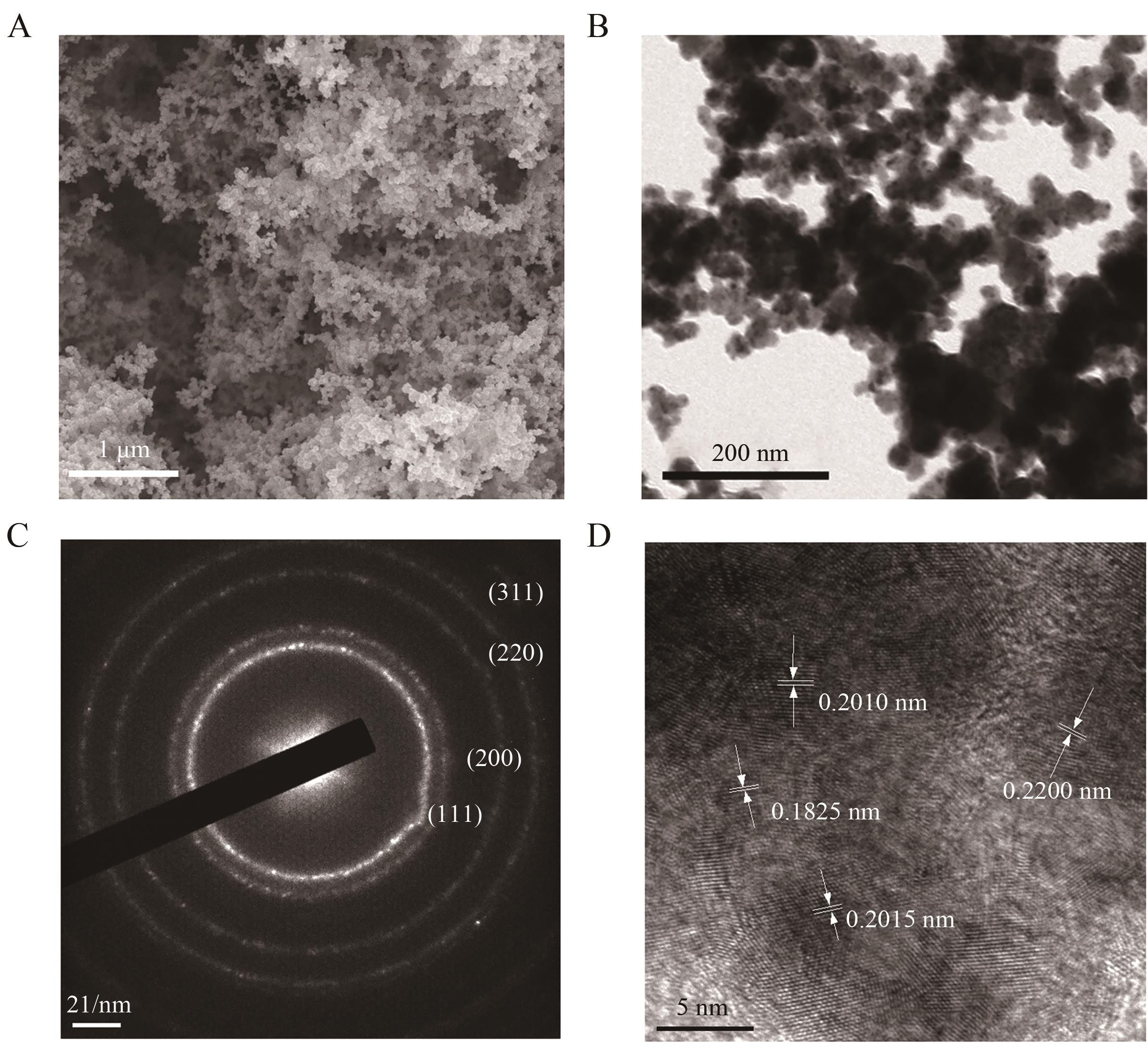
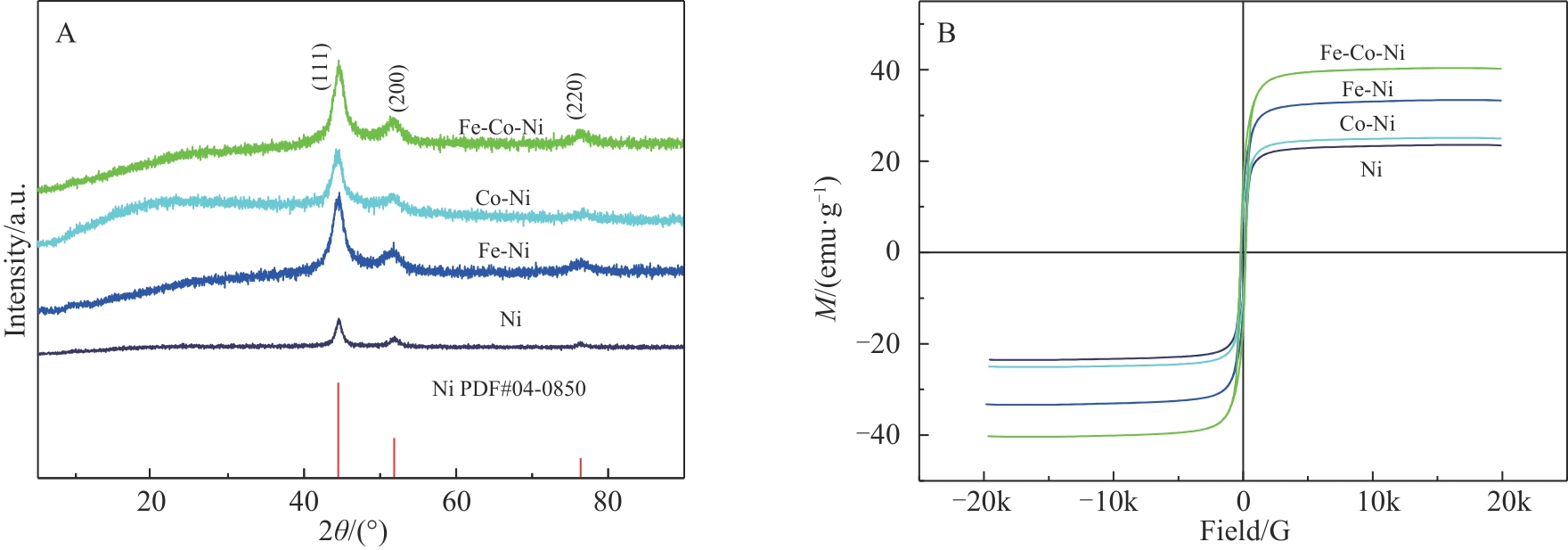
