
应用化学 ›› 2024, Vol. 41 ›› Issue (1): 39-59.DOI: 10.19894/j.issn.1000-0518.230264
近红外二区激活型小分子荧光探针研究进展
- 湖南大学化学化工学院,化学生物传感与计量学国家重点实验室,长沙 410082
-
收稿日期:2023-09-01接受日期:2023-11-17出版日期:2024-01-01发布日期:2024-01-30 -
通讯作者:袁林 -
基金资助:国家自然科学基金(22074036)
Research Progress in Activatable NIR-Ⅱ Small Molecule Fluorescent Probes
Xin-Yu ZHAO, Zuo-Jia QIN, Xiao-Bing ZHANG, Lin YUAN( )
)
- State Key Laboratory of Chemo/Biosensing and Chemometrics,College of Chemistry and Chemical Engineering,Hunan University,Changsha 410082,China
-
Received:2023-09-01Accepted:2023-11-17Published:2024-01-01Online:2024-01-30 -
Contact:Lin YUAN -
About author:lyuan@hnu.edu.cn
-
Supported by:the National Natural Science Foundation of China(22074036)
摘要:
近红外二区(NIR-Ⅱ)荧光成像具有优良的组织穿透深度和空间分辨率,广泛应用于生物医学领域。与传统的常亮型荧光探针相比,激活型探针只有当特定的生物标志物存在情况下才会发生荧光信号变化,因此具有更高的信背比,会进一步提高区分病变与正常组织的准确度和可靠性。基于此,本文按照生物疾病模型进行分类,系统总结了近5年报道的近红外二区激活型小分子探针的结构、响应机理以及活体应用,并对目前激活型小分子探针面临的问题及发展方向进行简要介绍。
中图分类号:
引用本文
赵欣雨, 秦作佳, 张晓兵, 袁林. 近红外二区激活型小分子荧光探针研究进展[J]. 应用化学, 2024, 41(1): 39-59.
Xin-Yu ZHAO, Zuo-Jia QIN, Xiao-Bing ZHANG, Lin YUAN. Research Progress in Activatable NIR-Ⅱ Small Molecule Fluorescent Probes[J]. Chinese Journal of Applied Chemistry, 2024, 41(1): 39-59.
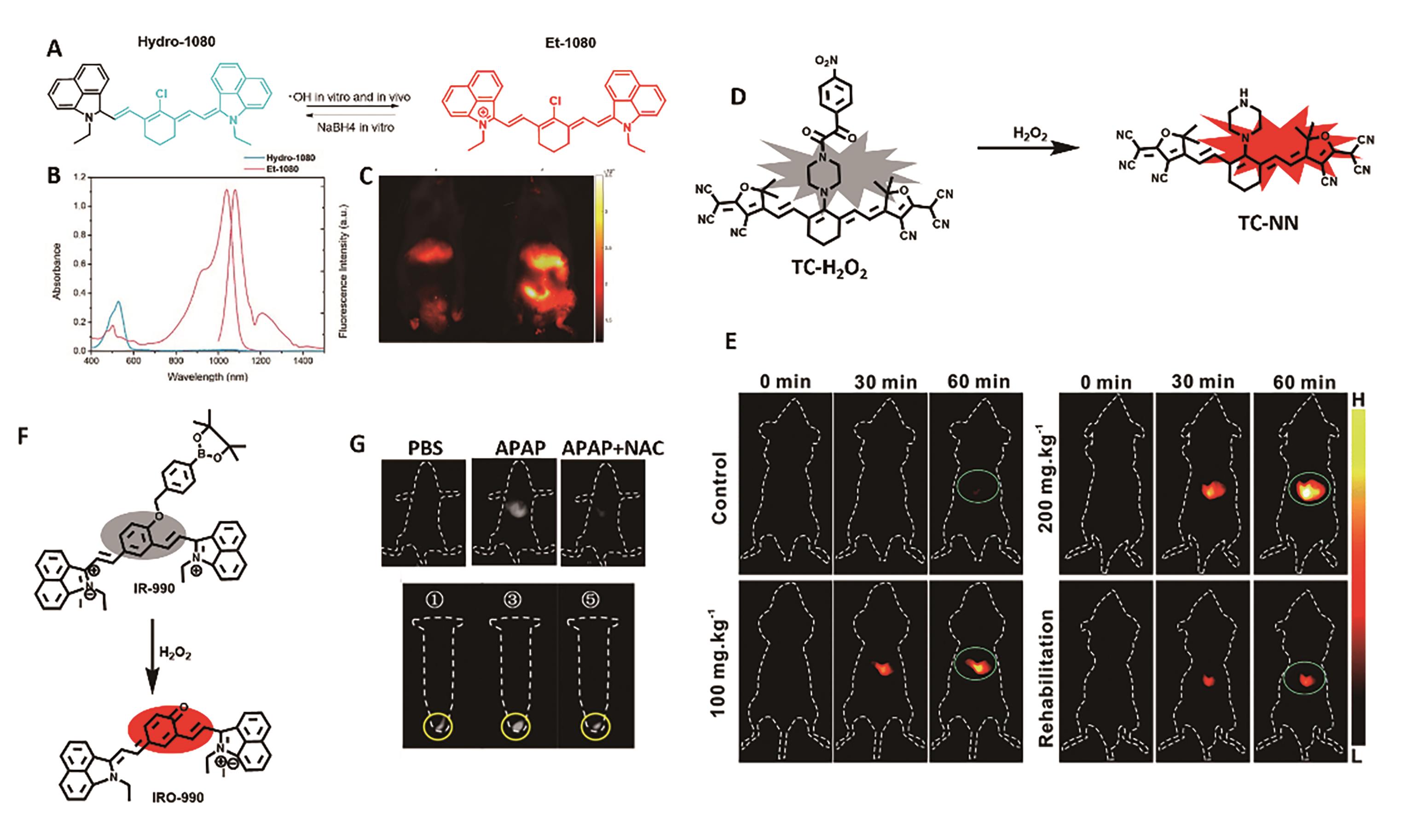
图1 用于肝脏炎症成像的代表性探针。 (A)探针Hydro-1080的结构及响应机理[23]。 (B)Hydro-1080和Et-1080的吸收和发射光谱图[23]。 (C)患有肝炎小鼠NIR-Ⅱ荧光成像[23]。 (D)探针TC-H2O2的结构及响应机理[24]。 (E)小鼠肝损伤模型的MOST和荧光成像[24]。 (F)探针IR-990的结构及响应机理[26]。 (G)HepG2细胞及APAP诱导的肝损伤模型中H2O2的NIR-Ⅱ荧光成像[26]
Fig.1 Representative probe for liver inflammation imaging. (A) Structure and response mechanism of Hydro-1080[23]. (B) Absorption and fluorescence spectra of Hydro-1080 and Et-1080 in DMSO[23]. (C) NIR-Ⅱ fluorescence images of mice liver injury[23]. (D) Structure and response mechanism of probe TC-H2O2[24]. (E) MOST and fluorescence imaging of mice liver injury model[24]. (F) Structure and response mechanism of IR-990[26]. (G) NIR-Ⅱ fluorescence imaging of H2O2 in HepG2 cells and APAP-induced mice liver injury models[26]
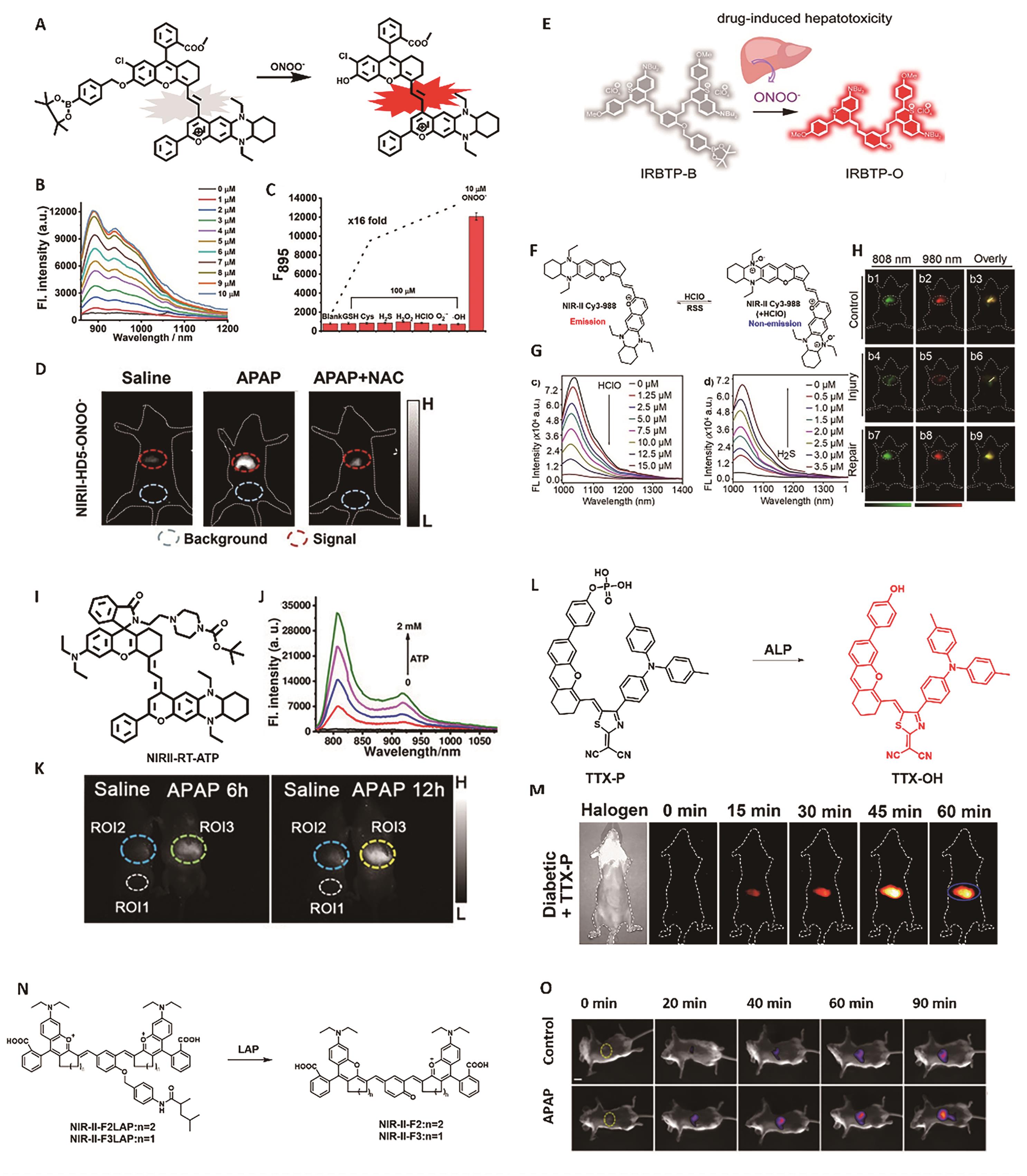
图2 用于肝脏炎症成像的代表性探针。 (A)探针NIRII-HD5-ONOO-的结构及响应机理[30]。 (B) NIRII-HD5-ONOO-探针(5 μmol/L)与不同浓度ONOO-共孵育后的荧光光谱[30]。 (C) NIRII-HD5-ONOO-对不同分析物的荧光响应[30]。 (D)经APAP处理后小鼠肝脏脏的NIR-Ⅱ荧光成像[30]。 (E)探针IRBTP-B的结构及响应机理[31]。 (F) NIR-Ⅱ Cy3-988探针的响应机理[33]。 (G)随HClO(0~15 μmol/L)和H2S(0~3.5 μmol/L)浓度的变化NIR-Ⅱ Cy3-988探针(5 μmol/L)荧光强度的变化[33]。 (H)小鼠肝脏损伤/修复模型的NIR-Ⅱ荧光成像[33]。 (I)探针NIRII-RT-ATP的结构[34]。 (J)NIRII-RT-ATP探针(5 μmol/L)与不同浓度ATP共孵育后的荧光光谱[34]。 (K)经APAP处理后小鼠肝脏的NIR-Ⅱ荧光成像[34]。 (L)探针TTX-P的结构及响应机理 [35]。 (M)小鼠肝脏的NIR-Ⅱ荧光成像[35]。 (N)探针NIR-Ⅱ-LAP的结构及响应机理[36]。 (O)经APAP处理后小鼠肝脏的NIR-Ⅱ荧光成像[36]
Fig.2 Representative probes for liver inflammation imaging. (A) Structure and response mechanism of NIRII-HD5-ONOO-[30]. (B) NIR-Ⅱ fluorescence spectra of NIRII-HD5-ONOO- (5 μmol/L) after reaction with various concentrations of ONOO-. (C) NIR-Ⅱ fluorescence response of NIRII-HD5-ONOO- (5 μmol/L) toward different analytes[30]. (D) NIR-Ⅱ fluorescence imaging of liver injury after APAP treatment[30]. (E) Structure and response mechanism of IRBTP-B[31]. (F) Response mechanism of probe NIR-Ⅱ Cy3-988[33]. (G) Fluorescence intensity changes of NIR-Ⅱ Cy3-988 (5 μmol/L) after reaction with various concentrations of HClO (0~15 μmol/L) and H2S (0~3.5 μmol/L)[33]. (H) NIR-Ⅱ fluorescence imaging of mice liver injury and repair[33]. (I) Structure of probe NIRII-RT-ATP[34]. (J) NIR-Ⅱ fluorescence spectra of NIRII-RT-ATP (5 μmol/L) after reaction with various concentrations of ATP[34]. (K) Fluorescence imaging of liver injury after APAP treatment[34]. (L) Structure and response mechanism of TTX-P[35]. (M) NIR-Ⅱ fluorescence imaging of liver injury[35]. (N) Structure and response mechanism of NIR-Ⅱ-LAP[36]. (O) Fluorescence imaging of liver injury after APAP treatment[36]

