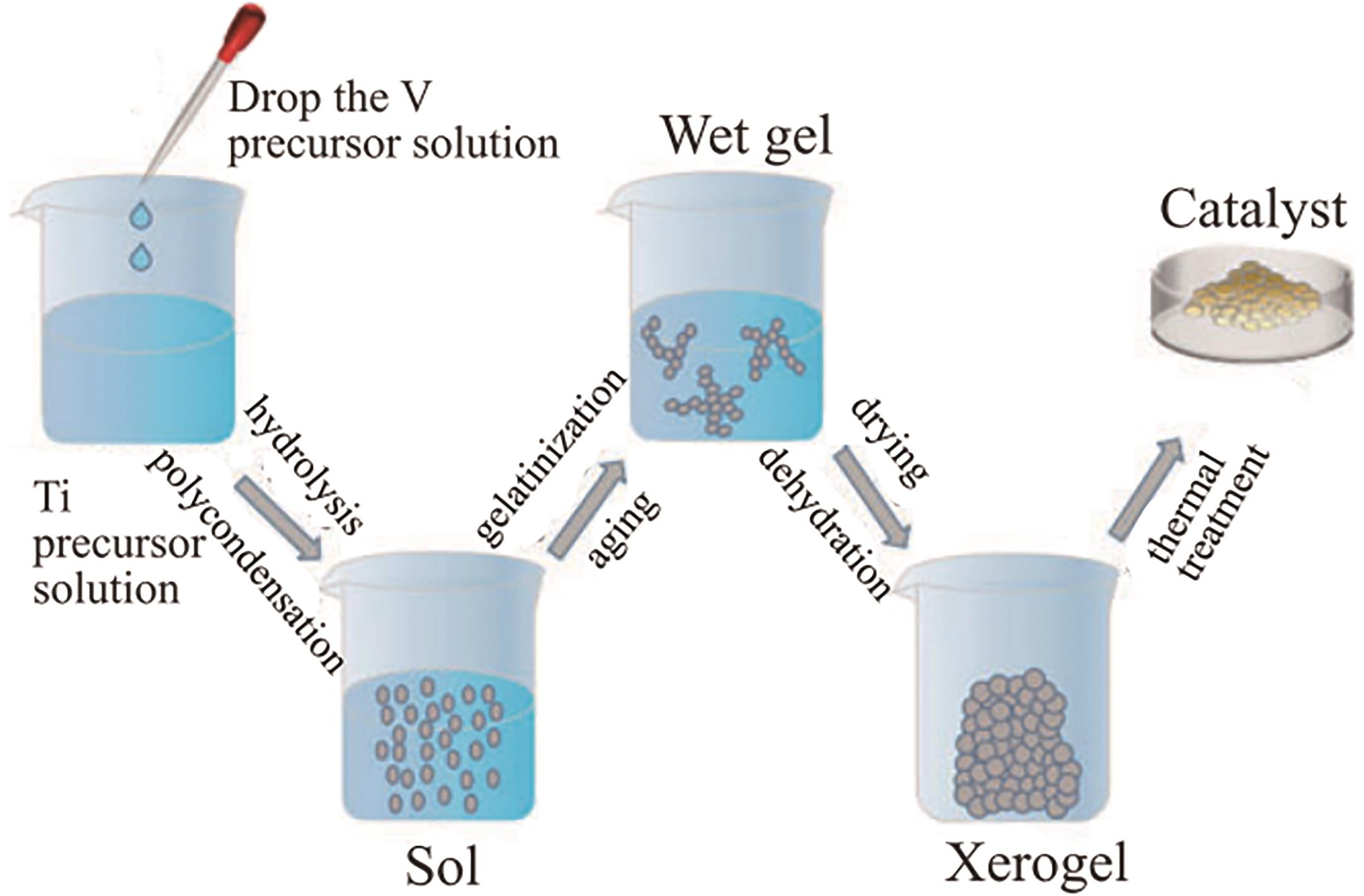
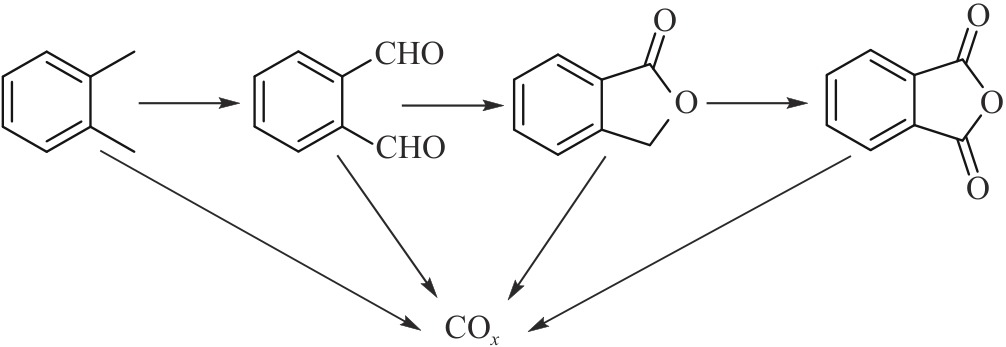
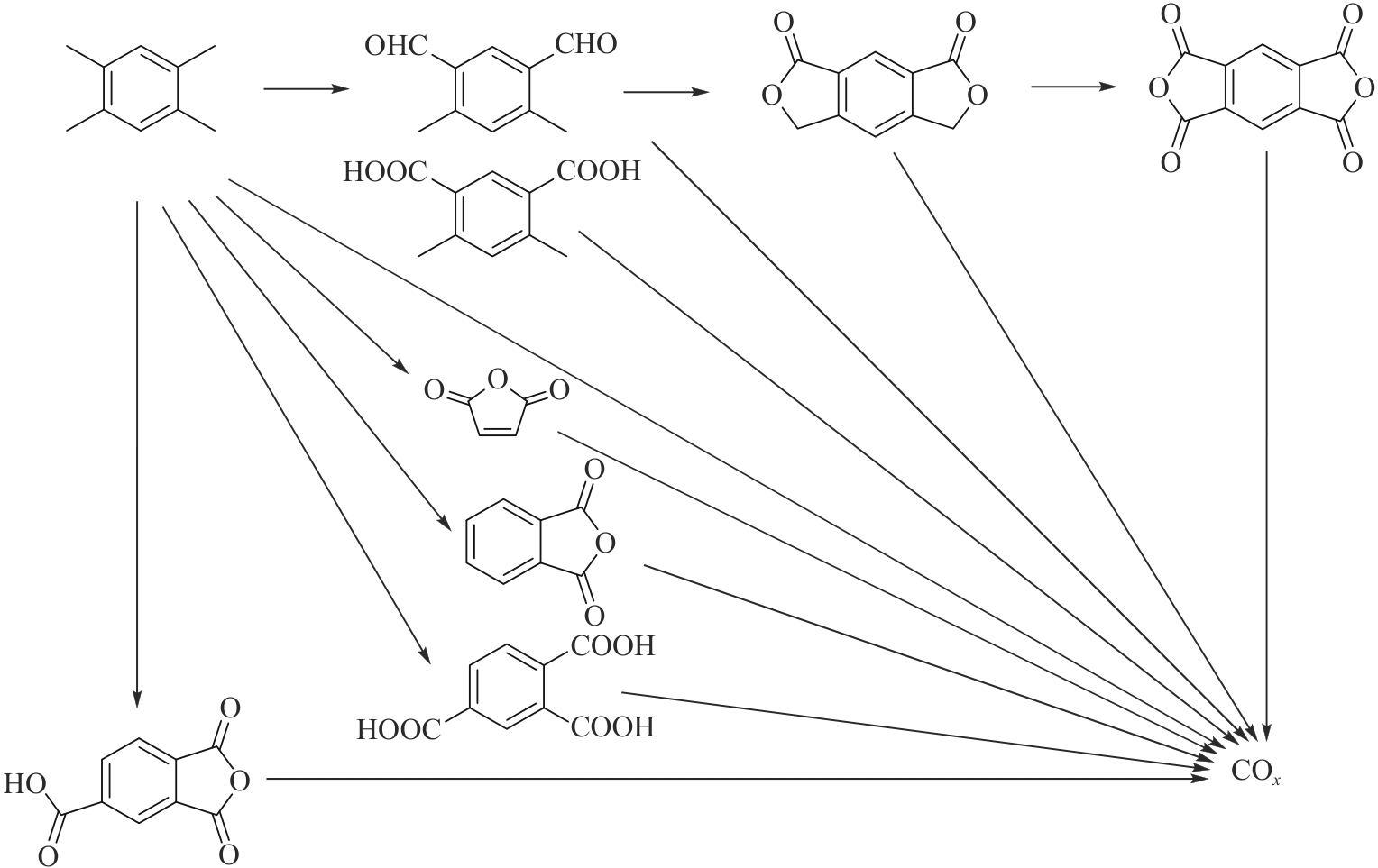
应用化学 ›› 2026, Vol. 43 ›› Issue (2): 167-181.DOI: 10.19894/j.issn.1000-0518.250143
• 综合评述 • 上一篇
均四甲苯选择性氧化制备均苯四甲酸二酐的研究进展
张国辉1, 贺滨2, 章伟3, 王秀2, 张昊天4, 罗琛4, 闫瑞一2( ), 周明东1(
), 周明东1( ), 刘瑞霞2
), 刘瑞霞2
- 1.沈阳化工大学化学工程学院,沈阳 110142
2.中国科学院过程工程研究所,北京 100190
3.(中国石油大学(北京)化学工程与环境学院,北京 102249 )
4.中国石油石油化工研究院,北京 102206
-
收稿日期:2025-04-17接受日期:2025-10-10出版日期:2026-02-01发布日期:2026-03-06 -
通讯作者:闫瑞一,周明东 -
基金资助:中国科学院战略性先导科技专项(XDA0390302);国家自然科学基金(22222813)
Research Progress in Selective Oxidation of Durene to Pyromellitic Acid Dianhydride
Guo-Hui ZHANG1, Bin HE2, Wei ZHANG3, Xiu WANG2, Hao-Tian ZHANG4, Chen LUO4, Rui-Yi YAN2( ), Ming-Dong ZHOU1(
), Ming-Dong ZHOU1( ), Rui-Xia LIU2
), Rui-Xia LIU2
- 1.School of Chemical Engineering,Shenyang University of Chemical Technology,Shenyang 110142,China
2.Institute of Process Engineering,Chinese Academy of Sciences,Beijing 100190,China
3.(School of Chemical Engineering and Environment,China University of Petroleum (Beijing),Beijing 102249,China )
