应用化学 ›› 2024, Vol. 41 ›› Issue (6): 878-889.DOI: 10.19894/j.issn.1000-0518.240021
去乙酰化槐糖脂生物表面活性剂的结构鉴定和理化性质
杨昊1,2, 马孝萌1,2, 黄金华1,2, 李俊峰3, 梁生康1,2( )
)
- 1.中国海洋大学化学化工学院,青岛 266100
2.中国海洋大学海洋化学理论与工程技术教育部重点实验室,青岛 266100
3.青岛科技大学海洋科学与生物工程学院,青岛 266045
-
收稿日期:2024-01-22接受日期:2024-05-04出版日期:2024-06-01发布日期:2024-07-09 -
通讯作者:梁生康 -
基金资助:山东省重大科技创新工程项目(2021CXGC010705);崂山实验室“十四五”重大项目(2022QNLM040002)
Structural Identification and Physicochemical Properties of Nonacetylated Sophorolipids Biosurfactant
Hao YANG1,2, Xiao-Meng MA1,2, Jin-Hua HUANG1,2, Jun-Feng LI3, Sheng-Kang LIANG1,2( )
)
- 1.College of Chemistry and Chemistry Engineering,Ocean University of China,Qingdao 266100,China
2.The Key Laboratory of Marine Chemistry Theory and Technology,Ministry of Education,Ocean University of China,Qingdao 266100,China
3.College of Marine Science and Biological Engineering,Qingdao University of Science and Technology,Qingdao 266045,China
-
Received:2024-01-22Accepted:2024-05-04Published:2024-06-01Online:2024-07-09 -
Contact:Sheng-Kang LIANG -
About author:liangsk@ouc.edu.cn
-
Supported by:the Shandong Province Major Science and Technology Innovation Engineering Project(2021CXGC010705);the Laoshan Laboratory's 14th Five Year Plan Major Project(2022QNLM040002)
摘要:
槐糖脂亲水基上的乙酰化程度对其表界面活性和理化性质有重要影响,为了查明不同菌株发酵得到的乙酰化/去乙酰化槐糖脂(SLs)在结构和理化性能上的差异,使用HPLC-MS/MS解析了去乙酰化SLs的结构,并与野生菌株所产乙酰化SLs的表界面活性等理化性质进行比较。 结果表明: 去乙酰化菌株所产的去乙酰化SLs疏水基主要为十八烯酸,亲水基主要为去乙酰化槐糖,其中,内酯型、酸性和具有bola型结构的槐糖脂质量分数分别为26.99%、49.98%和23.03%。 野生菌株所产槐糖脂以乙酰化槐糖脂为主,且内酯型和酸型质量分数分别为97.86%和2.14%; 其疏水基主要为十八烯酸。 去乙酰化SLs在水中的溶解度高达485.8 g/L,较乙酰化SLs提高了14倍,且具有更好的发泡性能和泡沫稳定性,同时,去乙酰化SLs乳化性能较乙酰化SLs提高26.7倍。 去乙酰化SLs的表面活性稍低,临界表面张力为41.0 mN/m,高于乙酰化SLs的36.2 mN/m; 亲水-亲油平衡值为13,高于乙酰化SLs的11。 2种槐糖脂具有良好的抗硬水性,但耐酸碱性均较弱。
中图分类号:
引用本文
杨昊, 马孝萌, 黄金华, 李俊峰, 梁生康. 去乙酰化槐糖脂生物表面活性剂的结构鉴定和理化性质[J]. 应用化学, 2024, 41(6): 878-889.
Hao YANG, Xiao-Meng MA, Jin-Hua HUANG, Jun-Feng LI, Sheng-Kang LIANG. Structural Identification and Physicochemical Properties of Nonacetylated Sophorolipids Biosurfactant[J]. Chinese Journal of Applied Chemistry, 2024, 41(6): 878-889.

