
Chinese Journal of Applied Chemistry ›› 2023, Vol. 40 ›› Issue (12): 1630-1642.DOI: 10.19894/j.issn.1000-0518.230203
• Review • Previous Articles Next Articles
Research Progress on Preparation and Adsorption of Water Pollutants of Bimetallic Metal-Organic Framework
Ke-Xin YANG1,2, Jie ZHOU2, Yu-Shan HOU1,2, Yao-Wei ZHANG1, Chen YIN2,3, Dong-Hui XU2, Guang-Yang LIU2( )
)
- 1.College of Horticulture and Landscape Architecture,Northeast Agricultural University,Harbin 150030,China
2.Institute of Vegetables and Flowers,Chinese Academy of Agricultural Sciences,Beiing 100081,China
3.College of Agriculture,Forestry and Technology,Hebei North University,Zhangjiakou 075132,China
-
Received:2023-07-13Accepted:2023-11-06Published:2023-12-01Online:2024-01-03 -
Contact:Guang-Yang LIU -
About author:liuguangyang@caas.cn
-
Supported by:the National Key Research and Development Program of China(2022YFF0606800);the Special Fund of the Construction of the National Modern Agricultural Technology Industrial System(CARS-23-E03)
CLC Number:
Cite this article
Ke-Xin YANG, Jie ZHOU, Yu-Shan HOU, Yao-Wei ZHANG, Chen YIN, Dong-Hui XU, Guang-Yang LIU. Research Progress on Preparation and Adsorption of Water Pollutants of Bimetallic Metal-Organic Framework[J]. Chinese Journal of Applied Chemistry, 2023, 40(12): 1630-1642.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.230203

Fig.2 (A) One-step microwave-assisted synthesis of Co/Zn-MOF[26]; (B) Mechanochemical synthesis of bimetallic CoZn-ZIF[31]; (C) Post-synthesis modification synthesis of Fe/Ni-MOF[35]; (D) Synthesis of Cu-TMA by in-situ metal substitution method[36]
| Simple operation, high yield, high crystallinity, low cost, easy to form crystals with large specific surface area | The binding kinetics between the two metal ions and the ligands are different, resulting in an unstable topology of the obtained metal frameworks | Precise control of reaction conditions. Such as molar ratio, reaction time, solubility of metal ions and pH of the reactant solution, etc | ||
| Microwave irradiation method | The use of electric or magnetic fields to induce high-speed collisions between charged particles to produce high-purity crystals | The crystal particles formed are too small and the cost and yield are not always proportional | The reaction conditions were controlled by changing the irradiation power, reaction time and temperature | |
