应用化学 ›› 2022, Vol. 39 ›› Issue (11): 1746-1756.DOI: 10.19894/j.issn.1000-0518.220069
亚铁氰化铜-聚丙烯酰胺/羧甲基纤维素/石墨烯复合水凝胶的制备及铷吸附性能
- 合肥工业大学化学与化工学院,合肥 230009
-
收稿日期:2022-03-14接受日期:2022-06-08出版日期:2022-11-01发布日期:2022-11-09 -
通讯作者:袁定坤 -
基金资助:科技部“科技助力经济2020”重点专项(SQ2020YFF0412719)
Preparation of Copper Ferrocyanide⁃Polyacrylamide/ Carboxymethyl Cellulose/Graphene Composite Hydrogel and Its Adsorption Performance of Rubidium
Ding-Kun YUAN( ), Wei-Fan CHU, Jia-Hui NI
), Wei-Fan CHU, Jia-Hui NI
- School of Chemistry and Chemical Engineering,Hefei University of Technology,Hefei 230009,China
-
Received:2022-03-14Accepted:2022-06-08Published:2022-11-01Online:2022-11-09 -
Contact:Ding-Kun YUAN -
About author:yuandingkun1124@163.com
-
Supported by:the 2020 Key Science and Technology Ministry of Power Economy Special(SQ2020YFF0412719)
摘要:
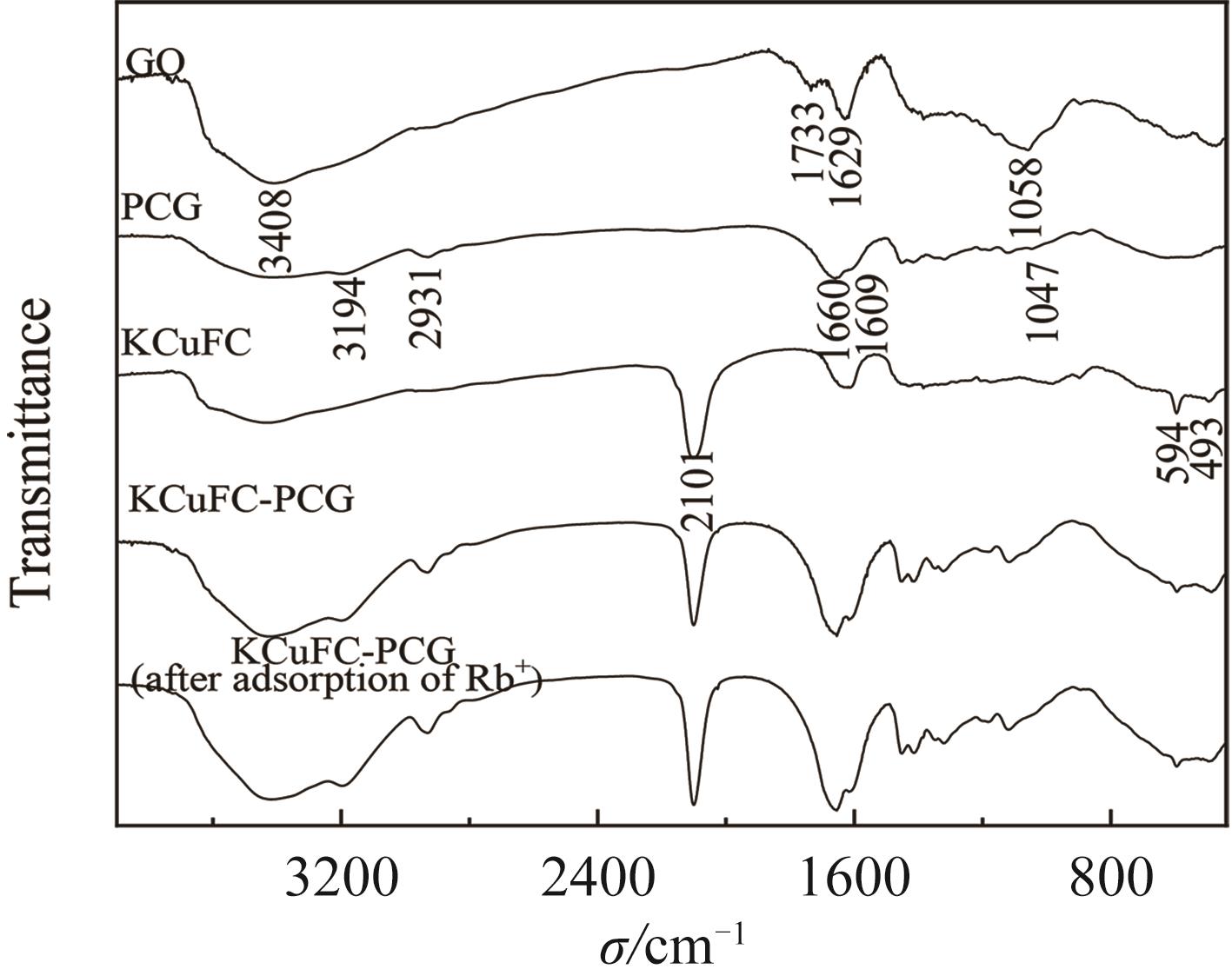
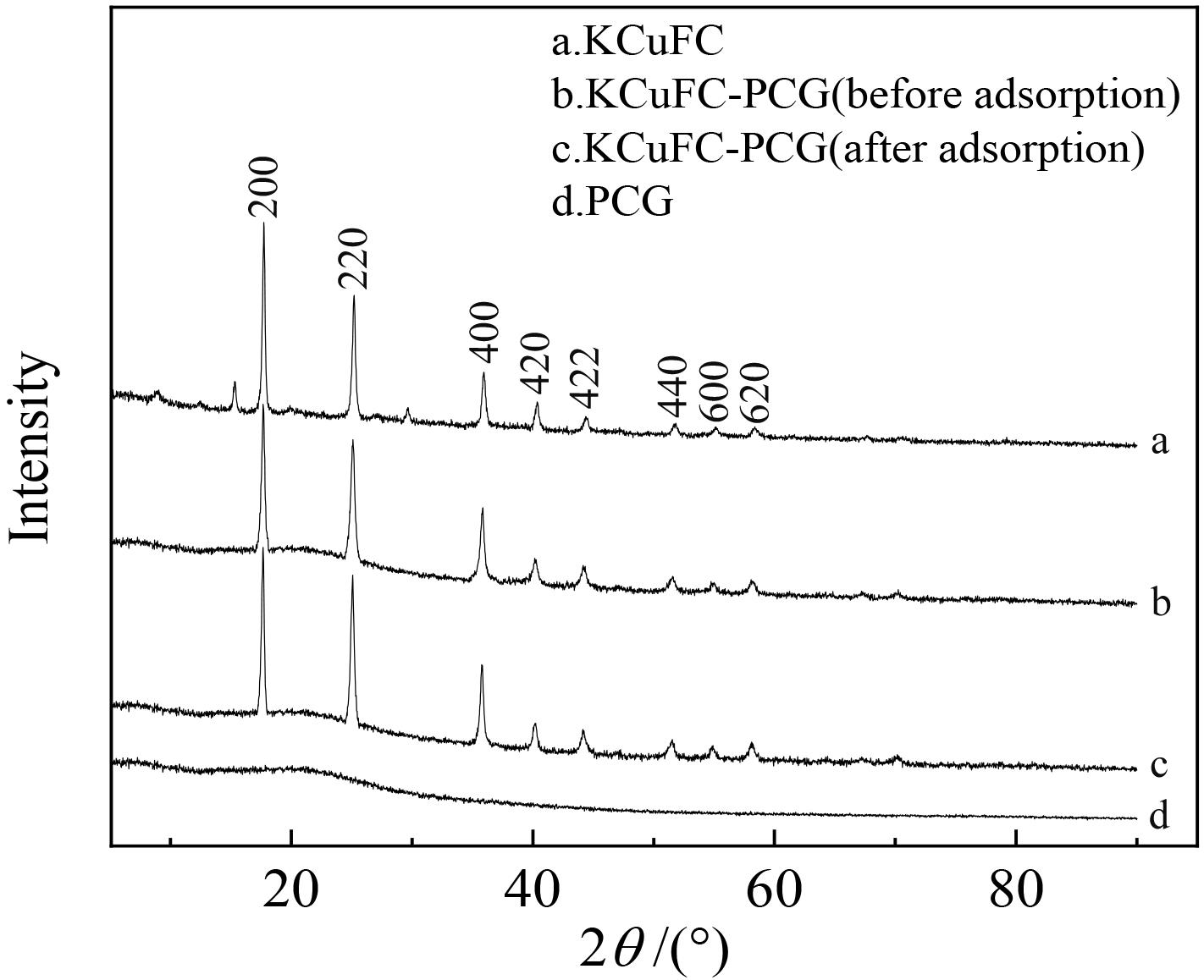
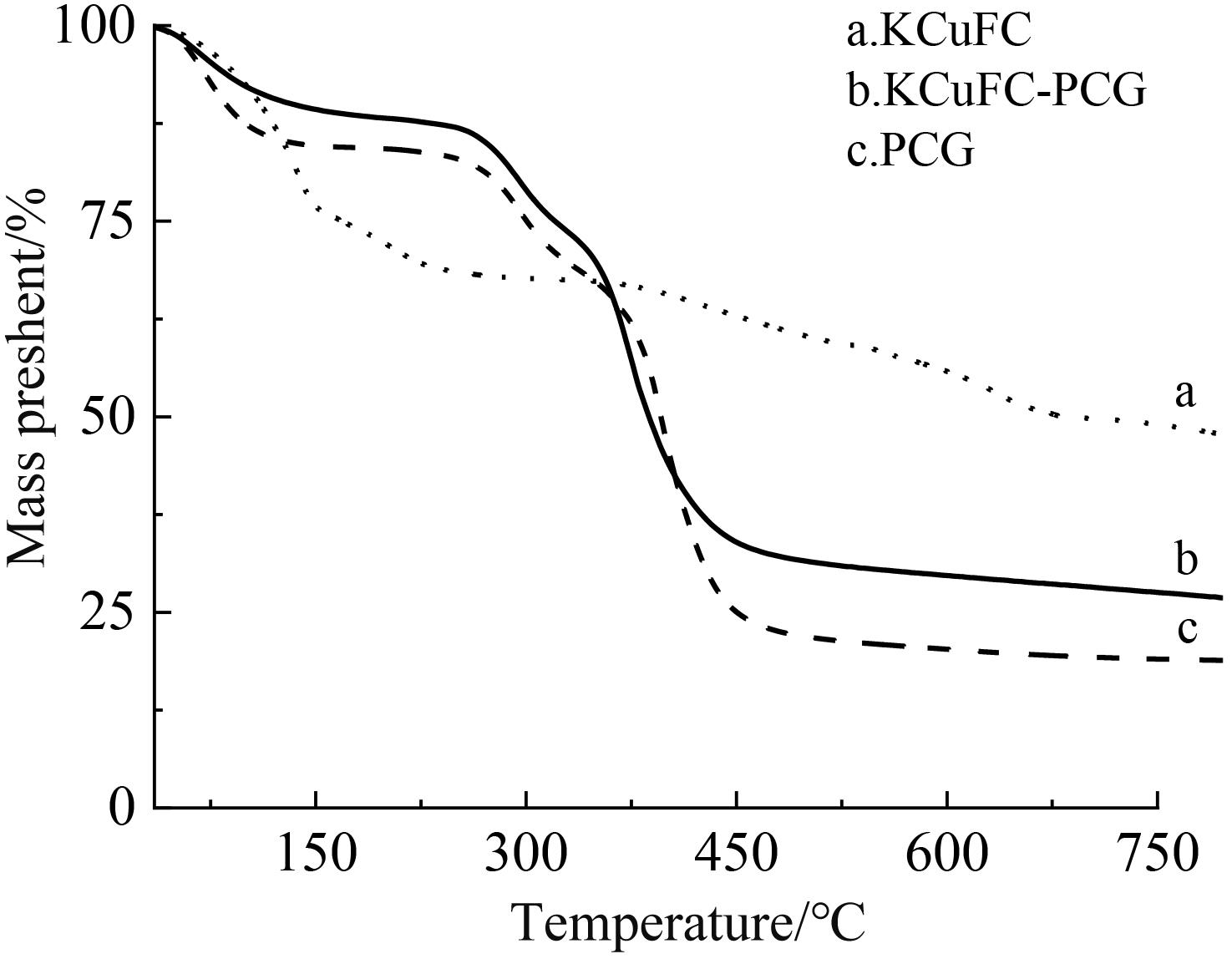
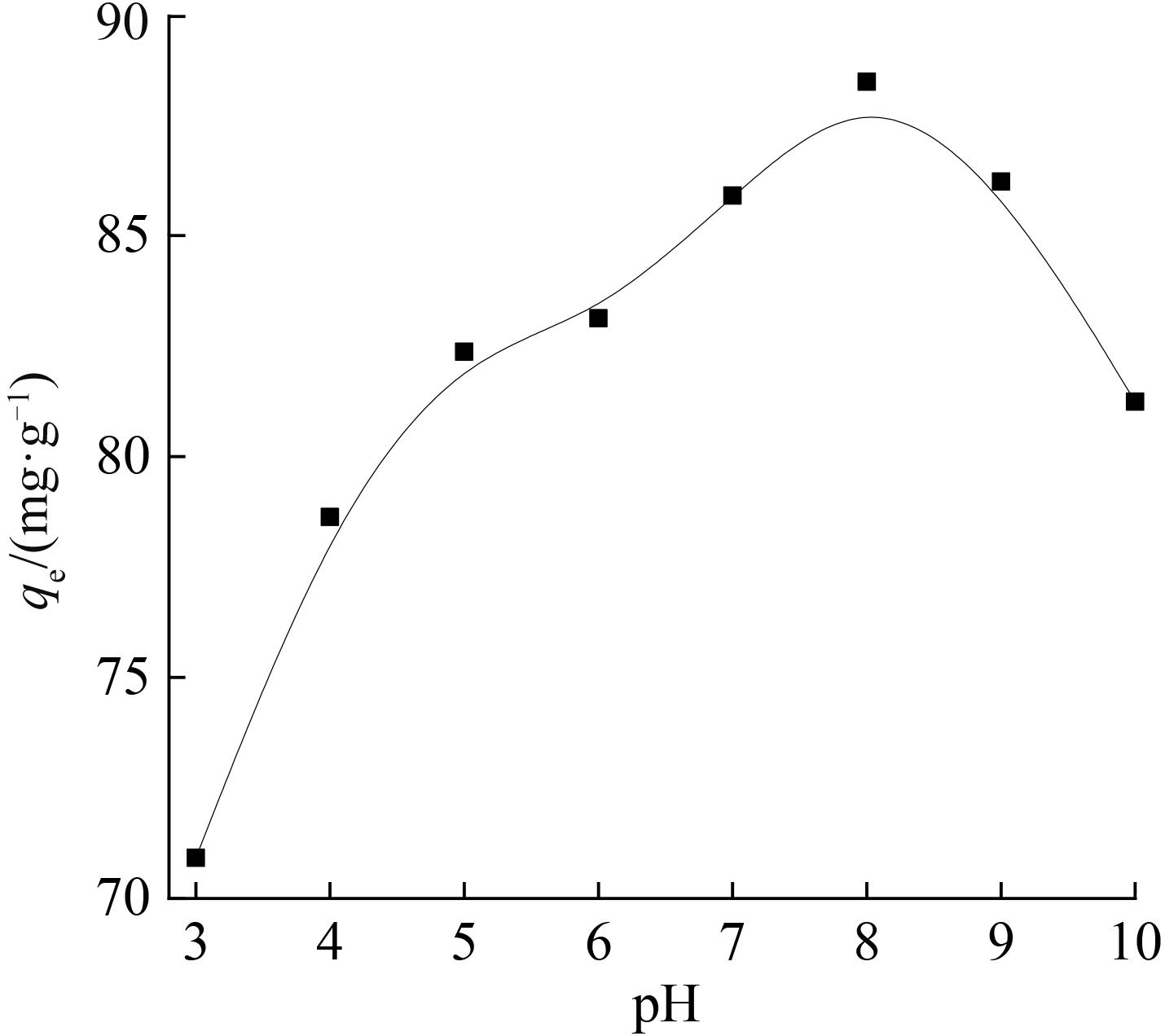
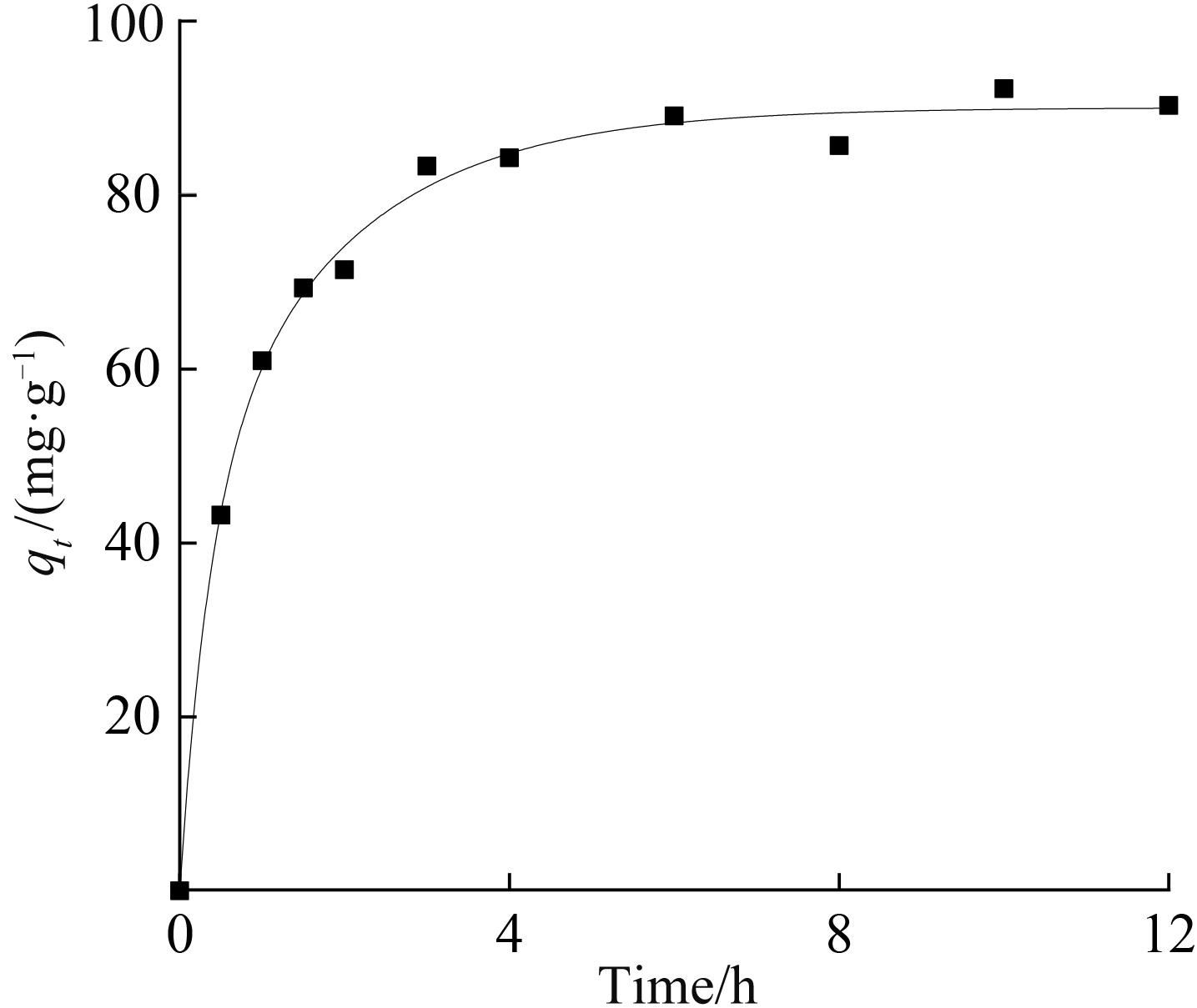
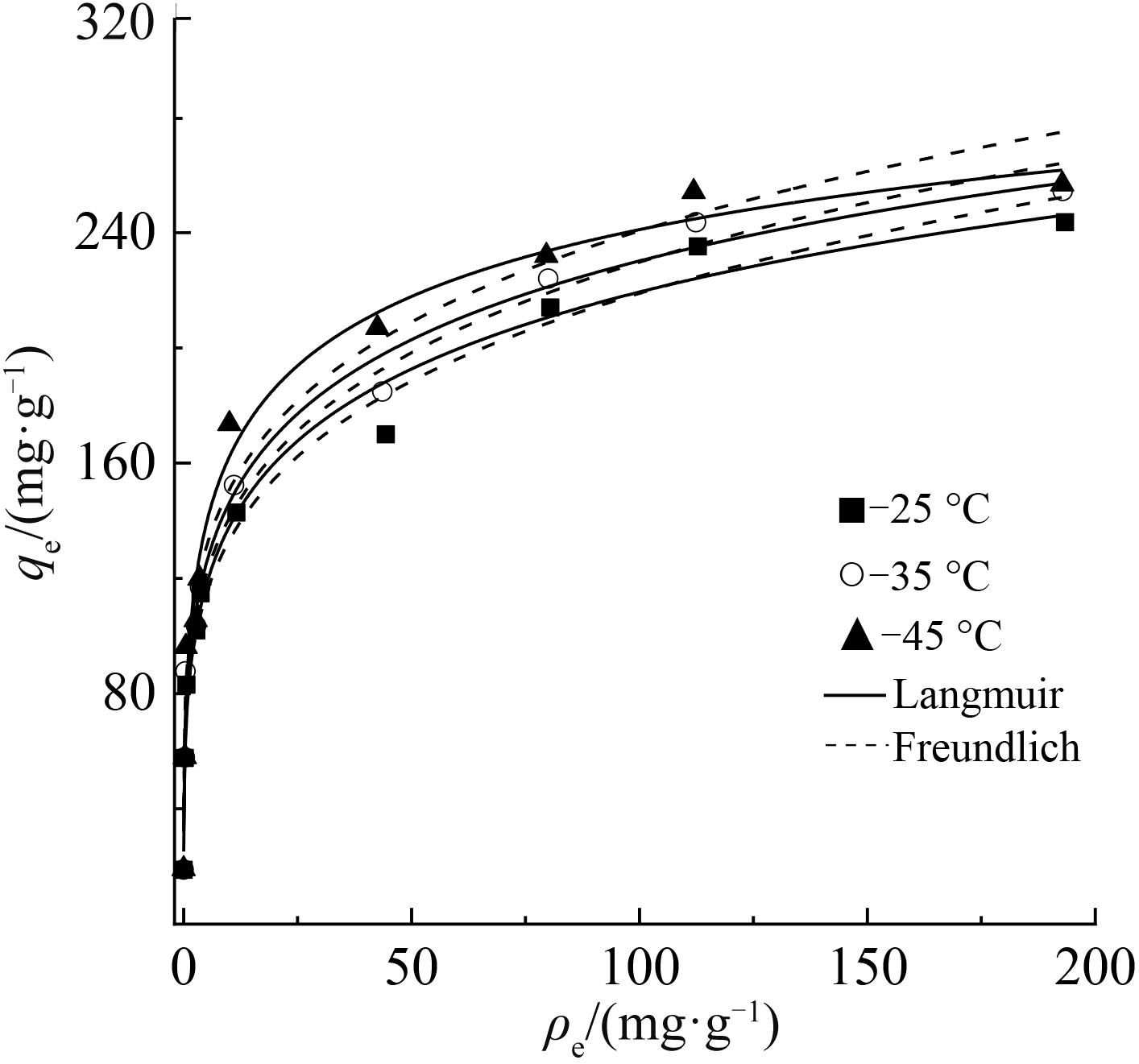
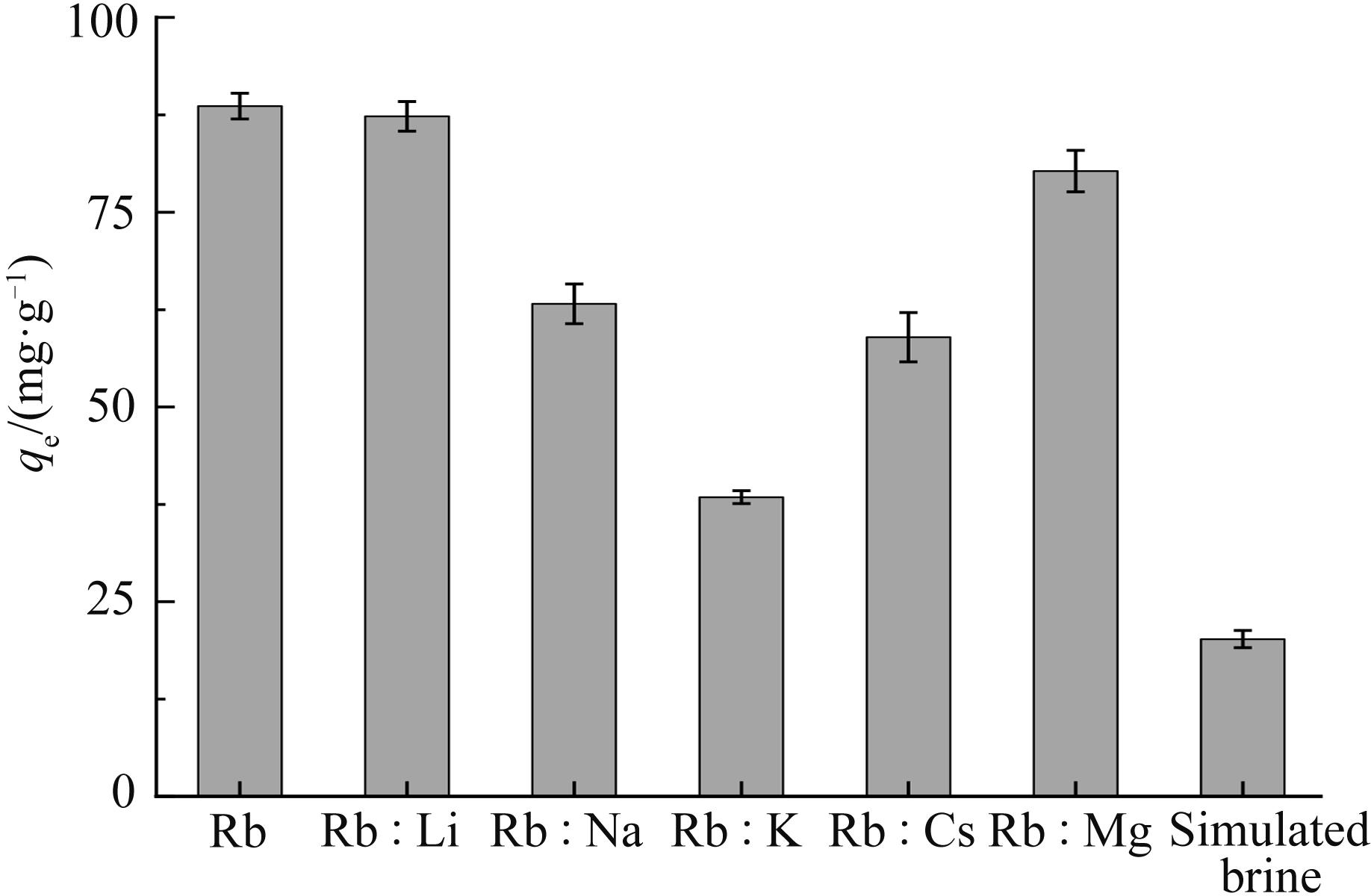
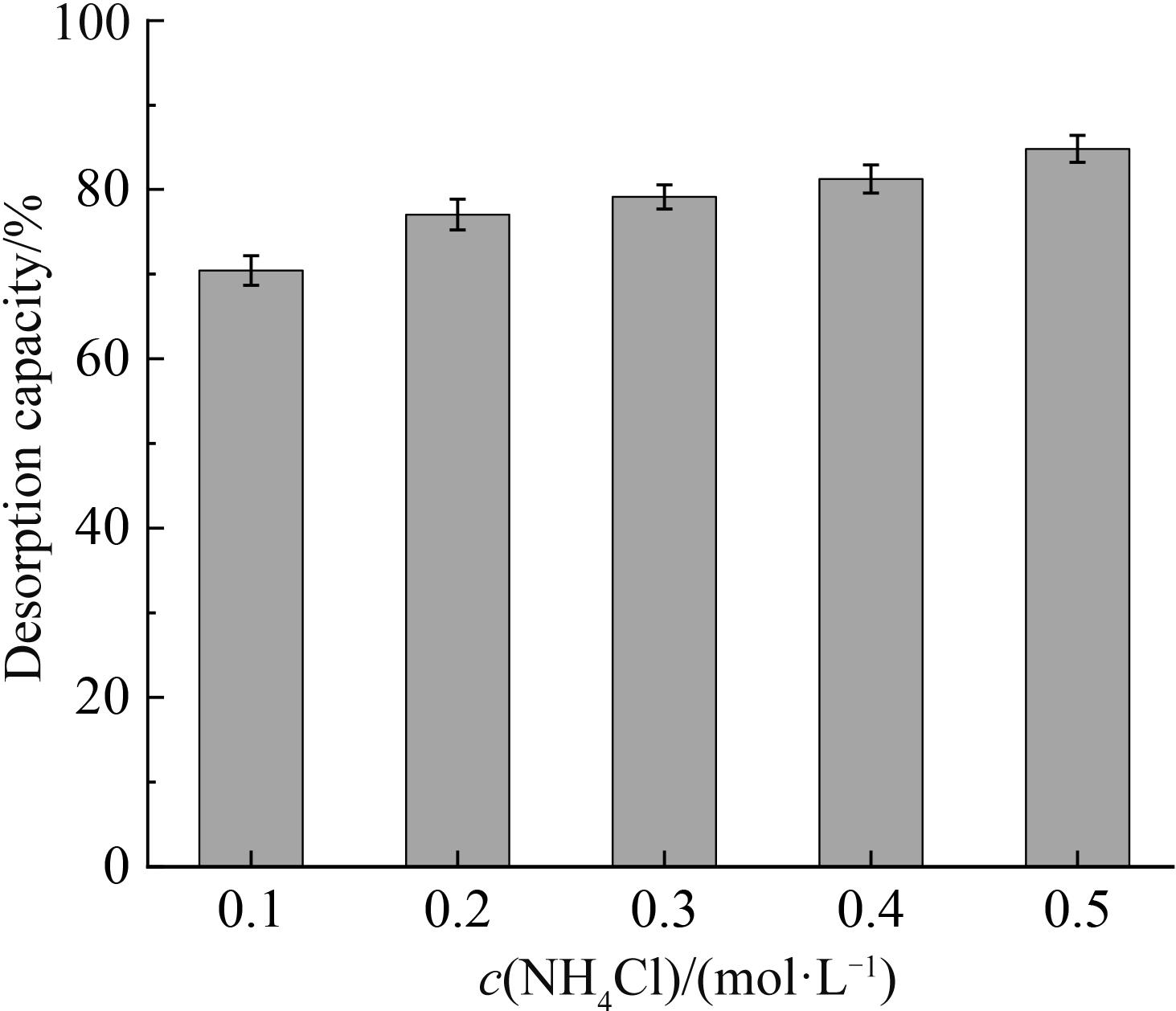
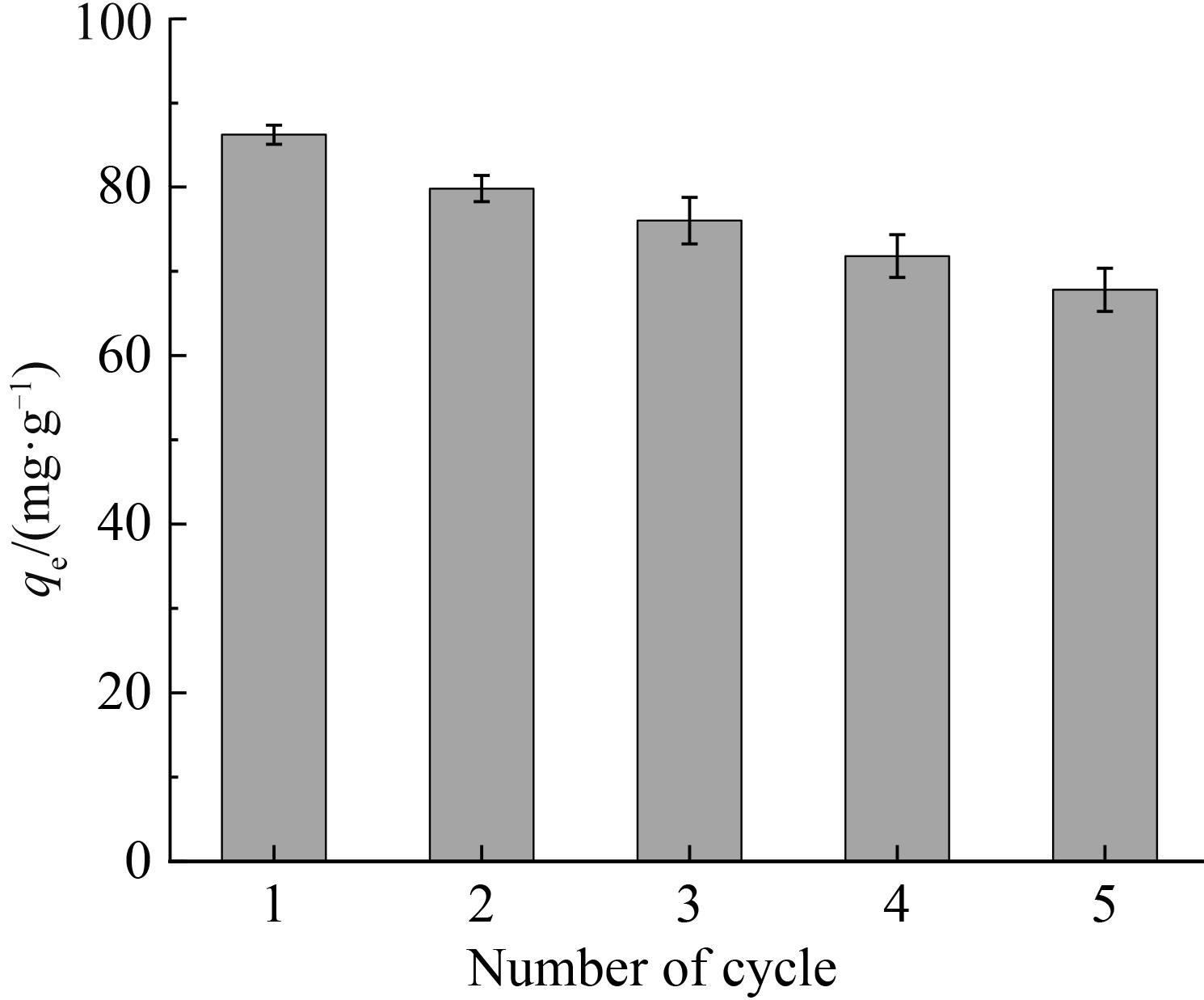
铷是一种稀贵的碱金属,具有很高的经济价值和广阔应用前景,从卤水中有效提取铷具有重要意义。通过热引发聚合法合成水凝胶基质(聚丙烯酰胺/羧甲基纤维素/氧化石墨烯水凝胶,PCG)固定亚铁氰化铜(KCuFC),制备了一种新型的铷(Rb+)吸附剂(KCuFC-PCG)。采用物理化学方法对KCuFC-PCG的结构和性质进行了表征。通过批量吸附实验,研究了pH值、吸附时间、Rb+初始质量浓度、温度和竞争离子对吸附的影响。结果表明,KCuFC-PCG吸附剂在pH值(5~9)范围内表现出良好的吸附能力,pH=8时表现最佳;在Rb+质量浓度为5 mg/L,pH=8,吸附6 h达到吸附平衡,KCuFC-PCG的对Rb+的吸附量为89.12 mg/g;动力学行为可用准二级动力学模型来描述,表明化学吸附为速率控制步骤;吸附过程符合Langmuir等温吸附模型,为单分子层吸附,最大吸附量为258.4 mg/g。以0.2 mol/L NH4Cl作为解吸剂,解吸3 h,解吸率为77%。