4.Petrochina Petrochemical Research Institute,Beijing 102206,China
-
Received:2025-04-17Accepted:2025-10-10Published:2026-02-01Online:2026-03-06 -
Contact:Rui-Yi YAN,Ming-Dong ZHOU -
About author:mingdong.zhou@syuct.edu.cn
ryyan@ipe.ac.cn;
-
Supported by:the Strategic Priority Research Program of the Chinese Academy of Sciences(XDA0390302);the National Natural Science Foundation of China(22222813)
摘要:
均四甲苯气相氧化法是当前生产均苯四甲酸二酐(PMDA)的主流生产工艺。 本文系统总结了均四甲苯选择性氧化制备PMDA所用催化剂的研究进展。 首先,从催化剂的结构与性能强化策略出发,全面总结了通过调控掺杂元素、优化合成方法等手段提升PMDA收率的研究成果。 其次,分析了反应机理、催化剂体相组成和载体类型等对催化性能的影响。 与此同时,从反应条件角度(如均四甲苯进料负荷、反应温度及空速等)讨论了各参数对催化效率的调控作用,为高效催化剂的开发及工艺优化提供了理论支持与实践参考,对于推动PMDA相关行业的发展具有重要的意义。
中图分类号:
引用本文
张国辉, 贺滨, 章伟, 王秀, 张昊天, 罗琛, 闫瑞一, 周明东, 刘瑞霞. 均四甲苯选择性氧化制备均苯四甲酸二酐的研究进展[J]. 应用化学, 2026, 43(2): 167-181.
Guo-Hui ZHANG, Bin HE, Wei ZHANG, Xiu WANG, Hao-Tian ZHANG, Chen LUO, Rui-Yi YAN, Ming-Dong ZHOU, Rui-Xia LIU. Research Progress in Selective Oxidation of Durene to Pyromellitic Acid Dianhydride[J]. Chinese Journal of Applied Chemistry, 2026, 43(2): 167-181.
