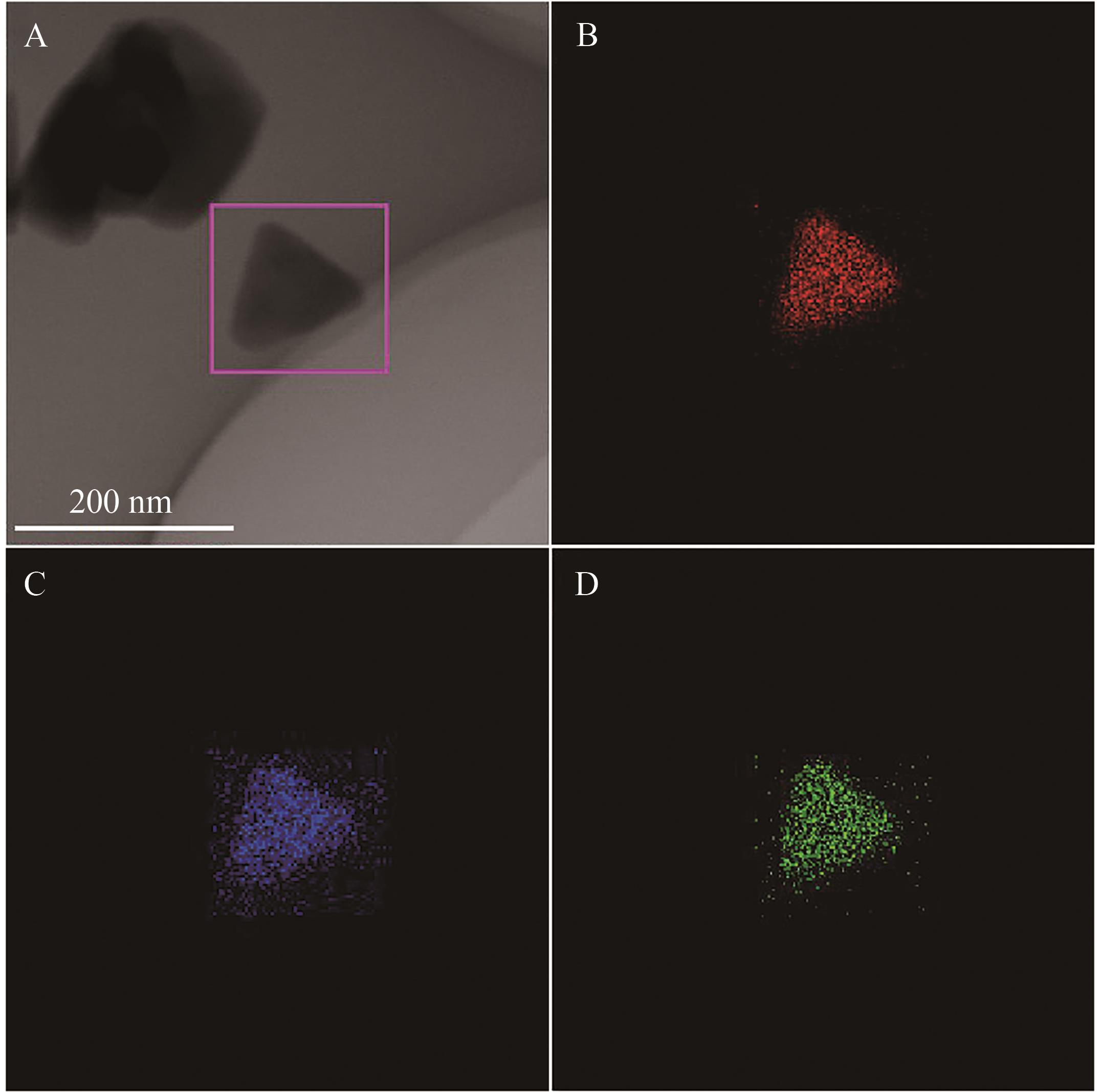
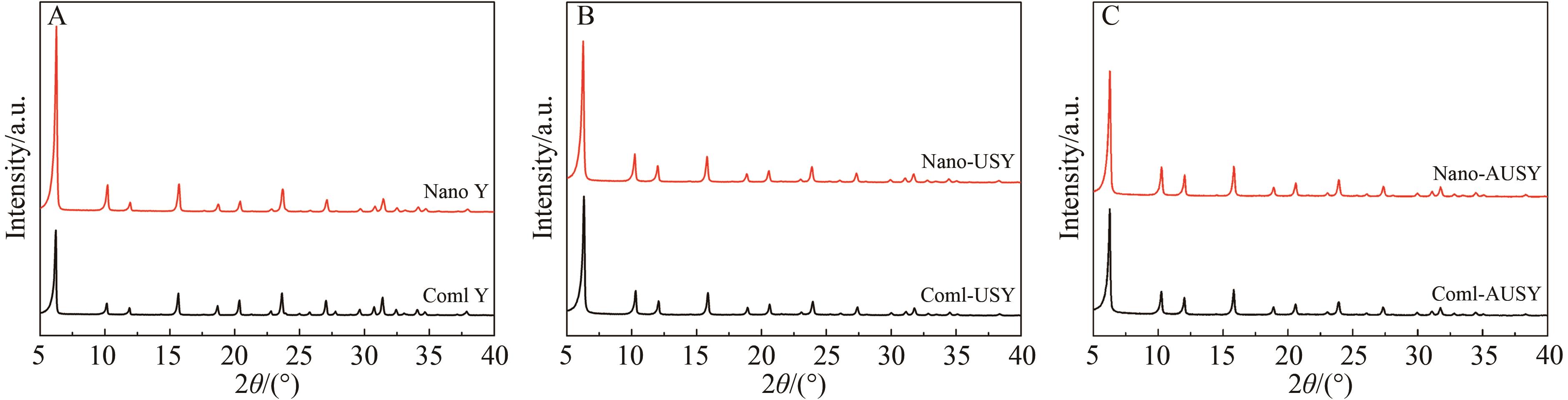
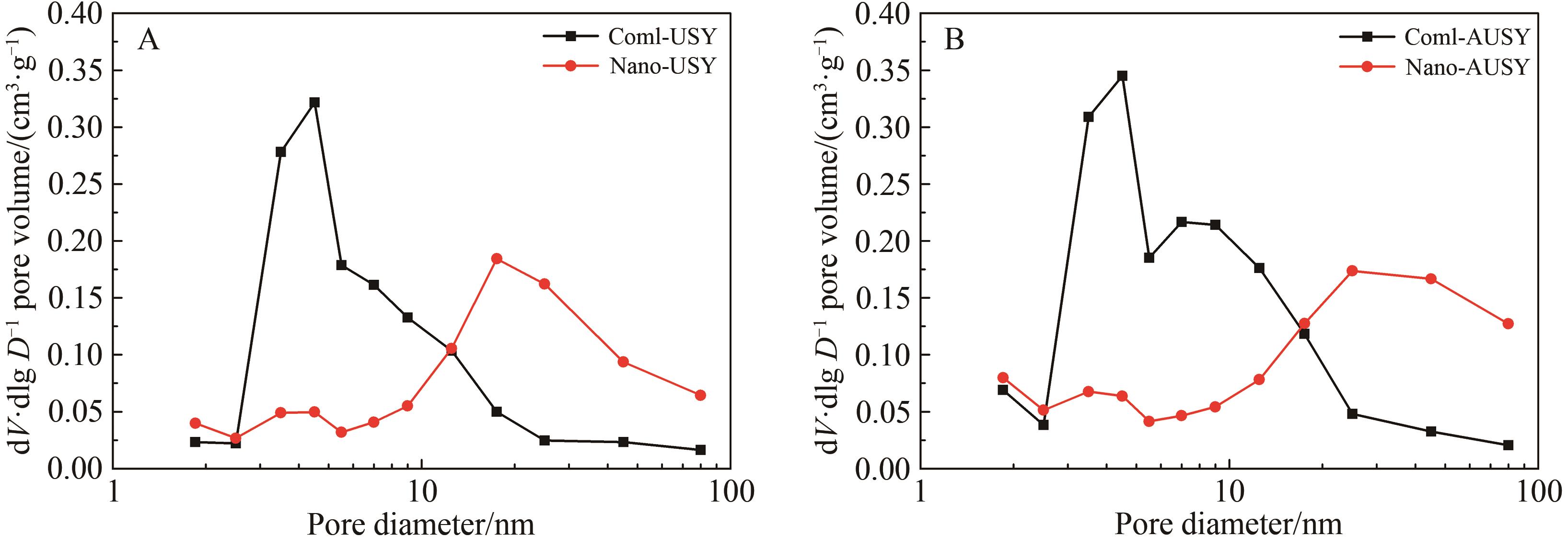
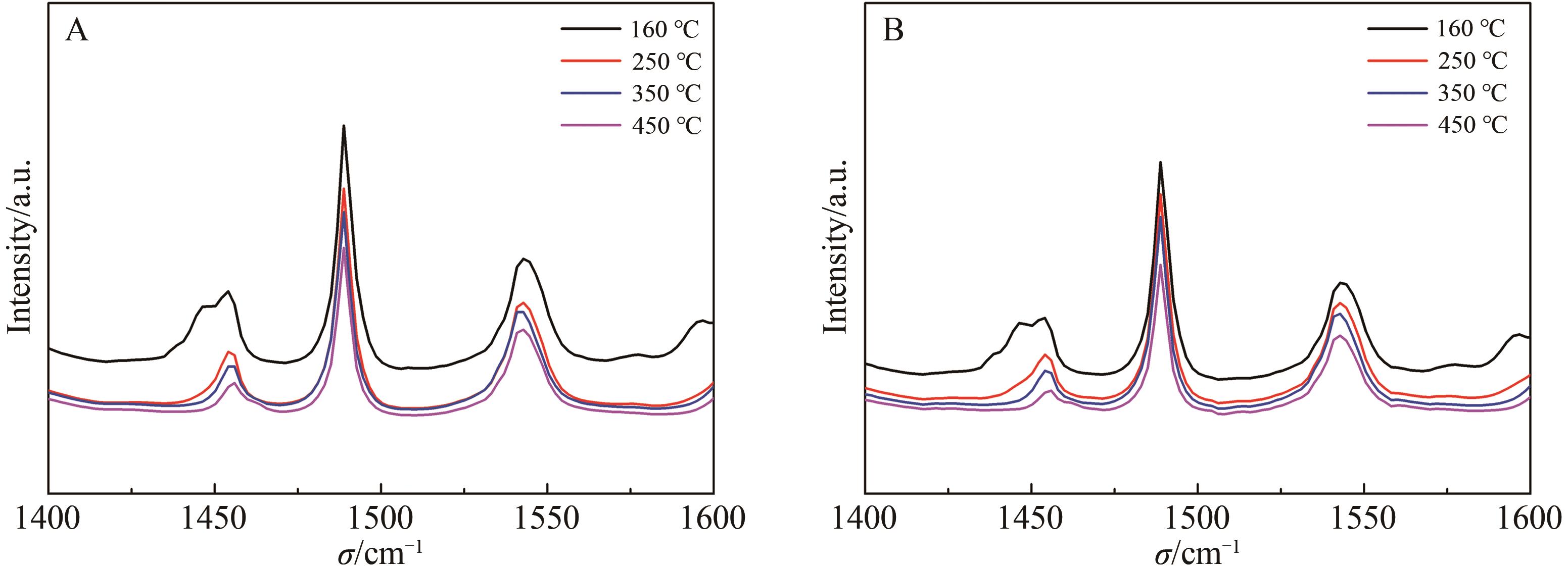
Chinese Journal of Applied Chemistry ›› 2026, Vol. 43 ›› Issue (3): 373-386.DOI: 10.19894/j.issn.1000-0518.250291
• Full Papers • Previous Articles Next Articles
Synthesis and Characterization of Highly Stable Nano-Sized Y Zeolite
Jing-Hang XUE( ), Bo QIN, Wei LIU, Tong ZHANG, Wen-Jie XIE
), Bo QIN, Wei LIU, Tong ZHANG, Wen-Jie XIE
- Sinopec Dalian Research Institute of Petroleum and Petrochemicals,Dalian 116045,China
-
Received:2025-07-20Accepted:2025-10-16Published:2026-03-01Online:2026-03-26 -
Contact:Jing-Hang XUE -
About author:xuejinghang.fshy@sinopec.com
-
Supported by:the Joint Funds of the National Natural Science Foundation of China(U24A202895)
CLC Number:
Cite this article
Jing-Hang XUE, Bo QIN, Wei LIU, Tong ZHANG, Wen-Jie XIE. Synthesis and Characterization of Highly Stable Nano-Sized Y Zeolite[J]. Chinese Journal of Applied Chemistry, 2026, 43(3): 373-386.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.250291
| Sample | SBET/(m2·g-1) | Vtotal/(cm3·g-1) | SAR |
|---|---|---|---|
| Nanoy-b | 858 | 0.37 | 5.3 |
| Nanoy-c | 861 | 0.40 | 5.1 |
| Nanoy-d | 856 | 0.37 | 4.9 |
| Nanoy-e | 859 | 0.40 | 4.6 |
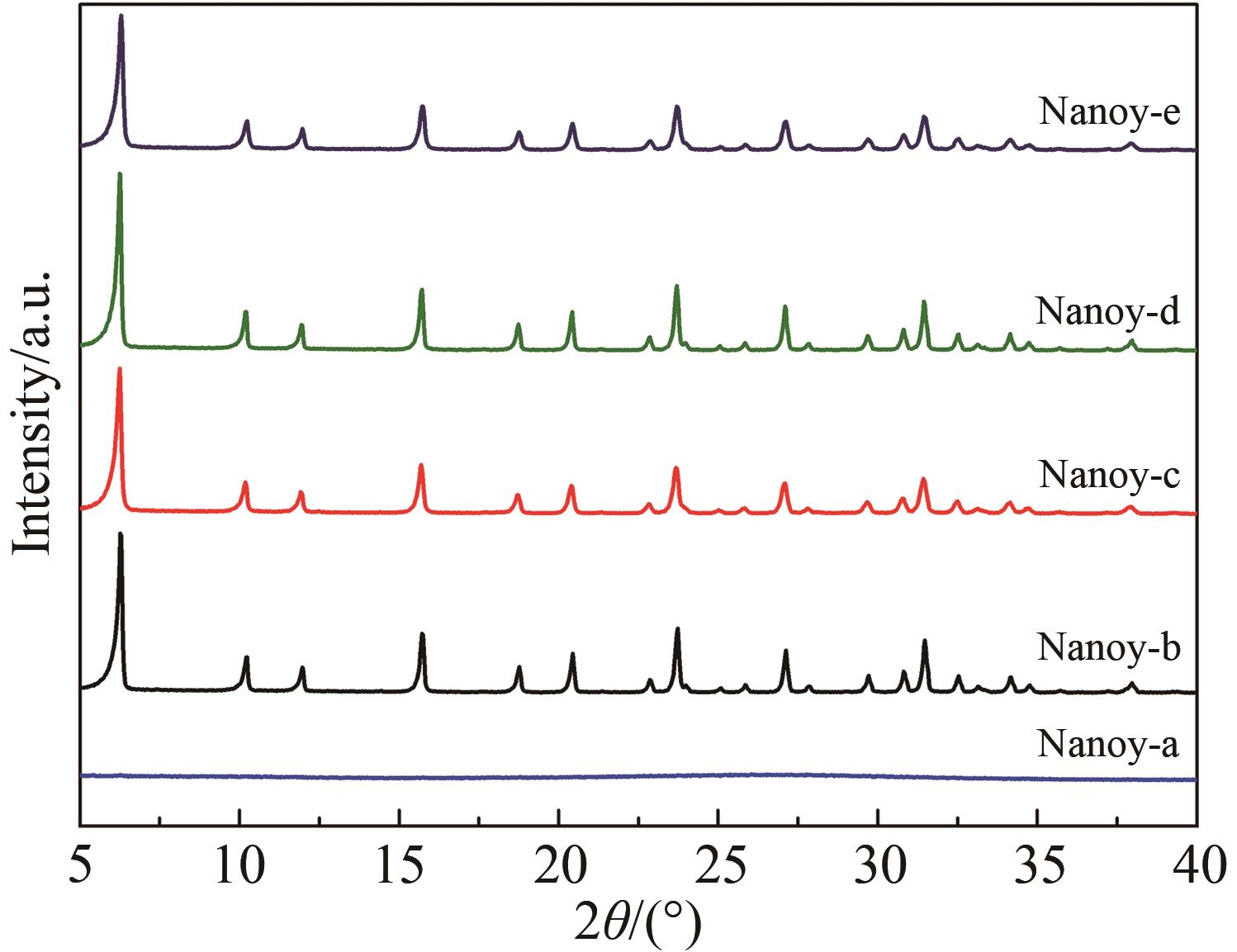