中图分类号:
引用本文
袁定坤, 褚维凡, 倪加惠. 亚铁氰化铜-聚丙烯酰胺/羧甲基纤维素/石墨烯复合水凝胶的制备及铷吸附性能[J]. 应用化学, 2022, 39(11): 1746-1756.
Ding-Kun YUAN, Wei-Fan CHU, Jia-Hui NI. Preparation of Copper Ferrocyanide⁃Polyacrylamide/ Carboxymethyl Cellulose/Graphene Composite Hydrogel and Its Adsorption Performance of Rubidium[J]. Chinese Journal of Applied Chemistry, 2022, 39(11): 1746-1756.
平衡吸附容量 实验值 qe,exp/(mg·g-1) | 准一阶模型 Pseudo?first?order model | 准二阶模型 Pseudo?second?order model | ||||
|---|---|---|---|---|---|---|
吸附速率常数 k1 | 平衡吸附容量计算值 qe1cal/(mg·g-1) | 线性决定系数 R2 | 吸附速率常数 k2 | 平衡吸附容量计算值 qe2cal/(mg·g-1) | 线性决定系数 R2 | |
| 90.07 | 0.449 8 | 45.959 | 0.714 | 0.018 9 | 95.147 | 0.998 |
表1 吸附Rb+的准一级和准二级动力学拟合参数
Table 1 Pseudo first and second order model parameters for the adsorption of Rb+
平衡吸附容量 实验值 qe,exp/(mg·g-1) | 准一阶模型 Pseudo?first?order model | 准二阶模型 Pseudo?second?order model | ||||
|---|---|---|---|---|---|---|
吸附速率常数 k1 | 平衡吸附容量计算值 qe1cal/(mg·g-1) | 线性决定系数 R2 | 吸附速率常数 k2 | 平衡吸附容量计算值 qe2cal/(mg·g-1) | 线性决定系数 R2 | |
| 90.07 | 0.449 8 | 45.959 | 0.714 | 0.018 9 | 95.147 | 0.998 |
温度 Temperature/℃ | 朗格缪尔吸附等温模型 Langmuir | 弗伦德里希吸附等温模型 | ||||
|---|---|---|---|---|---|---|
| Freundlich | ||||||
最大吸附量 qmax/(mg·g-1) | 吸附平衡常数 KL/(L·mg-1) | 线性决定系数 R2 | 非均质参数 n | 吸附平衡常数 KF | 线性决定系数 R2 | |
| 25 | 243.90 | 0.185 3 | 0.990 | 3.799 5 | 70.277 4 | 0.932 |
