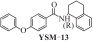
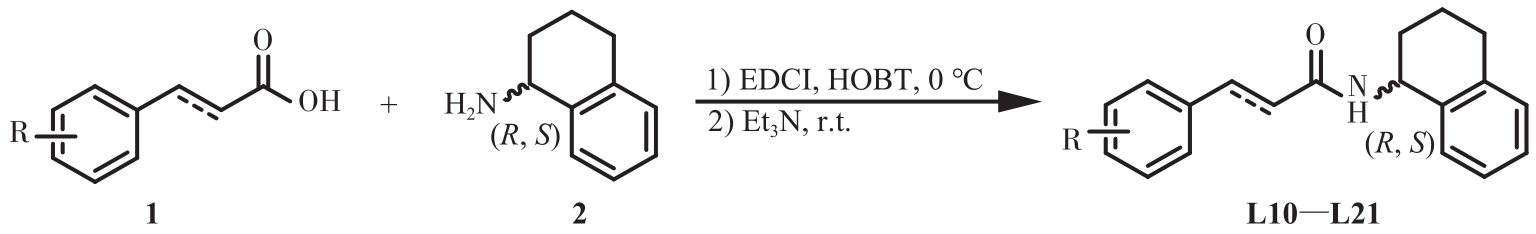| 1 |
SUNG H, FERLAY J, SIEGEL R L, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71(3): 209-249.
|
| 2 |
STANAWAY J D, AFSHIN A, GAKIDOU E, et al. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks for 195 countries and territories, 1990-2017: a systematic analysis for the Global burden of disease study 2017[J]. The Lancet, 2018, 392(10159): 1923-1994.
|
| 3 |
FITZMAURICE C, AKINYEMIJU T F,Al LAMI F H, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2017: a systematic analysis for the global burden of disease study[J]. JAMA Oncol, 2019, 5(12): 1749-1768.
|
| 4 |
国家卫生健康委办公厅. 原发性肝癌诊疗指南(2022年版)[J]. 临床肝胆病杂志, 2022, 38(2): 288-303.
|
|
General Office of National Health Commission. Standard for diagnosis and treatment of primary liver cancer (2022 edition)[J]. J Clin Hepatol, 2022, 38(2): 288-303.
|
| 5 |
FAN Y, XUE H, ZHENG H. Systemic therapy for hepatocellular carcinoma: current updates and outlook[J]. J Hepatocell Carcinoma, 2022, 9: 233-263.
|
| 6 |
AWOSIKA J, SOHAL D. A narrative review of systemic treatment options for hepatocellular carcinoma: state of the art review[J]. J Gastrointest Oncol, 2022, 13(1): 426-437.
|
| 7 |
ATWA S M, ODENTHAL M, EL TAYEBI H M. Genetic heterogeneity, therapeutic hurdle confronting sorafenib and immune checkpoint inhibitors in hepatocellular carcinoma[J]. Cancers, 2021, 13(17): 4343.
|
| 8 |
BAO M H, WONG C C. Hypoxia, metabolic reprogramming, and drug resistance in liver cancer[J]. Cells, 2021, 10(7): 1715.
|
| 9 |
TOVAR V, CORNELLA H, MOEINI A, et al. Tumour initiating cells and IGF/FGF signalling contribute to sorafenib resistance in hepatocellular carcinoma[J]. Gut, 2017, 66(3): 530-540.
|
| 10 |
RUWIZHI N, ADERIBIGBE B A. Cinnamic acid derivatives and their biological efficacy[J]. Int J Mol Sci, 2020, 21(16): 5712.
|
| 11 |
杨少梅. 二芳醚类酪氨酸激酶抑制剂的设计、合成及抗肿瘤活性研究[D]. 厦门: 厦门大学, 2014.
|
|
YANG S M. Design, synthesis of diaryl ther tyrosine kinases inhibitor and study of their antitumor activities[D]. Xiamen: Xiamen University, 2014.
|
| 12 |
VASCONCELOS N G, CRODA J, SIMIONATTO S. Antibacterial mechanisms of cinnamon and its constituents: a review[J]. Microb Pathog, 2018, 120: 198-203.
|
| 13 |
MINGOIA M, CONTE C, DI RIENZO A, et al. Synthesis and biological evaluation of novel cinnamic acid-based antimicrobials[J]. Pharmaceuticals, 2022, 15(2): 228.
|
| 14 |
SOVA M. Antioxidant and antimicrobial activities of cinnamic acid derivatives[J]. Mini Rev Med Chem, 2012, 12(8): 749-767.
|
| 15 |
ABAZARI M F, NASIRI N, KARIZI S Z, et al. An updated review of various medicinal applications of p-Co umaric acid: from antioxidative and anti-inflammatory properties to effects on cell cycle and proliferation[J]. Mini Rev Med Chem, 2021, 21(15): 2187-2201.
|
| 16 |
LAN J S, HOU J W, LIU Y, et al. Design, synthesis and evaluation of novel cinnamic acid derivatives bearing N-benzyl pyridinium moiety as multifunctional cholinesterase inhibitors for Alzheimer's disease[J]. J Enzyme Inhib Med Chem, 2017, 32(1): 776-788.
|
| 17 |
WANG R, YANG W, FAN Y, et al. Design and synthesis of the novel oleanolic acid-cinnamic acid ester derivatives and glycyrrhetinic acid-cinnamic acid ester derivatives with cytotoxic properties[J]. Bioorg Chem, 2019, 88: 102951.
|
| 18 |
VALE J A D, RODRIGUES M P, LIMA A M A, et al. Synthesis of cinnamic acid ester derivatives with antiproliferative and antimetastatic activities on murine melanoma cells[J]. Biomed Pharmacother, 2022, 148: 112689.
|
| 19 |
MALHEIRO J F, MAILLARD J Y, BORGES F, et al. Evaluation of cinnamaldehyde and cinnamic acid derivatives in microbial growth control[J]. Int Biodeterior Biodegrad, 2019, 141: 71-78.
|

 )
)