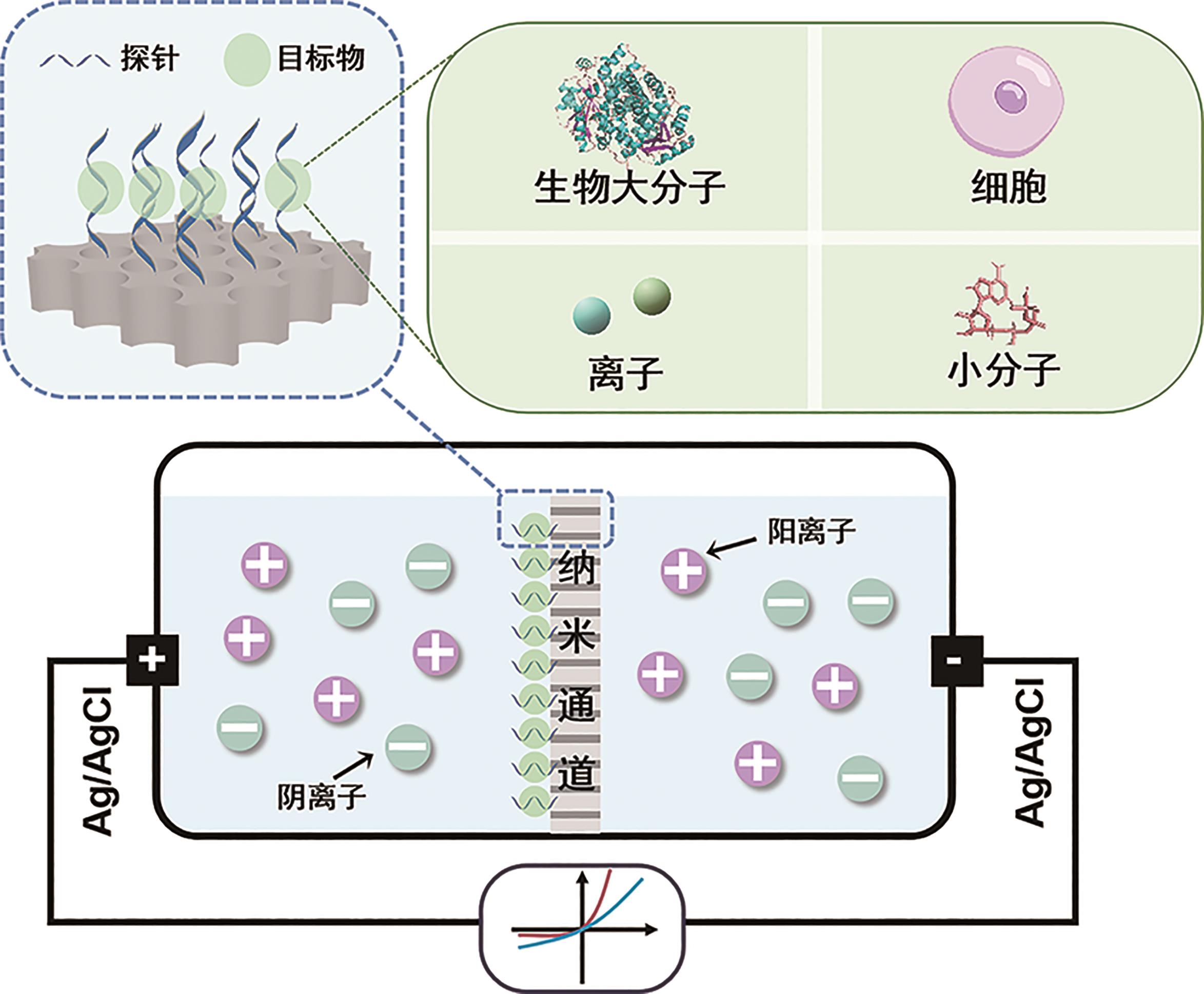
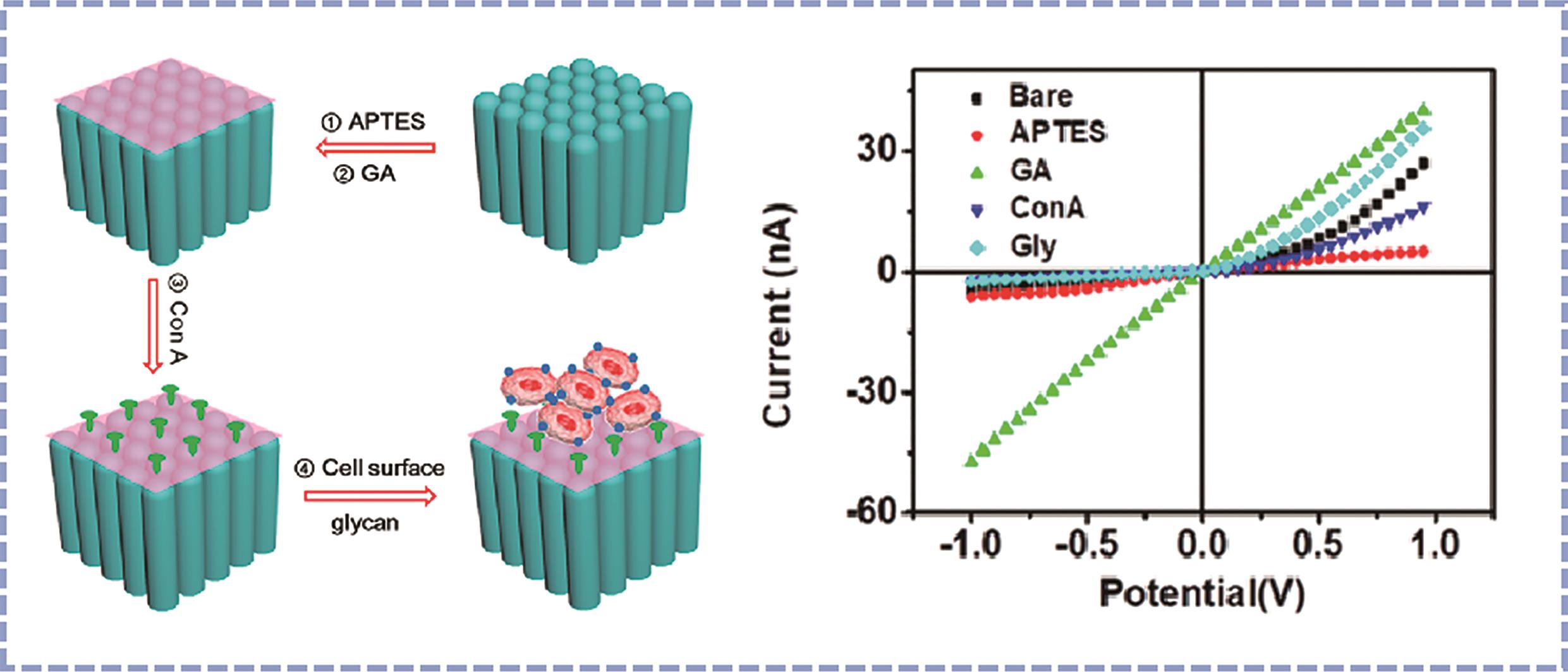
Chinese Journal of Applied Chemistry ›› 2024, Vol. 41 ›› Issue (1): 87-99.DOI: 10.19894/j.issn.1000-0518.230304
• Review • Previous Articles Next Articles
Development in Probes on Outer Surface of Nanochannels for Detecting Biomarkers
Li-Zhen YUAN, Ni-Ya LIN, Yun-Fan ZHANG, Jing-Jing HU( ), Xiao-Ding LOU, Fan XIA
), Xiao-Ding LOU, Fan XIA
- State Key Laboratory of Biogeology and Environmental Geology,Engineering Research Center of Nano-Geomaterials of Ministry of Education,Faculty of Materials Science and Chemistry,China University of Geosciences,Wuhan 430074,China
-
Received:2023-10-05Accepted:2023-12-15Published:2024-01-01Online:2024-01-30 -
Contact:Jing-Jing HU -
About author:hujingjing@cug.edu.cn
-
Supported by:the National Natural Science Foundation of China(22090050)
CLC Number:
Cite this article
Li-Zhen YUAN, Ni-Ya LIN, Yun-Fan ZHANG, Jing-Jing HU, Xiao-Ding LOU, Fan XIA. Development in Probes on Outer Surface of Nanochannels for Detecting Biomarkers[J]. Chinese Journal of Applied Chemistry, 2024, 41(1): 87-99.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.230304
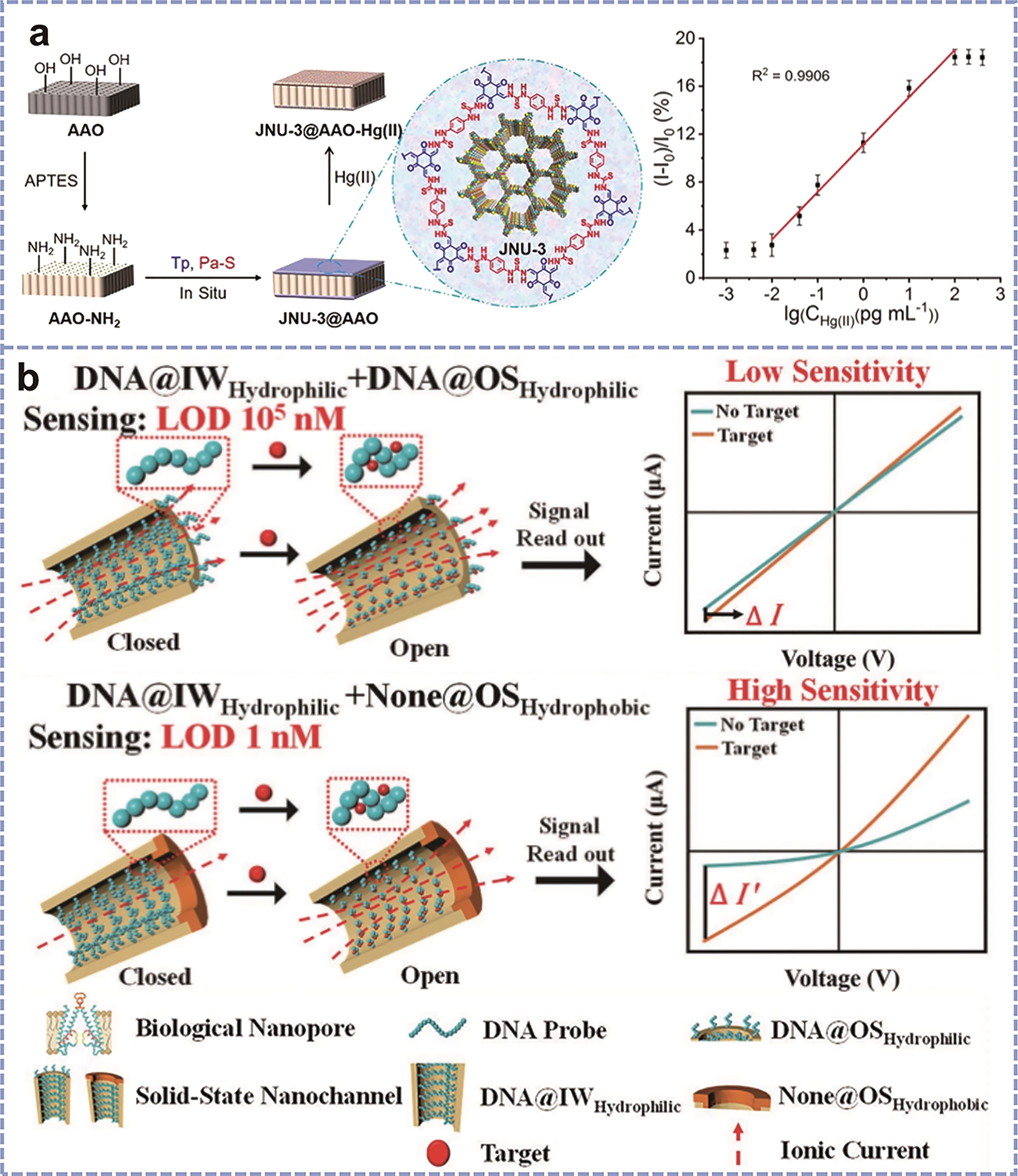
Fig.2 (a) Schematic illustration of JNU-3@AAO-based nanofluidic sensor for the detection of Hg(Ⅱ)[24]; (b) Schematic diagram of bio-inspired solid-state nanochannels (DNA@IWHydrophilic+DNA@OSHydrophilic and DNA@IWHydrophilic+None@OSHydrophobic) sensing target[25]
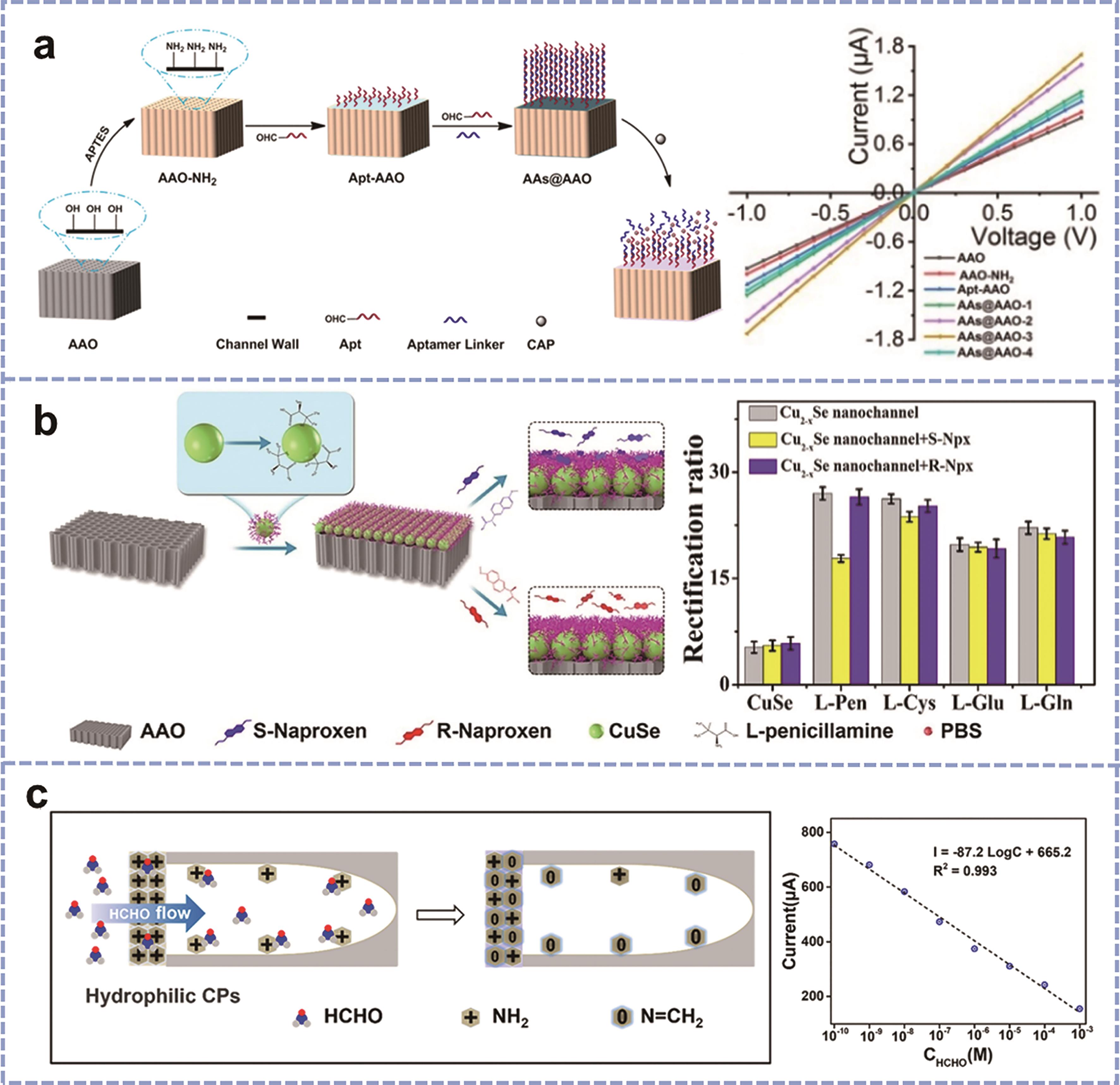
Fig.3 (a) Schematic illustration of JNU-3@AAO-based nanofluidic sensor for the detection of Hg(Ⅱ) and I-V plot of the test results[29]; (b) Schematic diagram of Cu2-x Se/AAO nanochannels for the detection of naproxen[30]; (c) Schematic diagram of CP/AAO solid-state nanochannels for the detection of formaldehyde[31]

