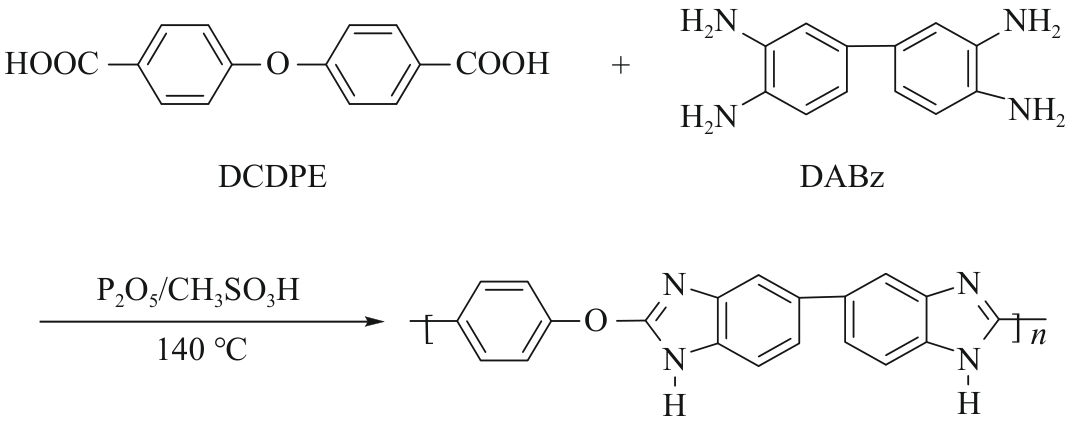
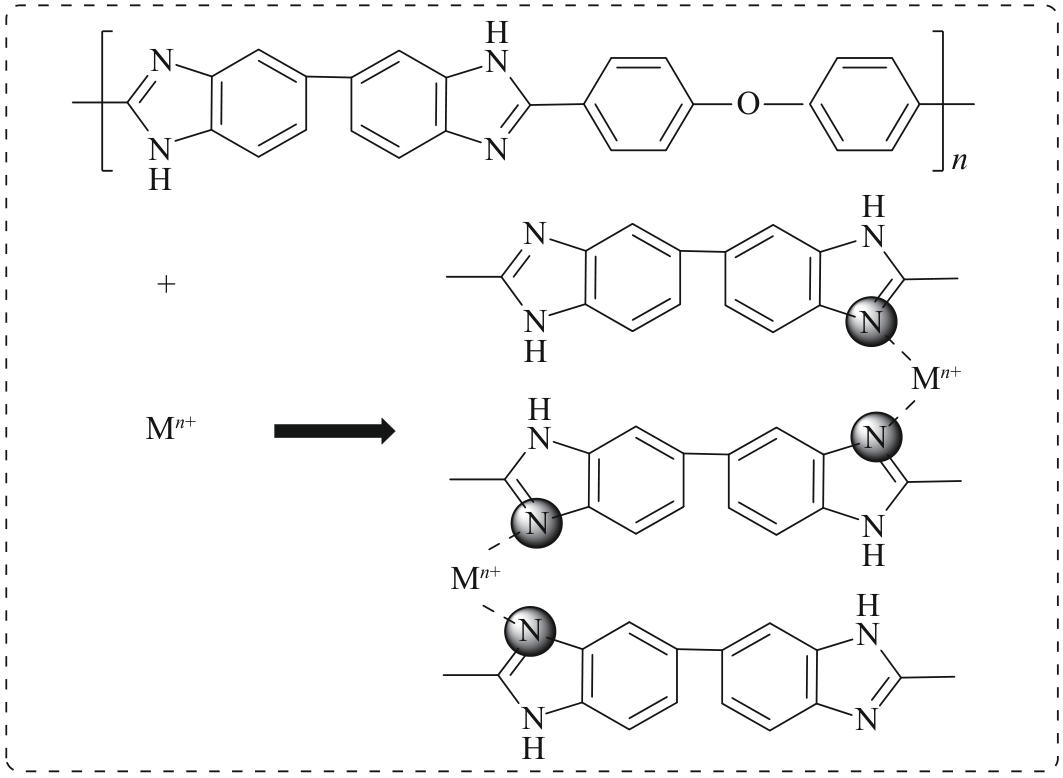
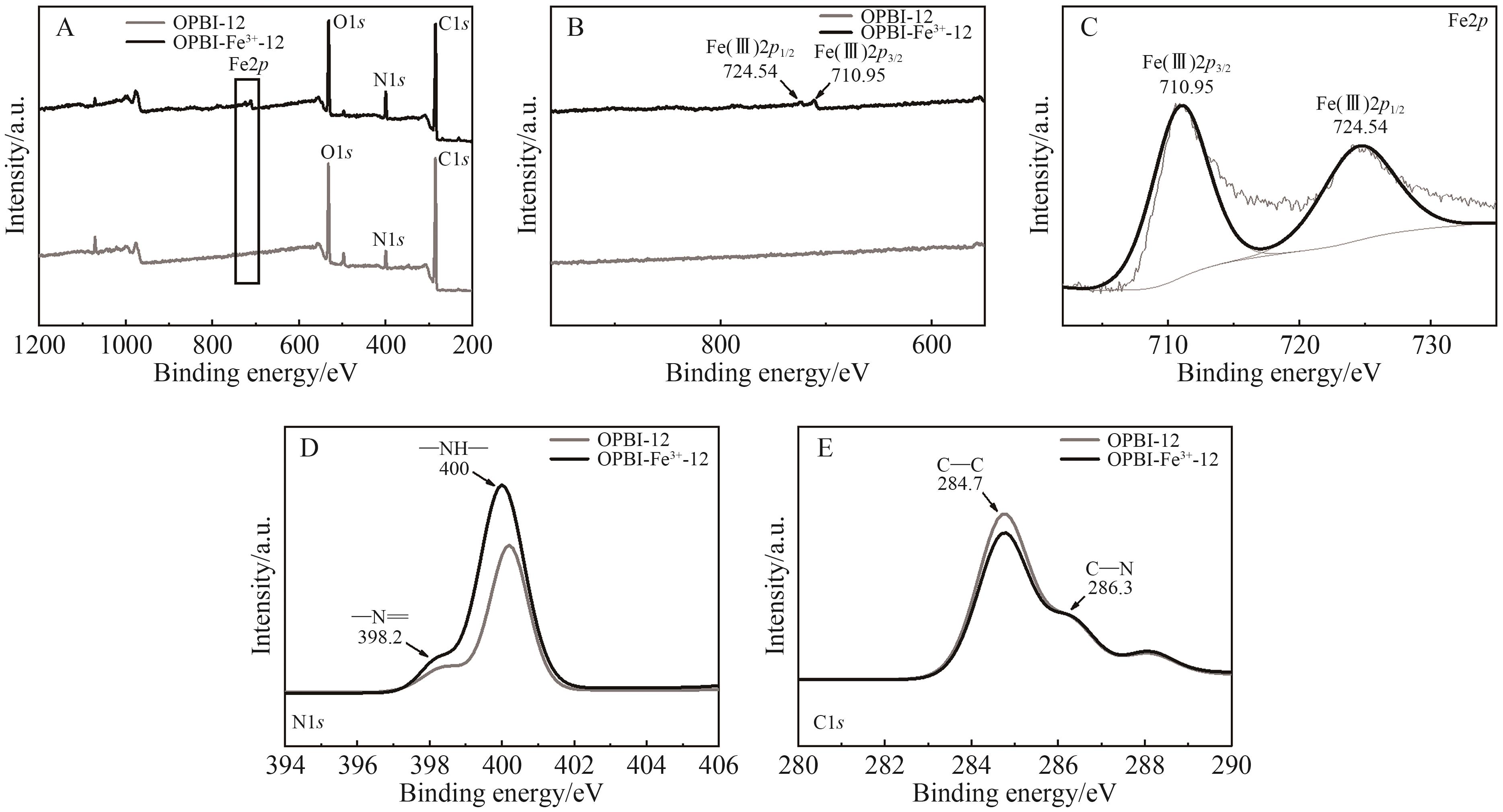
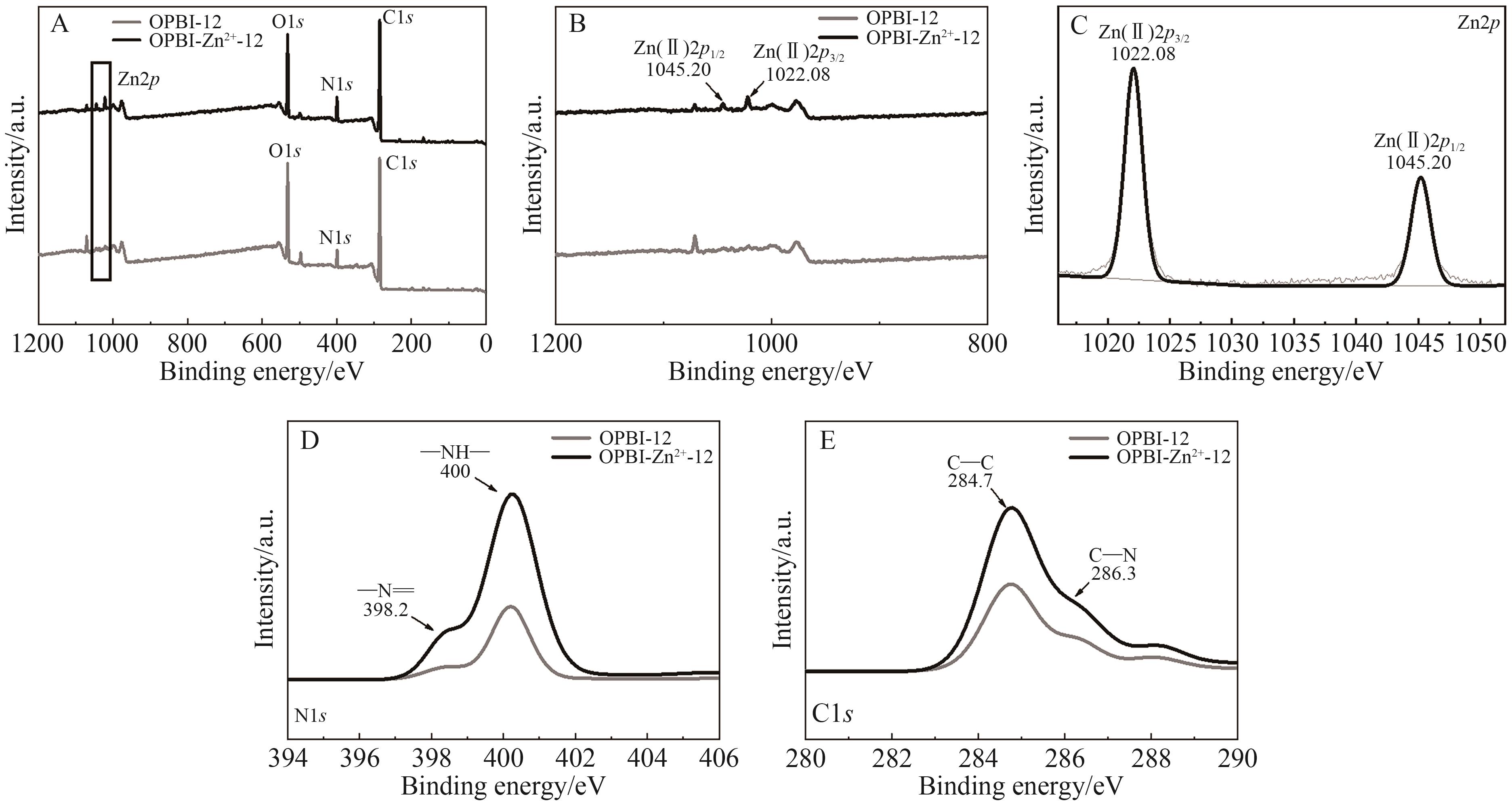
Chinese Journal of Applied Chemistry ›› 2026, Vol. 43 ›› Issue (2): 259-274.DOI: 10.19894/j.issn.1000-0518.250107
• Full Papers • Previous Articles Next Articles
Preparation and Properties of Modified Polybenzimidazole Solvent Resistant Nanofiltration Membranes
Li-Na JIAO, Chen-Yu WANG, Yu-Ting JIANG, Miao-Miao HE, Dong-Ju CHEN( )
)
- School of Chemistry and Chemical Engineering,Liaoning Normal University,Dalian 116029,China
-
Received:2025-03-14Accepted:2025-06-24Published:2026-02-01Online:2026-03-06 -
Contact:Dong-Ju CHEN -
About author:Dongjuchen_1978@lnnu.edu.cn
-
Supported by:the National Natural Science Foundation of China(22075121);the Open Project of State Key Laboratory of Fine Chemicals, Dalian University of Technology(KF2205)
CLC Number:
Cite this article
Li-Na JIAO, Chen-Yu WANG, Yu-Ting JIANG, Miao-Miao HE, Dong-Ju CHEN. Preparation and Properties of Modified Polybenzimidazole Solvent Resistant Nanofiltration Membranes[J]. Chinese Journal of Applied Chemistry, 2026, 43(2): 259-274.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.250107
| w(polymer)/% | 12% | 15% | 17.5% |
|---|---|---|---|
| Pristine membranes | OPBI-12 | OPBI-15 | OPBI-17.5 |
| FeCl3·6H2O crosslinked membranes | OPBI-Fe3+-12 | OPBI-Fe3+-15 | OPBI-Fe3+-17.5 |
| ZnSO4·7H2O crosslinked membranes | OPBI-Zn2+-12 | OPBI-Zn2+-15 | OPBI-Zn2+-17.5 |
Table 1 Naming of original and crosslinked membranes
| w(polymer)/% | 12% | 15% | 17.5% |
|---|---|---|---|
| Pristine membranes | OPBI-12 | OPBI-15 | OPBI-17.5 |
| FeCl3·6H2O crosslinked membranes | OPBI-Fe3+-12 | OPBI-Fe3+-15 | OPBI-Fe3+-17.5 |
| ZnSO4·7H2O crosslinked membranes | OPBI-Zn2+-12 | OPBI-Zn2+-15 | OPBI-Zn2+-17.5 |
| Solvent | Relative molecular mass/(g·mol-1) | Molar volume/(m3·mol-1) | Hansen solubility parameters/(MPa0.5) | η/(mPa·s) | ρ/(g·cm-3) |
|---|---|---|---|---|---|
| n-Heptane | 100.2 | 144.0 | 15.3 | 0.400 | 0.683 |
| n-Hexane | 86.2 | 127.5 | 14.9 | 0.326 | 0.659 |
| DCM | 84.9 | 67.8 | 20.3 | 0.430 | 1.325 |
| EA | 88.1 | 98.0 | 18.1 | 0.450 | 0.902 |
| MIBK | 100.1 | 125.0 | 16 | 0.542 | 0.796 |
| THF | 72.1 | 79.7 | 19.4 | 0.550 | 0.890 |
| IPA | 60.1 | 75.9 | 23.6 | 1.830 | 0.785 |
| NMP | 99.1 | 98.8 | 23.0 | 1.650 | 1.020 |
| Acetone | 58.1 | 75.1 | 19.9 | 0.290 | 0.791 |
| DMF | 73.1 | 77.0 | 24.9 | 0.802 | 0.947 |
| DMAc | 87.1 | 96.2 | 22.8 | 0.920 | 0.940 |