Table 1 Physicochemical properties of products obtained by different synthesis methods
| Sample | SBET/(m2·g-1) | Vtotal/(cm3·g-1) | SAR |
|---|---|---|---|
| Nanoy-b | 858 | 0.37 | 5.3 |
| Nanoy-c | 861 | 0.40 | 5.1 |
| Nanoy-d | 856 | 0.37 | 4.9 |
| Nanoy-e | 859 | 0.40 | 4.6 |
| Sample | SAR (XRF) | a0/? | SAR (XRD) a | Composition/% of Q4(nAl) | SAR (29Si NMR) b | |||
|---|---|---|---|---|---|---|---|---|
| 3 | 2 | 1 | 0 | |||||
| Nanoy-13.3-0.1 | 5.2 | 24.64 | 5.4 | 13.4 | 35.2 | 43.6 | 7.8 | 5.2 |
| Nanoy-13.3-0.3 | 5.3 | 24.64 | 5.4 | 12.5 | 33.9 | 44.9 | 8.7 | 5.3 |
| Nanoy-13.3-0.7 | 5.4 | 24.63 | 5.6 | 14.5 | 30.5 | 45.9 | 9.1 | 5.3 |
| Nanoy-13.3-1.5 | 5.7 | 24.60 | 6.1 | 9.8 | 33.0 | 46.7 | 10.5 | 5.6 |
| Nanoy-13.3-2.2 | 5.7 | 24.60 | 6.1 | 9.0 | 32.5 | 48.0 | 10.5 | 5.7 |
| Nanoy-13.3-3.0 | 5.9 | 24.61 | 6.0 | - | - | - | - | - |
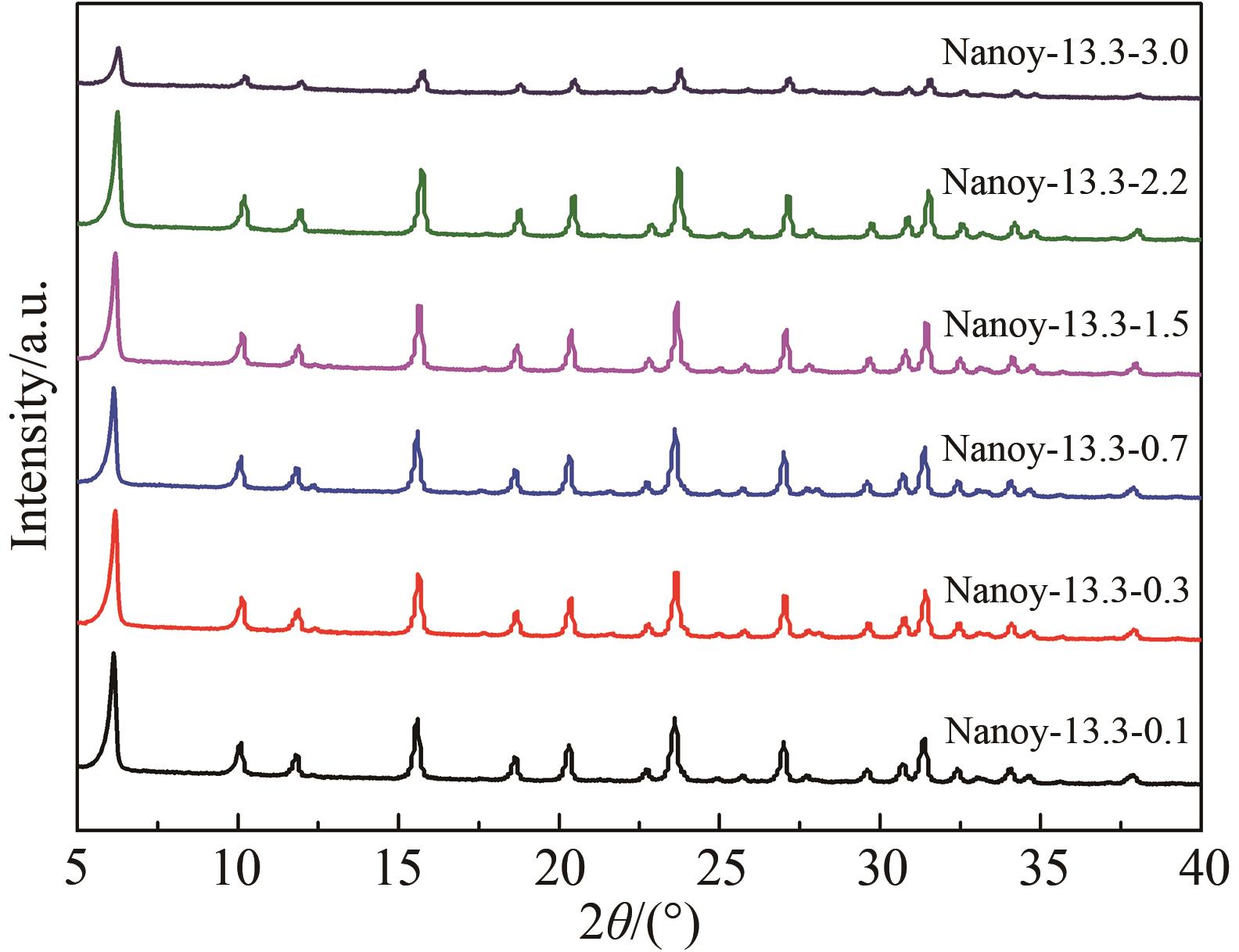
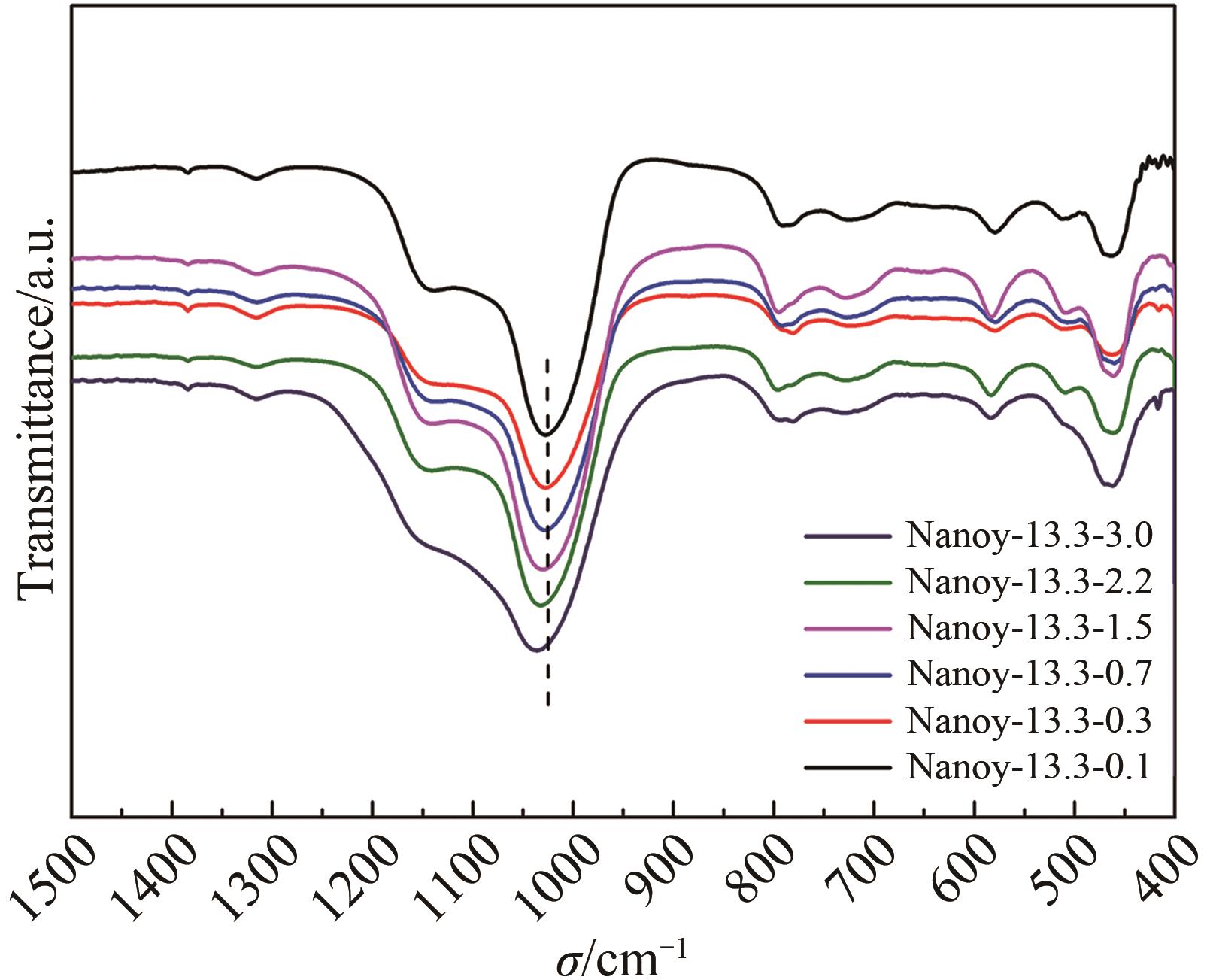
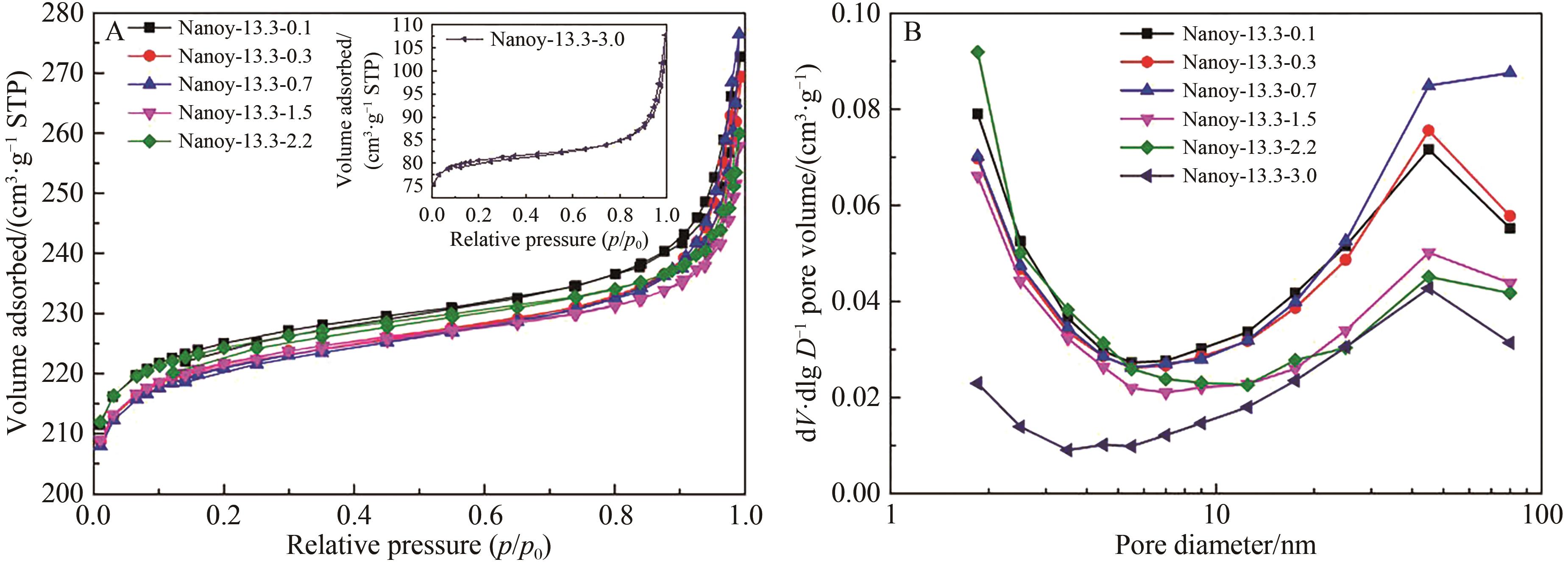
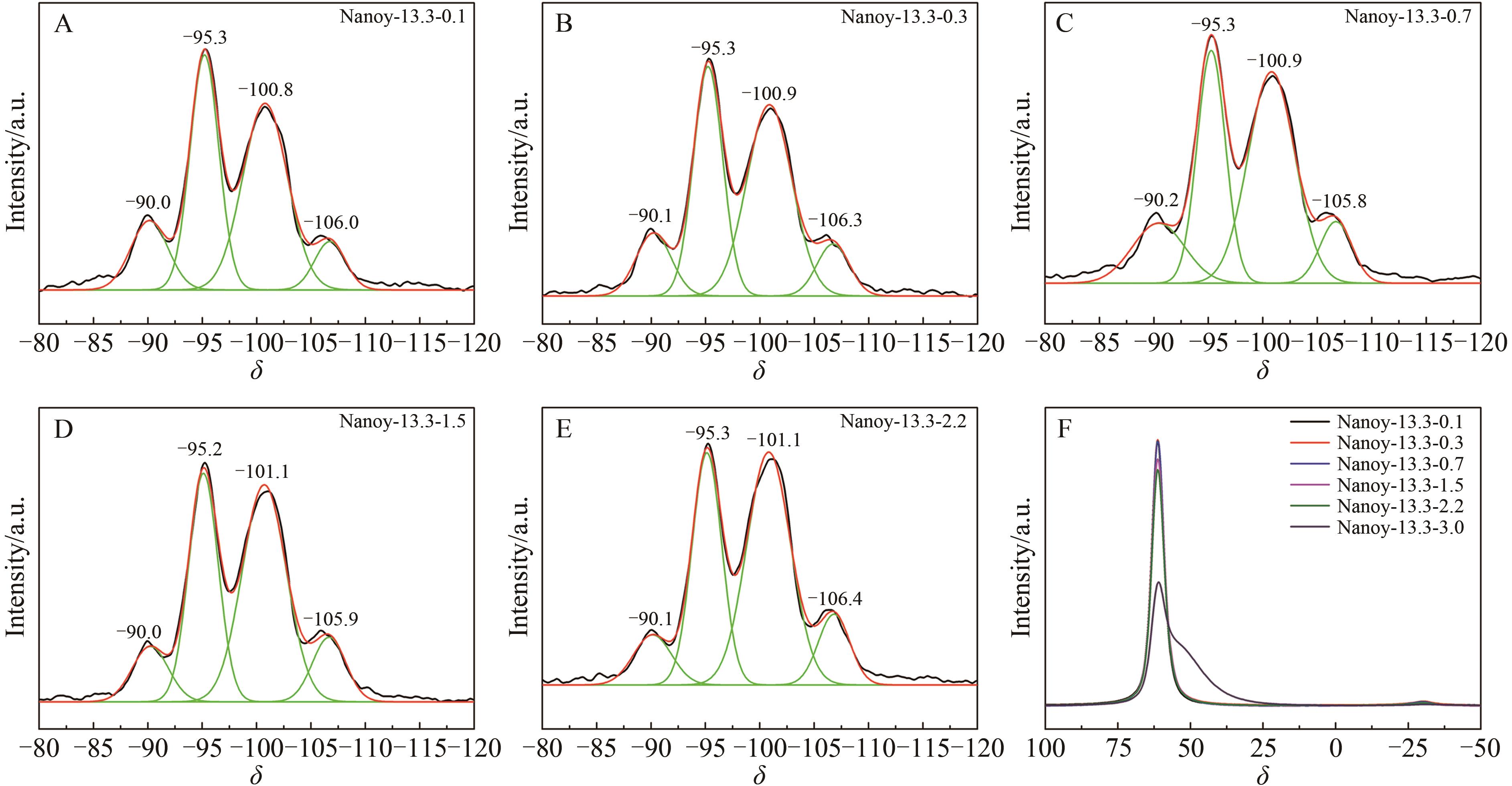
Table 2 The SAR of products with different n(AESA)∶n(Al2O3)
| Sample | SAR (XRF) | a0/? | SAR (XRD) a | Composition/% of Q4(nAl) | SAR (29Si NMR) b | |||
|---|---|---|---|---|---|---|---|---|
| 3 | 2 | 1 | 0 | |||||
| Nanoy-13.3-0.1 | 5.2 | 24.64 | 5.4 | 13.4 | 35.2 | 43.6 | 7.8 | 5.2 |
