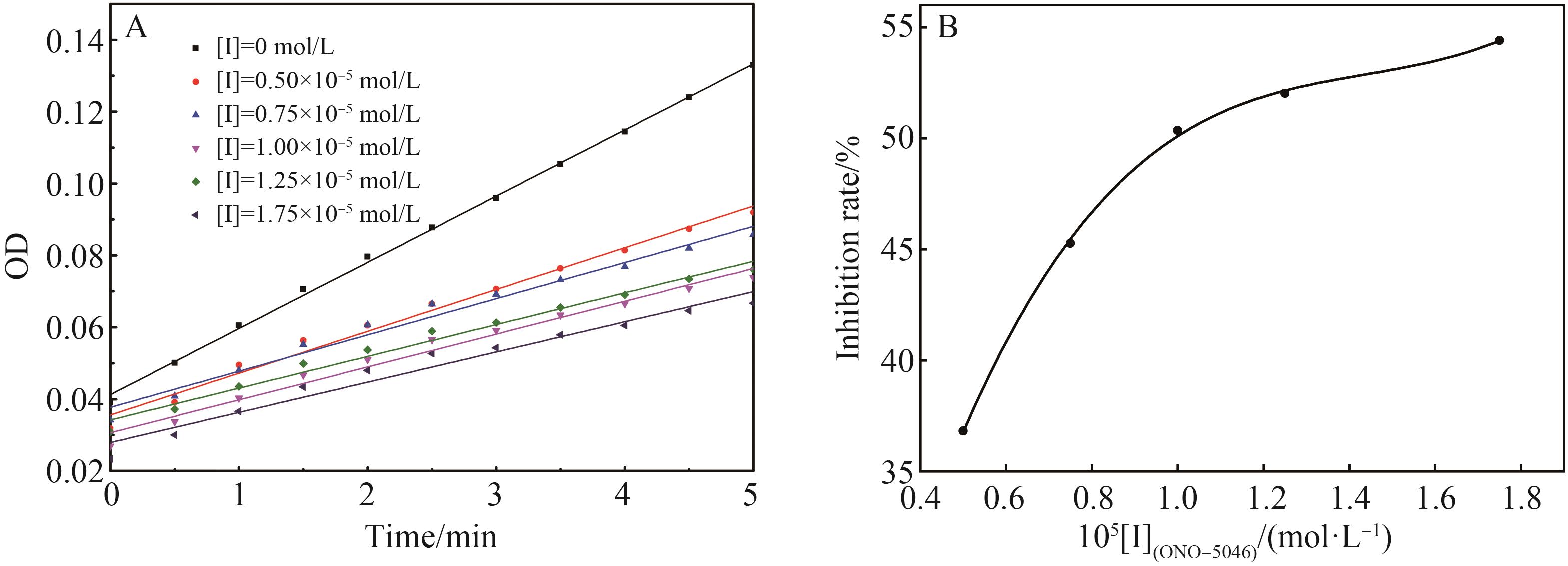
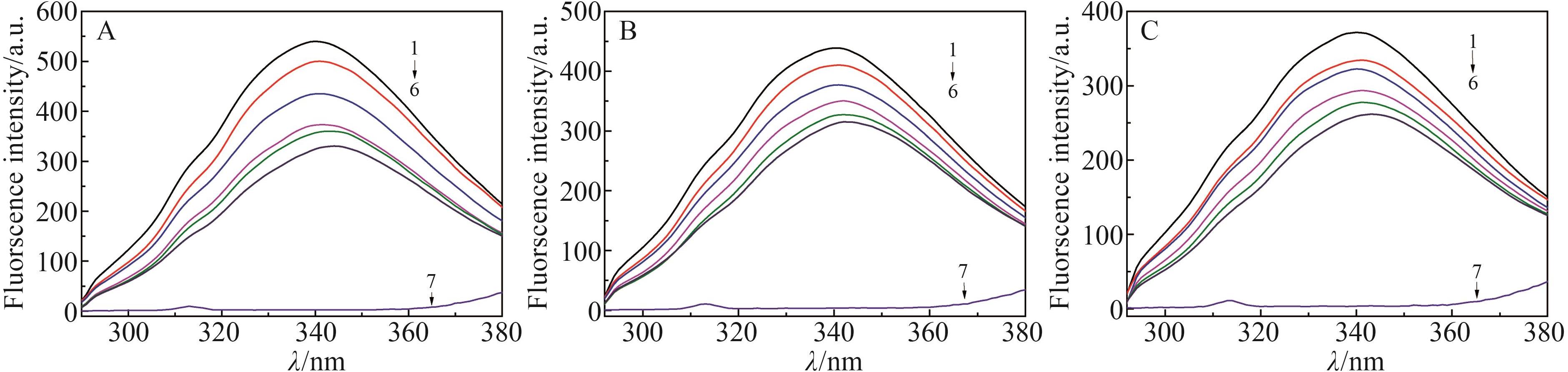
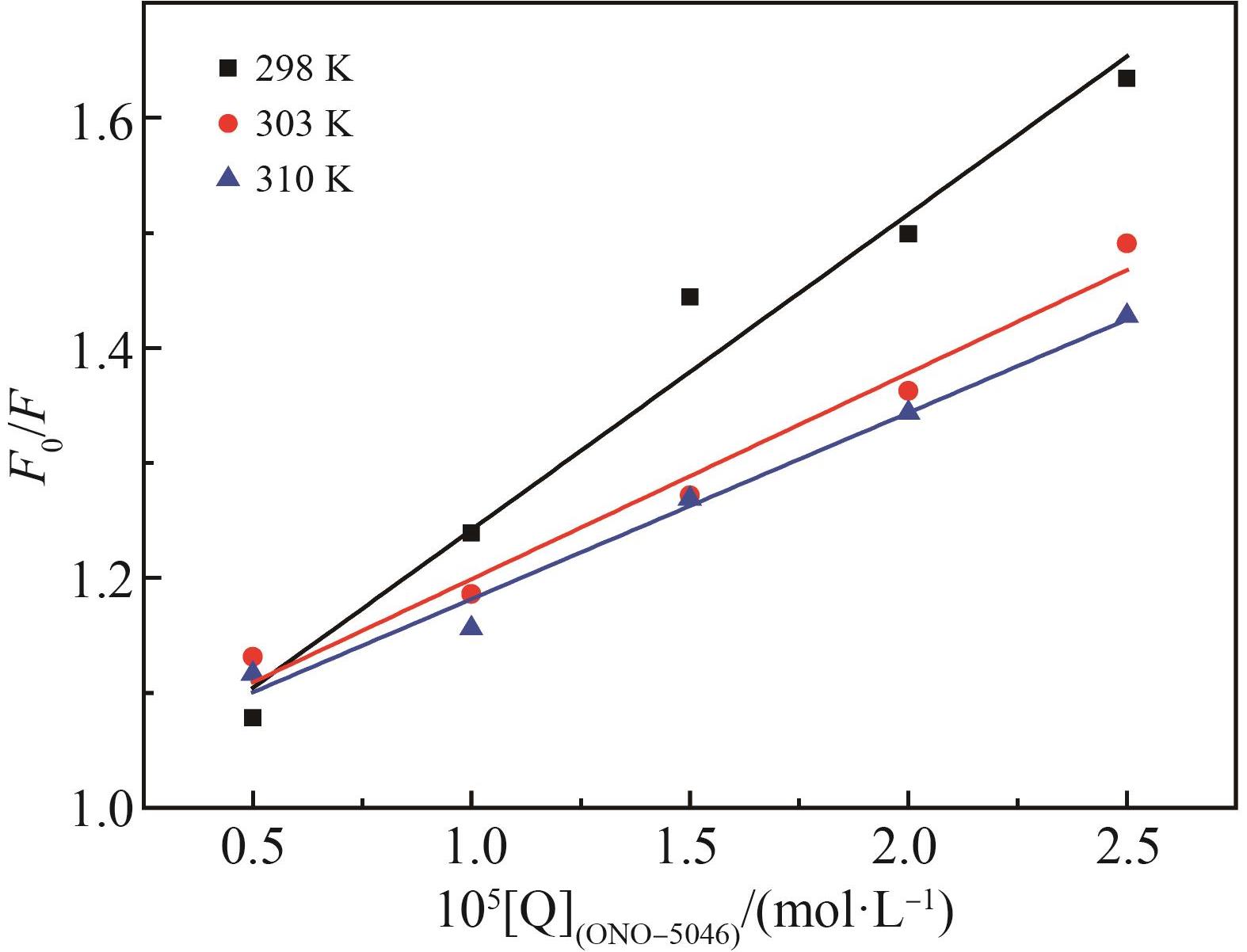
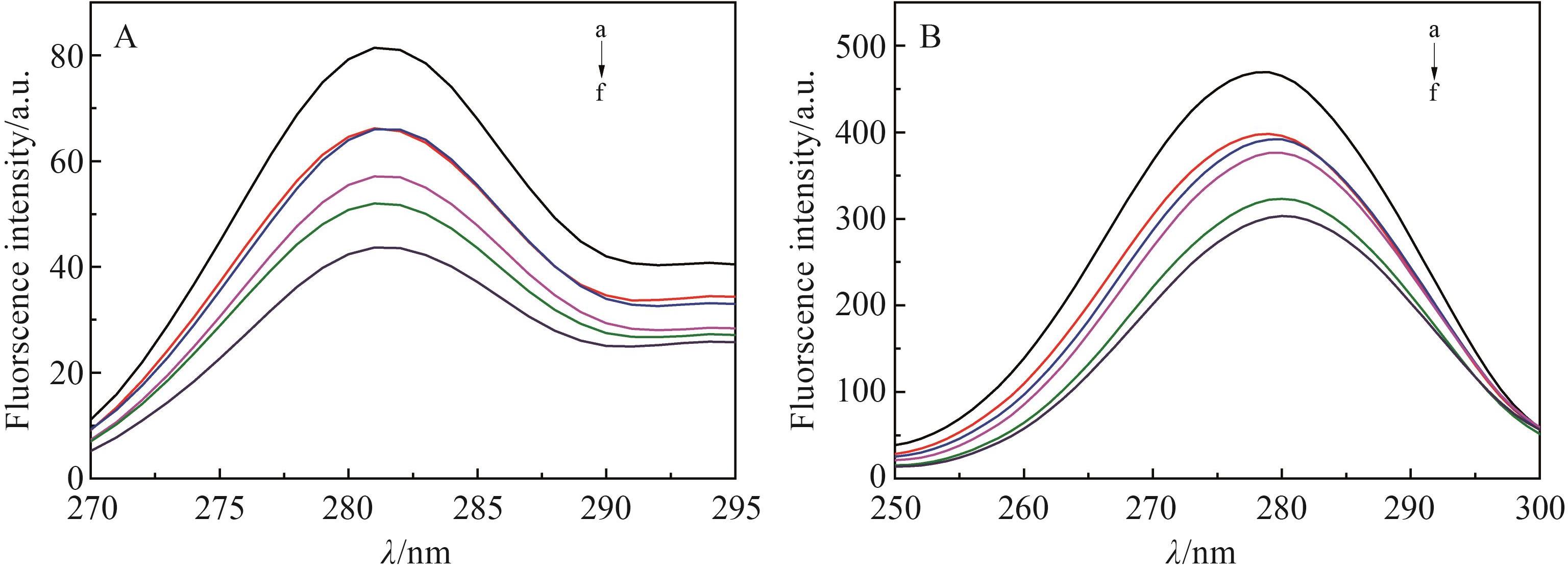
Chinese Journal of Applied Chemistry ›› 2022, Vol. 39 ›› Issue (6): 960-968.DOI: 10.19894/j.issn.1000-0518.210248
• Full Papers • Previous Articles Next Articles
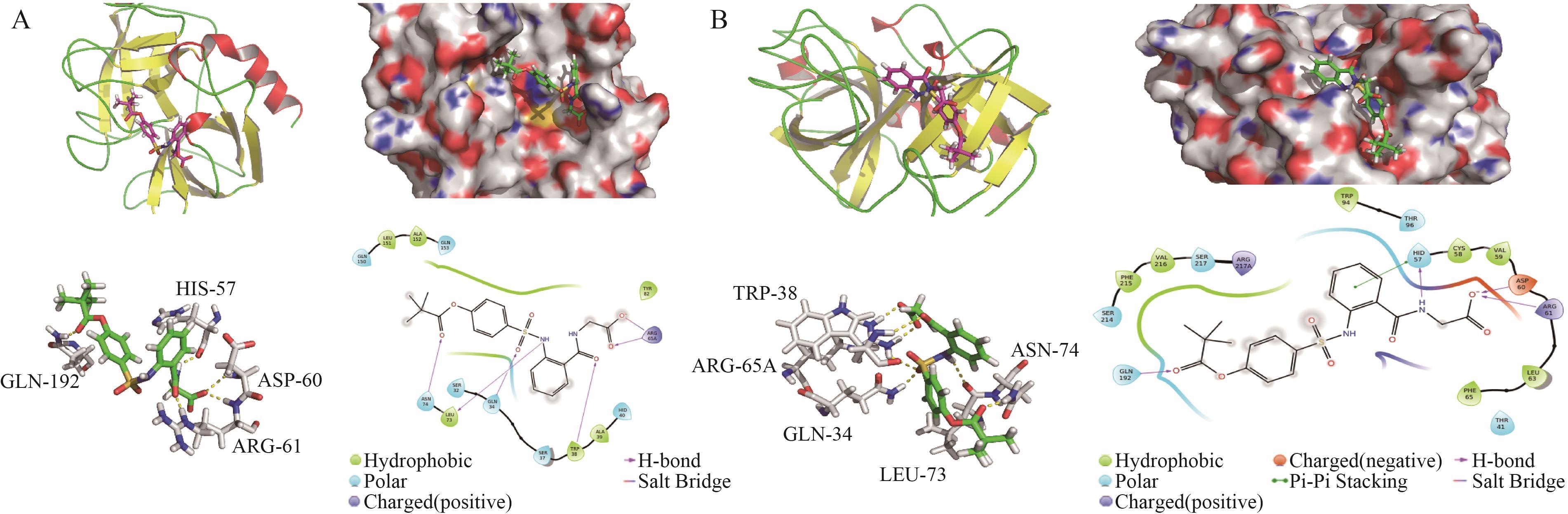
Interaction Between Sivelestat Sodium and Elastase by Multispectra and Molecular Docking
Guang-Ting YIN1, Xue-Jian ZHOU1, Hong-Liu YAO1, Jin-Feng FU1, Hong-Yu CAO2, Xue-Fang ZHENG2, Li-Hong SU1( )
)
- 1.College of Chemistry,Changchun Normal University,Changchun 130032,China
2.School of Life Science and Technology,Dalian University,Dalian 116622,China
-
Received:2021-05-19Accepted:2021-09-27Published:2022-06-01Online:2022-06-27 -
Contact:Li-Hong SU -
About author:bethsu@qq.com
-
Supported by:the Natural Science Foundation of Jilin Province of China(20140101043JC);the Foundation of Jilin Educational Committee ([2013]No.251) and Natural Science Foundation of Changchun Normal University ([2018]No.16)
CLC Number:
Cite this article
Guang-Ting YIN, Xue-Jian ZHOU, Hong-Liu YAO, Jin-Feng FU, Hong-Yu CAO, Xue-Fang ZHENG, Li-Hong SU. Interaction Between Sivelestat Sodium and Elastase by Multispectra and Molecular Docking[J]. Chinese Journal of Applied Chemistry, 2022, 39(6): 960-968.
