Chinese Journal of Applied Chemistry ›› 2024, Vol. 41 ›› Issue (3): 349-364.DOI: 10.19894/j.issn.1000-0518.230238
• Review • Previous Articles
Progress Research on Electrolyte Modification Strategy to Improve the Performance of Aqueous Zinc-Ion Batteries Within the Wide Temperature Range
Yu CHENG1, Ling-Jun HE1, Chu-Yuan LIN1, Hui LIN1, Fu-Yu XIAO1, Wen-Bin LAI1, Qing-Rong QIAN1,2, Xiao-Xia HUANG3( ), Qing-Hua CHEN1,2, Ling-Xing ZENG1,2(
), Qing-Hua CHEN1,2, Ling-Xing ZENG1,2( )
)
- 1.Engineering Research Center of Polymer Green Recycling of Ministry of Education,College of Environment and Resources,Fujian Normal University,Fuzhou 350007,China
2.(Key Laboratory of Advanced Energy Materials Chemistry (Ministry of Education),College of Chemistry,Nankai University,Tianjin 300071,China )
3.Fujian College of Water Conservancy and Electric Power,Sanming 366000,China
-
Received:2023-08-09Accepted:2024-01-01Published:2024-03-01Online:2024-04-09 -
Contact:Xiao-Xia HUANG,Ling-Xing ZENG -
Supported by:the National Key R&D Program Project of China(2023YFC3906300);the National Natural Science Foundation of China(21801251);Fujian Eagle Programme for Young Top Talents, Fujian Provincial Key Fund(2023J02013);the Provincial Science and Technology Innovation Key Project(2022G02022);Fuzhou City Foreign Science and Technology Cooperation Project(2022-Y-004)
CLC Number:
Cite this article
Yu CHENG, Ling-Jun HE, Chu-Yuan LIN, Hui LIN, Fu-Yu XIAO, Wen-Bin LAI, Qing-Rong QIAN, Xiao-Xia HUANG, Qing-Hua CHEN, Ling-Xing ZENG. Progress Research on Electrolyte Modification Strategy to Improve the Performance of Aqueous Zinc-Ion Batteries Within the Wide Temperature Range[J]. Chinese Journal of Applied Chemistry, 2024, 41(3): 349-364.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.230238
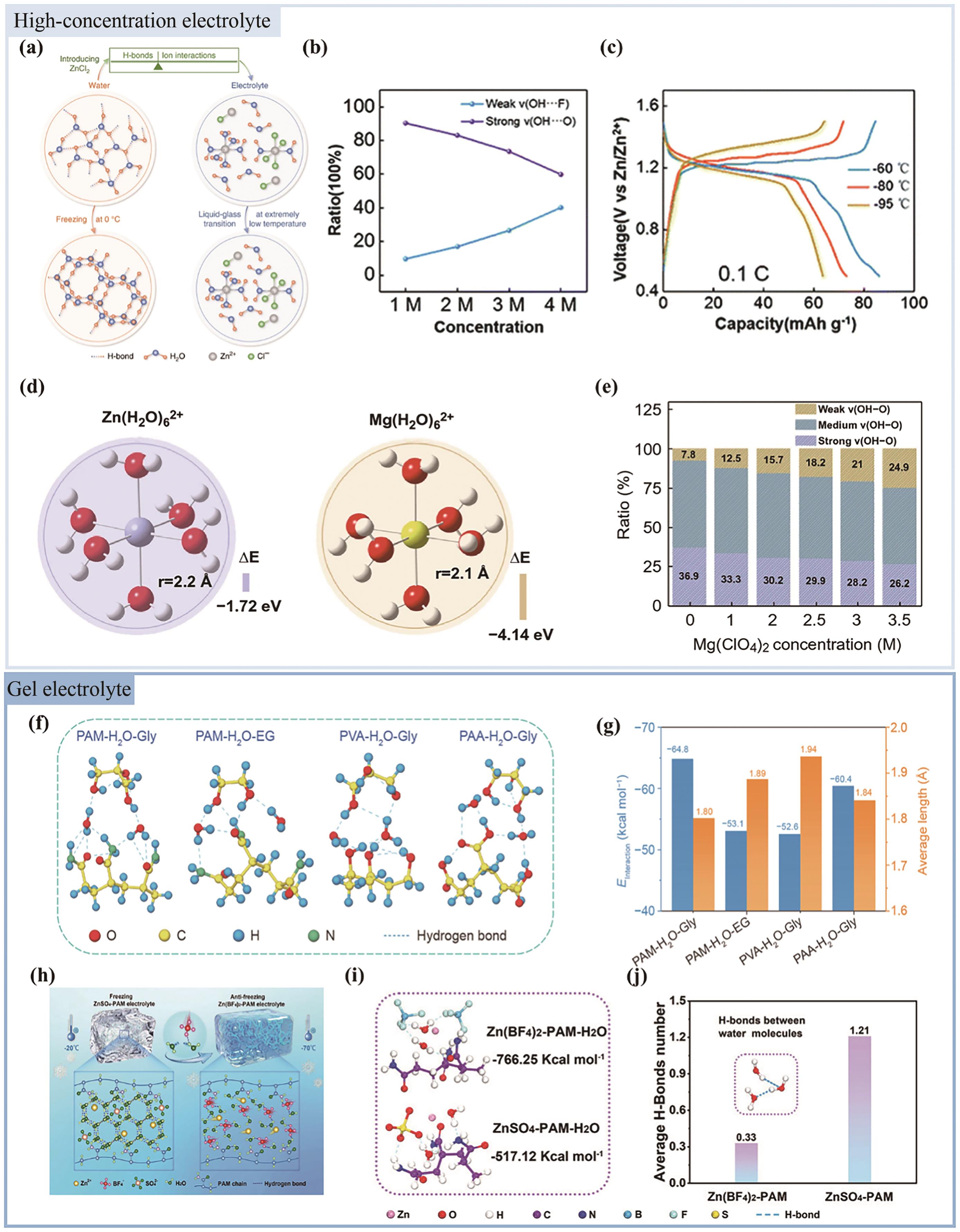
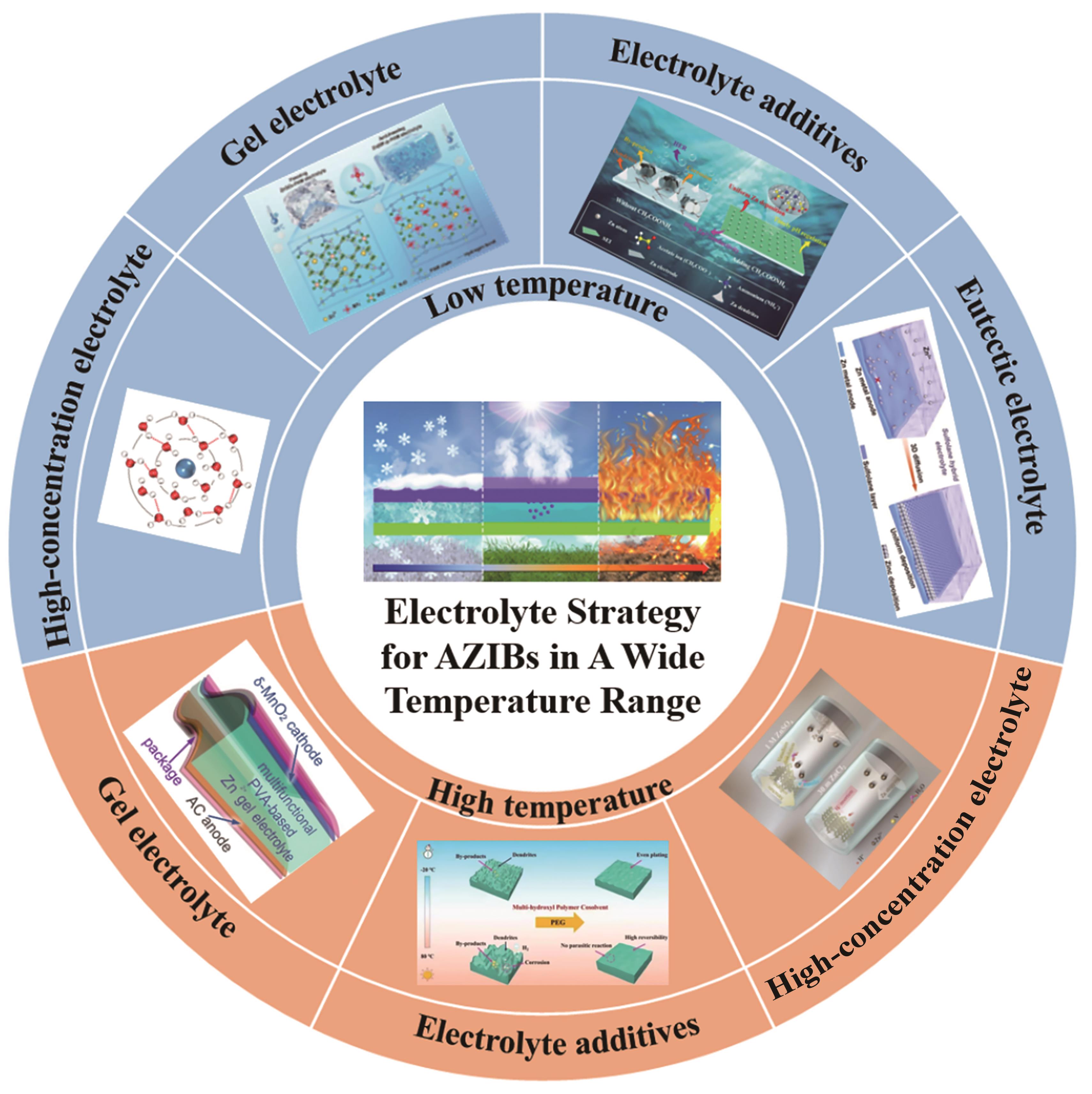
Fig.2 (a) The mechanism of “water-in-salt” electrolyte action[29]; (b) The proportion of two hydrogen bonds in different concentrations of Zn(BF4)2[38]; (c) The charge-discharge curves of Zn//TCBQ battery in the temperature range from -95 ℃ to -60 ℃ were obtained[38]; (d) The calculated formation energy and hydrated radius of Zn2+ solvation configuration and Mg2+ solvation configuration[51]; (e) The ratio of different types of HBs[37]; (f) DFT optimized structures of PAM-H2O-Gly, PAM-H2O-EG, PVA-H2O-Gly and PAA-H2O-Gly were obtained; (g) The interaction energies and the average length of hydrogen bonds of PAM-H2O-Gly, PAM-H2O-EG, PVA-H2O-Gly and PAA-H2O-Gly were calculated[51]; (h) The principle diagram and mechanism of antifreeze hydrogel electrolyte[54]; (i) The binding energy of Zn(BF4)2-PAM-H2O system and ZnSO4-PAM-H2O system obtained by DFT simulation[54]; (j) The average number of H-bonds formed between water molecules obtained by MD simulation[54]
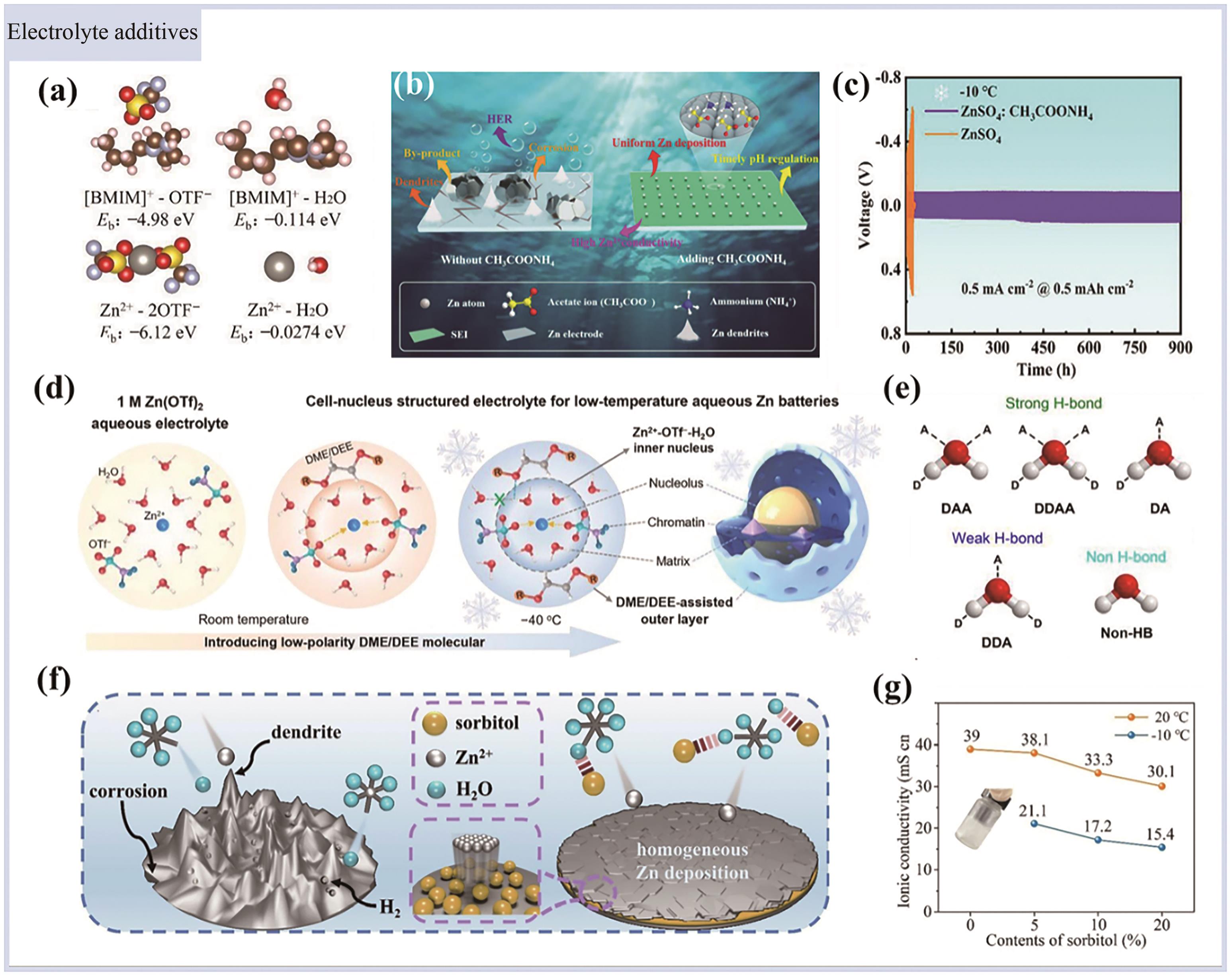
Fig.3 (a) Eb values of different couples[67]; (b) A schematic diagram of Zn surface evolution in electrolytes with/without CH3COONH4 additives[68]; (c) Long-life cycling performance of zinc symmetric cells in ZnSO4∶CH3COONH4 and ZnSO4 electrolytes at -10 ℃[68]; (d) Schematic illustration of the cell-nucleus structured electrolyte design for low temperature aqueous Zn batteries[69]; (e) A schematic pattern of H2O molecules with DAA, DDAA, DA, DDA and non-HB[69]; (f) Comparison of Zn deposition process with or without sorbitol additive[70]; (g) Ionic conductivity values of the electrolytes with different amounts of sorbitol, The inset in g shows the photograph of 2 mol/L ZnSO4 aqueous electrolyte without sorbitol at -10 ℃[70]