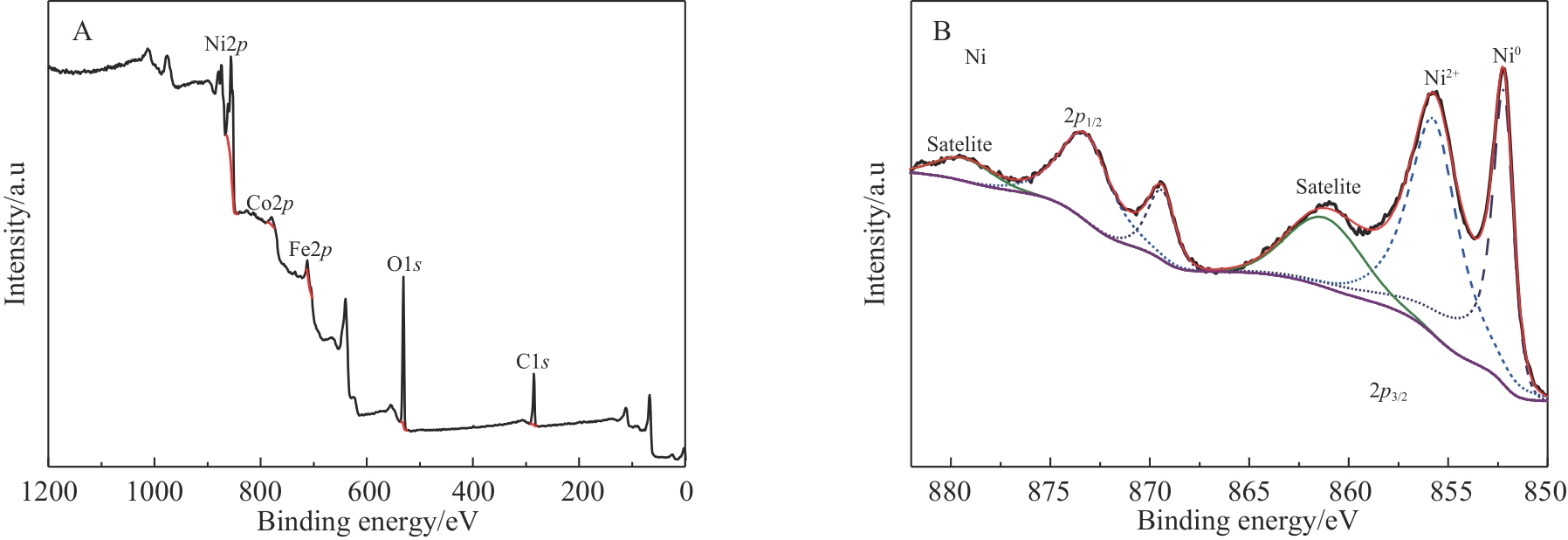
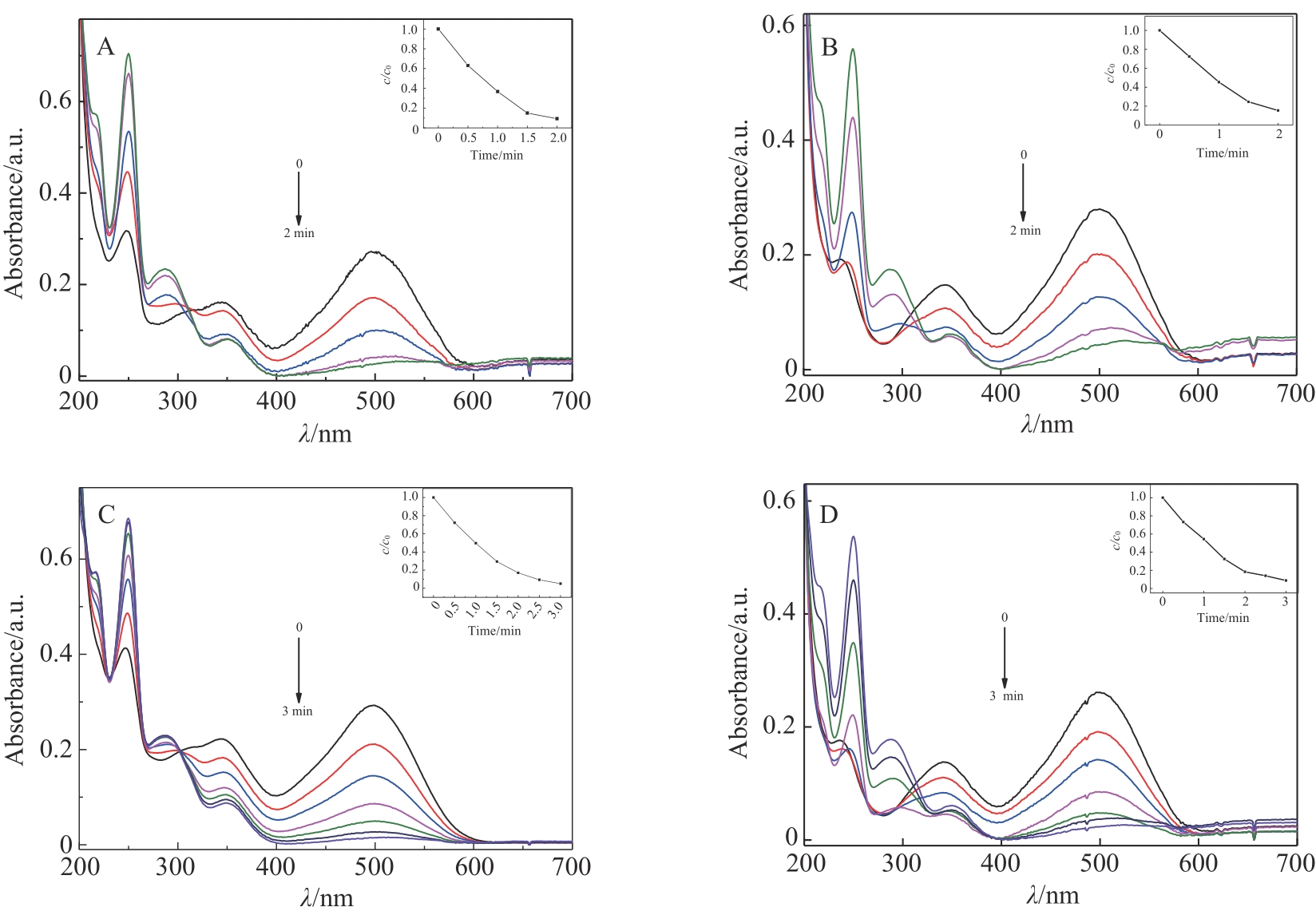