share this article
温度 T/K | 猝灭常数 Ksv/(L·mol-1) | 猝灭速率常数Kq/(L·mol-1·s-1) | 相关系数 R2 |
|---|---|---|---|
| 298 | 2.744×104 | 2.744×1012 | 0.971 0 |
| 303 | 1.791×104 | 1.791×1012 | 0.979 1 |
| 310 | 1.620×104 | 1.620×1012 | 0.985 6 |
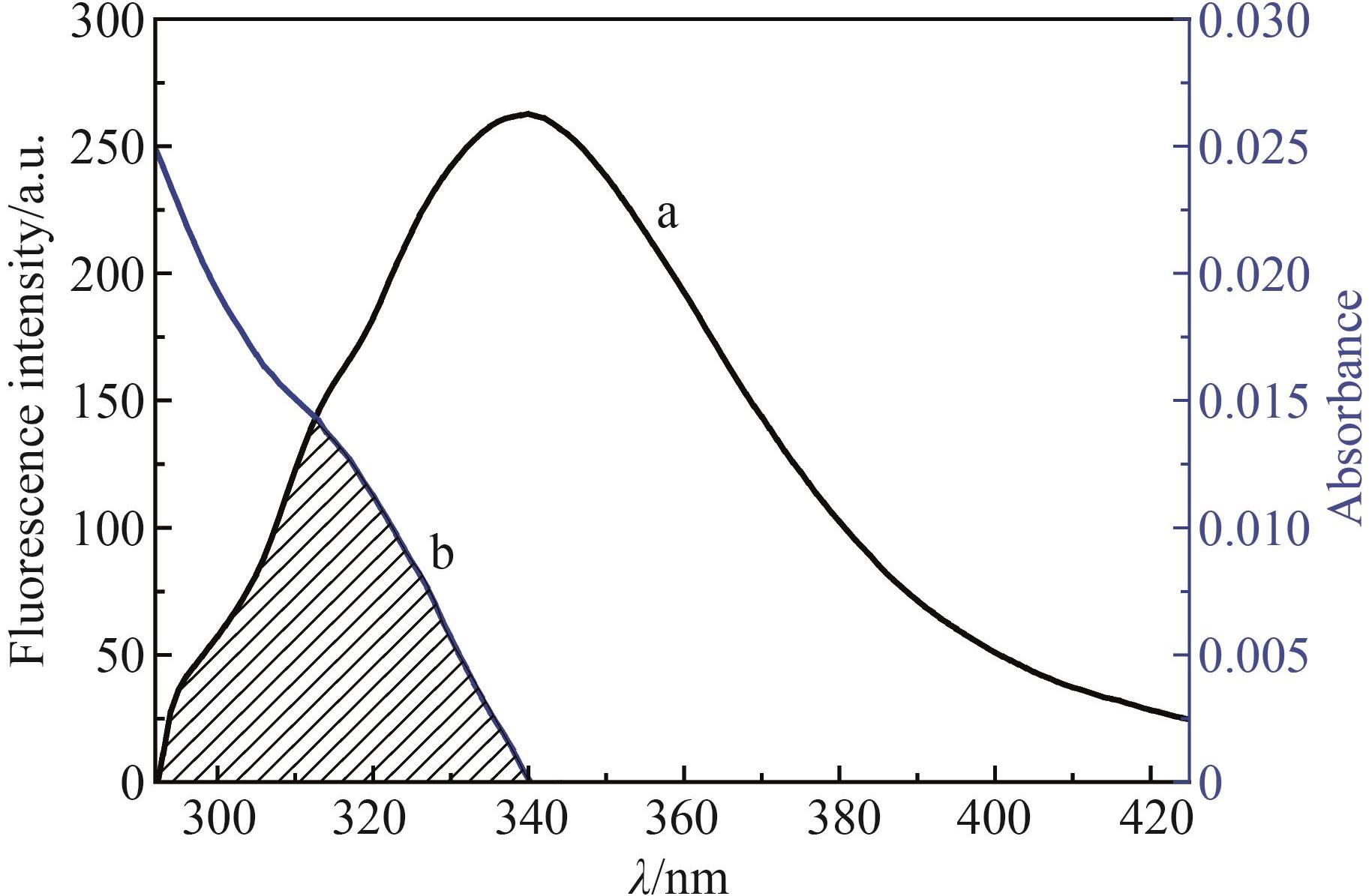
Table 1 Quenching rate constants and correlation coefficients of ONO?5046 and PPE
温度 T/K | 猝灭常数 Ksv/(L·mol-1) | 猝灭速率常数Kq/(L·mol-1·s-1) | 相关系数 R2 |
|---|---|---|---|
| 298 | 2.744×104 | 2.744×1012 | 0.971 0 |
| 303 | 1.791×104 | 1.791×1012 | 0.979 1 |
| 310 | 1.620×104 | 1.620×1012 | 0.985 6 |
温度 T/K | 结合常数 Ka/(L·mol-1) | 结合位点数 n | 自由能 ΔG/(kJ·mol-1) | 焓 ΔH/(kJ·mol-1) | 熵 ΔS/(J·mol-1·K-1) |
|---|---|---|---|---|---|
| 298 | 3.274×104 | 1.318 6 | -25.757 | -51.061 | -86.021 |
| 303 | 1.866×104 | 1.198 2 | -24.773 | -51.061 | -86.021 |
| 310 | 1.391×104 | 0.829 3 | -24.589 | -51.061 | -86.021 |
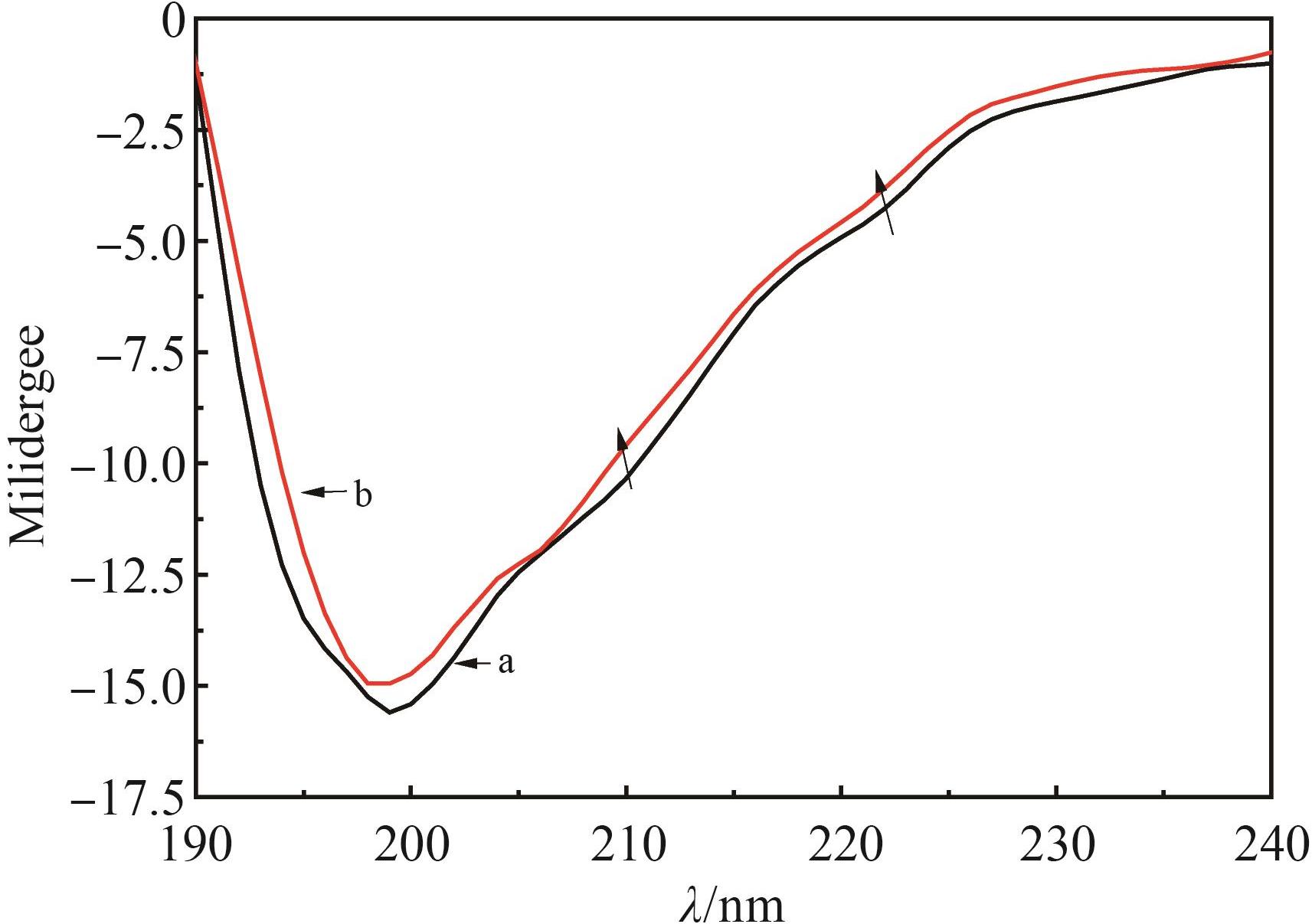
Table 2 Binding constant and thermodynamic constant of ONO?5046 and PPE
温度 T/K | 结合常数 Ka/(L·mol-1) | 结合位点数 n | 自由能 ΔG/(kJ·mol-1) | 焓 ΔH/(kJ·mol-1) | 熵 ΔS/(J·mol-1·K-1) |
|---|---|---|---|---|---|
| 298 | 3.274×104 | 1.318 6 | -25.757 | -51.061 | -86.021 |
| 303 | 1.866×104 | 1.198 2 | -24.773 | -51.061 | -86.021 |
