应用化学 ›› 2026, Vol. 43 ›› Issue (2): 259-274.DOI: 10.19894/j.issn.1000-0518.250107
改性聚苯并咪唑耐溶剂纳滤膜的制备及性能
- 辽宁师范大学化学化工学院,大连 116029
-
收稿日期:2025-03-14接受日期:2025-06-24出版日期:2026-02-01发布日期:2026-03-06 -
通讯作者:陈冬菊 -
基金资助:国家自然科学基金(22075121);大连理工大学精细化工国家重点实验室开放课题项目(KF2205)
Preparation and Properties of Modified Polybenzimidazole Solvent Resistant Nanofiltration Membranes
Li-Na JIAO, Chen-Yu WANG, Yu-Ting JIANG, Miao-Miao HE, Dong-Ju CHEN( )
)
- School of Chemistry and Chemical Engineering,Liaoning Normal University,Dalian 116029,China
-
Received:2025-03-14Accepted:2025-06-24Published:2026-02-01Online:2026-03-06 -
Contact:Dong-Ju CHEN -
About author:Dongjuchen_1978@lnnu.edu.cn
-
Supported by:the National Natural Science Foundation of China(22075121);the Open Project of State Key Laboratory of Fine Chemicals, Dalian University of Technology(KF2205)
摘要:
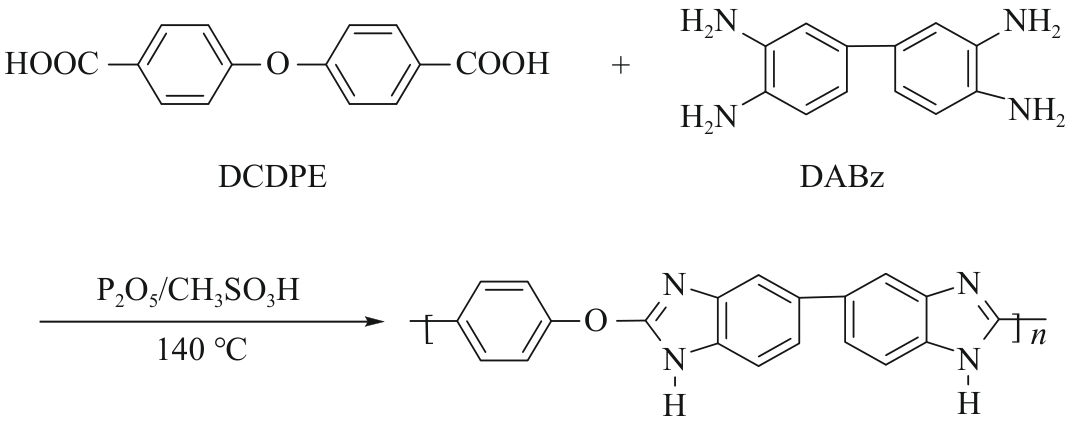