| Ambient stirring method | Fast and simple operation, avoids aggregation caused by in-situ solvent heat conditions, enables large-scale production | Poor stability performance | Strictly control the reaction conditions, such as the order of addition reaction time, etc | |
| Metal-substitution method | MOFs materials that cannot be prepared by conventional methods can be obtained by substitution reaction | Incorporation of a second metal usually produces a fragile framework | Selection of metals with similar ionic radii and coordination geometries, similar reactivity, size, electronegativity |
Table 1 The advantages and disadvantages of the preparation methods of bimetallic MOFs
| Simple operation, high yield, high crystallinity, low cost, easy to form crystals with large specific surface area | The binding kinetics between the two metal ions and the ligands are different, resulting in an unstable topology of the obtained metal frameworks | Precise control of reaction conditions. Such as molar ratio, reaction time, solubility of metal ions and pH of the reactant solution, etc | ||
| Microwave irradiation method | The use of electric or magnetic fields to induce high-speed collisions between charged particles to produce high-purity crystals | The crystal particles formed are too small and the cost and yield are not always proportional | The reaction conditions were controlled by changing the irradiation power, reaction time and temperature | |
| Ambient stirring method | Fast and simple operation, avoids aggregation caused by in-situ solvent heat conditions, enables large-scale production | Poor stability performance | Strictly control the reaction conditions, such as the order of addition reaction time, etc | |
| Metal-substitution method | MOFs materials that cannot be prepared by conventional methods can be obtained by substitution reaction | Incorporation of a second metal usually produces a fragile framework | Selection of metals with similar ionic radii and coordination geometries, similar reactivity, size, electronegativity |
