| 1 |
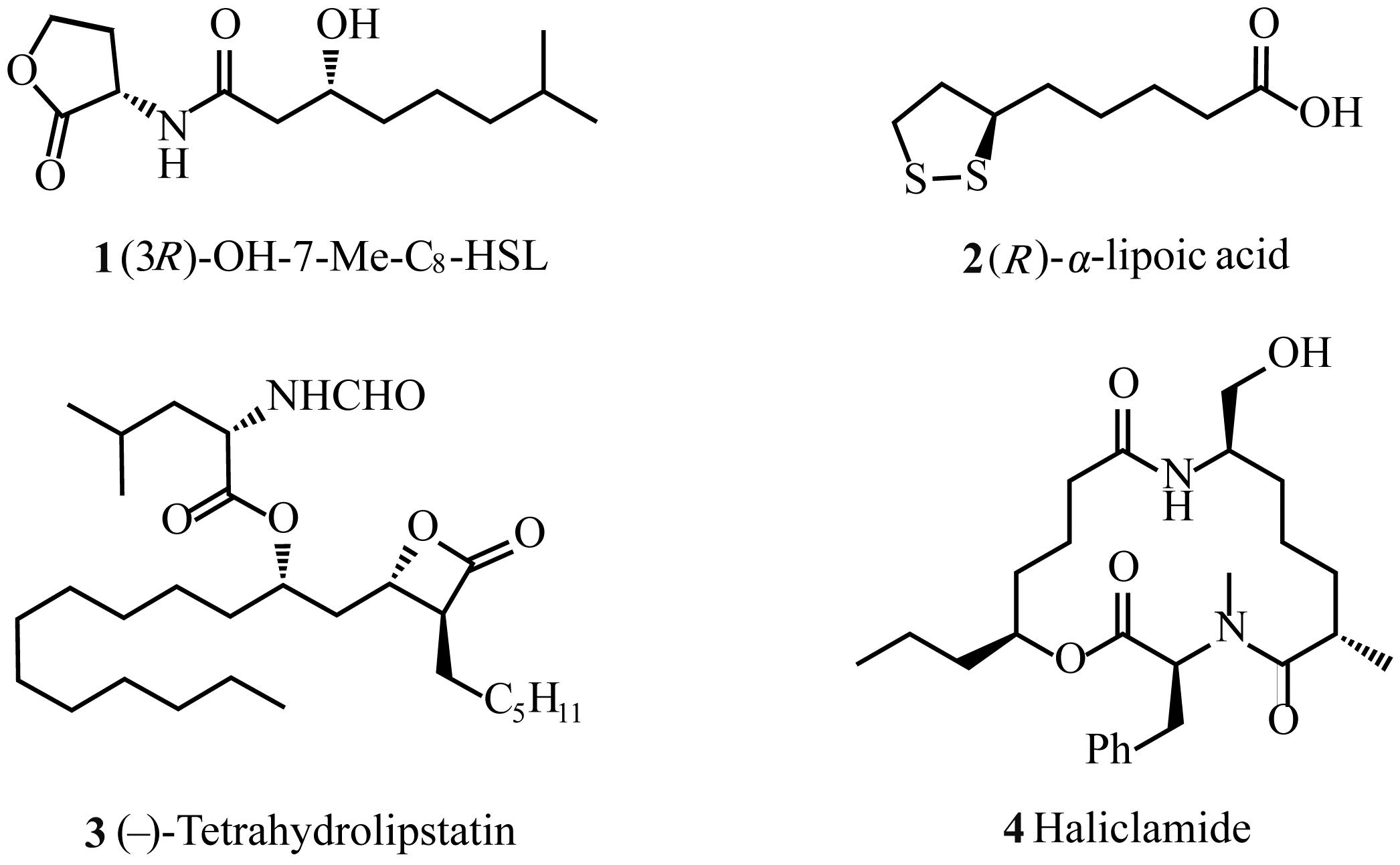
YANG X H, WANG K, ZHU S F, et al. Remote ester group leads to efficient kinetic resolution of racemic aliphatic alcohols via asymmetric hydrogenation[J]. J Am Chem Soc, 2014, 136(50): 17426-17429.
|
| 2 |
YAJIMA A. Recent progress in the chemistry and chemical biology of microbial signaling molecules: quorum-sensing pheromones and microbial hormones[J]. Tetrahedron Lett, 2014, 55(17): 2773-2780.
|
| 3 |
HAN X, KANG C Q. Advances in synthetic methods of (R)-α-lipoic acid[J]. Chinese J Appl Chem, 2020, 37(11): 1236-1248.
|
|
韩修, 康传清. (R)-α-硫辛酸合成方法的研究进展[J]. 应用化学, 2020, 37(11): 1236-1248.