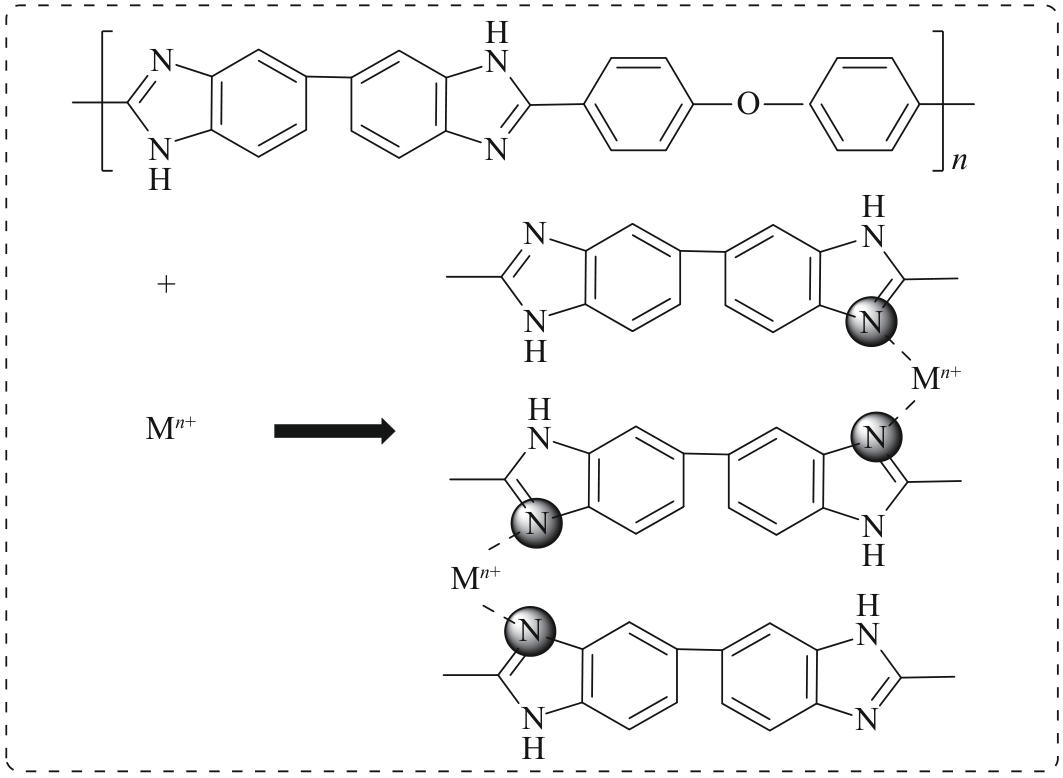
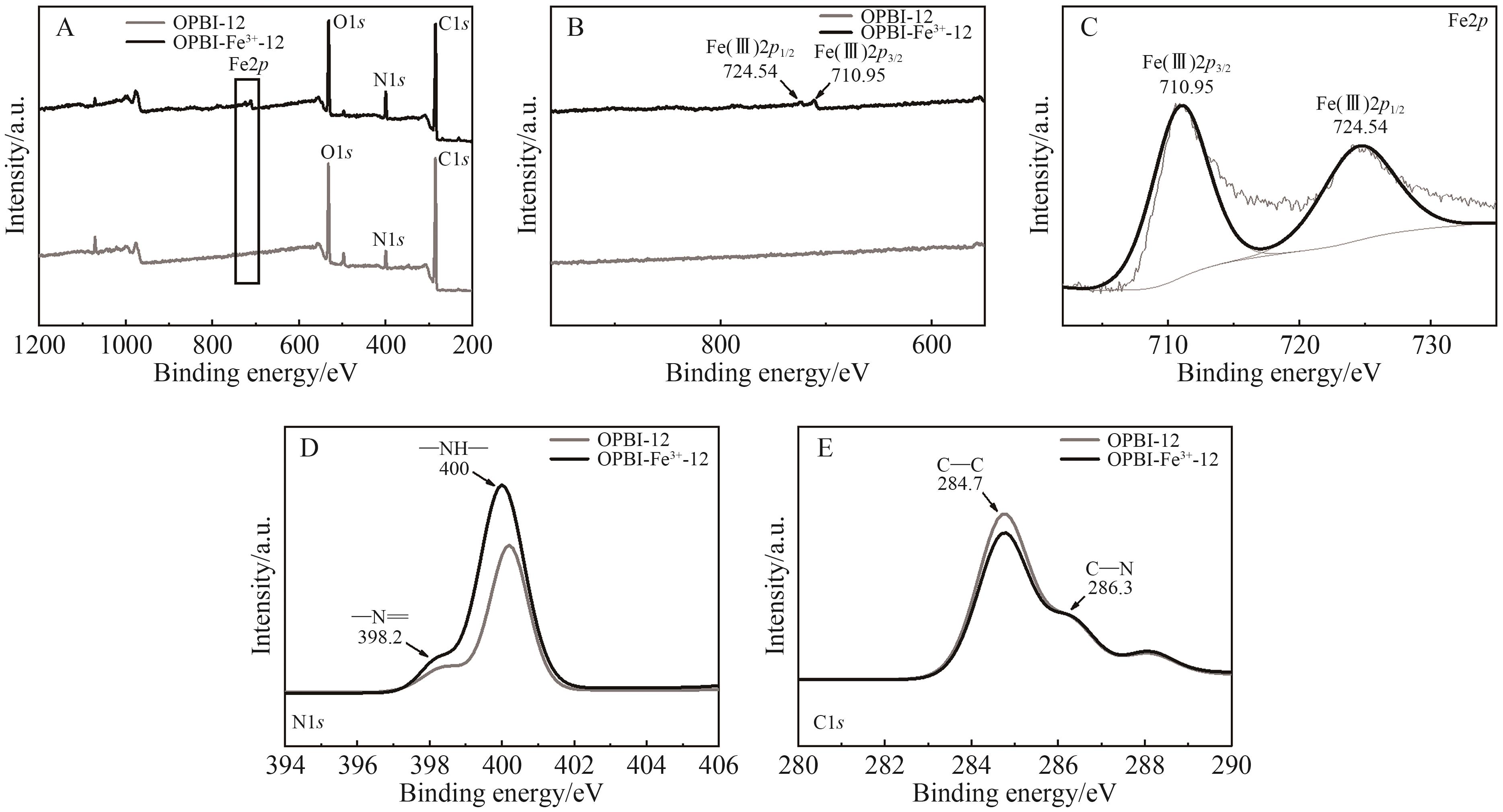
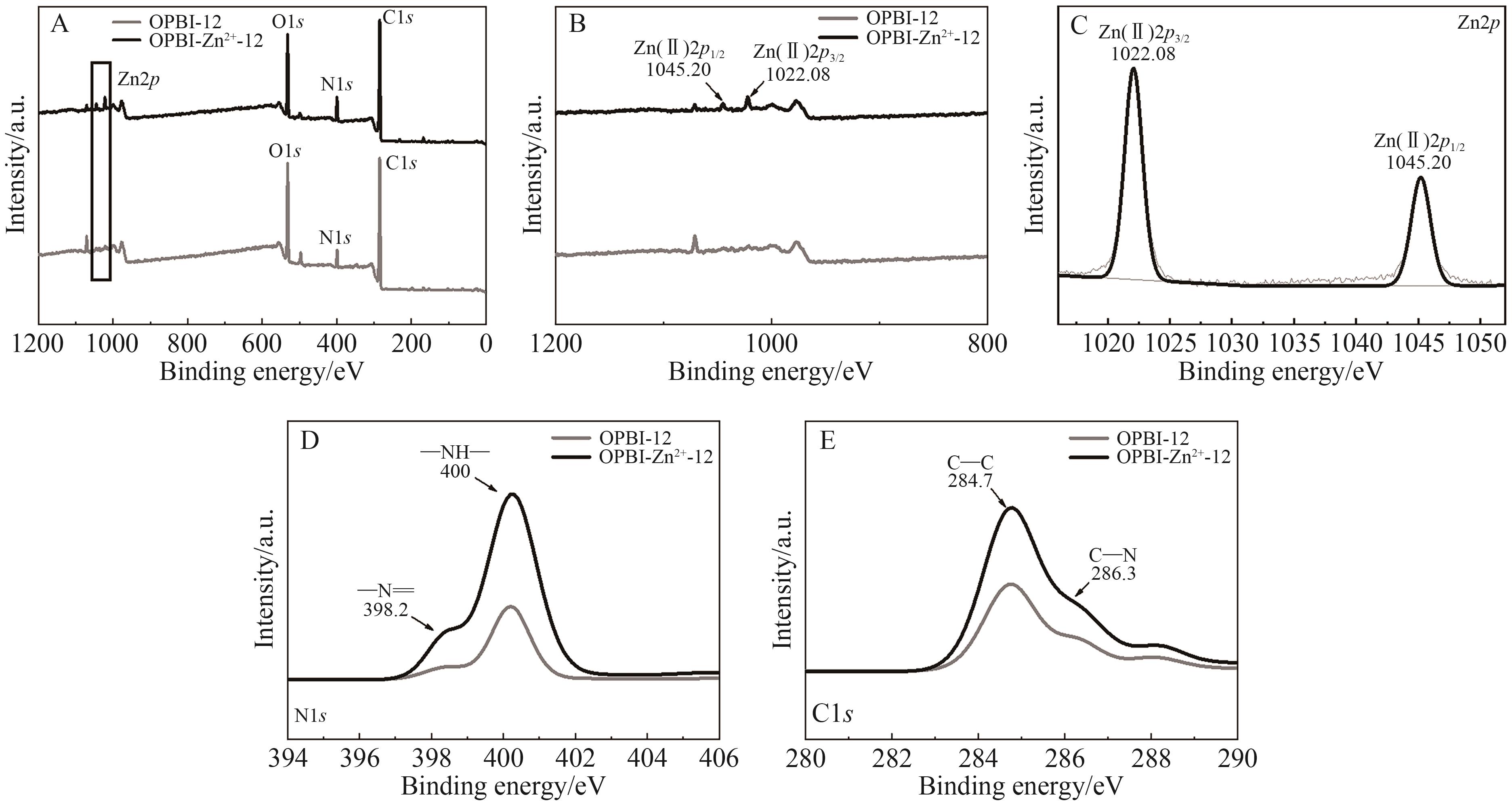
随着我国工业的不断的发展,产生了越来越多的有机废液。 因此,从有机溶液中分离溶质并回收溶剂引起人们的广泛关注。 相比传统的分离技术,纳滤因具有操作条件温和、能耗小等优点逐渐被应用于有机溶液中,但开发出能在有机溶剂中稳定存在的并具有良好分离性能的纳滤膜仍是一大挑战。 本研究选择FeCl3·6H2O和ZnSO4·7H2O溶液作为交联剂与聚苯并咪唑(Polybenzimidazole, OPBI)膜发生交联反应,制备了高性能的改性OPBI耐溶剂纳滤膜。 对改性前后纳滤膜的形貌结构以及分离性能进行探究。 结果表明,与原始OPBI膜相比,所有改性膜的分离性能均有显著提高。 交联后,OPBI膜在异丙醇(IPA)中对玫瑰红(RB)的截留率均达到了99%以上。 交联膜在药物和食品方面也有良好的应用前景,对IPA中的四环素和二氯甲烷(DCM)中的L-α-卵磷脂均具有优异的截留率。 此外,交联膜在有机溶剂中也具有优异的稳定性,在N,N-二甲基甲酰胺(DMF)、N,N-二甲基乙酰胺(DMAc)等强极性溶剂中也表现出良好的稳定性。
中图分类号:
引用本文
焦丽娜, 王晨钰, 江娱婷, 贺苗苗, 陈冬菊. 改性聚苯并咪唑耐溶剂纳滤膜的制备及性能[J]. 应用化学, 2026, 43(2): 259-274.
Li-Na JIAO, Chen-Yu WANG, Yu-Ting JIANG, Miao-Miao HE, Dong-Ju CHEN. Preparation and Properties of Modified Polybenzimidazole Solvent Resistant Nanofiltration Membranes[J]. Chinese Journal of Applied Chemistry, 2026, 43(2): 259-274.