图3 用于肾脏炎症成像的代表性探针。 (A) BOD-Ⅱ-NAG探针的结构及响应机理[39]。 (B)在静脉注射BOD-Ⅱ-NAG-NP(16 μmol/kg)后活鼠的NIR-Ⅱ荧光成像图[39]。 (C)经不同处理的小鼠在不同时间点静脉注射BOD-Ⅱ-NAG-NP后肾脏的荧光强度变化[39]。 (D) 经不同处理的小鼠体内KIM-1、NGAL、NAG、Cyst C、sCr和BUN含量的变化[39]。 (E) HP-N系列染料及探针HP-H2O2的结构及设计策略[40]。 (F)探针HP-H2O2(10 μmol/L)与不同浓度的H2O2孵育后的NIR-Ⅱ荧光光谱图[40]。 (G)探针HP-H2O2在937 nm处的荧光强度随时间变化关系图[40]。 (H)对照组小鼠和AKI小鼠静脉注射HP-H2O2后NIR-Ⅱ荧光图像[40]
Fig.3 Representative probes for imaging kidney inflammation. (A) Structure and response mechanism of BOD-Ⅱ-NAG[39]. (B) NIR-Ⅱ fluorescence images of living mice after injection of BOD-Ⅱ-NAG-NP (16 μmol/kg) at different time[39]. (C) The fluorescence intensity in different groups after intravenous injection of BOD-Ⅱ-NAG-NP[39]. (D) Changes in KIM-1, NGAL, NAG, Cyst C, sCr and BUN in living mice after different treatment[39]. (E) Structure and design strategy of HP-N dyes and HP-H2O2[40] (F) NIR-Ⅱ fluorescence spectra of HP-H2O2 after reaction with various concentrations of H2O2(10 μmol/L)[40]. (G) Fluorescence intensities of the probe HP-H2O2 at different time[40]. (H) NIR-Ⅱ fluorescence images of control mice and AKI mice[40]

图4 用于膀胱炎症成像的代表性探针。 (A) Chrodol系列染料的结构及光物理性质[42]。 (B)双激活型探针PN910的响应机理[42]。 (C) PN910及Chrodol-3的吸收和发射光谱[42]。 (D)在H2O2(左)和ONOO-(右)存在下探针的荧光随时间的变化[42]。 (E) BTPE-NO2@F127探针的结构和响应机制[43]。 (F)对照组和患有间质性膀胱炎小鼠在注射BTPE-NO2@F127后MOST成像图(左)和NIR-Ⅱ荧光成像图(右)[43]
Fig.4 Representative probe for imaging cystitis. (A) Structure and photophysical properties of Chrodol[42]. (B) Response mechanism of dual-activatable probe PN910[42]. (C) Absorption and fluorescence spectra of PN910 and Chrodol-3[42]. (D) Fluorescence intensity upon addition of H2O2 (left) and ONOO- (right)[42]. (E) Structure and response mechanism of BTPE-NO2@F127 probe[43]. (F) MOST images (left) and NIR-Ⅱ fluorescence images (right) of the control and the interstitial cystitis model[43]
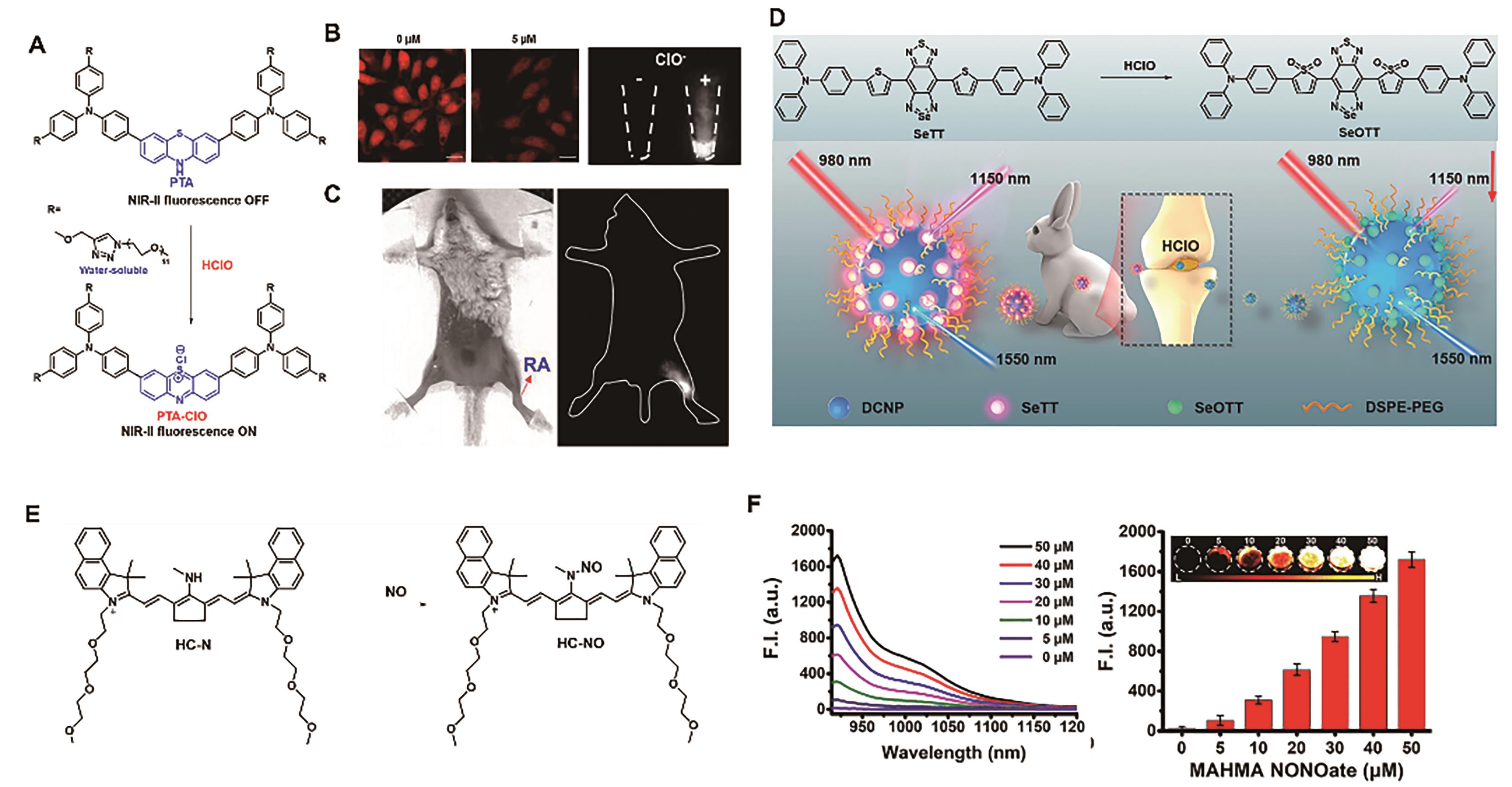
图5 用于关节炎成像的代表性探针。 (A)探针PTA的结构及响应机理[46]。 (B) HeLa细胞外源性和内源性HClO的荧光成像图[46]。(C) RA小鼠模型中HClO的荧光成像图[46]。(D)探针DNCP@SeTT的结构及响应机理[47]。 (E)探针HC-N的结构及响应机理[48]。 (F)探针HC-N(5 μmol/L)与不同浓度NO响应后的NIR-Ⅱ荧光光谱图(左)和位于923 nm处荧光强度(右)[48]
Fig.5 Representative probes for imaging joint inflammation. (A) Structure and response mechanism of probe PTA[46]. (B) Fluorescence images of exogenous and endogenous HClO in HeLa cell[46]. (C) Fluorescence imaging of HClO in RA mouse model[46]. (D) Structure and response mechanism of DNCP@SeTT[47]. (E) Structure and response mechanism of probe HC-N[48]. (F) NIR-Ⅱ fluorescence spectra (left) and fluorescence intensity at 923 nm (right) of HC-N (5 μmol/L) after reaction with various concentrations of NO[48]
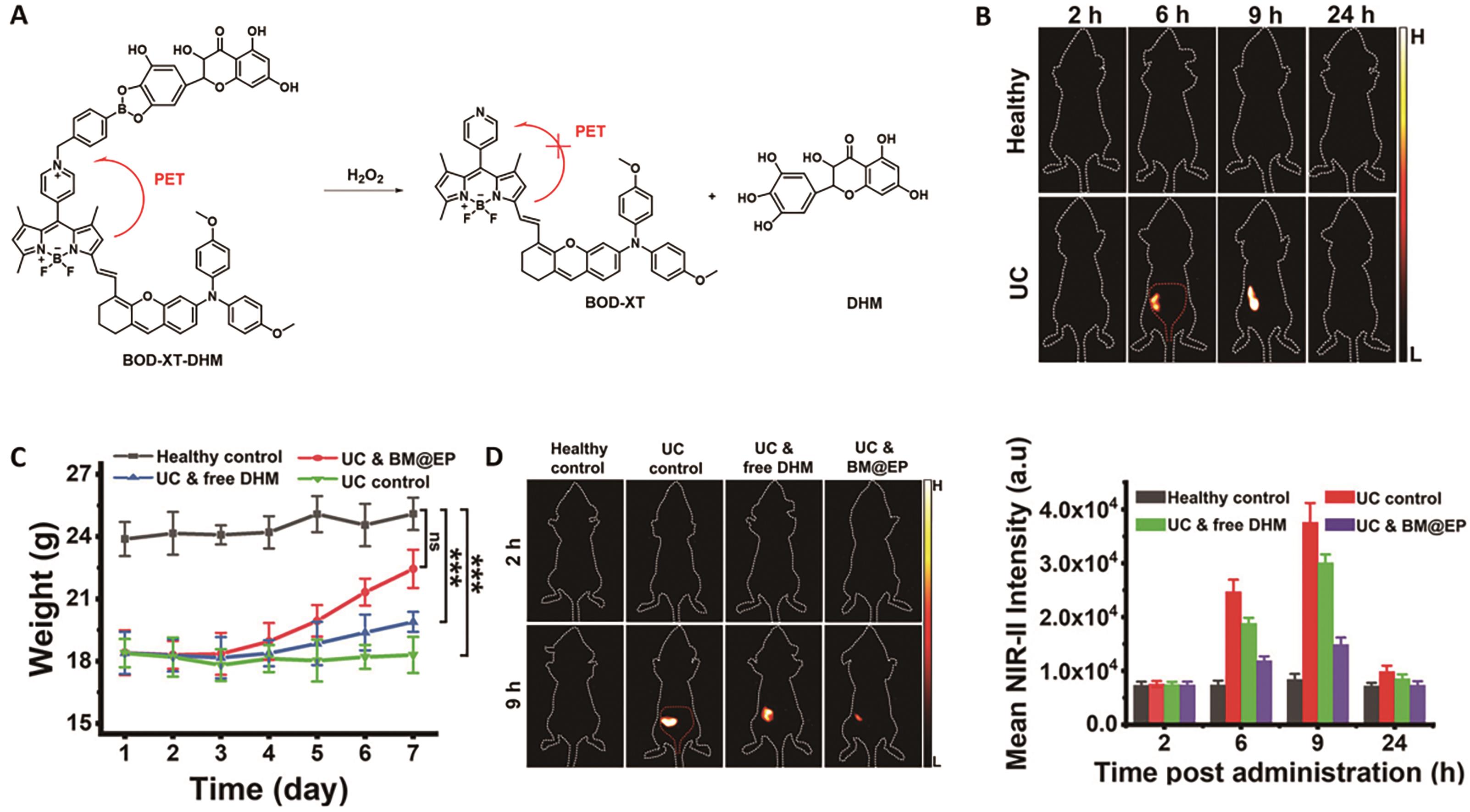
图6 用于溃疡性结肠炎成像的代表性探针[50]。 (A)探针BOD-XT-DHM的结构及响应机理。 (B)小鼠口服探针BM@EP(50 mg/kg)后的荧光成像图。 (C)经不同处理后的小鼠体重变化。 (D)经不同处理的小鼠在口服BM@EP探针(43.6 mg/kg)后的NIR-Ⅱ荧光成像图(左)和结肠区域的荧光强度(右)