| 310 | 1.391×104 | 0.829 3 | -24.589 | -51.061 | -86.021 |
| 1 | LI L Y, HUANG Q H, WANG D C, et al. Acute lung injury in patients with COVID-19 infection[J]. Clin Transl Med, 2020, 10(1): 20-27. |
| 2 | JOANNA S M, KATARZYNA Z, OKTAWIA O, et al. Differences in staining for neutrophil elastase and its controlling inhibitor SLPI reveal heterogeneity among neutrophils in psoriasis[J]. J Invest Dermatol, 2019, 140(7): 1371-1378. |
| 3 | RUAN Q, YANG K, WANG W, et al. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China[J]. Intensive Care Med, 2020, 46: 846-848. |
| 4 | YU X F, ZHAO L J, YU Z P, et al. Sivelestat sodium hydrate improves post-traumatic knee osteoarthritis through nuclear factor-κB in a rat model[J]. Exp Ther Med, 2017, 14(2): 1531-1537. |
| 5 | XIAO X G, ZU H G, LI Q G, et al. Cinnoline derivatives as human neutrophil elastase inhibitors[J]. Riv Eur Sci Med Farmacol, 2016, 20(3): 528-536. |
| 6 | CHEN K Y, ZHOU K L, LOU Y Y, et al. Exploring the binding interaction of calf thymus DNA with lapatinib, a tyrosine kinase inhibitor: multi-spectroscopic techniques combined with molecular docking[J]. J Biomol Struct Dyn, 2019, 37(3): 576-583. |