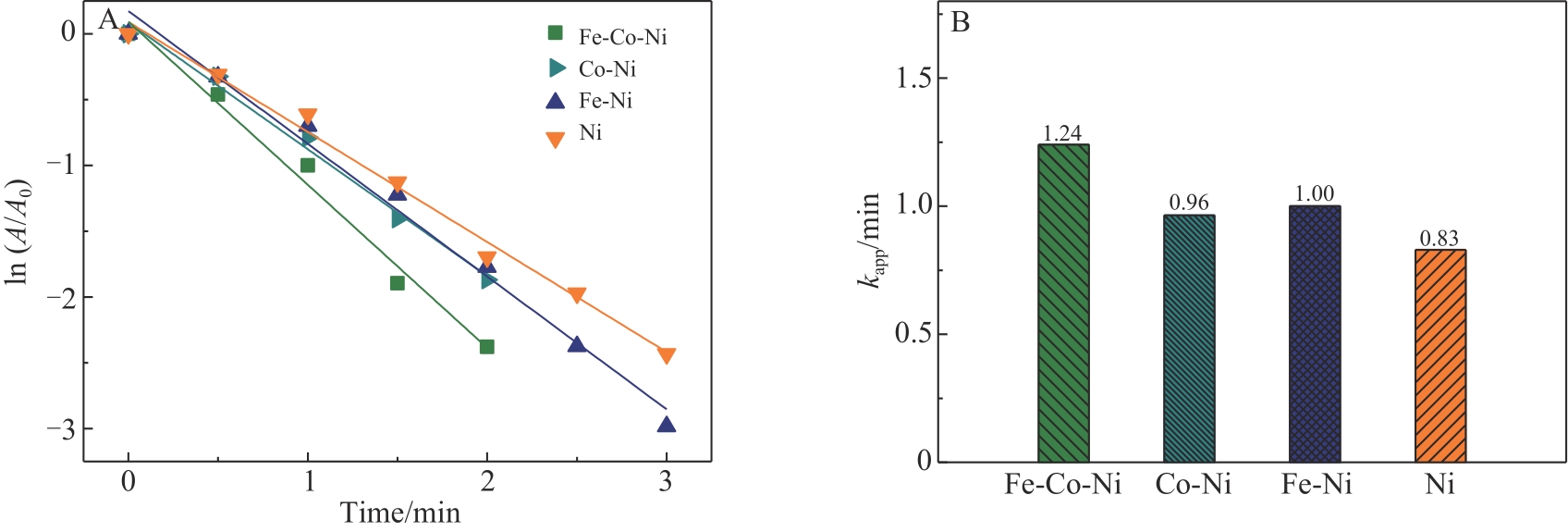
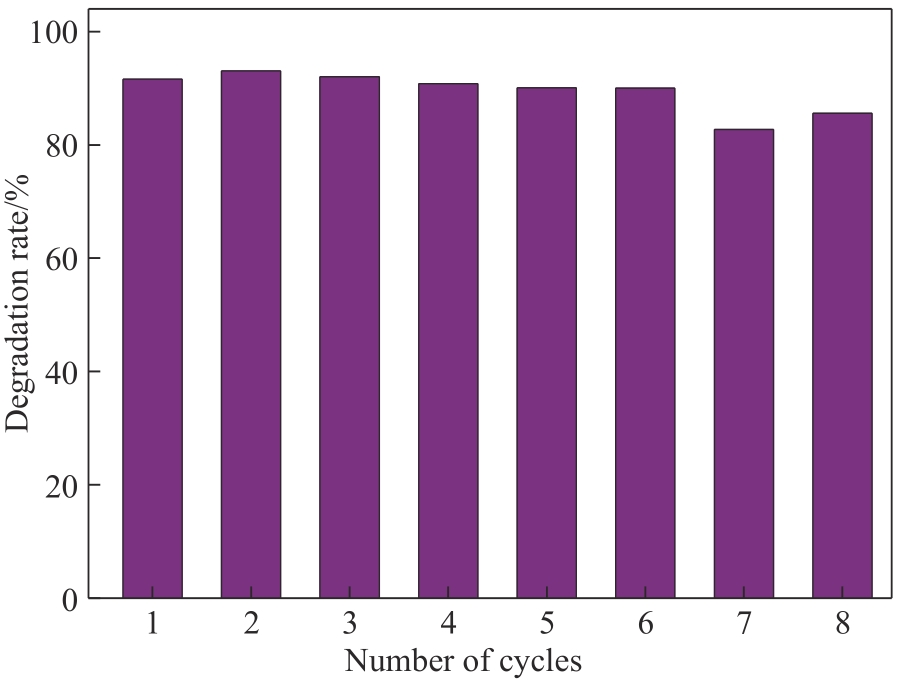
