
应用化学 ›› 2024, Vol. 41 ›› Issue (6): 783-799.DOI: 10.19894/j.issn.1000-0518.230400
• 综合评述 • 上一篇
基于Cu(Ⅰ)配合物的OLEDs发光材料研究进展
王冠中1,4, 宋世平2, 彭红珍2, 张瑜3, 诸颖2, 陈静2( )
)
- 1.中国科学院上海应用物理研究所,上海 201800
2.上海大学理学院材料生物学研究所,上海 200444
3.中国科学院上海高等研究院,上海同步辐射光源,上海 201210
4.中国科学院大学,北京 100049
-
收稿日期:2023-12-29接受日期:2024-04-21出版日期:2024-06-01发布日期:2024-07-09 -
通讯作者:陈静 -
基金资助:国家重点研发计划(2023YFC3404200);国家自然科学基金(21974147)
Research Progress in OLEDs Emissive Materials Based on Cu(Ⅰ) Complexes
Guan-Zhong WANG1,4, Shi-Ping SONG2, Hong-Zhen PENG2, Yu ZHANG3, Ying ZHU2, Jing CHEN2( )
)
- 1.Division of Physical Biology,CAS Key Laboratory of Interfacial Physics and Technology,Shanghai Institute of Applied Physics,Chinese Academy of Sciences,Shanghai 201800,China
2.Institute of Materiobiology,Department of Chemistry,College of Sciences,Shanghai University,Shanghai 200444,China
3.Shanghai Synchrotron Radiation Facility,Shanghai Advanced Research Institute,Chinese Academy of Sciences,Shanghai 201210,China
4.University of Chinese Academy of Sciences,Beijing 100049,China
-
Received:2023-12-29Accepted:2024-04-21Published:2024-06-01Online:2024-07-09 -
Contact:Jing CHEN -
About author:chenjing929@shu.edu.cn
-
Supported by:the National Key R&D Program of China(2023YFC3404200);the National Natural Science Foundation of China(21974147)
摘要:
由于有机材料丰富的发光特性,有机发光二极管(OLEDs)在平面显示器和照明中的应用获得了广泛的关注。传统的有机发光二极管发光层材料主要为有机小分子或贵金属配合物,其中贵金属配合物,如铂(Ⅱ)和铱(Ⅲ),存在热稳定性较差、载流子传输能力有限、获取难度大、性能受限和材料价格高昂等问题,严重限制了其应用和发展。Cu(Ⅰ)配合物具有优异的发光特性、发射可调性和相对较低的成本,为开发金属配合物发光材料提供了新的途径。本文总结了基于Cu(Ⅰ)配合物的OLEDs发光材料的研究进展,探讨了改变配位原子类型和配体结构调节Cu(Ⅰ)配合物电致发光的策略,对Cu(Ⅰ)配合物OLEDs发光材料的发展前景予以展望。
中图分类号:
引用本文
王冠中, 宋世平, 彭红珍, 张瑜, 诸颖, 陈静. 基于Cu(Ⅰ)配合物的OLEDs发光材料研究进展[J]. 应用化学, 2024, 41(6): 783-799.
Guan-Zhong WANG, Shi-Ping SONG, Hong-Zhen PENG, Yu ZHANG, Ying ZHU, Jing CHEN. Research Progress in OLEDs Emissive Materials Based on Cu(Ⅰ) Complexes[J]. Chinese Journal of Applied Chemistry, 2024, 41(6): 783-799.
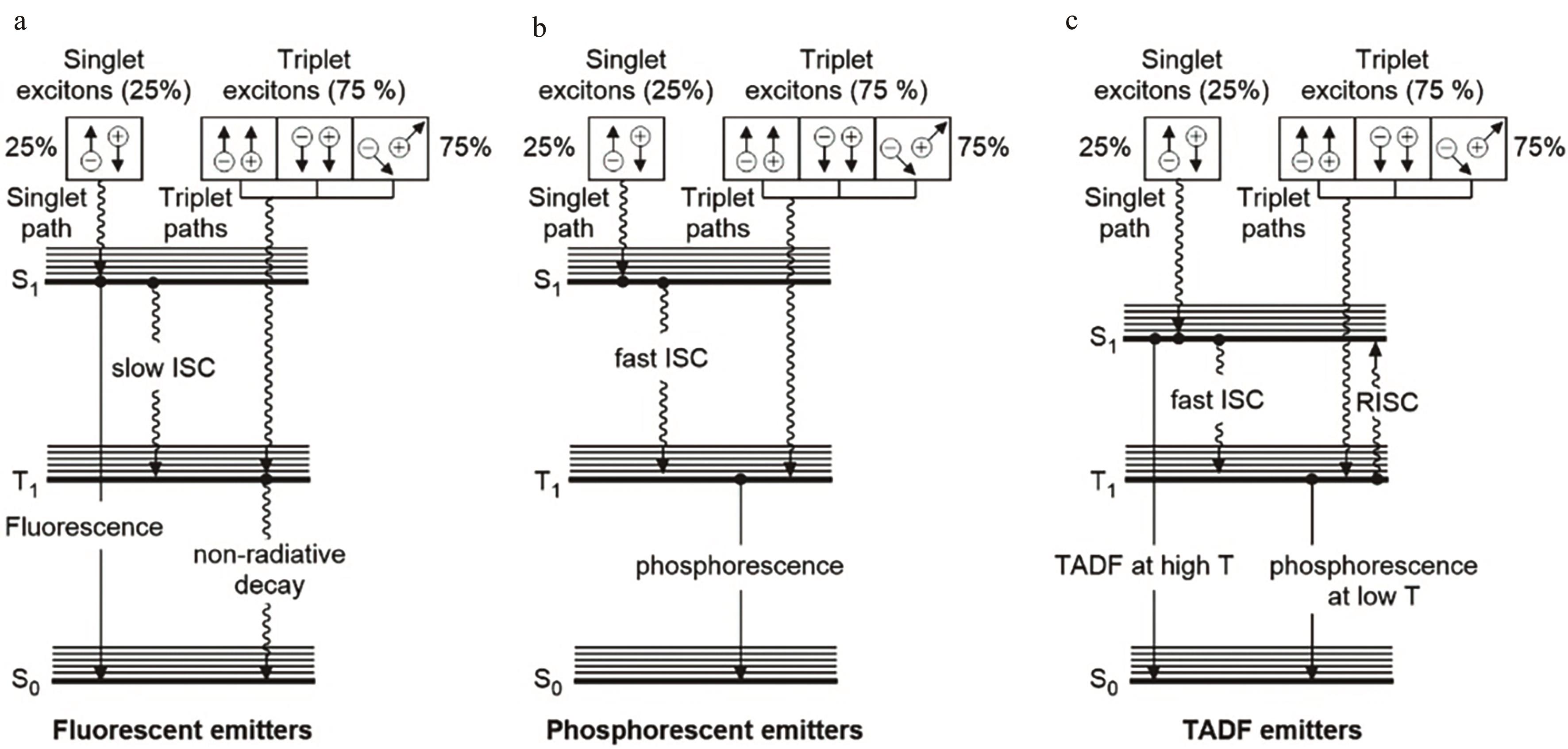
图1 荧光(a)、磷光(b)和热激活延迟荧光(c)的分子机制示意图[3]
Fig.1 Schematic diagram to illustrate the molecular mechanism of (a) fluorescent, (b) phosphorescent and (c) thermally activated delayed fluorescence[3]
| Ligands | ΔEST/eV | Emissions/nm a | PLQYs/% a | τ/μs a | EQE/% | Ref. |
|---|---|---|---|---|---|---|
| POP,ECAF | 0.09 | 550 | 22.4 | 5.7 | 14.81 | [ |
| POP,EHCAF | 0.09 | 549 | 18.5 | 5.7 | 11.17 | [ |
| POP,PCAF | 0.09 | 556 | 20.0 | 5.7 | 6.67 | [ |
| POP,DMAC-PyPI | 0.11 | 534 | 62 | 21.4 | - | [ |
| Xantphos,DMAC-PyPI | 0.08 | 533 | 71 | 24.1 | 5.91 | [ |
| POP,PXZ-PyPI | 0.07 | 565 | 48 | 5.8 | 7.96 | [ |
| Xantphos, PXZ-PyPI | 0.05 | 564 | 42 | 4.3 | - | [ |
| Cbz,BINAP | 0.08 | 564 | 22 | 5.0 (91), 46.0 (9) | - | [ |
| Cbz tBu,BINAP | 0.06 | 549 | 25 | 19.2 (74), 62.6 (26) | - | [ |
| POP,PNNA | 0.12 | 493 | 60.9 | 145 | 5.83 | [ |
| PNNA,Xantphos | 0.12 | 475 | 40.7 | 51 | 7.42 | [ |
| PPh3,pptz | 0.09 | 490 | 89.87 | 23.6 | - | [ |
表1 配位原子为N、P的单核Cu(Ⅰ)配合物的光学性质
Table 1 The optical properties of mononuclear Cu(Ⅰ) complexes with N and P coordination atoms
| Ligands | ΔEST/eV | Emissions/nm a | PLQYs/% a | τ/μs a | EQE/% | Ref. |
|---|---|---|---|---|---|---|
| POP,ECAF | 0.09 | 550 | 22.4 | 5.7 | 14.81 | [ |
| POP,EHCAF | 0.09 | 549 | 18.5 | 5.7 | 11.17 | [ |
| POP,PCAF | 0.09 | 556 | 20.0 | 5.7 | 6.67 | [ |
| POP,DMAC-PyPI | 0.11 | 534 | 62 | 21.4 | - | [ |
| Xantphos,DMAC-PyPI | 0.08 | 533 | 71 | 24.1 | 5.91 | [ |