| Nanoy-13.3-0.3 | 5.3 | 24.64 | 5.4 | 12.5 | 33.9 | 44.9 | 8.7 | 5.3 |
| Nanoy-13.3-0.7 | 5.4 | 24.63 | 5.6 | 14.5 | 30.5 | 45.9 | 9.1 | 5.3 |
| Nanoy-13.3-1.5 | 5.7 | 24.60 | 6.1 | 9.8 | 33.0 | 46.7 | 10.5 | 5.6 |
| Nanoy-13.3-2.2 | 5.7 | 24.60 | 6.1 | 9.0 | 32.5 | 48.0 | 10.5 | 5.7 |
| Nanoy-13.3-3.0 | 5.9 | 24.61 | 6.0 | - | - | - | - | - |
| Sample | SBET/(m2·g-1) | Smicro/(m2·g-1) | Smeso/(m2·g-1) | Vtotal/(cm3·g-1) | Vmicro/(cm3·g-1) | Vmeso/(cm3·g-1) | SAR | a0/? | Relative crystallinity/% |
|---|---|---|---|---|---|---|---|---|---|
| Nanoy-12.3-0.5 | 832 | 764 | 68 | 0.38 | 0.30 | 0.08 | 6.0 | 24.61 | 77.77 |
| Nanoy-12.8-0.5 | 874 | 799 | 75 | 0.40 | 0.32 | 0.08 | 6.0 | 24.61 | 75.21 |
| Nanoy-13.3-0.5 | 865 | 790 | 75 | 0.40 | 0.32 | 0.08 | 5.6 | 24.63 | 64.07 |
| Nanoy-13.8-0.5 | 859 | 783 | 76 | 0.40 | 0.31 | 0.09 | 5.4 | 24.64 | 65.49 |
| Nanoy-14.3-0.5 | 844 | 767 | 77 | 0.39 | 0.31 | 0.08 | 4.2 | 24.73 | 64.24 |
| Nanoy-14.8-0.5 | 651 | 597 | 54 | 0.33 | 0.24 | 0.09 | 4.1 | 24.74 | 47.08 |
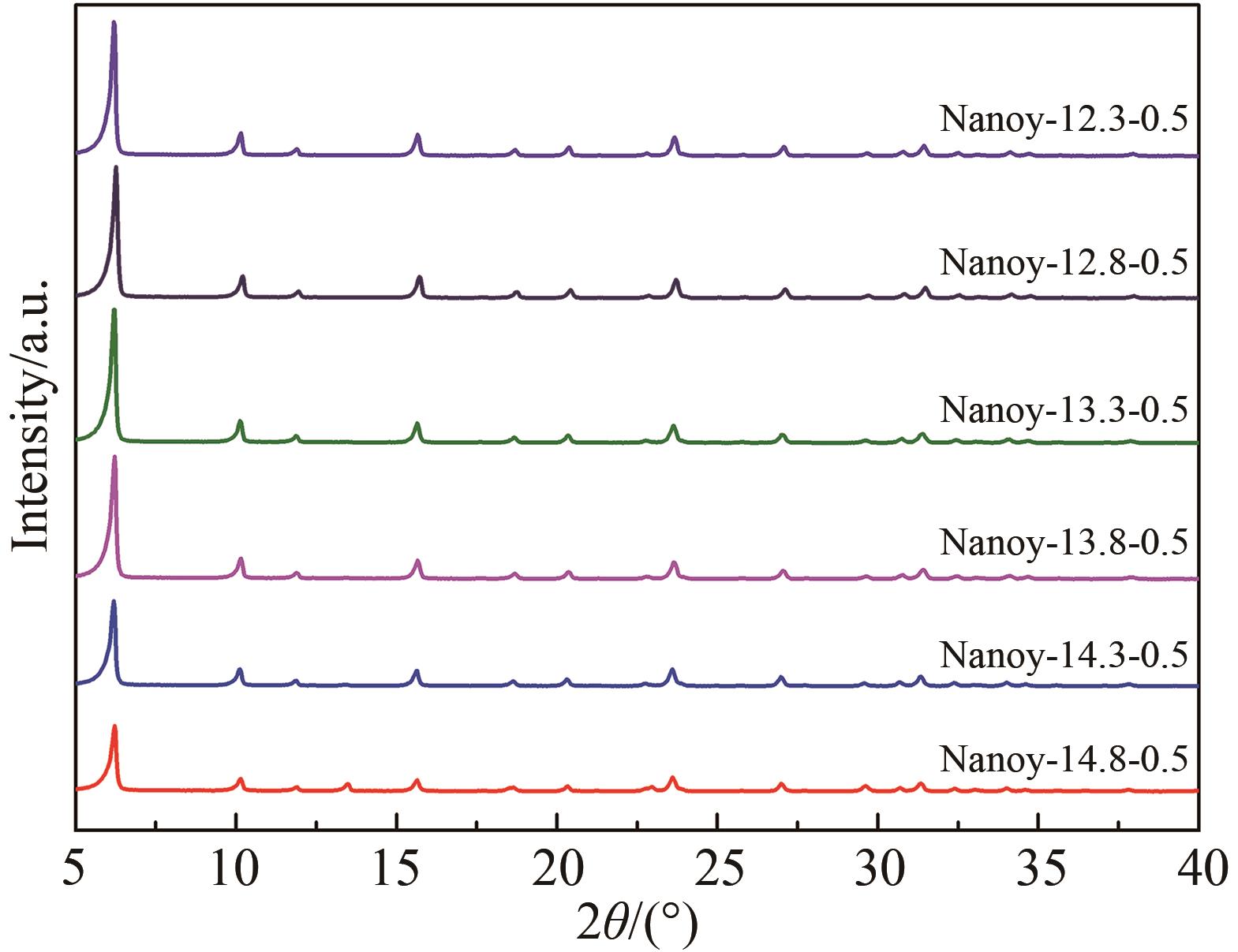
Table 3 Physicochemical properties of synthesized products with different n(Na2O)∶n(Al2O3)
| Sample | SBET/(m2·g-1) | Smicro/(m2·g-1) | Smeso/(m2·g-1) | Vtotal/(cm3·g-1) | Vmicro/(cm3·g-1) | Vmeso/(cm3·g-1) | SAR | a0/? | Relative crystallinity/% |
|---|---|---|---|---|---|---|---|---|---|
