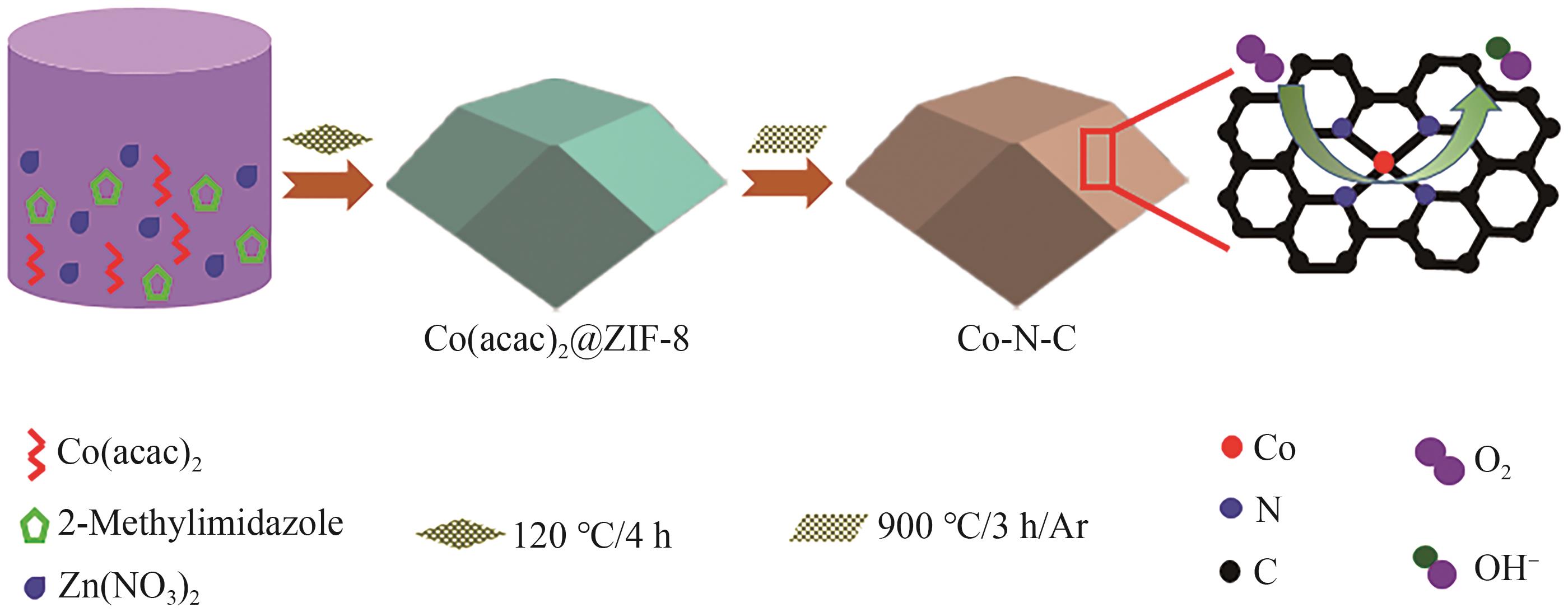
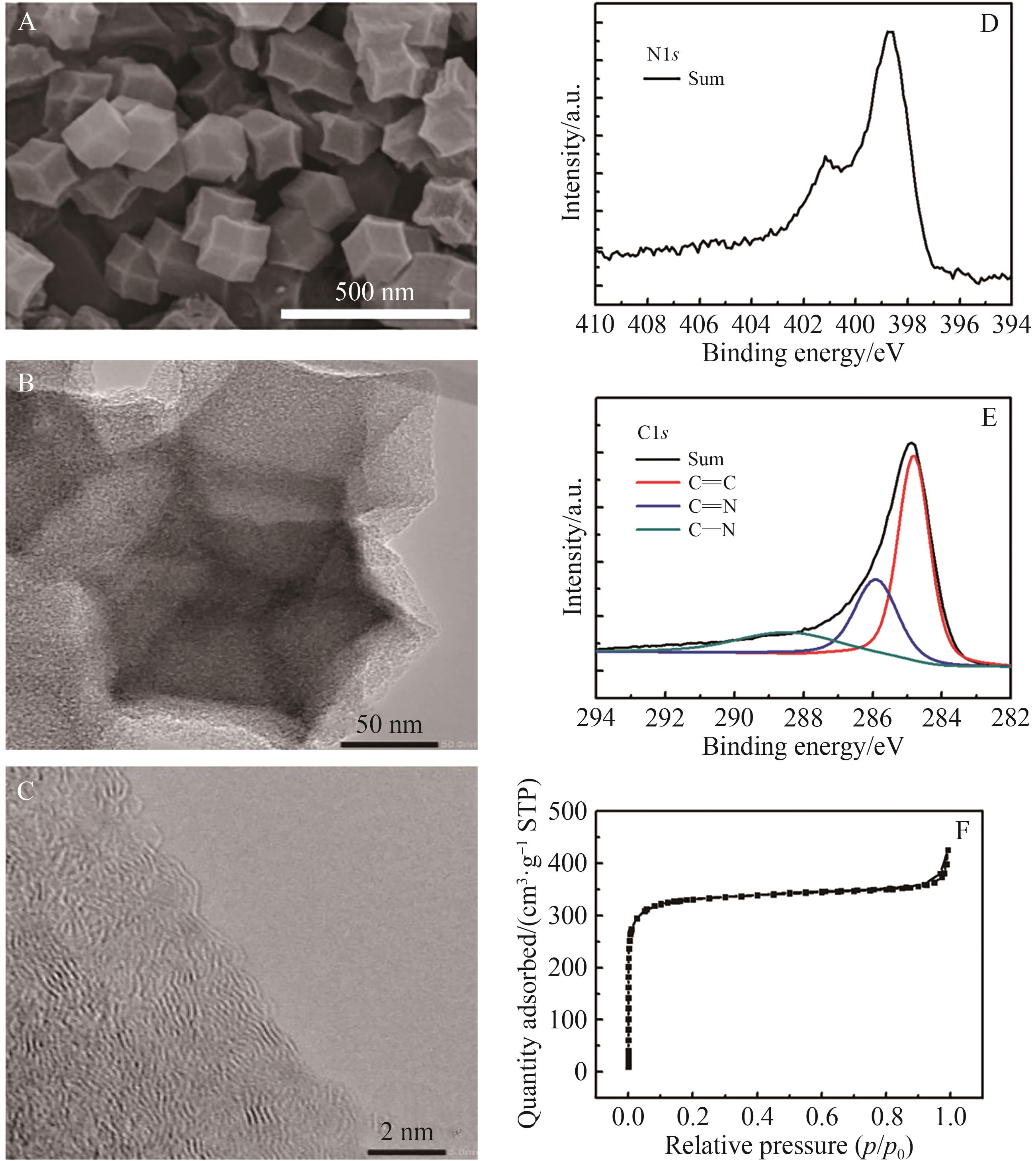
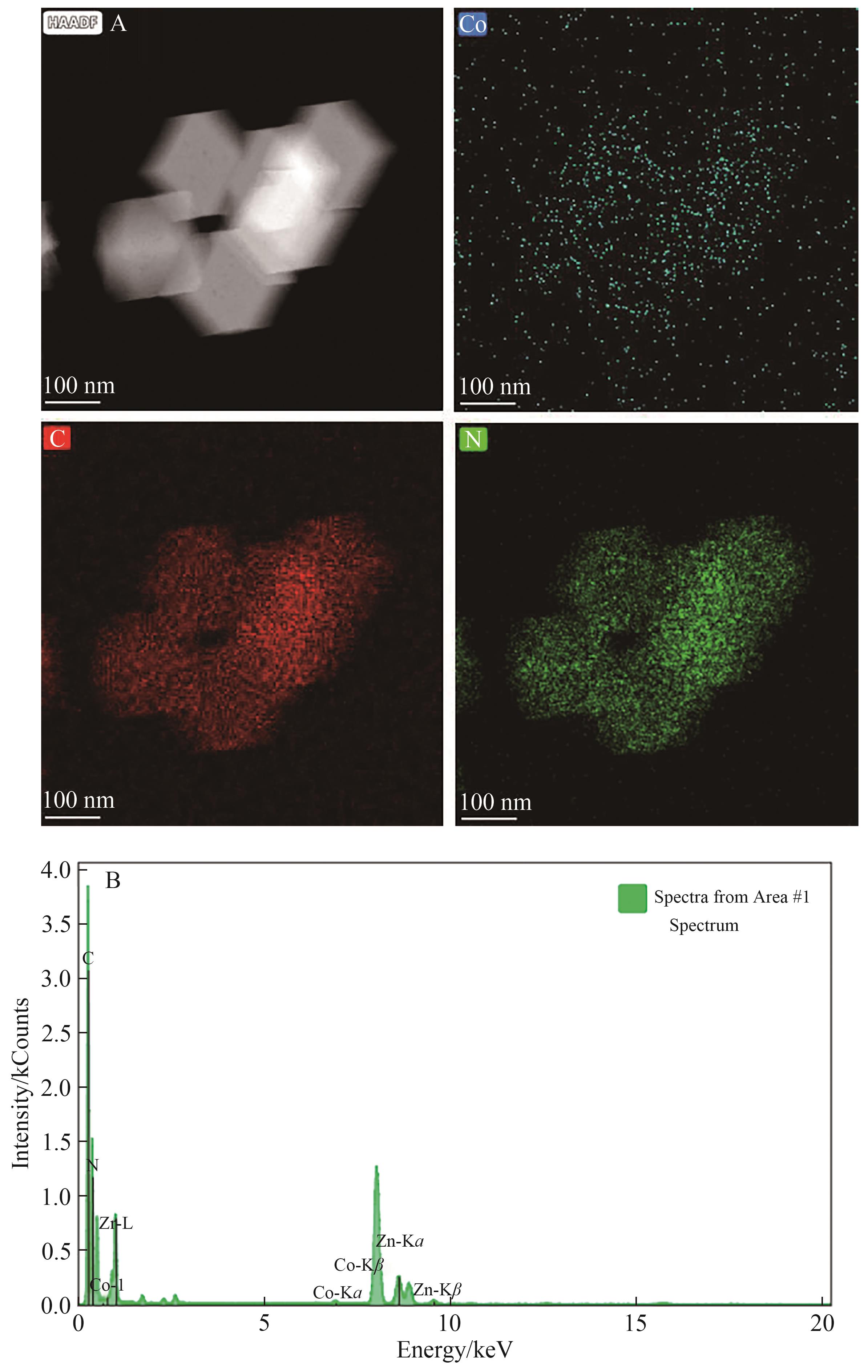
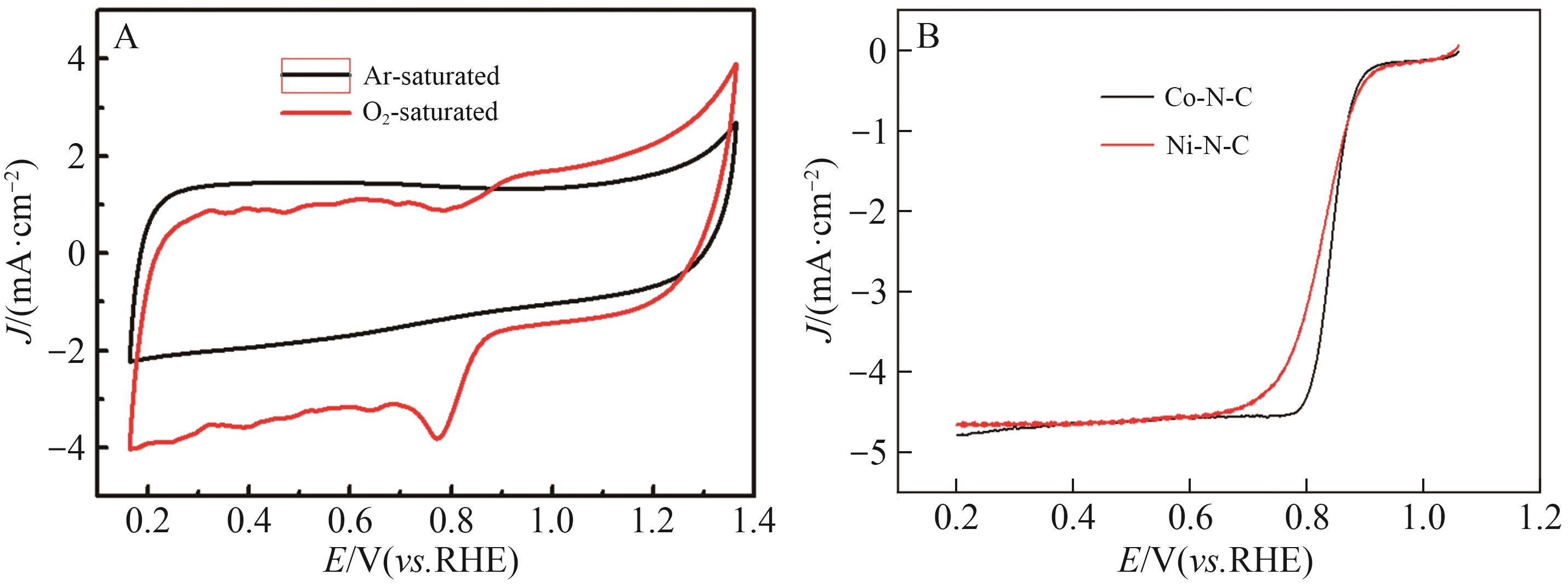
| [1] |
LIU D, FU L, HUANG X, et al. Influence of iron source type on the electrocatalytic activity toward oxygen reduction reaction in Fe-N/C for Al-air batteries[J]. J Electrochem Soc, 2018, 165: F662-F670.
|
| [2] |
PRIYA V K, KHARABE G P, BARIK S, et al. Co-incorporated N-doped micro-meso porous carbon as an electrocatalyst for oxygen reduction reaction and Zn-air battery[J]. Energy Fuels, 2024, 38: 7196-7207.
|
| [3] |