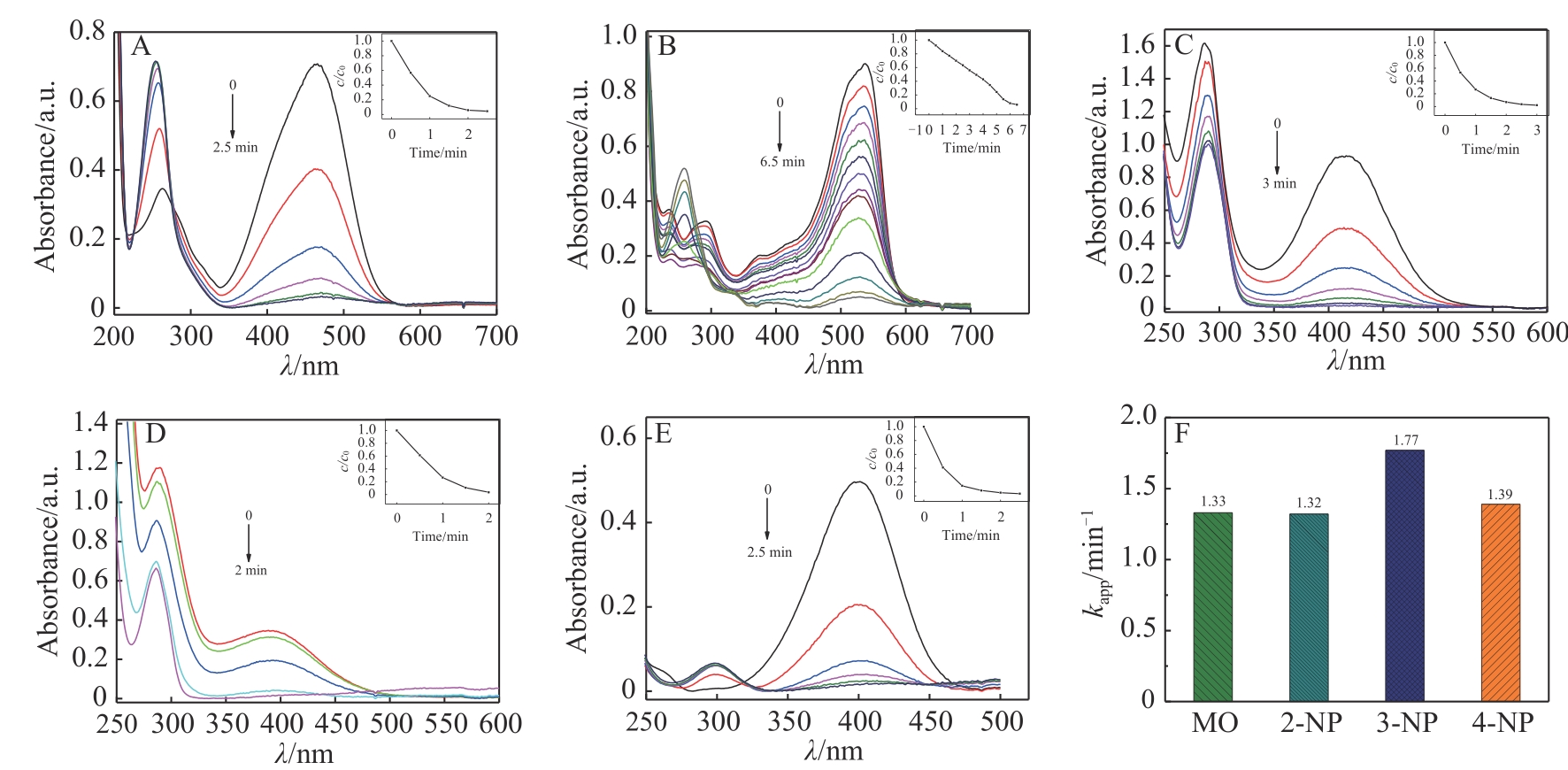
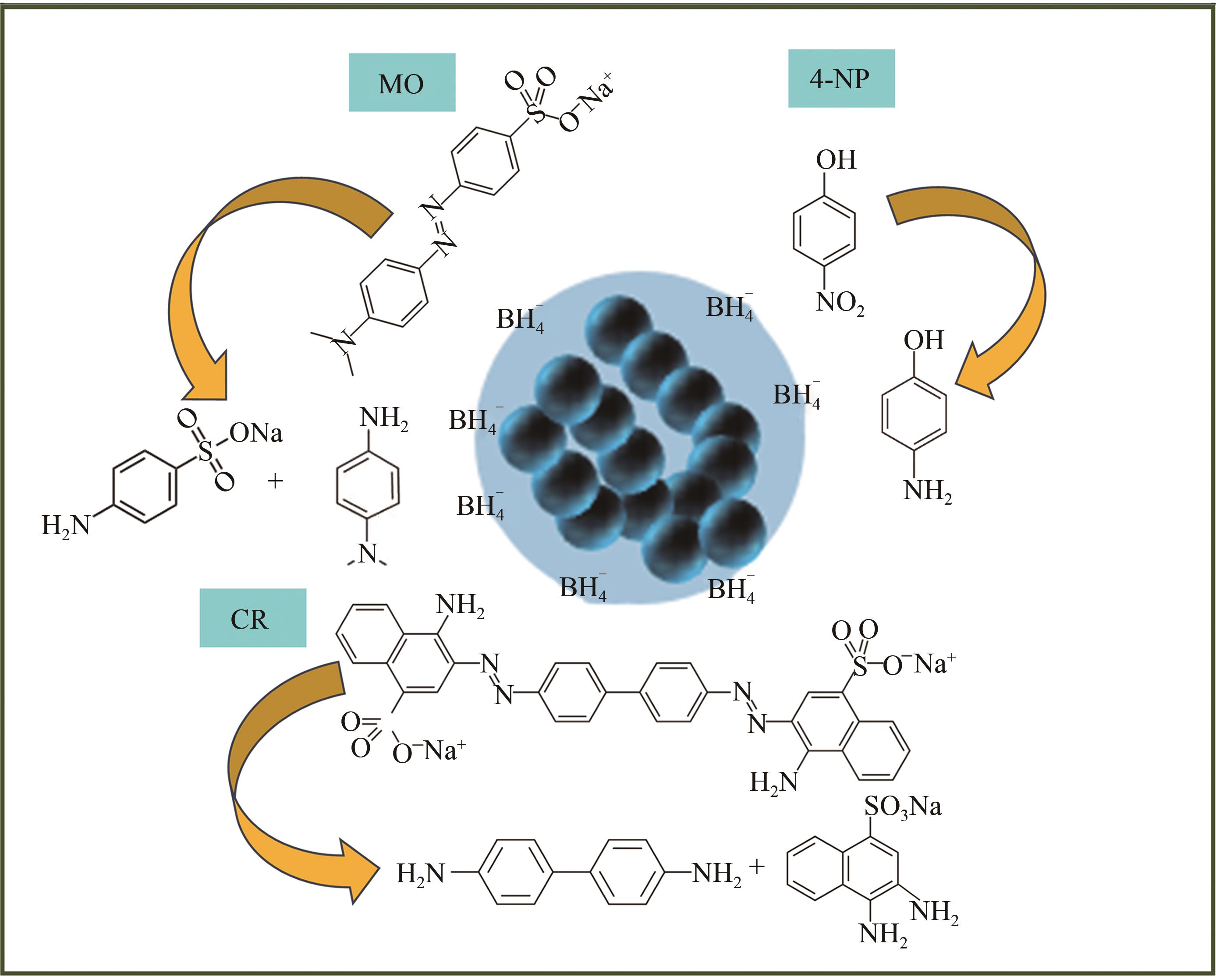
| 1 |
MAENO K, PATEL B R, ENDO T, et al. Angle-sensitive photonic crystals for simultaneous detection and photocatalytic degradation of hazardous diazo compounds[J]. Micromachines, 2020, 11(1): 93.