| w(polymer)/% | 12% | 15% | 17.5% |
|---|---|---|---|
| Pristine membranes | OPBI-12 | OPBI-15 | OPBI-17.5 |
| FeCl3·6H2O crosslinked membranes | OPBI-Fe3+-12 | OPBI-Fe3+-15 | OPBI-Fe3+-17.5 |
| ZnSO4·7H2O crosslinked membranes | OPBI-Zn2+-12 | OPBI-Zn2+-15 | OPBI-Zn2+-17.5 |
表1 原始膜和交联膜的名称
Table 1 Naming of original and crosslinked membranes
| w(polymer)/% | 12% | 15% | 17.5% |
|---|---|---|---|
| Pristine membranes | OPBI-12 | OPBI-15 | OPBI-17.5 |
| FeCl3·6H2O crosslinked membranes | OPBI-Fe3+-12 | OPBI-Fe3+-15 | OPBI-Fe3+-17.5 |
| ZnSO4·7H2O crosslinked membranes | OPBI-Zn2+-12 | OPBI-Zn2+-15 | OPBI-Zn2+-17.5 |
| Solvent | Relative molecular mass/(g·mol-1) | Molar volume/(m3·mol-1) | Hansen solubility parameters/(MPa0.5) | η/(mPa·s) | ρ/(g·cm-3) |
|---|---|---|---|---|---|
| n-Heptane | 100.2 | 144.0 | 15.3 | 0.400 | 0.683 |
| n-Hexane | 86.2 | 127.5 | 14.9 | 0.326 | 0.659 |
| DCM | 84.9 | 67.8 | 20.3 | 0.430 | 1.325 |
| EA | 88.1 | 98.0 | 18.1 | 0.450 | 0.902 |
| MIBK | 100.1 | 125.0 | 16 | 0.542 | 0.796 |
| THF | 72.1 | 79.7 | 19.4 | 0.550 | 0.890 |
| IPA | 60.1 | 75.9 | 23.6 | 1.830 | 0.785 |
| NMP | 99.1 | 98.8 | 23.0 | 1.650 | 1.020 |
| Acetone | 58.1 | 75.1 | 19.9 | 0.290 | 0.791 |
| DMF | 73.1 | 77.0 | 24.9 | 0.802 | 0.947 |
| DMAc | 87.1 | 96.2 | 22.8 | 0.920 | 0.940 |
表2 有机溶剂的性质
Table 2 Properties of solvents
| Solvent | Relative molecular mass/(g·mol-1) | Molar volume/(m3·mol-1) | Hansen solubility parameters/(MPa0.5) | η/(mPa·s) | ρ/(g·cm-3) |
|---|---|---|---|---|---|
| n-Heptane | 100.2 | 144.0 | 15.3 | 0.400 | 0.683 |
| n-Hexane | 86.2 | 127.5 | 14.9 | 0.326 | 0.659 |
| DCM | 84.9 | 67.8 | 20.3 | 0.430 | 1.325 |
| EA | 88.1 | 98.0 | 18.1 | 0.450 | 0.902 |
| MIBK | 100.1 | 125.0 | 16 | 0.542 | 0.796 |
| THF | 72.1 | 79.7 | 19.4 | 0.550 | 0.890 |
| IPA | 60.1 | 75.9 | 23.6 | 1.830 | 0.785 |
| NMP | 99.1 | 98.8 | 23.0 | 1.650 | 1.020 |
| Acetone | 58.1 | 75.1 | 19.9 | 0.290 | 0.791 |
| DMF | 73.1 | 77.0 | 24.9 | 0.802 | 0.947 |
| DMAc | 87.1 | 96.2 | 22.8 | 0.920 | 0.940 |
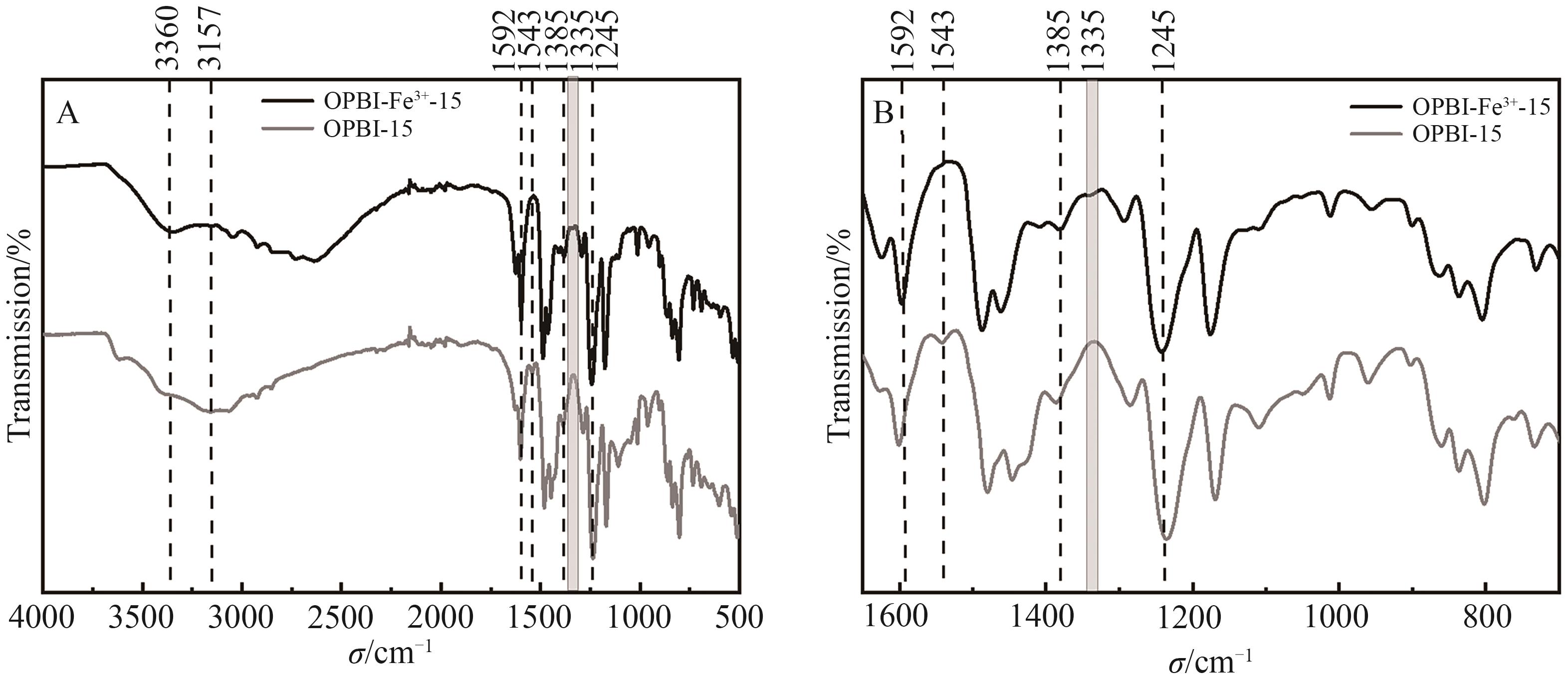
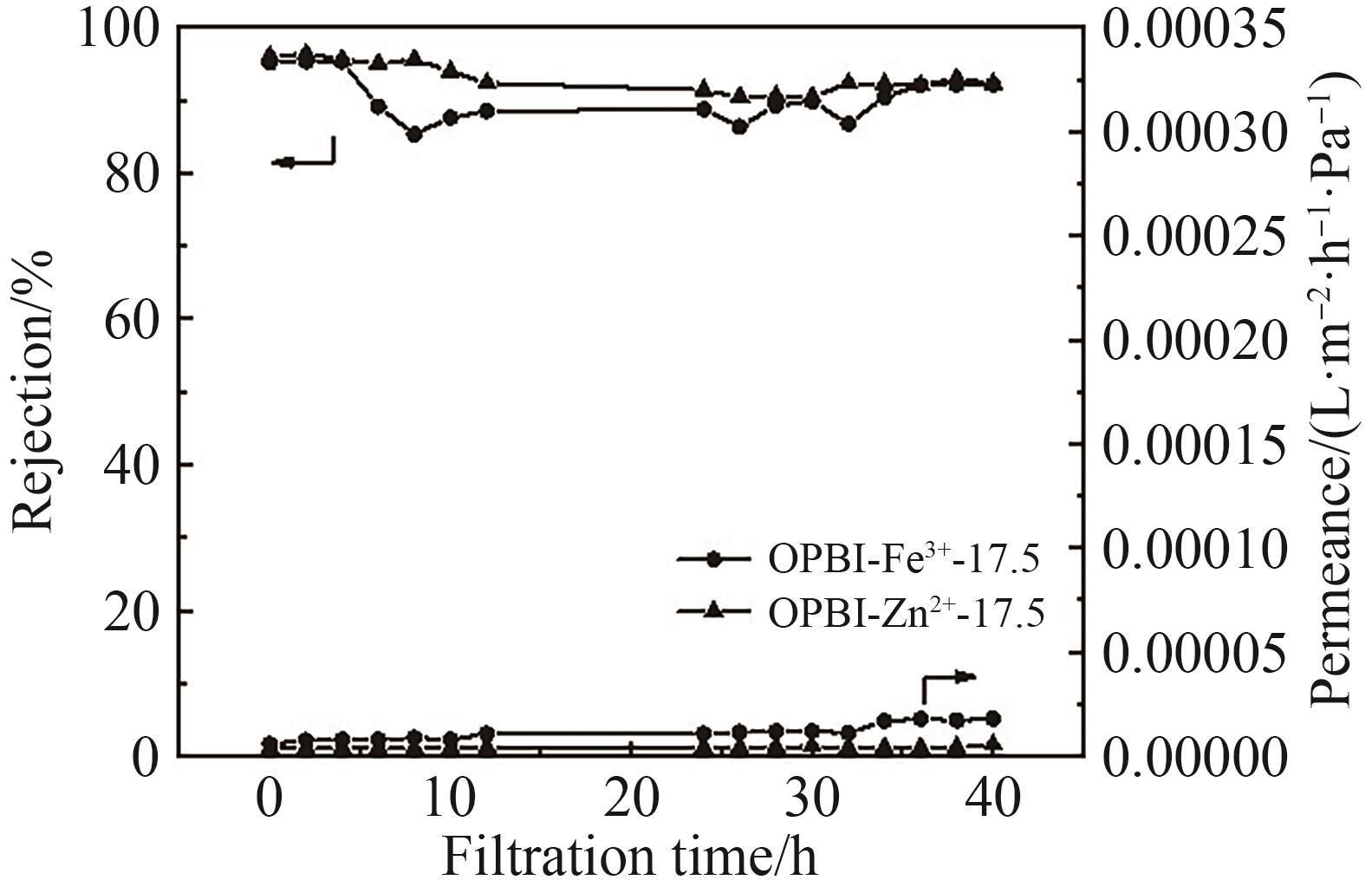
图1 (A) FeCl3·6H2O交联15% OPBI耐溶剂纳滤膜在500~4000 cm-1区间的FT-IR谱图; (B)为FeCl3·6H2O交联15% OPBI耐溶剂纳滤膜在700~1650 cm-1区间的FT-IR谱图
Fig.1 (A) FT-IR spectra of FeCl3·6H2O crosslinked 15% OPBI membrane in range of 500~4000 cm-1; (B) 700~1650 cm-1 FT-IR spectra of FeCl3·6H2O crosslinked 15% OPBI membrane in range of 700~1650 cm-1
图2 (A) ZnSO4·7H2O交联15% OPBI耐溶剂纳滤膜在500~4000 cm-1区间的FT-IR谱图; (B)ZnSO4·7H2O交联15% OPBI耐溶剂纳滤膜在200~1800 cm-1区间的FT-IR谱图
Fig.2 (A) FT-IR spectra of ZnSO4·7H2O crosslinked 15% OPBI membrane in range of 500~4000 cm-1; (B) FT-IR spectra of ZnSO4·7H2O crosslinked 15% OPBI membrane in range of 200~1800 cm-1
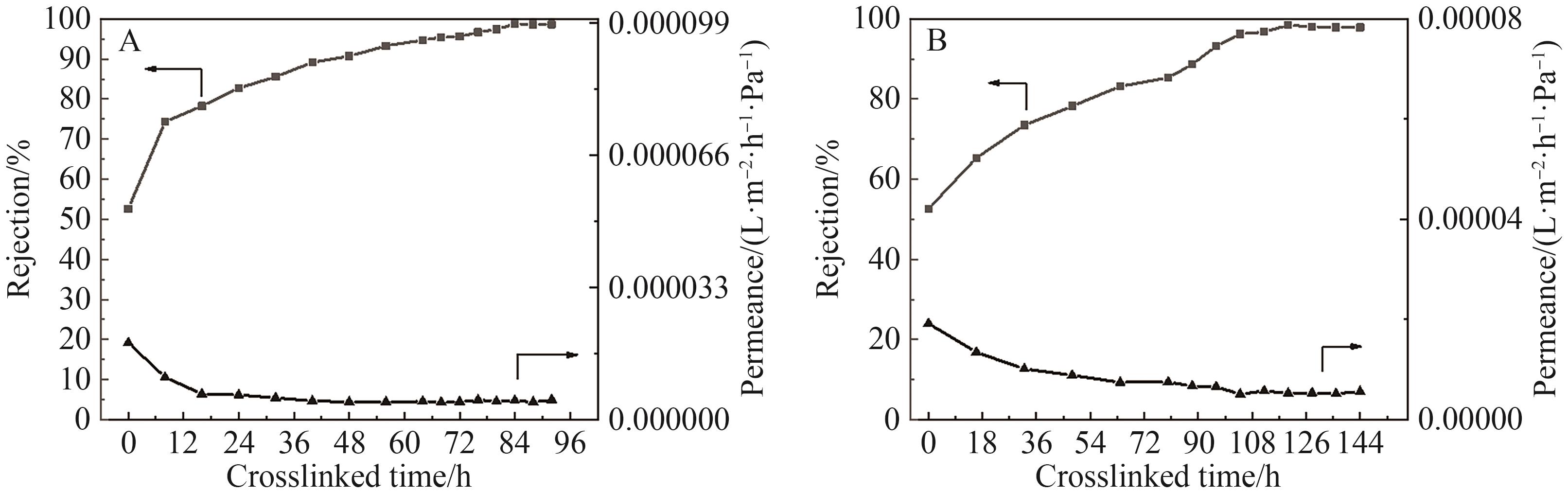
图8 不同交联时间对OPBI-Fe3+-15(A)和OPBI-Zn2+-15(B)膜分离性能的影响
Fig.8 Effect of different crosslinking times on the separation performance of OPBI-Fe3+-15 (A) and OPBI-Zn2+-15 (B) membrane
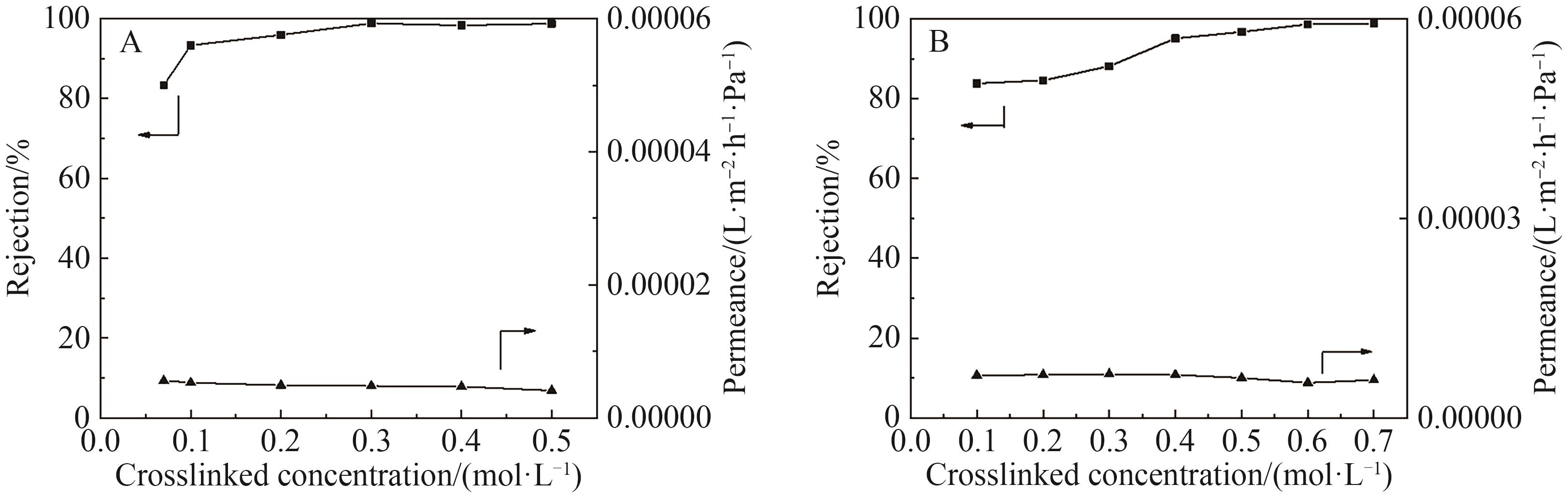
