
Chinese Journal of Applied Chemistry ›› 2026, Vol. 43 ›› Issue (3): 327-346.DOI: 10.19894/j.issn.1000-0518.250404
• Review • Previous Articles
Recent Advances in Nanozymes for Enhancing Plant Resistance to Abiotic Stress
Run-Xin HOU1,2, Na YIN1, Zhi-Yi WU3( ), Sheleg-Valery KONSTANTINOVIC4, Kravchuk-Marina ANATOLYEVN4, Ying-Hui WANG1
), Sheleg-Valery KONSTANTINOVIC4, Kravchuk-Marina ANATOLYEVN4, Ying-Hui WANG1
- 1.China-Belarus Belt and Road Joint Laboratory on Advanced Materials and Manufacturing,Changchun Institute of Applied Chemistry,Chinese Academy of Sciences,Changchun 130022,China
2.Department of Applied Chemistry and Engineering,University of Science and Technology of China,Hefei 230026,China
3.Department of Landscape Architecture,Northeast Forestry University,Harbin 150040,China
4.Department of Mechanical Engineering Technology,Belarusian National Technical University,Minsk 220070,Belarus
-
Received:2025-10-23Accepted:2025-12-11Published:2026-03-01Online:2026-03-26 -
Contact:Zhi-Yi WU -
Supported by:the National Natural Science Foundation of China(52272169)
CLC Number:
Cite this article
Run-Xin HOU, Na YIN, Zhi-Yi WU, Sheleg-Valery KONSTANTINOVIC, Kravchuk-Marina ANATOLYEVN, Ying-Hui WANG. Recent Advances in Nanozymes for Enhancing Plant Resistance to Abiotic Stress[J]. Chinese Journal of Applied Chemistry, 2026, 43(3): 327-346.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.250404
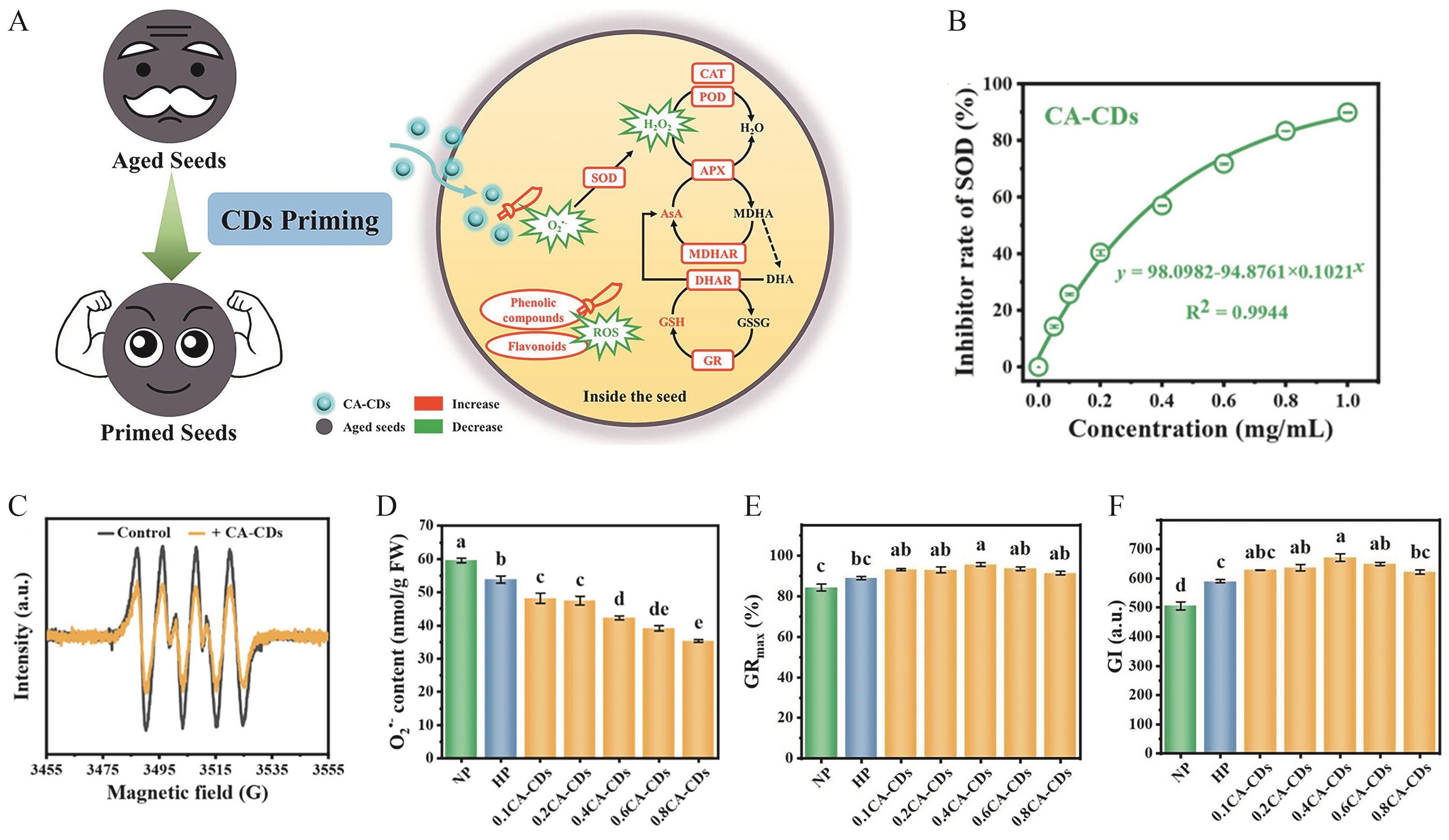
Fig.1 (A) Schematic illustration of the synergistic mechanism by which CA-CDs restore the vigor of naturally aged Chinese cabbage seeds through modulation of ROS homeostasis; (B) SOD-like activity of CA-CDs; (C) Eleetron paramagnetic resonance signal (EPR) spectra of spin adducts generated from O2·- and DMPO in the absence (control) and presence of 0.5 mg/mL CA-CDs; (D) Effect of CA-CDs on O2·- content in aged Chinese cabbage seeds; Rejuvenation effects: GRmax (E) and GI (F)[32]
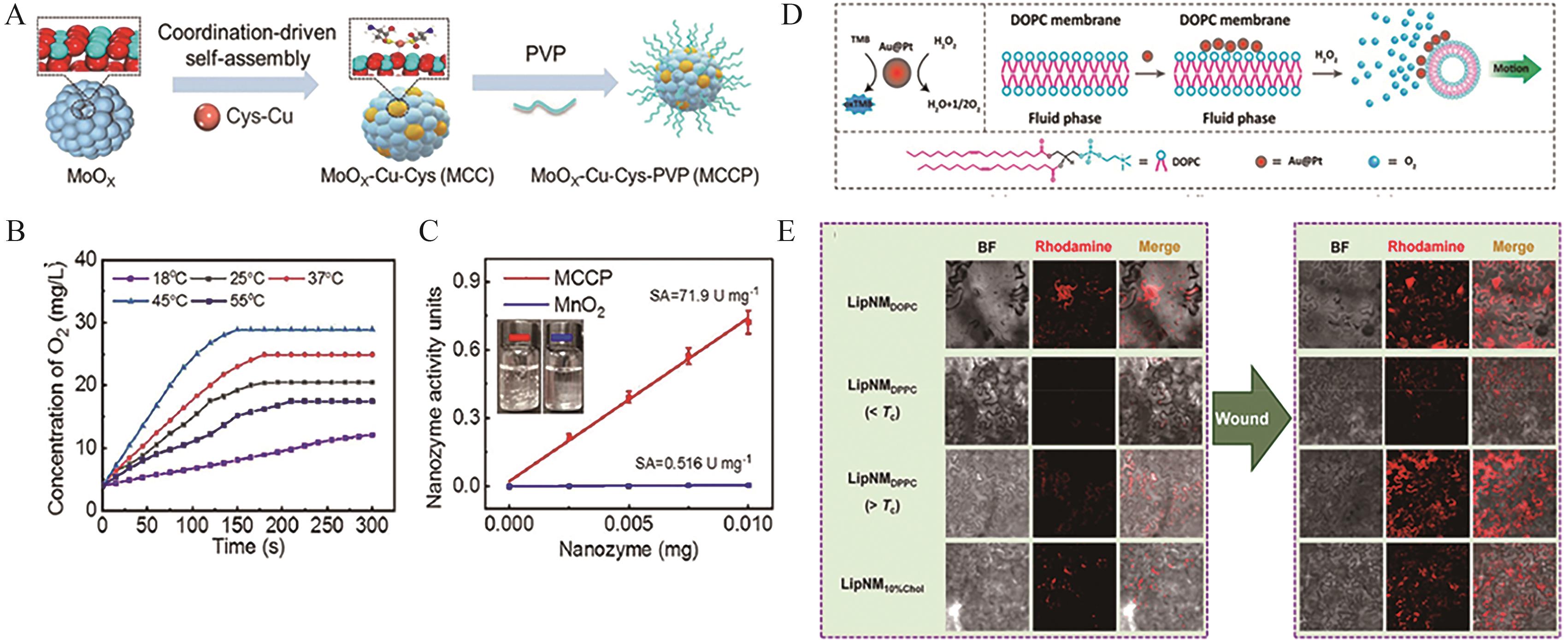
Fig.2 (A) Schematic illustration of the synthesis process of MCCP SAzymes[35]; (B) CAT-like catalytic activity of MCCP at different temperatures (ρ(MCCP)=50 μg/mL, c(H2O2)=1 mmol/L, pH=5.0)[35]; (C) Comparison of CAT-like specific activity (U/mg) between MnO2 nanozymes and MCCP at 25 ℃ (c(H2O2)=20 mmol/L, pH=5.0)[35]; (D) Schematic depiction of the heterogeneous distribution and directional movement of Au@Pt nanoparticles on liquid-phase DOPC liposomes[36]; (E) CLSM images of tobacco leaves infiltrated with LipNMDOPC, LipNMDOPC (>Tc or <Tc, and LipNM10%Chol) following wound treatment[36]
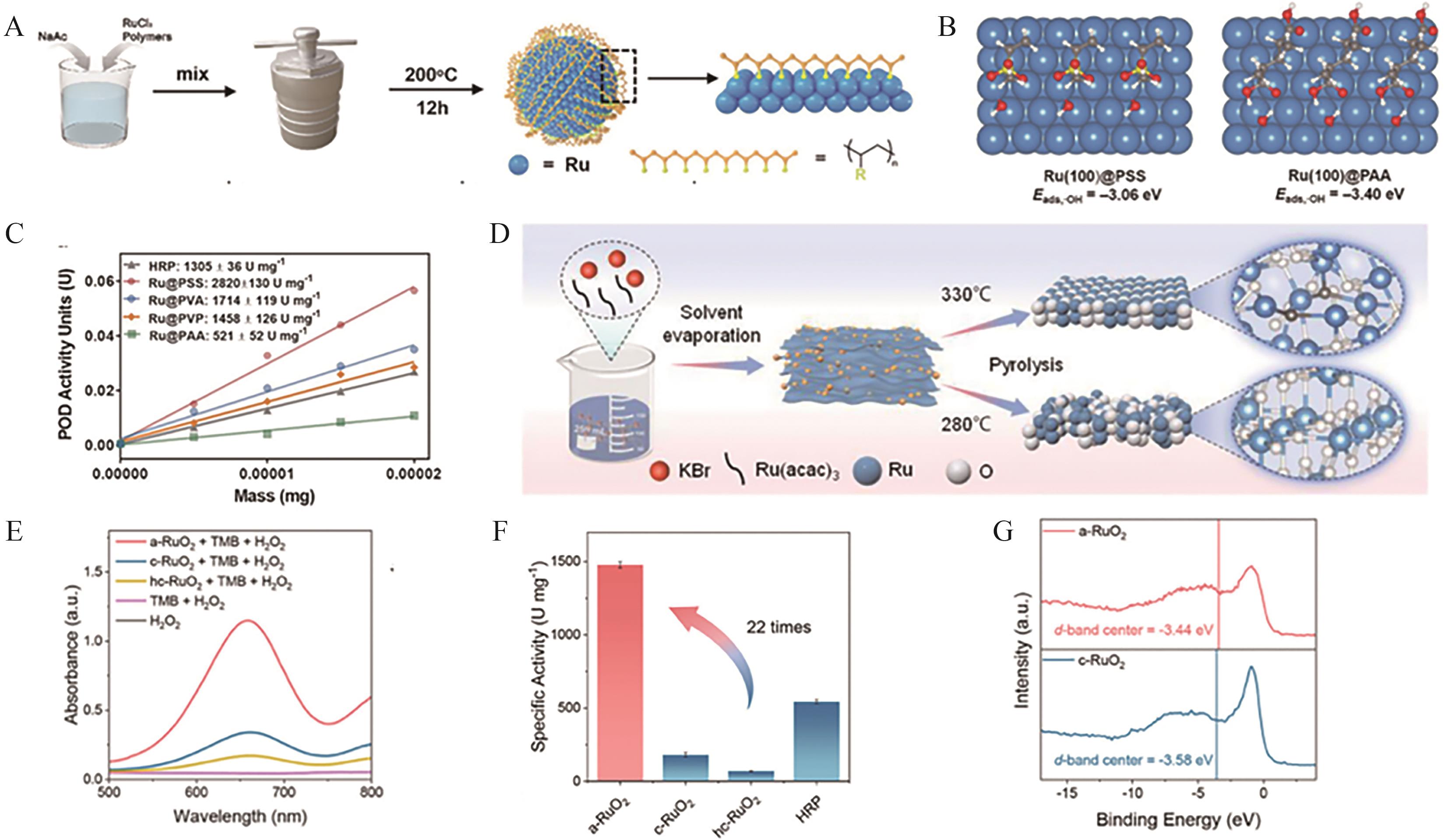
Fig.3 (A) Schematic illustration of the synthesis process of Ru-POD nanozymes with different polymer-coated ligands[42]; (B) Adsorption configurations and energy calculations of hydroxyl groups on Ru(100)@PAA and Ru(100)@PSS surfaces[42]; (C) Comparative analysis of specific activity (U/mg) between Ru-POD nanozymes and horseradish peroxidase (HRP)[42]; (D) Schematic representation of the synthesis of crystalline and amorphous RuO2; (E) UV-Vis absorption spectra of TMB and H2O2 catalyzed by RuO2 nanozymes[43]; (F) Comparison of peroxidase-like specific activity (U/mg) between RuO2 nanozymes and HRP[43]; (G) d-Band XPS valence band spectra of crystalline and amorphous RuO2 nanozymes[43]
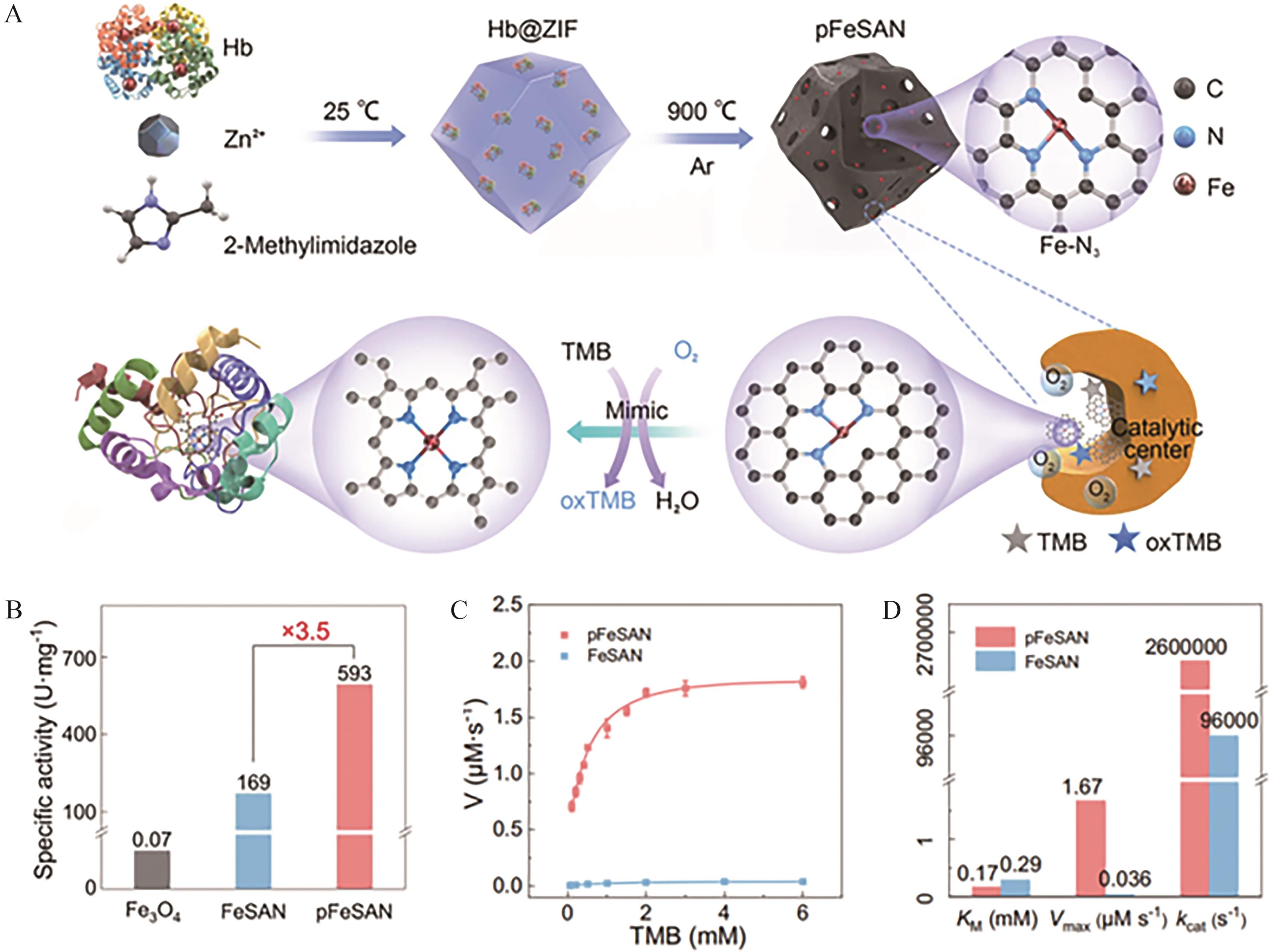
Fig.4 (A) Schematic illustration of the synthesis of porous pFeSAN with Fe-N3 sites and OXD-like activity using Hb@ZIF-8 as a precursor via a two-step method; (B) Comparative analysis of specific activity (U/mg) between pFeSAN and various OXD-like nanozymes; (C) Steady-state kinetic measurements of pFeSAN and FeSAN with TMB as substrate; (D) Kinetic comparison between pFeSAN and FeSAN[45]