Fig.4 (a) Schematic diagram of the principle of thrombin recognition detection[34]; (b) Schematic illustration of the detection of MUC1 based on the Au-AAO ion channel[35]; (c) Schematic illustration of the aptamer-functionalized nanochannels for one-step detection of SARS-CoV-2[36]

Fig.5 (a) Schematic diagram showing the peptide self-assembly on the block layer of PAA for dynamic monitoring of MMP activities at the biomimetic cell/ECM interface[40]; (b) Schematic of the solid-state nanochannel detection of MMP-2 by peptide probe functionalization[41]; (c) Schematic drawing of experimental procedures for the trypsin-PLL cleavage reaction[42]

Fig.8 (a) Illustration of surface modification and miRNA detection on the nanochannel-ionchannel hybrid[54]; (b) Schematic illustration of the RCA process triggered by DNA targets through the T4 polynucleotide kinase reaction illustration of surface modification and miRNA detection on the nanochannel-ionchannel hybrid[57]
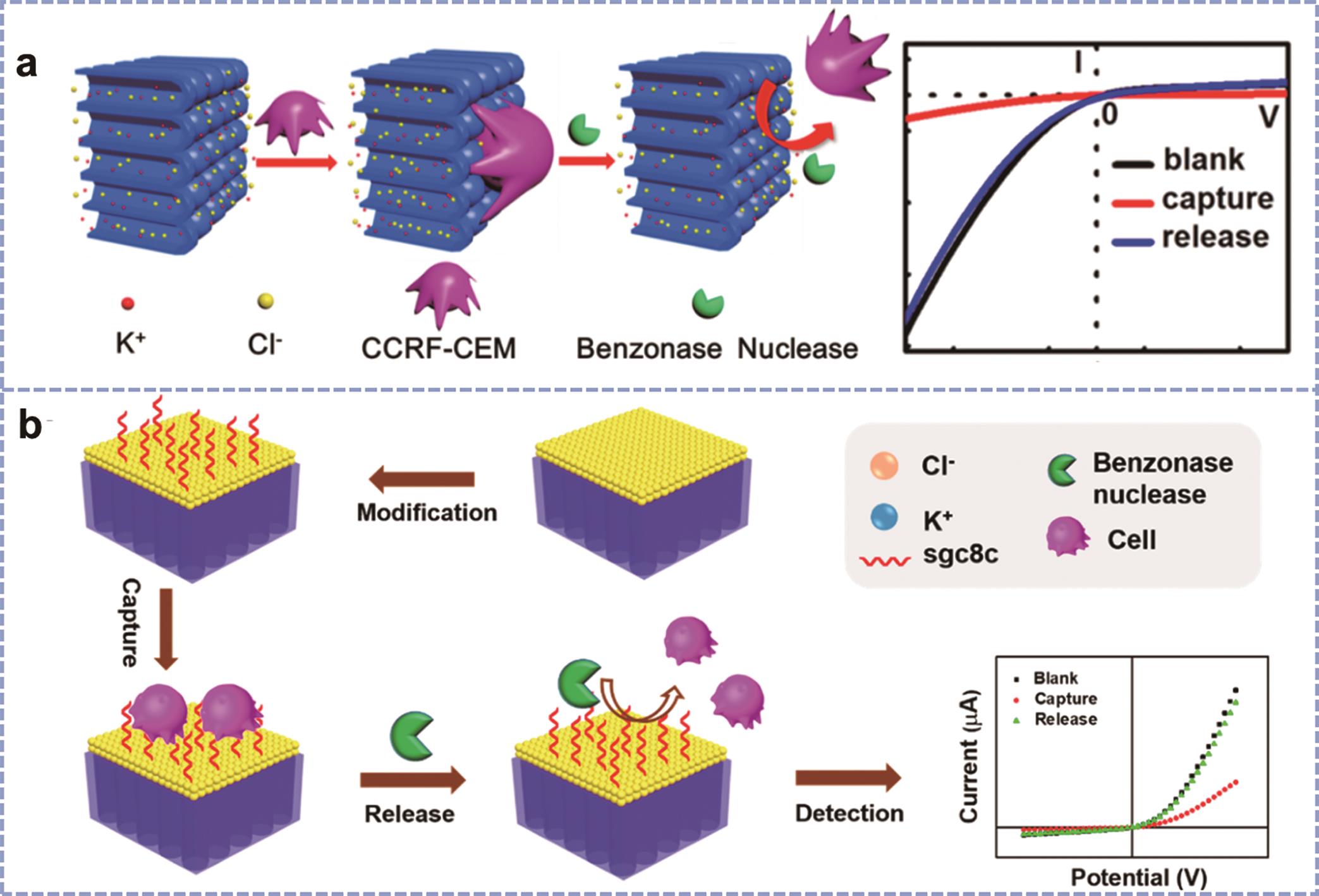
Fig.9 (a) Diagrammatical illustration of the CTCs capture and release process on the nanochannel-ion channel hybrid[61]; (b) Schematic demonstration of the CTCs capture and release processes and the corresponding I-V curves of the DAN/AAO hybrid[62]
| 1 | 淳彩璞, 王凌志, 赵宏, 等. 乳腺癌生物标志物研究新进展[J]. 兵团医学, 2023, 21(4): 57-59. |
| CHUN C P, WANG L Z, ZHAO H, et al. New progress of breast cancer biomarkers research[J]. J Bingtuan Med, 2023, 21(4): 57-59. | |
| 2 | CASTRO SESQUEN Y E, SARAF S L, GORDEUK V R, et al. Use of multiple urinary biomarkers for early detection of chronic kidney disease in sickle cell anemia patients[J]. Blood, 2020, 136(Supplement 1): 30. |
| 3 | YAN B. Photofunctional MOF-based hybrid materials for the chemical sensing of biomarkers[J]. J Mater Chem C, 2019, 7(27): 8155-8175. |
| 4 | XU D, XIAO H, WANG S, et al. Universal and sensitive drug assessment biosensing platform using optimal mechanical beating detection of single cardiomyocyte[J]. ACS Nano, 2022, 16(9): 15484-15494. |