ALLWYN N, GOKUlLNATH S, SATHISH M. In-situ nanoarchitectonics of Fe/Co LDH over cobalt-enriched N-doped carbon cookies as facile oxygen redox electrocatalysts for high-rate rechargeable zinc-air batteries[J]. ACS Appl Mater Interfaces, 2024, 16: 20360-20374.
|
| [4] |
NIE Y, LI L, WEI Z. Recent advancements in Pt and Pt-free catalysts for oxygen reduction reaction[J]. Chem Soc Rev, 2015, 44: 2168-2201.
|
| [5] |
LIU R, JIANG C, GUO J, et al. Activating Ru nanoclusters for robust oxygen reduction in aqueous wide-temperature zinc-air batteries[J] Matter, 2024, 7: 4031-4045.
|
| [6] |
VLIET D F V D, WANG C, TRIPKOVIC D, et al. Mesostructured thin films as electrocatalysts with tunable composition and surface morphology[J]. Nat Mater, 2012, 11: 1051-1058.
|
| [7] |
CHEN Y, JI S, WANG Y, et al. Isolated single iron atoms anchored on N-doped porous carbon as an efficient electrocatalyst for the oxygen reduction reaction[J]. Angew Chem Int Ed, 2017, 56: 6937-6941.
|
| [8] |
XIE L, ZHANG X, ZHAO B, et al. Enzyme-inspired iron porphyrins for improved electrocatalytic oxygen reduction and evolution reactions[J]. Angew Chem Int Ed, 2021, 60: 7576-7581.
|
| [9] |
郑锐雪, 孟庆磊, 张丽, 等. 分级孔结构的Fe-N-C催化剂用于高效电催化氧还原[J]. 应用化学, 2023, 40(8): 1187-1194.
|
|
ZHENG R X, MENG Q L, ZHANG L, et al. Hierarchically porous Fe-N-C catalysts for efficient electrocatalytic oxygen reduction reaction[J]. Chin J Appl Chem, 2023, 40(8): 1187-1194.
|
| [10] |
LI N, SONG X, WANG L, et al. Single-atom cobalt catalysts for electrocatalytic hydrodechlorination and oxygen reduction reaction for the degradation of chlorinated organic compounds[J]. ACS Appl Mater Interfaces, 2020, 12: 24019-24029.
|
| [11] |
ZHAO Y, GAO L, CHEN W, et al. The S-doped cobalt porphyrin-based molecular catalysts with large conjugated meso-substituents for enhanced electrocatalytic oxygen reduction and evolution reaction[J]. J Electrochem Soc, 2021, 168: 116502.