Fig.4 (a) Structure diagram of cocrystal solvation structure[8]; (b) The addition of sulfolane destroyed the bulk H—O—H…O—H hydrogen bond network and formed a new type of sulfolane-H2O hydrogen bond[72]; (c) The rate performance of Zn-VOH battery based on 1∶1 electrolyte at -20 ℃[72]; (d) The schematic diagram of the effect of different solvation structures on Zn coating at -20 ℃[74]
Fig.5 (a) The PVA-based gel electrolyte schematic[80]; (b) The cycle performance of the assembled antifreeze FZHSC at 80, 30, 0 and -30 ℃[80]; (c) Schematic diagram of synthesis process of the PDZ-H electrolyte[65]; (d) The ionic conductivity of the PDZ-H electrolyte at different temperature[65]; (e) The schematic diagram of the temperature-regulated hydrogel electrolyte mechanism[81]; (f) When the temperature rises from 30 ℃ to 100 ℃, the apparent volume expansion of the blank electrolyte (BE), the baseline hydrogel electrolyte (BHE) and the TRHE bagged cell[81]

Fig.6 (a) The coordination ratios of different reagents to Zn2+ in Zn(OTf)2-H2O and Zn(OTf)2-H2O/PD electrolytes at 25 ℃[82]; (b) Ignition test of Zn(OTf)2-PD electrolyte and Zn (OTf)2-H2O/PD electrolyte[82]; (c) The acidic environment of the 2 mol/L-P5W5 cosolvent electrolyte with a pH value as low as 3.23 may be beneficial to inhibit the growth of by-products at a wide temperature[76]; the zinc plating demonstration diagram in a water electrolyte without (d) or with (e) polyhydroxy polymer cosolvent[76]; (f) The electrochemical reaction diagram of 1 mol/L ZnSO4 and 30 mol/L ZnCl2 electrolyte[83]; (g) The charge retention and charge recovery of V2O5 cathode after high temperature storage (55 ℃) in various electrolytes[83]
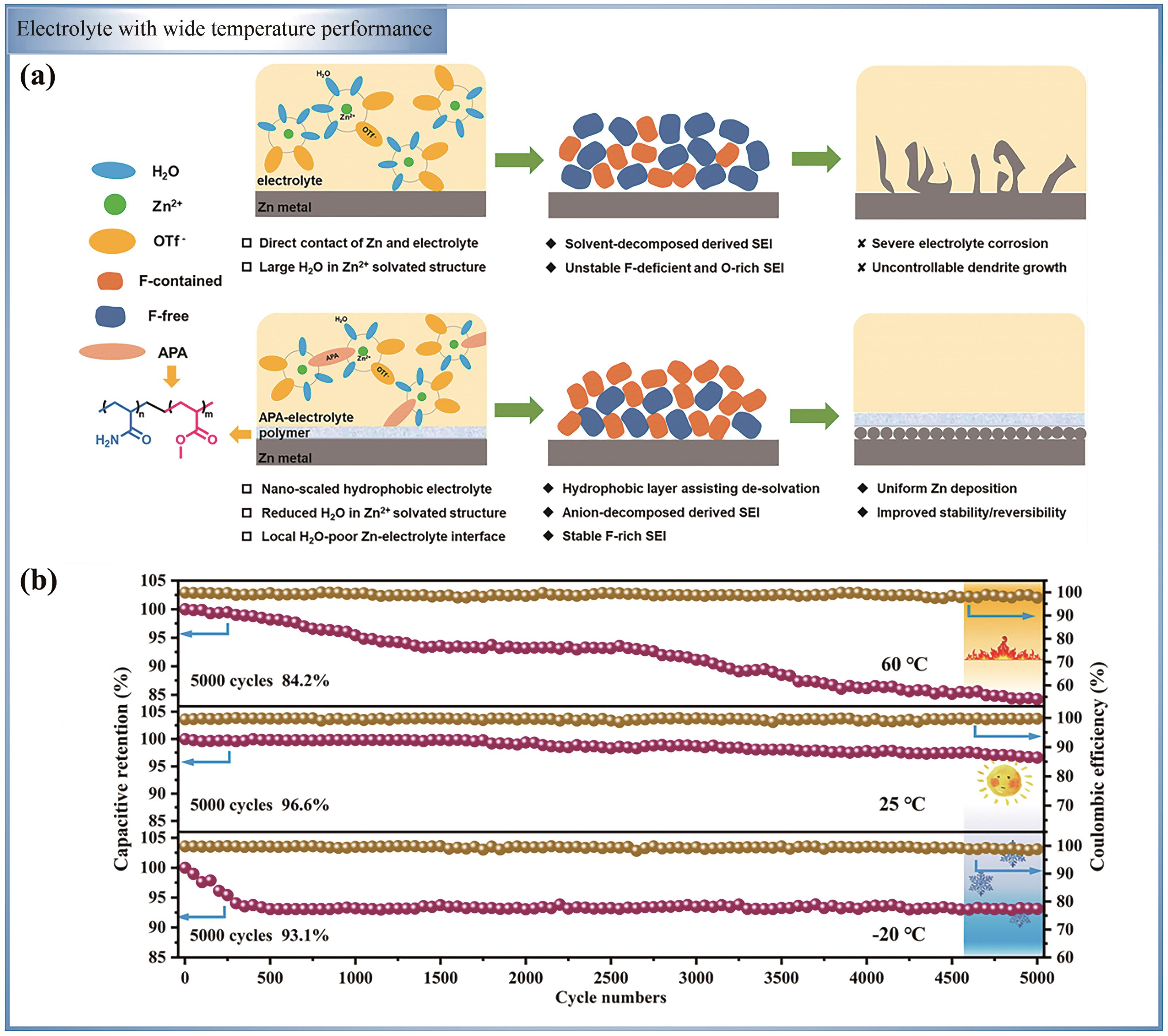
Fig.7 (a) The schematic diagram of the morphological evolution of Zn(OTf)2 aqueous electrolytes without and with APA[85]; (b) The cycle performance of ZIHC based on PMPG-25 GPE energy storage device at 5 A/g is studied[86]
| Electrolyte | Category | Cathode | Ionicconductivity/ (mS·cm-1·℃-1) | Operating temperature range/℃ | Freezing point/℃ | Capacity/((mA·h·g-1)|(A·g-1)) | Year |
|---|---|---|---|---|---|---|---|
| 4 mol/L Zn(BF4)2 | High-concentration electrolyte | TCBQ | 1.47/-70 | -95~25 | -122 | -95 ℃:63.5/0.22 | 2021[ |
| 3.5 mol/L Mg(ClO4)2+1 mol/L Zn(ClO4)2 | High-concentration electrolyte | PTO | 1.41/-70 | -70~25 | -121 | -70 ℃:101.5/0.2 | 2021[ |
| PAM-H2O-Gly-20% | Gel electrolyte | SWCNTs/PANI | 0.096 5/-40 | -40~20 | * | -40 ℃:52/1 | 2022[ |
| 3 mol/L Zn(BF4)2-PAM | Gel electrolyte | PANI | 2.38/-70 | -70~25 | * | -70 ℃:33.5/0.25 | 2023[ |
| 0.5 mol/L [BMIM]OTF+3 mol/L Zn(OTF)2 | Electrolyte additives | H11Al2V6O23.2(HAVO) | 27.7/-40 | -40~25 | * | -30 ℃:165/2 | 2022[ |
| ZnSO4∶CH3COONH4 | Electrolyte additives | Zn | * | -10~25 | * | * | 2022[ |
| DME(DME∶DME+H2O=0.15)+1 mol/L Zn(OTf)2 | Electrolyte additives | V2O5 | 1.06/-40 | -40~25 | -52.4 | -40 ℃:212.4/0.5 | 2023[ |
| 10% C6H14O6 | Electrolyte additives | MnO2 | 17.2/-10 | -10~20 | -18 | -10 ℃:101.9/5 | 2023[ |
| 30% 2-propanol/H2O/Zn(OTf)2 | Eutectic electrolyte | V2O5 | * | -20~25 | <-100 | * | 2022[ |
| Zn(TFSI)2-sulfolane-H2O(1∶1) | Eutectic electrolyte | V2O5·nH2O | * | -20~25 | <-80 | * | 2023[ |
| 3 mol/L ZnOAc1.2Cl1.8-DOL | Eutectic electrolyte | AC | * | -20~20 | * | * | 2023[ |
| PVA/Gly/ZnCl2 | Gel electrolyte | δ-MnO2 | 0.21/-50 | -30~80 | -105.73 | * | 2022[ |