| POP,PXZ-PyPI | 0.07 | 565 | 48 | 5.8 | 7.96 | [ |
| Xantphos, PXZ-PyPI | 0.05 | 564 | 42 | 4.3 | - | [ |
| Cbz,BINAP | 0.08 | 564 | 22 | 5.0 (91), 46.0 (9) | - | [ |
| Cbz tBu,BINAP | 0.06 | 549 | 25 | 19.2 (74), 62.6 (26) | - | [ |
| POP,PNNA | 0.12 | 493 | 60.9 | 145 | 5.83 | [ |
| PNNA,Xantphos | 0.12 | 475 | 40.7 | 51 | 7.42 | [ |
| PPh3,pptz | 0.09 | 490 | 89.87 | 23.6 | - | [ |
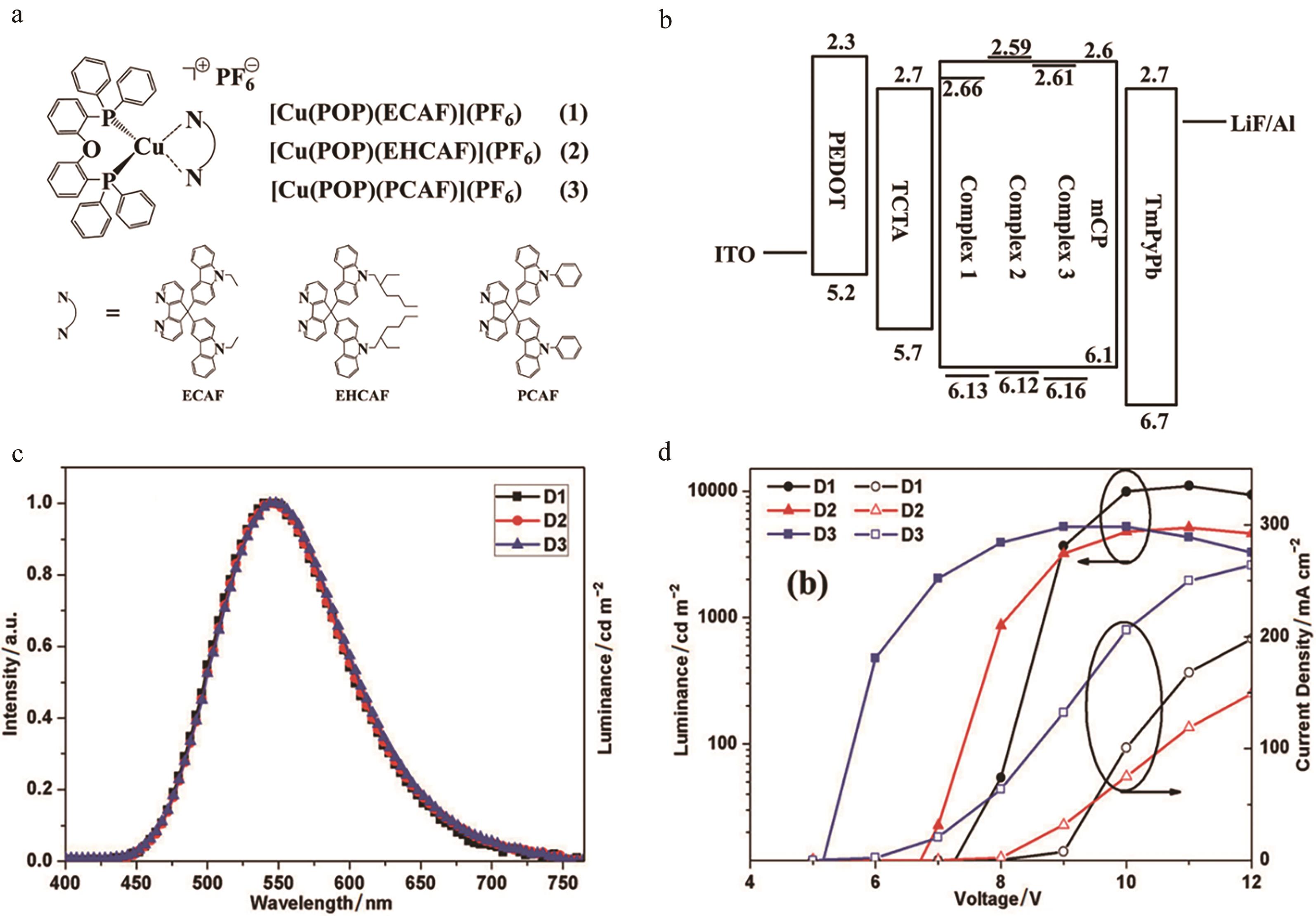
图2 (a)配合物结构图; (b) OLEDs器件结构图; (c) OLED器件发射光谱; (d) OLEDs器件的亮度和电流密度与电压关系[28]
Fig.2 (a) Structures of complexes; (b) Device architecture of OLEDs; (c) Normalized EL spectra of OLEDs; (d) Luminance and current density versus voltage for the OLEDs devices[28]
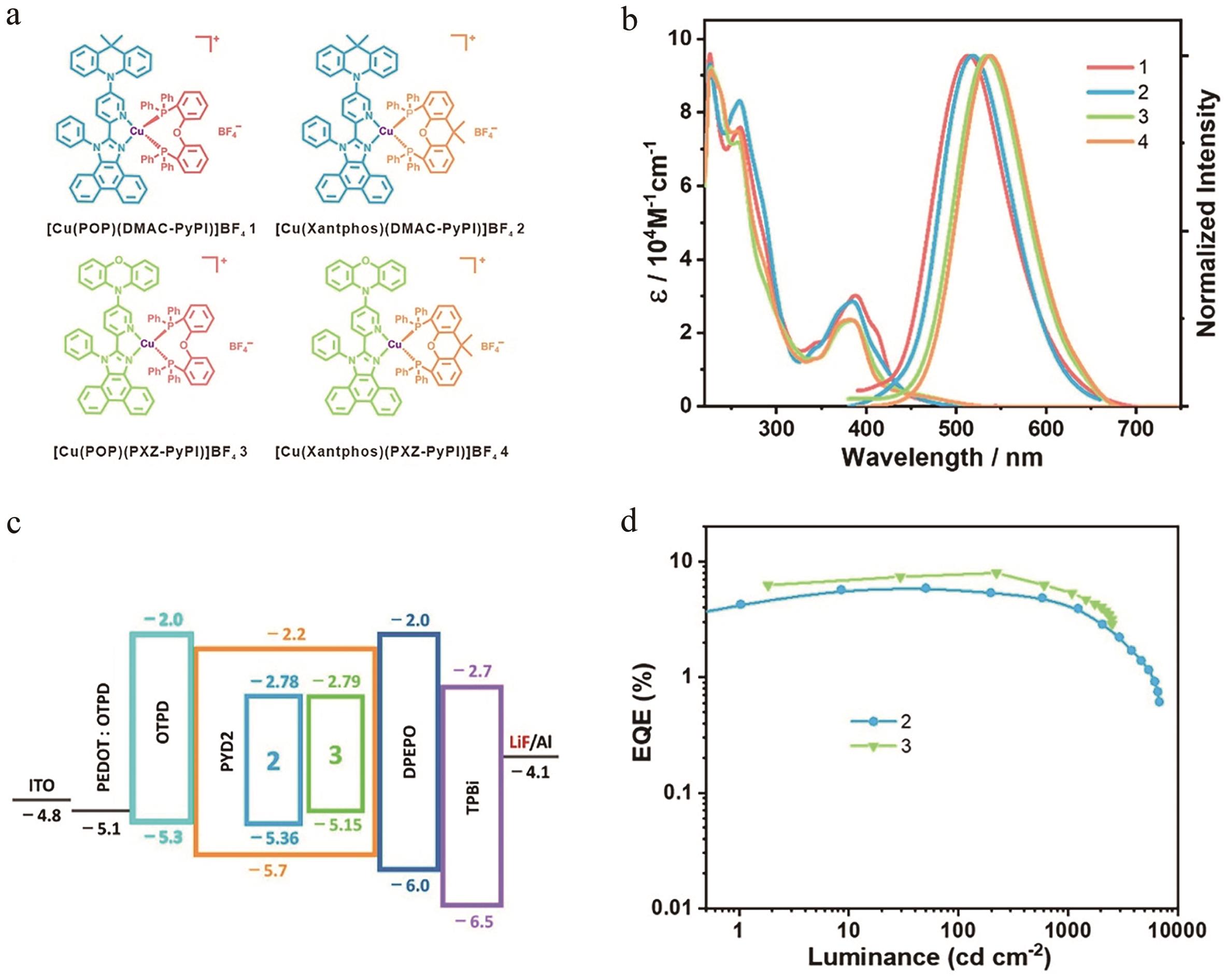
图3 (a)配合物结构图; (b)配合物在二氯甲烷溶液中的紫外-可见光吸收光谱及发射光谱; (c) OLEDs器件结构图; (d)配合物器件外量子效率与亮度关系[29]
Fig.3 (a) Structures of complexes; (b) UV-Vis absorption and emission spectra of complexes in CH2Cl2 solution; (c) Device architecture of OLEDs; (d) EQE-luminance characteristics of OLEDs[29]

图4 (a) Cu(CbzR)[(S/R)-BINAP]的结构图; (b)固体状态配合物变温辐射寿命及热激活延迟荧光过程; (c)四氢呋喃溶液中(S/R)-1及(S/R)-2圆二色谱(虚线)及圆偏振发光(实线)谱; (d) OLEDs器件结构及电致发光光谱[30]
Fig.4 (a) Structures of Cu(CbzR)[(S/R)-BINAP]; (b) Temperature dependence of the radiative lifetimes of complexes in the ground solid state, and state diagrams visualizing the TADF processes; (c) CD (dashed lines) and CPL (solid lines) spectra of (S/R)-1 and (S/R)-2 in THF; (d) General OLEDs stack architecture and EL spectra[30]
| ligands | ΔEST/eV | Emissions/nm | PLQYs/% | τ/μs a | EQE/% | Ref. |
|---|---|---|---|---|---|---|
| POP,pytzph | 0.09 | 509 | 43 | 5.5 | 7.6 | [ |
| POP,pytzphcf | 0.13 | 519 | 29 | 16.0 | 6.2 | [ |
| POP,pytzphcz | 0.09 | 503 | 79 | 5.5 | 8.3 | [ |
| DPEPhos,TzTz | - | 712 | 1 | 3.1 | 0.32 | [ |
| XantPhos,TzTz | - | 705 | 1 | 3.2 | 0.17 | [ |
| Tpbz,pz4B | 0.07 | 580 | 7 | 20.5 | - | [ |
| Tpbz,pz2BH2 | 0.08 | 569 | 28 | 11.9 | - | [ |
| Tpbz,tz2BH2 | 0.04 | 540 | 45 | 7.4 | - | [ |
| PymPPh2,MeCN | 0.09 | 515 | 47 | 27.0 | - | [ |
| PymPPh2 | - | 525 | 15 | 22 | - | [ |
| PymPPh2,PhCN | 0.06 | 550 | 65 | 9.6 | - | [ |
表2 配位原子为N、P的双核Cu(Ⅰ)配合物的光学性质
Table 2 The optical properties of binuclear copper complexes with N and P coordination atoms
| ligands | ΔEST/eV | Emissions/nm | PLQYs/% | τ/μs a | EQE/% | Ref. |
|---|---|---|---|---|---|---|
| POP,pytzph | 0.09 | 509 | 43 | 5.5 | 7.6 | [ |
| POP,pytzphcf | 0.13 | 519 | 29 | 16.0 | 6.2 | [ |
| POP,pytzphcz | 0.09 | 503 | 79 | 5.5 | 8.3 | [ |
| DPEPhos,TzTz | - | 712 | 1 | 3.1 | 0.32 | [ |
| XantPhos,TzTz | - | 705 | 1 | 3.2 | 0.17 | [ |
| Tpbz,pz4B | 0.07 | 580 | 7 | 20.5 | - | [ |
| Tpbz,pz2BH2 | 0.08 | 569 | 28 | 11.9 | - | [ |
| Tpbz,tz2BH2 | 0.04 | 540 | 45 | 7.4 | - | [ |
| PymPPh2,MeCN | 0.09 | 515 | 47 | 27.0 | - | [ |
| PymPPh2 | - | 525 | 15 | 22 | - | [ |
| PymPPh2,PhCN | 0.06 | 550 | 65 | 9.6 | - | [ |
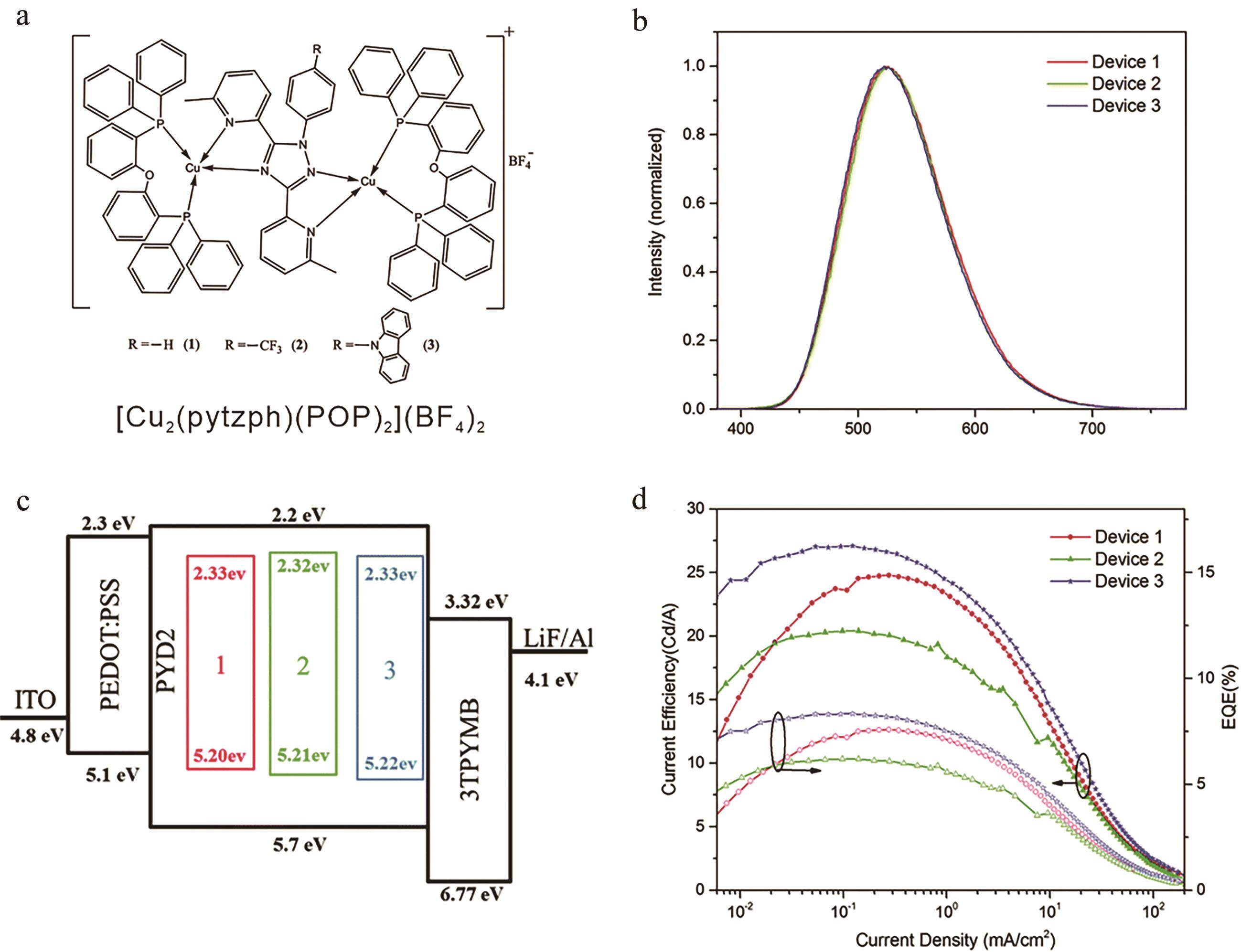
图5 (a) [Cu2(pytzph)(POP)2](BF4)2结构图; (b) OLEDs器件电致发光光谱; (c) OLEDs器件结构图; (d)配合物器件电流效率、外量子产率与电流密度关系[34]
Fig. 5 (a) Structures of [Cu2(pytzph)(POP)2](BF4)2; (b) Normalized EL spectra of OLEDs; (c) Device architecture of OLEDs; (d) Current efficiency and EQE versus current density for the OLEDs devices[34]
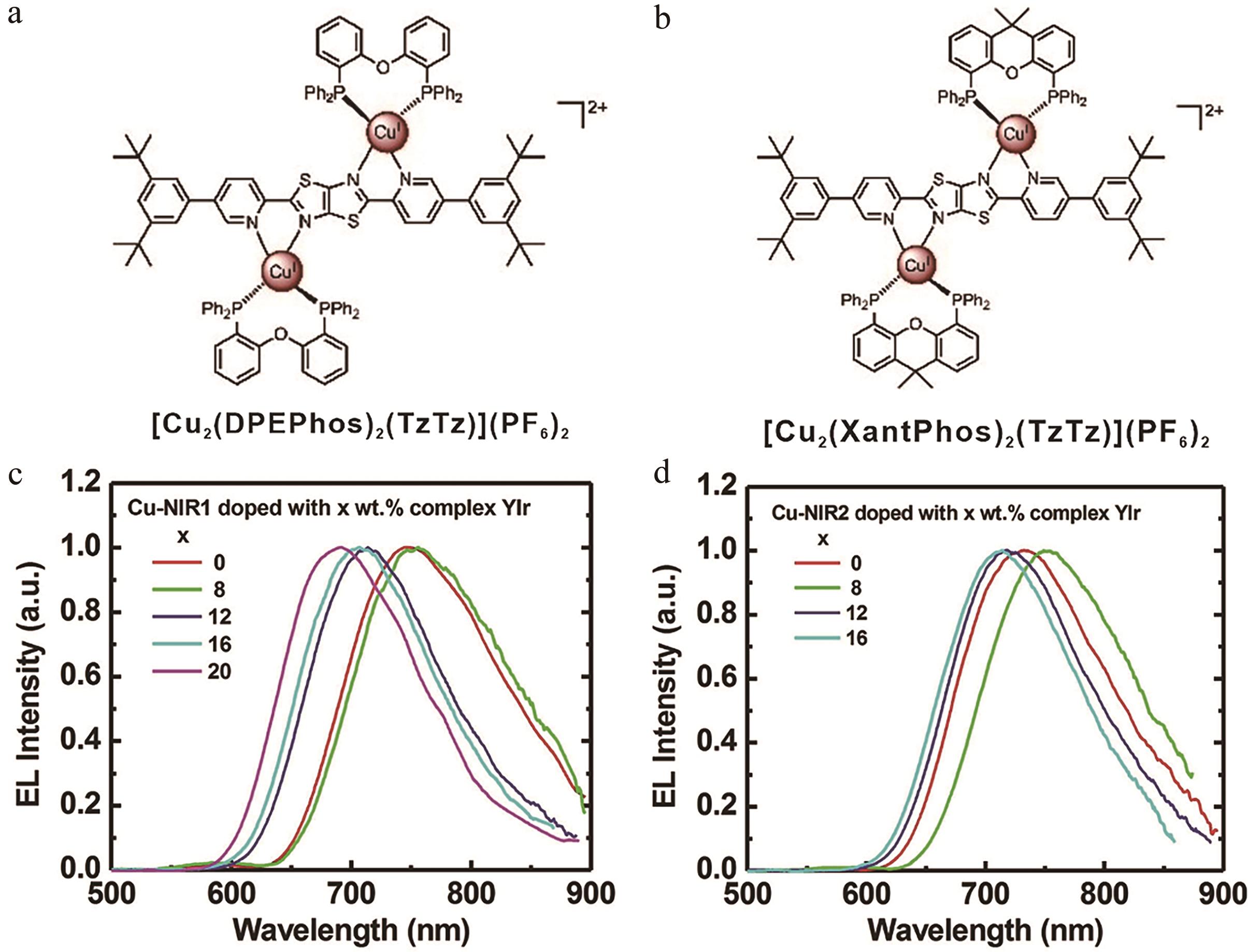
图6 (a) [Cu2(DPEPhos)2(TzTz)]2+结构图; (b) [Cu2(XantPhos)2(TzTz)]2+结构图; (c)基于 [Cu2(DPEPhos)2(TzTz)]2+分子OLEDs器件发射光谱; (d)基于[Cu2(XantPhos)2(TzTz)]2+分子OLEDs器件发射光谱[36]