Table 2 Properties of solvents
| Solvent | Relative molecular mass/(g·mol-1) | Molar volume/(m3·mol-1) | Hansen solubility parameters/(MPa0.5) | η/(mPa·s) | ρ/(g·cm-3) |
|---|---|---|---|---|---|
| n-Heptane | 100.2 | 144.0 | 15.3 | 0.400 | 0.683 |
| n-Hexane | 86.2 | 127.5 | 14.9 | 0.326 | 0.659 |
| DCM | 84.9 | 67.8 | 20.3 | 0.430 | 1.325 |
| EA | 88.1 | 98.0 | 18.1 | 0.450 | 0.902 |
| MIBK | 100.1 | 125.0 | 16 | 0.542 | 0.796 |
| THF | 72.1 | 79.7 | 19.4 | 0.550 | 0.890 |
| IPA | 60.1 | 75.9 | 23.6 | 1.830 | 0.785 |
| NMP | 99.1 | 98.8 | 23.0 | 1.650 | 1.020 |
| Acetone | 58.1 | 75.1 | 19.9 | 0.290 | 0.791 |
| DMF | 73.1 | 77.0 | 24.9 | 0.802 | 0.947 |
| DMAc | 87.1 | 96.2 | 22.8 | 0.920 | 0.940 |
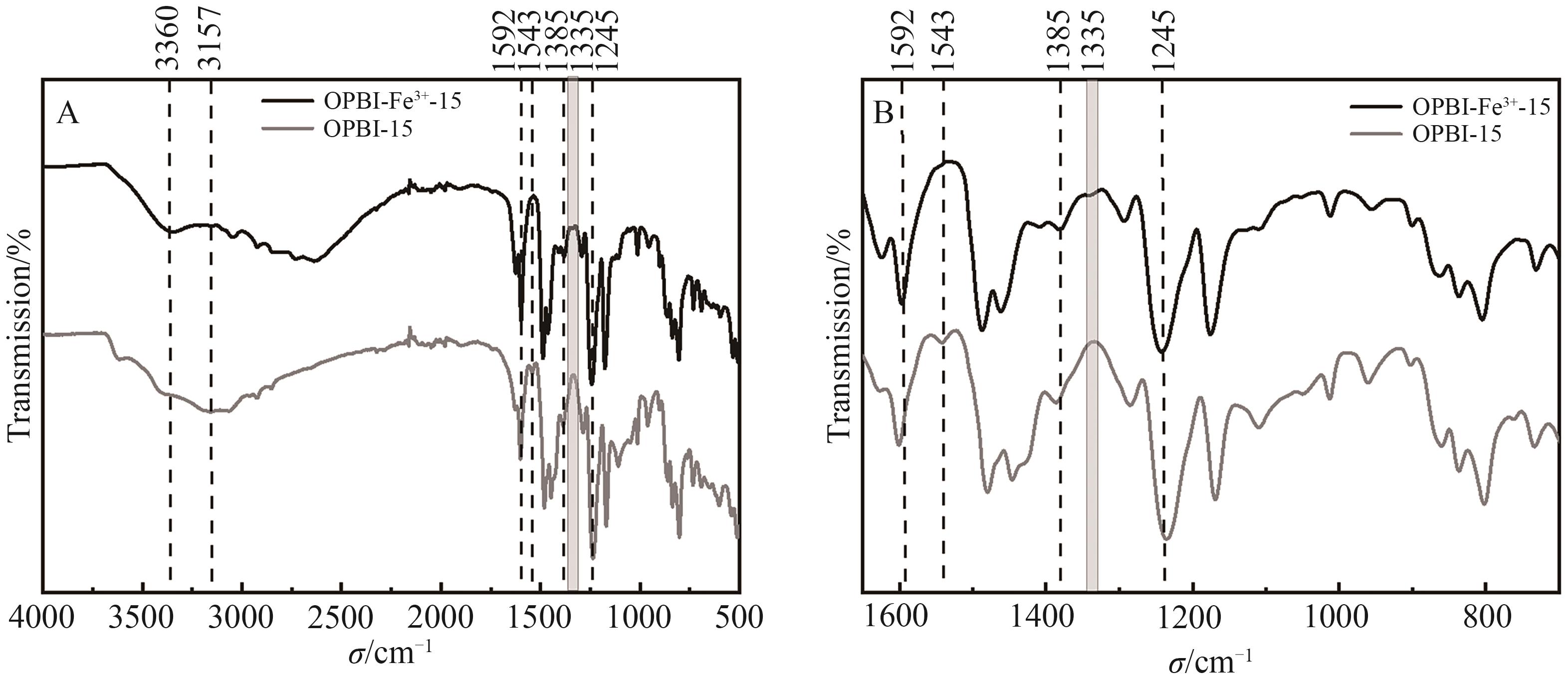
Fig.1 (A) FT-IR spectra of FeCl3·6H2O crosslinked 15% OPBI membrane in range of 500~4000 cm-1; (B) 700~1650 cm-1 FT-IR spectra of FeCl3·6H2O crosslinked 15% OPBI membrane in range of 700~1650 cm-1
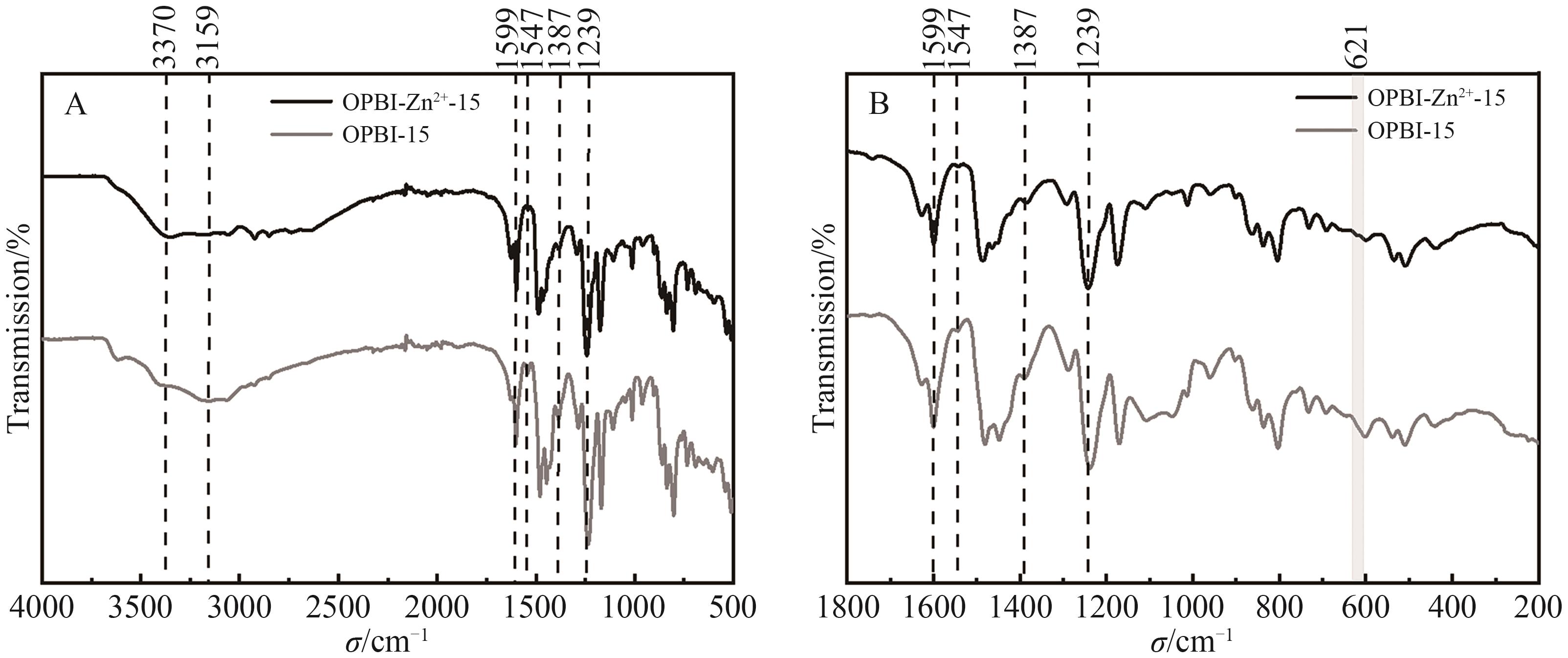
Fig.2 (A) FT-IR spectra of ZnSO4·7H2O crosslinked 15% OPBI membrane in range of 500~4000 cm-1; (B) FT-IR spectra of ZnSO4·7H2O crosslinked 15% OPBI membrane in range of 200~1800 cm-1
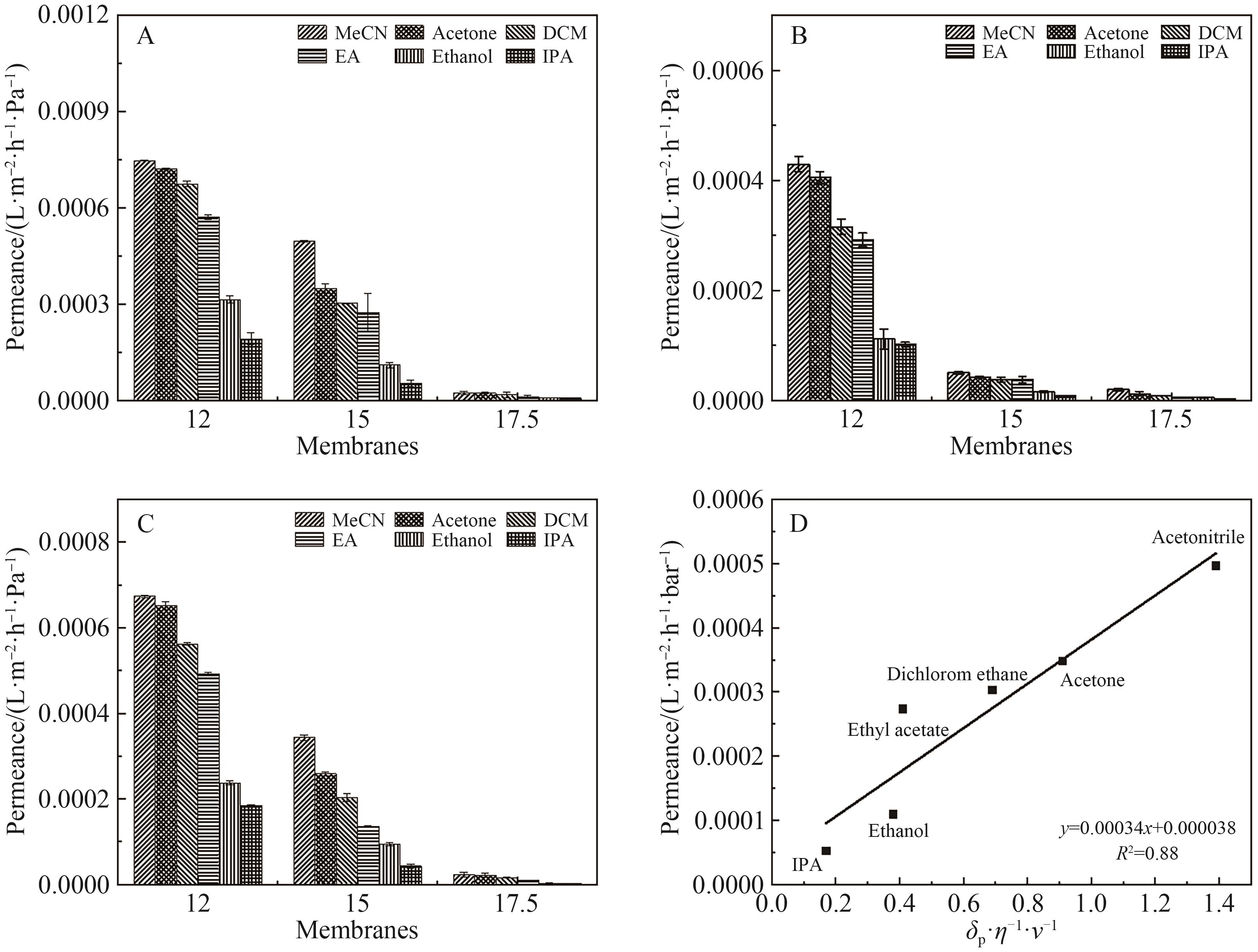
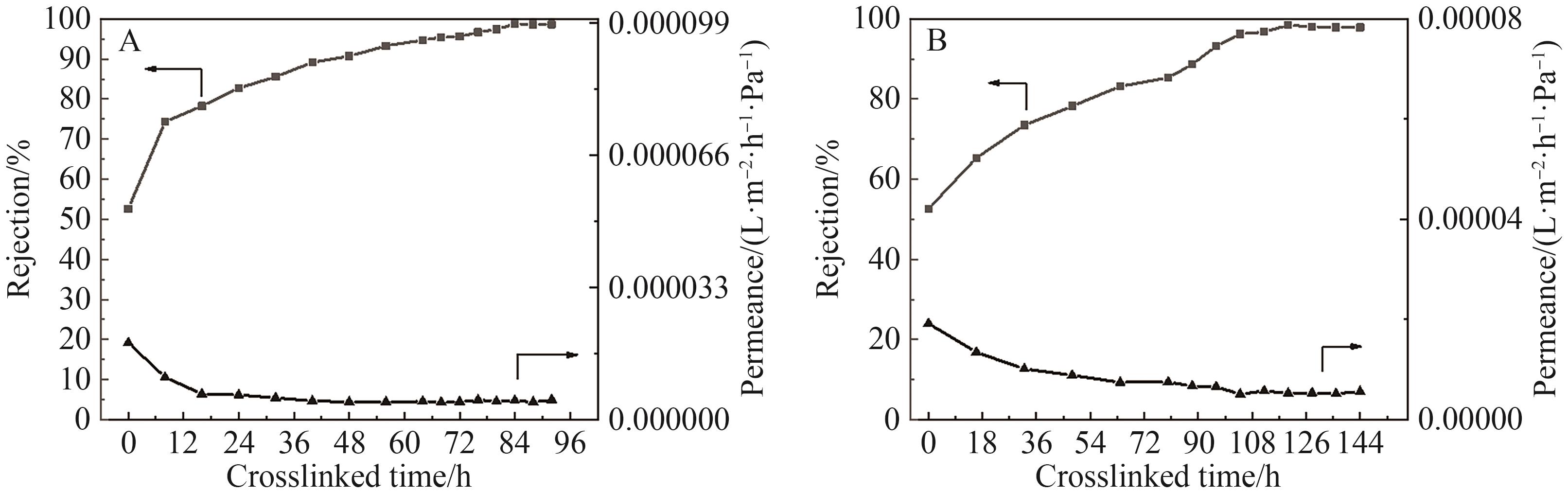
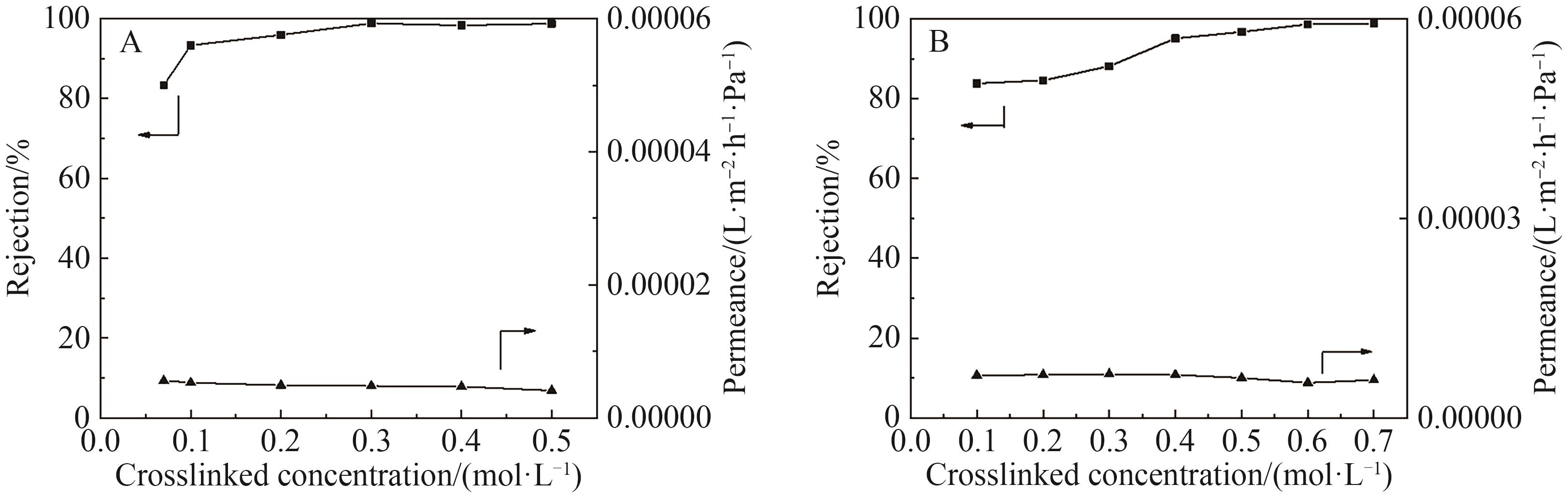
Fig.10 (A) The pure solvent permeation flux of the original OPBI membrane; (B) The pure solvent permeation flux of FeCl3·6H2O crosslinked OPBI membrane; (C) The pure solvent permeation flux of ZnSO4·7H2O crosslinked OPBI membrane; (D) The relationship between the solvent permeation flux of the OPBI-15 membrane and δp·η-1·v-1
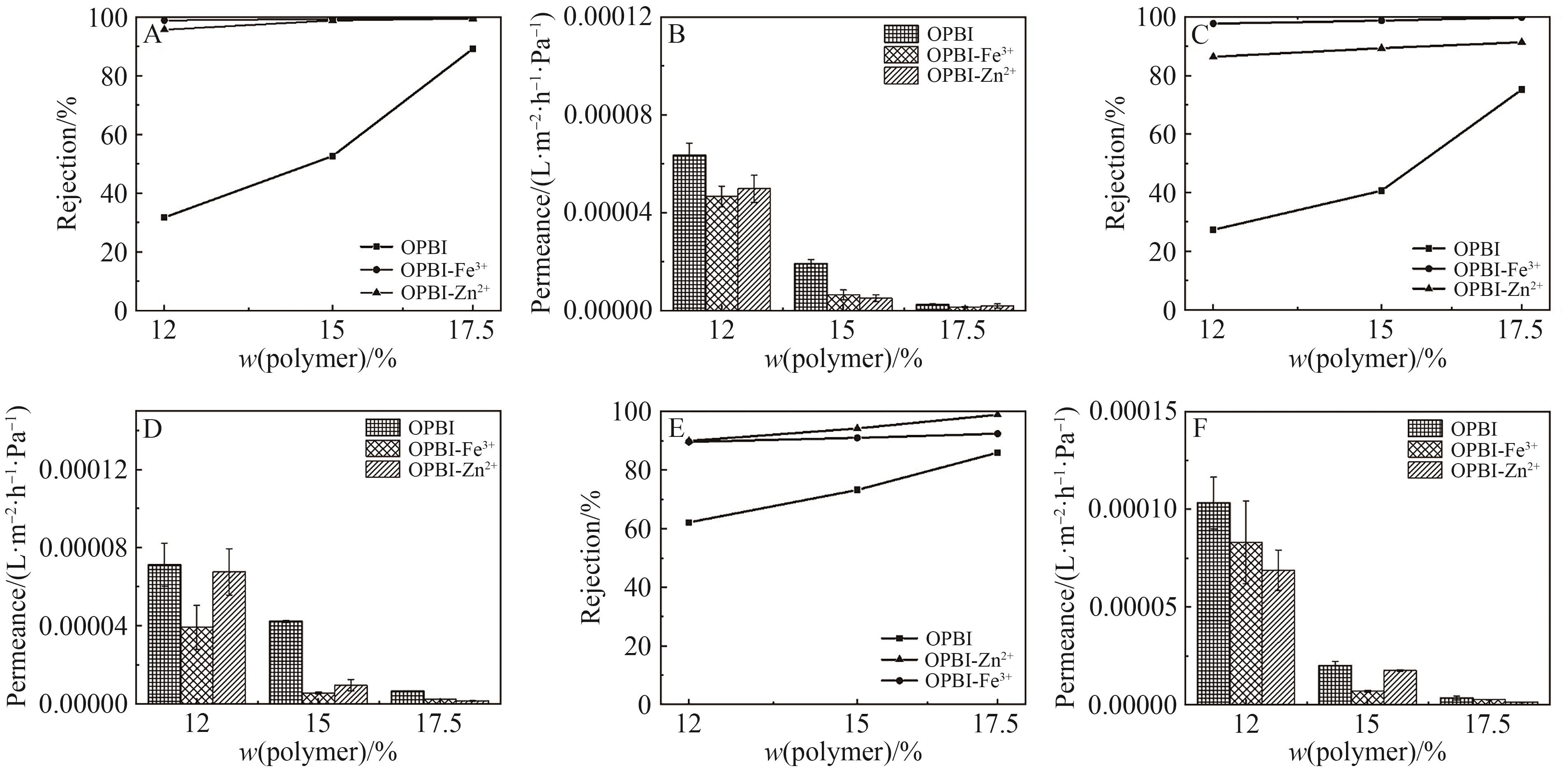
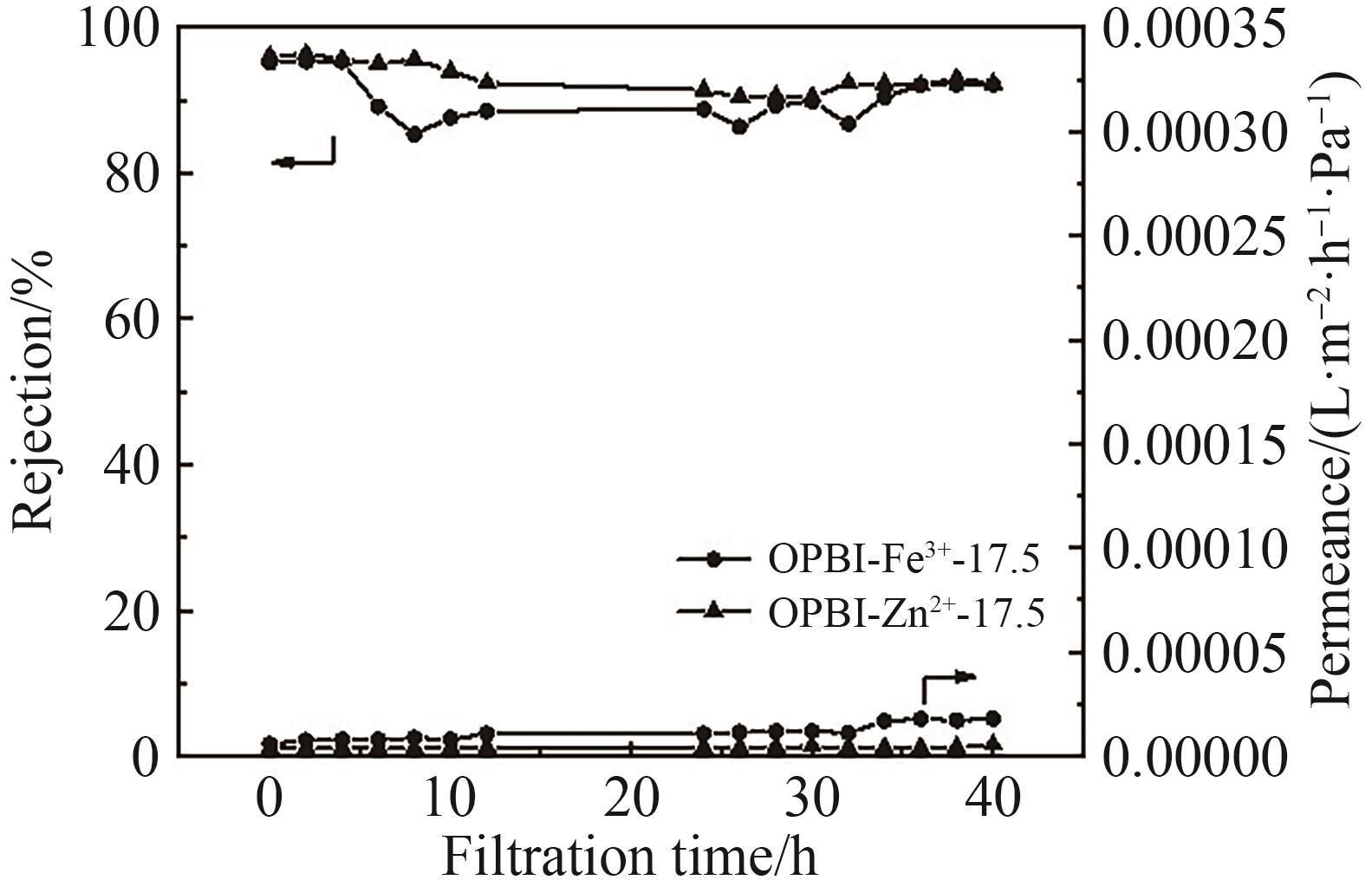
Fig.11 (A) Retention rates of RB in IPA by original and cross-linked OPBI membranes; (B) Permeation flux of RB in IPA by original and cross-linked OPBI membranes; (C) Retention rates of BTB in isopropanol by original and cross-linked OPBI membranes; (D) Permeation flux of BTB in isopropanol by original and cross-linked OPBI membranes; (E) Retention rates of MB in IPA by original and cross-linked OPBI membranes; (F) Permeation flux of MB in IPA by original and cross-linked OPBI membranes
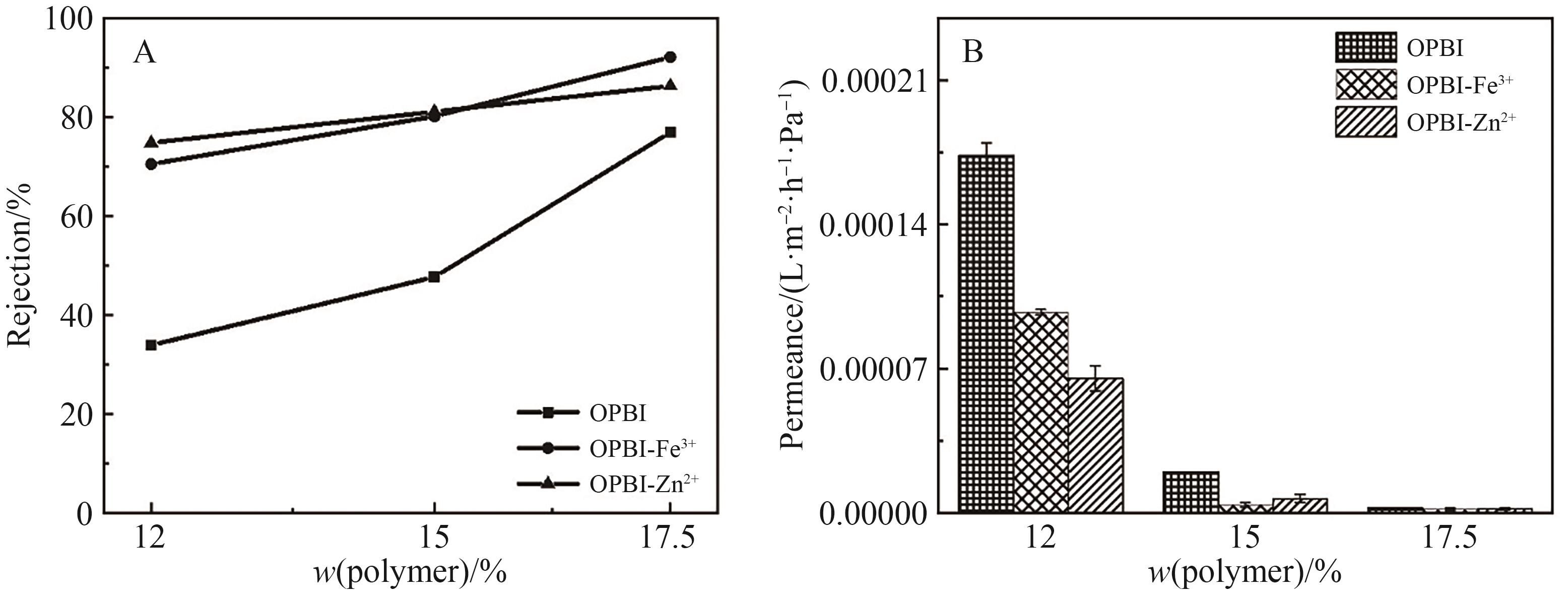
Fig.12 (A) Retention rates of tetracycline in IPA by original and cross-linked OPBI membranes; (B) Permeance of tetracycline in IPA by originaland cross-linked OPBI membranes
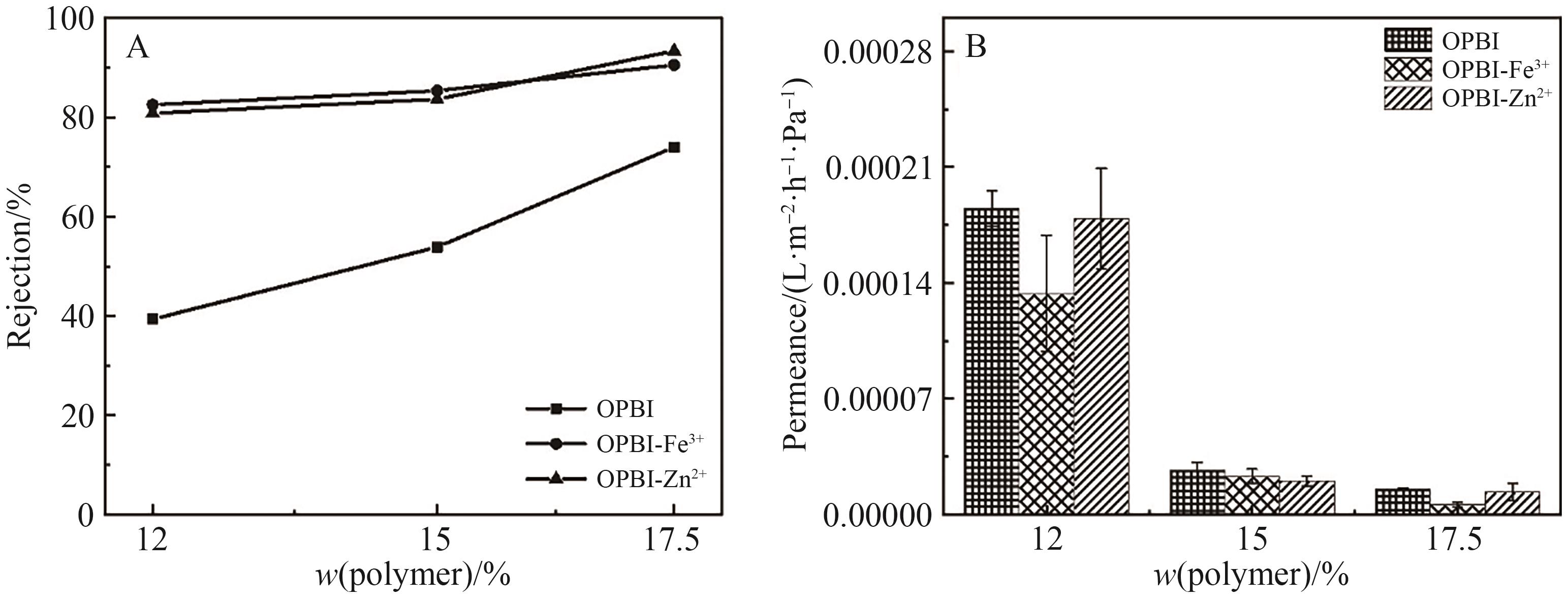