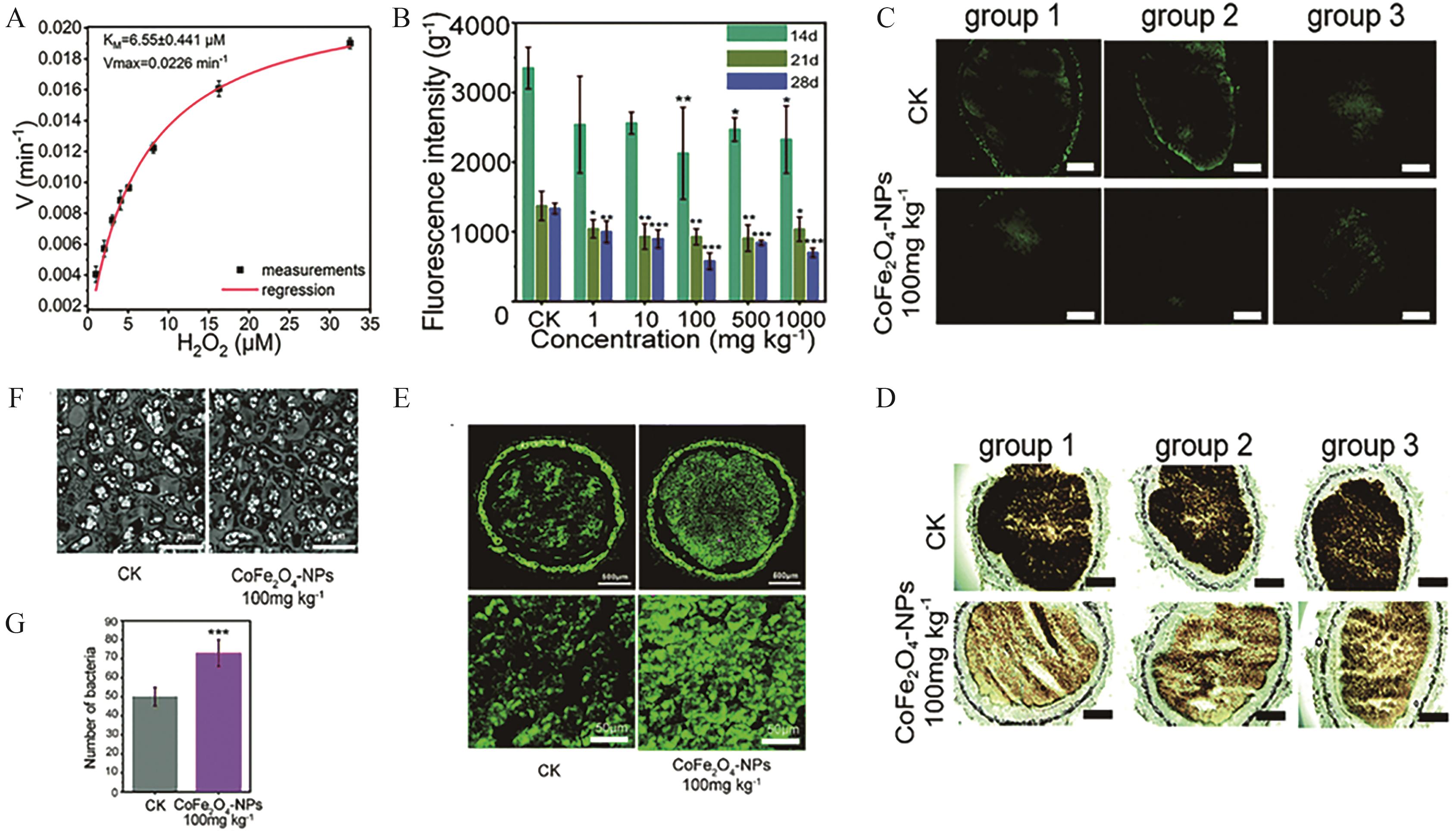
Fig.5 (A) CAT-like enzymatic kinetics of CoFe2O4 nanozyme; (B) ROS accumulation in nodules treated with CoFe2O4 nanozyme at concentrations of 1, 10, 100, 500 and 1000 mg/kg on days 14, 21 and 28 d (DCFH-DA probe); (C) In situ ROS fluorescence staining in nodules (DCFH-DA probe) (scale bar=500 μm); (D) In situ DAB staining of H2O2 in root nodules captured via optical microscopy (scale bar=500 μm); (E) Distribution of dead (red) and live (green) bacteria in root nodules stained with SYTO9/PI; (F) TEM images of rhizobial colonization in nodules; (G) Quantitative analysis of rhizobial colonization based on 100 randomly selected TEM fields[52]
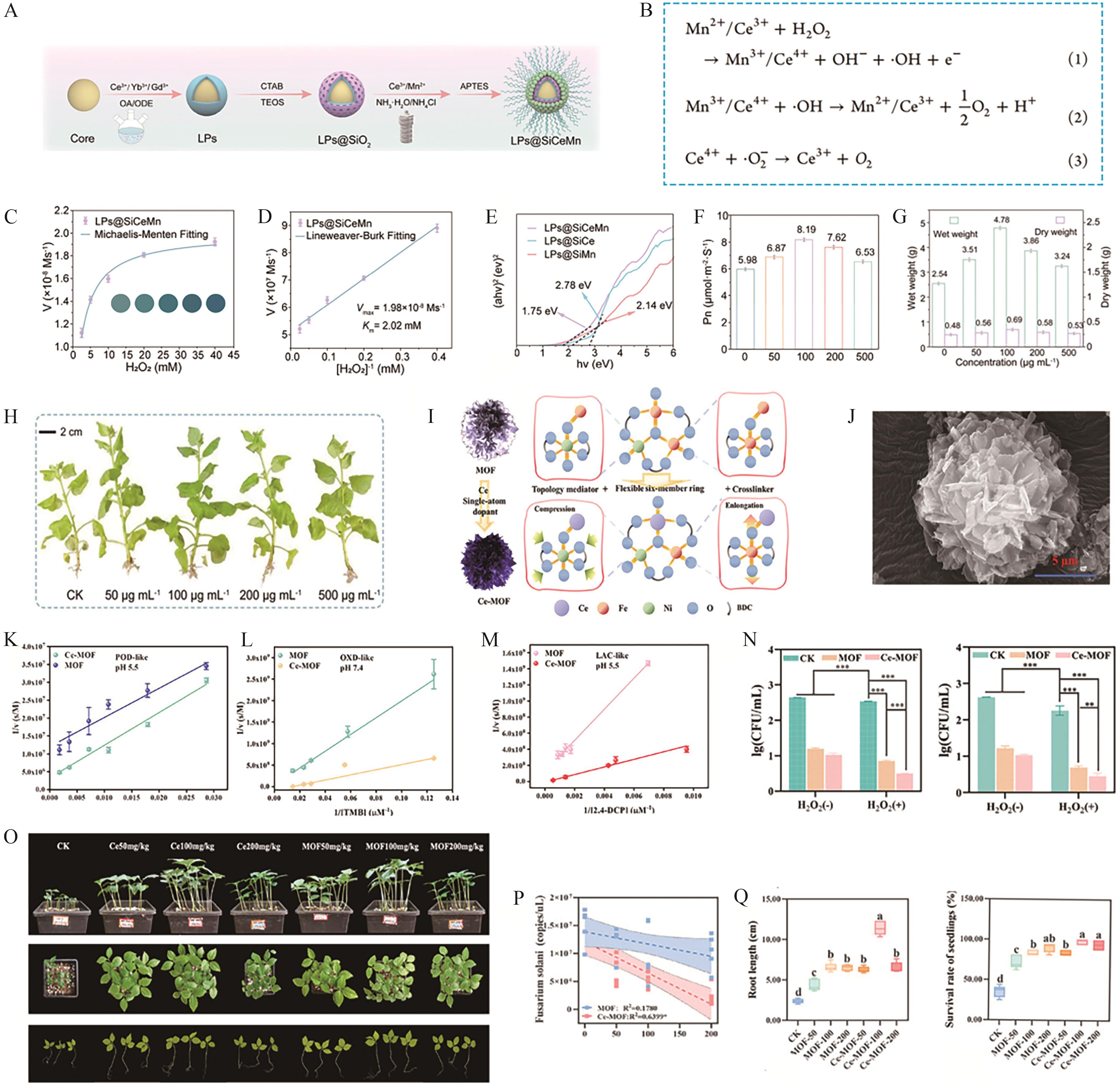
Fig.6 (A) Schematic illustration of the preparation process for LPs@SiCeMn[54]; (B) Redox cycling process (antioxidant mechanism) of LPs@SiCeMn[54]; (C, D) Michaelis-Menten kinetic analysis of LPs@SiCeMn in situ ROS fluorescence staining in nodules (DCFH-DA probe) (scale bar=500 μm)[54]; (E) Tauc plot of LPs@SiCeMn[54]; Nicotiana benthamiana treated with LPs@SiCeMn, its′ photosynthetic rate (Pn) (F), dry/wet mass (G) and Phenotype images of treated Nicotiana benthamiana (H)[54]; (I) Schematic diagram of Ce-doping effects on MOF structure[55]; (J) TEM schematic illustration of the morphology of Ce-MOF[55]; Michaelis-Menten kinetic analysis of multi-enzyme mimetic activities for Ce-MOF and pristine MOF: POD-like activity (K), OXD-like activity (L) and LAC-like activity (M)[55]; (N) In vitro antibacterial activity of Ce-MOF at pH 5.5 (left) and 7.4 (right)[55]; (O) Phenotype images of ginseng treated with Ce-MOF (planted in Fusarium solani-infected soil)[55]; (P) Linear relationship between pathogen density in rhizosphere soil and different concentrations of Ce-MOF[55]; (Q) Seedling root length (cm) and survival rate (%) under each treatment[55]
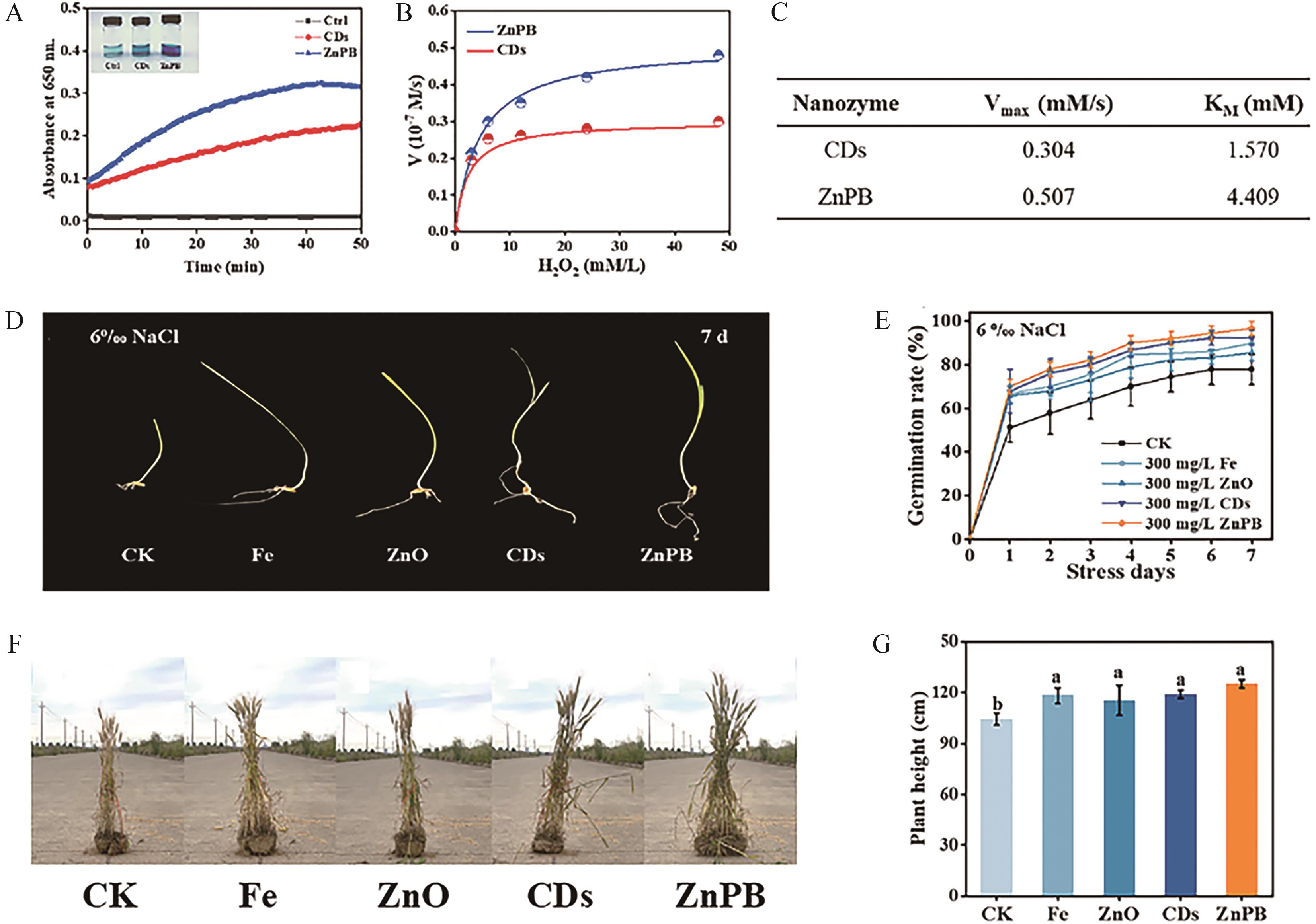
Fig.7 (A) UV-Vis absorption spectra of TMB chromogenic reaction with ZnPB and CDs nanozymes; (B) POD steady-state kinetics of ZnPB and CDs nanozymes at a concentration of 6 μg/mL (substrate: TMB, pH=4.8); (C) Kinetic parameters of POD-like enzyme activity of ZnPB and CDs (substrate: H2O2); (D) Final phenotypic images of wheat treated with four nanozyme materials after 7 days of 6% NaCl salinity stress; (E) Final germination status; (F) Phenotypic images of mature wheat at 18 weeks after sowing in saline-alkali soil, with application of Fe NPs, ZnO NPs, CDs NPs, and ZnPB nanozyme fertilizers, respectively; (G) Plant height of the final mature wheat[71]
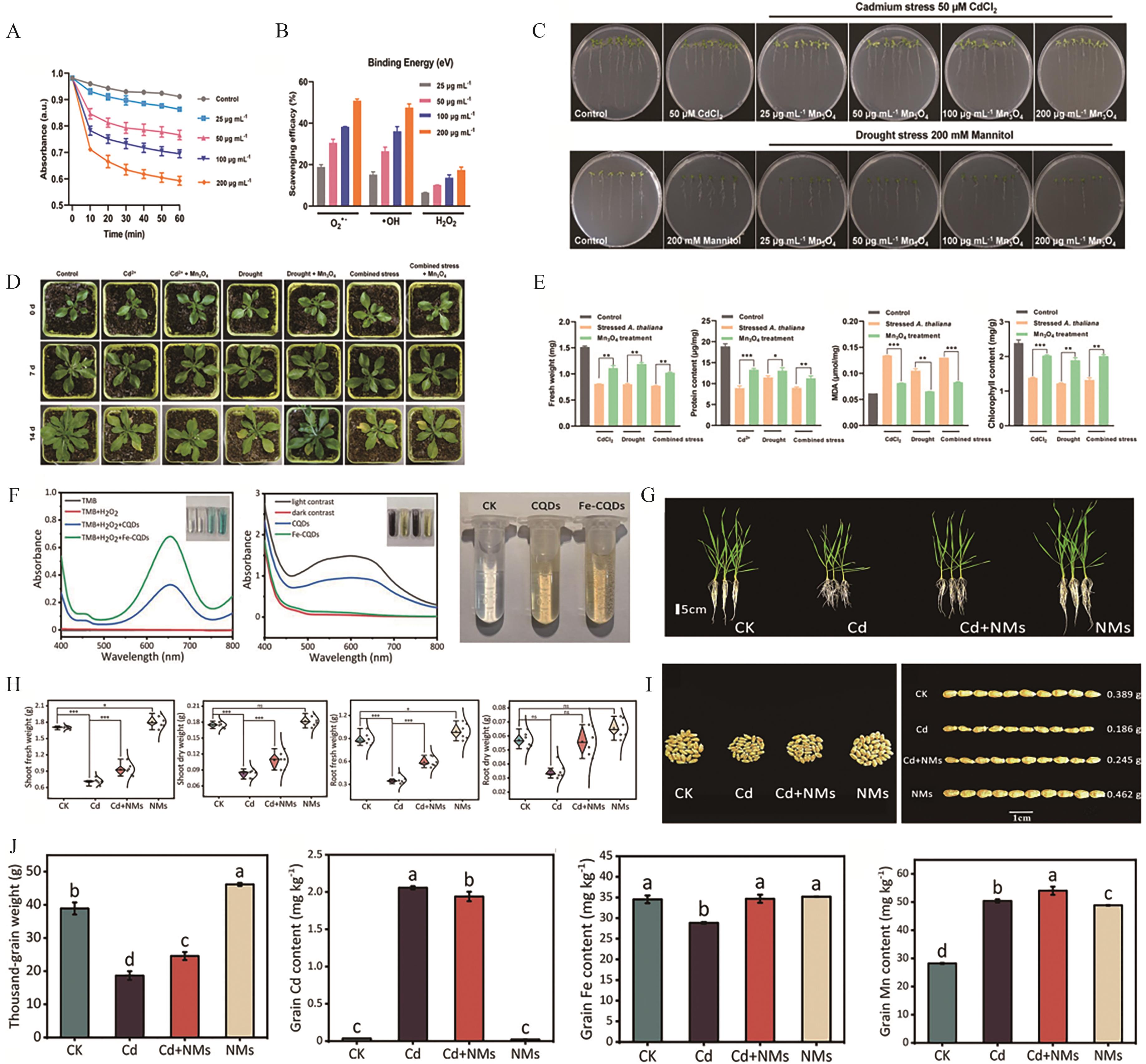
Fig.8 (A) The scavenging capacity of Mn3O4 nanozymes at different concentrations on ABTS radicals[25]; (B) The scavenging capacity of Mn3O4 at different concentrations on O2·-, ·OH, and H2O2[25]; (C) Phenotypic images showing the effects of Mn3O4 nanozyme fertilizer at different concentrations on the growth and development of Arabidopsis seedlings in MS medium containing 50 μmol/L cadmium (simulated cadmium stress) and 200 mmol/L mannitol (simulated drought stress)[25]; (D) Pot experiments of Arabidopsis treated with Mn3O4 nanozyme fertilizer under different treatment conditions[25]; (E) From left to right: fresh weight, protein content, MDA content, and chlorophyll content of Arabidopsis in the pot experiments[25]; (F) From left to right: Validation experiments of POD-like, SOD-like, and CAT-like activities of Fe-CQD[77]; (G) Phenotypic images of wheat seedlings after root application of Fe-CQD[77]; (H) From left to right: Fresh/dry weight of aerial parts and fresh/dry weight of underground parts of corresponding wheat seedlings[77]; (I) Phenotypic images of wheat grains treated with Fe-CQD and harvested in greenhouse[77]; (J) From left to right: thousand-kernel weight of wheat grains and contents of Cd, Fe and Mn trace elements in wheat grains[77]