| 35 | 254.45 | 0.201 3 | 0.993 | 3.852 5 | 75.010 6 | 0.924 |
| 45 | 258.40 | 0.270 3 | 0.997 | 3.849 7 | 79.653 0 | 0.934 |
表2 吸附Rb+的Langmuir和Freundlich吸附等温线拟合参数
Table 2 Rb+ adsorption parameters obtained from fitting with Langmuir equation and Freundlich equation
温度 Temperature/℃ | 朗格缪尔吸附等温模型 Langmuir | 弗伦德里希吸附等温模型 | ||||
|---|---|---|---|---|---|---|
| Freundlich | ||||||
最大吸附量 qmax/(mg·g-1) | 吸附平衡常数 KL/(L·mg-1) | 线性决定系数 R2 | 非均质参数 n | 吸附平衡常数 KF | 线性决定系数 R2 | |
| 25 | 243.90 | 0.185 3 | 0.990 | 3.799 5 | 70.277 4 | 0.932 |
| 35 | 254.45 | 0.201 3 | 0.993 | 3.852 5 | 75.010 6 | 0.924 |
| 45 | 258.40 | 0.270 3 | 0.997 | 3.849 7 | 79.653 0 | 0.934 |
组分 Components | PCG | KCuFC | KCuFC?PCG |
|---|---|---|---|
| qe/(mg·g-1) | 2.13 | 91.63 | 89.24 |
表3 各组分对Rb+的吸附性能
Table 3 Adsorption performance of each component on Rb+
组分 Components | PCG | KCuFC | KCuFC?PCG |
|---|---|---|---|
| qe/(mg·g-1) | 2.13 | 91.63 | 89.24 |
卤水组成 Composition | Na质量浓度 ρ(Na)/(g·L-1) | Mg质量浓度 ρ(Mg)/(g·L-1) | K质量浓度 ρ(K)/(g·L-1) | Li质量浓度 ρ(Li)/(mg·L-1) | Rb质量浓度 ρ(Rb)/(mg·L-1) | Cs质量浓度 ρ(Cs)/(mg·L-1) |
|---|---|---|---|---|---|---|
吸附前 Before adsorption | 1.12 | 0.91 | 2.88 | 2.81 | 10.9 | 0.2 |
吸附后 After adsorption | 1.11 | 0.88 | 2.93 | 2.72 | 4.85 | 0.01 |
解吸后 After desorption | 0.074 | 0.024 | 0.169 | 0.16 | 8.63 | 0.02 |
表4 吸附前后以及解吸后盐湖卤水组成
Table 4 Composition of salt lake brine before and after adsorption and after desorption