Fig.6 (a) Structures of [Cu2(DPEPhos)2(TzTz)]2+; (b) Structures of [Cu2(XantPhos)2(TzTz)]2+; (c) Normalized EL spectra of OLEDs based on [Cu2(DPEPhos)2(TzTz)]2+; (d) Normalized EL spectra of OLEDs based on [Cu2(XantPhos)2(TzTz)]2+[36]
| Ligands | ΔEST/eV | Emissions/nm | PLQYs/% | τ/μs a | EQE/% | Ref. |
|---|---|---|---|---|---|---|
| dppt1,I | 0.07 | 487 | 69 | 9.46 | 7.44 | [ |
| dppt2,I | 0.05 | 483 | 86 | 7.62 | 14.50 | [ |
| TTPP,Cl | 0.01 | 530 | 76 | 19 | 9.6 | [ |
| TTPP,Br | 0.04 | 523 | 79 | 16 | 12.4 | [ |
| TTPP,I | 0.05 | 521 | 83 | 11 | 16.3 | [ |
| DBFDP,I | 0.16 | 491 | 5 | 2 | 0.73 | [ |
| PPh3,HPBI,I | - | 581 | 38 | 8.2 | 3.4 | [ |
| PPh3,MOPBI,I | - | 557 | 34 | 22.6 | - | [ |
| PPh3,POPBI,I | - | 382 | 30 | 15.8 | - | [ |
| dpmb,I | 0.12 | 498 | 32 | 2.5 | 10.1 | [ |
| dpmb,Br | 0.12 | 511 | 28 | 12.5 | 7.3 | [ |
| dpmb,Cl | 0.14 | 527 | 29 | 4.8 | 8.3 | [ |
表3 配位原子为 N、P、X(卤素) 的Cu(I)配合物的光学性质
Table 3 The optical properties of Cu(I) complexes with N,P,X(halogen) coordination atoms
| Ligands | ΔEST/eV | Emissions/nm | PLQYs/% | τ/μs a | EQE/% | Ref. |
|---|---|---|---|---|---|---|
| dppt1,I | 0.07 | 487 | 69 | 9.46 | 7.44 | [ |
| dppt2,I | 0.05 | 483 | 86 | 7.62 | 14.50 | [ |
| TTPP,Cl | 0.01 | 530 | 76 | 19 | 9.6 | [ |
| TTPP,Br | 0.04 | 523 | 79 | 16 | 12.4 | [ |
| TTPP,I | 0.05 | 521 | 83 | 11 | 16.3 | [ |
| DBFDP,I | 0.16 | 491 | 5 | 2 | 0.73 | [ |
| PPh3,HPBI,I | - | 581 | 38 | 8.2 | 3.4 | [ |
| PPh3,MOPBI,I | - | 557 | 34 | 22.6 | - | [ |
| PPh3,POPBI,I | - | 382 | 30 | 15.8 | - | [ |
| dpmb,I | 0.12 | 498 | 32 | 2.5 | 10.1 | [ |
| dpmb,Br | 0.12 | 511 | 28 | 12.5 | 7.3 | [ |
| dpmb,Cl | 0.14 | 527 | 29 | 4.8 | 8.3 | [ |
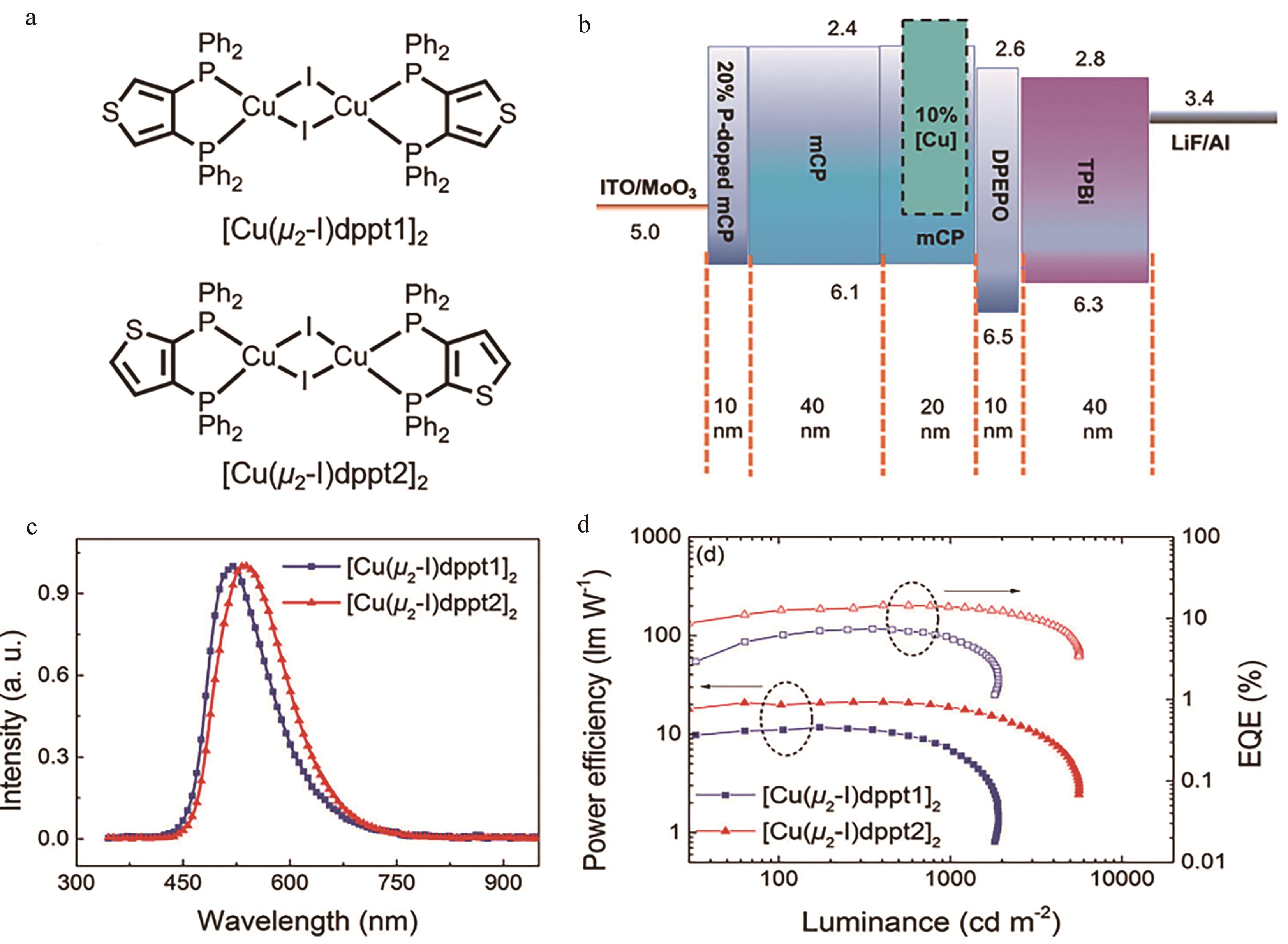
图7 (a) [Cu(μ2-I)dppt1]2及[Cu(μ2-I)dppt2]2结构图; (b) OLEDs器件结构图; (c) 12 V时2种OLEDs器件发射光谱; (d) 2种器件效率及外量子效率与亮度关系[45]
Fig.7 (a) Structures of [Cu(μ2-I)dppt1]2 and [Cu(μ2-I)dppt2]2; (b) Device architecture of OLEDs; (c) EL spectra of both devices at 12 V device architecture of OLEDs; (d) PE-L-EQE characteristics of both devices[45]
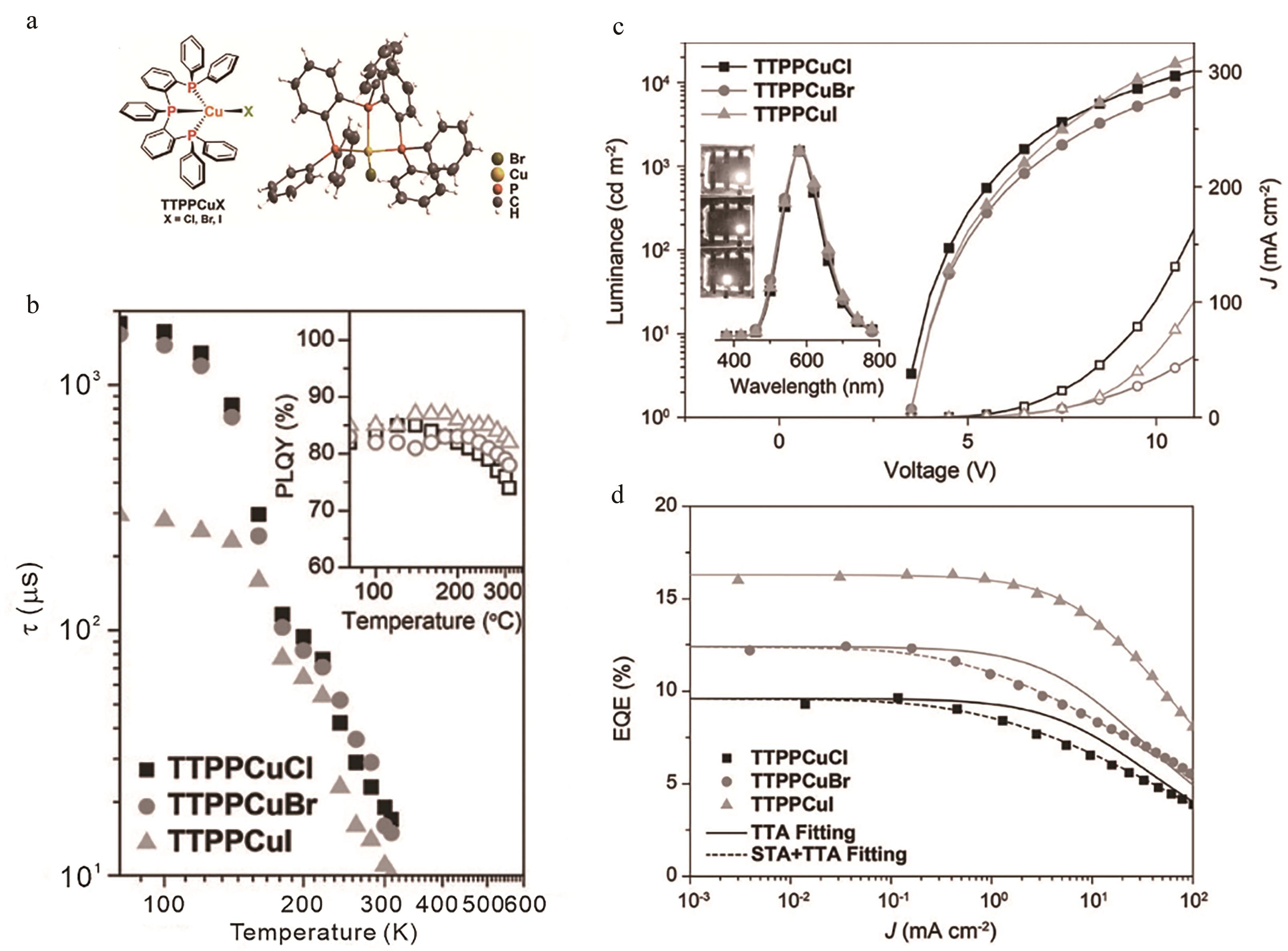
图8 (a) TTPPCuX(X= Cl,Br,I)化学式及TTPPCuBr单晶结构; (b) TTPPCuX变温寿命及变温量子产率; (c) 电流密度(中空)-亮度(实心)-电压曲线,1000 cd/m2时电致发光光谱和5 V时器件照片(插图); (d) 外量子产率与电流密度关系及根据单重态-三重态和三重态-三重态湮灭的拟合曲线[48]
Fig.8 (a) Chemical structure of TTPPCuX (X=Cl, Br, I) and single-crystal structure of TTPPCuBr; (b) Temperature dependence of emissive lifetime (τ) and PLQY for TTPPCuX; (c) Current density (J) (hollow)?-luminance (solid)-voltage curves, EL spectra at 1000 cd/m2 and device photos at 5 V (inset); (d) EQE-J correlations and the fitting curves according to TTA and STA+TTA models[48]
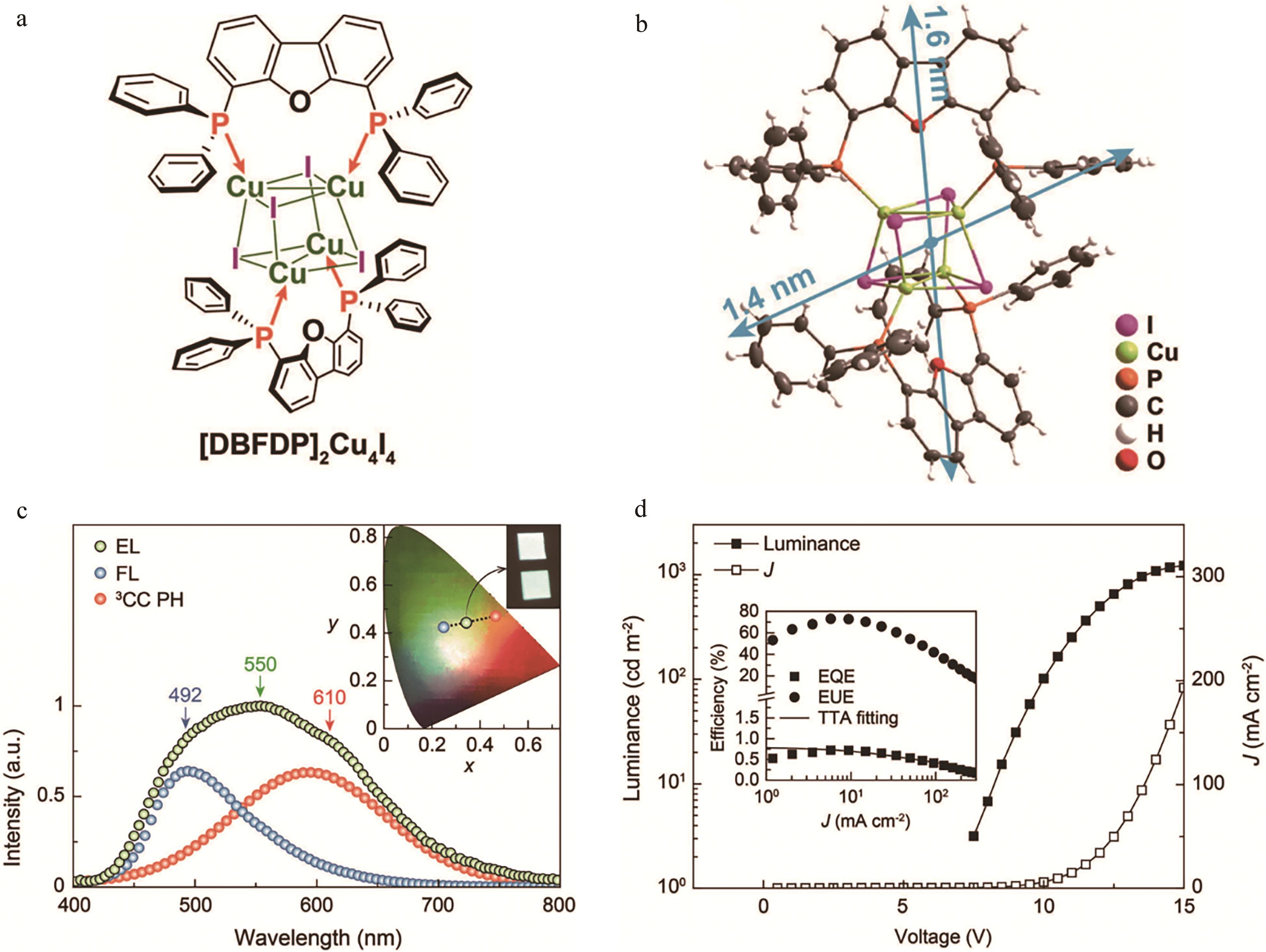
图9 (a) [DBFDP]2Cu4I4结构图; (b) [DBFDP]2Cu4I4单晶结构; (c)使用[DBFDP]2Cu4I4作为掺杂剂的OLEDs电致发光光谱、CIE坐标和照片(插图); (d)亮度-电流密度-电压关系曲线及外量子效率/激子利用率-电流密度曲线[49]
Fig.9 (a) Structures of [DBFDP]2Cu4I4; (b) Single-crystal structure and packing diagram of [DBFDP]2Cu4I4; (c) EL spectra, CIE coordinates and photographs (insets) of the OLEDs using [DBFDP]?2Cu4I4 as dopant; (d) Luminance-current density (J)-voltage relationship and EQE/EUE-J curves (inset)[49]
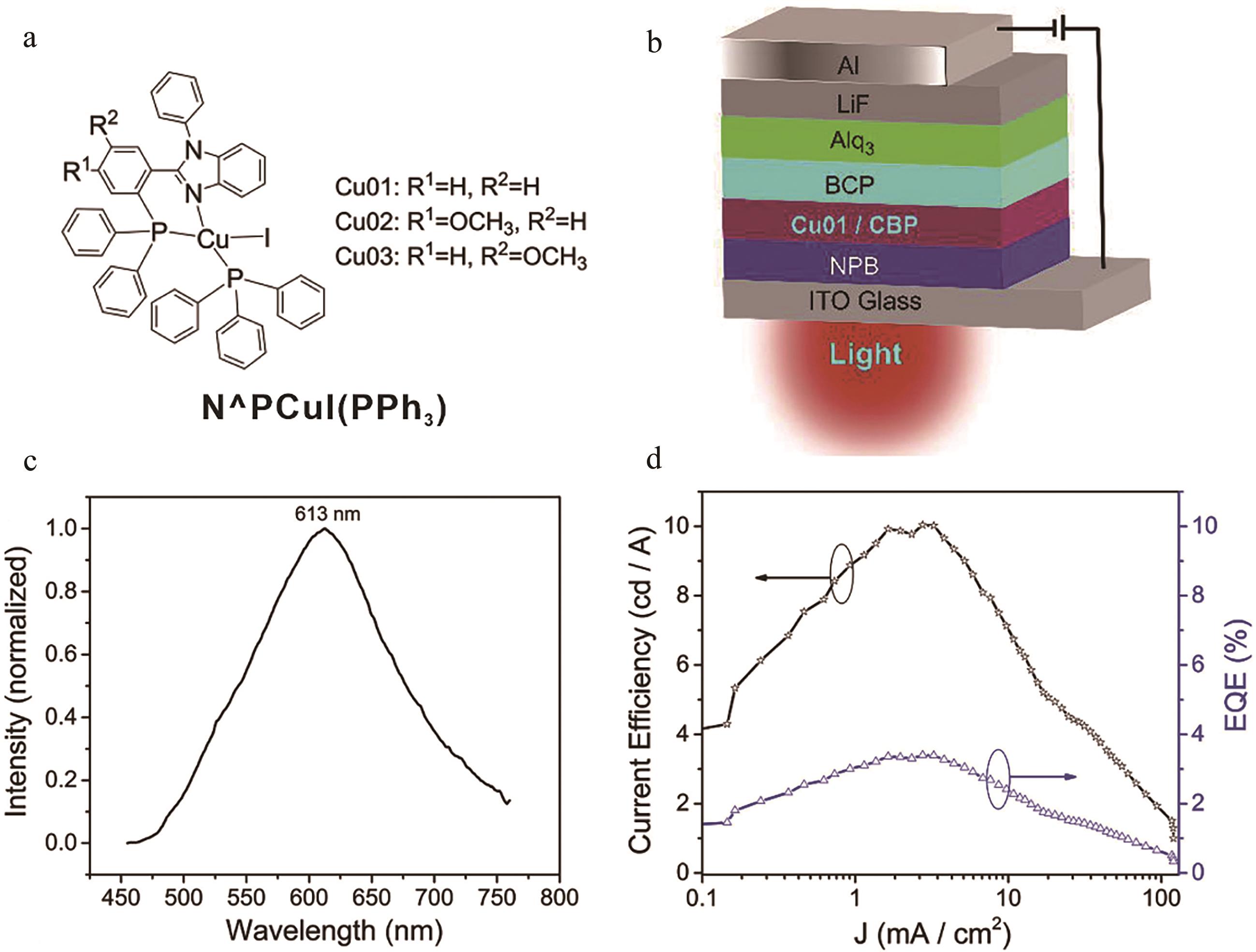
图10 (a) N^PCuI(PPh3)结构图; (b) OLEDs器件结构图; (c) 基于N^PCuI(PPh3)分子OLEDs器件发射光谱; (d) 电流效率、外量子产率与电流密度关系[51]
Fig.10 (a) Structures of N^PCuI(PPh3); (b) Device architecture of OLEDs; (c) Normalized EL spectra of OLEDs based on N^PCuI(PPh3); (d) Current efficiency-EQE-current density (J) relationship[51]