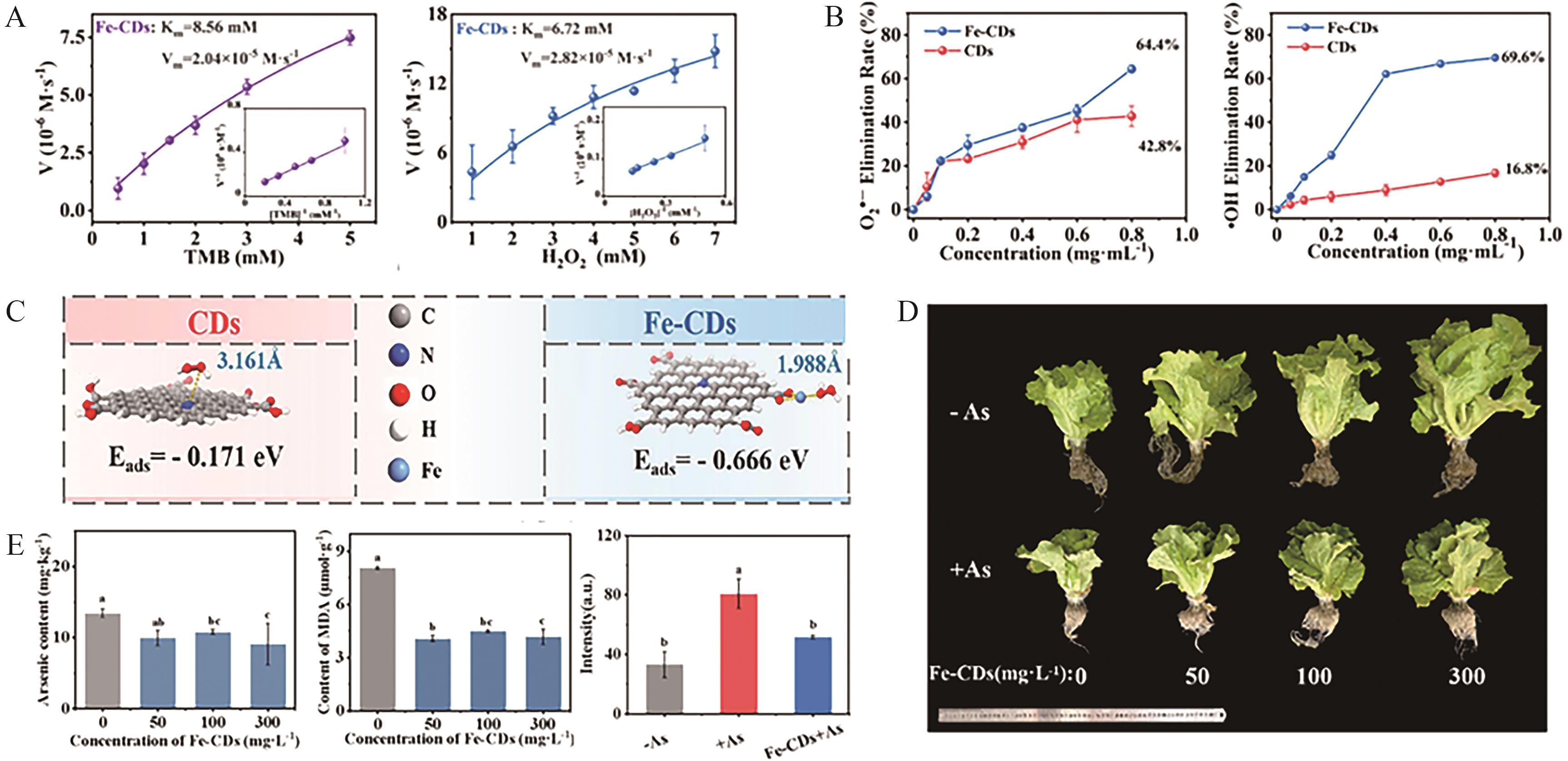
Fig.9 (A) Steady-state kinetic analysis of Fe-CDs nanozymes (with TMB and H2O2 as substrates, respectively); (B) Schematic illustration of the O2·- and ·OH scavenging activities of Fe-CDs nanozymes; (C) DFT calculation models of Fe-CDs nanozymes and CDs, along with their adsorption energies towards H2O2; (D) Morphological images of lettuce leaves following foliar application of Fe-CDs nanozymes at varying concentrations (0, 50, 100 and 300 mg/L), under both As-stressed and non-stressed conditions; (E) From left to right: arsenic content, MDA content, and average ROS fluorescence intensity in lettuce leaves, obtained using DCFH-DA ROS probe staining[82]
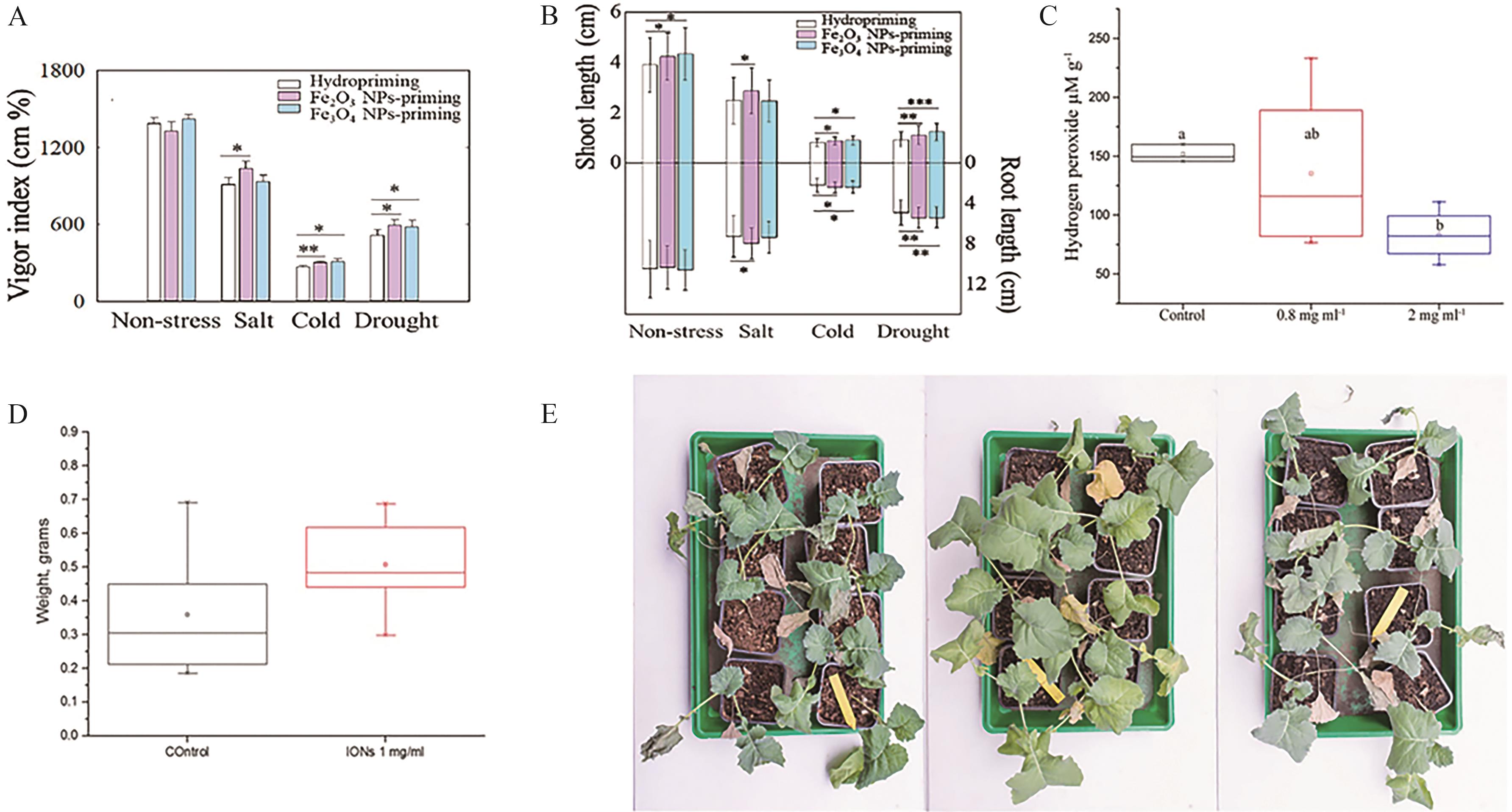
Fig.10 Effects of seed priming with Fe2O3 and Fe3O4 NPs on maize resistance to abiotic stresses (salinity, cold, and drought) during germination: Seedling vigor index (A), shoot and root length of seedlings(B)[92]; (C) Effects of ION-containing or ION-free nutrient solution on plant H2O2 content and fresh weight after 5-day drought stress[93]; (D) Impact of irrigation with or without ION on plant fresh weight[93]; (E) Phenotypic images of plants after rewatering following 5-day drought stress: control (nutrient solution), 0.8 mg/mL ION, 2 mg/mL ION[93]
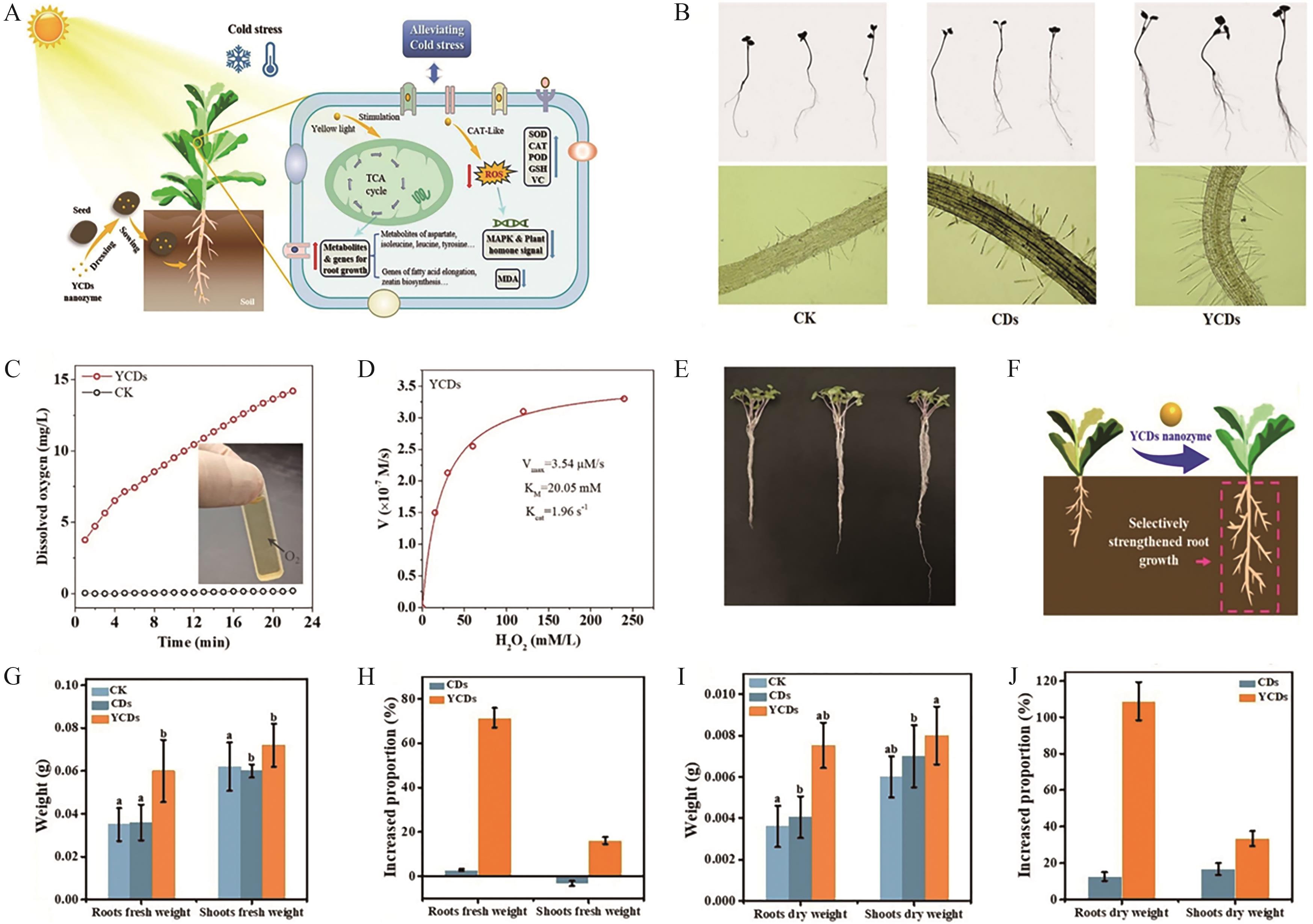
Fig.11 (A) Seed priming with ROS-generating Ag- and Fe-based NPs enhances drought and cold stress tolerance in maize seeds and seedlings; (B) Phenotypic morphology and root microstructural images of rapeseed under different treatment conditions (CK, CDs, and YCDs); (C) Validation of the CAT-like activity of YCDs through dissolved oxygen experiments; (D) Steady-state kinetic analysis of the CAT-like activity of YCDs (substrate: H2O2); (E) Phenotypic images of rapeseed plants after seed fortification with YCDs and subsequent 27-day cold stress treatment at 10 ℃; (F) Schematic model illustrating root growth enhancement in rapeseed; (G) Comparative analysis of final fresh weight; (H) Fresh weight growth rates of roots and shoots; (I) Comparative analysis of final dry weight; (J) Dry weight growth rates of roots and shoots[98]
| [1] | CASTRO B, CITTERICO M, KIMURA S, et al. Stress-induced reactive oxygen species compartmentalization, perception and signalling[J]. Nat Plants, 2021, 7(4): 403-412. |
| [2] | FICHMAN Y, MILLER G, MITTLER R. Whole-plant live imaging of reactive oxygen species[J]. Mol Plant, 2019, 12(9): 1203-1210. |
| [3] | MITTLER R. ROS are good[J]. Trends Plant Sci, 2017, 22(1): 11-19. |
| [4] | PETROV V, HILLE J, MUELLER-ROEBER B, et al. ROS-mediated abiotic stress-induced programmed cell death in plants[J]. Front Plant Sci, 2015, 6(69): 1-16. |
| [5] | ZHANG H, ZHU J, GONG Z, et al. Abiotic stress responses in plants[J]. Nat Rev Genet, 2021, 23(2): 104-119. |
| [6] | WAN N F, FU L W, DAINESE M, et al. Pesticides have negative effects on non-target organisms[J]. Nat Commun, 2025, 16(1): 1360. |
| [7] | BAHOJB N E, ZHANG H F, HE Q, et al. Alternatives for traditional pesticides: exploring the potential of metal complexes for crop management[J]. Coord Chem Rev, 2026, 546: 217061. |
