Chinese Journal of Applied Chemistry ›› 2024, Vol. 41 ›› Issue (3): 365-376.DOI: 10.19894/j.issn.1000-0518.230262
• Review • Previous Articles
Research Progress on Anodic Titanium‑Based Gas Diffusion Layer in Proton Exchange Membrane Electrolysis Cell
Ting-Ting GU1, Ke ZHANG1, Xin-Zhou ZHANG1, Yang LIU1, Wei-Cai SUN1, Ai-Dong TAN2, Jian-Guo LIU2( )
)
- 1.(China Titanium Guochuang (Qingdao) Technology Co. ,Ltd. ,Qingdao 266111,China )
2.North China Electric Power University,Beijing 100054,China
-
Received:2023-08-30Accepted:2023-12-08Published:2024-03-01Online:2024-04-09 -
Contact:Jian-Guo LIU -
About author:jianguoliu@ncepu.edu.cn
-
Supported by:the National Key Research and Development Program(2021YFB4000100)
CLC Number:
Cite this article
Ting-Ting GU, Ke ZHANG, Xin-Zhou ZHANG, Yang LIU, Wei-Cai SUN, Ai-Dong TAN, Jian-Guo LIU. Research Progress on Anodic Titanium‑Based Gas Diffusion Layer in Proton Exchange Membrane Electrolysis Cell[J]. Chinese Journal of Applied Chemistry, 2024, 41(3): 365-376.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.230262
| Type | Raw material | Raw material diameter/μm | Porosity ratio/% | Pore diameter/μm | Thickness/μm | Ref. |
|---|---|---|---|---|---|---|
Titanium powder sintered felt | Ordinary titanium powder | / | 10~33 | / | 253~526 | [ |
| 75~100 | 35~40 | 11~25 | 1 200~1 400 | [ | ||
| 380~520 | / | 100 | 1 000 | |||
| 100~200 | 34.3 | 16 | 1 300 | [ | ||
| 330~780 | 32.7 | 60 | 1 200 | |||
| 20 | 50~70 | 10~35 | 200/300 | [ | ||
| Hydrogenated dehydrogenation titanium powder | / | 10~55 | / | 246~515 | [ | |
| / | 54 | / | 513 | [ | ||
| 40 | 28 | 8 | 800 | [ | ||
| Titanium fiber sintered felt | Titanium fiber | / | 53 | / | 1 000 | [ |
| 20 | 75 | 50 | 300 | [ | ||
| 80 | 75 | 180 | 300 | |||
| 20 | 50 | 28 | 300 | |||
| / | 75 | / | 300 | [ | ||
| 20 | 50 | 12.7 | 350/500/1 000 | [ | ||
| Porous titanium foil | Titanium foil | / | / | 1~25 | 50.8 | [ |
Table 1 Structural parameters of various porous titanium materials
| Type | Raw material | Raw material diameter/μm | Porosity ratio/% | Pore diameter/μm | Thickness/μm | Ref. |
|---|---|---|---|---|---|---|
Titanium powder sintered felt | Ordinary titanium powder | / | 10~33 | / | 253~526 | [ |
| 75~100 | 35~40 | 11~25 | 1 200~1 400 | [ | ||
| 380~520 | / | 100 | 1 000 | |||
| 100~200 | 34.3 | 16 | 1 300 | [ | ||
| 330~780 | 32.7 | 60 | 1 200 | |||
| 20 | 50~70 | 10~35 | 200/300 | [ | ||
| Hydrogenated dehydrogenation titanium powder | / | 10~55 | / | 246~515 | [ | |
| / | 54 | / | 513 | [ | ||
| 40 | 28 | 8 | 800 | [ | ||
| Titanium fiber sintered felt | Titanium fiber | / | 53 | / | 1 000 | [ |
| 20 | 75 | 50 | 300 | [ | ||
| 80 | 75 | 180 | 300 | |||