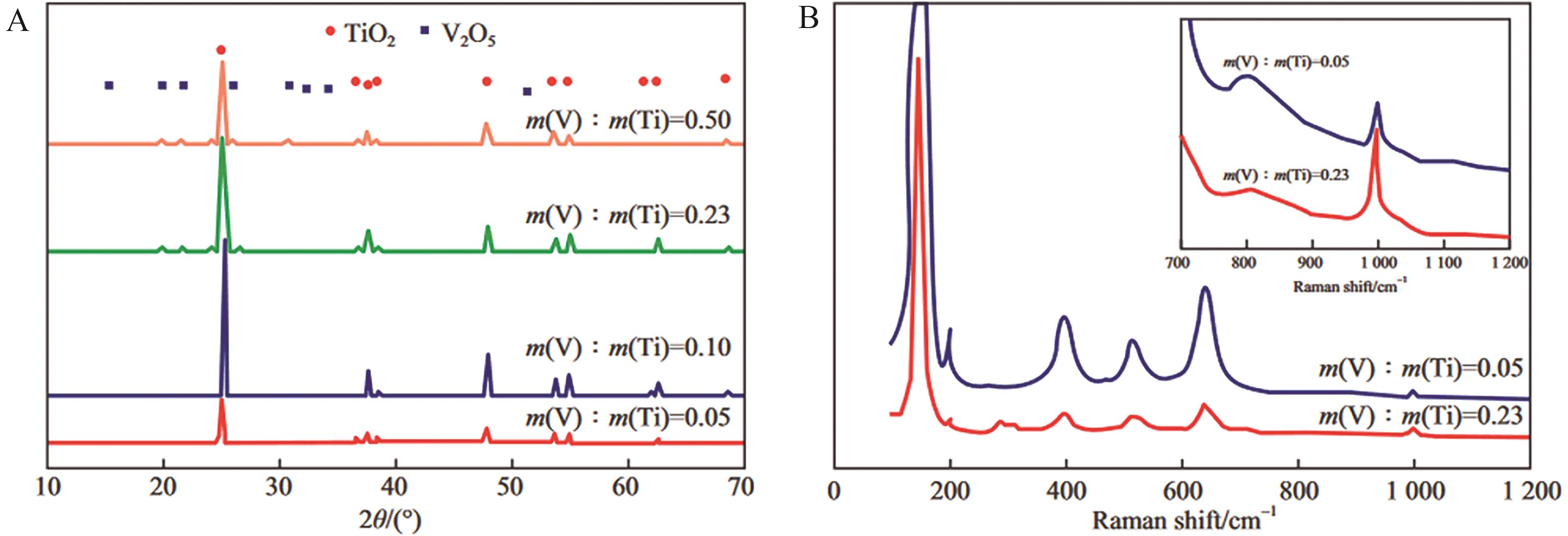
图1 (A)不同m(V)/m(Ti)催化剂的XRD谱图及(B) m(V)/m(Ti)为0.05和0.23催化剂的拉曼谱图[16]
Fig.1 (A) XRD patterns of catalysts with different m(V)/m(Ti) and (B) Raman spectra of catalysts with m(V)/m(Ti) equal to 0.05 and 0.23[16]
| Material | |||||
|---|---|---|---|---|---|
| a1 | b1 | c1 | d1 | ||
| PMDA | -2 200.08 | -0.16 | 8.354×10-5 | -1.84×10-8 | -7.81×10-10 |
| CO2 | -5 568.89 | -1.141×10-2 | -1.1629×10-4 | 7.51×10-8 | -1.67×10-11 |
| PA | -2 471.11 | 0.03 | 3.474×10-5 | -6.144×10-8 | 2.465×10-11 |
| CO | -2 719.96 | 0.257 | -3.31×10-4 | 1.134×10-7 | -1.029×10-8 |
| TMA | -2 296.42 | -0.14 | 4.053×10-5 | -1.53×10-8 | -5.15×10-12 |
| MA | -3 149.11 | -0.0334 | 5.673×10-5 | -7.378×10-8 | 2.775×10-11 |
| 4,6-DMIPA | -1 150.51 | -0.133 | 1.447×10-5 | -4.611×10-9 | -1.66×10-12 |
表1 各物质反应焓变与温度关系式[40]
Table 1 Relationship between reaction enthalpy and temperature of each substance[40]
| Material | |||||
|---|---|---|---|---|---|
| a1 | b1 | c1 | d1 | ||
| PMDA | -2 200.08 | -0.16 | 8.354×10-5 | -1.84×10-8 | -7.81×10-10 |
| CO2 | -5 568.89 | -1.141×10-2 | -1.1629×10-4 | 7.51×10-8 | -1.67×10-11 |
| PA | -2 471.11 | 0.03 | 3.474×10-5 | -6.144×10-8 | 2.465×10-11 |
| CO | -2 719.96 | 0.257 | -3.31×10-4 | 1.134×10-7 | -1.029×10-8 |
| TMA | -2 296.42 | -0.14 | 4.053×10-5 | -1.53×10-8 | -5.15×10-12 |
| MA | -3 149.11 | -0.0334 | 5.673×10-5 | -7.378×10-8 | 2.775×10-11 |
| 4,6-DMIPA | -1 150.51 | -0.133 | 1.447×10-5 | -4.611×10-9 | -1.66×10-12 |
| Material | ||||||
|---|---|---|---|---|---|---|
| a2 | b2 | c2 | d2 | e2 | ||
| PMDA | -2 200.08 | -0.427 | 0.164 | -8.355×10-5 | 9.372×10-9 | 2.605×10-10 |
| CO2 | -5 568.89 | -0.089 5 | 1.141×10-2 | 1.162 9×10-4 | -3.76×10-8 | 5.58×10-12 |
| PA | -2 471.11 | -0.272 | -0.031 2 | -3.474×10-5 | 3.072×10-8 | -8.21×10-12 |
| CO | -2 719.96 | -0.242 | -0.257 | 3.31×10-4 | -5.673×10-8 | 3.432×10-9 |
| TMA | -2 296.42 | -0.749 | 0.135 | -4.053×10-5 | 7.67×10-9 | 1.72×10-12 |
| MA | -3 149.11 | -0.025 | 0.033 4 | -5.673×10-5 | 3.689×10-8 | -9.25×10-12 |
| 4,6-DMIPA | -1 014.85 | -0.531 | 0.032 3 | 1.872 9×10-4 | -2.257×10-8 | -0.3×10-13 |
表2 各物质吉布斯自由能变与温度关系式[40]
Table 2 The relationship between Gibbs free energy change and temperature of each substance[40]
| Material | ||||||
|---|---|---|---|---|---|---|
| a2 | b2 | c2 | d2 | e2 | ||
| PMDA | -2 200.08 | -0.427 | 0.164 | -8.355×10-5 | 9.372×10-9 | 2.605×10-10 |
| CO2 | -5 568.89 | -0.089 5 | 1.141×10-2 | 1.162 9×10-4 | -3.76×10-8 | 5.58×10-12 |
| PA | -2 471.11 | -0.272 | -0.031 2 | -3.474×10-5 | 3.072×10-8 | -8.21×10-12 |
| CO | -2 719.96 | -0.242 | -0.257 | 3.31×10-4 | -5.673×10-8 | 3.432×10-9 |
| TMA | -2 296.42 | -0.749 | 0.135 | -4.053×10-5 | 7.67×10-9 | 1.72×10-12 |
| MA | -3 149.11 | -0.025 | 0.033 4 | -5.673×10-5 | 3.689×10-8 | -9.25×10-12 |
| 4,6-DMIPA | -1 014.85 | -0.531 | 0.032 3 | 1.872 9×10-4 | -2.257×10-8 | -0.3×10-13 |