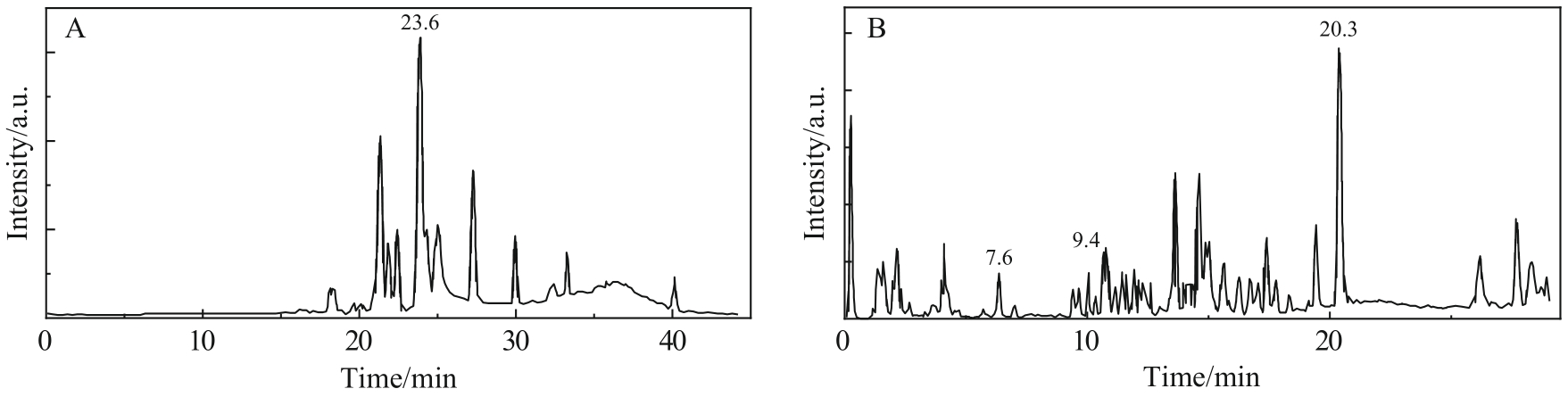
图2 (A)乙酰化SLs保留时间为23.6 min 组分的质谱图. (B)去乙酰化SLs保留时间为20.3 min组分的质谱图. (C)去乙酰化SLs保留时间为9.4 min组分的质谱图. (D)去乙酰化SLs保留时间为7.6 min组分的质谱图
Fig.2 (A) The mass spectrum of components with acetylated SLs retention time of 23.6 min. (B) The mass spectrum of components with nonacetylated SLs retention time of 20.3 min. (C) The mass spectrum of components with nonacetylated SLs retention time of 9.4 min. (D) The mass spectrum of components with nonacetylated SLs retention time of 7.6 min
| Type | Molecular formula | m/z of [M+H]+ | Full name of component | w/% | |||
|---|---|---|---|---|---|---|---|
| Fatty acid groups | L/A/B* | Measured value | Simulation value | ||||
| Nonacetylated | C18∶2 | A | C30H52O13 | 621.342 1 | 621.340 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid | 0.32 |
| Monoacetylated | C16∶0 | L | C30H52O13 | 621.743 5 | 621.340 8 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanoic acid-1′,4"- lactone-6"-monoacetyl ester | 0.11 |
| C18∶0 | L | C32H56O13 | 649.369 7 | 649.372 2 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanoic acid-1′,4"- lactone-6"-monoacetyl ester | 0.18 | |
| C18∶1 | L | C32H54O13 | 647.358 1 | 647.356 5 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1′,4"-lactone-6"- monoacetyl ester | 5.29 | |
| C18∶2 | L | C32H52O13 | 645.341 9 | 645.340 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-1′,4"-lactone-6"-monoacetyl ester | 1.60 | |
| Diacetylated | C16∶0 | L | C32H54O14 | 663.360 4 | 663.351 4 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanoic acid-1′,4"- lactone-6′,6"-diacetyl ester | 12.27 |
| C16∶1 | L | C32H52O14 | 661.338 6 | 661.335 7 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecenoic acid-1′,4"-lactone-6',6"-diacetyl ester | 0.15 | |
| C18∶0 | L | C34H58O14 | 691.384 9 | 691.382 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanoic acid-1′,4"-lactone-6′,6"-diacetyl ester | 16.97 | |
| C18∶1 | L | C34H56O14 | 689.375 4 | 689.367 1 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1′,4"-lactone-6′,6"-diacetyl ester | 42.47 | |
| C18∶2 | L | C34H54O14 | 687.356 3 | 687.351 4 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-1′,4"-lactone-6',6"-diacetyl ester | 18.82 | |
| C16∶2 | A | C32H52O15 | 677.331 9 | 677.330 6 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanodienoic acid-6′,6″-diacetyl ester | 1.29 | |
| C18∶1 | A | C34H58O15 | 707.382 5 | 707.377 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-6′,6″-diacetylester | 0.19 | |
| C18∶2 | A | C34H56O15 | 705.351 6 | 705.362 0 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-6′,6″- diacetyl ester | 0.18 | |
| C22∶1 | A | C38H66O15 | 745.432 9 | 745.429 9 | 21-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]- docosenoic acid -6′,6″- diacetyl ester | 0.16 | |
表1 乙酰化SLs同系物组分和质量分数 (Continued from previous page)