| 7 | WANG Q, ZHANG S R, JI X H, et al. Investigation of interaction of antibacterial drug sulfamethoxazole with human serum albumin by molecular modeling and multi-spectroscopic method[J]. Spectrochim Acta A, 2014, 124: 84-90. |
| 8 | FAHIMAH K, MAHSA P, MAHYA S, et al. Multi-spectroscopic and molecular docking studies on the interaction of neotame with calf thymus DNA[J]. Nucleosides, Nucleotides Nucleic Acids, 2020, 39(5): 699-714. |
| 9 | KABIRU A M, TING N, SAHARUDDIN B M, et al. Intermolecular recognition between pyrimethamine, an antimalarial drug and human serum albumin: spectroscopic and docking study[J]. J Mol Liq, 2020, 311: 658-664. |
| 10 | YANG R, YU L L,ZENG H J, et al. The interaction of flavonoid-lysozyme and the relationship between molecular structure of flavonoids and their binding activity to lysozyme [J]. J Fluoresc, 2012, 22(6): 1449-1459. |
| 11 | RAZIQUE A, KHALID I A, SHAFIUL H, et al. Unravelling the interaction of glipizide with human serum albumin using various spectroscopic techniques and molecular dynamics studies[J]. J Biomol Struct Dyn, 2021, 39(1): 336-347. |
| 12 | 张红梅, 王彦卿, 张根成. 荧光光谱法研究茶碱与牛血清白蛋白的相互作用[J]. 应用化学, 2006, 23(8): 866-870. |
| ZHANG H M, WANG Y Q, ZHANG G C. Study of interaction between theophylline and bovine serum album in by fluorescence spectroscopy[J]. Chinese J Appl Chem, 2006, 23(8): 866-870. | |