| Ligands | ΔEST/eV | Emissions/nm | PLQYs/% | τ/μs a | EQE/% | Ref. |
|---|---|---|---|---|---|---|
| CAAC,Cz | 0.01 | 474 | 65 | 1.3 | 9.0 | [ |
| MAC*,CzCN2 | - | 438 | 5 | 0.37(33%) 1.8(67%) | - | [ |
| MAC*,CzCN | - | 474 | 76 | 0.75 | - | [ |
| MAC*,Cz | 0.06 | 492 | 53 | 0.84 | 19.4 | [ |
| MAC*,DPAC | 0.05 | 609 | 24 | 0.42 | 21.1 | [ |
| MAC*,CzP | 0.06 | 559 | 40 | 0.4 | 15.2 | [ |
| MAC*,CNCzP | 0.11 | 518 | 48 | 2.58 (77%) 34.1 (23%) | 13.2 | [ |
| Ipr,dpa | 0.10 | 463 | 22 | 13 | - | [ |
| Ipr,dpym | 0.12 | 473 | 15 | 6 | - | [ |
| Ipr,dpyp | 0.10 | 474 | 73 | 14 | - | [ |
| Ipr,PPhpy2 | 0.10 | 503 | 86 | 13 | - | [ |
表4 配位原子为N、C的Cu(Ⅰ)配合物的光学性质
Table 4 The optical properties of Cu(Ⅰ) complexes with N, C coordination atoms
| Ligands | ΔEST/eV | Emissions/nm | PLQYs/% | τ/μs a | EQE/% | Ref. |
|---|---|---|---|---|---|---|
| CAAC,Cz | 0.01 | 474 | 65 | 1.3 | 9.0 | [ |
| MAC*,CzCN2 | - | 438 | 5 | 0.37(33%) 1.8(67%) | - | [ |
| MAC*,CzCN | - | 474 | 76 | 0.75 | - | [ |
| MAC*,Cz | 0.06 | 492 | 53 | 0.84 | 19.4 | [ |
| MAC*,DPAC | 0.05 | 609 | 24 | 0.42 | 21.1 | [ |
| MAC*,CzP | 0.06 | 559 | 40 | 0.4 | 15.2 | [ |
| MAC*,CNCzP | 0.11 | 518 | 48 | 2.58 (77%) 34.1 (23%) | 13.2 | [ |
| Ipr,dpa | 0.10 | 463 | 22 | 13 | - | [ |
| Ipr,dpym | 0.12 | 473 | 15 | 6 | - | [ |
| Ipr,dpyp | 0.10 | 474 | 73 | 14 | - | [ |
| Ipr,PPhpy2 | 0.10 | 503 | 86 | 13 | - | [ |
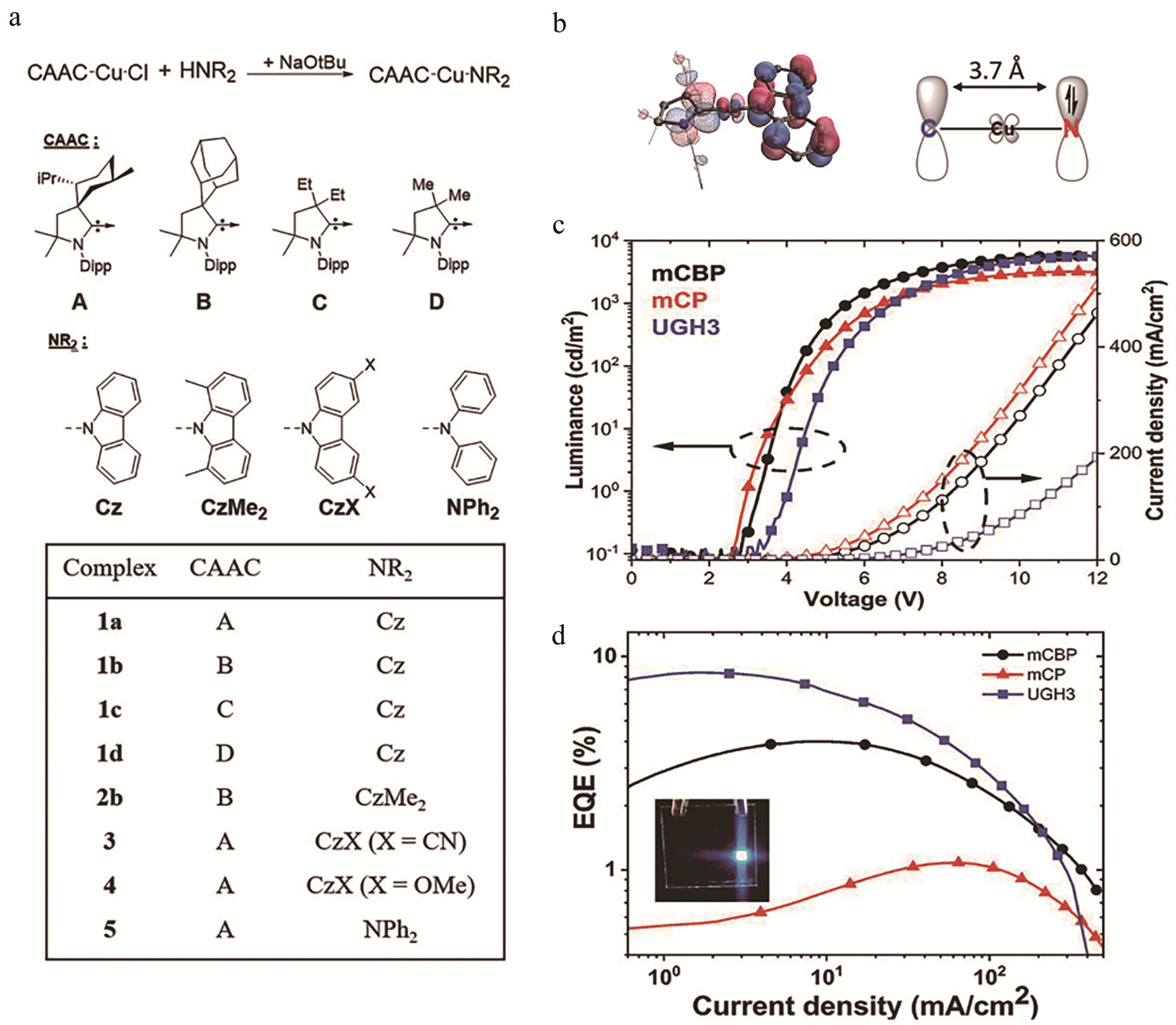
图11 (a) CAAC-Cu-NR2结构图; (b)配合物1a的HOMO和LUMO图; (c)电流-电压-亮度关系; (d)使用不同主体材料的EQE曲线(插图是基于1a的OLEDs照片) [57]
Fig.11 (a) Structures of CAAC-Cu-NR2; (b) HOMO (solid) and LUMO (mesh) surfaces of complex 1a; (c) Current (J)-voltage (V)-luminance (L) traces; (d) EQE traces of devices using different hosts (The inset is a photograph of a 1a-based device)[57]
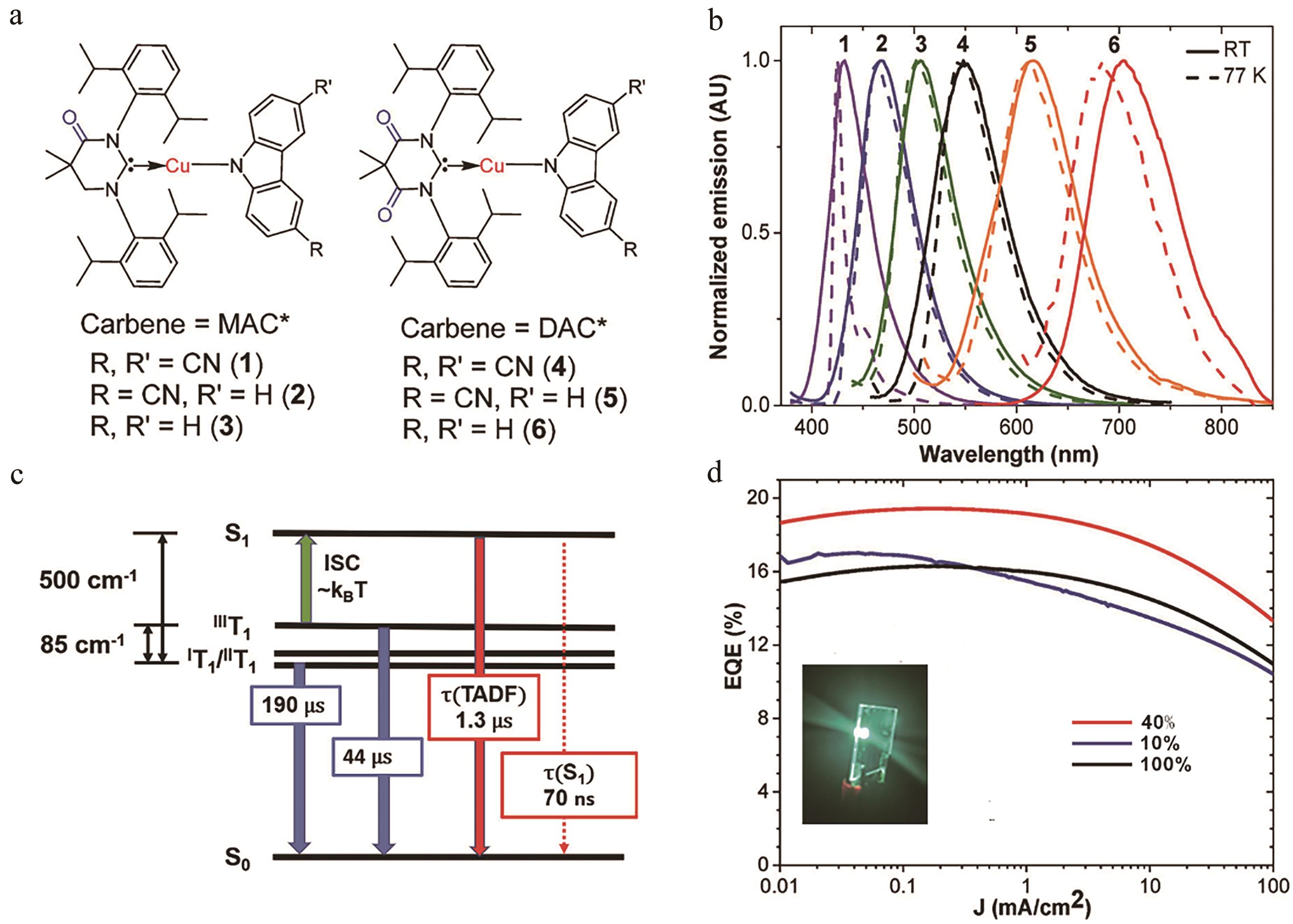
图12 (a) (MAC*)Cu(CzRR')及(DAC*)Cu(CzRR')结构图; (b)室温及77 K发射光谱; (c)配合物3能级图;(d)外量子效率及基于配合物3的器件图(插图),配合物掺杂质量分数10%、40%和100%[63]
Fig.12 (a) Structures of (MAC*)Cu(CzRR') and (DAC*)Cu(CzRR'); (b) The emission spectra at room temperature (RT) and 77 K; (c) Energy level diagram for complex (MAC*)Cu(Cz); (d) External quantum efficiency. Inset: photograph of a (MAC*)Cu(Cz)-based device, doping mass concentrations of 3 are 10%, 40% and 100%[63]
| 1 | ZHANG N, QU L, HU H, et al. Sky blue and yellow cluster light-emitting diodes based on asymmetric Cu4I4 nanocubes[J]. Research,2022(2022): 0005. |
| 2 | TANG C, WVANSLYKE S. A organic electroluminescent diodes[J]. Appl Phys Lett,1987, 51(12): 913-915. |
| 3 | SANDOVAL-PAUKER C, SANTANDER-NELLI MDREYSE P. Thermally activated delayed fluorescence in luminescent cationic copper(Ⅰ) complexes[J]. RSC Adv, 2022, 12(17): 10653-10674. |
| 4 | FU Y, LIU H, YANG D, et al. Boosting external quantum efficiency to 38.6% of sky-blue delayed fluorescence molecules by optimizing horizontal dipole orientation[J]. Sci Adv,2021, 7(43): eabj2504. |
| 5 | UOYAMA H, GOUSHI K, SHIZU K, et al. Highly efficient organic light-emitting diodes from delayed fluorescence[J]. Nature,2012, 492(7428): 234-238. |
| 6 | MA Y G, ZHANG H Y, SHEN J C, et al. Electroluminescence from triplet metal-ligand charge-transfer excited state of transition metal complexes[J]. Synth Met,1998, 94(3): 245-248. |
| 7 | KIDO JIIZUMI Y. Fabrication of highly efficient organic electroluminescent devices[J]. Appl Phys Lett,1998, 73(19): 2721-2723. |