| [8] | SCHULZ R, BUB S, PETSCHICK L L, et al. Applied pesticide toxicity shifts toward plants and invertebrates, even in GM crops[J]. Science, 2021, 372(6537): 81-84. |
| [9] | SU Y, ZHOU X, MENG H, et al. Cost-benefit analysis of nanofertilizers and nanopesticides emphasizes the need to improve the efficiency of nanoformulations for widescale adoption[J]. Nat Food, 2022, 3(12): 1020-1030. |
| [10] | WEI L F, LIU J Y, JIANG G B. Nanoparticle-specific transformations dictate nanoparticle effects associated with plants and implications for nanotechnology use in agriculture[J]. Nat Commun, 2024, 15(1): 7389. |
| [11] | GAO R, XU L G, SUN M Z, et al. Site-selective proteolytic cleavage of plant viruses by photoactive chiral nanoparticles[J]. Nat Catal, 2022, 5(8): 694-707. |
| [12] | WANG D J, SALEH N B, BYRO A, et al. Nano-enabled pesticides for sustainable agriculture and global food security[J]. Nat Nanotechnol, 2022, 17(4): 347-360. |
| [13] | LI C X, HAN Y P, GAO T T, et al. Insecticidal activity of metallic nanopesticides synthesized from natural resources: a review[J]. Environ Chem Lett, 2023, 21(2): 1141-1176. |
| [14] | ZHANG Z Y, LI J Q, YU C, et al. Enhanced control of tomato bacterial wilt using a triple-responsive nanopesticide with self-supplying reactive oxygen species[J]. Adv Funct Mater, 2025: 2504824. |
| [15] | XIANG S Y, WANG J, SHEN Y, et al. Manganese-enhanced nanopesticides augment plant resistance against viral infection via an untapped macroautophagy pathway[J]. Adv Funct Mater, 2025: e00538. |
| [16] | YANG J H, LIU K J, CHEN Y Z, et al. A supramolecular bactericidal material for preventing and treating plant-associated biofilms[J]. Nat Commun, 2025, 16(1): 2627. |
| [17] | ZHAO L J, BAI T H, WEI H, et al. Nanobiotechnology-based strategies for enhanced crop stress resilience[J]. Nat Food, 2022, 3(10): 829-836. |
| [18] | WU H H, LI Z. Recent advances in nano-enabled agriculture for improving plant performance[J]. Crop J, 2022, 10(1): 1-12. |
| [19] | GAO L Z, ZHUANG J, NIE L, et al. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles[J]. Nat Nanotechnol, 2007, 2(9): 577-583. |
| [20] | CHI Y L, CHENG H, SUN D W, et al. Food nanozymology: nanozyme engineering toward future food[J]. Adv Mater, 2025, 37(35): 2505552. |
| [21] | ZHANG R F, YAN X Y, GAO L Z, et al. Nanozymes expanding the boundaries of biocatalysis[J]. Nat Commun, 2025, 16(1): 6817. |
| [22] | LIN Z T, YUAN J L, NIU L X, et al. Oxidase mimicking nanozyme: classification, catalytic mechanisms and sensing applications[J]. Coord Chem Rev, 2024, 520: 216166. |
| [23] | WANG Y Y, GU X Y, ZHAO L J, et al. Nanozymes: an innovative approach to regulating heavy metal accumulation in plants and alleviating toxicity-a comprehensive review[J]. Crit Rev Environ Sci Technol, 2025, 55(10): 732-756. |
| [24] | SHEN X, YANG Z Y, DAI X Y, et al. Calcium hexacyanoferrate nanozyme enhances plant stress resistance by oxidative stress alleviation and heavy metal removal[J]. Adv Mater, 2024, 36(30): 2402745. |
| [25] | YAN Y, WU S C, KANG S M, et al. Mn3O4 nanozyme-based nanofertilizer for plant acclimation to a combination of drought and cadmium stresses[J]. Chem Eng J, 2025, 519: 165545. |
| [26] | WU H H, TITO N, GIRALDO J P. Anionic cerium oxide nanoparticles protect plant photosynthesis from abiotic stress by scavenging reactive oxygen species[J]. ACS Nano, 2017, 11(11): 11283-11297. |
| [27] | LIU Y L, XIAO Z G, CHEN F R, et al. Metallic oxide nanomaterials act as antioxidant nanozymes in higher plants: trends, meta-analysis, and prospect[J]. Sci Total Environ, 2021, 780: 146578. |