| 20 | 50 | 28 | 300 | |||
| / | 75 | / | 300 | [ | ||
| 20 | 50 | 12.7 | 350/500/1 000 | [ | ||
| Porous titanium foil | Titanium foil | / | / | 1~25 | 50.8 | [ |
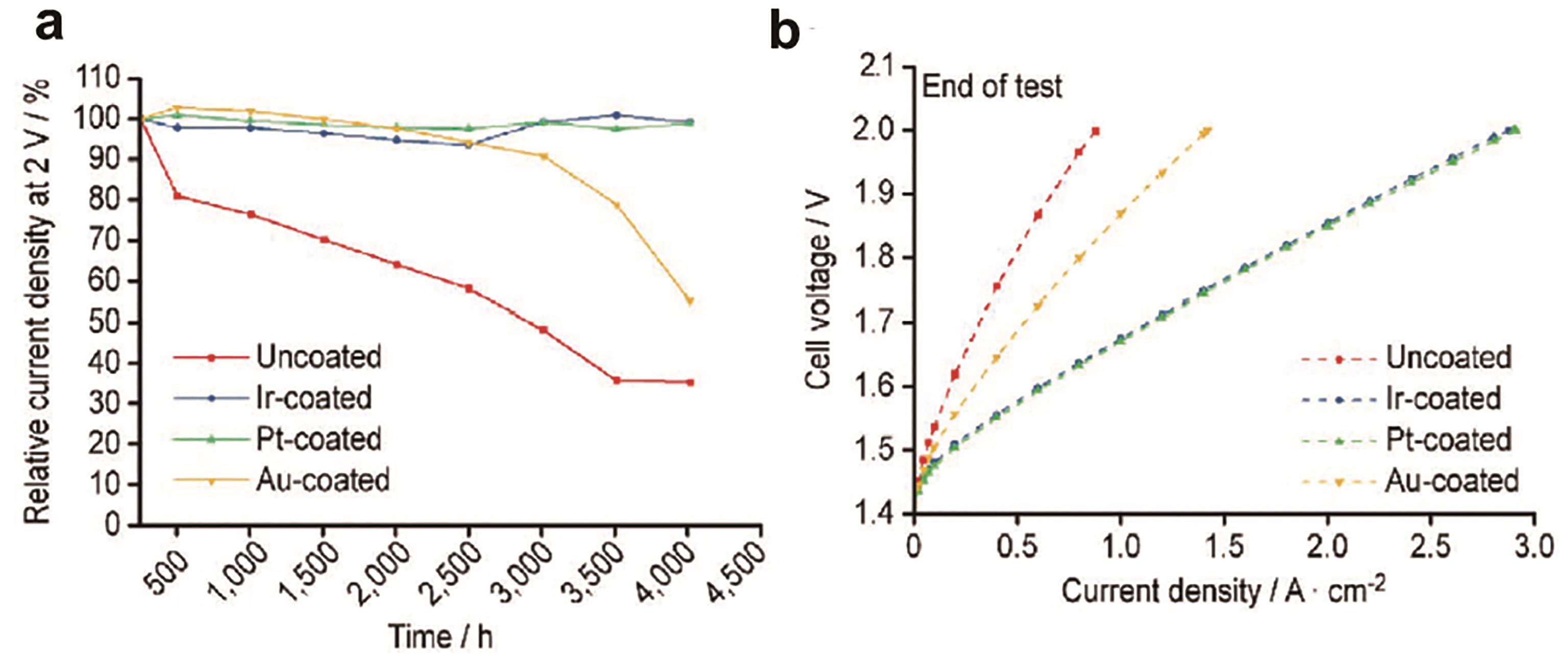
Fig.4 The results of stability test and polarization test: (a) The voltage change of Au, Ir and Pt as coating materials and uncoated titanium felt after 4000 h operation; (b) The polarization curve of titanium felt with Au, Ir and Pt as coating material after 4000 h operation[43]

Fig.5 Test results of contact resistance and polarization curves: (a) Contact resistance of uncoated gas transport layer and coated gas transport layer with Ir mass of 0.005, 0.013, 0.025 and 0.025 mg/cm2; (b) Polarization curve of uncoated gas transport layer and coated gas transport layer with Ir mass of 0.005, 0.013, 0.025 and 0.05 mg/cm2[47]
| Coating | Contact resistance | Load capacity | Electrolytic performance | Stability | Ref. |
|---|---|---|---|---|---|
| IrO2 | / | 1.0 mg/cm2 | 0.36 A/cm2@1.60 V | 53 h@1.52 A/cm2 | [ |
| / | 0.60 mg/cm2 | 2.72 A/cm2@2.0 V | / | [ | |
| Ir | 11.5 mΩ·cm2@0.5 MPa | 0.10 mg/cm2 | 2.0 A/ cm2@1.86 V | / | [ |
| 4.0 mΩ·cm2@3 MPa | 0.10 mg/cm2 | 2.38 A/cm2@1.90 V | 4 000 h@2.0 V | [ | |
| 3.1 mΩ·cm2@3 MPa | 0.025 mg/cm2 | 1.0 A/cm2@1.70 V | / | [ | |
| Pt | 6.0 mΩ·cm2@3 MPa | 0.16 mg/cm2 | 2.4 A/cm2@1.90 V | 4 000 h@2.0 V | [ |
| / | 200 nm (thickness) | 2.82 A/cm2@2.0 V | 1 100 h@2.0 A/cm2 | [ | |
| Au | 2.0 mΩ·cm2@3 MPa | 0.18 mg/cm2 | 2.35 A/cm2@1.90 V | 2 000 h@2.0 V | [ |
| / | 180 nm (thickness) | 2.0 A/cm2 @1.63 V | 100 h@0.2 A/cm2 | [22] | |
| Ir0.7Ru0.3O2 | 6.5 mΩ·cm2@2 MPa | 1.0 mg/cm2 | 1.84 V@2.0 A/cm2 | / | [ |
| IrO2?RuO2?TaO x | / | 1.0 mg (Ir+Ru)/cm2 | 1.84 V@2.0 A/cm2 | / | [ |
| STN?RuTi | / | STN (thickness)99 nm | / | 225 h@0.10A/cm2 | [ |
| NbN_500_60 | / | 1.0 μm (thickness) | / | / | [ |
Table 2 Electrochemical energy table of gas diffusion layer treated by various coatings in literature
| Coating | Contact resistance | Load capacity | Electrolytic performance | Stability | Ref. |
|---|---|---|---|---|---|
| IrO2 | / | 1.0 mg/cm2 | 0.36 A/cm2@1.60 V | 53 h@1.52 A/cm2 | [ |
| / | 0.60 mg/cm2 | 2.72 A/cm2@2.0 V | / | [ | |
| Ir | 11.5 mΩ·cm2@0.5 MPa | 0.10 mg/cm2 | 2.0 A/ cm2@1.86 V | / | [ |
| 4.0 mΩ·cm2@3 MPa | 0.10 mg/cm2 | 2.38 A/cm2@1.90 V | 4 000 h@2.0 V | [ | |
| 3.1 mΩ·cm2@3 MPa | 0.025 mg/cm2 | 1.0 A/cm2@1.70 V | / | [ | |
| Pt | 6.0 mΩ·cm2@3 MPa | 0.16 mg/cm2 | 2.4 A/cm2@1.90 V | 4 000 h@2.0 V | [ |
| / | 200 nm (thickness) | 2.82 A/cm2@2.0 V | 1 100 h@2.0 A/cm2 | [ | |
| Au | 2.0 mΩ·cm2@3 MPa | 0.18 mg/cm2 | 2.35 A/cm2@1.90 V | 2 000 h@2.0 V | [ |
| / | 180 nm (thickness) | 2.0 A/cm2 @1.63 V | 100 h@0.2 A/cm2 | [22] | |