| 13 | MUNTJEEB M S, POOJA J D, MOHAN V K. Alizarin interaction with sickle hemoglobin: elucidation of their anti-sickling properties by multi-spectroscopic and molecular modeling techniques[J]. J Biomol Struct Dyn, 2019, 37(17): 4614-4631. |
| 14 | 郭兴家, 郭闯, 姜玉春, 等. 光谱法研究氨基己酸与牛血清白蛋白的相互作用[J]. 应用化学, 2010, 27(10): 1192-1198. |
| GUO X J, GUO C, JIANG Y C, et al. Spectroscopic studies on the interaction between amidocaproic acid and bovine serum albumin[J]. Chinese J Appl Chem, 2010, 27(10): 1192-1198. | |
| 15 | 吕茜茜, 高苏亚, 夏冬辉, 等. 荧光光谱法研究双醋瑞因与人血清白蛋白的相互作用[J]. 应用化学, 2011, 28(7): 836-841. |
| LV Q Q, GAO S Y, XIA D H, et al. Fluorescence spectroscopy on the interaction of diacerein and human serum albumin[J]. Chinese J Appl Chem, 2011, 28(7): 836-841. | |
| 16 | SWARUP R. An insight of binding interaction between tryptophan, tyrosine and phenylalanine separately with green gold nanoparticles by fluorescence quenching method[J]. Opt Morya, 2017, 138: 280-288. |
| 17 | ZOU W, WANG M, ZHU Z Z, et al. Fluorescence and circular dichroism spectroscopy to understand the interactions between cyclodextrins and α-galactosidase from green coffee beans[J]. Food Biosci, 2017, 20: 110-115. |