| Nanoy-12.3-0.5 | 832 | 764 | 68 | 0.38 | 0.30 | 0.08 | 6.0 | 24.61 | 77.77 |
| Nanoy-12.8-0.5 | 874 | 799 | 75 | 0.40 | 0.32 | 0.08 | 6.0 | 24.61 | 75.21 |
| Nanoy-13.3-0.5 | 865 | 790 | 75 | 0.40 | 0.32 | 0.08 | 5.6 | 24.63 | 64.07 |
| Nanoy-13.8-0.5 | 859 | 783 | 76 | 0.40 | 0.31 | 0.09 | 5.4 | 24.64 | 65.49 |
| Nanoy-14.3-0.5 | 844 | 767 | 77 | 0.39 | 0.31 | 0.08 | 4.2 | 24.73 | 64.24 |
| Nanoy-14.8-0.5 | 651 | 597 | 54 | 0.33 | 0.24 | 0.09 | 4.1 | 24.74 | 47.08 |
| Sample | SBET/(m2·g-1) | Smicro/(m2·g-1) | Smeso/(m2·g-1) | Vtotal/(cm3·g-1) | Vmicro/(cm3·g-1) | Vmeso/(cm3·g-1) | SAR | a0/? | Relative crystallinity/% | Reduction rate of relative crystallinity x /% |
|---|---|---|---|---|---|---|---|---|---|---|
| Coml Y | 850 | 797 | 53 | 0.36 | 0.32 | 0.04 | 5.0 | 24.67 | 93.0 | - |
| Nano Y | 887 | 807 | 80 | 0.41 | 0.32 | 0.09 | 5.8 | 24.62 | 73.0 | - |
| Coml-USY | 537 | 431 | 106 | 0.33 | 0.17 | 0.16 | 5.0 | 24.39 | 51.2 | 44.9 |
| Nano-USY | 623 | 549 | 74 | 0.36 | 0.22 | 0.14 | 5.8 | 24.39 | 61.3 | 16.0 |
| Coml-AUSY | 730 | 579 | 151 | 0.45 | 0.23 | 0.22 | 14.7 | 24.39 | 54.9 | 41.0 |
| Nano-AUSY | 786 | 671 | 115 | 0.44 | 0.27 | 0.17 | 15.6 | 24.38 | 67.8 | 7.1 |
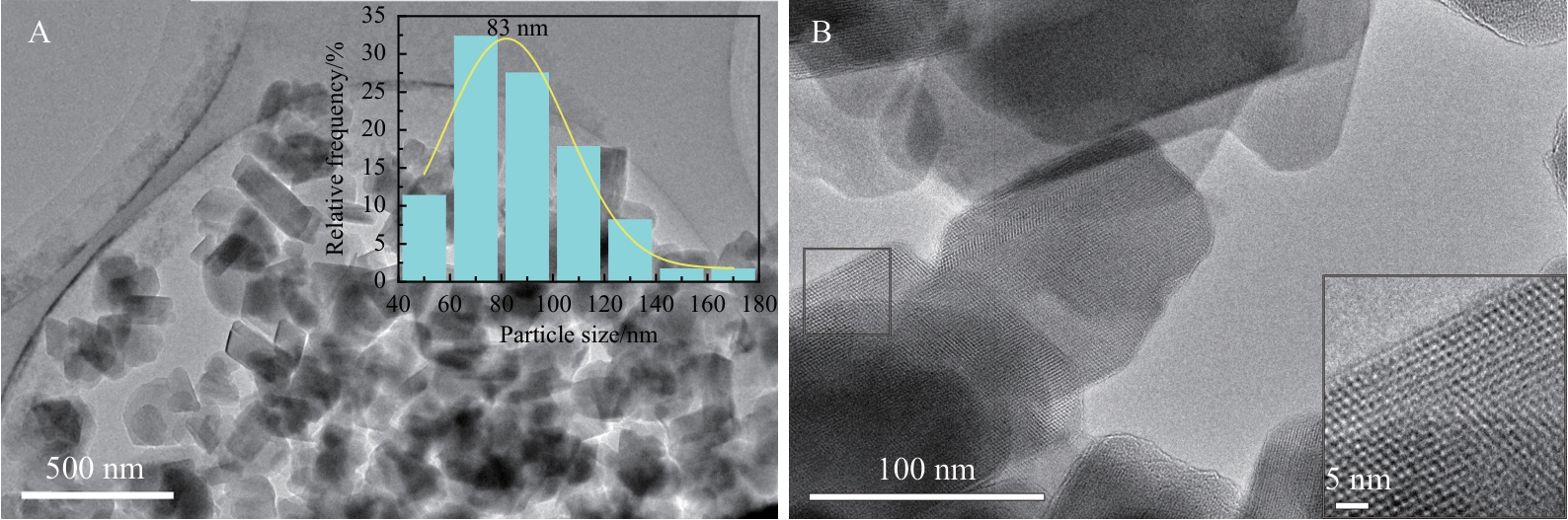
Table 4 Comparison of the physicochemical properties of commercial Y and nanosized Y zeolite