| PAAm/DMSO/Zn(CF3SO3)2 | Gel electrolyte | Zn3V2O8 | * | -40~60 | <-40 | * | 2022[ |
| PEG+AGr | Gel electrolyte | Na5V12O32(NVO) | * | 30~100 | * | * | 2022[ |
| Zn(OTf)2-H2O/PD | Electrolyte additives | Te | 12.8/100 | 30~100 | * | 100 ℃:195.7/2C | 2022[ |
| 50%PEG+50%H2O+2 mol/L Zn(OTf)2 | Electrolyte additives | PANI@V2O5 | 87/100 | -20~80 | -93.04 | 60 ℃:310/2 | 2022[ |
| 30 mol/L ZnCl2 | High-concentration electrolyte | V2O5 | * | 55 | * | * | 2021[ |
| 1% APA+3 mol/L Zn(OTf)2 | Electrolyte additives | Zn | * | -30~50 | * | * | 2023[ |
| PMPG-25 GPE+2 mol/L ZnCl2 | Gel electrolyte | AC/CC | * | -20~60 | * | * | 2023[ |
Table 1 Comparison of physical and chemical properties and electrochemical properties of aqueous zinc ion batteries
| Electrolyte | Category | Cathode | Ionicconductivity/ (mS·cm-1·℃-1) | Operating temperature range/℃ | Freezing point/℃ | Capacity/((mA·h·g-1)|(A·g-1)) | Year |
|---|---|---|---|---|---|---|---|
| 4 mol/L Zn(BF4)2 | High-concentration electrolyte | TCBQ | 1.47/-70 | -95~25 | -122 | -95 ℃:63.5/0.22 | 2021[ |
| 3.5 mol/L Mg(ClO4)2+1 mol/L Zn(ClO4)2 | High-concentration electrolyte | PTO | 1.41/-70 | -70~25 | -121 | -70 ℃:101.5/0.2 | 2021[ |
| PAM-H2O-Gly-20% | Gel electrolyte | SWCNTs/PANI | 0.096 5/-40 | -40~20 | * | -40 ℃:52/1 | 2022[ |
| 3 mol/L Zn(BF4)2-PAM | Gel electrolyte | PANI | 2.38/-70 | -70~25 | * | -70 ℃:33.5/0.25 | 2023[ |
| 0.5 mol/L [BMIM]OTF+3 mol/L Zn(OTF)2 | Electrolyte additives | H11Al2V6O23.2(HAVO) | 27.7/-40 | -40~25 | * | -30 ℃:165/2 | 2022[ |
| ZnSO4∶CH3COONH4 | Electrolyte additives | Zn | * | -10~25 | * | * | 2022[ |
| DME(DME∶DME+H2O=0.15)+1 mol/L Zn(OTf)2 | Electrolyte additives | V2O5 | 1.06/-40 | -40~25 | -52.4 | -40 ℃:212.4/0.5 | 2023[ |
| 10% C6H14O6 | Electrolyte additives | MnO2 | 17.2/-10 | -10~20 | -18 | -10 ℃:101.9/5 | 2023[ |
| 30% 2-propanol/H2O/Zn(OTf)2 | Eutectic electrolyte | V2O5 | * | -20~25 | <-100 | * | 2022[ |
| Zn(TFSI)2-sulfolane-H2O(1∶1) | Eutectic electrolyte | V2O5·nH2O | * | -20~25 | <-80 | * | 2023[ |
| 3 mol/L ZnOAc1.2Cl1.8-DOL | Eutectic electrolyte | AC | * | -20~20 | * | * | 2023[ |
| PVA/Gly/ZnCl2 | Gel electrolyte | δ-MnO2 | 0.21/-50 | -30~80 | -105.73 | * | 2022[ |
| PAAm/DMSO/Zn(CF3SO3)2 | Gel electrolyte | Zn3V2O8 | * | -40~60 | <-40 | * | 2022[ |
| PEG+AGr | Gel electrolyte | Na5V12O32(NVO) | * | 30~100 | * | * | 2022[ |
| Zn(OTf)2-H2O/PD | Electrolyte additives | Te | 12.8/100 | 30~100 | * | 100 ℃:195.7/2C | 2022[ |
| 50%PEG+50%H2O+2 mol/L Zn(OTf)2 | Electrolyte additives | PANI@V2O5 | 87/100 | -20~80 | -93.04 | 60 ℃:310/2 | 2022[ |
| 30 mol/L ZnCl2 | High-concentration electrolyte | V2O5 | * | 55 | * | * | 2021[ |
| 1% APA+3 mol/L Zn(OTf)2 | Electrolyte additives | Zn | * | -30~50 | * | * | 2023[ |
| PMPG-25 GPE+2 mol/L ZnCl2 | Gel electrolyte | AC/CC | * | -20~60 | * | * | 2023[ |
| 1 | WANG Y, XIAO F, CHEN X, et al. Extraordinarily stable and wide‐temperature range sodium/potassium-ion batteries based on 1D SnSe2‐SePAN composite nanofibers[J]. Info Mat, 2023, 5: e12467. |
| 2 | 王丹, 侯现飚, 汪兴坤, 等. 应用于锌空气电池的碳包覆铁基纳米颗粒电催化剂研究进展[J]. 应用化学, 2022, 39(10): 1488-1500. |
| WANG D, HOU X B, WANG X K, et al. Research progress of carbon-encapsulated iron‑based nanoparticles electrocatalysts for zinc‑air batteries[J]. Chin J Appl Chem, 2022, 39(10): 1488-1500. | |
| 3 | 宋林虎, 李世友, 王洁, 等. 锂离子电池电解液除酸除水添加剂的研究进展[J]. 应用化学, 2022, 39(5): 697-706. |