| Ir0.7Ru0.3O2 | 6.5 mΩ·cm2@2 MPa | 1.0 mg/cm2 | 1.84 V@2.0 A/cm2 | / | [ |
| IrO2?RuO2?TaO x | / | 1.0 mg (Ir+Ru)/cm2 | 1.84 V@2.0 A/cm2 | / | [ |
| STN?RuTi | / | STN (thickness)99 nm | / | 225 h@0.10A/cm2 | [ |
| NbN_500_60 | / | 1.0 μm (thickness) | / | / | [ |
| Coating material | Electrical conductivity | Corrosion resistance | OER catalytic performance |
|---|---|---|---|
| Ir/IrO2 | ++ | +++ | +++ |
| Pt | ++ | +++ | + |
| Au | +++ | + | - |
| RuO2 | - | - | +++ |
| Other metallic materials | + | + | - |
Table 3 Comparison table of properties of various coating materials
| Coating material | Electrical conductivity | Corrosion resistance | OER catalytic performance |
|---|---|---|---|
| Ir/IrO2 | ++ | +++ | +++ |
| Pt | ++ | +++ | + |
| Au | +++ | + | - |
| RuO2 | - | - | +++ |
| Other metallic materials | + | + | - |
| 1 | NOYAN O F, HASAN M M, PALA N. A global review of the hydrogen energy ECO-system[J]. Energies, 2023, 16(3): 1462-1484. |
| 2 | MAIER M, SMITH K, DODWELL J, et al. Mass transport in PEM water electrolysers: a review[J]. Int J Hydrogen Energy, 2022, 47(1): 30-56. |
| 3 | MO J, STEEN S M, RETTERER S T, et al. Mask-patterned wet etching of thin titanium liquid gas diffusion layers for a PEMEC[J]. ECS Transact, 2015, 66(24): 3-10. |
| 4 | LEE J K, LAU G Y, SABHARWAL M, et al. Titanium porous-transport layers for PEM water electrolysis prepared by tape casting[J]. J Power Sources, 2023, 559(1): 232606. |
| 5 | MO J, KANG Z, RETTERER S T, et al. Discovery of true electrochemical reactions for ultrahigh catalyst mass activity in water splitting[J]. Sci Adv, 2016, 2(11): 1600690. |
| 6 | WANG C, WU Y, BODACH A, et al. A novel electrode for value-generating anode reactions in water electrolyzers at industrial current densities[J]. Angew Chem Int Ed Engl, 2023, 62(7): e202215804. |
| 7 | XUE F, SU J, LI P, et al. Application of proton exchange membrane electrolysis of water hydrogen production technology in power plant[J]. IOP Conference Series: Earth Environ Sci, 2021, 631(1): 012079. |
| 8 | DONG S, ZHANG C, YUE Z, et al. Overall design of anode with gradient ordered structure with low Iridium loading for proton exchange membrane water electrolysis[J]. Nano Lett, 2022, 22(23): 9434-9440. |
| 9 | PUTHIYAPURA V K, MAMLOUK M, PASUPATHI S, et al. Physical and electrochemical evaluation of ATO supported IrO2 catalyst for proton exchange membrane water electrolyser[J]. J Power Sources, 2014, 269(10): 451-460. |
| 10 | FENG Q, YUAN X Z, LIU G, et al. A review of proton exchange membrane water electrolysis on degradation mechanisms and mitigation strategies[J]. J Power Sources, 2017, 366(31): 33-55. |