| Sample | SBET/(m2·g-1) | Smicro/(m2·g-1) | Smeso/(m2·g-1) | Vtotal/(cm3·g-1) | Vmicro/(cm3·g-1) | Vmeso/(cm3·g-1) | SAR | a0/? | Relative crystallinity/% | Reduction rate of relative crystallinity x /% |
|---|---|---|---|---|---|---|---|---|---|---|
| Coml Y | 850 | 797 | 53 | 0.36 | 0.32 | 0.04 | 5.0 | 24.67 | 93.0 | - |
| Nano Y | 887 | 807 | 80 | 0.41 | 0.32 | 0.09 | 5.8 | 24.62 | 73.0 | - |
| Coml-USY | 537 | 431 | 106 | 0.33 | 0.17 | 0.16 | 5.0 | 24.39 | 51.2 | 44.9 |
| Nano-USY | 623 | 549 | 74 | 0.36 | 0.22 | 0.14 | 5.8 | 24.39 | 61.3 | 16.0 |
| Coml-AUSY | 730 | 579 | 151 | 0.45 | 0.23 | 0.22 | 14.7 | 24.39 | 54.9 | 41.0 |
| Nano-AUSY | 786 | 671 | 115 | 0.44 | 0.27 | 0.17 | 15.6 | 24.38 | 67.8 | 7.1 |
| Sample | Acid amount(160 ℃)/(mmol·g-1) | Acid amount(450 ℃)/(mmol·g-1) | ||||||
|---|---|---|---|---|---|---|---|---|
| B | L | B+L | B/L | B | L | B+L | B/L | |
| Coml-AUSY | 0.52 | 0.24 | 0.76 | 2.17 | 0.29 | 0.07 | 0.36 | 4.14 |
| Nano-AUSY | 0.61 | 0.26 | 0.87 | 2.35 | 0.37 | 0.06 | 0.43 | 6.17 |
Table 5 Py-FT-IR acid content of Y zeolite after acid treatment
| Sample | Acid amount(160 ℃)/(mmol·g-1) | Acid amount(450 ℃)/(mmol·g-1) | ||||||
|---|---|---|---|---|---|---|---|---|
| B | L | B+L | B/L | B | L | B+L | B/L | |
| Coml-AUSY | 0.52 | 0.24 | 0.76 | 2.17 | 0.29 | 0.07 | 0.36 | 4.14 |
