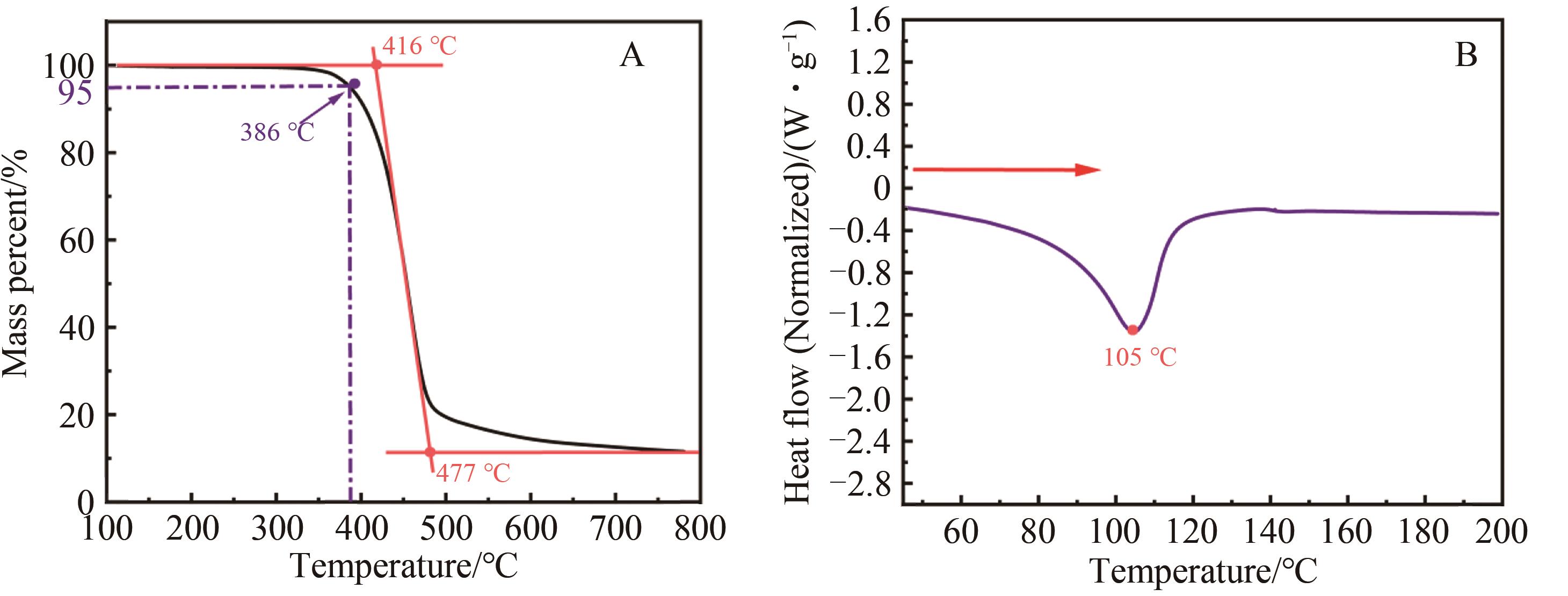
Chinese Journal of Applied Chemistry ›› 2023, Vol. 40 ›› Issue (9): 1322-1329.DOI: 10.19894/j.issn.1000-0518.230019
• Full Papers • Previous Articles Next Articles
Synthesis, Photophysical Properties, Theoretical Calculation and Cell Imaging of a Tetraphenylethene Imidazole Compound with Methoxy Group
Da-Wei TONG1, Ming KONG1( ), Yu-Bin XIANG2
), Yu-Bin XIANG2
- 1.Nanjing Institute of Product Quality Inspection,Nanjing 210019,China
2.School of Chemistry and Materials Science,Nanjing University of Information Science & Technology,Nanjing 210044,China
-
Received:2023-02-09Accepted:2023-05-24Published:2023-09-01Online:2023-09-14 -
Contact:Ming KONG -
About author:mingkong@smail.nju.edu.cn
-
Supported by:the Key Science and Technology Program of Nanjing Administration for Market Regulation(Kj2021021);the Shuangchuang Program of Jiangsu Province(JSSCBS20221736);the National Natural Science Foundation of China(22205114);the Natural Science Foundation of Jiangsu Province(BK20190776)
CLC Number:
Cite this article
Da-Wei TONG, Ming KONG, Yu-Bin XIANG. Synthesis, Photophysical Properties, Theoretical Calculation and Cell Imaging of a Tetraphenylethene Imidazole Compound with Methoxy Group[J]. Chinese Journal of Applied Chemistry, 2023, 40(9): 1322-1329.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.230019

Fig.2 The crystal structure of BTI (Thermal ellipsoids shown at the 30% probability level and the hydrogen atoms has been omitted for clarity); (B) The hydrogen bonding interactions between adjacent molecules
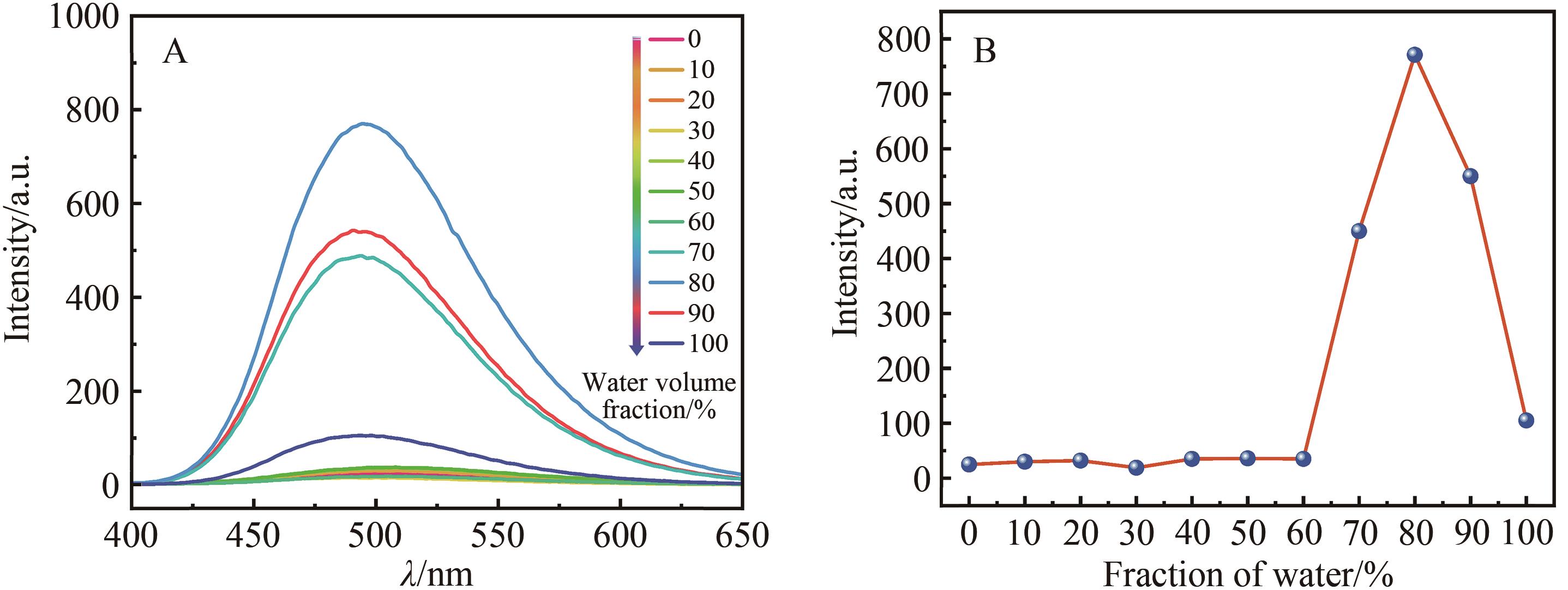
Fig.5 The fluorescence emission spectra of BTI in different volume fraction of ethanol and water mixed solution (A) and the variation of fluorescence intensity according to the water volume fraction (B) (concentration: 1×10-5 mol/L, slit width: 5 nm)