| 11 | LOPATA J, KANG Z, YOUNG J, et al. Effects of the transport/catalyst layer interface and catalyst loading on mass and charge transport phenomena in polymer electrolyte membrane water electrolysis devices[J]. J Electrochem Soc, 2020, 167(6): 064507. |
| 12 | 徐滨, 王锐, 苏伟, 等. 质子交换膜电解水技术关键材料的研究进展与展望[J]. 储能科学与技术, 2022, 11(11): 3510-3520. |
| XU B, WANG R, SU W, et al. Research progress and prospect of key materials of proton exchange membrane water electrolysis[J]. Energy Storage Sci Technol, 2022, 11(11): 3510-3520. | |
| 13 | ARBABI F, KALANTARIAN A, ABOUATALLAH R, et al. Feasibility study of using microfluidic platforms for visualizing bubble flows in electrolyzer gas diffusion layers[J]. J Power Sources, 2014, 258(15): 142-149. |
| 14 | 阳柳. 质子交换膜燃料电池气体扩散层及流场板结构的模拟研究[D]. 北京: 北京交通大学, 2020. |
| YANG L. Simulation study on the structure of gas diffusion layer and flow field plate in Proton exchange membrane fuel cell[D]. Beijing: Beijing Jiaotong University, 2020. | |
| 15 | ATHANASAKI G, JAYAKUMAR A, KANNAN A M. Gas diffusion layers for PEM fuel cells: materials, properties and manufacturing-a review[J]. Int J Hydrog Energy, 2023, 48(6): 2294-2313. |
| 16 | DOAN T L, LEE H E, SHAH S S H, et al. A review of the porous transport layer in polymer electrolyte membrane water electrolysis[J]. Int J Energy Res, 2021, 45(10): 14207-14220. |
| 17 | 刘春轩, 王骏斌, 高平平, 等. PEMFC钛金属双极板表面改性研究进展[J]. 稀有金属与硬质合金, 2019, 47(6): 6-9. |
| LIU C X, WANG J B, GAO P P, et al. Research progress on surface modification of PEMFC titanium metal bipolar plate[J]. Rare Metal Cemented Carbides, 2019, 47(6): 6-9. | |
| 18 | 张萍俊. 质子交换膜水电解池的性能优化及动态响应的研究[D]. 大连: 大连交通大学, 2019. |
| ZHANG J P. Performance optimization and dynammic response of proton exchange membrane water electolysis cell[D]. Dalian: Dalian Jiaotong University, 2019. | |
| 19 | BORGARDT E, PANCHENKO O, HACKEMÜLLER F J, et al. Mechanical characterization and durability of sintered porous transport layers for polymer electrolyte membrane electrolysis[J]. J Power Sources, 2018, 374(15): 84-91. |
| 20 | BAUTKINOVA T, UTSCH N, BYSTRON T, et al. Introducing titanium hydride on porous transport layer for more energy efficient water electrolysis with proton exchange membrane[J]. J Power Sources, 2023, 565(1): 232913. |
| 21 | PANCHENKO O, BORGARDT E, ZWAYGARDT W, et al. In‑situ two-phase flow investigation of different porous transport layer for a polymer electrolyte membrane (PEM) electrolyzer with neutron spectroscopy[J]. J Power Sources, 2018, 390: 108-115. [22] KANG Z, MO J, YANG G, et al. Thin film surface modifications of thin/tunable liquid/gas diffusion layers for high-efficiency proton exchange membrane electrolyzer cells[J]. Appl Energy, 2017, 206(15): 983-990. |