Table 1 Composition and mass fraction of acetylated SLs
| Type | Molecular formula | m/z of [M+H]+ | Full name of component | w/% | |||
|---|---|---|---|---|---|---|---|
| Fatty acid groups | L/A/B* | Measured value | Simulation value | ||||
| Nonacetylated | C18∶2 | A | C30H52O13 | 621.342 1 | 621.340 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid | 0.32 |
| Monoacetylated | C16∶0 | L | C30H52O13 | 621.743 5 | 621.340 8 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanoic acid-1′,4"- lactone-6"-monoacetyl ester | 0.11 |
| C18∶0 | L | C32H56O13 | 649.369 7 | 649.372 2 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanoic acid-1′,4"- lactone-6"-monoacetyl ester | 0.18 | |
| C18∶1 | L | C32H54O13 | 647.358 1 | 647.356 5 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1′,4"-lactone-6"- monoacetyl ester | 5.29 | |
| C18∶2 | L | C32H52O13 | 645.341 9 | 645.340 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-1′,4"-lactone-6"-monoacetyl ester | 1.60 | |
| Diacetylated | C16∶0 | L | C32H54O14 | 663.360 4 | 663.351 4 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanoic acid-1′,4"- lactone-6′,6"-diacetyl ester | 12.27 |
| C16∶1 | L | C32H52O14 | 661.338 6 | 661.335 7 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecenoic acid-1′,4"-lactone-6',6"-diacetyl ester | 0.15 | |
| C18∶0 | L | C34H58O14 | 691.384 9 | 691.382 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanoic acid-1′,4"-lactone-6′,6"-diacetyl ester | 16.97 | |
| C18∶1 | L | C34H56O14 | 689.375 4 | 689.367 1 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1′,4"-lactone-6′,6"-diacetyl ester | 42.47 | |
| C18∶2 | L | C34H54O14 | 687.356 3 | 687.351 4 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-1′,4"-lactone-6',6"-diacetyl ester | 18.82 | |
| C16∶2 | A | C32H52O15 | 677.331 9 | 677.330 6 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanodienoic acid-6′,6″-diacetyl ester | 1.29 | |
| C18∶1 | A | C34H58O15 | 707.382 5 | 707.377 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-6′,6″-diacetylester | 0.19 | |
| C18∶2 | A | C34H56O15 | 705.351 6 | 705.362 0 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-6′,6″- diacetyl ester | 0.18 | |
| C22∶1 | A | C38H66O15 | 745.432 9 | 745.429 9 | 21-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]- docosenoic acid -6′,6″- diacetyl ester | 0.16 | |
| Type | Molecular formula | m/z of [M+H]+ | Full name of component | w/% | |||
|---|---|---|---|---|---|---|---|
| Fatty acid groups | L/A/B* | Measured value | Simulation value | ||||
| Nonacetylated | C16∶2 | L | C28H46O12 | 575.310 4 | 575.298 8 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-hexadecanodienoic acid-1′,4″-lactone | 8.73 |
| C18∶1 | L | C30H52O12 | 605.354 5 | 605.345 9 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-oleic acid-1′,4″-lactone | 16.50 | |