Fig.6 Representative probe for imaging ulcerative colitis[50]. (A) Structure and response mechanism of probe BOD-XT-DHM. (B) NIR-Ⅱ fluorescent images of mice after oral administration of BM@EP (50 mg/kg). (C) Changes in body mass of different groups' mice. (D) NIR-Ⅱ fluorescence images of mice (left) and average fluorescent intensities at mice′s colon region (right) after oral dosing of BM@EP (43.6 mg/kg)
| Liver injury | ?OH | Hydro-1080 | |||
| H2O2 | TC-H2O2 | ||||
| IR-990 | 0.59 μ | ||||
| BHC-Lut | |||||
| NIRII-HD5-ONOO- | |||||
| IRBTP-B | 55.9 n | ||||
| HClO | NIR Ⅱ Cy3-988 | ||||
| NIRII-RT-ATP | |||||
| TTX-P | |||||
| NIR-Ⅱ-F2LAP | 0.063 U/L | ||||
| Kidney injury | NAG | BOD-II-NAG-NP | |||
| HP-H2O2 | |||||
| Cystitis | ROS/RNS and base | PN910 | |||
| H2O2 | BTPE-NO2 | ||||
| Joint inflammation | HClO | ||||
| HC-N | |||||
| Colitis | pH/ROS | BM@EP |
表1 用于炎症模型的NIR-Ⅱ激活型探针
Table 1 NIR-Ⅱ activatable probes for inflammation models
| Liver injury | ?OH | Hydro-1080 | |||
| H2O2 | TC-H2O2 | ||||
| IR-990 | 0.59 μ | ||||
| BHC-Lut | |||||
| NIRII-HD5-ONOO- | |||||
| IRBTP-B | 55.9 n | ||||
| HClO | NIR Ⅱ Cy3-988 | ||||
| NIRII-RT-ATP | |||||
| TTX-P | |||||
| NIR-Ⅱ-F2LAP | 0.063 U/L | ||||
| Kidney injury | NAG | BOD-II-NAG-NP | |||
| HP-H2O2 | |||||
| Cystitis | ROS/RNS and base | PN910 | |||
| H2O2 | BTPE-NO2 | ||||
| Joint inflammation | HClO | ||||
| HC-N | |||||
| Colitis | pH/ROS | BM@EP |

图7 用于肝癌成像的代表性探针[52]。 (A)探针BH-NO2@BSA的结构及响应机理。 (B)小鼠肝肿瘤的3D MOST成像(中)和NIR-Ⅱ荧光成像导航的肝肿瘤切除(右)
Fig.7 Representative probe for liver cancer imaging[52]. (A) Structure and response mechanism of probe BH-NO2@BSA. (B) 3D MOST imaging of mouse liver tumors (middle) and NIR-Ⅱ fluorescence image-guided resection of liver tumors (right)
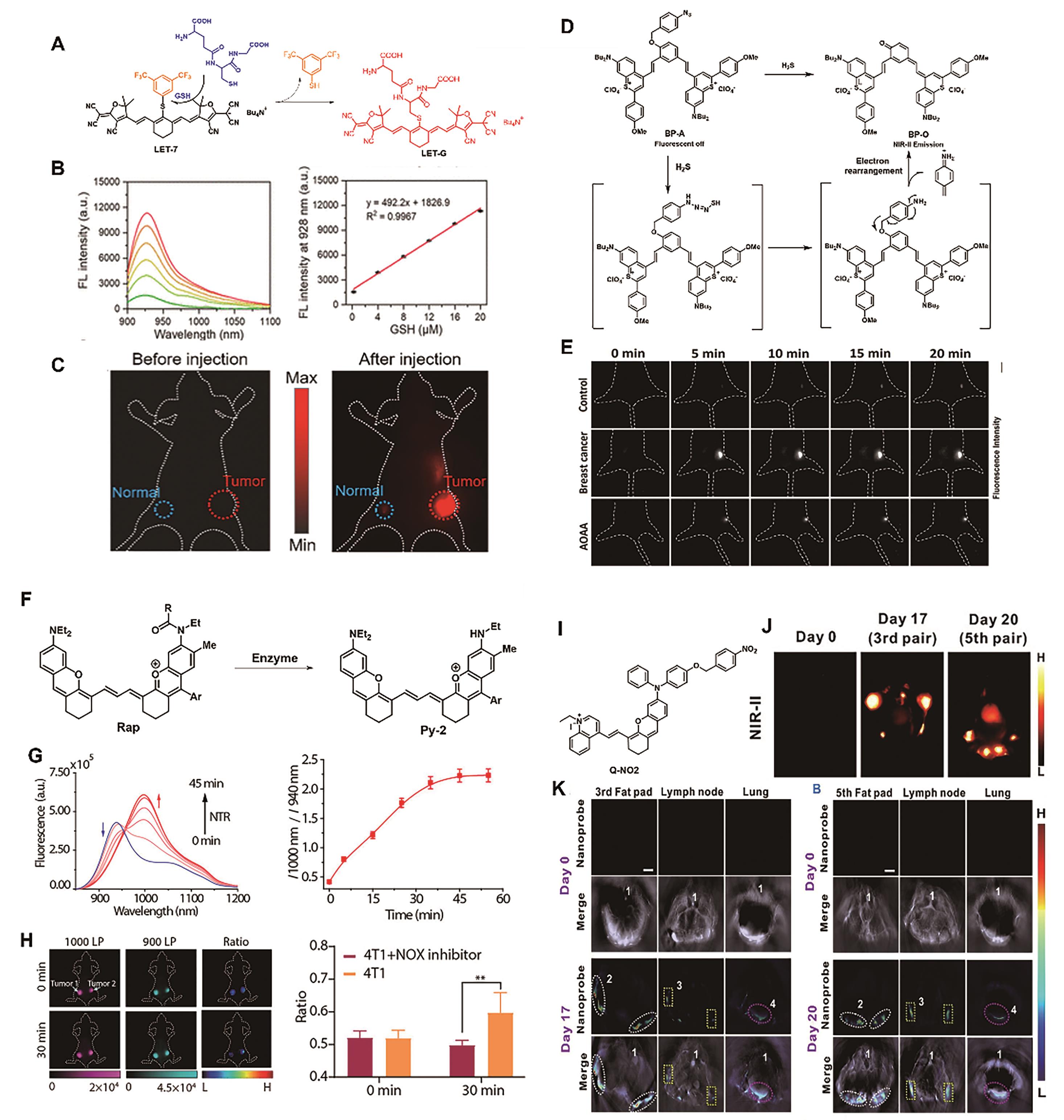
图8 用于乳腺癌成像的代表性探针。 (A)探针LET-7的结构及响应机理[53]。 (B)探针LET-7(2 μmol/L)与不同浓度GSH响应后的NIR-Ⅱ荧光光谱图(左)及相应的线性关系(右)[53]。 (C)小鼠的NIR-Ⅱ荧光成像图[53]。 (D)探针BP-A的结构及响应机理[55]。 (E)经不同处理的小鼠的在注射探针LET-7(200 μmol/L)后的NIR-Ⅱ荧光成像图[55]。 (F)比例型探针Rap-N的设计策略[57]。 (G)探针与NTR(10 μg/mL)共孵育后荧光强度(左)和比例荧光信号I1010 nm/I940 nm(右)随时间的变化情况[57]。 (H)乳腺癌小鼠模型(左侧肿瘤被注射NTR抑制剂双香豆素)的比例荧光成像(左)和信号强度(右)[57]。 (I)分子探针Q-NO2的结构[59]。 (J)小鼠乳腺肿瘤局部转移和远端转移的NIR-Ⅱ成像和(K)MOST成像[59]
Fig.8 Representative probes for breast cancer imaging. (A) Structure and response mechanism of probe LET-7[53]. (B) NIR-Ⅱ fluorescence spectra of probe LET-7 (2 μmol/L) after reaction with various concentrations of GSH (left) and corresponding linear relationships (right) [53]. (C) NIR-Ⅱ fluorescence images of mice[53]. (D) Structure and response mechanism of probe BP-A[55]. (E) Different groups′ mice′s NIR-Ⅱ fluorescence images after intratumoral injection of BP-A (200 μmol/L)[55]. (F) Design strategy of the ratiometric probe Rap-N[57]. (G) Time dependence of fluorescence spectra (left) and I1000 nm/I940 nm ratio of Rap-N (right) incubation with NTR (10 μg/mL)[57]. (H) Ratiometric fluorescence images (left) and signal intensity (right) of mice bearing breast tumors (the left tumor injected with the NTR inhibitor dicoumarol)[57]. (I) Structure of the molecular probe Q-NO2 [59]. (J)NIR-Ⅱ images and (K) MOST images of regional and distant breast cancer metastases in mice[59]

图9 用于肺癌成像的代表性探针。 (A)酶激活型探针的结构及响应机理[61]。 (B)探针与NTR(20 mg/mL)共孵育后荧光强度随时间变化情况[61]。 (C)经不同处理的小鼠的在注射探针NTR-InD(30 nmol)后的NIR-Ⅱ荧光成像[61]。 (D)探针RHC-NO2的结构及响应机理[62]。 (E)探针RHC-NO2(10 μmol/L)与不同浓度NTR响应后的NIR-Ⅱ荧光光谱图[62]。 (F)尾静脉注射RHC-NO2(500 μmol/L)后小鼠的NIR-Ⅱ荧光成像图[62]