|
| 4 |
REED L J, KOIKE M, LEVITCH M E, et al. Studies on the nature and reactions of protein bound lipoic acid[J]. Biol Chem, 1958, 232(1): 143-158.
|
| 5 |
BAST A, HANEN G. Interplay between lipoic acid and glutathione in the protection against microsomal lipid-peroxidation[J]. Biochem Biophys Acta, 1988, 963(3): 558-561.
|
| 6 |
REED L J, NIU C I. Syntheses of DL-α-lipoic acid[J]. J Am Chem Soc, 1955, 77(2): 416-419.
|
| 7 |
K.GHOSH A, SHURRUSH K, KULKARNI S. Asymmetric synthesis of anti-aldol segments via a nonaldol route: synthetic applications to statines and (–)- tetrahydrolipstatin[J]. J Org Chem, 2009, 74(12): 4508-4518.
|
| 8 |
MULZER M, TIEGS B, WANG Y P, et al. Total synthesis of tetrahydrolipstatin and stereoisomers via a highly regio- and diastereoselective carbonylation of epoxyhomoallylic alcohols[J]. J Am Chem Soc, 2014, 136(30): 10814-10820.
|
| 9 |
PFEIFFER B, GISLER S S, BARANDUN L, et al. Total synthesis and configurational assignment of the marine natural product haliclamide[J]. J Org Chem, 2013, 78(6): 2553-2563.
|
| 10 |
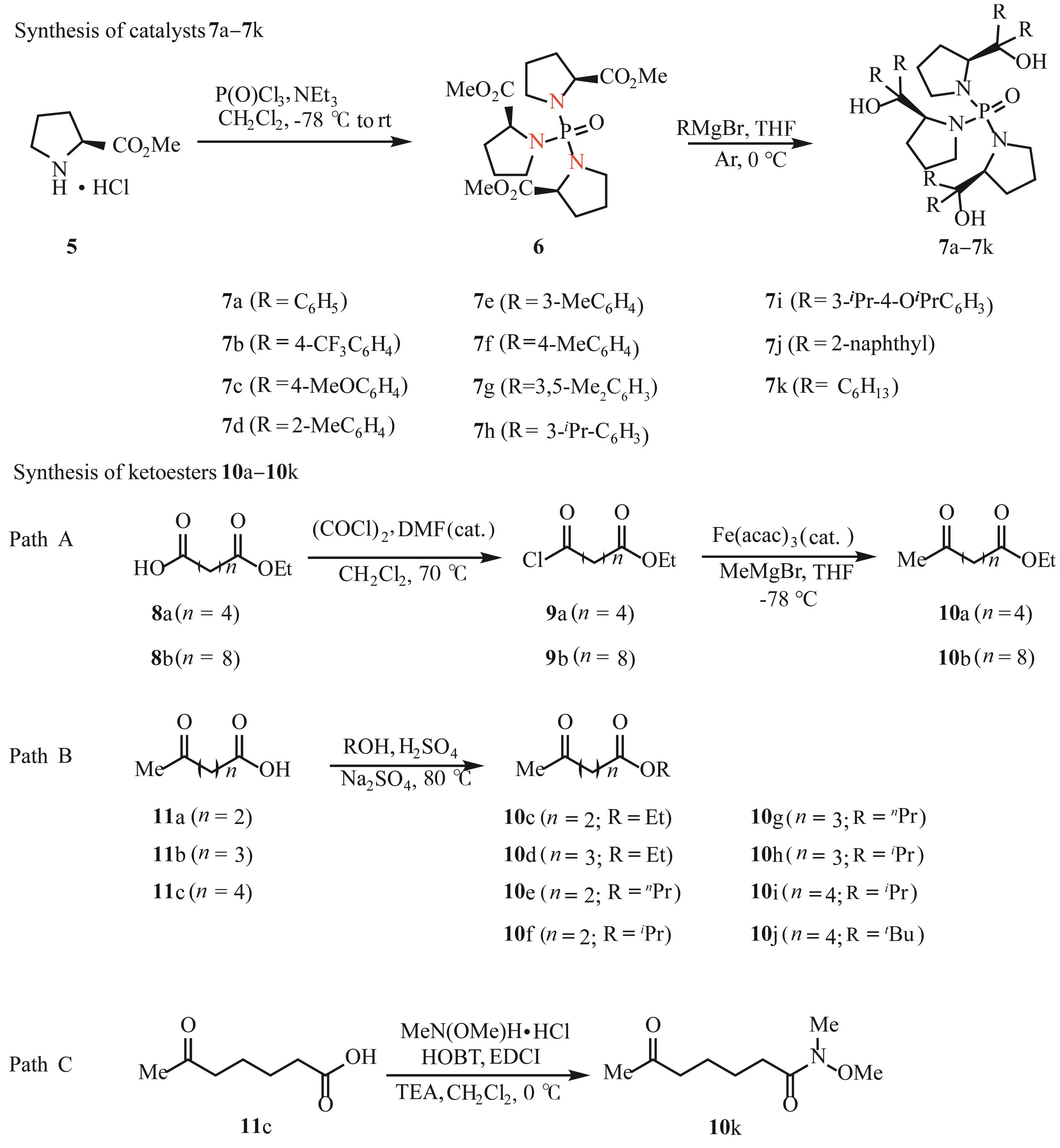
ZHANG Y J, ZHANG W X, ZHENG G W, et al. Identification of an ε-keto ester reductase for the efficient synthesis of an (R)-α-lipoic acid precursor[J]. Adv Synth Catal, 2015, 357(8): 1697-1702.