| 5 | MUNAWAR J, KHAN M S, SYEDA S E Z, et al. Metal-organic framework-based smart nanoplatforms for biosensing, drug delivery, and cancer theranostics[J]. Inorg Chem Commun, 2023, 147: 110145. |
| 6 | SHEN J, LI Y, GU H, et al. Recent development of sandwich assay based on the nanobiotechnologies for proteins, nucleic acids, small molecules, and ions[J]. Chem Rev, 2014, 114(15): 7631-7677. |
| 7 | WANG H, WANG J, MA G, et al. TICT-based turn-on deep-red fluorescent probe for endoplasmic reticulum targeted detection of serum albumin in kidney diseases[J]. Chem Eng J, 2023, 464: 142551. |
| 8 | YU R J, MA W, LIU X Y, et al. Metal-linked immunosorbent assay (MeLISA): the enzyme-free alternative to ELISA for biomarker detection in serum[J]. Theranostics, 2016, 6(10): 1732. |
| 9 | YE H, YANG K, TAO J, et al. An enzyme-free signal amplification technique for ultrasensitive colorimetric assay of disease biomarkers[J]. ACS Nano, 2017, 11(2): 2052-2059. |
| 10 | PARVIN R, ZHANG L, ZU Y, et al. Photothermal responsive digital polymerase chain reaction resolving exosomal micrornas expression in liver cancer[J]. Small, 2023: 2207672. |
| 11 | DAS S, DEVIREDDY R, GARTIA M R. Surface plasmon resonance (SPR) sensor for cancer biomarker detection[J]. Biosensors, 2023, 13(3): 396. |
| 12 | SONG X T, YIN Y D, WU G R, et al. Nanopore-based metal ion detection and metal ion-mediated nanopore sensing[J]. Chin J Chem, 2023, 41(20): 2746-2757. |
| 13 | HUANG Y, LIU L, LUO C, et al. Solid-state nanochannels for bio-marker analysis[J]. Chem Soc Rev, 2023, 52(18): 6270-6293. |
| 14 | MA Q, SI Z, LI Y, et al. Functional solid-state nanochannels for biochemical sensing[J]. Trac-Trends Anal Chem, 2019, 115: 174-186. |
| 15 | FRIED J P, SWETT J L, NADAPPURAM B P, et al. In situ solid-state nanopore fabrication[J]. Chem Soc Rev, 2021, 50(8): 4974-4992. |