| [28] | HUANG Y W, PENG S, LIU Y F, et al. Emerging roles of nanozymes in plant and environmental sectors[J]. J Agric Food Chem, 2024, 72(42): 23008-23023. |
| [29] | COHU C M, ABDEL-GHANY S E, GOGOLIN REYNOLDS K A, et al. Copper delivery by the copper chaperone for chloroplast and cytosolic copper/zinc-superoxide dismutases: regulation and unexpected phenotypes in an Arabidopsis mutant[J]. Mol Plant, 2009, 2(6): 1336-1350. |
| [30] | JIANG J M, ZHANG N, SRIVASTAVA A K, et al. Superoxide dismutase positively regulates Cu/Zn toxicity tolerance in Sorghum bicolor by interacting with Cu chaperone for superoxide dismutase[J]. J Hazard Mater, 2024, 480: 135828. |
| [31] | CHEN F Q, ZHOU Z Y, YANG N, et al. Carbon dots based cascading nanozymes mitigate phytotoxicity in lettuces under imidacloprid stress[J]. Food Chem, 2025, 464: 141926. |
| [32] | ZHOU Z Y, LI W, ZHANG X J, et al. Carbon dot nanozymes rejuvenate aged seeds through ROS homeostasis regulation: athree-dimensional coupled seed priming strategy[J]. ACS Appl Mater Interfaces, 2025, 17(38): 53910-53923. |
| [33] | MHAMDI A, QUEVAL G, CHAOUCH S, et al. Catalase function in plants: a focus on Arabidopsis mutants as stress-mimic models[J]. J Exp Bot, 2010, 61(15): 4197-4220. |
| [34] | WANG B, XUE P, ZHANG Y, et al. OsCPK12 phosphorylates OsCATA and OsCATC to regulate H2O2 homeostasis and improve oxidative stress tolerance in rice[J]. Plant Commun, 2024, 5(3): 100780. |
| [35] | ZHOU J, XU D T, TIAN G, et al. Coordination-driven self-assembly strategy-activated Cu single-atom nanozymes for catalytic tumor-specific therapy[J]. J Am Chem Soc, 2023, 145(7): 4279-4293. |
| [36] | WANG Z F, YAN Y, LI C, et al. Fluidity-guided assembly of Au@Pt on liposomes as a catalase-powered nanomotor for effective cell uptake in cancer cells and plant leaves[J]. ACS Nano, 2022, 16(6): 9019-9030. |
| [37] | LIU J H, LV Y X, LI M, et al. Peroxidase in plant defense: novel insights for cadmium accumulation in rice (Oryza sativa L.)[J]. J Hazard Mater, 2024, 474: 134826. |
| [38] | REHMAN S, ABBAS G, SHAHID M, et al. Effect of salinity on cadmium tolerance, ionic homeostasis and oxidative stress responses in conocarpus exposed to cadmium stress: implications for phytoremediation[J]. Ecotox Environ Safe, 2019, 171: 146-153. |
| [39] | MA L M, WANG Y, CHEN Y Y, et al. Optimizing the electronic structure of iron oxides via nonmetallic dopants for enhanced peroxidase mimetic catalysis[J]. ACS Nano, 2025, 19(31): 28410-28421. |
| [40] | LYU Z Y, DING S C, DU D, et al. Recent advances in biomedical applications of 2D nanomaterials with peroxidase-like properties[J]. Adv Drug Deliv Rev, 2022, 185: 114269. |
| [41] | DONG H J, DU W, DONG J, et al. Depletable peroxidase-like activity of Fe3O4 nanozymes accompanied with separate migration of electrons and Iron ions[J]. Nat Commun, 2022, 13(1): 5365. |
| [42] | FAN H Z, ZHENG J J, XIE J Y, et al. Surface ligand engineering ruthenium nanozyme superior to horseradish peroxidase for enhanced immunoassay[J]. Adv Mater, 2024, 36(10): 2300387. |
| [43] | QIN Z R, JIAO L, HU L J, et al. Amorphous RuO2 nanozymes with an excellent catalytic efficiency superior to natural peroxidases[J]. Small, 2025, 21(24): 2502158. |
| [44] | FU Q R, WEI C, WANG M Z. Transition-metal-based nanozymes: synthesis, mechanisms of therapeutic action, and applications in cancer treatment[J]. ACS Nano, 2024, 18(19): 12049-12095. |
| [45] | CHEN D, XIA Z M, GUO Z X, et al. Bioinspired porous three-coordinated single-atom Fe nanozyme with oxidase-like activity for tumor visual identification via glutathione[J]. Nat Commun, 2023, 14(1): 7127. |