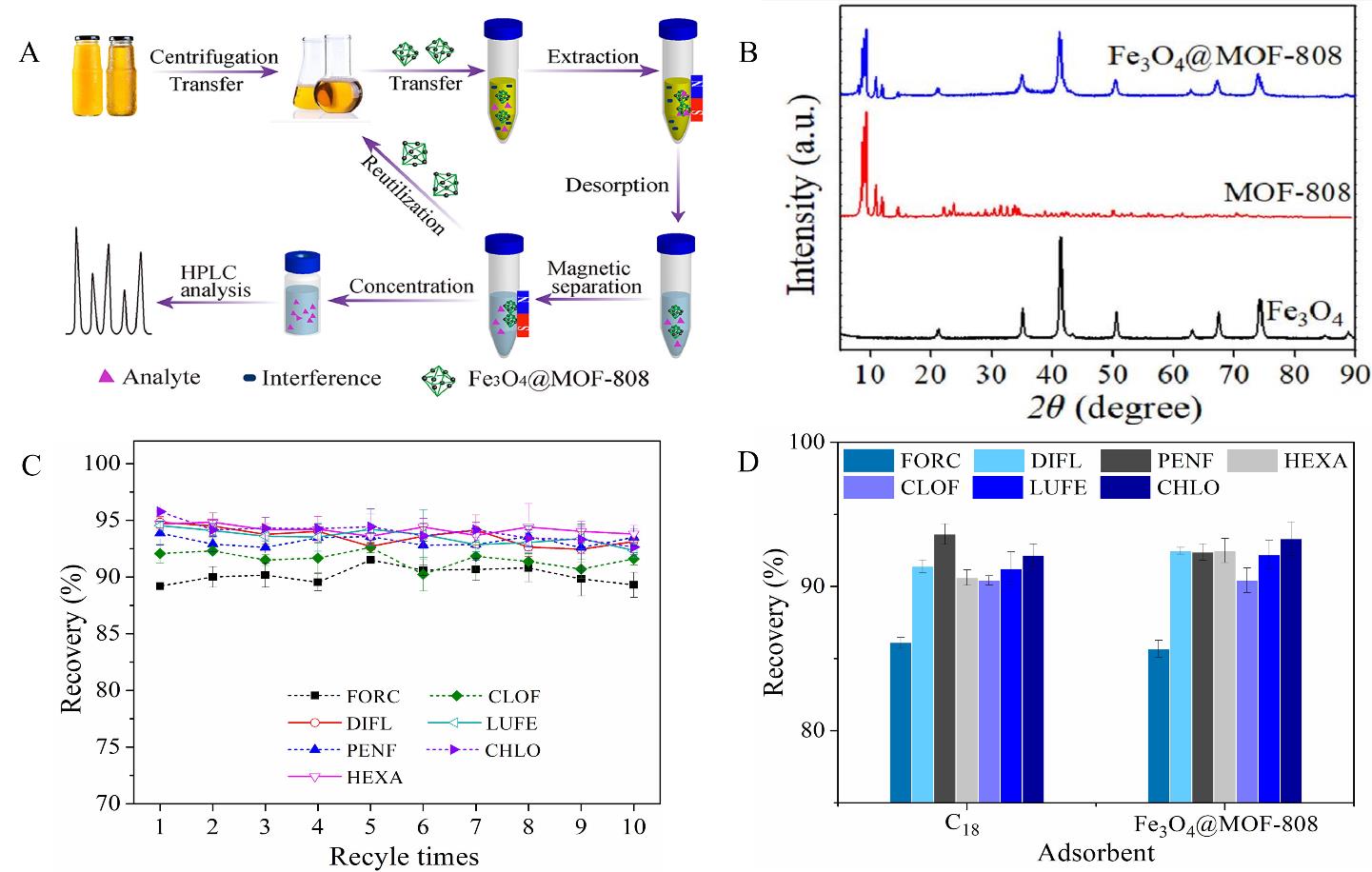
Fig.3 (A) Schematic of the MSPE procedure for BUs from tea beverages and juice samples based on Fe3O4@MOF-808[48]; (B) XRD patterns of Fe3O4@MOF-808, MOF-808 and Fe3O4[48]; (C) The effect of recycle times of Fe3O4@MOF-808 on the recoveries of BUs[48]; (D) Comparison of the recoveries of Fe3O4@MOF-808 with commercial C18 sorbent for BUs[48]
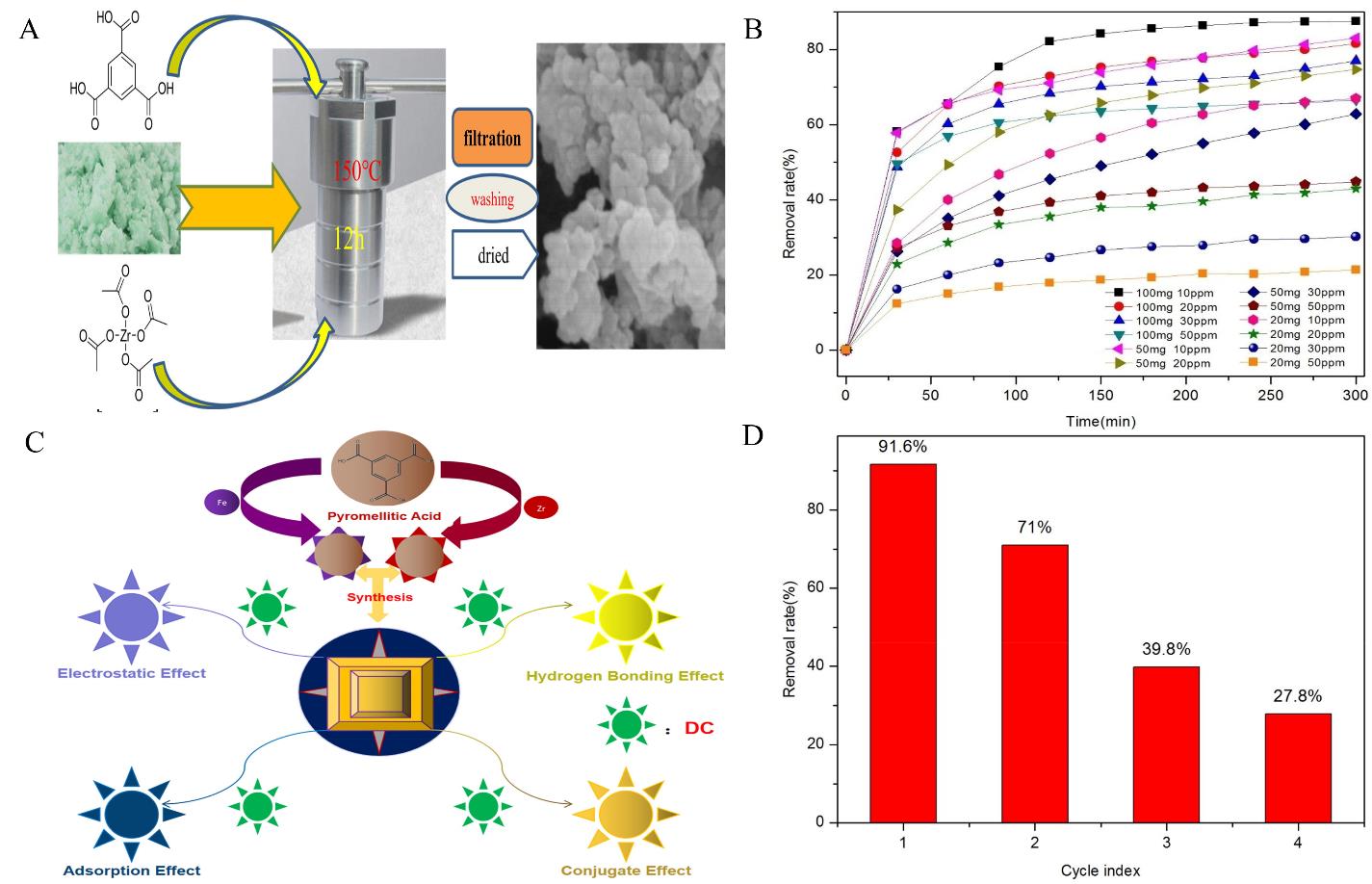
Fig.4 (A) The preparation process of the Fe/Zr-MOFs[57]; (B) Removal rate of Fe/Zr-MOFs[57]; (C) Reusability of Fe/Zr-MOFs for the removal of DC[57]; (D) The adsorption mechanism of CR by Fe/Zr-MOFs[57]

Fig.5 (A) ?SEM images of ZIF-ZnCo-8∶?1[69]?; (B) FT-IR and XRD of ZIF-ZnCo-8∶?1 before and after photocatalytic[69]; (C) Photocatalytic performances of ZIF-ZnCo to Cr6+ and mixed of Cr6+ and CR[69]
| 1 | SVENSSON G E, CHACON-GARCIA A J, ROJAS S, et al. Removal of pharmaceutical pollutants from effluent by a plant-based metal-organic framework[J]. Nat Water, 2023, 1(5): 433-442. |
| 2 | HUANG D, WU J, WANG L, et al. Novel insight into adsorption and co-adsorption of heavy metal ions and an organic pollutant by magnetic graphene nanomaterials in water[J]. Chem Eng J, 2019, 358: 1399-1409. |
| 3 | MARTINEZ-HUITLE C A, PANIZZA M. Electrochemical oxidation of organic pollutants for wastewater treatment[J]. Curr Opin Electrochem, 2018, 11: 62-71. |
| 4 | CHEN Z, YANG B, WEN Q, et al. Evaluation of enhanced coagulation combined with densadeg-ultrafiltration process in treating secondary effluent: organic micro-pollutants removal, genotoxicity reduction, and membrane fouling alleviation[J]. J Hazard Mater, 2020, 396: 122697. |
| 5 | KANG W, CHEN S, YU H, et al. Photocatalytic ozonation of organic pollutants in wastewater using a flowing through reactor[J]. J Hazard Mater, 2021, 405: 124277. |
| 6 | Li X, YUAN H, QUAN X, et al. Effective adsorption of sulfamethoxazole, bisphenol A and methyl orange on nanoporous carbon derived from metal-organic frameworks[J]. J Environ Sci, 2018, 63: 250-259. |
| 7 | KUMAR S, JAIN S, NEHRA M, et al. Green synthesis of metal-organic frameworks: a state-of-the-art review of potential environmental and medical applications[J]. Coordin Chem Rev, 2020, 420: 213407. |