| 16 | CAIRNS GIBSON D F, COCKROFT S L. Functionalised nanopores: chemical and biological modifications[J]. Chem Sci, 2022, 13(7): 1869-1882. |
| 17 | MA Q, LI Y, WANG R, et al. Towards explicit regulating-ion-transport: nanochannels with only function-elements at outer-surface[J]. Nat Commun, 2021, 12(1): 1573. |
| 18 | MA Q, LIU T, XU R, et al. Revealing the critical role of probe grafting density in nanometric confinement in ionic signal via an experimental and theoretical study[J]. Anal Chem, 2021, 93(4): 1984-1990. |
| 19 | MA Q, WANG R, GAO P, et al. Revealing the role of surface wettability in ionic detection signals of nanofluidic-based chemical sensors[J]. Anal Chem, 2022, 94(47): 16411-16417. |
| 20 | SI Z, XU H, LIN M, et al. Polydopamine-induced modification on the highly charged surface of asymmetric nanofluidics: a strategy for adjustable ion current rectification properties[J]. Anal Chem, 2022, 94(5): 2493-2501. |
| 21 | WU X, LI Y, XU H, et al. Exponential increase in an ionic signal: a dominant role of the space charge effect on the outer surface of nanochannels[J]. Anal Chem, 2021, 93(40): 13711-13718 |
| 22 | MAO H, MA Q, XU H, et al. Exploring the contribution of charged species at the outer surface to the ion current signal of nanopores: a theoretical study[J]. Analyst, 2021, 146(16): 5089-5094. |
| 23 | 马群. 精准分区功能化纳米孔道构筑及其性能研究[D]. 武汉: 中国地质大学, 2022. |
| MA Q. Study of the construction and properties of precisely functional nanochannels with explicit spatial partition[D]. Wuhan: China University of Geosciences, 2022. | |
| 24 | RAN X Q, QIAN H L, YAN X P. Integrating ordered two-dimensional covalent organic frameworks to solid-state nanofluidic channels for ultrafast and sensitive detection of mercury[J]. Anal Chem, 2022, 94(23): 8533-8538. |