| [1] | Xing-Yu TAO, Xuan HU, Tong-Chuan WU, Fei ZHENG, Hao YUE, Yu-Lin DAI. Research Progress on the Application and Function of Portulaca Oleracea L. in Cosmetics [J]. Chinese Journal of Applied Chemistry, 2023, 40(6): 820-832. |
| [2] | Xi-Yu SONG, Lin-Bo GAO, Li HOU, Ai-Qin HOU, Hong-Fei QIAN. Synthesis and azo-Hydrazone Heterochromia Mechanism of Disperse Dyes Containing Pyridone Groups [J]. Chinese Journal of Applied Chemistry, 2023, 40(3): 430-435. |
| [3] | Ling-Yu ZHANG, Su-Xin HOU, Wen-Wei ZHANG, Shan JIANG, Jun-Tong LIU, Hao YUE, Nan ZHANG. Research Progress on Chemical Constituents and Extraction and Separation of Lonicera edulis [J]. Chinese Journal of Applied Chemistry, 2022, 39(11): 1629-1640. |
| [4] | GAO Zi-Ting, LI Zhan-Chao, JIANG Qian, GAO Jun-Li, LIU Xing-Yu, WANG Zi-Ming. A Method for Preparing Environment-friendly Snow-melting Agent from Wood Vinegar [J]. Chinese Journal of Applied Chemistry, 2021, 38(8): 1022-1024. |
| [5] | LI Zi-Yue, YANG Tong-Ren, YANG Ge, HUANG Yuan-Yu. Research Progresses of Nucleic Acid Based Detection of Pathogenic Microorganisms [J]. Chinese Journal of Applied Chemistry, 2021, 38(5): 592-604. |
| [6] | TUO Suxing*, WU Mingjian, DAI Yunhui. Reducing Polycyclic Aromatic Hydrocarbons in Cigarette Smoke by Pyrene Molecular Imprinted Polymers [J]. Chinese Journal of Applied Chemistry, 2014, 31(01): 89-95. |