图9 不同交联剂浓度对OPBI-Fe3+ -15(A)和OPBI-Zn2+-15(B)膜分离性能的影响
Fig.9 The effect of different crosslinking concentrations on the separation performance of OPBI-Fe3+-15 (A) and OPBI-Zn2+-15 (B) membrane
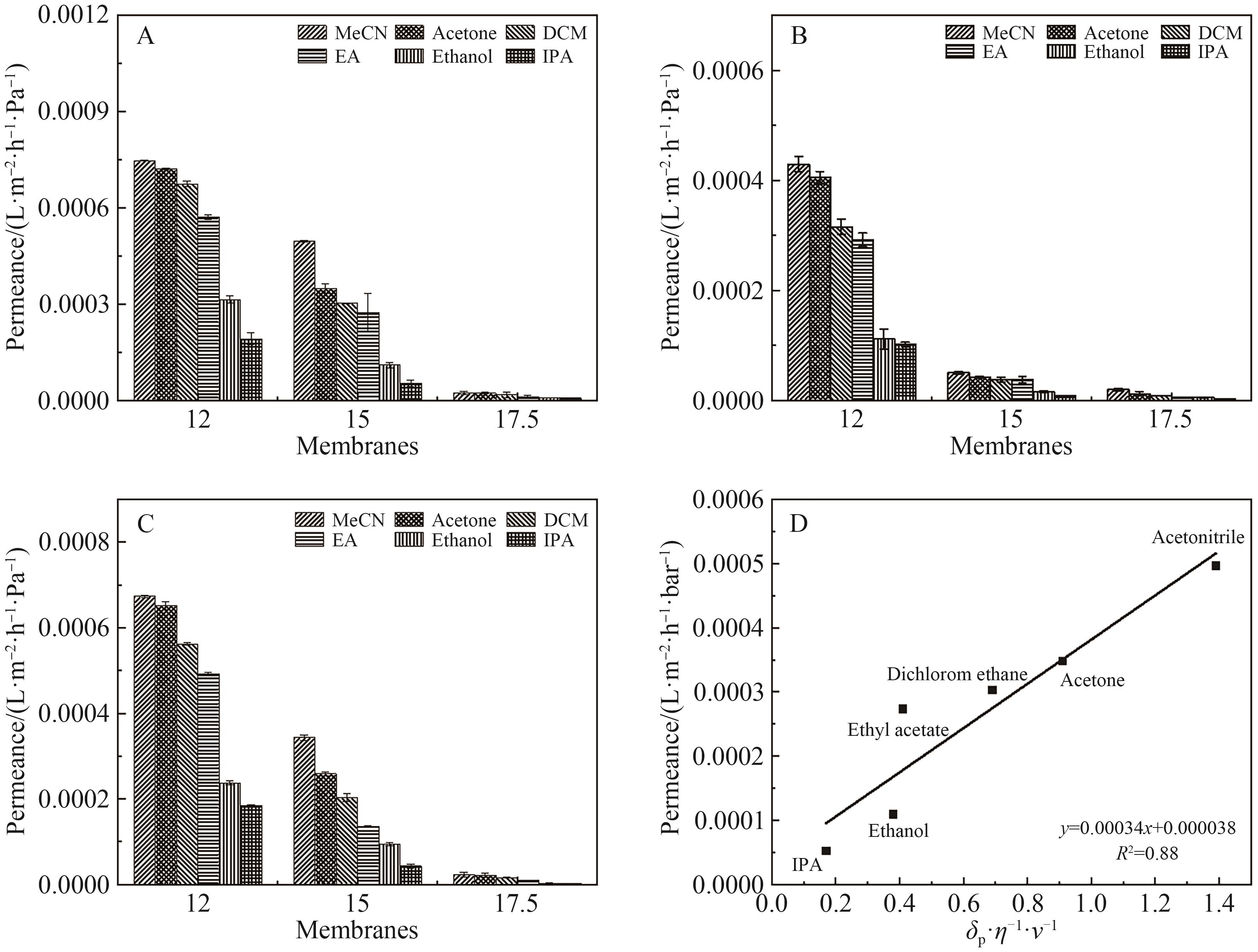
图10 (A)原始OPBI膜的纯溶剂渗透通量; (B) FeCl3·6H2O交联OPBI纳滤膜的纯溶剂渗透通量; (C) ZnSO4·7H2O交联OPBI纳滤膜的纯溶剂渗透通量; (D) OPBI-15膜对溶剂的渗透通量与δp·η-1·v-1的关系
Fig.10 (A) The pure solvent permeation flux of the original OPBI membrane; (B) The pure solvent permeation flux of FeCl3·6H2O crosslinked OPBI membrane; (C) The pure solvent permeation flux of ZnSO4·7H2O crosslinked OPBI membrane; (D) The relationship between the solvent permeation flux of the OPBI-15 membrane and δp·η-1·v-1
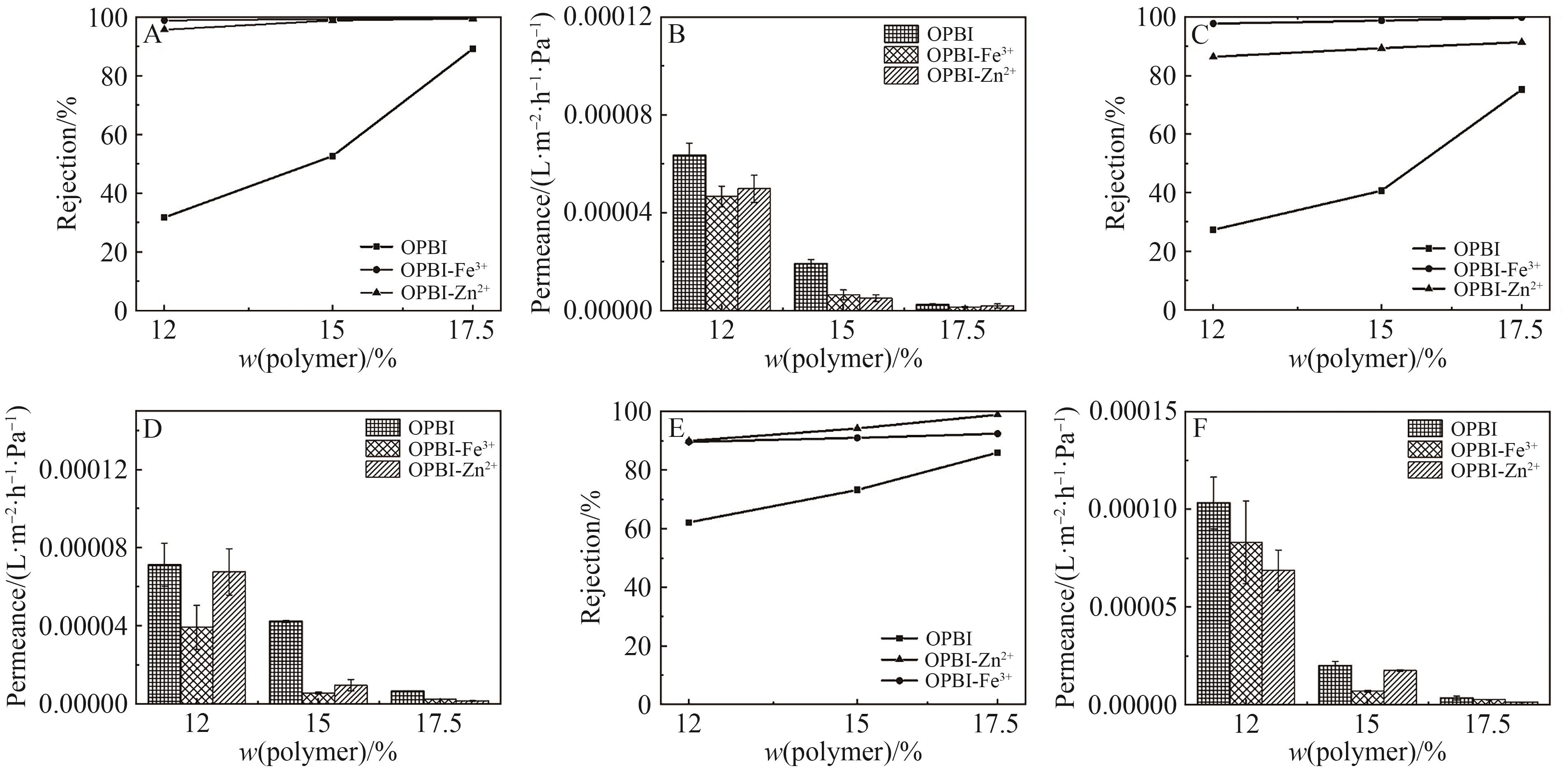
图11 (A)原始和交联OPBI膜对IPA中RB的截留率; (B)原始膜和交联OPBI膜对异丙醇的渗透通量; (C)原始膜和交联膜对IPA中BTB的截留率; (D)原始膜和交联膜对IPA的渗透通量; (E)原始膜和交联膜对IPA中MB的截留率; (F)原始膜和交联膜对IPA的渗透通量
Fig.11 (A) Retention rates of RB in IPA by original and cross-linked OPBI membranes; (B) Permeation flux of RB in IPA by original and cross-linked OPBI membranes; (C) Retention rates of BTB in isopropanol by original and cross-linked OPBI membranes; (D) Permeation flux of BTB in isopropanol by original and cross-linked OPBI membranes; (E) Retention rates of MB in IPA by original and cross-linked OPBI membranes; (F) Permeation flux of MB in IPA by original and cross-linked OPBI membranes
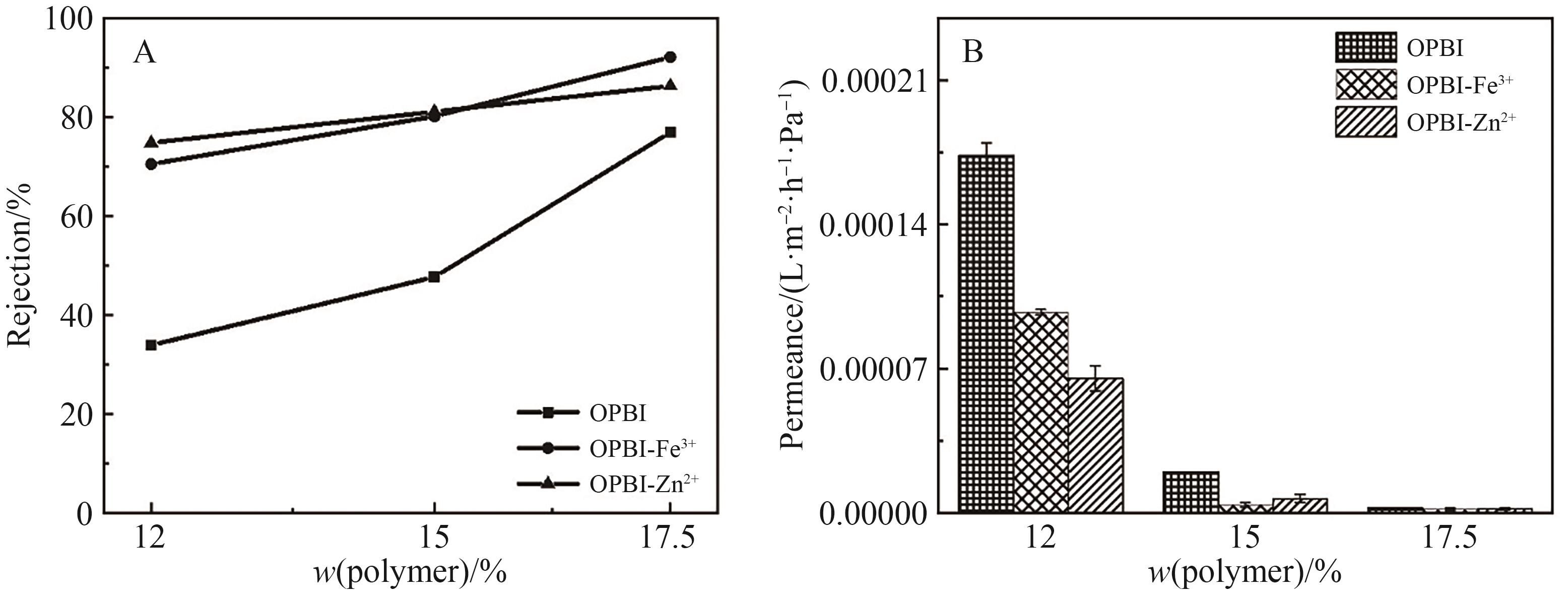
图12 (A)原始膜和交联膜对IPA中四环素的截留率; (B)原始膜和交联膜对IPA的渗透通量
Fig.12 (A) Retention rates of tetracycline in IPA by original and cross-linked OPBI membranes; (B) Permeance of tetracycline in IPA by originaland cross-linked OPBI membranes
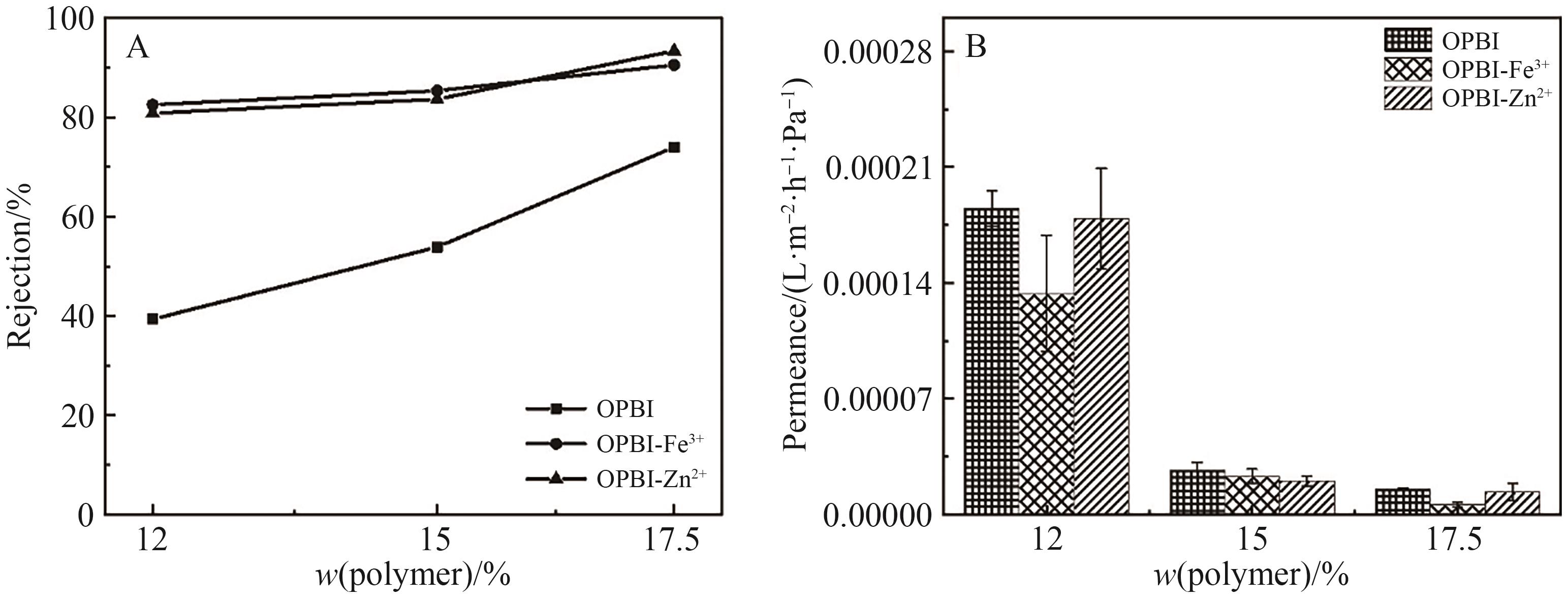
图13 (A)原始膜和交联膜对DCM中L-α-卵磷脂的截留率; (B)原始膜和交联膜对DCM的渗透通量
Fig.13 (A) Retention rates of L-α-lecithin in DCM by original and cross-linked OPBI membranes; (B) Permeance of L-α-lecithin in DCM by originaland cross-linked OPBI membranes
| Membrane | n-Heptane | DCM | EA | n-Hexane | IPA | Ethanol | Acetone | MIBK | MeCN |
|---|---|---|---|---|---|---|---|---|---|
| OPBI-12 | — | — | — | — | — | — | — | — | — |
| OPBI-Fe3+-12 | — | — | — | — | — | — | — | — | — |
| OPBI-Zn2+-12 | — | — | — | — | — | — | — | — | — |
| OPBI-15 | — | — | — | — | — | — | — | — | — |
| OPBI-Fe3+-15 | — | — | — | — | — | — | — | — | — |
| OPBI-Zn2+-15 | — | — | — | — | — | — | — | — | — |
| OPBI-17.5 | — | — | — | — | — | — | — | — | — |
| OPBI-Fe3+-17.5 | — | — | — | — | — | — | — | — | — |
| OPBI-Zn2+-17.5 | — | — | — | — | — | — | — | — | — |
表3 原始OPBI纳滤膜和交联OPBI纳滤膜在有机溶剂中的稳定性
Table 3 Stability of original and cross-linked OPBI nanofiltration membranes in organic solvents
| Membrane | n-Heptane | DCM | EA | n-Hexane | IPA | Ethanol | Acetone | MIBK | MeCN |
|---|---|---|---|---|---|---|---|---|---|
| OPBI-12 | — | — | — | — | — | — | — | — | — |
| OPBI-Fe3+-12 | — | — | — | — | — | — | — | — | — |
| OPBI-Zn2+-12 | — | — | — | — | — | — | — | — | — |
| OPBI-15 | — | — | — | — | — | — | — | — | — |