卤水组成 Composition | Na质量浓度 ρ(Na)/(g·L-1) | Mg质量浓度 ρ(Mg)/(g·L-1) | K质量浓度 ρ(K)/(g·L-1) | Li质量浓度 ρ(Li)/(mg·L-1) | Rb质量浓度 ρ(Rb)/(mg·L-1) | Cs质量浓度 ρ(Cs)/(mg·L-1) |
|---|---|---|---|---|---|---|
吸附前 Before adsorption | 1.12 | 0.91 | 2.88 | 2.81 | 10.9 | 0.2 |
吸附后 After adsorption | 1.11 | 0.88 | 2.93 | 2.72 | 4.85 | 0.01 |
解吸后 After desorption | 0.074 | 0.024 | 0.169 | 0.16 | 8.63 | 0.02 |
质量比 Mass ratio | m(Na)/m(Rb) | m(Mg)/m(Rb) | m(K)/m(Rb) | m(Li)/m(Rb) | m(Cs)/m(Rb) |
|---|---|---|---|---|---|
吸附前 Before adsorption | 103 | 83 | 264 | 0.3 | 0.02 |
解吸后 After desorption | 9 | 3 | 20 | 0.02 | 0.002 |
表5 吸附前和解吸后各离子与Rb+的质量比
Table 5 Mass ratio of each ion to Rb+ before adsorption and after desorption
质量比 Mass ratio | m(Na)/m(Rb) | m(Mg)/m(Rb) | m(K)/m(Rb) | m(Li)/m(Rb) | m(Cs)/m(Rb) |
|---|---|---|---|---|---|
吸附前 Before adsorption | 103 | 83 | 264 | 0.3 | 0.02 |
解吸后 After desorption | 9 | 3 | 20 | 0.02 | 0.002 |
| 1 | SARKAR A, JACOBS G, JI Y Y, et al. Fischer-tropsch synthesis: characterization Rb promoted iron catalyst[J]. Catal Lett, 2008, 121(1): 1-11. |
| 2 | REN W, SUN Y G, WANG B, et al. Highly reliable optical system for a rubidium space cold atom clock[J]. Appl Opt, 2016, 55(13): 3607-3614. |
| 3 | RAHIMI N, HASSANIPOUR M, YARMOHAMMADI F, et al. Nitric oxide and glutamate are contributors of anti-seizure activity of rubidium chloride: a comparison with lithium[J]. Neurosci Lett, 2019, 708: 134349. |
| 4 | JOSEPH K, PARKS G. Development of a deep space nuclear electric propulsion (NEP) system-a NuAER plasma NEP reactor[C]. AIAA Propulsion and Energy 2020 Forum, 2020: 3540. |