| [46] | DICENZO G C, TESI M, PFAU T, et al. Genome-scale metabolic reconstruction of the symbiosis between a leguminous plant and a nitrogen-fixing bacterium[J]. Nat Commun, 2020, 11(1): 2574. |
| [47] | WANG J, CAO X S, WANG C X, et al. Fe-based nanomaterial-induced root nodulation is modulated by flavonoids toimprove soybean (Glycine max) growth and quality[J]. ACS Nano, 2022, 16(12): 21047-21062. |
| [48] | CAO X S, YUE L, WANG C X, et al. Foliar application with iron oxide nanomaterials stimulate nitrogen fixation, yield, and nutritional quality of soybean[J]. ACS Nano, 2022, 16(1): 1170-1181. |
| [49] | SUJKOWSKA-RYBKOWSKA M. Reactive oxygen species production and antioxidative defense in pea (Pisum sativum L.) root nodules after short-term aluminum treatment[J]. Acta Physiol Plant, 2012, 34(4): 1387-1400. |
| [50] | LI M S, ZHANG P, GUO Z L, et al. Molybdenum nanofertilizer boosts biological nitrogen fixation and yield of soybean through delaying nodule senescence and nutrition enhancement[J]. ACS Nano, 2023, 17(15): 14761-14774. |
| [51] | LI M, ZHANG P, GUO Z, et al. Dynamic transformation of nano-MoS2 in a soil-plant system empowers its multifunctionality on soybean growth[J]. Environ Sci Technol, 2024, 58(2): 1211-1222. |
| [52] | MA J, SONG Z Y, YANG J H, et al. Cobalt ferrite nanozyme for efficient symbiotic nitrogen fixation via regulating reactive oxygen metabolism[J]. Environ Sci-Nano, 2021, 8(1): 188-203. |
| [53] | CHEN Z Q, YE Y H, PENG T, et al. Iron-single sites confined by graphene lattice for ammonia synthesis under mild conditions[J]. ACS Catal, 2023, 13(21): 14385-14394. |
| [54] | HUANG Y R, WANG Q, LI C S, et al. Sunlight broad spectrum-capturing nanophotofertilizer for plant growth multiregulation and second near-infrared imaging[J]. ACS Nano, 2025, 19(37): 33632-33643. |
| [55] | LI Q, CHA Y Y, ZHAN Y, et al. Atomic ce-induced adaptive synergism for self-optimized multi-enzymatic nanozyme design for soil amendment[J]. Small, 2025, 21(39): 2503939. |
| [56] | MITTLER R, ZANDALINAS S I, FICHMAN Y, et al. Reactive oxygen species signalling in plant stress responses[J]. Nat Rev Mol Cell Biol, 2022, 23(10): 663-679. |
| [57] | WASZCZAK C, CARMODY M, KANGASJÄRVI J. Reactive oxygen species in plant signaling[J]. Annu Rev Plant Biol, 2018, 69: 209-236. |
| [58] | SINGH V P, JAISWAL S, WANG Y Y, et al. Evolution of reactive oxygen species cellular targets for plant development[J]. Trends Plant Sci, 2024, 29(8): 865-877. |
| [59] | LIU L L, GUEGUEN-CHAIGNON V, GONÇALVES I R, et al. A secreted metal-binding protein protects necrotrophic phytopathogens from reactive oxygen species[J]. Nat Commun, 2019, 10(1): 4853. |
| [60] | SHEN X, YANG Z Y, DAI X Y, et al. Calcium hexacyanoferrate nanozyme enhances plant stress resistance by oxidative stress alleviation and heavy metal removal[J]. Adv Mater, 2024, 36(30): 2402745. |
| [61] | DING Z H, LIU L W, SONG Q Q, et al. A redox-modulating coating with multi-enzyme activity on engineered nanoparticles: an effective strategy for enhancing the control of plant bacterial diseases[J]. Chem Eng J, 2025, 516: 163990. |
| [62] | YU J, HE J L, LIU Y H, et al. ZIF-8-derived nanozyme loaded with botanical fungicides as an eco-friendly nanopesticide for microenvironment-enhanced synergistic control of ginseng seedling blight[J]. J Agric Food Chem, 2025, 73(17): 10144-10156. |
| [63] | CHEN K Y, HAO M, YUAN T, et al. Biosynthesized Fe-C-dots nanozymes modulate growth, physiological and phytochemical peculiarity to improve saline-alkaline stress tolerance in wheat[J]. Plant Physiol Biochem, 2025, 222: 109777. |