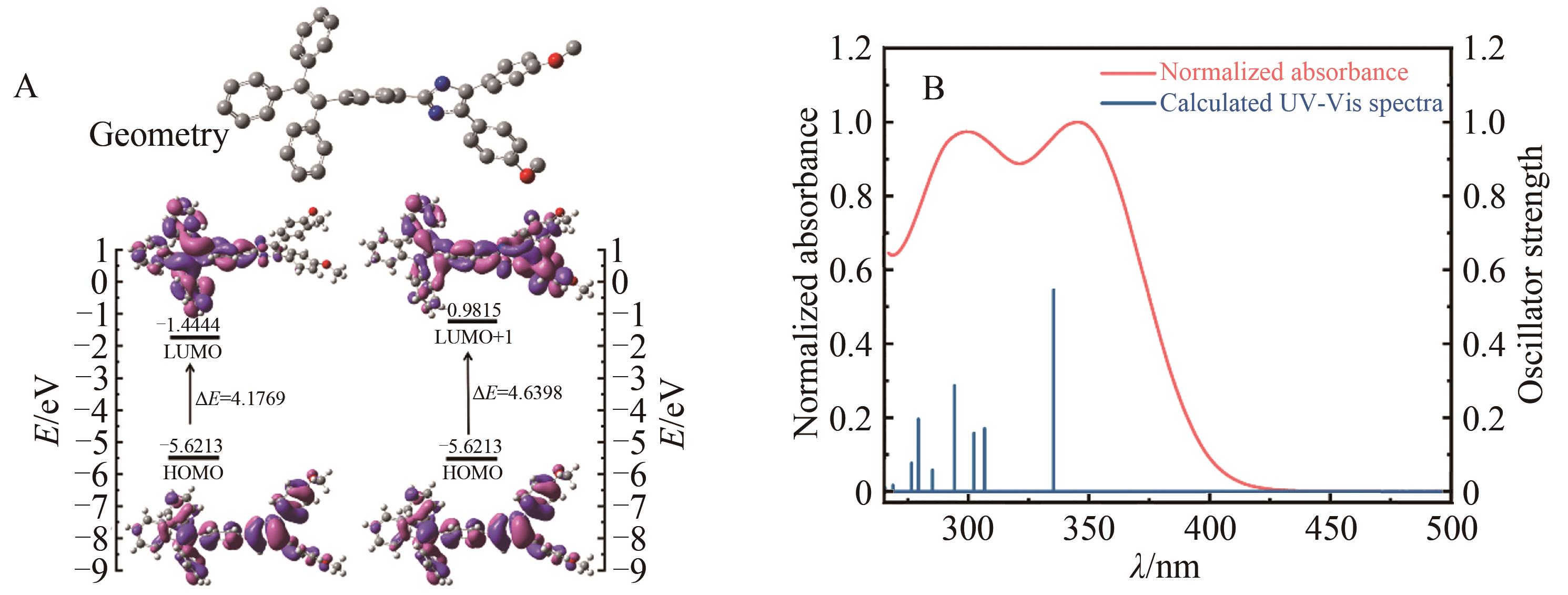
| Compound | OI a | ΔE/eV b | Cal.λmax/nm c | Obs.λmax/nm d | f e | Character |
|---|---|---|---|---|---|---|
| BTI | HOMO→LUMO | 4.176 9 | 335 | 344 | 0.5457 | ICT/π→π* |
| HOMO→LUMO+1 | 4.639 8 | 302 | 299 | 0.1578 | π→π* |
Table 1 The major electronic energy level of electron transitions and attributions for compound by TD-DFT calculations
| Compound | OI a | ΔE/eV b | Cal.λmax/nm c | Obs.λmax/nm d | f e | Character |
|---|---|---|---|---|---|---|
| BTI | HOMO→LUMO | 4.176 9 | 335 | 344 | 0.5457 | ICT/π→π* |
| HOMO→LUMO+1 | 4.639 8 | 302 | 299 | 0.1578 | π→π* |
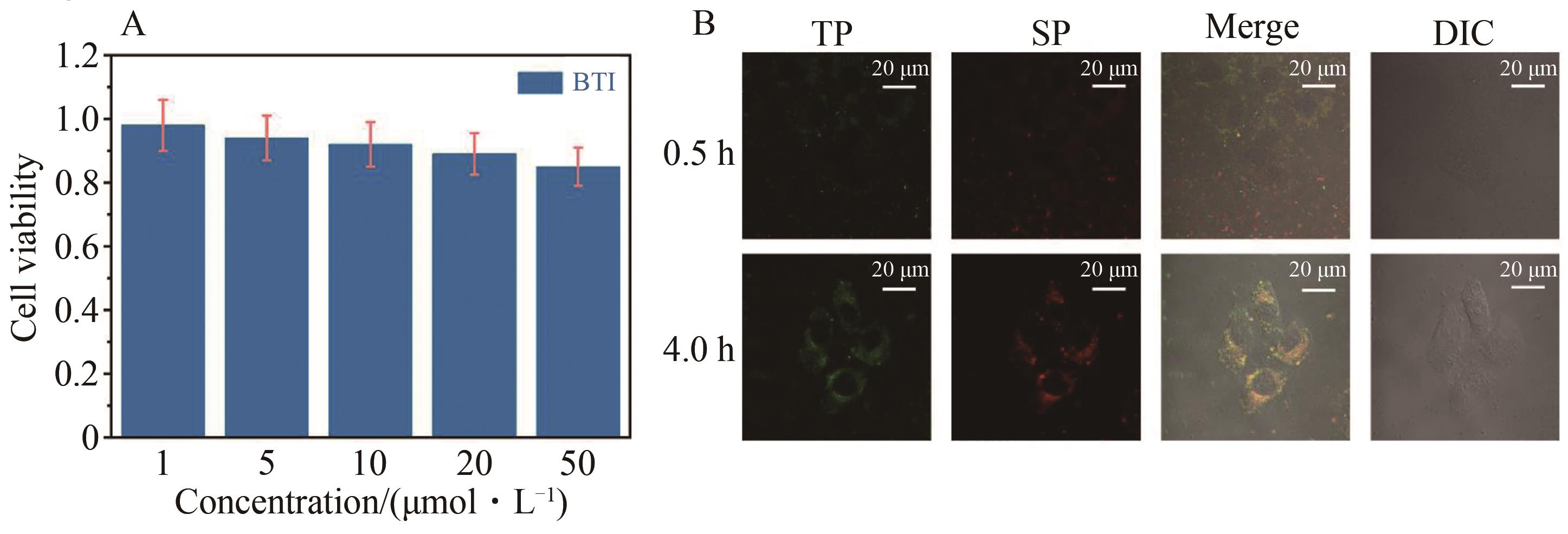
Fig.7 The cytotoxic diagram in HepG2 cells of BTI (A) and the one- and two-photon confocal fluorescence images of BTI in HepG2 cells at different time (B) (incubated with the concentrations 10 μmol/L, two-photon excited wavelength at 700 nm, confocal fluorescence images were received by collecting the emissions at 450~600 nm)
| 1 | 任飞, 石建兵, 佟斌, 等. 具有聚集诱导发光性质的近红外荧光染料[J]. 化学进展, 2021, 33(3): 341-354. |
| REN F, SHI J B, TONG B, et al. Near infrared fluorescent dyes with aggregation-induced emission[J]. Prog Chem, 2021, 33(3): 341-354. | |
| 2 | WU D, SEDGWICK A C, GUNNLAUGSSON T, et al. Fluorescent chemosensors: the past, present and future[J]. Chem Soc Rev, 2017, 46(23): 7105-7123. |
| 3 | 张鹏, 郭心洁, 张倩, 等. 有机染料聚集在光化学传感中的应用[J]. 化学进展, 2020, 32(2/3): 286-297. |
| ZHANG P, GUO X J, ZHANG Q, et al. Photochemical sensing based on the aggregation of organic dyes[J]. Prog Chem, 2020, 32(2/3): 286-297. | |
| 4 | 韩鹏博, 徐赫, 安众福, 等. 聚集诱导发光[J]. 化学进展, 2022, 34(1): 1-130. |
| HAN P B, XU H, AN Z F, et al. Aggregation-induced emission[J]. Prog Chem, 2022, 34(1): 1-130. | |