| 25 | LIU L, LUO C, ZHANG J, et al. Synergistic effect of bio-inspired nanochannels: hydrophilic dna probes at inner wall and hydrophobic coating at outer surface for highly sensitive detection[J]. Small, 2022, 18(37): 2201925. |
| 26 | MA X, LI Y, ZHANG J, et al. Metal-organic framework-decorated nanochannel electrode: integration of internal nanoconfined space and outer surface for small-molecule sensing[J]. ACS Appl Mater Interfaces, 2023, 15(22): 27034-27045. |
| 27 | LI X, ZHAI T, GAO P, et al. Role of outer surface probes for regulating ion gating of nanochannels[J]. Nat Commun, 2018, 9(1): 40. |
| 28 | ROUSHANI M, RAHMATI Z, HOSEINI S J, et al. Impedimetric ultrasensitive detection of chloramphenicol based on aptamer MIP using a glassy carbon electrode modified by 3-ampy-RGO and silver nanoparticle[J]. Colloids Surf B-Biointerfaces, 2019, 183: 110451. |
| 29 | RAN X Q, QIAN H L, YAN X P. Aptamer self-assembly-functionalized nanochannels for sensitive and precise detection of chloramphenicol[J]. Anal Chem, 2021, 93(42): 14287-14292. |
| 30 | MENG D, HAO C, CAI J, et al. Tailored chiral copper selenide nanochannels for ultrasensitive enantioselective recognition and detection[J]. Angew Chem Int Ed Engl, 2021, 60(47): 24997-25004. |
| 31 | ZHANG D, ZHANG X. Aquaporin-inspired CPs/AAO nanochannels for the effective detection of HCHO: importance of a hydrophilic/hydrophobic Janus device for high-performance sensing[J]. Nano Lett, 2022, 22(9): 3793-3800. |
| 32 | KIM M, JO H, JUNG G Y, et al. Molecular complementarity of proteomimetic materials for target-specific recognition and recognition-mediated complex functions[J]. Adv Mater, 2023, 35(22): 2208309. |
| 33 | LI Y, TAM W W, YU Y, et al. The application of aptamer in biomarker discovery[J]. Biomark Res, 2023, 11(1): 70. |
| 34 | ZHAO X P, ZHOU Y, ZHANG Q W, et al. Nanochannel-ion channel hybrid device for ultrasensitive monitoring of biomolecular recognition events[J]. Anal Chem, 2019, 91(1): 1185-1193.. |