| Temperature/℃ | Overall conversion/% | Yield(PMDA)/% | Yield(CO2)/% | Yield(other)/% |
|---|---|---|---|---|
| 410 | 97.04 | 48.30 | 18.59 | 30.15 |
| 420 | 98.11 | 49.47 | 20.23 | 28.41 |
| 435 | 99.04 | 54.55 | 24.50 | 20.09 |
| 445 | 99.17 | 58.30 | 28.49 | 13.27 |
表3 均四甲苯氧化反应主要物质转化率或收率数据[43]
Table 3 Conversion or yield of main substances during durene oxidation[43]
| Temperature/℃ | Overall conversion/% | Yield(PMDA)/% | Yield(CO2)/% | Yield(other)/% |
|---|---|---|---|---|
| 410 | 97.04 | 48.30 | 18.59 | 30.15 |
| 420 | 98.11 | 49.47 | 20.23 | 28.41 |
| 435 | 99.04 | 54.55 | 24.50 | 20.09 |
| 445 | 99.17 | 58.30 | 28.49 | 13.27 |
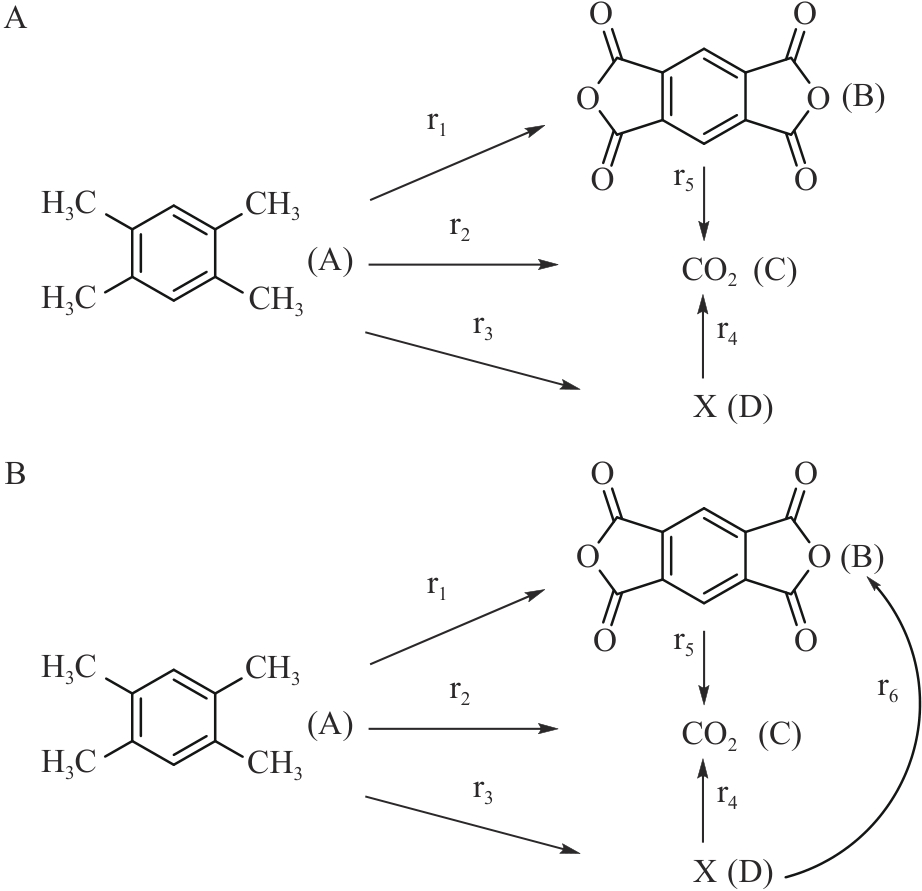
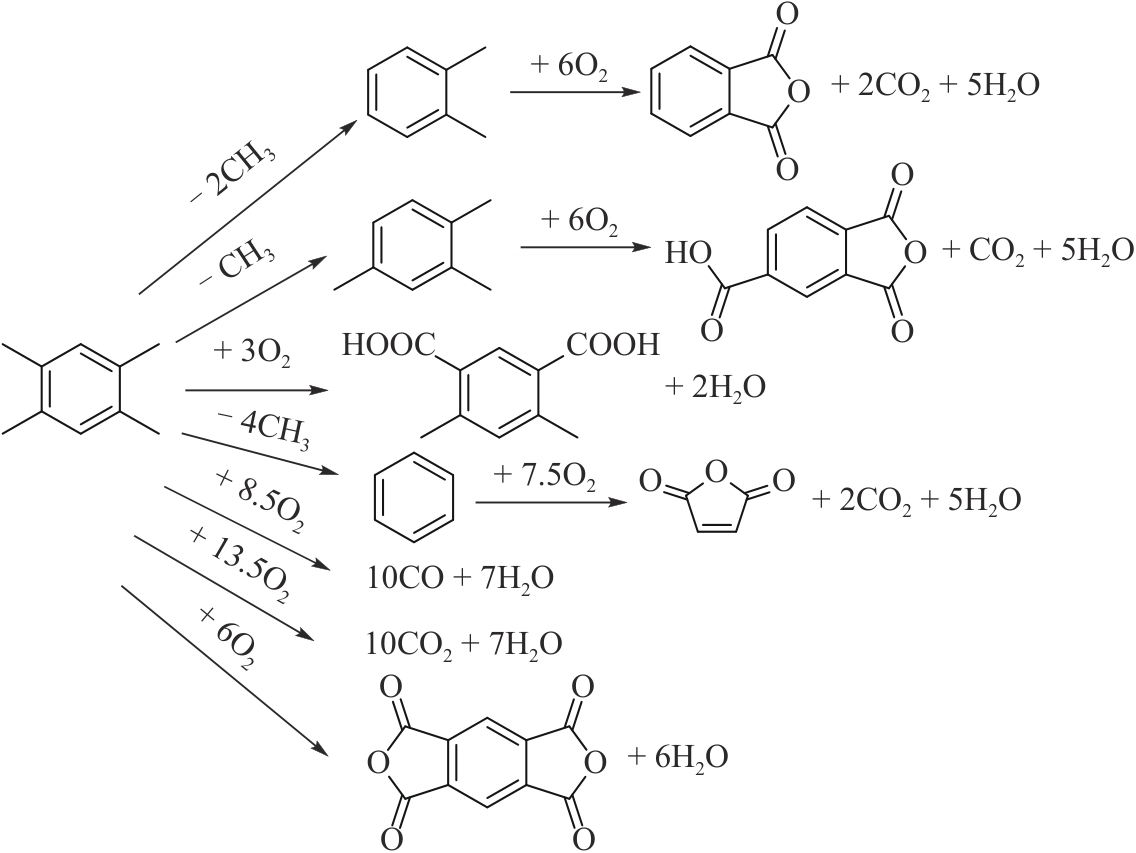
图6 (A)均四甲苯氧化制PMDA简化反应网络1[43]; (B)均四甲苯氧化制PMDA简化反应网络2[49]
Fig.6 (A) Reaction network 1 of durene to PMDA by oxidation[43]; (B) Reaction network 2 of durene to PMDA by oxidation[49]
| T≤673 K | 673 K≤T≤718 K | T≥718 K |
|---|---|---|
| k1=exp(-12 009.16/T+12.93) | k1=exp(-11 380.93/T+11.95) | k1=exp(-3 513.60/T+0.81) |
| k2=exp(-10 390.91/T+8.81) | k2=exp(-10 412.77/T+8.80) | k2=exp(-5 591.02/T+2.22) |
| k3=exp(-4 611.90/T+2.00) | k3=exp(-2 437.22/T-1.11) | k3=exp(-12 616.25/T+13.24) |
| k4=exp(-13 573.3/T+13.36) | k4=exp(-14 001.32/T+13.55) | k4=exp(-13 324.01/T+12.25) |
| k5=exp(-22 534.7/T+11.16) | k5=exp(-13 522.9/T+11.15) | k5=exp(-15 462.02/T+12.86) |
表4 旧反应网络的反应动力学参数[40]
Table 4 Reaction kinetic parameters of the old reaction network[40]
| T≤673 K | 673 K≤T≤718 K | T≥718 K |
|---|---|---|
| k1=exp(-12 009.16/T+12.93) | k1=exp(-11 380.93/T+11.95) | k1=exp(-3 513.60/T+0.81) |
| k2=exp(-10 390.91/T+8.81) | k2=exp(-10 412.77/T+8.80) | k2=exp(-5 591.02/T+2.22) |
| k3=exp(-4 611.90/T+2.00) | k3=exp(-2 437.22/T-1.11) | k3=exp(-12 616.25/T+13.24) |
| k4=exp(-13 573.3/T+13.36) | k4=exp(-14 001.32/T+13.55) | k4=exp(-13 324.01/T+12.25) |
| k5=exp(-22 534.7/T+11.16) | k5=exp(-13 522.9/T+11.15) | k5=exp(-15 462.02/T+12.86) |
| T≤673 K | 673 K≤T≤718 K | T≥718 K |
|---|---|---|
| k1=exp(-8 745.97/T+7.84) | k1=exp(-11 927.81/T+12.68) | k1=exp(-11 340.92/T+11.33) |
| k2=exp(-8 972.36/T+6.73) | k2=exp(-10 485.31/T+9.06) | k2=exp(-9 568.20/T+7.87) |
| k3=exp(-7 474.8/T+6.35) | k3=exp(-5 268.545/T+2.96) | k3=exp(-5 368.29/T+3.42) |
| k4=exp(-2 763.76/T-3.08) | k4=exp(-25 002.71/T+14.10) | k4=exp(-24 055.81/T+14.10) |
| k5=exp(-15 038.1/T+13.46) | k5=exp(-2 389.95/T-3.49) | k5=exp(-12 217.32/T+10.41) |
| k6=exp(-4 980.63/T-0.65) | k6=exp(-14 958.48/T+15.04) | k6=exp(-9 357.71/T+6.91) |
表5 新反应网络的反应动力学参数[40]
Table 5 Reaction kinetic parameters of the new reaction network[40]
| T≤673 K | 673 K≤T≤718 K | T≥718 K |
|---|---|---|
| k1=exp(-8 745.97/T+7.84) | k1=exp(-11 927.81/T+12.68) | k1=exp(-11 340.92/T+11.33) |
| k2=exp(-8 972.36/T+6.73) | k2=exp(-10 485.31/T+9.06) | k2=exp(-9 568.20/T+7.87) |
| k3=exp(-7 474.8/T+6.35) | k3=exp(-5 268.545/T+2.96) | k3=exp(-5 368.29/T+3.42) |