Fig.13 (A) Retention rates of L-α-lecithin in DCM by original and cross-linked OPBI membranes; (B) Permeance of L-α-lecithin in DCM by originaland cross-linked OPBI membranes
| Membrane | n-Heptane | DCM | EA | n-Hexane | IPA | Ethanol | Acetone | MIBK | MeCN |
|---|---|---|---|---|---|---|---|---|---|
| OPBI-12 | — | — | — | — | — | — | — | — | — |
| OPBI-Fe3+-12 | — | — | — | — | — | — | — | — | — |
| OPBI-Zn2+-12 | — | — | — | — | — | — | — | — | — |
| OPBI-15 | — | — | — | — | — | — | — | — | — |
| OPBI-Fe3+-15 | — | — | — | — | — | — | — | — | — |
| OPBI-Zn2+-15 | — | — | — | — | — | — | — | — | — |
| OPBI-17.5 | — | — | — | — | — | — | — | — | — |
| OPBI-Fe3+-17.5 | — | — | — | — | — | — | — | — | — |
| OPBI-Zn2+-17.5 | — | — | — | — | — | — | — | — | — |
Table 3 Stability of original and cross-linked OPBI nanofiltration membranes in organic solvents
| Membrane | n-Heptane | DCM | EA | n-Hexane | IPA | Ethanol | Acetone | MIBK | MeCN |
|---|---|---|---|---|---|---|---|---|---|
| OPBI-12 | — | — | — | — | — | — | — | — | — |
| OPBI-Fe3+-12 | — | — | — | — | — | — | — | — | — |
| OPBI-Zn2+-12 | — | — | — | — | — | — | — | — | — |
| OPBI-15 | — | — | — | — | — | — | — | — | — |
| OPBI-Fe3+-15 | — | — | — | — | — | — | — | — | — |
| OPBI-Zn2+-15 | — | — | — | — | — | — | — | — | — |
| OPBI-17.5 | — | — | — | — | — | — | — | — | — |
| OPBI-Fe3+-17.5 | — | — | — | — | — | — | — | — | — |
| OPBI-Zn2+-17.5 | — | — | — | — | — | — | — | — | — |
| Membrane | THF | DMF | DMAc | NMP |
|---|---|---|---|---|
| OPBI-12 | + | + | + | + |
| OPBI-Fe3+-12 | — | -4.3% | -3.7% | -23.6% |
| OPBI-Zn2+-12 | — | -3.4% | -2.0% | -3.8% |
| OPBI-15 | -4.5% | + | + | + |
| OPBI-Fe3+-15 | — | -1.2% | -2.3% | -16.3% |
| OPBI-Zn2+-15 | — | — | -1.2% | -3.1% |
| OPBI-17.5 | -2.2% | + | + | + |
| OPBI-Fe3+-17.5 | — | — | — | -7.8% |
| OPBI-Zn2+-17.5 | — | — | — | -2.6% |
Table 4 Stability of original and cross-linked OPBI nanofiltration membranes in polar organic solvents
| Membrane | THF | DMF | DMAc | NMP |
|---|---|---|---|---|
| OPBI-12 | + | + | + | + |
| OPBI-Fe3+-12 | — | -4.3% | -3.7% | -23.6% |
| OPBI-Zn2+-12 | — | -3.4% | -2.0% | -3.8% |
| OPBI-15 | -4.5% | + | + | + |
| OPBI-Fe3+-15 | — | -1.2% | -2.3% | -16.3% |
| OPBI-Zn2+-15 | — | — | -1.2% | -3.1% |
| OPBI-17.5 | -2.2% | + | + | + |
| OPBI-Fe3+-17.5 | — | — | — | -7.8% |
| OPBI-Zn2+-17.5 | — | — | — | -2.6% |
| [1] | GANIYU S O, HULLEBUSCH V E D, MARC C, et al. Coupling of membrane filtration and advanced oxidation processes for removal of pharmaceutical residues: a critical review[J]. Sep Purif Technol, 2015, 156: 891-914. |