| 5 | 程金华, 姜鸿基. 末端四苯乙烯荧光团标记法研究双亲性嵌段聚合物的自组装行为[J]. 应用化学, 2019, 36(4): 440-450. |
| CHENG J H, JIANG H J. Study on self-assembly behaviors of an amphiphilic block polymer by terminally grafting tetraphenylethene-based aggregation-induced emission active moietys[J]. Chin J Appl Chem, 2019, 36(4): 440-450. | |
| 6 | LUO J, XIE Z, LAM J W Y, et al. Aggregation-induced emission of 1-methyl-1,2,3,4,5-pentaphenylsilole[J]. Chem Commun, 2001(18): 1740-1741. |
| 7 | XIAO P, XIE W, ZHANG J, et al. De novo design of reversibly pH-switchable NIR-II aggregation-induced emission luminogens for efficient phototheranostics of patient-derived tumor xenografts[J]. J Am Chem Soc, 2023, 145(1): 334-344. |
| 8 | WU Y, LI J, SHEN Z, et al. Double-pronged antimicrobial agents based on a donor-π-acceptor type aggregation-induced emission luminogen[J]. Angew Chem Int Ed, 2022, 61(47): e202212386. |
| 9 | YANG J, FANG M, LI Z. Organic luminescent materials: the concentration on aggregates from aggregation-induced emission[J]. Aggregate, 2020, 1(1): 6-18. |
| 10 | NIE X, HUANG W, ZHOU D, et al. Kinetic and thermodynamic control of tetraphenylethene aggregation-induced emission behaviors[J]. Aggregate, 2022, 3(4): e165. |
| 11 | LIU J, ZHANG H, HU L, et al. Through-space interaction of tetraphenylethylene: what, where, and how[J]. J Am Chem Soc, 2022, 144(17): 7901-7910. |
| 12 | WU C, LI J, DUAN X. Enrichment of aggregation-induced emission aggregates using acoustic streaming tweezers in microfluidics for trace human serum albumin detection[J]. Anal Chem, 2023, 95(3): 2071-2078. |
| 13 | SUN Y, LEI Z, MA H. Twisted aggregation-induced emission luminogens (AIEgens) contribute to mechanochromism materials: a review[J]. J Mater Chem C, 2022, 10(40): 14834-14867. |
| 14 | XIE Y, LI Z. Development of aggregated state chemistry accelerated by aggregation-induced emission[J]. Natl Sci Rev, 2021, 8(6): nwaa199. |