| SONG L H, LI S Y, WANG J, et al. Research progress of additives for acid and water removal in electrolyte of lithium ion battery[J]. Chin J Appl Chem, 2022, 39(5): 697-706. | |
| 4 | ZHENG B, LIN X, ZHANG X, et al. Emerging functional porous polymeric and carbonaceous materials for environmental treatment and energy storage[J]. Adv Funct Mater, 2019, 30(41): 1907006. |
| 5 | LIN X, ZHOU G, LIU J, et al. Rechargeable battery electrolytes capable of operating over wide temperature windows and delivering high safety[J]. Adv Energy Mater, 2020, 10(43): 2001235. |
| 6 | LIN X, ZHOU G, ROBSON M J, et al. Hydrated deep eutectic electrolytes for high‐performance Zn-ion batteries capable of low‐temperature operation[J]. Adv Funct Mater, 2021, 32(14): 2109322. |
| 7 | YUAN Z, XIAO F, FANG Y, et al. Defect engineering on VO2(B) nanoleaves/graphene oxide for the high performance of cathodes of zinc-ion batteries with a wide temperature range[J]. J Power Sources, 2023, 559: 232688. |
| 8 | MA Q, GAO R, LIU Y, et al. Regulation of outer solvation shell toward superior low-temperature aqueous zinc-ion batteries[J]. Adv Mater, 2022, 34(49): 2207344. |
| 9 | YUAN Z, YANG X, LIN C, et al. Progressive activation of porous vanadium nitride microspheres with intercalation-conversion reactions toward high performance over a wide temperature range for zinc-ion batteries[J]. J Colloid Interface Sci, 2023, 640(15): 487-497. |
| 10 | XIONG P, ZHANG Y, ZHANG J, et al. Recent progress of artificial interfacial layers in aqueous Zn metal batteries[J]. Energy Chem, 2022, 4(4): 100076. |
| 11 | YAO Q, XIAO F, LIN C, et al. Regeneration of spent lithium manganate into cation-doped and oxygen-deficient MnO2 cathodes toward ultralong lifespan and wide-temperature-tolerant aqueous Zn-ion batteries[J]. Battery Energy, 2023, 2(4): 20220065. |
| 12 | KUNDU D, HOSSEINI VAJARGAH S, WAN L, et al. Aqueous vs. nonaqueous Zn-ion batteries: consequences of the desolvation penalty at the interface[J]. Energy Environ Sci, 2018, 11(4): 881-892. |
| 13 | SU Z, GUO H, ZHAO C. Rational design of electrode-electrolyte interphase and electrolytes for rechargeable proton batteries[J]. Nano-Micro Lett, 2023, 15(1): 96. |
| 14 | ZHANG Y, CHEN Z, QIU H, et al. Pursuit of reversible Zn electrochemistry: a time-honored challenge towards low-cost and green energy storage[J]. NPG Asia Mater, 2020, 12(1): 4. |
| 15 | ZHU C, ZHOU J, WANG Z, et al. Phase diagrams guided design of low-temperature aqueous electrolyte for Zn metal batteries[J]. Chem Eng J, 2023, 454: 140413. |
| 16 | HAN M M, LI T C, CHEN X, et al. Electrolyte modulation strategies for low-temperature Zn batteries[J]. Small, 2023: 2304901. |
| 17 | WANG N, DONG X, WANG B, et al. Zinc-organic battery with a wide operation-temperature window from -70 to 150 ℃[J]. Angew Chem Int Ed, 2020, 59(34): 14577-14583. |
| 18 | ZHANG W, DOONG Q, WANG J, et al. Failure mechanism, electrolyte design, and electrolyte/electrode interface regulation for low-temperature zinc-based batteries[J]. Small Methods, 2023: 2300324. |
| 19 | YUE F, TIE Z W, DENG S Z, et al. An ultralow temperature aqueous battery with proton chemistry[J]. Angew Chem Int Ed, 2021, 60(25): 13882-13886. |
| 20 | LI M, YANG J, SHI Y, et al. Soluble organic cathodes enable long cycle life, high rate, and wide-temperature lithium-ion batteries[J]. Adv Mater, 2022, 34(5): 2107226. |
| 21 | NIAN Q, LIU S, LIU J, et al. All-climate aqueous dual-ion hybrid battery with ultrahigh rate and ultralong life performance[J]. ACS Appl Energy Mater, 2019, 2(6): 4370-4378. |