| 5 | JANDOVA J, DVORAK P, FORMANEK J, et al. Recovery of rubidium and potassium alums from lithium-bearing minerals[J]. Hydrometallurgy, 2012, 119: 73-76. |
| 6 | 郭双华, 葛荣庭, 刘宇. 含铷多金属矿石浸出液中铷,钾的萃取分离[J]. 湿法冶金, 2019, 38(6): 473-475. |
| GUO S H, GE R T, LIU Y. Extraction and separation of rubidium and potassium in leaching solution of rubidium-containing polymetallic ore[J]. Hydrometallurgy (China), 2019, 38(6): 473-475. | |
| 7 | 曹冬梅, 王颖, 张雨山, 等. 磷钼酸铵提取铷离子的工艺研究[J]. 盐业与化工, 2014, 43(9): 22-24. |
| CAO D M, WANG Y, ZHANG Y S, et al. Research on the extraction technology for Rb+ by ammonium molybdophosphate[J]. J Salt Chem Ind, 2014, 43(9): 22-24. | |
| 8 | YU C, LU J, HOU Z Q, et al. Mixed matrix membranes for rubidium-dependent recognition and separation: a synergistic recombination design based on electrostatic interactions[J]. Sep Purif Technol, 2021, 255: 117727. |
| 9 | LIU X W, WANG Q, WANG Z, et al. Extraction of Rb(I) ions from aqueous solution using novel imprinting materials[J]. Ind Eng Chem Res, 2019, 58(13): 5269-5279. |
| 10 | NAIDU G, LOGANATHAN P, JEONG S, et al. Rubidium extraction using an organic polymer encapsulated potassium copper hexacyanoferrate sorbent[J]. Chem Eng J, 2016, 306: 31-42. |
| 11 | YANG H J, LI H Y, ZHAI J L, et al. Magnetic prussian blue/graphene oxide nanocomposites caged in calcium alginate microbeads for elimination of cesium ions from water and soil[J]. Chem Eng J, 2014, 246: 10-19. |