| 15 | ZHAO C X, LIU T, XU M, et al. A fundamental study on the fluorescence-quenching effect of nitro groups in tetraphenylethene AIE dyes with electron-withdrawing groups[J]. Chin Chem Lett, 2021, 32(6): 1925-1928. |
| 16 | HONG Y, LAM J W Y, TANG B Z. Aggregation-induced emission[J]. Chem Soc Rev, 2011,40: 5361-5388. |
| 17 | DING D, LI K, LIU B, et al. Bioprobes based on AIE fluorogens[J]. Acc Chem Res, 2013, 46(11): 2441-2453. |
| 18 | WU W, LIU B. Aggregation-induced emission: challenges and opportunities[J]. Nat Sci Rev, 2021, 8(6): nwaa222. |
| 19 | YANG J, CHI Z, ZHU W, et al. Aggregation-induced emission: a coming-of-age ceremony at the age of eighteen[J]. Sci China Chem, 2019, 62(9): 1090-1098. |
| 20 | 王涛, 马拉毛草, 马恒昌. 基于聚集诱导发光荧光探针的细胞成像研究进展[J]. 应用化学, 2018, 35(10): 1155-1165. |
| WANG T, MA L M C, MA H C. Research progress on cell imaging based on the aggregation-induced emission fluorescent probes[J]. Chin J Appl Chem, 2018, 35(10): 1155-1165. | |
| 21 | ZHENG Z, ZHANG T, LIU H, et al. Bright near-infrared aggregation-induced emission luminogens with strong two-photon absorption, excellent organelle specificity, and efficient photodynamic therapy potential[J]. ACS Nano, 2018, 12(8): 8145-8159. |
| 22 | QIN W, ZHANG P, LI H, et al. Ultrabright red AIEgens for two-photon vascular imaging with high resolution and deep penetration[J]. Chem Sci, 2018, 9(10): 2705-2710. |
| 23 | CHEN M, XIE W, LI D, et al. Utilizing a pyrazine-containing aggregation-induced emission luminogen as an efficient photosensitizer for imaging-guided two-photon photodynamic therapy[J]. Chem Eur J, 2018, 24(62): 16603-16608. |
| 24 | KIM M H, CHO B R. Small-molecule two-photon probes for bioimaging applications[J]. Chem Rev, 2015, 115(11): 5014-5055. |