| 22 | JIANG L, LU Y, ZHAO C, et al. Building aqueous K-ion batteries for energy storage[J]. Nat Energy, 2019, 4(6): 495-503. |
| 23 | YOU Y, YAO H, XIN S, et al. Subzero-temperature cathode for a sodium-ion battery[J]. Adv Mater, 2016, 28(33): 7243-7248. |
| 24 | NIAN Q, WANG J, LIU S, et al. Aqueous batteries operated at -50 ℃[J]. Angew Chem Int Ed, 2019, 58(47): 16994-16999. |
| 25 | GUO Z, WANG T, WEI H, et al. Ice as solid electrolyte to conduct various kinds of ions[J]. Angew Chem Int Ed, 2019, 58(36): 12569-12573. |
| 26 | WANG X, ZHANG Z, XI B, et al. Advances and perspectives of cathode storage chemistry in aqueous zinc-ion batteries[J]. ACS Nano, 2021, 15(6): 9244-9272. |
| 27 | VERMA V, KUMAR S, MANALASTAS W, et al. Undesired reactions in aqueous rechargeable zinc ion batteries[J]. ACS Energy Lett, 2021, 6(5): 1773-1785. |
| 28 | LIU C, XIE X, LU B, et al. Electrolyte strategies toward better zinc-ion batteries[J]. ACS Energy Lett, 2021, 6(3): 1015-1033. |
| 29 | LIU Z, LUO X, QIN L, et al. Progress and prospect of low-temperature zinc metal batteries[J]. Adv Powder Mater, 2022, 1(2): 100011. |
| 30 | TAMTOGL A, BAHN E, SACCHI M, et al. Motion of water monomers reveals a kinetic barrier to ice nucleation on graphene[J]. Nat Commun, 2021, 12(1): 3120. |
| 31 | RAMANUJAPURAM A, YUSHIN G. Understanding the exceptional performance of lithium-ion battery cathodes in aqueous electrolytes at subzero temperatures[J]. Adv Energy Mater, 2018, 8(35): 1802624. |
| 32 | NIAN Q, SUN T, LIU S, et al. Issues and opportunities on low-temperature aqueous batteries[J]. Chem Eng J, 2021, 423: 130253. |
| 33 | ZHANG Q, MA Y, LU Y, et al. Modulating electrolyte structure for ultralow temperature aqueous zinc batteries[J]. Nat Commun, 2020, 11(1): 4463. |
| 34 | LEONARD D P, WEI Z, CHEN G, et al. Water-in-salt electrolyte for potassium-ion batteries[J]. ACS Energy Lett, 2018, 3(2): 373-374. |
| 35 | LUKATSKAYA M R, FELDBLYUM J I, MACKANIC D G, et al. Concentrated mixed cation acetate “water-in-salt” solutions as green and low-cost high voltage electrolytes for aqueous batteries[J]. Energy Environ Sci, 2018, 11(10): 2876-2883. |
| 36 | ZHANG H, LIU X, LI H, et al. High-voltage operation of a V2O5 cathode in a concentrated gel polymer electrolyte for high-energy aqueous zinc batteries[J]. ACS Appl Mater Interfaces, 2020, 12(13): 15305-15312. |
| 37 | SUN T, ZHENG S, DU H, et al. Synergistic effect of cation and anion for low-temperature aqueous zinc-ion battery[J]. Nanomicro Lett, 2021, 13(1): 204. |
| 38 | SUN T, YUAN X, WANG K, et al. An ultralow-temperature aqueous zinc-ion battery[J]. J Mater Chem A, 2021, 9(11): 7042-7047. |
| 39 | PU Y, WANG C, FENG J, et al. Organics-free aqueous hybrid electrolyte for high-performance zinc ion hybrid capacitors operating at low temperature[J]. J Power Sources, 2023, 571: 233061. |
| 40 | BORODIN O, SELF J, PERSSON K A, et al. Uncharted waters: super-concentrated electrolytes[J]. Joule, 2020, 4(1): 69-100. |
| 41 | WANG Z, LI H, TANG Z, et al. Hydrogel electrolytes for flexible aqueous energy storage devices[J]. Adv Funct Mater, 2018, 28(48): 1804560. |
| 42 | ZHAO S, ZUO Y, LIU T, et al. Multi-functional hydrogels for flexible zinc-based batteries working under extreme conditions[J]. Adv Energy Mater, 2021, 11(34): 2101749. |
| 43 | LOU Z, SHEN G. Flexible image sensors with semiconducting nanowires for biomimic visual applications[J]. Small Struct, 2021, 2(7): 2000152. |