| [2] | 方彦彦, 涂丛慧, 王晓琳. 静电位阻模型在纳滤膜跨膜电位解析中的应用[J]. 高等学校化学学报, 2010, 31(4): 782-789. |
| FANG Y Y, TU C H, WANG X L. Application of the electrostatic and steric-hindrance model to analysis of transmembrane potential across nanofiltration membranes[J]. Chem J Chin Univ, 2010, 31(4): 782-789. | |
| [3] | RAVANCHI M T, KAGHAZCHI T, KARGARI A. Application of membrane separation processes in petrochemical industry: a review[J]. Desalination, 2009, 235: 199-244. |
| [4] | MARCHETTI P, SOLOMON M F J, SZEKELY G, et al. Molecular separation with organic solvent nanofiltration: a critical review[J]. Chem Rev, 2014, 114: 10735-10806. |
| [5] | XIAO H F, CHU C H, XU W T, et al. Amphibian inspired amino acid ionic liquid functionalized nanofiltration membranes with high water permeability and ion selectivity for pigment wastewater treatment[J]. J Membr Sci, 2019, 586: 44-52. |
| [6] | CONSTABLE D J, JIMENEZ-GONZALEZ C, HENDERSON R K. Perspective on solvent use in the pharmaceutical industry[J]. Org Process Res Dev, 2007, 11: 133-137. |
| [7] | SHI G M, FENG Y, LI B, et al. Recent progress of organic solvent nanofiltration membranes[J]. Prog Polym Sci, 2021, 123: 101470. |
| [8] | ASADI T A, FENG Y, WEBER M, et al. 110th anniversary: selection of cross-linkers and cross-linking procedures for the fabrication of solvent-resistant nanofiltration membranes: a review[J]. Ind Eng Chem Res, 2019, 58: 10678-10691. |
| [9] | XIAO H, FENG Y, GOUNDRY F R W, et al. Organic solvent nanofiltration in pharmaceutical applications[J]. Org Process Res Dev, 2024, 28: 891-923. |
| [10] | SZEKELY G, JIMENEZ-SOLOMON M F, MARCHETTI P, et al. Sustainability assessment of organic solvent nanofiltration: from fabrication to application[J]. Green Chem, 2014, 16: 4440-4473. |
| [11] | BUONMENNA M G, BAE J. Organic solvent nanofiltration in pharmaceutical industry[J]. Sep Purif Rev, 2015, 44: 157-182. |
| [12] | CHENG X Q, ZHANG Y L, WANG Z X, et al. Recent advances in polymeric solvent-resistant nanofiltration membranes[J]. Adv Polym Technol, 2014, 33: 1-24. |
| [13] | 张颖, 胡雁鸣, 李战胜, 等. 聚二苯基乙炔耐溶剂纳滤膜的制备与性能[J]. 应用化学, 2017, 34(4): 443-448. |
| ZHANG Y, HU Y M, LI Z S, et al. Preparation and performance of solvent resistance poly(diphenylacetylene) nanofiltration membranes[J]. Chin J Appl Chem, 2017, 34(4): 443-448. | |