| 25 | ZHENG K, CHEN H, XIAO Y, et al. A novel strategy to design and construct AIE-active mechanofluorochromic materials via regulation of molecular structure[J]. Chem Eur J, 2021, 27(60): 14964-14970. |
| 26 | 李亚雯, 敖宛彤, 金慧琳, 等. 四苯乙烯衍生物与大环主体在主客体相互作用下的聚集诱导发光[J]. 化学进展, 2019, 31(1): 121-134. |
| LI Y W, AO W T, JIN H L, et al. Aggregation-induced emission of tetraphenylethene derivatives with macrocycles via host-guest interactions[J]. Prog Chem, 2019, 31(1): 121-134. | |
| 27 | MENG Q, CUI L, LIAO Q, et al. Chiral cyclic architectonics with tetraphenylethylenes: conformation immobilization, optical resolution and circularly polarized luminescence[J]. Chem Commun, 2022, 58(77): 10384-10387. |
| 28 | WANG X, SONG Y, PAN G, et al. Exploiting radical-pair intersystem crossing for maximizing singlet oxygen quantum yields in pure organic fluorescent photosensitizers[J]. Chem Sci, 2020,11(40): 10921-10927. |
| 29 | YUAN Y X, JIA J H, SONG Y P, et al. Fluorescent TPE macrocycle relayed light-harvesting system for bright customized-color circularly polarized luminescence[J]. J Am Chem Soc, 2022, 144(12): 5389-5399. |
| 30 | MELHUISH W H. Quantum efficiencies of fluorescence of organic substances: effect of solvent and concentration of the fluorescent solute[J]. J Phys Chem, 1961, 65(2): 229-235. |
| 31 | FRISCH M J, TRUCKS G W, SCHLEGEL H B, et al. Gaussian 09, Revision B[CP]. 01, Gaussian, Inc., Wallingford CT, 2010. |
| 32 | 石玉芳, 王迎进, 孙金鱼, 等. 含多甲氧基芳香基双查尔酮的合成、表征及光热性能[J]. 应用化学, 2022, 39(2): 235-240. |
| SHI Y F, WANG Y J, SUN J Y, et al. Synthesis, characterization, photothermal properties of dichalcone containing polymethoxy aromatic group[J]. Chin J Appl Chem, 2022, 39(2): 235-240. |