| 44 | MA L, CHEN S, WANG D, et al. Super-stretchable zinc-air batteries based on an alkaline-tolerant dual-network hydrogel electrolyte[J]. Adv Energy Mater, 2019, 9(12): 1803046. |
| 45 | MA L, ZHAO Y, JI X, et al. A usage scenario independent “air chargeable” flexible zinc ion energy storage device[J]. Adv Energy Mater, 2019, 9(19): 1900509. |
| 46 | MO F, LIANG G, MENG Q, et al. A flexible rechargeable aqueous zinc manganese-dioxide battery working at -20 ℃[J]. Energy Environ Sci, 2019, 12(2): 706-715. |
| 47 | HAN L, LIU K, WANG M, et al. Mussel-inspired adhesive and conductive hydrogel with long-lasting moisture and extreme temperature tolerance[J]. Adv Funct Mater, 2018, 28(3): 1704195. |
| 48 | LIU T, LIU M, DOU S, et al. Triboelectric-nanogenerator-based soft energy-harvesting skin enabled by toughly bonded elastomer/hydrogel hybrids[J]. ACS Nano, 2018, 12(3): 2818-2826. |
| 49 | YANG J, GAO L, LIU M, et al. Advanced biotechnology for cell cryopreservation[J]. Trans Tianjin Univ, 2019, 26(6): 409-423. |
| 50 | YUK H, ZHANG T, PARADA G A, et al. Skin-inspired hydrogel-elastomer hybrids with robust interfaces and functional microstructures[J]. Nat Commun, 2016, 7: 12028. |
| 51 | WANG R, YAO M, HUANG S, et al. An anti-freezing and anti-drying multifunctional gel electrolyte for flexible aqueous zinc-ion batteries[J]. Sci China Mater, 2022, 65(8): 2189-2196. |
| 52 | LIU L, DOU Q, SUN Y, et al. A moisture absorbing gel electrolyte enables aqueous and flexible supercapacitors operating at high temperatures[J]. J Mater Chem A, 2019, 7(35): 20398-20404. |
| 53 | SUN Y, MA H, ZHANG X, et al. Salty ice electrolyte with superior ionic conductivity towards low‐temperature aqueous zinc ion hybrid capacitors[J]. Adv Funct Mater, 2021, 31(28): 2101277. |
| 54 | SHI Y, WANG R, BI S, et al. An anti‐freezing hydrogel electrolyte for flexible zinc‐ion batteries operating at -70 ℃[J]. Adv Funct Mater, 2023, 33(24): 2214546. |
| 55 | CHEN M, CHEN J, ZHOU W, et al. Realizing an all-round hydrogel electrolyte toward environmentally adaptive dendrite-free aqueous Zn-MnO2 batteries[J]. Adv Mater, 2021, 33(9): 2007559. |
| 56 | YAN C, WANG Y, DENG X, et al. Cooperative chloride hydrogel electrolytes enabling ultralow-temperature aqueous zinc ion batteries by the hofmeister effect[J]. Nanomicro Lett, 2022, 14(1): 98. |
| 57 | GAO J, XIE X, LIANG S, et al. Inorganic colloidal electrolyte for highly robust zinc-ion batteries[J]. Nanomicro Lett, 2021, 13(1): 69. |
| 58 | LI C, XIE X, LIU H, et al. Integrated 'all-in-one' strategy to stabilize zinc anodes for high-performance zinc-ion batteries[J]. Natl Sci Rev, 2022, 9(3): nwab177. |
| 59 | TANG Y, LIU C, ZHU H, et al. Ion-confinement effect enabled by gel electrolyte for highly reversible dendrite-free zinc metal anode[J]. Energy Storage Mater, 2020, 27: 109-116. |
| 60 | CURSARU B, STANESCU P O, TEODORESCU M. The states of water in hydrogels synthesized from diepoxy-terminated poly(ethylene glycol)s and aliphatic polyamines[J]. UPB Sci Bull Ser B, 2010, 72(4): 99-15. |
| 61 | JIANG H, SHIN W, MA L, et al. A high‐rate aqueous proton battery delivering power below -78 ℃ via an unfrozen phosphoric acid[J]. Adv Energy Mater, 2020, 10(28): 2000968. |
| 62 | LU C, CHEN X. All-temperature flexible supercapacitors enabled by antifreezing and thermally stable hydrogel electrolyte[J]. Nano Lett, 2020, 20(3): 1907-1914. |
| 63 | WANG A, ZHOU W, HUANG A, et al. Developing improved electrolytes for aqueous zinc-ion batteries to achieve excellent cyclability and antifreezing ability[J]. J Colloid Interface Sci, 2021, 586: 362-370. |
