QIU Liping. Aptamer-based Fluorescence Probe for Bioanalysis and Bioimaging [J]. Chinese Journal of Applied Chemistry, 2018, 35(1): 28-39. |
| [3] | Jia ZHOU, Yun NI, Chengwu ZHANG, Xinghan QIU, Yanfei ZHAO, Lei BAI, Gaobin ZHANG, Lin LI. Design and Synthesis of Pyrimidine Based Two-Photon Fluorescence Probe and Its Application in Bioimaging [J]. Chinese Journal of Applied Chemistry, 2017, 34(12): 1450-1456. |
| [4] | Jia ZHOU, Yun NI, Chengwu ZHANG, Xinghan QIU, Yanfei ZHAO, Lei BAI, Gaobin ZHANG, Lin LI. Design and Synthesis of Pyrimidine Based Two-Photon Fluorescence Probe and Its Application in Bioimaging [J]. Chinese Journal of Applied Chemistry, 2017, 34(12): 0-0. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||