| k4=exp(-2 763.76/T-3.08) | k4=exp(-25 002.71/T+14.10) | k4=exp(-24 055.81/T+14.10) |
| k5=exp(-15 038.1/T+13.46) | k5=exp(-2 389.95/T-3.49) | k5=exp(-12 217.32/T+10.41) |
| k6=exp(-4 980.63/T-0.65) | k6=exp(-14 958.48/T+15.04) | k6=exp(-9 357.71/T+6.91) |
| [1] | 赵帆, 张刚, 秦少雄, 等. 均苯四甲酸二酐的性质及应用[J]. 精细石油化工, 1998(4): 53-55. |
| ZHAO F, ZHANG G, QIN S X, et al. Properties and applications of pyromellitic acid dianhydride[J]. Adv Fine Petrochem, 1998(4): 53-55. | |
| [2] | 姚宁, 李俊华, 苗伟涛. 均苯四甲酸二酐催化剂制备的研究进展[J]. 河南化工, 2020, 37(2): 9-12. |
| YAO N, LI J H, MIAO W T. Research progress in preparation of pyromellitic acid dianhydride catalyst[J]. Henan Chem Ind, 2020, 37(2): 9-12. | |
| [3] | GOUZMAN I, GROSSMAN E, VERKER R, et al. Advances in polyimide-based materials for space applications[J]. Adv Mater, 2019, 31(18): 1807738. |
| [4] | 任小龙, 董占林, 张俊丽, 等. 国外聚酰亚胺薄膜产品及应用进展[J]. 绝缘材料, 2013, 46(3): 28-32. |
| REN X L, DONG Z L, ZHANG J L, et al. Foreign polyimide film products and application progress[J]. Insul Mater, 2013, 46(3): 28-32. | |
| [5] | 陈韶辉, 刘经伟, 李泽壮, 等. 均苯四甲酸二酐的合成工艺研究进展[J]. 石油化工, 2020, 49(4): 398-402. |
| CHEN S H, LIU J W, LI Z Z, et al. Progress in the synthesis of pyromellitic acid dianhydride[J]. Petrochem, 2020, 49(4): 398-402. | |
| [6] | 曹龙海, 王晶, 何文秀, 等. 均四甲苯在钒钛催化剂上气相氧化反应的途径和步骤[J]. 化学与粘合, 2001(6): 254-255. |
| CAO L H, WANG J, HE W X, et al. Pathways and steps of gas-phase oxidation of durene over vanadium-titanium catalysts[J]. Chem Bonding, 2001(6): 254-255. | |
| [7] | LONG S, XU L, LIU G. Preparation and modification of heterogeneous vanadium-titanium-based catalysts[J]. Russ J Gen Chem, 2016, 91(3): 464-487. |
| [8] | 丁志平, 杨晓东, 朱智清. 精均苯四甲酸二酐的研制[J]. 涂料工业, 2006(11): 38-41. |
| DING Z P, YANG X D, ZHU Z Q. Development of refined pyromellitic acid dianhydride[J]. Coat Ind, 2006(11): 38-41. | |
| [9] | 鲁爽, 李仁娜, 翁小乐. 钒基催化剂协同控制氮氧化物和含氯有机物研究进展与展望[J]. 科学通报, 2025, 70(14): 2070-2084. |
| LU S, LI R N, WENG X L. Research progress and prospect of vanadium-based catalysts for synergistic control of nitrogen oxides and chlorinated organic compounds[J]. Sci Bull, 2025, 70(14): 2070-2084. | |
| [10] | 张利杰, 曾贤君, 李贺, 等. 均四甲苯气相氧化催化剂研究进展[J]. 工业催化, 2022, 30(11): 13-18. |
| ZHANG L J, ZENG X J, LI H, et al. Advances in catalysts for gas-phase oxidation of durene[J]. Ind Catal, 2022, 30(11): 13-18. | |
| [11] | 方敏, 顾龙勤, 王昂, 等. 焙烧温度对均四甲苯气相氧化制均苯四甲酸二酐钒钛氧化物催化剂的影响[J]. 工业催化, 2024, 32(7): 49-53. |
| FANG M, GU L Q, WANG A, et al. Effect of calcination temperature on vanadium-titanium oxide catalysts for vapor-phase oxidation of mesitylene to pyromellitic dianhydride[J]. Ind Catal, 2024, 32(7): 49-53. | |
| [12] | DIAS C R, PORTELA M F, GALANFERERES M, et al. Selective oxidation of o-xylene to phthalic anhydride on V2O5 supported on TiO2-coated SiO2[J]. Catal Lett, 1997, 43(1/2): 117-121. |