| [1] | Yue YANG, Shi-Wen HUANG, Yue TONG, Ze-Da CHEN, Ben-Hua MA, Chuan-Dong DOU. Donor-Acceptor Type Chiral Tetracoordinate Organoboranes and Their Optical Properties [J]. Chinese Journal of Applied Chemistry, 2023, 40(5): 743-748. |
| [2] | Zhi-Peng DU, Yang ZHOU, San-Gen ZHAO. Synthesis, Crystal Structure and Birefringence Properties of Silver Cluster Compound Ag3B6O10I [J]. Chinese Journal of Applied Chemistry, 2023, 40(2): 229-235. |
| [3] | Jin-Jian LIU, Na LIU, Feng-Yi YANG. Synthesis and Photochromic Properties of Two Isostructural Viologen Coordination Polymers [J]. Chinese Journal of Applied Chemistry, 2023, 40(2): 245-251. |
| [4] | Ai-Ping YIN, Jin-Yu SUN, Yu-Fang SHI, Ying-Jin WANG, Ming-Gen ZHAO. Linear Optical Properties and Ultrafast Nonlinear Optical Response of Trithiophenyl Chalcone Derivatives [J]. Chinese Journal of Applied Chemistry, 2022, 39(7): 1138-1146. |
| [5] | Jin-Ping SONG, Qi MA, Xiao-Min LIANG, Jian-Peng SHANG, Chuan DONG. Neodymium and Nitrogen Co‑doped Carbon Dots with High Fluorescence Quantum Yield for Detection of Sulfasalazine and Hela Cell Imaging [J]. Chinese Journal of Applied Chemistry, 2022, 39(11): 1726-1734. |
| [6] | Yu-Fang SHI, Ying-Jin WANG, Jin-Yu SUN, Ming-Gen ZHAO. Synthesis, Characterization, Photothermal Properties of Dichalcone Containing Polymethoxy Aromatic Group [J]. Chinese Journal of Applied Chemistry, 2022, 39(02): 235-240. |
| [7] | DONG Ziyue, ZHOU Xiaoxia, ZHAO Xiaohui, YE Daying, AN Yue. A Heterocyclic Aromatic Halide Small Molecule Fluorescent Probe for the Detection of 2,4,6-Trinitrophenol [J]. Chinese Journal of Applied Chemistry, 2020, 37(3): 332-339. |
| [8] | LI Di, SUN Jianqiang, XU Bingsong, CHEN Hao, ZHOU Le, HU Lei, WANG Hui. Design and Synthesis of Chalcone-Based Derivative for Sensing Cu(Ⅱ) Ion [J]. Chinese Journal of Applied Chemistry, 2020, 37(11): 1268-1275. |