| [7] | WU Yunxia1,2, HUANG Jing1,2, YIN Zhengzhi2, ZENG Yanbo2, ZHANG Zulei2, LI Lei2*. Properties of Rhodamine B′ Magnetic Molecularly Imprinted Polymers Prepared from the Suspension Polymerization Method [J]. Chinese Journal of Applied Chemistry, 2013, 30(12): 1481-1488. |
| [8] | LI Junfen1, LI Jinzeng1, ZHANG Caihong1, HAN Yuying1, QIN Xi1, DONG Chuan2*. Synthesis and Luminescence Behavior of a Novel Charge Transfer Compound Containing Carbazole Phenylhydrazone [J]. Chinese Journal of Applied Chemistry, 2013, 30(10): 1139-1143. |
| [9] | XUE Shuping1*, WANG Haijuan2. Synthesis and Structure of H2O to O2 of [M (2,2'–bpy)2]2SiW12O40(M = Mn , Co; bpy=bipyridine) and Its Electrocatalytic Oxidation [J]. Chinese Journal of Applied Chemistry, 2013, 30(03): 305-309. |
| [10] | XIONG Xiaoqin*, ZHAI Qiuge, XUE Lingfen, ZHENG Fei, XIE Xu. Synthesis of Styrylpyrimidine and Its Spectral Characteristic as a pH Probe [J]. Chinese Journal of Applied Chemistry, 2012, 29(11): 1262-1265. |
| [11] | ZENG Zhi1,2*, FU Lin1, YE Xuening1, ZHANG Tao3, MENG Shaojin1, MENG Chunyan1. Comparative of Volatile Oil Components from Fructus Amomi Rotundus, Fructus Galangae, Semen Alpiniae Katsumadai and Semen Myristicae [J]. Chinese Journal of Applied Chemistry, 2012, 29(11): 1316-1323. |