| 12 | SOMEDA H H, ELZAHHAR A A, SHEHATA M K, et al. Supporting of some ferrocyanides on polyacrylonitrile (PAN) binding polymer and their application for cesium treatment[J]. Sep Purif Technol, 2002, 29(1): 53-61. |
| 13 | VINCENT C, HERTZ A, VINCENT T, et al. Immobilization of inorganic ion-exchanger into biopolymer foams-application to cesium sorption[J]. Chem Eng J, 2014, 236: 202-211. |
| 14 | WEI P D, WANG L, XIE F, et al. Strong and tough cellulose-graphene oxide composite hydrogels by multi-modulus components strategy as photothermal antibacterial platform[J]. Chem Eng J, 2022, 431: 133964. |
| 15 | THAKUR A, KAUR H. Synthetic chemistry of cellulose hydrogels-a review[J]. Mater Today: Proc, 2022, 48(5): 1431-1438. |
| 16 | WANG Y, GONG Y S, LIN N P, et al. Cellulose hydrogel coated nanometer zero-valent iron intercalated montmorillonite (CH-MMT-nFe0) for enhanced reductive removal of Cr(VI): characterization, performance, and mechanisms[J]. J Mol Liq, 2021: 118355. |
| 17 | 张飞雄, 党奉娜, 吴金风, 等. 玉米秸秆/羧甲基纤维素复配水凝胶水分释放行为[J]. 应用化学, 2016, 33(2): 181-189. |
| ZHANG F X, DANG F N, WU J F, et al. Water release behavior of raw corn straw/carboxymethyl cellulose composite hydrogel[J]. Chinese J Appl Chem, 2016, 33(2): 181-189. | |
| 18 | KIM Y, KIM Y K, KIM S, et al. Nanostructured potassium copper hexacyanoferrate-cellulose hydrogel for selective and rapid cesium adsorption[J]. Chem Eng J, 2017, 313: 1042-1050. |