| 8 | ENDO A, OGASAWARA M, TAKAHASHI. A, et al. Thermally activated delayed fluorescence from Sn4+-porphyrin complexes and their application to organic light-emitting diodes-a novel mechanism for electroluminescence[J]. Adv Mater,2009, 21(47): 4802-4806. |
| 9 | BALDO M A, O'BRIEN D F, YOU Y, et al. Highly efficient phosphorescent emission from organic electroluminescent devices[J]. Nature,1998, 395(6698): 151-154. |
| 10 | ZHANG Z, WU P, WANG K, et al. Manipulation of Pt┈Pt interaction in platinum complex by methyl group to achieve single-doped white OLEDs: an approach to simulation of daylight from dawn until dusk[J]. ACS Mater Lett,2023, 5(4): 920-927. |
| 11 | YANG X, XU S, ZHANG Y, et al. Narrowband pure near-infrared (NIR) Ir(Ⅲ) complexes for solution-processed organic light-emitting diode (OLED) with external quantum efficiency over 16%[J]. Angew Chem Int Ed,2023, 62(41): e202309739. |
| 12 | WEN Z, XU Y, SONG X F, et al. Approaching the shortest intermetallic distance of half-lantern diplatinum(Ⅱ) complexes for efficient and stable deep-red organic light-emitting diodes[J]. Adv Opt Mater,2023, 11(14): 2300201. |
| 13 | FUSELLA M A, SARAMAK R, BUSHATI R, et al. Plasmonic enhancement of stability and brightness in organic light-emitting devices[J]. Nature,2020, 585(7825): 379-382. |
| 14 | MA Y G, CHE C M, CHAO H Y, et al. High luminescence gold(Ⅰ) and copper(Ⅰ) complexes with a triplet excited state for use in light-emitting diodes[J]. Adv Mater,1999, 11(10): 852-857. |
| 15 | SCHULZ G, LHOLDCROFT S. Conjugated polymers bearing iridium complexes for triplet photovoltaic devices[J]. Chem Mater,2008, 20(16): 5351-5355. |
| 16 | LI C, REN ZYAN S. Thermally activated delayed fluorescence materials in OLEDs devices:design, synthesis and applications[J]. Chin Sci Bull,2015, 60(31): 2989-3004. |
| 17 | ZHANG Y, SCHULZ M, WAECHTLER M, et al. Heteroleptic diimine-diphosphine Cu(Ⅰ) complexes as an alternative towards noble-metal based photosensitizers: design strategies, photophysical properties and perspective applications[J]. Coord Chem Rev,2018, 356: 127-146. |
| 18 | ZHAN L, YING A, QI Y, et al. Copper(Ⅰ) complex as sensitizer enables high-performance organic light-emitting diodes with very low efficiency roll-off[J]. Adv Funct Mater,2021, 31(48): 2106345. |
| 19 | GERNERT M, BALLES-WOLF L, KERNER F, et al. Cyclic (amino)(aryl)carbenes enter the field of chromophore ligands: expanded π system leads to unusually deep red emitting CuⅠ compounds[J]. J Am Chem Soc,2020, 142(19): 8897-8909. |
| 20 | BIZZARRI C, HUNDEMER F, BUSCH J, et al. Triplet emitters versus TADF emitters in OLEDs: a comparative study[J]. Polyhedron,2018, 140: 51-66. |
| 21 | ALCONCHEL A, CRESPO OGIMENO M C. Thermally activated delayed fluorescence in neutral and cationic copper(Ⅰ) complexes with the 2-(4-thiazolyl)benzimidazole ligand[J]. Inorg Chem, 2023, 62(26): 10431-10439. |
| 22 | LI C, LI W, HENWOOD A F, et al. Luminescent dinuclear copper(Ⅰ) complexes bearing an imidazolylpyrimidine bridging ligand[J]. Inorg Chem,2020, 59(20): 14772-14784. |
| 23 | CZERWIENIEC R, YUJYERSIN H. Correction to blue-light emission of Cu(Ⅰ) complexes and singlet harvesting[J]. Inorg Chem,2012, 51(3): 1975-1975. |
| 24 | ALCONCHEL A, CRESPO O, GARCIA-ORDUNA P, et al. Closo- or nido-carborane diphosphane as responsible for strong thermochromism or time activated delayed fluorescence (TADF) in Cu(N^N)(P^P)0/+[J]. Inorg Chem,2021, 60(23): 18521-18528. |