| C18∶3 | L | C30H48O12 | 637.365 2 | 637.335 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]- octadecanotrienoic acid-1′,4″-lactone | 1.76 | |
| C16∶0 | A | C28H52O13 | 597.509 0 | 597.340 8 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-hexadecenoic acid | 16.96 | |
| C16∶0 | A | C28H52O13 | 597.347 9 | 597.340 8 | 16-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-hexadecenoic acid | 1.74 | |
| C18∶0 | A | C30H56O13 | 625.386 3 | 625.372 2 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-octadecanoic acid | 6.37 | |
| C18∶1 | A | C30H54O13 | 623.364 2 | 623.356 5 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-oleic acid | 10.18 | |
| C18∶2 | A | C30H52O13 | 621.356 7 | 621.340 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-octadecanodienoic acid | 1.38 | |
| C20∶1 | A | C32H56O12 | 651.395 8 | 651.387 9 | 19-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-eicosenoic acid | 3.59 | |
| C24∶0 | A | C36H74O12 | 709.496 9 | 709.466 4 | 23-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-lignoceric acid | 1.49 | |
| C16∶0 | B | C40H72O23 | 921.450 2 | 921.446 4 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanoic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 6.60 | |
| C16∶1 | B | C40H70O23 | 901.435 5 | 919.430 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]- hexadecenoic acid -1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 10.16 | |
| C18∶1 | B | C42H74O23 | 947.469 3 | 947.462 1 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 2.74 | |
| C18∶2 | B | C42H72O23 | 945.485 5 | 945.446 4 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 2.36 | |
| Monoacetylated | C18∶1 | A | C32H56O14 | 669.365 0 | 665.367 1 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-6″-monoacetyl ester | 8.27 |
| C18∶1 | B | C44H76O23 | 989.478 5 | 989.472 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester-6″-monoacetyl ester | 0.36 | |
| C26∶0 | B | C52H94O23 | 1103.664 2 | 1103.614 0 | 25-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexacosanoic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester-6″-monoacetyl ester | 0.81 | |
表2 去乙酰化SLs同系物组分和质量分数 (Continued from previous page)
Table 2 Composition and mass fraction of nonacetylated SLs
| Type | Molecular formula | m/z of [M+H]+ | Full name of component | w/% | |||
|---|---|---|---|---|---|---|---|
| Fatty acid groups | L/A/B* | Measured value | Simulation value | ||||
| Nonacetylated | C16∶2 | L | C28H46O12 | 575.310 4 | 575.298 8 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-hexadecanodienoic acid-1′,4″-lactone | 8.73 |
| C18∶1 | L | C30H52O12 | 605.354 5 | 605.345 9 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-oleic acid-1′,4″-lactone | 16.50 | |
| C18∶3 | L | C30H48O12 | 637.365 2 | 637.335 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]- octadecanotrienoic acid-1′,4″-lactone | 1.76 | |