| 35 | PAN M, CAI J, LI S, et al. Aptamer-gated ion channel for ultrasensitive mucin 1 detection[J]. Anal Chem, 2021, 93(11): 4825-4831. |
| 36 | SHI L, WANG L, MA X, et al. Aptamer-functionalized nanochannels for one-step detection of SARS-CoV-2 in samples from COVID-19 patients[J]. Anal Chem, 2021, 93(49): 16646-16654. |
| 37 | BHAGWAT S R, HAJELA K, KUMAR A. Proteolysis to identify protease substrates: cleave to decipher[J]. Proteomics, 2018, 18(13): 1800011. |
| 38 | SINGH A. Nanopore-detectable reporter proteins[J]. Nat Methods, 2021, 18(10): 1149. |
| 39 | SHI L, KUANG D, MA X, et al. Peptide assembled in a nano-confined space as a molecular rectifier for the availability of ionic current modulation[J].Nano Lett, 2022, 22(3): 1083-1090. |
| 40 | WANG L, LI H, SHI L, et al. In situ peptide self-assembly on ionic nanochannel for dynamic monitoring of MMPs in extracellular matrix[J]. Biosens Bioelectron, 2022, 195: 113671. |
| 41 | HU J J, JIANG W, QIAO Y, et al. Enzyme regulating the wettability of the outer surface of nanochannels[J]. ACS Nano, 2023, 17(12): 11935-11945. |
| 42 | DUAN C, ALIBAKHSHI M A, KIM D K, et al. Label-free electrical detection of enzymatic reactions in nanochannels[J]. ACS Nano, 2016, 10(8): 7476-7484. |
| 43 | QIAO Y, HU J J, HU Y, et al. Detection of unfolded cellular proteins using nanochannel arrays with probe-functionalized outer surfaces[J]. Angew Chem Int Ed Engl, 2023, 62(43): e202309671. |
| 44 | PIPERIGKOU Z, MOHR B, KARAMANOS N, et al. Shed proteoglycans in tumor stroma[J]. Cell Tissue Res, 2016, 365(3): 643-655. |
| 45 | MAGALHÃES A, DUARTE H O, REIS C A. Aberrant glycosylation in cancer: a novel molecular mechanism controlling metastasis[J]. Cancer Cell, 2017, 31(6): 733-735. |
| 46 | LIU X, MCNALLY D J, NOTHAFT H, et al. Mass spectrometry-based glycomics strategy for exploring n-linked glycosylation in eukaryotes and bacteria[J]. Anal Chem, 2006, 78(17): 6081-6087. |