|
| 11 |
CHEN R J, ZHENG G W, NI Y, et al. Efficient synthesis of an ε-hydroxy ester in a space-time yield of 1580 g L-1 d-1 by a newly identified reductase RhCR[J]. Tetrahedron: Asymmetry, 2014, 25(23): 1501-1504.
|
| 12 |
NOYORI R, OHKUMA T, KITAMURA M, et al. A novel enantiospecific synthesis of (S)-(-)-methyl 6,8-dihydroxyoctanoate, a precursor of (R)-(+)-α-lipoic acid[J]. J Chem Soc Chem Commun, 1990, (10): 729-730.
|
| 13 |
BENINCORI T, PICCOLO O, RIZZO S, et al. 3,3'-Bis(diphenylphosphino)-1,1'-disubstituted-2,2'-biindoles: easily accessible, electron-rich, chiral diphosphine ligands for homogeneous enantioselective hydrogenation of oxoesters[J]. J Org Chem, 2000, 65(24): 8340-8347.
|
| 14 |
SUN X F, ZHOU L, LI W, et al. Convenient divergent strategy for the synthesis of TunePhos-type chiral diphosphine ligands and their applications in highly enantioselective Ru-catalyzed hydrogenations[J]. J Org Chem, 2008, 73(3): 1143-1146.
|
| 15 |
XIE J H, LIU X Y, YANG X Y, et al. Chiral iridium catalysts bearing spiro pyridine-aminophosphine ligands enable highly efficient asymmetric hydrogenation of β-aryl-β-ketoesters[J]. Angew Chem Int Ed, 2012, 51(1): 201-203.
|
| 16 |
AKIO F, SHOHEI H, NOBUYUKI U, et al. Ruthenium(II)-catalyzed asymmetric transfer hydrogenation of ketones using a formic acid-triethylamine mixture[J]. J Am Chem Soc, 1996, 118(10): 2521-2522.
|
| 17 |
YANG J W, LIST B. Catalytic asymmetric transfer hydrogenation of α-ketoesters with Hantzsch esters[J]. Org Lett, 2006, 8(24): 5653-5655.
|
| 18 |
ZHOU W J, NI Y, ZHENG G W, et al. Enzymatic resolution of a chiral chlorohydrin precursor for (R)-α-lipoic acid synthesis via lipase catalyzed enantioselective transacylation with vinyl acetate[J]. J Mol Catal B: Enzym, 2004, 99: 102-107.
|
| 19 |
COREY E J, HELAL C J. Reduction of carbonyl compounds with chiral oxazaborolidine catalysts: a new paradigm for enantioselective catalysis and a powerful new synthetic method[J]. Angew Chem Int Ed, 1998, 37(15): 1986-2012.
|
| 20 |
TOSHIHARU T, MIYUKI M, TADASHI N, et al. Total synthesis of (–)-centrolobine[J]. Tetrahedron Lett, 2008, 49(45): 6462-6465.
|
| 21 |
DU Z J, GUAN J, WU G J, et al. Pd(II)-catalyzed enantioselective synthesis of P-stereogenic phosphinamides via desymmetric C—H arylation[J]. J Am Chem Soc, 2015, 137(2): 632-635.
|
| 22 |
CHEN Y H, QIN X L, HAN F S. Efficient synthesis of cyclic P-stereogenic phosphinamides from acyclic chiral precursors via radical oxidative intramolecular aryl C—H phosphinamidation[J]. Chem Commun, 2017, 53(43): 5826-5829.
|
| 23 |
CHEN Y H, QIN X L, GUAN J, et al. Pd-catalyzed enantioselective C—H arylation of phosphinamides with boronic acids for the synthesis of P-stereogenic compounds[J]. Tetrahedron: Asymmetry, 2017, 28(4): 522-531.