| 23 | MO J, DEHOFF R R, PETER W H, et al. Additive manufacturing of liquid/gas diffusion layers for low-cost and high-efficiency hydrogen production[J]. Int J Hydrog Energy, 2016, 41(4): 3128-3135. |
| 24 | ARISETTY S, PRASAD A K, ADVANI S G. Metal foams as flow field and gas diffusion layer in direct methanol fuel cells[J]. J Power Sources, 2007, 165(1): 49-57. |
| 25 | BAROUTAJI A, CARTON J G, STOKES J, et al. Application of open pore cellular foam for air breathing PEM fuel cell[J]. Int J Hydrog Energy, 2017, 42(40): 25630-25638. |
| 26 | MI B, WANG Q, QI T, et al. Performance and structure of Ti-doped amorphous carbon/CrN/Ti multilayer coating deposited on 316L stainless steel for use as bipolar plate in proton exchange membrane fuel cell[J]. J Alloys Compd, 2023, 943(15): 169080. |
| 27 | SCHULER T, SCHMIDT T J, BÜCHI F N. Polymer electrolyte water electrolysis: correlating performance and porous transport layer structure: part II. electrochemical performance analysis[J]. J Electrochem Soc, 2019, 166(10): 555-565. |
| 28 | YUAN S, ZHAO C, CAI X, et al. Bubble evolution and transport in PEM water electrolysis: mechanism, impact, and management[J]. Prog Energy Combust Sci, 2023, 96(1): 101075. |
| 29 | HWANG C M, ISHIDA M, ITO H, et al. Influence of properties of gas diffusion layers on the performance of polymer electrolyte-based unitized reversible fuel cells[J]. Int J Hydrog Energy, 2011, 36(2): 1740-1753. |
| 30 | ITO H, MAEDA T, NAKANO A, et al. Influence of different gas diffusion layers on the water management of polymer electrolyte unitized reversible fuel cell[J]. ECS Trans, 2010, 33(1): 945-954. |
| 31 | GRIGORIEV S A, MILLET P, VOLOBUEV S A, et al. Optimization of porous current collectors for PEM water electrolysers[J]. Int J Hydrogen Energy, 2009, 34(11): 4968-4973. |
| 32 | JUDE O M, FRANCESCO L, PAUL R S, et al. Effect of microstructure of porous transport layer on performance in polymer electrolyte membrane water electrolyser[J]. Energy Procedia, 2018, 151: 111-119. |
| 33 | LEE J K, LEE C H, BAZYLAK A. Pore network modelling to enhance liquid water transport through porous transport layers for polymer electrolyte membrane electrolyzers[J]. J Power Sources, 2019, 437: 226910. |
| 34 | SCHULER T, CICCONE J M, KRENTSCHER B, et al. Hierarchically structured porous transport layers for polymer electrolyte water electrolysis[J]. Adv Energy Mater, 2019, 10(2): 1903216. |
| 35 | 唐英伦, 杨小涛, 赵军, 等. 叠层扩散层对质子交换膜电解池性能影响的实验研究[J]. 工程热物理学报, 2023, 44(2): 304-310. |
| TANG Y L, YANG X T, ZHAO J, et al. Experimental study on the effects of stacked diffusion layer on the performance of proton-exchange membrane electrolysis cells[J]. J Eng Thermophys, 2023, 44(2): 304-310. | |