| [64] | YU Z P, DUAN X B, LUO L, et al. How plant hormones mediate salt stress responses[J]. Trends Plant Sci,2020, 25(11): 1117-1130. |
| [65] | PERRI S, MOLINI A, HEDIN L O, et al. Contrasting effects of aridity and seasonality on global salinization[J]. Nat Geosci, 2022, 15(5): 375-381. |
| [66] | ZELM E V, ZHANG Y X, TESTERINK C. Salt tolerance mechanisms of plants[J]. Annu Rev Plant Biol, 2020, 71: 403-433. |
| [67] | WAADT R, SELLER C A, HSU P K, et al. Plant hormone regulation of abiotic stress responses[J]. Nat Rev Mol Cell Biol, 2022, 23(10): 680-694. |
| [68] | SZYMAŃSKA K P, POLKOWSKA-KOWALCZYK L, LICHOCKA M, et al. SNF1-related protein kinases SnRK2.4 and SnRK2.10 modulate ROS homeostasis in plant response to salt stress[J]. International Int J Mol Sci, 2019, 20(1): 143. |
| [69] | KAPPACHERY S, ALHOSANI M, KHAN T A, et al. Modulation of antioxidant defense and PSII components by exogenously applied acetate mitigates salinity stress in Avena sativa[J]. Sci Rep, 2024, 14(1): 620. |
| [70] | MA X X, ZENG X Y, HUANG Y R, et al. Visualizing plant salt stress with a NaCl-responsive fluorescent probe[J]. Nat Protoc, 2025, 20(4): 902-933. |
| [71] | XU W L, FENG Y C, DING Z X, et al. Peroxidase like Zn doped prussian blue facilitates salinity tolerance in winter wheat through seed dressing 11 electronic supplementary information (ESI) available[J]. Int J Biol Macromol, 2024, 267: 131477. |
| [72] | FENG Z, JI S Y, PING J F, et al. Recent advances in metabolomics for studying heavy metal stress in plants[J]. Trac-Trends Anal Chem, 2021, 143: 116402. |
| [73] | GHUGE S A, NIKALJE G C, KADAM U S, et al. Comprehensive mechanisms of heavy metal toxicity in plants, detoxification, and remediation[J]. J Hazard Mater, 2023, 450: 131039. |
| [74] | XU Z H, ZHANG T T, XU Z H, et al. Research progress and prospects of nanozymes in alleviating abiotic stress of crops[J]. J Agric Food Chem, 2025, 73(15): 8694-8714. |
| [75] | YU Y, FOTOPOULOS V, ZHOU K J, et al. The role of gasotransmitter hydrogen sulfide in plant cadmium stress responses[J]. Trends Plant Sci, 2025, 30(1): 35-53. |
| [76] | EL RASAFI T, OUKARROUM A, HADDIOUI A, et al. Cadmium stress in plants: a critical review of the effects, mechanisms, and tolerance strategies[J]. Crit Rev Environ Sci Technol, 2022, 52(5): 675-726. |
| [77] | LU Z W, LI Y, CHEN K Y, et al. Multi-activity ferruginated carbon quantum dots nanozyme improves wheat seedling growth and Cd tolerance[J]. Crop J, 2025, 13(2): 510-523. |
| [78] | SUN D, ZHANG X, YIN Z Y, et al. As-hyperaccumulator Pteris vittata and non-hyperaccumulator Pteris ensiformis under low As-exposure: transcriptome analysis and implication for As hyperaccumulation[J]. J Hazard Mater, 2023, 458: 132034. |
| [79] | LEW T T S, PARK M, CUI J Q, et al. Plant nanobionic sensors for Arsenic detection[J]. Adv Mater, 2021, 33(1): 2005683. |
| [80] | ZULFIQAR F, ASHRAF M. Antioxidants as modulators of arsenic-induced oxidative stress tolerance in plants: an overview[J]. J Hazard Mater, 2022, 427: 127891. |
| [81] | ZHANG J, HAMZA A, XIE Z M, et al. Arsenic transport and interaction with plant metabolism: clues for improving agricultural productivity and food safety[J]. Environ Pollut, 2021, 290: 117987. |
| [82] | XIE M H, LI F Y, LI Y Y, et al. Iron-doped carbon dots nanozyme scavenged reactive oxygen species system for inhibiting effectively the uptake of arsenic in lettuce[J]. Chem Eng J, 2025, 506: 159956. |
| [83] | MUNNÉ-BOSCH S, VILLADANGOS S. Cheap, cost-effective, and quick stress biomarkers for drought stress detection and monitoring in plants[J]. Trends Plant Sci, 2023, 28(5): 527-536. |