| [14] | VALTCHEVA I B, MARCHETTI P, LIVINGSTON A G. Crosslinked polybenzimidazole membranes for organic solvent nanofiltration (OSN): analysis of cross-linking reaction mechanism and effects of reaction parameter[J]. J Membr Sci, 2015, 493: 568-579. |
| [15] | FARAHANI M H D A, CHUNG T S. A novel crosslinking technique towards the fabrication of high-flux polybenzimidazole (PBI) membranes for organic solvent nanofiltration (OSN)[J]. Sep Purif Technol, 2019, 209: 182-192. |
| [16] | FEI F, CSERI L, SZEKELY G, et al. Robust covalently cross-linked polybenzimidazole graphene oxide membranes for high flux organic solvent nanofiltration[J]. ACS Appl Mater Interfaces, 2018, 10: 16140-16147. |
| [17] | KIM S D, WON G Y, SHAH A A, et al. Reinforcing the polybenzimidazole membrane surface by an ultrathin co-cross-linked polydopamine layer for organic solvent nanofiltration applications[J]. J Membr Sci, 2021, 636: 119587. |
| [18] | LEE J, YANG H, PARK G, et al. Highly stable epoxy-crosslinked polybenzimidazole membranes for organic solvent nanofiltration under strongly basic conditions[J]. J Membr Sci, 2022, 661: 120951. |
| [19] | YANG H, HONG Y, LEE Y, et al. Advancing polybenzimidazole organic solvent nanofiltration membranes with oxygen-rich crosslinking for rapid polar solvent flux[J]. Sep Purif Technol, 2025, 356: 129976. |
| [20] | SHIN S J, PARK Y I, PARK H, et al. Solvent-resistant crosslinked polybenzimidazole membrane for use in enhanced molecular separation[J]. J Membr Sci, 2024, 695: 122463. |
| [21] | CHEN D J, YAN C, LI X N, et al. A highly stable PBI solvent resistant nanofiltration membrane prepared via versatile and simple crosslinking process[J]. Sep Purif Technol, 2019, 224: 15-22. |
| [22] | CHEN D J, YU S S, YANG M, et al. Solvent resistant nanofiltration membranes based on crosslinked polybenzimidazole[J]. RSC Adv, 2016, 6(21): 16925-16932. |
| [23] | 尚尔慧, 陈慧龄, 贺苗苗, 等. 具有图灵结构的聚苯并咪唑耐溶剂纳滤膜的制备及性能[J]. 高等学校化学学报, 2023, 44(6): 211-220. |
| SHANG E H, CHEN H L, HE M M, et al. Preparation and performance of turning structured polybenzimidazole solvent-resistant nanofiltration membrane [J]. Chem J Chin Univ, 2023, 44(6): 211-220. | |
| [24] | YUAN F, YANG Y, WANG R, et al. Poly(vinylidene fluoride) grafted polystyrene (PVDF-g-PS) membrane based on in situ polymerization for solvent resistant nanofiltration[J]. RSC Adv, 2017, 7(53): 33201-33207. |
| [25] | CHEN D J, LIU X, LI D D, et al. Highly stable polysulfone solvent resistant nanofiltration membranes with internal cross-linking networks[J]. RSC Adv, 2016, 6(35): 29570-29575. |