| OPBI-Fe3+-15 | — | — | — | — | — | — | — | — | — |
| OPBI-Zn2+-15 | — | — | — | — | — | — | — | — | — |
| OPBI-17.5 | — | — | — | — | — | — | — | — | — |
| OPBI-Fe3+-17.5 | — | — | — | — | — | — | — | — | — |
| OPBI-Zn2+-17.5 | — | — | — | — | — | — | — | — | — |
| Membrane | THF | DMF | DMAc | NMP |
|---|---|---|---|---|
| OPBI-12 | + | + | + | + |
| OPBI-Fe3+-12 | — | -4.3% | -3.7% | -23.6% |
| OPBI-Zn2+-12 | — | -3.4% | -2.0% | -3.8% |
| OPBI-15 | -4.5% | + | + | + |
| OPBI-Fe3+-15 | — | -1.2% | -2.3% | -16.3% |
| OPBI-Zn2+-15 | — | — | -1.2% | -3.1% |
| OPBI-17.5 | -2.2% | + | + | + |
| OPBI-Fe3+-17.5 | — | — | — | -7.8% |
| OPBI-Zn2+-17.5 | — | — | — | -2.6% |
表4 原始OPBI纳滤膜和交联OPBI纳滤膜在极性非质子溶剂中的稳定性
Table 4 Stability of original and cross-linked OPBI nanofiltration membranes in polar organic solvents
| Membrane | THF | DMF | DMAc | NMP |
|---|---|---|---|---|
| OPBI-12 | + | + | + | + |
| OPBI-Fe3+-12 | — | -4.3% | -3.7% | -23.6% |
| OPBI-Zn2+-12 | — | -3.4% | -2.0% | -3.8% |
| OPBI-15 | -4.5% | + | + | + |
| OPBI-Fe3+-15 | — | -1.2% | -2.3% | -16.3% |
| OPBI-Zn2+-15 | — | — | -1.2% | -3.1% |
| OPBI-17.5 | -2.2% | + | + | + |
| OPBI-Fe3+-17.5 | — | — | — | -7.8% |
| OPBI-Zn2+-17.5 | — | — | — | -2.6% |
| [1] | GANIYU S O, HULLEBUSCH V E D, MARC C, et al. Coupling of membrane filtration and advanced oxidation processes for removal of pharmaceutical residues: a critical review[J]. Sep Purif Technol, 2015, 156: 891-914. |
| [2] | 方彦彦, 涂丛慧, 王晓琳. 静电位阻模型在纳滤膜跨膜电位解析中的应用[J]. 高等学校化学学报, 2010, 31(4): 782-789. |
| FANG Y Y, TU C H, WANG X L. Application of the electrostatic and steric-hindrance model to analysis of transmembrane potential across nanofiltration membranes[J]. Chem J Chin Univ, 2010, 31(4): 782-789. | |
| [3] | RAVANCHI M T, KAGHAZCHI T, KARGARI A. Application of membrane separation processes in petrochemical industry: a review[J]. Desalination, 2009, 235: 199-244. |
| [4] | MARCHETTI P, SOLOMON M F J, SZEKELY G, et al. Molecular separation with organic solvent nanofiltration: a critical review[J]. Chem Rev, 2014, 114: 10735-10806. |
| [5] | XIAO H F, CHU C H, XU W T, et al. Amphibian inspired amino acid ionic liquid functionalized nanofiltration membranes with high water permeability and ion selectivity for pigment wastewater treatment[J]. J Membr Sci, 2019, 586: 44-52. |
| [6] | CONSTABLE D J, JIMENEZ-GONZALEZ C, HENDERSON R K. Perspective on solvent use in the pharmaceutical industry[J]. Org Process Res Dev, 2007, 11: 133-137. |
| [7] | SHI G M, FENG Y, LI B, et al. Recent progress of organic solvent nanofiltration membranes[J]. Prog Polym Sci, 2021, 123: 101470. |
| [8] | ASADI T A, FENG Y, WEBER M, et al. 110th anniversary: selection of cross-linkers and cross-linking procedures for the fabrication of solvent-resistant nanofiltration membranes: a review[J]. Ind Eng Chem Res, 2019, 58: 10678-10691. |