| [13] | KUTEPOV B I, BAL′ZHINIMAEV B S. Reasons for the deactivation of vanadia-titania catalysts for partial durene oxidation during prolonged performance[J]. Kinet Catal, 2001, 42(3): 291-300. |
| [14] | 邓国才, 穆瑞才, 马洪涛, 等. Nd-Ti-V催化剂的制备及在均酐制备上的应用[J]. 中国稀土学报, 1998(3): 89-92. |
| DENG G C, MU R C, MA H T, et al. Preparation of Nd-Ti-V catalyst and its application in the preparation of homo-anhydride[J]. J Chin Soc Rare Earths, 1998(3): 89-92. | |
| [15] | MANUPUTTY M Y, LINDBERG C S, DREYER J A H, et al. Understanding the anatase-rutile stability in flame-made TiO2[J]. Combust Flame, 2021, 226: 347-361. |
| [16] | 方敏, 徐俊峰, 曾炜, 等. 均四甲苯气相氧化制均苯四甲酸二酐钒钛氧化物催化剂[J]. 石油化工, 2020, 49(5): 422-425. |
| FANG M, XU J F, ZENG W, et al. Vanadium titanium oxide catalyst for vapor phase oxidation of durene to pyromellitic acid dianhydride[J]. Petrochem Ind, 2020, 49(5): 422-425. | |
| [17] | 颜志鹏, 崇明本, 程党国, 等. 纯与掺杂CeO2的氧化还原性质及其催化领域的应用[J]. 化学进展, 2008(Z2): 1037-1043. |
| YAN Z P, CHONG M B, CHENG D G, et al. Redox properties of pure and doped CeO2 and their applications in catalysis[J]. Chem Prog, 2008(Z2): 1037-1043. | |
| [18] | 余鸿敏, 卢晗锋, 陈银飞. Pt掺杂对Cu-Mn-Ce复合氧化物催化燃烧性能的影响[J]. 化工学报, 2011, 62(4): 947-952. |
| YU H M, LU H F, CHEN Y F. Effect of Pt doping on the catalytic combustion performance of Cu-Mn-Ce mixed oxides[J]. Chem J, 2011, 62(4): 947-952. | |
| [19] | 甘洪宇, 冯燕, 杨德鸿, 等. 杂原子掺杂生物质碳催化丙烷直接脱氢制丙烯[J]. 无机材料学报, 2022, 37(10): 1058-1065. |
| GAN H Y, FENG Y, YANG D H, et al. Heteroatom-doped biomass carbon catalyzed direct dehydrogenation of propane to propylene[J]. Inorg Mater, 2022, 37(10): 1058-1065. | |
| [20] | 管银涛. 掺杂功能高分子材料的铁基催化剂在常温常压合成氨领域的开发与应用[D]. 济南: 山东师范大学, 2024. |
| GUAN Y T. Development and application of iron-based catalysts doped with functional polymer materials in the field of ammonia synthesis at room temperature and pressure[D]. Jinan: Shandong Normal University, 2024. | |
| [21] | 董华青, 潘西, 谢琴, 等. CO在金属掺杂TiO2纳米管阵列中的吸附及氧化[J]. 物理化学学报, 2012, 28(1): 44-50. |
| DONG H Q, PAN X, XIE Q, et al. CO adsorption and oxidation in metal-doped TiO2 nanotube arrays[J]. Acta Phys Chim Sin, 2012, 28(1): 44-50. | |
| [22] | 仓理, 杨晓东. 气相氧化法生产均苯四甲酸二酐催化剂进展[J]. 化工进展, 2008(12): 1878-1881. |
| CANG L, YANG X D. Catalyst progress in the production of pyromellitic acid dianhydride by gas phase oxidation[J]. Chem Prog, 2008(12): 1878-1881. | |
| [23] | KE D, WANG M, RUAN J, et al. Efficient, continuous oxidation of durene to pyromellitic dianhydride mediated by a V-Ti-P ternary catalyst: the remarkable doping effect[J]. Chin J Chem Eng, 2023, 55: 156-164. |
| [24] | QIU Y, LIU B, DU J, et al. The monolithic cordierite supported V2O5-MoO3/TiO2 catalyst for NH3-SCR[J]. Chem Eng J, 2016, 294: 264-272. |
| [25] | 孔杨, 张泽凯, 李浙飞, 等. 近十年来(2011-2021)钒基NH3-SCR催化剂的研究进展[J]. 工业催化, 2022, 30(12): 16-33. |
| KONG Y, ZHANG Z K, LI Z F, et al. Research progress of vanadium-based NH3-SCR catalysts in the past decade (2011-2021)[J]. Ind Catal, 2022, 30(12): 16-33. | |