|
| 2 |
DIHOM H R, AL-SHAIBANI M M, MOHAMED R M S R, et al. Photocatalytic degradation of disperse azo dyes in textile wastewater using green zinc oxide nanoparticles synthesized in plant extract: a critical review[J]. J Water Proc Eng, 2022, 47: 102705.
|
| 3 |
LIN S H, PENG C F. Continuous treatment of textile wastewater by combined coagulation, electrochemical oxidation and activated sludge[J]. Water Res, 1996, 30(3): 587-592.
|
| 4 |
PAŹDZIOR K, BILIŃSKA L, LEDAKOWICZ S. A review of the existing and emerging technologies in the combination of AOPs and biological processes in industrial textile wastewater treatment[J]. Chem Eng J, 2019, 376: 120597.
|
| 5 |
WU J, WANG T. Ozonation of aqueous azo dye in a semi-batch reactor[J]. Water Res, 2001, 35(4): 1093-1099.
|
| 6 |
RATHI B S, KUMAR P S. Sustainable approach on the biodegradation of azo dyes: a short review[J]. Curr Opin Green Sustainable Chem, 2022, 33: 100578.
|
| 7 |
NDLOVU L N, MALATJIE K I, DONGA C, et al. Catalytic degradation of methyl orange using beta cyclodextrin modified polyvinylidene fluoride mixed matrix membranes imbedded with in-situ generated palladium nanoparticles[J]. J Appl Polym Sci, 2023, 140(1): e53270.
|
| 8 |
于宗龙, 贾春晓, 路鑫, 等. 离子液体@TiO2纳米复合材料的制备及其光催化性能[J]. 应用化学, 2016, 33(4): 459-465.
|
|
YU Z L, JIA C X, LU X, et al. Synthesis of ionic liquid@TiO2 nanocomposites and their photocatalytic performance[J]. Chin J Appl Chem, 2016, 33(4): 459-465.
|
| 9 |
李文进, 姚威龙, 徐嘉鑫, 等. Ho3+掺杂铁酸铋的制备及光催化性[J]. 应用化学, 2019, 36(1): 91-96.
|
|
LI W J, YAO W L, XU J X, et al. Preparation and photocatalytic properties of Ho3+ doping BiFeO3[J]. Chin J Appl Chem, 2019, 36(1): 91-96.
|
| 10 |
SUN L, MO Y, ZHANG L. A mini review on bio-electrochemical systems for the treatment of azo dye wastewater: state-of-the-art and future prospects[J]. Chemosphere, 2022, 294: 133801.
|
| 11 |
NAZ M, RAFIQ A, IKRAM M, et al. Elimination of dyes by catalytic reduction in the absence of light: a review[J]. J Mater Sci, 2021, 56: 15572-15608.
|
| 12 |
ZHU Y, WANG W, NI J, et al. Cultivation of granules containing anaerobic decolorization and aerobic degradation cultures for the complete mineralization of azo dyes in wastewater[J]. Chemosphere, 2020, 246: 125753.
|
| 13 |
ZHENG L Q, YU X D, XU J J, et al. Reversible catalysis for the reaction between methyl orange and NaBH4 by silver nanoparticles[J]. Chem Commun, 2015, 51(6): 1050-1053.
|
| 14 |
MEI S, PAN M, GAO S, et al. Organic-inorganic bimetallic hybrid particles with controllable morphology for the catalytic degradation of organic dyes[J]. New J Chem, 2020, 44(20): 8366-8378.
|
| 15 |
SHAH S A, AHMAD Z, KHAN S A, et al. Biomass impregnated zero-valent Ag and Cu supported-catalyst: evaluation in the reduction of nitrophenol and discoloration of dyes in aqueous medium[J]. J Organomet Chem, 2021, 938: 121756.