| 25 | GIOBBIO G, CAVINATO L M M, FRESTA E, et al. Design rule hidden from the eye in S/N-bridged ancillary ligands for copper(Ⅰ) complexes applied to light-emitting electrochemical cells[J]. Adv Funct Mater,2023, 33(50): 2304668. |
| 26 | FRESTA E, MAHORO G U, CAVINATO L M, et al. Novel red-emitting copper(Ⅰ) complexes with pyrazine and pyrimidinyl ancillary ligands for white light-emitting electrochemical cells[J]. Adv Opt Mater,2022, 10(3): 2101999. |
| 27 | ARNOSTI N, BRUNNER F, SUSIC I, et al. Remote modification of bidentate phosphane ligands controlling the photonic properties in their complexes: enhanced performance of [Cu(RN-xantphos)(N^N)][PF6] in light-emitting electrochemical cells[J]. Adv Opt Mater,2020, 8(10): 1901689. |
| 28 | ZHANG F, GUAN Y, CHEN X, et al. Syntheses, photoluminescence, and electroluminescence of a series of sublimable bipolar cationic cuprous complexes with thermally activated delayed fluorescence[J]. Inorg Chem,2017, 56(7): 3742-3753. |
| 29 | TENG T, XIONG J, CHENG G, et al. Solution-processed OLEDs based on thermally activated delayed fluorescence copper(Ⅰ) complexes with intraligand charge-transfer excited state[J]. Molecules,2021, 26(4): 1125. |
| 30 | MUTHIG A M T, MROZEK O, FERSCHKE T, et al. Mechano-stimulus and environment-dependent circularly polarized TADF in chiral copper(Ⅰ) complexes and their application in OLEDs[J]. J Am Chem Soc,2023, 145(8): 4438-4449. |
| 31 | LIANG D, CHEN X L, LIAO J Z, et al. Highly efficient cuprous complexes with thermally activated delayed fluorescence for solution-processed organic light-emitting devices[J]. Inorg Chem,2016, 55(15): 7467-7475. |
| 32 | XU H, YANG T, WANG F, et al. Thermally activated delayed fluorescence of copper(Ⅰ) complexes using N,N'-heteroaromatic of 2-(5-phenyl-1,2,3-triazole)pyridine as ligand[J]. J Lumin,2019, 205: 82-86. |
| 33 | ZHANG R, LIU J W, ZHONG W Y, et al. Mechanochromic and selective vapochromic solid-state luminescence of a dinuclear cuprous complex[J]. Inorg Chem,2023, 62(29): 11510-11517. |
| 34 | LIN L, CHEN D H, YU R, et al. Photo- and electro-luminescence of three TADF binuclear Cu(Ⅰ) complexes with functional tetraimine ligands[J]. J Mater Chem C,2017, 5(18): 4495-4504. |
| 35 | GRUPE M, BODEN P, DI MARTINO-FUMO P, et al. Time-resolved spectroscopy and electronic structure of mono- and dinuclear pyridyl-triazole/dpephos-based Cu(Ⅰ) complexes[J]. Chem Eur J,2021, 27(61): 15251-15270. |
| 36 | JOUAITI A, BALLERINI L, SHEN H L, et al. Binuclear copper(Ⅰ) complexes for near-infrared light-emitting electrochemical cells[J]. Angew Chem Int Ed,2023, 62(38): e202305569. |
| 37 | CHEN J, TENG T, WANG J Y, et al. Synthesis, structure, and characterization of emissive neutral dinuclear CuI complexes with a tetraphosphane bridging ligand[J]. Eur J Inorg Chem,2016(18): 3036-3041. |
| 38 | ARTEM'EV A V, DAVYDOVA M P, BEREZIN A S, et al. Dicopper(Ⅰ) paddle-wheel complexes with thermally activated delayed fluorescence adjusted by ancillary ligands[J]. Inorg Chem,2020, 59(15): 10699-10706. |
| 39 | XU K, CHEN B L, YANG F, et al. Largely color-tuning prompt and delayed fluorescence: dinuclear Cu(Ⅰ) halide complexes with tert-amines and phosphines[J]. Inorg Chem,2021, 60(7): 4841-4851. |