| [26] | LIU T M, HUANG H, WANG Y, et al. Super strong and tough polybenzimidazole metal ions coordination networks: reinforcing mechanism recyclability and anti-counterfeiting applications[J]. Macromol Rapid Commun, 2022, 43(3): 2100643. |
| [27] | WU J, LIAO C Y, LI T Y, et al. Metal-coordinated polybenzimidazole membranes with preferential K+ transport[J]. Nat Commun, 2023, 14(1): 1149. |
| [28] | HARDIAN R, POGANY P, LEE Y M, et al. Molecular sieving using metal-polymer coordination membranes in organic media[J]. Mater Chem A, 2021, 9: 14400-14410. |
| [29] | ASADI T A, LUO L, CHUNG T S, et al. Performance enhancement in organic solvent nanofiltration by double crosslinking technique using sulfonated polyphenylsulfone (sPPSU) and polybenzimidazole (PBI)[J]. J Membr Sci, 2018, 551: 204-213. |
| [30] | HU J, HARDIAN R, GEDE M, et al. Reversible crosslinking of polybenzimidazole-based organic solvent nanofiltration membranes using difunctional organic acids: toward sustainable crosslinking approaches[J]. J Membr Sci, 2022, 648: 120383. |
| [31] | 吴宏超. 含吡啶交联聚苯并咪唑高温质子交换膜的制备与性能研究[D]. 兰州: 兰州大学, 2022. |
| WU H C. Preparation and properties of high temperature proton exchange membranes containing pyridine crosslinked polybenzimidazole[D]. Lanzhou: Lanzhou University, 2022. | |
| [32] | WANG J, LIU X, SUN Y, et al. Synthesis, crystal structures, thermal properties, and DNA-binding studies of transition metal complexes with imidazole ligands[J]. J Coord Chem, 2011, 64(9): 1554-1565. |
| [33] | WU H Q, ANG J M, KONG J H, et al. One-pot synthesis of polydopamine-Zn complex antifouling coatings on membranes for ultrafiltration under harsh conditions[J]. RSC Adv, 2016, 6(105): 103390-103398. |
| [34] | SHIN S J, PARK Y I, PARK H, et al. A facile crosslinking method for polybenzimidazole membranes toward enhanced organic solvent nanofiltration performance[J]. Sep Purif Technol, 2022, 299: 121783. |
| [35] | BESHAHWORED S S, HUANG Y H, ABDI Z G, et al. Polybenzimidazole (PBI) membranes cross-linked with various cross-linkers and impregnated with 4-sulfocalix [4]arene (SCA4) for organic solvent nanofiltration (OSN)[J]. J Membr Sci, 2022, 663: 121039. |
| [36] | DAHE G J, SINGH R P, DUDECK K W, et al. Influence of non-solvent chemistry on polybenzimidazole hollow fiber membrane preparation[J]. J Membr Sci, 2019, 577: 91-103. |
| [37] | AKBAR A T, NIECK E B. Covalent organic polymers for aqueous and organic solvent nanofiltration[J]. Sep Purif Technol, 2022, 298: 121589. |