| [9] | GAO Man, HE Xin, CUI Jingnan, LIU Tao, TIAN Zhenhao, HE Shengui. A Coumarin-Based Fluorescent Probe for Rapid Detection of Endogenous Formaldehyde [J]. Chinese Journal of Applied Chemistry, 2019, 36(9): 1053-1060. |
| [10] | CHENG Jinhua, JIANG Hongji. Study on Self-assembly Behaviors of an Amphiphilic Block Polymer by Terminally Grafting Tetraphenylethene-Based Aggregation-Induced Emission Active Moietys [J]. Chinese Journal of Applied Chemistry, 2019, 36(4): 440-450. |
| [11] | TANG Jing, LIN Xiaoyi, ZHOU Wenyan, HUANG Qiuying. Synthesis, Crystal Structure and Recognition Properties of One-Dimensional Luminescent Cd Coordination Polymer [J]. Chinese Journal of Applied Chemistry, 2019, 36(4): 459-464. |
| [12] | ZHU Qingwen, YU Haotian, LIU Mengqin, ZHANG Fuxing, KUANG Daizhi, TAN Yuxing, JIANG Wujiu. Syntheses, Crystal Structures and Biological Activity of Binuclear Di-2,4-dichlorobenzyltin Complexes Based on Arylformylhydrazone Ligand [J]. Chinese Journal of Applied Chemistry, 2019, 36(12): 1387-1396. |
| [13] | SUN Jinyu,WANG Guilin,SHI Yufang,LIU Chengqi,ZHAO Minggen. Synthesis, Theoretical Investigation and Ultrafast Third-Order Nonlinear Optical Response of 2-(Pyren-1-yl)-1,8-naphthyridine [J]. Chinese Journal of Applied Chemistry, 2019, 36(10): 1172-1178. |
| [14] | Feng HONG, Yueru YIN, Yupeng TIAN, Jieying WU. A Novel Third-Order Non-Linear Optical Ruthenium(Ⅱ) Complex Based on Imidazo-phenanthroline Derivative:Design, Synthesis and DNA Binding Properties [J]. Chinese Journal of Applied Chemistry, 2018, 35(7): 818-824. |
| [15] | HAN Wenjing,BAIYIN Menghe. Solvothermal Synthesis and Characterization of Cs8Mn4Sn4Se16 and [Ni(1,2-dap)3]2Cd2Sn2S8 (1,2-dap:1,2-Propanediamine)Chains [J]. Chinese Journal of Applied Chemistry, 2018, 35(6): 700-707. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||