| 8 | HASAN Z, JHUNG S H. Removal of hazardous organics from water using metal-organic frameworks (MOFs): plausible mechanisms for selective adsorptions[J]. J Hazard Mater, 2015, 283: 329-339. |
| 9 | CHEN L, WANG H F, LI C, et al. Bimetallic metal-organic frameworks and their derivatives[J]. Chem Sci, 2020, 11(21): 5369-5403. |
| 10 | ZHANG H, WANG J, TENG Y, et al. Ce-MOF composite electrospinning as antibacterial adsorbent for the removal of 2,4-dichlorophenoxyacetic acid[J]. Chem Eng J, 2023, 462: 142195. |
| 11 | ZHAI Q G, BU X, MAO C, et al. Systematic and dramatic tuning on gas sorption performance in heterometallic metal-organic frameworks[J]. J Am Chem Soc, 2016, 138(8): 2524-2527. |
| 12 | 高宇航, 高明坤, 黄晓冬, 等. 磁性金属有机骨架材料的合成及其在食品检测前处理中的应用研究[J]. 食品科技, 2020, 45(7): 324-331. |
| GAO Y H, GAO M K, HUANG X D, et al. Application of magnetic metal-organic framework composites in food quality safety and inspection[J]. Food Sci Technol, 2020, 45(7): 324-331. | |
| 13 | 花雨薇, 刘广洋, 刘中笑, 等. MOFs在高级氧化降解环境污染物中的应用进展[J]. 工业水处理, 2022: 1-19. |
| HUA Y W, LIU G Y, LIU Z X, et al. Applioncation progress of MOFs in advanced oxidation process of environmental pollutants[J]. Ind Water Treat, 2022: 1-19. | |
| 14 | GAO M, LIU G, GAO Y, et al. Recent advances in metal-organic frameworks/membranes for adsorption and removal of metal ions[J]. TrAC Trends Anal Chem, 2021, 137: 116226. |
| 15 | LIU G, ZHANG X, LU M, et al. Adsorption and removal of organophosphorus pesticides from Chinese cabbages and green onions by using metal organic frameworks based on the mussel-inspired adhesive interface[J]. Food Chem, 2022, 393: 133337. |
| 16 | LOLOEI M, KALIAGUINE S, RODRIGUE D. CO2-selective mixed matrix membranes of bimetallic Zn/Co-ZIF vs. ZIF-8 and ZIF-67[J]. Separat Purif Technol, 2022, 296: 121391. |
| 17 | MPHUTHI L E, ERASMUS E, LANGNER E H G. Metal exchange of ZIF-8 and ZIF-67 nanoparticles with Fe(Ⅱ) for enhanced photocatalytic performance[J]. ACS Omega, 2021, 6(47): 31632-31645. |
| 18 | 王雨萌, 杨蓉, 邓七九, 等. 双金属MOFs及其衍生物在电化学储能领域中的应用[J]. 化学进展, 2022, 34(2): 460-473. |
| WANG Y M, YANG R, DENG Q J, et al. Application of bimetallic MOFs and their derivatives in electrochemical energy storage[J]. Prog Chem, 2022, 34(2): 460-473. | |