| 47 | NISHIKAZE T, KANESHIRO K, KAWABATA S I, et al. Structural analysis of N-glycans by the glycan-labeling method using 3-aminoquinoline-based liquid matrix in negative-ion MALDI-MS[J]. Anal Chem, 2012, 84(21): 9453-9461. |
| 48 | PARK S, SUNG J W, SHIN I. Fluorescent glycan derivatives: their use for natural glycan microarrays[J]. ACS Chem Biol, 2009, 4(9): 699-701. |
| 49 | LIM S H, MUSTO C J, PARK E, et al. A colorimetric sensor array for detection and identification of sugars[J]. Org Lett, 2008, 10(20): 4405-4408. |
| 50 | CHEN X, HE Y, ZHANG Y, et al. Ultrasensitive detection of cancer cells and glycan expression profiling based on a multivalent recognition and alkaline phosphatase-responsive electrogenerated chemiluminescence biosensor[J]. Nanoscale, 2014, 6(19): 11196-11203. |
| 51 | LIU F F, ZHAO X P, LIAO X W, et al. Ultrasensitive and label-free detection of cell surface glycan using nanochannel-ionchannel hybrid coupled with electrochemical detector[J].Anal Chem, 2020, 92(7): 5509-5516. |
| 52 | ARENZ C. MicroRNAs-future drug targets?[J]. Angew Chem Int Ed Engl, 2006, 45(31): 5048-5050. |
| 53 | LEE J M, CHO H, JUNG Y. Fabrication of a structure-specific RNA binder for array detection of label-free microRNA[J]. Angew Chem Int Ed Engl, 2010, 49(46): 8662-8665. |
| 54 | ZHAO X P, LIU F F, HU W C, et al. Biomimetic nanochannel-ionchannel hybrid for ultrasensitive and label-free detection of microRNA in cells[J]. Anal Chem, 2019, 91(5): 3582-3589. |
| 55 | PARK J S, PISANIC T, ZHANG Y, et al. Ligation-enabled fluorescence-coding PCR for high-dimensional fluorescence-based nucleic acid detection[J]. Anal Chem, 2021, 93(4): 2351-2358. |
| 56 | ALI M M, LI F, ZHANG Z, et al. Rolling circle amplification: a versatile tool for chemical biology, materials science and medicine[J]. Chem Soc Rev, 2014, 43(10): 3324-3341. |