| 36 | MO J, STEEN S, HAN B, et al. Investigation of titanium felt transport parameters for energy storage and hydrogen/oxygen production[M]. 13th International Energy Conversion Engineering Conference, 2015. |
| 37 | LI H, FUJIGAYA T, NAKAJIMA H, et al. Optimum structural properties for an anode current collector used in a polymer electrolyte membrane water electrolyzer operated at the boiling point of water[J]. J Power Sources, 2016, 332(15): 16-23. |
| 38 | OMRANI R, SHABANI B. Review of gas diffusion layer for proton exchange membrane-based technologies with a focus on unitised regenerative fuel cells[J]. Int J Hydrogen Energy, 2019, 44(7): 3834-3860. |
| 39 | BYSTRON T, VESELY M, PAIDAR M, et al. Enhancing PEM water electrolysis efficiency by reducing the extent of Ti gas diffusion layer passivation[J]. J Appl Electrochem, 2018, 48(6): 713-723. |
| 40 | TAN A, ZHANG Y, SHI X, et al. The poisoning effects of Ti-ion from porous transport layers on the membrane electrode assembly of proton exchange membrane water electrolyzers[J]. Chem Eng J, 2023, 471(1): 144624-144631. |
| 41 | 欧阳韬, 杨扬, 张亮, 等. 酸处理钛毡对质子交换膜电解槽性能的影响[J]. 中国电机工程学报, 2023: 1-12. |
| OU-YANG T, YANG Y, ZHANG L, et al. Effect of acid-treated titanium felt on the performance of proton exchange electrolyzer[J]. Proc CSEE, 2023: 1-12. | |
| 42 | LIU C, CARMO M, BENDER G, et al. Performance enhancement of PEM electrolyzers through iridium-coated titanium porous transport layers[J]. Electrochem Commun, 2018, 97(1): 96-100. |
| 43 | LIU C, SHVIRO M, GAGO A S, et al. Exploring the interface of skin-layered titanium fibers for electrochemical water splitting[J]. Adv Energy Mater, 2021, 11(8): 2002926. |
| 44 | LIU C, SHVIRO M, BENDER G, et al. Degradation effects at the porous transport layer/catalyst layer interface in polymer electrolyte membrane water electrolyzer[J]. J Electrochem Soc, 2023, 170(3): 034508. |
| 45 | KULKARNI D, HUYNH A, SATJARUTANUN P, et al. Elucidating effects of catalyst loadings and porous transport layer morphologies on operation of proton exchange membrane water electrolyzers[J]. Appl Catal B, 2022, 308(5): 121213. |
| 46 | DOAN T L, LEE H E, KIM M, et al. Influence of IrO2/TiO2 coated titanium porous transport layer on the performance of PEM water electrolysis [J]. J Power Sources, 2022, 533(15): 231370. |
| 47 | LIU C, WIPPERMANN K, RASINSKI M, et al. Constructing a multifunctional interface between membrane and porous transport layer for water electrolyzers[J]. ACS Appl Mater Interfaces, 2021, 13(14): 16182-16196. |
| 48 | ROST U, PODLESCHNY P, SCHUMACHER M, et al. Long-term stable electrodes based on platinum electrocatalysts supported on titanium sintered felt for the use in PEM fuel cells[J]. IOP Conference Series: Mater Sci Eng, 2018, 416(1): 012013. |