| Nano-AUSY | 0.61 | 0.26 | 0.87 | 2.35 | 0.37 | 0.06 | 0.43 | 6.17 |
| [1] | LI T S, LIU H Y, FAN Y, et al. Synthesis of zeolite Y from natural aluminosilicate minerals for fluid catalytic cracking application[J]. Green Chem, 2012, 14(12): 3255-3259. |
| [2] | DE JONG K P, ZECEVIC J, FRIEDRICH H, et al. Zeolite Y crystals with trimodal porosity as ideal hydrocracking catalysts[J]. Angew Chem Int Ed, 2010, 49(52): 10074-10078. |
| [3] | WEI L J, PENG P, QIAO K, et al. Hierarchical Y zeolite with accessible intracrystalline mesopores and enhanced hydrocracking performances via sequential multi-carboxyl acid and fluoroborate post-treatments[J]. Fuel, 2023, 354: 129289. |
| [4] | MASTROPIETRO T F, DRIOLI E, POERIO T. Low temperature synthesis of nanosized NaY zeolite crystals from organic-free gel by using supported seeds[J]. RSC Adv, 2014, 4(42): 21951-21957. |
| [5] | WANG B, LI Y Z, SHAO C R, et al. Rapid and high yield synthesis method of colloidal nano faujasite[J]. Microporous Mesoporous Mater, 2016, 230: 89-99. |
| [6] | AWALA H, GILSON J P, RETOUX R, et al. Template-free nanosized faujasite-type zeolites[J]. Nat Mater, 2015, 14(4): 447-451. |
| [7] | RADMAN H M, DABBAWALA A A, ISMAIL I, et al. Influence of salt on nanozeolite-Y particles size synthesized under organic template-free condition[J]. Microporous Mesoporous Mater, 2019, 282: 73-81. |