| C16∶0 | A | C28H52O13 | 597.509 0 | 597.340 8 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-hexadecenoic acid | 16.96 | |
| C16∶0 | A | C28H52O13 | 597.347 9 | 597.340 8 | 16-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-hexadecenoic acid | 1.74 | |
| C18∶0 | A | C30H56O13 | 625.386 3 | 625.372 2 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-octadecanoic acid | 6.37 | |
| C18∶1 | A | C30H54O13 | 623.364 2 | 623.356 5 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-oleic acid | 10.18 | |
| C18∶2 | A | C30H52O13 | 621.356 7 | 621.340 8 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-octadecanodienoic acid | 1.38 | |
| C20∶1 | A | C32H56O12 | 651.395 8 | 651.387 9 | 19-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-eicosenoic acid | 3.59 | |
| C24∶0 | A | C36H74O12 | 709.496 9 | 709.466 4 | 23-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)- O-]-lignoceric acid | 1.49 | |
| C16∶0 | B | C40H72O23 | 921.450 2 | 921.446 4 | 15-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexadecanoic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 6.60 | |
| C16∶1 | B | C40H70O23 | 901.435 5 | 919.430 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]- hexadecenoic acid -1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 10.16 | |
| C18∶1 | B | C42H74O23 | 947.469 3 | 947.462 1 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 2.74 | |
| C18∶2 | B | C42H72O23 | 945.485 5 | 945.446 4 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-octadecanodienoic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester | 2.36 | |
| Monoacetylated | C18∶1 | A | C32H56O14 | 669.365 0 | 665.367 1 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-6″-monoacetyl ester | 8.27 |
| C18∶1 | B | C44H76O23 | 989.478 5 | 989.472 7 | 17-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-oleic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester-6″-monoacetyl ester | 0.36 | |
| C26∶0 | B | C52H94O23 | 1103.664 2 | 1103.614 0 | 25-L-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-hexacosanoic acid-1-[(2′-O-β-D-glucopyranosyl-β-D-glucopyranosyl)-O-]-ester-6″-monoacetyl ester | 0.81 | |
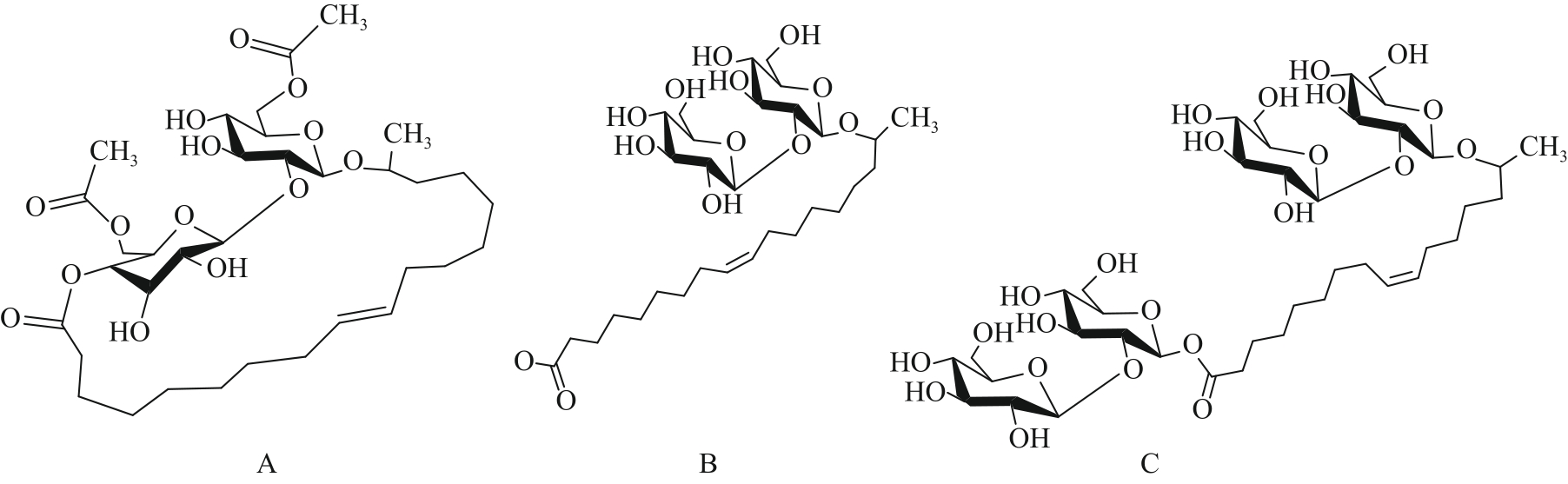
图3 3种有代表性的槐糖脂结构式: (A)双乙酰化内脂型槐糖脂; (B)去乙酰化酸型槐糖脂; (C)去乙酰化bola型槐糖脂
Fig.3 Three representative structural formulas of sophorolipids: (A) Diacetylated lactone type SLs; (B) Nonacetylated acid type SLs; (C) Nonacetylated bola type SLs
| Acetylated SLs | Nonacetylated SLs | |
|---|---|---|
| Solubility/(g·L-1) | 33.8 | 485.8 |
| Cloud point/℃ | 36 | Not detected |
| Acid resistance | Conditional acid resistance | Non acid resistance |
| Alkali resistance | Non alkali resistance | Non alkali resistance |
| Hard water resistance/grade | 5 | 5 |
表3 乙酰化SLs和去乙酰化SLs的溶解性能和抗酸抗碱性
Table 3 Solubility and acid and alkali resistance of acetylated SLs and nonacetylated SLs
| Acetylated SLs | Nonacetylated SLs | |
|---|---|---|