| 57 | WU X, CHE C, WANG X, et al. Ionic signal enhancement by the space charge effect through the DNA rolling circle amplification on the outer surface of nanochannels[J]. Anal Chem, 2021, 93(48): 16043-16050. |
| 58 | YOON H J, KOZMINSKY M, NAGRATH S. Emerging role of nanomaterials in circulating tumor cell isolation and analysis[J]. ACS Nano, 2014, 8(3): 1995-2017. |
| 59 | LIN E, CAO T, NAGRATH S, et al. Circulating tumor cells: diagnostic and therapeutic applications[J]. Annu Rev Biomed Eng, 2018, 20(1): 329-352. |
| 60 | MOCELLIN S, KEILHOLZ U, ROSSI C R, et al. Circulating tumor cells: the ‘leukemic phase’ of solid cancers[J]. Trends Mol Med, 2006, 12(3): 130-139. |
| 61 | CAO J, ZHAO X P, YOUNIS M R, et al. Ultrasensitive capture, detection, and release of circulating tumor cells using a nanochannel-ion channel hybrid coupled with electrochemical detection technique[J]. Anal Chem, 2017, 89(20): 10957-10964. |
| 62 | WANG C, ZHAO X P, LIU F F, et al. Dendrimer-Au nanoparticle network covered alumina membrane for ion rectification and enhanced bioanalysis[J]. Nano Lett, 2020, 20(3): 1846-1854. |
| [1] | ZHANG Nan, LI Tie, YANG Guang, HUANG Xin, YUE Hao, WANG Yang, LIU Jun-Tong, LIU Shu-Ying, WANG Fu-Chun. Screening for Urine Metabolic Biomarkers of Transient Ischemic Attack [J]. Chinese Journal of Applied Chemistry, 2021, 38(3): 305-314. |
| [2] | YANG Xian, MIN Lingli, ZHU Yinglin, CAO Liuxuan, XIE Yanbo, HOU Xu. Recent Research Progress on Nanopores and Nanochannels Based Electrokinetical Energy Conversion Systems [J]. Chinese Journal of Applied Chemistry, 2018, 35(6): 613-624. |
| [3] | Zhang Gongcheng, Tan Zhen, Wei Shujuan, Li Yulin. SYNTHESIS 0F BI0MARKERS Ⅲ.SYNTHESIS OF 5α-CHOLESTANE AND 5β-CHOLESTANE [J]. Chinese Journal of Applied Chemistry, 1989, 0(4): 50-53. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||