| [9] | XIAO H, FENG Y, GOUNDRY F R W, et al. Organic solvent nanofiltration in pharmaceutical applications[J]. Org Process Res Dev, 2024, 28: 891-923. |
| [10] | SZEKELY G, JIMENEZ-SOLOMON M F, MARCHETTI P, et al. Sustainability assessment of organic solvent nanofiltration: from fabrication to application[J]. Green Chem, 2014, 16: 4440-4473. |
| [11] | BUONMENNA M G, BAE J. Organic solvent nanofiltration in pharmaceutical industry[J]. Sep Purif Rev, 2015, 44: 157-182. |
| [12] | CHENG X Q, ZHANG Y L, WANG Z X, et al. Recent advances in polymeric solvent-resistant nanofiltration membranes[J]. Adv Polym Technol, 2014, 33: 1-24. |
| [13] | 张颖, 胡雁鸣, 李战胜, 等. 聚二苯基乙炔耐溶剂纳滤膜的制备与性能[J]. 应用化学, 2017, 34(4): 443-448. |
| ZHANG Y, HU Y M, LI Z S, et al. Preparation and performance of solvent resistance poly(diphenylacetylene) nanofiltration membranes[J]. Chin J Appl Chem, 2017, 34(4): 443-448. | |
| [14] | VALTCHEVA I B, MARCHETTI P, LIVINGSTON A G. Crosslinked polybenzimidazole membranes for organic solvent nanofiltration (OSN): analysis of cross-linking reaction mechanism and effects of reaction parameter[J]. J Membr Sci, 2015, 493: 568-579. |
| [15] | FARAHANI M H D A, CHUNG T S. A novel crosslinking technique towards the fabrication of high-flux polybenzimidazole (PBI) membranes for organic solvent nanofiltration (OSN)[J]. Sep Purif Technol, 2019, 209: 182-192. |
| [16] | FEI F, CSERI L, SZEKELY G, et al. Robust covalently cross-linked polybenzimidazole graphene oxide membranes for high flux organic solvent nanofiltration[J]. ACS Appl Mater Interfaces, 2018, 10: 16140-16147. |
| [17] | KIM S D, WON G Y, SHAH A A, et al. Reinforcing the polybenzimidazole membrane surface by an ultrathin co-cross-linked polydopamine layer for organic solvent nanofiltration applications[J]. J Membr Sci, 2021, 636: 119587. |
| [18] | LEE J, YANG H, PARK G, et al. Highly stable epoxy-crosslinked polybenzimidazole membranes for organic solvent nanofiltration under strongly basic conditions[J]. J Membr Sci, 2022, 661: 120951. |
| [19] | YANG H, HONG Y, LEE Y, et al. Advancing polybenzimidazole organic solvent nanofiltration membranes with oxygen-rich crosslinking for rapid polar solvent flux[J]. Sep Purif Technol, 2025, 356: 129976. |
| [20] | SHIN S J, PARK Y I, PARK H, et al. Solvent-resistant crosslinked polybenzimidazole membrane for use in enhanced molecular separation[J]. J Membr Sci, 2024, 695: 122463. |
| [21] | CHEN D J, YAN C, LI X N, et al. A highly stable PBI solvent resistant nanofiltration membrane prepared via versatile and simple crosslinking process[J]. Sep Purif Technol, 2019, 224: 15-22. |
| [22] | CHEN D J, YU S S, YANG M, et al. Solvent resistant nanofiltration membranes based on crosslinked polybenzimidazole[J]. RSC Adv, 2016, 6(21): 16925-16932. |
| [23] | 尚尔慧, 陈慧龄, 贺苗苗, 等. 具有图灵结构的聚苯并咪唑耐溶剂纳滤膜的制备及性能[J]. 高等学校化学学报, 2023, 44(6): 211-220. |
| SHANG E H, CHEN H L, HE M M, et al. Preparation and performance of turning structured polybenzimidazole solvent-resistant nanofiltration membrane [J]. Chem J Chin Univ, 2023, 44(6): 211-220. | |