| Solubility/(g·L-1) | 33.8 | 485.8 |
| Cloud point/℃ | 36 | Not detected |
| Acid resistance | Conditional acid resistance | Non acid resistance |
| Alkali resistance | Non alkali resistance | Non alkali resistance |
| Hard water resistance/grade | 5 | 5 |
| Sophorolipid | CMC/(mg·L-1) | γCMC/(mN·m-1) | Гmax/(μmol·m-2) | Amin/nm2 |
|---|---|---|---|---|
| Acetylated-SLs | 30.3 | 36.2 | 2.55 | 0.653 |
| Nonacetylated SLs | 152.5 | 41.0 | 3.01 | 0.551 |
表4 乙酰化SLs和去乙酰化SLs在气液界面上的吸附参数
Table 4 The adsorption parameters of acetylated SLs and nonacetylated SLs
| Sophorolipid | CMC/(mg·L-1) | γCMC/(mN·m-1) | Гmax/(μmol·m-2) | Amin/nm2 |
|---|---|---|---|---|
| Acetylated-SLs | 30.3 | 36.2 | 2.55 | 0.653 |
| Nonacetylated SLs | 152.5 | 41.0 | 3.01 | 0.551 |

图6 乙酰化SLs和去乙酰化SLs的泡沫性能 (A)在纯水中的30 s和5 min泡沫量,(B)不同盐度下的泡沫性能
Fig.6 The foam performance of acetylated SLs and nonacetylated SLs (A) Foam volume of 30 s and 5 min in pure water, (B) foam properties at different salinities
| 1 | PAL S, CHATTERJEE N, DAS A K, et al. Sophorolipids: a comprehensive review on properties and applications[J]. Adv Colloid Interface Sci, 2023, 313: 102856. |
| 2 | MADANKAR C S, BORDE P K. Review on sophorolipids-a promising microbial bio-surfactant[J]. Tenside Surfact Det, 2023, 60(2): 95-105. |
| 3 | CHO W Y, NG J F, YAP W H, et al. Bio-based antimicrobial formulating agents for applications in food and health[J]. Molecules, 2022, 27(17): 5556. |
| 4 | DEVELTER D W G, LAURYSSEN L M L. Properties and industrial applications of sophorolipids[J]. Eur J Lipid Sci Technol, 2010, 112(6): 628-638. |
| 5 | ASMER H J, LANG S, WAGNER F, et al. Microbial production, structure elucidation and bioconversion of sophorose lipids[J]. J Am Oil Chem Soc, 1988, 65(9): 1460-1466. |
| 6 | DENGLE PULATE V, BHAGWAT S, PRABHUNE A. Microbial oxidation of medium chain fatty alcohol in the synthesis of sophorolipids by candida bombicola and its physicochemical characterization[J]. J Surfactants Deterg, 2013, 16(2): 173-181. |
| 7 | KONISHI M, FUKUOKA T, MORITA T, et al. Production of new types of sophorolipids by by candida batistae[J]. J Oleo Sci, 2008, 57(6): 359-369. |
| 8 | VAN BOGAERT, SAERENS K, DE MUYNCK C, et al. Microbial production and application of sophorolipids[J]. Appl Microbiol Biot, 2007, 76(1): 23-34. |
| 9 | SEN A R, KARIM S A, SANTRA A. Effect of defaunation on carcass and meat characteristics of finisher lambs[J]. Indian J Anim Sci, 2000, 70(6): 659-661. |
| 10 | OTTO R T, DANIEL H J, PEKIN G, et al. Production of sophorolipids from whey[J]. Appl Microbiol Biot, 1999, 52(4): 495-501. |
| 11 | LI S, QIAN X, XU L, et al. Biological tailoring of novel sophorolipid molecules and their derivatives[J]. Biofuel Bioprod Bior, 2021, 15(6): 1938-1949. |
| 12 | SHAO L, SONG X, MA X,et al. Bioactivities of sophorolipid with different structures against human esophageal cancer cells[J]. J Surg Res, 2012, 173(2): 286-291. |