|
| [12] |
DINDA S, ROY S, PATRA S C, et al. Polyaromatic hydrocarbon derivatized azo-oximes of cobalt(Ⅲ) for the ligand-redox controlled electrocatalytic oxygen reduction reaction[J]. New J Chem, 2020, 44: 3737-3747.
|
| [13] |
ABE Y, SATOH I, SAITO T, et al. Oxygen reduction reaction catalytic activities of pure Ni-based perovskite-related structure oxides[J]. Chem Mater, 2020, 32: 8694-8699.
|
| [14] |
KARIUKI N N, CANSIZOGLU M F, BEGUM M, et al. SAD-GLAD Pt-Ni@Ni nanorods as highly active oxygen reduction reaction electrocatalysts[J]. ACS Catal, 2016, 6: 3478-3485.
|
| [15] |
TODOROKI N, KATO T, HAYASHI T, et al. Pt-Ni nanoparticle-stacking thin film: highly active electrocatalysts for oxygen reduction reaction[J]. ACS Catal, 2015, 5: 2209-2212.
|
| [16] |
YANG J, LI J, DING R, et al. Kinetic effects of temperature on Fe-N-C catalysts for 2e- and 4e- oxygen reduction reactions[J]. J Electrochem Soc, 2021, 168: 096502.
|
| [17] |
BAI J, ZHANG K, GAO J, et al. Co-NC catalyst with rich coordinated nitrogen and hierarchical porous structure for oxygen reduction reaction[J]. J Alloy Compd, 2024, 992: 174599.
|
| [18] |
LI P, QI Z, YAN D, Rare earth Er-Nd dual single-atomic catalysts for efficient visible-light induced CO2 reduction to CnH2 n+1OH (n=1, 2)[J]. Angew Chem Int Ed, 2024, 63: e202411000.
|
| [19] |
JASINSKI R. A new fuel cell cathode catalyst[J]. Nature, 1964, 201: 1212-1213.
|
| [20] |
LI M, BO X, ZHANG Y, et al. Comparative study on the oxygen reduction reaction electrocatalytic activities of iron phthalocyanines supported on reduced graphene oxide, mesoporous carbon vesicle, and ordered mesoporous carbon[J]. J Power Sources, 2014, 264: 114-122.
|
| [21] |
REN C, LI H, LI R, et al. Electrocatalytic study of a 1,10-phenanthroline-cobalt(Ⅱ) metal complex catalyst supported on reduced graphene oxide towards oxygen reduction reaction[J]. RSC Adv, 2016, 6: 33302-33307.
|
| [22] |
WEI Y, LI Y, FENG L, et al. Effects of electrolyte pH on oxygen reduction properties using a cobalt porphyrin-coated carbon composite[J]. Electrochim Acta, 2024, 489: 144293.
|
| [23] |
CHEN S, LUO T, LI X, et al.Identification of the highly active Co-N4 coordination motif for selective oxygen reduction to hydrogen peroxide[J]. J Am Chem Soc, 2022, 144: 14505-14516.
|
| [24] |
LV M, CUI C, HUANG N, et al. Precisely engineering asymmetric atomic CoN4 by electron donating and extracting for oxygen reduction reaction[J]. Angew Chem Int Ed, 2024, 63: e202315802.
|
| [25] |
JIN X, FU J, ZHAO C, et al. FeN4- and CoN4-centered transition metallomacrocyles as highly efficient non-noble metal electrocatalysts for oxygen reduction reaction[J]. J Electroanal Chem, 2024, 967: 118398.
|
| [26] |
YANG H, LI Z, KOU S, et al. A complex-sequestered strategy to fabricate Fe single-atom catalyst for efficient oxygen reduction in a broad pH-range[J]. Appl Catal B-Environ, 2020, 278: 119270.
|
| [27] |
DENG L, QIU L, HU R, et al. Restricted diffusion preparation of fully-exposed Fe single-atom catalyst on carbon nanospheres for efficient oxygen reduction reaction[J]. Appl Catal B-Environ, 2022, 305: 121058.
|
| [28] |
YE W, YANG Y, ARIF M, et al. Fe, Mo-N/C hollow porous nitrogen-doped carbon nanorods as an effective electrocatalyst for N2 reduction reaction[J]. ACS Sustainable Chem Eng, 2020, 8: 15946-15952.
|