| [24] | YUAN F, YANG Y, WANG R, et al. Poly(vinylidene fluoride) grafted polystyrene (PVDF-g-PS) membrane based on in situ polymerization for solvent resistant nanofiltration[J]. RSC Adv, 2017, 7(53): 33201-33207. |
| [25] | CHEN D J, LIU X, LI D D, et al. Highly stable polysulfone solvent resistant nanofiltration membranes with internal cross-linking networks[J]. RSC Adv, 2016, 6(35): 29570-29575. |
| [26] | LIU T M, HUANG H, WANG Y, et al. Super strong and tough polybenzimidazole metal ions coordination networks: reinforcing mechanism recyclability and anti-counterfeiting applications[J]. Macromol Rapid Commun, 2022, 43(3): 2100643. |
| [27] | WU J, LIAO C Y, LI T Y, et al. Metal-coordinated polybenzimidazole membranes with preferential K+ transport[J]. Nat Commun, 2023, 14(1): 1149. |
| [28] | HARDIAN R, POGANY P, LEE Y M, et al. Molecular sieving using metal-polymer coordination membranes in organic media[J]. Mater Chem A, 2021, 9: 14400-14410. |
| [29] | ASADI T A, LUO L, CHUNG T S, et al. Performance enhancement in organic solvent nanofiltration by double crosslinking technique using sulfonated polyphenylsulfone (sPPSU) and polybenzimidazole (PBI)[J]. J Membr Sci, 2018, 551: 204-213. |
| [30] | HU J, HARDIAN R, GEDE M, et al. Reversible crosslinking of polybenzimidazole-based organic solvent nanofiltration membranes using difunctional organic acids: toward sustainable crosslinking approaches[J]. J Membr Sci, 2022, 648: 120383. |
| [31] | 吴宏超. 含吡啶交联聚苯并咪唑高温质子交换膜的制备与性能研究[D]. 兰州: 兰州大学, 2022. |
| WU H C. Preparation and properties of high temperature proton exchange membranes containing pyridine crosslinked polybenzimidazole[D]. Lanzhou: Lanzhou University, 2022. | |
| [32] | WANG J, LIU X, SUN Y, et al. Synthesis, crystal structures, thermal properties, and DNA-binding studies of transition metal complexes with imidazole ligands[J]. J Coord Chem, 2011, 64(9): 1554-1565. |
| [33] | WU H Q, ANG J M, KONG J H, et al. One-pot synthesis of polydopamine-Zn complex antifouling coatings on membranes for ultrafiltration under harsh conditions[J]. RSC Adv, 2016, 6(105): 103390-103398. |
| [34] | SHIN S J, PARK Y I, PARK H, et al. A facile crosslinking method for polybenzimidazole membranes toward enhanced organic solvent nanofiltration performance[J]. Sep Purif Technol, 2022, 299: 121783. |
| [35] | BESHAHWORED S S, HUANG Y H, ABDI Z G, et al. Polybenzimidazole (PBI) membranes cross-linked with various cross-linkers and impregnated with 4-sulfocalix [4]arene (SCA4) for organic solvent nanofiltration (OSN)[J]. J Membr Sci, 2022, 663: 121039. |
| [36] | DAHE G J, SINGH R P, DUDECK K W, et al. Influence of non-solvent chemistry on polybenzimidazole hollow fiber membrane preparation[J]. J Membr Sci, 2019, 577: 91-103. |
| [37] | AKBAR A T, NIECK E B. Covalent organic polymers for aqueous and organic solvent nanofiltration[J]. Sep Purif Technol, 2022, 298: 121589. |