| 13 | RAU U, HECKMANN R, WRAY V, et al. Enzymatic conversion of a sophorolipid into a glucose lipid[J]. Biotechnol Lett, 1999, 21(11): 973-977. |
| 14 | BORSANYIOVA M, PATIL A, MUKHERJI R, et al. Biological activity of sophorolipids and their possible use as antiviral agents[J]. Folia Microbiol, 2016, 61(1): 85-89. |
| 15 | KASTURE M, SINGH S, PATEL P, et al. Multiutility sophorolipids as nanoparticle capping agents: synthesis of stable and water dispersible Co nanoparticles[J]. Langmuir, 2007, 23(23): 11409-11412. |
| 16 | LYDON H L, BACCILE N, CALLAGHAN B, et al. Adjuvant antibiotic activity of acidic sophorolipids with potential for facilitating wound healing[J]. Antimicrob Agents Chemother, 2017, 61(5): E02547. |
| 17 | SAERENS K M J, SAEY L, SOETAERT W. One-step production of unacetylated sophorolipids by an acetyltransferase negative Candida bombicola[J]. Biotechnol Bioeng, 2011, 108(12): 2923-2931. |
| 18 | VAN B, BUYST D, MARTINS J C, et al. Synthesis of bolaform biosurfactants by an engineered starmerella bombicola yeast[J]. Biotechnol Bioeng, 2016, 113(12): 2644-2651. |
| 19 | 马孝萌. 去乙酰化槐糖脂基因工程菌株的构建及发酵条件优化[D]. 青岛: 中国海洋大学, 2023. |
| MA X M. Construction of genetically engineered strains producing deacetylated sophorolipids and its optimization of fermentation conditions[D]. Qingdao: Ocean University of China, 2023. | |
| 20 | 宋丹丹, 梁生康, 王江涛. 槐糖脂生物表面活性剂的结构特征及理化性质初探[J]. 环境化学, 2011, 30(8): 1474-1479. |
| SONG D D, LIANG S K, WANG J T. Structural characteristics and physical-chemical properties of sophorolipid biosurfactants[J]. Environ Chem, 2011, 30(8): 1474-1479. | |
| 21 | 孙旭璐, 陈慧, 郑伟. 改性大豆磷脂的HLB值检测方法优化研究[J]. 粮食与油脂, 2015, 28(4): 47-48. |
| SUN X L, CHEN H, ZHENG W. Study on the optimal method for detecting of modified soybean phospholipids HLB value[J]. Cereals Oils, 2015, 28(4): 47-48. | |
| 22 | 周家华, 崔英德. 表面活性剂HLB值的分析测定与计算Ⅰ∶ HLB值的分析测定[J]. 精细石油化工, 2001(2): 11-14. |
| ZHOU J H, CUI Y D. Measurement and calculation of HLB value of surfactants: the measurement of HLB value[J]. Spec Petrochem, 2001(2): 11-14. | |
| 23 | DAVID J T, ROBERT L R. Commercial synthesis of monoalkly phosphates[J]. J Surfactants Deterg, 2002, 5(2): 169-172. |
| 24 | 李双阳, 王佳颖, 郭高强, 等. BOLA型表面活性剂的研究进展[J]. 广州化工, 2015, 43(5): 18-20. |
| LI S Y, WANG J Y, GUO G Q, et al. Progress in research work with respect to bolaform surfactants[J]. Guangzhou Chem Ind, 2015, 43(5): 18-20. | |
| 25 | 周家华, 崔英德, 吴雅红. 表面活性剂HLB值的分析测定与计算 Ⅱ.HLB值的计算[J]. 精细石油化工, 2001(4): 38-41. |
| ZHOU J H, CUI Y D, WU Y H. Measurement and calculation of HLB value of surfactants: the calculation of HLB value[J]. Spec Petrochem, 2001(4): 38-41. | |
| 26 | KRAYNIK A M. The structure of random foam[J]. Adv Eng Mater, 2006, 8(9): 900-906. |