| 19 | LI G C, ZHAO Y X, ZHANG L Z, et al. Preparation of graphene oxide/polyacrylamide composite hydrogel and its effect on Schwann cells attachment and proliferation[J]. Colloids Surf B, 2016, 143: 547-556. |
| 20 | INAGAKI M, KIM Y A, ENDO M. Graphene: preparation and structural perfection[J]. J Mater Chem, 2011, 21(10): 3280-3294. |
| 21 | SIVASELVAM S, SELVAKUMAR R, VISWANATHAN C, et al. Rapid one-pot synthesis of PAM-GO-Ag nanocomposite hydrogel by gamma-ray irradiation for remediation of environment pollutants and pathogen inactivation[J]. Chemosphere, 2021, 275: 130061. |
| 22 | GODIYA C B, CHENG X, LI D W, et al. Carboxymethyl cellulose/polyacrylamide composite hydrogel for cascaded treatment/reuse of heavy metal ions in wastewater[J]. J Hazard Mater, 2019, 364: 28-38. |
| 23 | PASTA M, WESSELLS C D, LIU N, et al. Full open-framework batteries for stationary energy storage[J]. Nat Commun, 2014, 5(1): 1-9. |
| 24 | YANG J Y, LUO X G, YAN T S, et al. Recovery of cesium from saline lake brine with potassium cobalt hexacyanoferrate-modified chrome-tanned leather scrap adsorbent[J]. Colloids Surf A, 2018, 537: 268-280. |
| 25 | NAIDU G, NUR T, LOGANATHAN P, et al. Selective sorption of rubidium by potassium cobalt hexacyanoferrate[J]. Sep Purif Technol, 2016, 163: 238-246. |
| 26 | KRYS P, TESTA F, TROCHIMCZUK A, et al. Encapsulation of ammonium molybdophosphate and zirconium phosphate in alginate matrix for the sorption of rubidium(I)[J]. J Colloid Interface Sci, 2013, 409: 141-150. |