| [8] | BOREL M, DODIN M, DAOU T J, et al. SDA-free hydrothermal synthesis of high-silica ultra-nanosized zeolite Y[J]. Crystal Growth Design, 2017, 17(3): 1173-1179. |
| [9] | HOLMBERG B A, WANG H T, NORBECK J M, et al. Controlling size and yield of zeolite Y nanocrystals using tetramethylammonium bromide[J]. Microporous Mesoporous Mater, 2003, 59(1): 13-28. |
| [10] | QI J Y, GUO Y N, JIA H Q, et al. Unpredictable properties of industrial HY zeolite for tetralin hydrocracking[J]. Fuel Process Technol, 2023, 240: 107586. |
| [11] | GARCIA-MARTINEZ J, JOHNSON M, VALLA J, et al. Mesostructured zeolite Y-high hydrothermal stability and superior FCC catalytic performance[J]. Catal Sci Technol, 2012, 2(5): 987-994. |
| [12] | WANG J, WANG L Y, ZHU D L, et al. Progress in direct synthesis of high silica zeolite Y[J]. Chem J Chin Univ, 2021, 42(1): 10. |
| [13] | PENG P, ZHANG Z Q, WANG Y H, et al. Hierarchical molecular sieves: synthesis and catalytic applications[J]. Prog Chem, 2013, 25(12): 2028-2037. |
| [14] | ZHANG X W, GUO Q, QIN B, et al. Structural features of binary microporous zeolite composite Y-beta and its hydrocracking performance[J]. Catal Today, 2010, 149(1/2): 212-217. |
| [15] | LI Q, CREASER D, STERTE J. An investigation of the nucleation/crystallization kinetics of nanosized colloidal faujasite zeolites[J]. Chem Mater, 2002, 14(3):1319-1324. |