| [38] | SAIZA C A, DARVISHMANESHB S, BUEKENHOUDT A, et al. Shortcut applications of the hansen solubility parameter for organic solvent nanofiltration[J]. J Membr Sci, 2018, 546: 120-170. |
| [39] | ZHANG Y Y, WANG L, LI L, et al. Insight into the influences of thermal crosslinking on the transition from polyacrylonitrile based ultrafiltration membrane to organic solvent nanofiltration membrane[J]. J Membr Sci, 2023, 679: 121694. |
| [40] | JI C H, XUE S M, LIN C W, et al. Ultrapermeable organic solvent nanofiltration membranes with precisely tailored support layers fabricated using thin-film liftoff[J]. ACS Appl Mater Interfaces, 2020, 12(27): 30796-30804. |
| [41] | NG L Y, MOHAMMAD A W, NG C Y. A review on nanofiltration membrane fabrication and modification using polyelectrolytes: effective ways to develop membrane selective barriers and rejection capability[J]. Adv Colloid Interface Sci, 2013,197/198: 85-107. |
| [42] | 杨炎福, 王齐齐, 张辉, 等. 以聚偏氟乙烯为基底的高渗透选择性聚酰胺/酯纳膜的制备与表征[J]. 高等学校化学学报, 2023, 44(4): 20220574. |
| YANG Y F, WANG Q Q, ZHANG H, et al. Preparation and characterization of a high perm-selectivity poly(amide/ester) nanofiltration membrane using a poly(vinylidene fluoride) substrate[J]. Chem Res Chin Univ, 2023, 44(4): 20220574. | |
| [43] | LUO X F, WANG Z G, WU S S, et al. Metal ion cross-linked nanoporous polymeric membranes with improved organic solvent resistance for molecular separation [J]. J Membr Sci,2021, 621: 119002. |
| [1] | 王昊, 王熙宇, 熊英, 崔俊硕. 萘基反应型石墨烯分散剂及其在增强聚乙烯醇复合薄膜中的应用[J]. 应用化学, 2024, 41(12): 1712-1720. |
| [2] | 胡生隆, 雷振隆, 陈德军. 超交联聚萘基微孔炭的制备及其电化学性能[J]. 应用化学, 2024, 41(12): 1742-1750. |
| [3] | 李靖威, 郑睿鹏, 杨明, 赵娟, 郑帅, 李娅珂, 戚云霞, 徐义全, 刘佰军. 辐照聚乙烯热熔带的性能及在电缆绝缘层修复中的应用[J]. 应用化学, 2024, 41(11): 1648-1655. |
| [4] | 刘嘉辉, 安佰超, 闫秋艳, 栾世方. 贻贝启发的抗菌骨粘合剂的制备与性能[J]. 应用化学, 2023, 40(9): 1258-1266. |
| [5] | 颜丽娟, 高添贺, 施冬健, 陈明清. 丁香酚/改性聚乙烯醇抗菌复合膜的制备与性能[J]. 应用化学, 2023, 40(4): 527-535. |
| [6] | 石碧茹, 武浩浩, 谢昊圃, 田新欣, 孙莹潞, 刘向东, 杨宇明. Diels-Alder型自修复聚氨酯胶粘剂的制备及其性能[J]. 应用化学, 2023, 40(2): 277-287. |
| [7] | 姚競, 戴明明. 基于乘用车轮胎胎面胶粉的再生橡胶的制备及性能[J]. 应用化学, 2023, 40(1): 52-58. |
| [8] | 陈炳刚, 刘三荣, 蒋子江, 于喜飞. 水性聚硅氧烷和聚乙烯醇复合物制备及其作为皮肤屏障材料的性能[J]. 应用化学, 2022, 39(8): 1224-1236. |
| [9] | 薛晨怡, 刘林佳, 王婷, 宫磊, 金龙, 韩建刚, 李胎花. 荧光法检测重金属铅离子的研究进展[J]. 应用化学, 2022, 39(7): 1039-1051. |
| [10] | 曹从军, 马含笑, 侯成敏, 丁小健, 管飙. 乙基纤维素磁性复合材料对溶液中铜离子的吸附性能[J]. 应用化学, 2022, 39(6): 969-979. |
| [11] | 冷冰冰, 朱春卉, 石埕荧, 王志鹏, 刘洋, 张宏岩, 许文革, 刘佰军. 含环三磷腈衍生物的辐照交联聚乙烯基复合材料的制备及阻燃性能[J]. 应用化学, 2022, 39(11): 1672-1679. |
| [12] | 王静雯, 吕雅文, 尚亚卓, 刘洪来. 大米淀粉膜的制备及其性能[J]. 应用化学, 2022, 39(11): 1693-1702. |
| [13] | 焦元昊, 崔洪燕, 张留伟, 曾爽, 王浩, 张明, 王静云, 陈麒先. 基于原位聚合技术构建细胞内微环境响应型DNA递送系统[J]. 应用化学, 2022, 39(10): 1510-1522. |
| [14] | 杨洋, 陈奇, 李治鹏, 谷卓然, 叶世贵, 佘雪. 用于压裂液改性的耐高温锆硼交联剂的制备及评价[J]. 应用化学, 2021, 38(4): 431-438. |
| [15] | 肖海梅, 蔡蕾, 张朝晖, 陈珊, 周姝, 符金利. 磁性氧化石墨烯/MIL-101(Cr)表面金属离子印迹聚合物制备及其对Cu(Ⅱ)和Pb(Ⅱ)选择性吸附[J]. 应用化学, 2020, 37(9): 1076-1086. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||