| 40 | SUN C, LLANOS L, ARCE P, et al. Nuclearity control for efficient thermally activated delayed fluorescence in a CuI complex and its halogen-bridged dimer[J]. Chem Mater,2021, 33(16): 6383-6393. |
| 41 | GUO B K, YANG F, WANG Y Q, et al. Efficient TADF-OLEDs with ultra-soluble copper(Ⅰ) halide complexes containing non-symmetrically substituted bidentate phosphine and PPh3 ligands[J]. J Lumin,2020, 220: 116963. |
| 42 | ZHANG N, QU L, DAI S, et al. Intramolecular charge transfer enables highly-efficient X-ray luminescence in cluster scintillators[J]. Nat Commun,2023, 14(1): 2901. |
| 43 | ZHANG N, LI Y, HAN S, et al. Cluster light-emitting diodes containing copper iodine cube with 100% exciton utilization using host-cluster synergy[J]. Angew Chem Int Ed,2023, 62(27): e202305018. |
| 44 | ZHANG N, HU H, QU L, et al. Overcoming efficiency limitation of cluster light-emitting diodes with asymmetrically functionalized biphosphine Cu4I4 cubes[J]. J Am Chem Soc,2022, 144(14): 6551-6557. |
| 45 | LI X, ZHANG J, ZHAO Z, et al. Bluish-green Cu(Ⅰ) dimers chelated with thiophene ring-introduced diphosphine ligands for both singlet and triplet harvesting in OLEDs[J]. ACS Appl Mater Interfaces,2019, 11(3): 3262-3270. |
| 46 | LI Y, ZHANG X, MAN Y, et al. Interfacial passivation enormously enhances electroluminescence of triphenylphosphine Cu4I4 cube[J]. Adv Mater,2023, 35(36): 2302984. |
| 47 | LI Y, XU S, ZHANG X, et al. Bulk passivation enables hundredfold-enhanced electroluminescence of monophosphine Cu4I4 cubes[J]. Angew Chem Int Ed,2023, 62(39): e202308410. |
| 48 | ZHANG J, DUAN C, HAN C, et al. Balanced dual emissions from tridentate phosphine-coordinate copper(Ⅰ) complexes toward highly efficient yellow OLEDs[J]. Adv Mater,2016, 28(28): 5975-5979. |
| 49 | XIE M, HAN C, ZHANG J, et al. White electroluminescent phosphine-chelated copper iodide nanoclusters[J]. Chem Mater,2017, 29(16): 6606-6610. |
| 50 | YANG L, XU X, ZHANG P, et al. Photophysical properties and stability of binuclear emissive copper(Ⅰ) complexes co-deposited with CuX (X=Cl, Br,Ⅰ) and aza-9,9'-spirobifluorenes[J]. Dyes Pigm,2019, 161: 296-302. |
| 51 | JIAO B, WANG J, HUANG J, et al. Design and synthesis of stable cuprous complexes bearing P^N-type ligands for vapor-deposited organic light-emitting device[J]. Org Electron,2019, 64: 158-165. |
| 52 | HONG X, WANG B, LIU L, et al. Highly efficient blue-green neutral dinuclear copper(Ⅰ) halide complexes containing bidentate phosphine ligands[J]. J Lumin,2016, 180: 64-72. |
| 53 | ZOBEL J P, WERNBACHER A, MGONZALEZ L. Efficient reverse intersystem crossing in carbene-copper-amide TADF emitters via an intermediate triplet state[J]. Angew Chem Int Ed,2023, 62(15): e202217620. |
| 54 | YING A, GONG S. A rising star: luminescent carbene-metal-amide complexes[J]. Chem Eur J,2023, 29(59): e202301885. |
| 55 | MUNIZ C N, SCHAAB J, RAZGONIAEV A, et al. Pi-extended ligands in two-coordinate coinage metal complexes[J]. J Am Chem Soc,2022, 144(39): 17916-17928. |
| 56 | MUNIZ C N, ARCHER C A, APPLEBAUM J S, et al. Two-coordinate coinage metal complexes as solar photosensitizers[J]. J Am Chem Soc,2023, 145(25): 13846-13857. |
| 57 | HAMZE R, PELTIER J L, SYLVINSON D, et al. Eliminating nonradiative decay in Cu(Ⅰ) emitters: >99% quantum efficiency and microsecond lifetime[J]. Science,2019, 363(6427): 601-606. |