| [38] | SAIZA C A, DARVISHMANESHB S, BUEKENHOUDT A, et al. Shortcut applications of the hansen solubility parameter for organic solvent nanofiltration[J]. J Membr Sci, 2018, 546: 120-170. |
| [39] | ZHANG Y Y, WANG L, LI L, et al. Insight into the influences of thermal crosslinking on the transition from polyacrylonitrile based ultrafiltration membrane to organic solvent nanofiltration membrane[J]. J Membr Sci, 2023, 679: 121694. |
| [40] | JI C H, XUE S M, LIN C W, et al. Ultrapermeable organic solvent nanofiltration membranes with precisely tailored support layers fabricated using thin-film liftoff[J]. ACS Appl Mater Interfaces, 2020, 12(27): 30796-30804. |
| [41] | NG L Y, MOHAMMAD A W, NG C Y. A review on nanofiltration membrane fabrication and modification using polyelectrolytes: effective ways to develop membrane selective barriers and rejection capability[J]. Adv Colloid Interface Sci, 2013,197/198: 85-107. |
| [42] | 杨炎福, 王齐齐, 张辉, 等. 以聚偏氟乙烯为基底的高渗透选择性聚酰胺/酯纳膜的制备与表征[J]. 高等学校化学学报, 2023, 44(4): 20220574. |
| YANG Y F, WANG Q Q, ZHANG H, et al. Preparation and characterization of a high perm-selectivity poly(amide/ester) nanofiltration membrane using a poly(vinylidene fluoride) substrate[J]. Chem Res Chin Univ, 2023, 44(4): 20220574. | |
| [43] | LUO X F, WANG Z G, WU S S, et al. Metal ion cross-linked nanoporous polymeric membranes with improved organic solvent resistance for molecular separation [J]. J Membr Sci,2021, 621: 119002. |
| [1] | Sheng-Long HU, Zhen-Long LEI, De-Jun CHEN. Preparation of Hypercrosslinked Poly(Naphthalene-based) Microporous Carbon and Its Electrochemical Properties [J]. Chinese Journal of Applied Chemistry, 2024, 41(12): 1742-1750. |
| [2] | Cong-Jun CAO, Han-Xiao MA, Cheng-Min HOU, Xiao-Jian DING, Biao GUAN. Adsorption of Cu(Ⅱ) from Solution by Modified Magnetic Ethyl Cellulose [J]. Chinese Journal of Applied Chemistry, 2022, 39(6): 969-979. |
| [3] | LIU Shiwei, LIANG Liang, LI Chenyang, LIU Changpeng, XING Wei, DONG Xiandui. Multilayered Anode Catalytic Electrode in High-Temperature Proton Exchange Membrane Fuel Cell [J]. Chinese Journal of Applied Chemistry, 2019, 36(9): 1085-1090. |
| [4] | JI Fance, LIU Xiangdong, SHENG Dekun, YANG Yuming. Photothermal-Induced Shape Memory Polymers Prepared by Reactive Blending [J]. Chinese Journal of Applied Chemistry, 2019, 36(8): 904-908. |
| [5] | Cheng TANG,Zhijuan ZOU,Kunpeng SONG. Preparation of Ni-P Co-doped Hyper-Crosslinked Polymer and Used for Reduction of 4-Nitrophenol [J]. Chinese Journal of Applied Chemistry, 2019, 36(7): 782-789. |