| [1] | 元宁, 马洁, 张晋玲, 张建胜. 蒸气辅助合成PCN-6(M)双金属有机框架材料及其CH4和CO2吸附性能[J]. 应用化学, 2023, 40(6): 896-903. |
| [2] | 郝晨丽, 丁庆伟, 贾世昌, 毛泱博, 王松柏, 马骏. 硫辛酸修饰钛酸纳米管吸附亚甲基蓝的性能[J]. 应用化学, 2023, 40(5): 749-757. |
| [3] | 赵金丽, 于宗仁, 苏伯民. 墓葬壁画中蛋清胶结材料的热裂解-气质联用分析[J]. 应用化学, 2023, 40(4): 562-570. |
| [4] | 熊波, 黎泰华, 周武平, 刘长宇, 徐晓龙. 一步热聚合法制备Cu2O/CuO-g-C3N4吸附剂及其对甲基橙吸附的性能[J]. 应用化学, 2023, 40(3): 420-429. |
| [5] | 张琴, 刘文彬, 樊利娇, 谢宇铭, 黄国林. 功能化介孔二氧化硅的制备及其吸附分离水中铀的研究进展[J]. 应用化学, 2023, 40(2): 169-187. |
| [6] | 陈玉竹, 刘思思, 张蒙蒙, 林祥德, 曾冬冬. 基于抗菌性壳聚糖/羧甲基纤维素复合药物涂层的聚氨酯敷料[J]. 应用化学, 2023, 40(2): 252-260. |
| [7] | 黄旭娟, 王婷, 丁正青, 杨欣欣, 蔡照胜, 商士斌. 脱氢枞氧基聚氧乙烯缩水甘油醚接枝羟乙基壳聚糖水凝胶制备及其性能[J]. 应用化学, 2022, 39(9): 1421-1428. |
| [8] | 赵跃华, 王大鹏. 氨基化氧化石墨烯和脂肪酸在水-油界面的共吸附动力学[J]. 应用化学, 2022, 39(8): 1274-1284. |
| [9] | 曹从军, 马含笑, 侯成敏, 丁小健, 管飙. 乙基纤维素磁性复合材料对溶液中铜离子的吸附性能[J]. 应用化学, 2022, 39(6): 969-979. |
| [10] | 薛松松, 解正峰, 何佳伟, 张天怡, 夏保平, 李雨芹. 高选择性快速识别汞(Ⅱ)离子的磺酰腙型探针的合成及在吸附中的应用[J]. 应用化学, 2022, 39(5): 760-768. |
| [11] | 王雪, 王意波, 王显, 祝建兵, 葛君杰, 刘长鹏, 邢巍. 酸性电解水过程中氧析出反应的机理及铱基催化剂的研究进展[J]. 应用化学, 2022, 39(4): 616-628. |
| [12] | 张健爽, 高美珍, 王梦瑶, 石琪, 董晋湘. 沸石咪唑酯骨架结构材料ZIF-71用于低浓度生物基2,3-丁二醇/1,3-丙二醇的吸附分离性能[J]. 应用化学, 2022, 39(11): 1735-1745. |
| [13] | 赵常利, 秦明高, 窦晓秋, 冯传良. 纳米颗粒增强的手性超分子水凝胶成骨性能[J]. 应用化学, 2022, 39(1): 177-187. |
| [14] | 刘旭, 李杨可欣, 杜黎, 于健, 王佳程, 耿阳, 韩广, 孙宽, 李猛. 水凝胶的制备及仿生设计在能源领域应用的研究进展[J]. 应用化学, 2022, 39(1): 35-54. |
| [15] | 李胜男, 付俊. 水凝胶仿生柔性电子学[J]. 应用化学, 2022, 39(1): 55-73. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||