| [12] | ZHANG Daihui1,2*, LU Lijun1, ZHANG Xun1, MU Jun1, ZHOU Xiao1, HU Tingting1, HAN Dachuan1. Simultaneous Determination of Ten Kinds of Perfluorinated Compounds in Lubricant Samples by High-performance Liquid Chromatography-Tandem Mass Spectrometry [J]. Chinese Journal of Applied Chemistry, 2012, 29(10): 1163-1170. |
| [13] | ZENG Zhi1,2*, YE Xuening1, PANG Shimin1, ZHANG Tao2,3, SHEN Miaoting1. Comparison of the Volatile Oil Components from Atractylodes chinensis (DC.) Koidz. and Atractylodes lancea (Thunb.) DC. [J]. Chinese Journal of Applied Chemistry, 2012, 29(04): 470-476. |
| [14] | ZENG Zhi1,2*, XIE Runqian1, TAN Lixian1, ZHANG Tao3. GC-MS Analysis and Determination of Chemical Components of the Extracts from Ligusticum Chuanxiong Hort. by Steam Distillation and Supercritical CO2 Extraction [J]. Chinese Journal of Applied Chemistry, 2011, 28(08): 956-962. |
| [15] | ZHANG Yuan, LI Yang, CHEN Bo, ZHU Bingyu, LIU Feng*. Effects of Bentonite Modified by Organic Additives on the Physical Stability of Hexaconazole Suspension [J]. Chinese Journal of Applied Chemistry, 2011, 28(05): 565-570. |
| Viewed | ||||||||||||||||||||||||||||||||||||||||||||||||||
|
Full text 408
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|
Abstract 530
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||