| 64 | HUANG S, HE S, LI Y, et al. Hydrogen bond acceptor lined hydrogel electrolyte toward dendrite-free aqueous Zn ion batteries with low temperature adaptability[J]. Chem Eng J, 2023, 464: 142607. |
| 65 | LU H, HU J, WANG L, et al. Multi-component crosslinked hydrogel electrolyte toward dendrite-free aqueous Zn ion batteries with high temperature adaptability[J]. Adv Funct Mater, 2022, 32(19): 2112540. |
| 66 | WANG J, ZHU Q, LI F, et al. Low-temperature and high-rate Zn metal batteries enabled by mitigating Zn2+ concentration polarization[J]. Chem Eng J, 2022, 433: 134589. |
| 67 | CHEN J, ZHOU W, QUAN Y, et al. Ionic liquid additive enabling anti-freezing aqueous electrolyte and dendrite-free Zn metal electrode with organic/inorganic hybrid solid electrolyte interphase layer[J]. Energy Storage Mater, 2022, 53: 629-637. |
| 68 | LIN C, YANG X, XIONG P, et al. High-rate, large capacity, and long life dendrite-free Zn metal anode enabled by trifunctional electrolyte additive with a wide temperature range[J]. Adv Sci, 2022, 9(21): 2201433. |
| 69 | DONG Y, ZHANG N, WANG Z, et al. Cell-nucleus structured electrolyte for low-temperature aqueous zinc batteries[J]. J Energy Chem, 2023, 83: 324-332. |
| 70 | QUAN Y, YANG M, CHEN M, et al. Electrolyte additive of sorbitol rendering aqueous zinc-ion batteries with dendrite-free behavior and good anti-freezing ability[J]. Chem Eng J, 2023, 458: 141392. |
| 71 | JIANG L, DONG D, LU Y. Design strategies for low temperature aqueous electrolytes[J]. Nano Res Energy, 2022, 1(1): 9120003. |
| 72 | LI M, WANG X, HU J, et al. Comprehensive H2O molecules regulation via deep eutectic solvents for ultra-stable zinc metal anode[J]. Angew Chem Int Ed, 2023, 62(8): e202215552. |
| 73 | ZHAO Y, LIU H, HUYAN Y, et al. In situ construction of a stable composite solid electrolyte interphase for dendrite-free Zn batteries[J]. J Energy Chem, 2023, 79: 450-458. |
| 74 | LU X, LIU Z, AMARDEEP A, et al. Ultra-stable zinc metal anodes at -20 ℃ through eutectic solvation sheath in chlorine‐functionalized eutectic electrolytes with 1,3‐dioxolane[J]. Angew Chem Int Ed, 2023, 62(33): e202307475. |
| 75 | ZHANG C, ZHANG L, YU G. Eutectic electrolytes as a promising platform for next-generation electrochemical energy storage[J]. Acc Chem Res, 2020, 53(8): 1648-1659. |
| 76 | ZHOU J, YUAN H, LI J, et al. Highly reversible and stable Zn metal anode under wide temperature conditions enabled by modulating electrolyte chemistry[J]. Chem Eng J, 2022, 442: 136218. |
| 77 | HOU Z, LU Z, CHEN Q, et al. Realizing wide-temperature Zn metal anodes through concurrent interface stability regulation and solvation structure modulation[J]. Energy Storage Mater, 2021, 42: 517-525. |
| 78 | XIE X, FU H, FANG Y, et al. Manipulating ion concentration to boost two‐electron Mn4+/Mn2+ redox kinetics through a colloid electrolyte for high‐capacity zinc batteries[J]. Adv Energy Mater, 2021, 12(5): 2102393. |
| 79 | WANG H, CHEN Z, JI Z, et al. Temperature adaptability issue of aqueous rechargeable batteries[J]. Mater Today Energy, 2021, 19: 100577. |
| 80 | JIANG Y, MA K, SUN M, et al. All‐climate stretchable dendrite‐free Zn-ion hybrid supercapacitors enabled by hydrogel electrolyte engineering[J]. Energy Environ Mater, 2022, 6(2): e12357. |
| 81 | MENG Y, ZHANG L, PENG M, et al. Developing thermoregulatory hydrogel electrolyte to overcome thermal runaway in zinc-ion batteries[J]. Adv Funct Mater, 2022, 32(46): 2206653. |