Fig.9 Representative probes for lung cancer imaging. (A) Structure and response mechanism of enzyme-activated probes[61]. (B) Time dependence of fluorescence spectra of Rap-N (20 mg/mL) incubation with NTR[61]. (C) Different groups' mice's NIR-Ⅱ fluorescence images after injection of the probe NTR-InD (30 nmol)[61]. (D) Structure and response mechanism of probe RHC-NO2[62]. (E) NIR-Ⅱ fluorescence spectra of probe RHC-NO2 (10 μmol/L) after reaction with various concentrations of NTR[62]. (F) NIR-Ⅱ images of lung tumor-bearing mice after tail vein injection of RHC-NO2 (500 μmol/L)[62]
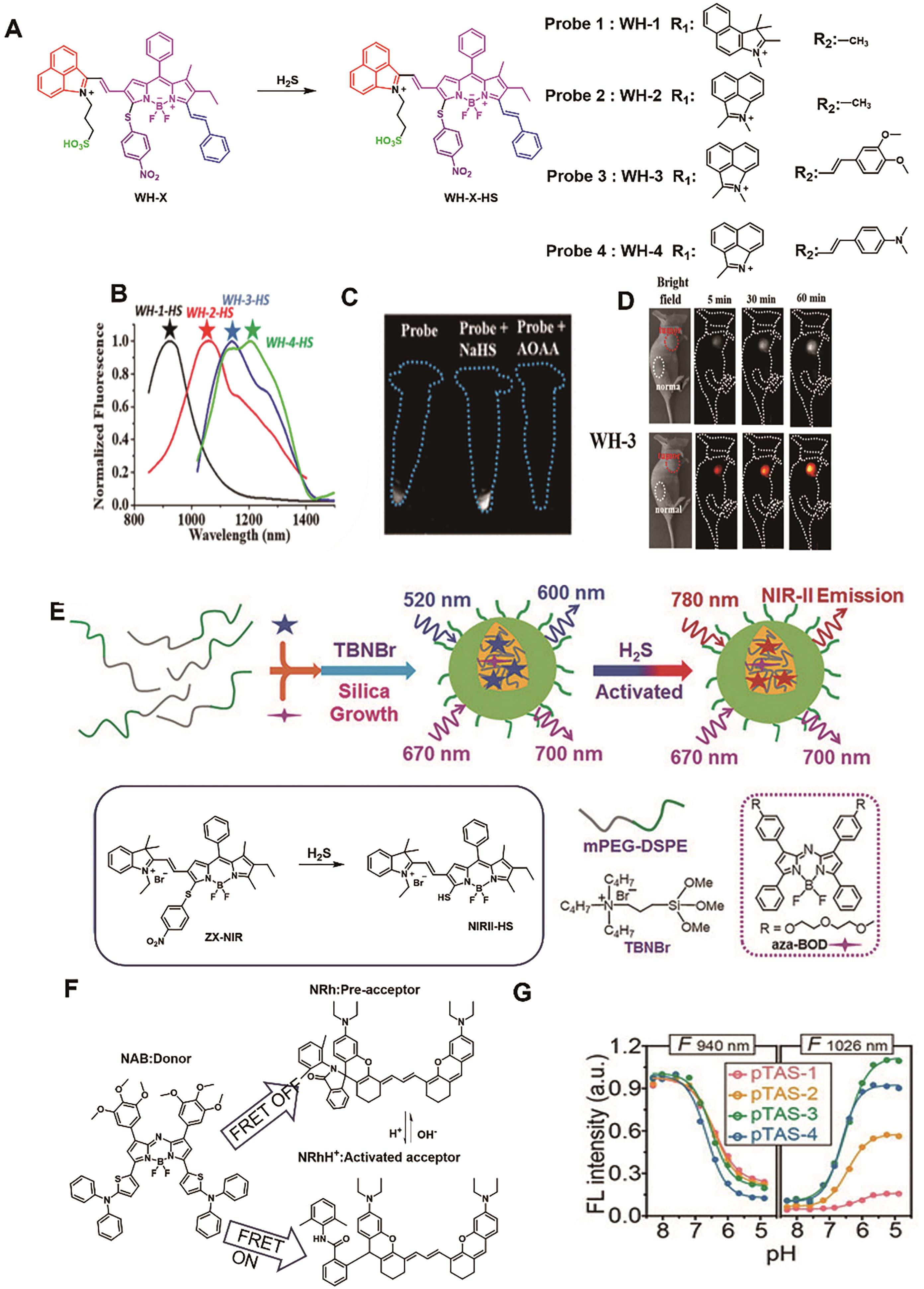
图10 用于结肠癌成像的代表性探针。 (A) WH-X系列探针的结构及响应机理[64]。 (B) WH-X探针被H2S激活后的荧光光谱。 (C) 经不同处理的细胞与探针WH-3(500 μmol/L)共孵育后的NIR-Ⅱ荧光成像[64]。 (D) 注射探针WH-3(20 μL, 1 mmol/L)后小鼠的NIR-Ⅱ荧光成像图[64]。 (E) 探针NIRII@Si的结构及响应机理[65]。 (F) FRET供体NAB和受体NRh的结构[67]。 (G) 探针pTAS在940和1026 nm处的荧光强度随pH值变化关系图[67]
Fig.10 Representative probes for colon cancer imaging. (A) Structure and response mechanism of WH-X probes[64]. (B) Fluorescence spectra of WH-X probe[64]. (C) Fluorescence images of different treated cells with probe WH-3 [64]. (D) NIR-Ⅱ fluorescence images of mice. after injection of probe WH-3 (20 μL, 1 mmol/L)[64]. (E) Structure and response mechanism of NIRII@Si[65]. (F) The structure of FRET donor NAB and acceptor NRh[67]. (G) Fluorescence intensity at 940 and 1026 nm of probe pTAS as a function of pH[67]

图11 用于卵巢癌成像的代表性探针。 (A)BOD-M-βGal结构及响应机理[70]。 (B)注射探针BOD-M-βGal(30 nmol)后小鼠的NIR-Ⅱ荧光成像图[70]。 (C)荧光团Flavchromenes的结构[71]。 (D)探针Flavchrom-4的响应机理[71]。 (E)探针与β-Gal共孵育后吸收光谱和发射光谱[71]。 (F)注射探针Flavchrom-4后小鼠的NIR-Ⅱ荧光成像图(左)和MOST成像图(右)[71]
Fig.11 Representative probes for imaging ovarian cancer. (A) Structure and response mechanism of BOD-M-βGal[70]. (B) NIR-Ⅱ fluorescence images of mice after injection of probe WH-3 (30 nmol)[70]. (C) Structure of Flavchromenes[71]. (D) Response mechanism of Flavchrom-4 probe[71]. (E) Absorption and fluorescence spectra of Flavchrom-4 after incubation with β-Gal[71]. (F) MOST images (left) and NIR-Ⅱ fluorescence images (right) of the ovarian cancer model after injection of Flavchrom-4[71]
| BH-NO2 | |||||
| LET-7 | |||||
| BP-A | |||||
| Rap-N | |||||
| NP-Q-NO2 | |||||
| NTR-InD | |||||
| RHC-NO2 | |||||
| Colorectal cancer | WH-3 | ||||
| NIR-Ⅱ@Si | 780/900 | 37 n | |||
| pTAS | |||||
| Ovarian cancers | βGal | BOD-M-βGal | |||
| Flavchrom-4 | 0.037 U/m |
表2 用于癌症模型的NIR-Ⅱ激活型探针 (Continued from preoious page)
Table 2 NIR-Ⅱ activatable probes for cancer models
| BH-NO2 | |||||
| LET-7 | |||||
| BP-A | |||||
| Rap-N | |||||
| NP-Q-NO2 | |||||
| NTR-InD | |||||
| RHC-NO2 | |||||
| Colorectal cancer | WH-3 | ||||
| NIR-Ⅱ@Si | 780/900 | 37 n | |||
| pTAS | |||||
| Ovarian cancers | βGal | BOD-M-βGal | |||
| Flavchrom-4 | 0.037 U/m |
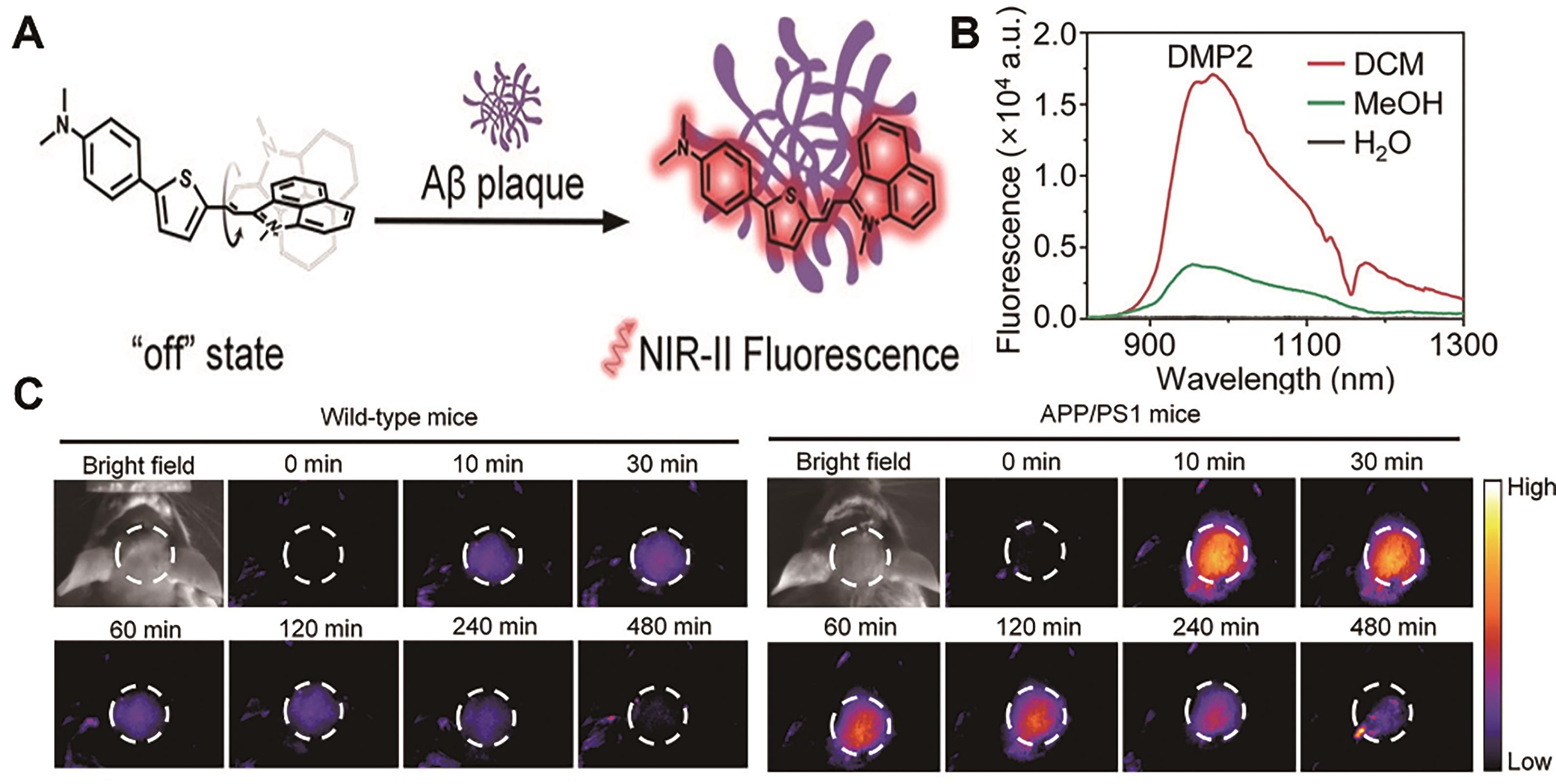
图12 用于阿尔兹海默症成像的代表性探针[73]。 (A) DMP2结构及响应机理。 (B) DMP2在不同极性溶剂中的荧光光谱。 (C)注射探针DMP2(2 mg/kg)后健康小鼠(左)和患有AD小鼠(右)的脑部NIR-Ⅱ荧光成像图
Fig.12 Representative probes for imaging Alzheimer′s disease[73]. (A) Structure and response mechanism of DMP2. (B) Fluorescence spectra of DMP2 in different solvents. (C) NIR-Ⅱ fluorescence imaging of the brain of healthy mice (left) and AD-model mice (right) after injection of probe DMP2 (2 mg/kg)
| 1 | HONG G, ANTARIS A L, DAI H. Near-infrared fluorophores for biomedical imaging[J]. Nat Biomed Eng, 2017, 1(1): 0010. |
| 2 | ANTARIS A L, CHEN H, CHENG K, et al. A small-molecule dye for NIR-Ⅱ imaging[J]. Nat Mater, 2015, 15(2): 235-242. |