| 27 | KUMAR V, PAL N, JANGIR A K, et al. Dynamic interfacial properties and tuning aqueous foamability stabilized by cationic surfactants in terms of their structural hydrophobicity, free drainage and bubble extent[J]. Colloid Surface A, 2020, 588: 124362. |
| 28 | WANG C, FANG H, GONG Q,et al. Roles of catanionic surfactant mixtures on the stability of foams in the presence of oil[J]. Energ Fuel, 2016, 30(8): 6355-6364. |
| 29 | 赵修太, 王泉, 王增宝, 等. 高温高盐油藏常用起泡剂研究进展[J]. 材料导报, 2016, 30(5): 75-80. |
| ZHAO X T, WANG Q, WANG Z B, et al. Commonly used foaming agents in high temperature and high salinity reservoir[J]. Mater Rep, 2016, 30(5): 75-80. | |
| 30 | SUN L, SUN X H, ZHANG Y C, et al. Stability of high-salinity-enhanced foam: surface behavior and thin-film drainage[J]. Petrol Sci, 2023, 20(4): 2343-2353. |
| 31 | 刘晓敏. 驱油用泡沫体系研究[D]. 无锡: 江南大学, 2023. |
| LIU X M. Study on foam systems for enhanced oil recovery[D]. Wuxi: Jiangnan University, 2023. | |
| 32 | 胡贝贝, 袁悦, 周小平, 等. 基于丝氨酸的Bola型表面活性剂的合成及理化性质[J]. 沈阳药科大学学报, 2016, 33(6): 419-425. |
| HU B B, YUAN Y, ZHOU X P, et al. Synthesis of a novel Bola amphiphile based on serine and investigation of its physicochemical properties[J]. J Shenyang Pharm Univ, 2016, 33(6): 419-425. | |
| 33 | VAN RENTERGHEM L, ROELANTS S, BACCILE N, et al. From lab to market: an integrated bioprocess design approach for new-to-nature biosurfactants produced by Starmerella bombicola[J]. Biotechnol Bioeng, 2018, 115(5): 1195-1206. |
| [1] | 焦陈斯帆, 郑少波, 许鹏军, 王炜, Takebe Hiromichi, Mukai Kusuhiro, 余仲达. 评价表面活性剂溶液泡沫性能的方法—真球气泡法[J]. 应用化学, 2022, 39(7): 1108-1118. |
| [2] | 王春雨, 赵辉, 代正华, 李伟锋, 刘海峰. 表面活性剂对亲水性颗粒悬浮液流变性的影响[J]. 应用化学, 2021, 38(4): 398-406. |
| [3] | 刘佳佳, 谢益诚, 许虎君. 一种阳离子Gemini型沥青乳化剂的合成及性能[J]. 应用化学, 2018, 35(5): 552-558. |
| [4] | 丁伟, 宋成龙, 李博洋. 壬基酚甜菜碱两性表面活性剂的合成和抗温耐盐性能[J]. 应用化学, 2015, 32(8): 922-930. |
| [5] | 李燕, 刘晓艳, 张顺, 刘亭, 李勇军, 兰燕宇, 王爱民, 王永林, 何彬. 一种香豆素荧光标记多肽的合成及应用[J]. 应用化学, 2014, 31(12): 1413-1418. |
| [6] | 温福山, 孔祥双, 王维, 胡松青, 夏政, 张军. 一种新型阳离子三硅氧烷表面活性剂的合成及其表面性能[J]. 应用化学, 2014, 31(10): 1216-1221. |
| [7] | 丁伟, 李金红, 于涛, 邢欣欣, 张微, 杨柳. 新型磺基甜菜碱/聚丙烯酰胺体系表面及界面性能[J]. 应用化学, 2013, 30(11): 1270-1275. |
| [8] | 王平, 郭祥峰, 贾丽华, 马淳淼, 钱琳. 溴化1-十二烷基-3-甲基咪唑与甲基橙的相互作用[J]. 应用化学, 2013, 30(07): 840-845. |
| [9] | 于涛, 史雷城, 丁伟, 曲广淼, 栾和鑫, 刘小军. 芳基油酸酰胺羟丙基磺基甜菜碱的合成及性能[J]. 应用化学, 2012, 29(11): 1302-1308. |
| [10] | 于涛, 袁丹丹, 丁伟, 李淑杰, 刘坤, 曲广淼, 王会敏, 罗石琼, 陈玉萍. 新型孪尾Gemini两性离子表面活性剂应用性能[J]. 应用化学, 2012, 29(10): 1182-1188. |
| [11] | 赵琴琴, 史鸿鑫, 武宏科, 陈立军, 项菊萍. p-全氟壬烯氧基苯磺酸聚乙二醇酯表面活性剂的合成及表面活性[J]. 应用化学, 2012, 29(08): 906-909. |
| [12] | 贾金英, 闫杰, 安悦, 崔冬梅, 杨陈希, 陈晓峰, 王拓, 佟妍. 琥珀酸酯磺酸盐Gemini表面活性剂的合成及表面活性[J]. 应用化学, 2011, 28(10): 1184-1188. |
| [13] | 于涛, 刘宏彬, 丁伟, 史鹏, 魏继军, 毛程, 任娅妮, 曲广淼. 烷基芳基磺酸盐相对分子质量及其分布与表面性能的关系[J]. 应用化学, 2011, 28(07): 831-835. |
| [14] | 曲广淼, 魏继军, 于涛, 丁伟, 孙颖, 史鹏, 刘宏彬, 董志龙. 系列磺丁基甜菜碱的表征及表面活性[J]. 应用化学, 2011, 28(06): 716-721. |
| [15] | 张建, 邱宇, 于道永. 同步荧光光谱法测定十二烷基苯磺酸钠的临界胶束浓度[J]. 应用化学, 2009, 26(12): 1480-1483. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||