| 49 | BERNT M, SCHRAMM C, SCHRÖTER J, et al. Effect of the IrOx conductivity on the anode electrode/porous transport layer interfacial resistance in PEM water electrolyzers[J]. J Electrochem Soc, 2021, 168(8): 084513. |
| 50 | RAKOUSKY C, KEELEY G P, WIPPERMANN K, et al. The stability challenge on the pathway to high-current-density polymer electrolyte membrane water electrolyzers[J]. Electrochim Acta, 2018, 278(10): 324-331. |
| 51 | 范芷萱, 俞红梅, 姜广, 等. PEM水电解池低成本阳极钛纤维毡扩散层研究[J]. 电源技术, 2019, 44(7): 933-937. |
| FAN Z X, YU H M, JIANG G, et al. A low-cost Ti felt anode gas diffusion layer for PEM water electrolysis[J]. Chin J Power Sources, 2019, 44(7): 933-937. | |
| 52 | FAN Z X, YU H M, JIANG G, et al. Low precious metal loading porous transport layer coating and anode catalyst layer for proton exchange membrane water electrolysis[J]. Int J Hydrogen Energy, 2022, 47(44): 18963-18971. |
| 53 | 卫飞彬. 高电密下高耐蚀性钛基阳极的制备及其电催化性能研究[D]. 北京: 北京化工大学, 2022. |
| WEI F B. Study on preparation of high corrosion resistance Ti-based anode and its electrocatalytic performance in acid[D]. Beijing: Beijing University of Chemical Technology, 2022. | |
| 54 | DAUDT N F, SCHNEIDER A D, ARNEMANN E R, et al. Fabrication of NbN-coated porous titanium sheets for PEM electrolyzers[J]. J Mater Eng Perform, 2020, 29(8): 5174-5183. |
| 55 | 黄敏, 刘高阳, 王新东. PEM水电解用多层钛网析氧阳极制备及性能研究[J]. 有色金属科学与工程, 2016, 7(3): 1-5. |
| HUANG M, LIU G Y, WANG X D. Synthesis and performance study of multilayer titanium mesh oxygen evolution anode in the polymer exchange membrane water electrolysis[J]. Nonferrous Met Sci Eng, 2016, 7(3): 1-5. |
| [1] | Yu CHENG, Ling-Jun HE, Chu-Yuan LIN, Hui LIN, Fu-Yu XIAO, Wen-Bin LAI, Qing-Rong QIAN, Xiao-Xia HUANG, Qing-Hua CHEN, Ling-Xing ZENG. Progress Research on Electrolyte Modification Strategy to Improve the Performance of Aqueous Zinc-Ion Batteries Within the Wide Temperature Range [J]. Chinese Journal of Applied Chemistry, 2024, 41(3): 349-364. |
| [2] | Si-Bei LIU, Jun-Qiao MEI, Jin-Yu XIE, Yi-Jun LIU, Feng-Xia DENG, Shan QIU. Activated Carbon Boosted Performance of a Titanium-Based Air Diffusion Electrode in Electro-Peroxone Oxidation and Ibuprofen Degradation [J]. Chinese Journal of Applied Chemistry, 2024, 41(2): 279-296. |
| [3] | Ding ZHANG, Wei-Wei YANG, Song-Song MIAO, Yi SU. Au Nanoparticles Confined in N-Doped Porous Carbon for Detection of Chlorine Dioxide in Liquid Phase [J]. Chinese Journal of Applied Chemistry, 2023, 40(11): 1572-1580. |
| [4] | Jun-Jie GE. Preface to Special Issue “Hydrogen Energy and Fuel Cells” [J]. Chinese Journal of Applied Chemistry, 2023, 40(8): 1061-1062. |