| 3 | DIAO S, HONG G, ANTARIS A L, et al. Biological imaging without autofluorescence in the second near-infrared region[J]. Nano Res, 2015, 8(9): 3027-3034. |
| 4 | LI C, CHEN G, ZHANG Y, et al. Advanced fluorescence imaging technology in the near-infrared-Ⅱ window for biomedical applications[J]. J Am Chem Soc, 2020, 142(35): 14789-14804. |
| 5 | LEI Z, ZHANG F. Molecular Engineering of NIR-Ⅱ fluorophores for improved biomedical detection[J]. Angew Chem Int Ed, 2021, 60(30): 16294-16308. |
| 6 | YI H J, GHOSH D, HAM M H, et al. M13 Phage-functionalized single-walled carbon nanotubes as nanoprobes for second near-infrared window fluorescence imaging of targeted tumors[J]. Nano Lett, 2012, 12(3): 1176-1183. |
| 7 | LI Y, ZHANG P, TANG W, et al. Bright, magnetic NIR-Ⅱ quantum dot probe for sensitive dual-modality imaging and intensive combination therapy of cancer[J]. ACS Nano, 2022, 16(5): 8076-8094. |
| 8 | SONG Y, LU M, XIE Y, et al. Deep learning fluorescence imaging of visible to NIR-Ⅱ based on modulated multimode emissions lanthanide nanocrystals[J]. Adv Funct Mater, 2022, 32(45): 2206802. |
| 9 | TANG Y, LI Y, LU X, et al. Bio-erasable intermolecular donor-acceptor interaction of organic semiconducting nanoprobes for activatable NIR-Ⅱ fluorescence imaging[J]. Adv Funct Mater, 2019, 29(10): 1807376. |
| 10 | LIU X, YU B, SHEN Y, et al. Design of NIR-Ⅱ high performance organic small molecule fluorescent probes and summary of their biomedical application[J]. Coord Chem Rev, 2022, 468: 214609. |
| 11 | LI C, GUAN X, ZHANG X, et al. NIR-Ⅱ bioimaging of small molecule fluorophores: from basic research to clinical applications[J]. Biosens Bioelectron, 2022, 216: 114620. |
| 12 | SHOU K, QU C, SUN Y, et al. Multifunctional biomedical imaging in physiological and pathological conditions using a NIR-Ⅱ probe[J]. Adv Funct Mater, 2017, 27(23): 1700995. |
| 13 | TANG Y, PEI F, LU X, et al. Recent advances on activatable NIR-Ⅱ fluorescence probes for biomedical imaging[J]. Adv Opt Mater, 2019, 7(21): 1900917. |
| 14 | HUANG J, PU K. Activatable molecular probes for second near-infrared fluorescence, chemiluminescence, and photoacoustic imaging[J]. Angew Chem Int Ed, 2020, 59(29): 11717-11731. |
| 15 | LI D, PAN J, XU S, et al. Activatable second near-infrared fluorescent probes: a new accurate diagnosis strategy for diseases[J]. Biosensors, 2021, 11(11): 436. |
| 16 | ZHAO M, LI B, ZHANG H, et al. Activatable fluorescence sensors for in vivo bio-detection in the second near-infrared window[J]. Chem Sci, 2021, 12(10): 3448-3459. |
| 17 | ZHANG X, AN L, TIAN Q, et al. Tumor microenvironment-activated NIR-> reagents for tumor imaging and therapy[J]. J Mater Chem B, 2020, 8(22): 4738-4747. |
| 18 | NETEA M G, BALKWILL F, CHONCHOL M, et al. A guiding map for inflammation[J]. Nat Immunol, 2017, 18(8): 826-831. |
| 19 | KOTAS MAYA E, MEDZHITOV R. Homeostasis, inflammation, and disease susceptibility[J]. Cell, 2015, 160(5): 816-827. |
| 20 | TU Z, ZHONG Y, HU H, et al. Design of therapeutic biomaterials to control inflammation[J]. Nat Rev Mater, 2022, 7(7): 557-574. |
| 21 | METUSHI I, UETRECHT J, PHILLIPS E. Mechanism of isoniazid-induced hepatotoxicity: then and now[J]. Br J Clin Pharmacol, 2016, 81(6): 1030-1036. |
| 22 | YAMAMOTO H, WATANABE T, MIZUNO H, et al. In vivo evidence for accelerated generation of hydroxyl radicals in liver of Long-Evans Cinnamon (LEC) rats with acute hepatitis[J]. Free Radic Biol Med, 2001, 30(5): 547-554. |