| [84] | GUPTA A, RICO-MEDINA A, CAÑO-DELGADO A I. The physiology of plant responses to drought[J]. Science, 2020, 368(6488): 266-269. |
| [85] | CHEN Q, CAO X F, NIE X K, et al. Alleviation role of functional carbon nanodots for tomato growth and soil environment under drought stress[J]. J Hazard Mater, 2022, 423: 127260. |
| [86] | LU Z W, ZHAO A, CHEN K, et al. The alleviating effect of straw-derived nitrogen-doped SiO2 nanozymes for wheat growth under drought stress[J]. ACS Mater Lett, 2025, 7: 3652-3659. |
| [87] | NILE S H, THIRUVENGADAM M, WANG Y, et al. Nano-priming as emerging seed priming technology for sustainable agriculture—recent developments and future perspectives[J]. J Nanobiotechnol, 2022, 20(1): 254. |
| [88] | GUHA T, DAS H, MUKHERJEE A, et al. Elucidating ROS signaling networks and physiological changes involved in nanoscale zero valent iron primed rice seed germination sensu stricto[J]. Free Radic Biol Med, 2021, 171: 11-25. |
| [89] | PAGANO L, MARMIROLI M, VILLANI M, et al. Engineered nanomaterial exposure affects organelle genetic material replication in Arabidopsis thaliana[J]. ACS Nano, 2022, 16(2): 2249-2260. |
| [90] | LIN J J, SUN M Q, SU B L, et al. Immobilization of cadmium in polluted soils by phytogenic iron oxide nanoparticles[J]. Sci Total Environ, 2019, 659: 491-498. |
| [91] | BHATIA A, KHATRI A, YADAV M, et al. Potential of iron oxide nanoparticles in enhancing growth and development of plants: a review[J]. Physiologic Mol Plant Pathol, 2025, 139: 102746. |
| [92] | CHEN S, LIU H L, YANGZONG Z X, et al. Seed priming with reactive oxygen species-generating nanoparticles enhanced maize tolerance to multiple abiotic stresses[J]. Environ Sci Technol, 2023, 57(48): 19932-19941. |
| [93] | PALMQVIST N G M, SEISENBAEVA G A, SVEDLINDH P, et al. Maghemite nanoparticles Acts as nanozymes, improving growth and abiotic stress tolerance in Brassica napus[J]. Nanoscale Res Lett, 2017, 12(1): 631. |
| [94] | KIDOKORO S, SHINOZAKI K, YAMAGUCHI-SHINOZAKI K. Transcriptional regulatory network of plant cold-stress responses[J]. Trends Plant Sci, 2022, 27(9): 922-935. |
| [95] | LI W X, WEN Y S, QUAN J J, et al. Regulation of jasmonic acid signalling in tomato cold stress response: insights into the MYB15-LOXD and MYB15-MYC2-LOXD regulatory modules[J]. Plant Biotechnol J, 2025, 23(10): 4246-4260. |
| [96] | DING Y L, SHI Y T, YANG S H. Advances and challenges in uncovering cold tolerance regulatory mechanisms in plants[J]. New Phyto, 2019, 222(4): 1690-1704. |
| [97] | CHEN S, PAN Z, ZHAO W, et al. Engineering climate-resilient rice using a nanobiostimulant-based “stress training” strategy[J]. ACS Nano, 2023, 17(11): 10760-10773. |
| [98] | DING Z, XU W, FENG Y, et al. Selective photosynthetically active nanozymes alleviate crops cold stress via exclusively enhancing root proliferation[J]. Chem Eng J, 2025, 508: 160428. |
| [1] | Shuang LIU, Si-Ying TENG, Peng HUI, Ya-Bin SUN. Research Progress on Nanozymes in the Treatment of Ophthalmic Diseases [J]. Chinese Journal of Applied Chemistry, 2024, 41(12): 1679-1696. |
| [2] | Jian-Tian LU, Jin-Hui ZOU, Bo-Lin ZHAO, Yu-Wei ZHANG. Research Progress on the Application of Inorganic Nanoparticle Enzyme in the Field of Analytical Sensing [J]. Chinese Journal of Applied Chemistry, 2024, 41(1): 60-86. |
| [3] | LI Junrong,SHEN Aiguo,HU Jiming. Research Progress of Nanozymes and Its Application in Analysis [J]. Chinese Journal of Applied Chemistry, 2016, 33(11): 1245-1252. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||