| 58 | STRELNIK I D, DAYANOVA I, GERASIMOVA T P, et al. Deep-blue emissive copper(Ⅰ) complexes based on p-thiophenylethyl-substituted cyclic bisphosphines displaying photoinduced structural transformations of the excited states[J]. Inorg Chem,2022, 61(42): 16596-16606. |
| 59 | WANG D N, HU W H, LIU C M, et al. Electronic tuning of photoexcited dynamics in heteroleptic Cu(Ⅰ) complex photosensitizers[J]. J Phys Chem Lett,2023, 14(45): 10137-10144. |
| 60 | YING A, AI Y, YANG C, et al. Aggregation-dependent circularly polarized luminescence and thermally activated delayed fluorescence from chiral carbene-CuI-amide enantiomers[J]. Angew Chem Int Ed,2022, 61(45): e202210490. |
| 61 | WANG H J, LIU Y, YU B, et al. A configurationally confined thermally activated delayed fluorescent two-coordinate CuⅠ complex for efficient blue electroluminescence[J]. Angew Chem Int Ed,2023, 62(7): e202217195. |
| 62 | LI J, WANG L, ZHAO Z, et al. Two-coordinate copper(Ⅰ)/NHC complexes: dual emission properties and ultralong room-temperature phosphorescence[J]. Angew Chem Int Ed,2020, 59(21): 8210-8217. |
| 63 | SHI S, JUNG M C, COBURN C, et al. Highly efficient photo- and electroluminescence from two-coordinate Cu(Ⅰ) complexes featuring nonconventional N-heterocyclic carbenes[J]. J Am Chem Soc,2019, 141(8): 3576-3588. |
| 64 | YING A, HUANG Y H, LU C H, et al. High-efficiency red electroluminescence based on a carbene-Cu(Ⅰ)-acridine complex[J]. ACS Appl Mater Interfaces,2021, 13(11): 13478-13486. |
| 65 | YING A, ZHAN L, TAN Y, et al. Copper(Ⅰ) complexes with planar chirality realize efficient circularly polarized electroluminescence[J]. Sci China Chem,2023, 66(8): 2274-2282. |
| 66 | ELIE M, WEBER M D, DI MEO F, et al. Role of the bridging group in bis-pyridyl ligands: enhancing both the photo- and electroluminescent features of cationic (IPr)CuⅠ complexes[J]. Chem Eur J,2017, 23(64): 16328-16337. |
| 67 | OLARU M, RYCHAGOVA E, KETKOV S, et al. A small cationic organo-copper cluster as thermally robust highly photo- and electroluminescent material[J]. J Am Chem Soc,2020, 142(1): 373-381. |
| [1] | 陈栎岩, 赵梓茗, 陶金琪, 张玉贞, 程刚, 沈云军. 单、双核含双硫配体金属有机铂磷光材料合成及其OLED应用[J]. 应用化学, 2023, 40(2): 236-244. |
| [2] | 郭海泉, 杨正华, 高连勋. 光敏聚酰亚胺光刻胶研究进展[J]. 应用化学, 2021, 38(9): 1119-1137. |
| [3] | 霍冉,吴雨萱,杨煜,朴树清,张治城,肖佶海,史翎. 石墨烯电子器件的研究进展[J]. 应用化学, 2019, 36(3): 245-258. |
| [4] | 刘伟强,崔荣朕,武瑞霞,李云辉,杨秀云,周亮. 蓝色延迟荧光材料及器件的研究进展[J]. 应用化学, 2019, 36(1): 1-9. |
| [5] | 邵世洋, 丁军桥, 王利祥. 高分子热活化延迟荧光材料研究进展[J]. 应用化学, 2018, 35(9): 993-1004. |
| [6] | 袁廷,孟婷,李淑花,范楼珍. 基于磷光材料的电致发光二极管研究进展[J]. 应用化学, 2018, 35(8): 871-880. |
| [7] | 关晓琳, 孟丽, 贾天明, 靳其军, 路宝翠, 来守军. 液晶性直线型共轭芳炔衍生物的合成及电致发光性质[J]. 应用化学, 2018, 35(4): 426-435. |
| [8] | 赵学森,崔荣朕,李云辉,高莹,崔成哲. 红色铱配合物磷光材料及器件的研究进展[J]. 应用化学, 2016, 33(9): 1002-1008. |
| [9] | 关晓琳, 张扬, 范红婷, 张东海, 贾天明, 来守军, 雷自强. 新型含萘基和二乙炔基高双折射液晶分子的合成及光电性能[J]. 应用化学, 2016, 33(5): 533-541. |
| [10] | 关晓琳, 张扬, 范红婷, 张东海, 贾天明, 来守军, 雷自强. 新型含萘基和二乙炔基高双折射液晶分子的合成及光电性能[J]. 应用化学, 2016, 33(5): 0-0. |
| [11] | 胡峥勇, 李善佳, 董新荣. 一种环金属铱、铂双核配合物的合成及光电性能[J]. 应用化学, 2016, 33(12): 1428-1434. |
| [12] | 崔荣朕, 唐艳茹, 马玉芹, 杨秀云, 耿丽华, 李云辉. 蓝色有机电致发光材料及器件的研究进展[J]. 应用化学, 2015, 32(8): 855-872. |
| [13] | 代岩峰,张智强,刘一鹏,马东阁. 无间隔层结构的高效率荧光/磷光混合型白光有机发光二极管[J]. 应用化学, 2015, 32(10): 1139-1145. |
| [14] | 吕剑虹, 马志华, 丁军桥, 王利祥. 基于氟代苯并咪唑配体的绿光铱配合物的合成与表征[J]. 应用化学, 2014, 31(10): 1177-1184. |
| [15] | 史常生, 陈江山, 马东阁. 超薄铝/碳酸锂修饰氧化铟锡阴极制备蓝色磷光反转底发光有机发光二极管[J]. 应用化学, 2012, 29(12): 1412-1416. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||