|
| 16 |
KHAN N, SHAHIDA B, KHAN S A, et al. Anchoring Zero-Valent Cu and Ni nanoparticles on carboxymethyl cellulose-polystyrene-block polyisoprene-block polystyrene composite films for nitrophenol reduction and dyes degradation[J]. J Polym Environ, 2023, 31(2): 608-620.
|
| 17 |
HABAS S E, LEE H, RADMILOVIC V, et al. Shaping binary metal nanocrystals through epitaxial seeded growth[J]. Nat Mater, 2007, 6(9): 692-697.
|
| 18 |
PANDEY P C, MITRA M D, TIWARI A K, et al. Synthetic incorporation of palladium-nickel bimetallic nanoparticles within mesoporous silica/silica nanoparticles as efficient and cheaper catalyst for both cationic and anionic dyes degradation[J]. J Environ Sci Health, Part A, 2021, 56(4): 460-472.
|
| 19 |
ARIF M. Catalytic degradation of azo dyes by bimetallic nanoparticles loaded in smart polymer microgels[J]. RSC Adv, 2023, 13(5): 3008-3019.
|
| 20 |
GONG Z, MA T, LIANG F. Syntheses of magnetic blackberry-like Ni@Cu@Pd nanoparticles for efficient catalytic reduction of organic pollutants[J]. J Alloys Compd, 2021, 873: 159802.
|
| 21 |
ALSHEHRI A A, MALIK M A. Facile one-pot biogenic synthesis of Cu-Co-Ni trimetallic nanoparticles for enhanced photocatalytic dye degradation[J]. Catalysts, 2020, 10(10): 1138.
|
| 22 |
AHMAD I, MANZOOR K, AALAM G, et al. Facile synthesis of L-tryptophan functionalized magnetic nanophotocatalyst supported by copper nanoparticles for selective reduction of organic pollutants and degradation of azo dyes[J]. Catal Lett, 2022: 1-20.
|
| 23 |
DEOKAR G K, INGALE A G. Exploring effective catalytic degradation of organic pollutant dyes using environment benign, green engineered gold nanoparticles[J]. Inorg Chem Commun, 2023, 151: 110649.
|
| 24 |
ISMAIL M, KHAN M I, KHAN S B, et al. Green synthesis of plant supported Cu single bond Ag and Cu single bond Ni bimetallic nanoparticles in the reduction of nitrophenols and organic dyes for water treatment[J]. J Mol Liq, 2018, 260: 78-91.
|
| 25 |
CHEN R, WANG W, ZHAO X, et al. Rapid hydrothermal synthesis of magnetic CoxNi1- xFe2O4 nanoparticles and their application on removal of Congo red[J]. Chem Eng J, 2014, 242: 226-233.
|
| 26 |
ADITYA T, PAL A, PAL T. Nitroarene reduction: a trusted model reaction to test nanoparticle catalysts[J]. Chem Commun, 2015, 51(46): 9410-9431.
|
| 27 |
AMIR M, KURTAN U, BAYKAL A. Rapid color degradation of organic dyes by Fe3O4@His@Ag recyclable magnetic nanocatalyst[J]. J Ind Eng Chem, 2015, 27: 347-353.
|
| 28 |
VIDHU V K, PHILIP D. Catalytic degradation of organic dyes using biosynthesized silver nanoparticles[J]. Micron, 2014, 56: 54-62.
|
| 29 |
LUO S, QIN P, SHAO J, et al. Synthesis of reactive nanoscale zero valent iron using rectorite supports and its application for orange II removal[J]. Chem Eng J, 2013, 223: 1-7.
|
| 30 |
YANG Y, ZHANG H, HUANG H, et al. Iron-loaded carbon nanotube-microfibrous composite for catalytic wet peroxide oxidation of m-cresol in a fixed bed reactor[J]. Environ Sci Pollut Res, 2020, 27: 6338-6351.
|
| 31 |
BHOL P, MOHANTY P S. Smart microgel-metal hybrid particles of PNIPAM-co-PAA@AgAu: synthesis, characterizations and modulated catalytic activity[J]. J Phys: Condens Mat, 2020, 33(8): 084002.
|

 ), Fu LIU4(
), Fu LIU4( )
)