| [26] | 余鼎章. 流化床邻二甲苯多相催化氧化制苯酐催化剂的改进[J]. 催化学报, 1991(3): 235-239. |
| YU D Z. Improvement of catalyst for heterogeneous catalytic oxidation of o-xylene in fluidized bed to phthalic anhydride[J]. J Catal, 1991(3): 235-239. | |
| [27] | 丁志平, 杨晓东, 朱智清. Ce-Mo-Ti-V催化制备均苯四甲酸二酐[J]. 化工进展, 2006(12): 1443-1446. |
| DING Z P, YANG X D, ZHU Z Q. Ce-Mo-Ti-V catalyzed preparation of pyromellitic acid dianhydride[J]. Chem Prog, 2006(12): 1443-1446. | |
| [28] | 王昂, 顾龙勤, 方敏, 等. 高分散Ce-VOX/TiO2均苯四甲酸二酐催化剂研究[J]. 化学反应工程与工艺, 2022, 38(3): 193-200. |
| WANG A, GU L Q, FANG M, et al. Highly dispersed Ce-VOX/TiO2 pyromellitic acid dianhydride catalyst[J]. Chem React Eng Technol, 2022, 38(3): 193-200. | |
| [29] | 张文杰, 何红波, 邓国才. 均四甲苯空气氧化制均酐[J]. 沈阳工业学院学报, 1998(3): 35-39. |
| ZHANG W J, HE H B, DENG G C. Oxidation of durene to homo-anhydride by air[J]. J Shenyang Inst Technol, 1998(3): 35-39. | |
| [30] | 刘玉芬, 安欣, 袁滨. 均苯四甲酸二酐合成催化剂的研究进展[J]. 精细与专用化学品, 2021, 29(9): 32-35. |
| LIU Y F, AN X, YUAN B. Research progress of catalysts for the synthesis of pyromellitic acid dianhydride[J]. Fine Spec Chem, 2021, 29(9): 32-35. | |
| [31] | 张淑琴, 钟占文, 王文彬, 等. 均苯四酸二酐新型催化剂的应用研究[J]. 石油化工, 1995(4): 249-251. |
| ZHANG S Q, ZHONG Z W, WANG W B, et al. Application of new catalysts for pyromellitic acid dianhydride[J]. Petrochem Ind, 1995(4): 249-251. | |
| [32] | 安欣, 袁滨, 刘玉芬. 萘气相氧化制苯酐催化剂的研究[J]. 精细与专用化学品, 2021, 29(9): 40-42. |
| AN X, YUAN B, LIU Y F. Study on catalysts for gas-phase oxidation of naphthalene to phthalic anhydride[J]. Fine Spec Chem, 2021, 29(9): 40-42. | |
| [33] | 王旭红, 汤忠俊, 柴文. 一种催化剂及其制备方法: 中国, 102319580A[P], 2012-01-18. |
| WANG X H, TANG Z J, CHAI W. A catalyst and its preparation method: CN, 102319580A[P], 2012-01-18. | |
| [34] | 姚霞喜, 惠洋, 王旭红, 等. 用于均四甲苯气相氧化制取均苯四甲酸二酐的催化剂体系: 中国, 201710916092.1[P], 2020-05-15. |
| YAO X X, HUI Y, WANG X H, et al. Catalyst system for vapor phase oxidation of durene to pyromellitic acid dianhydride: CN, 201710916092.1[P], 2020-05-15. | |
| [35] | SONG P, LI M, ZHU S, et al. Role of cerium-layered double hydroxide-derived oxide dopants in vanadium phosphorus oxide for selective oxidation of n-butane[J]. Ind Eng Chem Res, 2024, 63(44): 19329-19338. |
| [36] | YU J, ZHANG E, WANG L, et al. The interaction of NH4HSO4 with vanadium-titanium catalysts modified with molybdenum and tungsten[J]. Energy Fuels, 2020, 34(2): 2107-2116. |
| [37] | 张步江, 闫征, 刘祖愉. V2O5基催化剂的应用及催化机理[J]. 山西化工, 2008(5): 33-36. |
| ZHANG B J, YAN Z, LIU Z Y. Application and catalytic mechanism of V2O5-based catalysts[J]. Shanxi Chem Ind, 2008(5): 33-36. | |
| [38] | GRZYBOWSKA-SWIERKOSZ B. Effect of additives on the physicochemical and catalytic properties of oxide catalysts in selective oxidation reactions[J]. Top Catal, 2002, 21(1/2/3): 35-46. |
| [39] | GRZYBOWSKA-SWIERKOSZ B. Thirty years in selective oxidation on oxides: what have we learned[J]. Top Catal, 2000, 11(14): 23-42. |