| 23 | FENG W, ZHANG Y, LI Z, et al. Lighting up NIR-Ⅱ fluorescence in vivo: an activable probe for noninvasive hydroxyl radical imaging[J]. Anal Chem, 2019, 91(24): 15757-15762. |
| 24 | CHEN L, CHEN J, FANG Y, et al. A turn-on probe for detecting antituberculotic drug-induced liver injury in mice via NIR-Ⅱ fluorescence/optoacoustic imaging[J]. Chem Commun, 2021, 57(63): 7842-7845. |
| 25 | OU Y F, REN T B, YUAN L, et al. Molecular design of NIR-Ⅱ polymethine fluorophores for bioimaging and biosensing[J]. Chem Biomed Imaging, 2023, 1(3): 220-233. |
| 26 | TIAN Y, LIU S, CAO W, et al. H2O2-activated NIR-Ⅱ fluorescent probe with a large stokes shift for high-contrast imaging in drug-induced liver injury mice[J]. Anal Chem, 2022, 94(32): 11321-11328. |
| 27 | FERYSIUK K, WÓJCIAK K M. Reduction of nitrite in meat products through the application of various plant-based ingredients[J]. Antioxidants, 2020, 9(8): 711. |
| 28 | GEORGE J, TSUCHISHIMA M, TSUTSUMI M. Molecular mechanisms in the pathogenesis of N-nitrosodimethylamine induced hepatic fibrosis[J]. Cell Death Dis, 2019, 10(1): 18. |
| 29 | ZENG C, OUYANG J, SUN L, et al. An activatable probe for detection and therapy of food-additive-related hepatic injury via NIR-Ⅱ fluorescence/optoacoustic imaging and biomarker-triggered drug release[J]. Anal Chim Acta, 2022, 1208: 339831. |
| 30 | QIN Z, REN T B, ZHOU H, et al. NIRII-HDs: a versatile platform for developing activatable NIR-Ⅱ fluorogenic probes for reliable in vivo analyte sensing[J]. Angew Chem Int Ed, 2022, 61(19): e202201541. |
| 31 | LI D, WANG S, LEI Z, et al. Peroxynitrite activatable NIR-Ⅱ fluorescent molecular probe for drug-induced hepatotoxicity monitoring[J]. Anal Chem, 2019, 91(7): 4771-4779. |
| 32 | GRIFFITHS H R, GAO D, PARARASA C. Redox regulation in metabolic programming and inflammation[J]. Redox Biol, 2017, 12: 50-57. |
| 33 | HE L, HE L H, XU S, et al. Engineering of reversible NIR-Ⅱ redox-responsive fluorescent probes for imaging of inflammation in vivo[J]. Angew Chem Int Ed, 2022, 61(46): e202211409. |
| 34 | REN T B, WANG Z Y, XIANG Z, et al. A General strategy for development of activatable NIR-Ⅱ fluorescent probes for in vivo high-contrast bioimaging[J]. Angew Chem Int Ed, 2020, 60(2): 800-805. |
| 35 | CHEN Z, ZHANG Z, ZENG F, et al. Visualizing detection of diabetic liver injury by a biomarker-activatable probe via NIR-Ⅱ fluorescence imaging[J]. Chem Biomed Imaging, 2023: https://doi.org/10.1021/cbmi.3c00030. |
| 36 | ZHANG X, SHEN S, LIU D, et al. Combination of changeable π-conjugation and hydrophilic groups for developing water-soluble small-molecule NIR-Ⅱ fluorogenic probes[J]. Chem Sci, 2023, 14(11): 2928-2934. |
| 37 | DARMON M, OSTERMANN M, CERDA J, et al. Diagnostic work-up and specific causes of acute kidney injury[J]. Intensive Care Med, 2017, 43(6): 829-840. |
| 38 | VAIDYA V S, OZER J S, DIETERLE F, et al. Kidney injury molecule-1 outperforms traditional biomarkers of kidney injury in preclinical biomarker qualification studies[J]. Nat Biotechnol, 2010, 28(5): 478-485. |
| 39 | TAN J, YIN K, OUYANG Z, et al. Real-time monitoring renal impairment due to drug-induced AKI and diabetes-caused CKD using an NAG-activatable NIR-Ⅱ nanoprobe[J]. Anal Chem, 2021, 93(48): 16158-16165. |