| [5] | Er-Gui LUO, Tao TANG, Yi WANG, Jun-Ming ZHANG, Yu-Hong CHANG, Tian-Jun HU, Jian-Feng JIA. Progress on Tuning the Geometric and Electronic Structure of Precious Metal Catalysts for Hydrogen Peroxide Production via Two-Electron Oxygen Reduction [J]. Chinese Journal of Applied Chemistry, 2023, 40(8): 1063-1076. |
| [6] | Yi-Ning DONG, He LI, Xue GONG, Ce HAN, Ping SONG, Wei-Lin XU. Research Progress of Non-Pt-Based Catalysts in Cathode Oxygen Reduction Reaction of Proton Exchange Membrane Fuel Cells [J]. Chinese Journal of Applied Chemistry, 2023, 40(8): 1077-1093. |
| [7] | Cui-Ying TAN, Wei-Chao DING, Ting-Ting MA, Yao XIAO, Jian LIU. Research Progress on Superhydrophilic/Superaerophobic Electrocatalysts for Water Splitting [J]. Chinese Journal of Applied Chemistry, 2023, 40(8): 1109-1125. |
| [8] | Chun YIN, Jia-Xin LI, Li-Gang FENG. Basics of Research Progress for Urea Electrolysis for Hydrogen Generation and Urea Fuel Cells [J]. Chinese Journal of Applied Chemistry, 2023, 40(8): 1158-1174. |
| [9] | Rui-Xue ZHENG, Qing-Lei MENG, Li ZHANG, Chang-Peng LIU, Wei XING, Mei-Ling XIAO. Hierarchically Porous Fe-N-C Catalysts for Efficient Electrocatalytic Oxygen Reduction Reaction [J]. Chinese Journal of Applied Chemistry, 2023, 40(8): 1187-1194. |
| [10] | Lian-Cheng HUI, Jian-Xing ZHUANG, Shun XIAO, Mei-Ping LI, Meng-Yuan JIN, Qing LYU. Nickel-Nitrogen-Doped Graphdiyne as an Efficient Catalyst for Oxygen Reduction [J]. Chinese Journal of Applied Chemistry, 2023, 40(8): 1205-1213. |
| [11] | Xiao-Fang XU, Qiang CHEN, Hai-Bao ZHANG. Recent Progress in Nitrogen Fixation via Gliding Arc Plasma [J]. Chinese Journal of Applied Chemistry, 2023, 40(7): 923-937. |
| [12] | Feng ZHU, Xiao-Lian PENG, Wen-Bin ZHANG. Research Progress in the Effects of Proton Acceptor/Donor on Electrocatalytic Reactions [J]. Chinese Journal of Applied Chemistry, 2023, 40(5): 666-680. |
| [13] | Fang-Zheng HU, Xing GAO, Lei LIU, Tian-Heng YUAN, Ning CAO, Kai LI, Ya-Tao WANG, Jian-Hua LI, Hui-Qin LIAN, Xiao-Dong WANG, Xiu-Guo CUI. Advances in Black Phosphorus Anode Advantages and Optimization in Li-ion Battery Anodes [J]. Chinese Journal of Applied Chemistry, 2023, 40(4): 571-582. |
| [14] | Wen-Jun SHI, Zhong-Hui SUN, Zhong-Qian SONG, XU-Jia NAN, Dong-Xue HAN, Li NIU. Research Progress of Layered Transition Metal Oxides Cathode Materials for Sodium-ion Batteries [J]. Chinese Journal of Applied Chemistry, 2023, 40(4): 583-596. |
| [15] | Xue-Jian SHI, Wan-Qiang LIU, Chun-Li WANG, Yong CHENG, Li-Min WANG. Research Progress of Sb-based Anode Materials for Potassium Ion Batteries [J]. Chinese Journal of Applied Chemistry, 2023, 40(2): 210-228. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||