| [40] | 曹茂洪. 均四甲苯选择性氧化制均苯四甲酸二酐动力学及固定床反应器的模拟研究[D]. 厦门: 厦门大学, 2014. |
| CAO M H. Kinetics of selective oxidation of durene to pyromellitic acid dianhydride and simulation of a fixed-bed reactor[D]. Xiamen: Xiamen University, 2014. | |
| [41] | MARX R, WÖLK H J, MESTL G, et al. Reaction scheme of o-xylene oxidation on vanadia catalyst[J]. Appl Catal A Gen, 2011, 398(1): 37-43. |
| [42] | DIAS C R, PORTELA M F, BOND G C. Synthesis of phthalic anhydride: catalysts, kinetics, and reaction modeling[J]. Catal Rev, 1997, 39(3): 169-207. |
| [43] | 薛来奇, 曹玲, 陈秉辉, 等. 均四甲苯氧化制二酐宏观动力学分析[J]. 化学研究与应用, 2018, 30(5): 696-700. |
| XUE L Q, CAO L, CHEN B H, et al. Macrokinetic analysis of oxidation of durene to dianhydride[J]. Chem Res Appl, 2018, 30(5): 696-700. | |
| [44] | 傅献彩. 物理化学[M]. 第五版. 北京: 高等教育出版社, 2005: 481-483. |
| FU X C. Physical chemistry[M]. Fifth Edition. Beijing: Higher Education Press, 2005: 481-483. | |
| [45] | 刘光启, 马连相, 刘杰. 化学化工物性数据手册(有机卷)[M]. 北京: 化学工业出版社, 2004: 57-58. |
| LIU G Q, MA L X, LIU J. Chemical and chemical properties data handbook (organic volume)[M]. Beijing: Chemical Industry Press, 2004: 57-58. | |
| [46] | ONGIRO A, UGURSAL V I, ALTAWEEL A M, et al. Thermodynamic simulation and evaluation of a steam CHP plant using aspen plus[J]. Appl Therm Eng, 1996, 16(3): 263-271. |
| [47] | LI Y J, ZHAO C S, DUAN L B. A thermodynamic analysis of coal combustion products in O2/CO2 atmosphere[J]. J Eng Therm Energy Power, 2007, 22(3): 332. |
| [48] | 曹玲, 薛来奇, 陈秉辉, 等. 均四甲苯氧化制均苯四甲酸二酐热力学分析和宏观动力学的研究[J]. 化学反应工程与工艺, 2018, 34(5): 467-472. |
| CAO L, XUE L Q, CHEN B H, et al. Thermodynamic analysis and macroscopic kinetics of the oxidation of durene to pyromellitic acid dianhydride[J]. Chem React Eng Technol, 2018, 34(5): 467-472. | |
| [49] | FROMENT G. Fixed-bed catalytic reactors technological and fundamental design aspects[J]. Chem Ing Technol, 1974, 46(9): 374-386. |
| [50] | CALDERBANK P, CHANDRASEKHARAN K, FUMAGALLI C. The prediction of the performance of packed-bed catalytic reactors in the air-oxidation of o-xylene[J]. Chem Eng Sci, 1977, 32(12): 1435-1443. |
| [1] | 张丹, 尚润梅, 赵振涛, 李君华, 邢锦娟. V/Ce-Al2O3催化甲醇选择性氧化制备二甲氧基甲烷[J]. 应用化学, 2022, 39(9): 1429-1436. |
| [2] | 谢志琦, 张婷, 李英崴, 王周君, 代飞, 张瑞锐, 刘瑞霞. 钒磷氧合成条件对丁烷选择性氧化催化性能的影响[J]. 应用化学, 2021, 38(4): 414-421. |
| [3] | 陈川,徐迎迪,薛叙明,周鹏鹏. 金属卟啉/MgAl水滑石催化醇选择性氧化制备羰基化合物[J]. 应用化学, 2019, 36(11): 1286-1293. |
| [4] | 刘杰,周维友,吴中,孙富安,何明阳,陈群. 钴镁铝类水滑石液相选择性催化苯甲醇氧化合成苯甲醛[J]. 应用化学, 2015, 32(9): 1033-1039. |
| [5] | 曹明鸣, 周维友, 仲霞, 孙富安, 何明阳. 钴铝类水滑石液相选择性催化甲苯氧化合成苯甲醛[J]. 应用化学, 2014, 31(05): 541-547. |
| [6] | 刘长辉, 文瑞明, 雷雪松, 张哲. 茶螺烷酮合成的新工艺[J]. 应用化学, 2010, 27(10): 1238-1240. |
| [7] | 胡炳成, 吕春绪, 蔡超君. 2-甲酰基-3-乙酰基-4-甲基-5-苄氧羰基吡咯的合成[J]. 应用化学, 2008, 25(2): 233-236. |
| [8] | 高原, 张茂根, 王昉, 王炳祥, 沈健. PMDA-BPDA-HAB聚苯并噁唑的合成及耐热性[J]. 应用化学, 2005, 22(10): 1096-1099. |
| [9] | 岳国仁, 王建华, 张正. 选择性氧化含氰基的对硝基苯硫醚[J]. 应用化学, 1995, 0(4): 93-95. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||