| 40 | OUYANG J, SUN L, ZENG F, et al. Rational design of stable heptamethine cyanines and development of a biomarker-activatable probe for detecting acute lung/kidney injuries via NIR-Ⅱ fluorescence imaging[J]. Analyst, 2022, 147(3): 410-416. |
| 41 | TYAGI P, MOON C H, JANICKI J, et al. Recent advances in imaging and understanding interstitial cystitis[J]. F1000Research, 2018, 7: 1771. |
| 42 | ZHANG X, CHEN Y, HE H, et al. ROS/RNS and base dual activatable merocyanine-based NIR-Ⅱ fluorescent molecular probe for in vivo biosensing[J]. Angew Chem Int Ed, 2021, 60(50): 26337-26341. |
| 43 | CHEN J, CHEN L, WU Y, et al. A H2O2-activatable nanoprobe for diagnosing interstitial cystitis and liver ischemia-reperfusion injury via multispectral optoacoustic tomography and NIR-Ⅱ fluorescent imaging[J]. Nat Commun, 2021, 12(1): 6870. |
| 44 | WU W, YANG Y, YANG Y, et al. Molecular engineering of an organic NIR-Ⅱ fluorophore with aggregation-induced emission characteristics for in vivo imaging[J]. Small, 2019, 15(20): 1805549. |
| 45 | HUNTER D J, BIERMA-ZEINSTRA S. Osteoarthritis[J]. Lancet, 2019, 393(10182): 1745-1759. |
| 46 | WU P, ZHU Y, CHEN L, et al. A Fast-responsive OFF-ON near-infrared-Ⅱ fluorescent probe for in vivo detection of hypochlorous acid in rheumatoid arthritis[J]. Anal Chem, 2021, 93(38): 13014-13021. |
| 47 | GE X, LOU Y, SU L, et al. Single wavelength laser excitation ratiometric NIR-Ⅱ fluorescent probe for molecule imaging in vivo[J]. Anal Chem, 2020, 92(8): 6111-6120. |
| 48 | SHE Z, CHEN J, SUN L, et al. An NO-responsive probe for detecting acute inflammation using NIR-Ⅱ fluorescence/optoacoustic imaging[J]. Chem Commun, 2022, 58(94): 13123-13126. |
| 49 | KOBAYASHI T, SIEGMUND B, LE BERRE C, et al. Ulcerative colitis[J]. Nat Rev Dis Prim, 2020, 6(1): 74. |
| 50 | ZENG Z, OUYANG J, SUN L, et al. A Biomarker-responsive nanosystem with colon‐targeted delivery for ulcerative colitis's detection and treatment with optoacoustic/NIR-Ⅱ fluorescence imaging[J]. Adv Healthc Mater, 2022, 11(22): 2201544. |
| 51 | LLOVET J M, KELLEY R K, VILLANUEVA A, et al. Hepatocellular carcinoma[J]. Nat Rev Dis Prim, 2021, 7(1): 6. |
| 52 | ZENG Z, OUYANG J, SUN L, et al. Activatable nanocomposite probe for preoperative location and intraoperative navigation for orthotopic hepatic tumor resection via MSOT and aggregation-induced Near-IR-Ⅰ/Ⅱ fluorescence imaging[J]. Anal Chem, 2020, 92(13): 9257-9264. |
| 53 | PAN Y, LEI S, ZHANG J, et al. Activatable NIR-Ⅱ fluorescence probe for highly sensitive and selective visualization of glutathione in vivo[J]. Anal Chem, 2021, 93(51): 17103-17109. |
| 54 | WALLACE J L, WANG R. Hydrogen sulfide-based therapeutics: exploiting a unique but ubiquitous gasotransmitter[J]. Nat Rev Drug Discov, 2015, 14(5): 329-345. |
| 55 | YANG L, JIANG L, XU F, et al. Hydrogen sulfide activatable NIR-Ⅱ fluorescent probe for highly specific imaging of breast cancer[J]. Sens Actuators B: Chem, 2023, 379: 133251. |
| 56 | XU S, WANG Q, ZHANG Q, et al. Real time detection of ESKAPE pathogens by a nitroreductase-triggered fluorescence turn-on probe[J]. Chem Commun, 2017, 53(81): 11177-11180. |
| 57 | LAN Q, YU P, YAN K, et al. Polymethine molecular platform for ratiometric fluorescent probes in the second near-infrared window[J]. J Am Chem Soc, 2022, 144(46): 21010-21015. |
| 58 | SCHNEBLE E J, GRAHAM L J, SHUPE M P, et al. Future directions for the early detection of recurrent breast cancer[J]. J Cancer, 2014, 5(4): 291-300. |