| [16] | CHAVES T F, PASTORE H O, CARDOSO D. A simple synthesis procedure to prepare nanosized faujasite crystals[J]. Microporous Mesoporous Mater, 2012, 161: 67-75. |
| [17] | TANG T, ZHANG L, DONG H, et al. Organic template-free synthesis of zeolite Y nanoparticle assemblies and their application in the catalysis of the Ritter reaction[J]. RSC Adv, 2017, 7(13): 7711-7717. |
| [18] | HUO Q, DOU T, ZHAO Z, et al. Synthesis and application of a novel mesoporous zeolite L in the catalyst for the HDS of FCC gasoline[J]. Appl Catal A Gen, 2010, 381(1/2): 101-108. |
| [1] | LUO Caiwu, ZHAO Yong, JIANG Fuliang, LI Xiangyang, CHAO Zisheng. Synthesis of Pyridine and 3-Picoline from Acrolein Diethyl Acetal and Ammonia over La- and KF-modified Y Type Zeolite Catalysts [J]. Chinese Journal of Applied Chemistry, 2018, 35(5): 559-563. |
| [2] | Ma Dengjun, Tan Junra, Chen Xiuzeng, Wang Shuzi, Li Jinsheng. Photochemical Preparation and Quantum Size Effect of Nano-Grade TiO2 Micromicellae [J]. Chinese Journal of Applied Chemistry, 1996, 0(6): 11-14. |
| [3] | Jiao Qingze, Guo Jinfu, Mao Liqiu, Jiang Dazhen. Ion Exchange in NaY Zeolite withi La3+ under Microwave Radiation [J]. Chinese Journal of Applied Chemistry, 1994, 0(3): 84-85. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||