| [6] | XING Chenli, WANG Jing, ZHANG Zhaohui, XIE Dandan, LÜ Piaopiao. Multiple Metal Ion Imprinted Electrochemical Sensor with Enhanced Sensitivity by Graphene Oxide-C60 Composite [J]. Chinese Journal of Applied Chemistry, 2019, 36(3): 341-348. |
| [7] | ZHANG Ying, HU Yanming, LI Zhansheng, XU Jie, XIA Yuping, MASUDA Toshio. Preparation and Performance of Solvent Resistance Poly(diphenylacetylene) Nanofiltration Membranes [J]. Chinese Journal of Applied Chemistry, 2017, 34(4): 443-448. |
| [8] | HUANG Guangcheng, QIU Bite, LIU Yadong, ZHAO Hui, JI Shengxiang. Synthesis and Characterization of Crosslinked Porous Poly(styrene-co-divinylbenzene) Microspheres with Tunable Particle and Pore Diameters [J]. Chinese Journal of Applied Chemistry, 2016, 33(4): 406-411. |
| [9] | WANG Dongsheng,LI Wentao,YANG Xiaofang,AN Guangyu. Ferrates:Green Oxidants and Coagulants in Water Treatment [J]. Chinese Journal of Applied Chemistry, 2016, 33(11): 1221-1233. |
| [10] | YAO Qingxin, XIE Jianjun, LIU Junxia, TANG Liping, LIU Yuan. Effect of Ionic Strength on the Adsorption of Pb2+ and Cu2+ onto Bentonite/Sodium Lignosulfonate Graft-Polymerized with Acrylamide and Maleic Anhydride [J]. Chinese Journal of Applied Chemistry, 2015, 32(8): 940-947. |
| [11] | WANG Wei, CHEN Ji, LIU Hongzhao, YANG Hualing, CAO Yaohua, GAO Zhaoguo, ZHANG Bo. Research Progress of Task-specific Ionic Liquids Used in Metal Ions Extraction [J]. Chinese Journal of Applied Chemistry, 2015, 32(7): 733-742. |
| [12] | WANG Bo, ZHANG Fan*, HUANG Fu. Preparation of Reduced Graphene Oxide And Its Adsorption Property for Heavy Metal Ions [J]. Chinese Journal of Applied Chemistry, 2014, 31(04): 502-504. |
| [13] | XU Siyuan, LEI Ping, JIN Guanping*. Determination of Pb(Ⅱ), Cd(Ⅱ) with Melamine Chelating Resin/Multi-walled Carbon Nanotubes Composites Modified Waxed Graphite Electrode [J]. Chinese Journal of Applied Chemistry, 2014, 31(02): 206-211. |
| [14] | DING Hao, GAO Baojiao*, CHENG Wei. Synthesis, Immobilization and Characterization of Chiral Metal-Salen Complex on Chloromethylated Crosslinked Polystyrene Microspheres [J]. Chinese Journal of Applied Chemistry, 2013, 30(03): 276-282. |
| [15] | DAI Xin, GAO Baojiao*, DING Hao, FANG Xiaolin. Aminomethyl Group-Modification of Chloromethylated Polystyrene Microspheres and Preparation of Schiff Base-Type Chelating Resin Microspheres [J]. Chinese Journal of Applied Chemistry, 2012, 29(04): 383-391. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||