|
| 24 |
QIN X L, LI A, HAN F S. Desymmetric enantioselective reduction of cyclic 1,3-diketones catalyzed by a recyclable P-chiral phosphinamide organocatalyst[J]. J Am Chem Soc, 2021, 143(7): 2994-3002.
|
| 25 |
DU D M, FANG T, XU J X, et al. Structurally well-defined, recoverable C3-symmetric tris(β-hydroxyphosphoramide)-catalyzed enantioselective borane reduction of ketones[J]. Org Lett, 2006, 8(7): 1327-1330.
|
| 26 |
SCHEIPER B, BONNEKESSEL M, KRAUSE H, et al. Selective iron-catalyzed cross-coupling reactions of Grignard reagents with enol triflates, acid chlorides, and dichloroarenes[J]. J Org Chem, 2004, 69(11): 3943-3949.
|
| 27 |
HUSSEIN M A, HUYNH T V, HOMMELSHEIM R, et al. An efficient method for retro-Claisen-type C—C bond cleavage of diketones with tropylium catalyst[J]. Chem Commun, 2018, 54(92): 12970-12973.
|
| 28 |
LATLI B, WOOD E, CASIDA E J. Insecticidal quinazoline derivatives with (trifluoromethyl)-diazirinyl and azido substituents as NADH: ubiquinone oxidoreductase inhibitors and candidate photoaffinity probes[J]. Chem Res Toxicol, 1996, 9(7): 445-450.
|
| 29 |
CHEN J Z, ZHAO G Y, CHEN L M. Efficient production of 5-hydroxymethylfurfural and alkyl levulinate from biomass carbohydrate using ionic liquid-based polyoxometalate salts[J]. RSC Adv, 2014, 4(8): 4194-4202.
|
| 30 |
KUO C H, POYRAZ A S, JIN L, et al. Heterogeneous acidic TiO2 nanoparticles for efficient conversion of biomass derived carbohydrates[J]. Green Chem, 2014, 16(2): 785-791.
|
| 31 |
HE L Y, MIYUKI K, HORIUCHI C A. Oxidation of 2-alkylcycloalkanones with iodine-cerium(IV) salts in alcohols[J]. J Chem Res, Synop, 1999, (2): 122-123.
|
| 32 |
HE L Y, HORIUCHI C A. Oxidation of 2-substituted cycloalkanones with cerium(IV) sulfate tetrahydrate in alcohols and acetic acid[J]. Bull Chem Soc Jpn, 1999, 72(11): 2515-2521.
|
| 33 |
MASAHARU I, ATSUO N, SUSUMU K. Highly stereoselective construction of spiro[4.5]decanes by SmI2-promoted ketyl radical mediated tandem cyclization[J]. Org Lett, 2007, 9(3): 469-472.
|
| 34 |
YANG Y S, GUO J Y, NG H M, et al. Formal hydration of non-activated terminal olefins using tandem catalysts[J]. Chem Commun, 2014, 50(20): 2608-2611.
|
| 35 |
NICOLAOU K C, PASTOR J, BARLUENGA S, et al. Polymer-supported selenium reagents for organic synthesis[J]. Chem Commun, 1998, (18): 1947-1948.
|
| 36 |
BAPTISTE A, DAMIEN J, GIANLUCA P, et al. Dual benzophenone/copper-photocatalyzed Giese-type alkylation of C(sp 3)—H bonds[J]. Chem Eur J, 2019, 25(70): 16120-16127.
|
| 37 |
TSUTOMU O, YUYA T. Facile synthesis of optically-active γ-valerolactone from levulinic acid and its esters using a heterogeneous enantio-selective catalyst[J]. Catal Lett, 2018, 148(3): 824-830.
|
| 38 |
ANTOINE B V, WANG D, VINCENT D, et al. Transfer hydrogenation of carbonyl derivatives catalyzed by an inexpensive phosphine-free manganese precatalyst[J]. Org Lett, 2017, 19(13): 3656-3659.
|
| 39 |
DO H Q, BACHMAN S, BISSEMBER A C, et al. Photoinduced, copper-catalyzed alkylation of amides with unactivated secondary alkyl halides at room temperature[J]. J Am Chem Soc, 2014, 136(5): 2162-2167.
|
| 40 |
GAMBLE M P, SMITH A R C, WILLS M. A novel phosphinamide catalyst for the asymmetric reduction of ketones by borane[J]. J Org Chem, 1998, 63(17): 6068-6071.
|
| 41 |
WANG L L, CAO S S, DU Z J, et al. 4-Dimethylaminopyridine-catalyzed dynamic kinetic resolution in asymmetric synthesis of P-chirogenic 1,3,2-oxazaphospholidine-2-oxides[J]. RSC Adv, 2016, 6(92): 89665-89670.
|

 ), Fu-She HAN1,2(
), Fu-She HAN1,2( )
)