| 82 | WANG J, YANG Y, WANG Y, et al. Working aqueous Zn metal batteries at 100 ℃[J]. ACS Nano, 2022, 16(10): 15770-15778. |
| 83 | TANG X, WANG P, BAI M, et al. Unveiling the reversibility and stability origin of the aqueous V2O5-Zn batteries with a ZnCl2 “Water-in-Salt” electrolyte[J]. Adv Sci, 2021, 8(23): 2102053. |
| 84 | SUN L, SONG Z, DENG C, et al. Electrolytes for aqueous Zn-ion batteries working in wide-temperature range: progress and perspective[J]. Batteries, 2023, 9(7): 386. |
| 85 | NIU B, LI Z, LUO D, et al. Nano-scaled hydrophobic confinement of aqueous electrolyte by a nonionic amphiphilic polymer for long-lasting and wide-temperature Zn-based energy storage[J]. Energy Environ Sci, 2023, 16(4): 1662-1675. |
| 86 | WAN L, ZHANG H, QU M, et al. Polyanion-induced single zinc-ion gel polymer electrolytes for wide-temperature and interfacial stable zinc-ion hybrid capacitors[J]. Energy Storage Mater, 2023, 63: 102982. |
| [1] | Yu-Hua XIONG, Lei ZHOU, Shi-Zhong YANG, Bo-Zhong MU. Enzymatic Properties and Gel Breaking Performance of Low-temperature β-Mannanase [J]. Chinese Journal of Applied Chemistry, 2023, 40(1): 134-145. |
| [2] | LIU Xide, ZHOU Di, ZHANG Zhaoshun. Wastewater Treatment and Recycle of Zinc Chloride in 1,2,3,9-Tetrahydro-4H-carbazol-4-one Production Process [J]. Chinese Journal of Applied Chemistry, 2020, 37(1): 117-122. |
| [3] | HAO Bin,ZHAO Wenwu,YU Jianyuan,LIU Jinqiang,LIU Jian,DONG Xiuzhen,WANG Xiuwen. Preparation and Luminescence Property of Ba5-3x/2B4O11∶xEu3+ Phosphor [J]. Chinese Journal of Applied Chemistry, 2019, 36(5): 548-553. |
| [4] | ZHANG Qingxuan, LI Jintao, ZHANG Meng. Characteristics of Pyrolysis-Oxidation Reactions of Merey Crude Oil in Air and Nitrogen [J]. Chinese Journal of Applied Chemistry, 2018, 35(12): 1470-1477. |
| [5] | YE Xuefang, YI Shouzhi, XIAO Qinggui, SUN Randi, TIAN Ying, XU Hongbin, ZHANG Yi. Measurement of the Metastable Zone of Sodium Perborate Tetrahydrate [J]. Chinese Journal of Applied Chemistry, 2016, 33(3): 350-356. |
| [6] | BAI Tian, GUO Anru, LI Ruijie, LIU Chang, XIAO Dehai. Effect of Diluent on Performances of High-Temperature Resistance Epoxy Resin [J]. Chinese Journal of Applied Chemistry, 2016, 33(12): 1401-1407. |
| [7] | DENG Mengmeng1, WANG Yongli1, ZHANG Zhiyong1*, LIU Yun1, WEI Benmei1, XUAN Li2. Synthesis and Low-temperature Viscosity Characters of Difluorometheneoxy-bridged Alkylbenzene Compounds [J]. Chinese Journal of Applied Chemistry, 2012, 29(09): 1093-1095. |
| [8] | ZHANG Hai-Jiao1, LIU Jie-Xiang1, ZHANG Xiao-Guang2*, HAN En-Shan1. The Influence of Toluene on Conductivity Behavior of Water/AOT/Alkanol Reverse Microemulsions [J]. Chinese Journal of Applied Chemistry, 2010, 27(11): 1359-1361. |
| [9] | SHI Zai-Feng*, SONG Xue-Fang, LI Juan. Synthesis of Fe3+-doped Nanosized TiO2 Photocatalyst with Sol-gel Based Self-propagating High-temperature Synthesis Process [J]. Chinese Journal of Applied Chemistry, 2010, 27(10): 1177-1181. |
| [10] | YU Yao1, TONG Wei1, SU Ya-Bin2, DING Wei1*. Effect of Additives and Structure on Adsorption of Pentadecylaryl Sulfonates on Oil Sands [J]. Chinese Journal of Applied Chemistry, 2010, 27(04): 466-469. |
| [11] | AI Lun-Hong*, JIANG Jing. Low-temperature Synthesis, Spectral Characterization and Magnetic Properties of CoFe2O4 Nanocrystals [J]. Chinese Journal of Applied Chemistry, 2010, 27(01): 78-81. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||