| 59 | OUYANG J, SUN L, ZENG Z, et al. Nanoaggregate probe for breast cancer metastasis through multispectral optoacoustic tomography and aggregation-induced NIR-Ⅰ/Ⅱ fluorescence imaging[J]. Angew Chem Int Ed, 2019, 59(25): 10111-10121. |
| 60 | THAI A A, SOLOMON B J, SEQUIST L V, et al. Lung cancer[J]. Lancet, 2021, 398(10299): 535-554. |
| 61 | WANG R, CHEN J, GAO J, et al. A molecular design strategy toward enzyme-activated probes with near-infrared Ⅰ and Ⅱ fluorescence for targeted cancer imaging[J]. Chem Sci, 2019, 10(30): 7222-7227. |
| 62 | ZHANG X, LI X, SHI W, et al. Sensitive imaging of tumors using a nitroreductase-activated fluorescence probe in the NIR-Ⅱ window[J]. Chem Commun, 2021, 57(66): 8174-8177. |
| 63 | SUO Y, WU F, XU P, et al. NIR-Ⅱ fluorescence endoscopy for targeted imaging of colorectal cancer[J]. Adv Healthc Mater, 2019, 8(23): 1900974. |
| 64 | DOU K, FENG W, FAN C, et al. Flexible designing strategy to construct activatable NIR-Ⅱ fluorescent probes with emission maxima beyond 1200 nm[J]. Anal Chem, 2021, 93(8): 4006-4014. |
| 65 | XU G, YAN Q, LV X, et al. Imaging of colorectal cancers using activatable nanoprobes with second near-infrared window emission[J]. Angew Chem Int Ed, 2018, 57(14): 3626-3630. |
| 66 | XUAN W, XIA Y, LI T, et al. Molecular self-assembly of bioorthogonal aptamer-prodrug conjugate micelles for hydrogen peroxide and pH-independent cancer chemodynamic therapy[J]. J Am Chem Soc, 2019, 142(2): 937-944. |
| 67 | ZHAO M, WANG J, LEI Z, et al. NIR-Ⅱ pH sensor with a FRET adjustable transition point for in situ dynamic tumor microenvironment visualization[J]. Angew Chem Int Ed, 2021, 60(10): 5091-5095. |
| 68 | JESSMON P, BOULANGER T, ZHOU W, et al. Epidemiology and treatment patterns of epithelial ovarian cancer[J]. Expert Rev Anticancer Ther, 2017, 17(5): 427-437. |
| 69 | KIM E J, KUMAR R, SHARMA A, et al. In vivo imaging of β-galactosidase stimulated activity in hepatocellular carcinoma using ligand-targeted fluorescent probe[J]. Biomaterials, 2017, 122: 83-90. |
| 70 | CHEN J A, PAN H, WANG Z, et al. Imaging of ovarian cancers using enzyme activatable probes with second near-infrared window emission[J]. Chem Commun, 2020, 56(18): 2731-2734. |
| 71 | ZHANG L, ZHANG Y, CHI W, et al. “Crossbreeding” small-molecular weight NIR-Ⅱ flavchromenes endows activatable multiplexed in vivo imaging[J]. ACS Mater Lett, 2022, 4(8): 1493-1502. |
| 72 | SCHELTENS P, DE STROOPER B, KIVIPELTO M, et al. Alzheimer's disease[J]. Lancet, 2021, 397(10284): 1577-1590. |
| 73 | MIAO J, MIAO M, JIANG Y, et al. An activatable NIR-Ⅱ fluorescent reporter for in vivo imaging of amyloid-β plaques[J]. Angew Chem Int Ed, 2023, 62(7): e202216351. |
| [1] | 杜慧, 姚晨阳, 彭皓, 姜波, 李顺祥, 姚俊烈, 郑方, 杨方, 吴爱国. 过渡金属掺杂磁性纳米粒子在生物医学领域中的研究进展[J]. 应用化学, 2022, 39(3): 391-406. |
| [2] | 王涛,马拉毛草,马恒昌. 基于聚集诱导发光荧光探针的细胞成像研究进展[J]. 应用化学, 2018, 35(10): 1155-1165. |
| [3] | 黄子珂, 刘超, 付强强, 李进, 邹建梅, 谢斯滔, 邱丽萍. 核酸适配体荧光探针在生化分析和生物成像中的研究进展[J]. 应用化学, 2018, 35(1): 28-39. |
| [4] | 周佳, 倪赟, 张承武, 仇兴汉, 赵燕菲, 白磊, 张高宾, 李林. 嘧啶基双光子荧光染料的设计合成与生物成像[J]. 应用化学, 2017, 34(12): 1450-1456. |
| [5] | 周佳, 倪赟, 张承武, 仇兴汉, 赵燕菲, 白磊, 张高宾, 李林. 嘧啶基双光子荧光染料的设计合成与生物成像[J]. 应用化学, 2017, 34(12): 0-0. |
| [6] | 李海东,姚起超,樊江莉,杜健军,彭孝军. 钯离子荧光探针的研究进展[J]. 应用化学, 2016, 33(10): 1099-1114. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||