| 19 | ZHANG H, WANG J, TENG Y, et al. Ce-MOF composite electrospinning as antibacterial adsorbent for the removal of 2,4-dichlorophenoxyacetic acid[J]. Chem Eng J, 2023, 462: 142195. |
| 20 | DAI W, WANG X, CHEN G, et al. Facile synthesis of 2D europium-metal organic frameworks nanosheets for highly efficient electrochemiluminescence in DNA detection[J]. Chem Eng J, 2023, 465: 143037. |
| 21 | GLOWNIAK S, SZCZESNIAK B, CHOMA J, et al. Mechanochemistry: toward green synthesis of metal-organic frameworks[J]. Mater Today, 2021, 46: 109-124. |
| 22 | YU K, LEE Y R, SEO J Y, et al. Sonochemical synthesis of Zr-based porphyrinic MOF-525 and MOF-545: enhancement in catalytic and adsorption properties[J]. Microporous Mesoporous Mat, 2021, 316: 110985. |
| 23 | RAZA N, KUMAR T, SINGH V, et al. Recent advances in bimetallic metal-organic framework as a potential candidate for supercapacitor electrode material[J]. Coordin Chem Rev, 2021, 430: 213660. |
| 24 | LING J, ZHOU A, WANG W, et al. One-pot method synthesis of bimetallic MgCu-MOF-74 and its CO2 adsorption under visible light[J]. ACS Omega, 2022, 7(23): 19920-19929. |
| 25 | VILLAJOS J A, OREAJO G, MARTOS C, et al. Co/Ni mixed-metal sited MOF-74 material as hydrogen adsorbent[J]. Int J Hydrogen Energy, 2015, 40(15): 5346-5352. |
| 26 | LI X, DONG H, FAN Q, et al. One-pot, rapid microwave-assisted synthesis of bimetallic metal-organic framework for efficient enzyme-free glucose detection[J]. Microchem J, 2022, 179: 107468. |
| 27 | NGUYEN H T T, TRAN K N T, TAN L V, et al. Microwave-assisted solvothermal synthesis of bimetallic metal-organic framework for efficient photodegradation of organic dyes[J]. Mater Chem Phys, 2021, 272: 125040. |
| 28 | AHMED M. Recent advancement in bimetallic metal organic frameworks (M′MOFs): synthetic challenges and applications[J]. Inorg Chem Front, 2022, 9(12): 3003-3033. |
| 29 | YUAN W, O'CONNOR J, JAMES S L. Mechanochemical synthesis of homo- and hetero-rare-earth(Ⅲ) metal-organic frameworks by ball milling[J]. CrystEngComm, 2010, 12(11): 3515-3517. |
| 30 | PANEQUE A, FERNANDEZ B J, REGUERA E, et al. Mechanochemical synthesis of hemin-imidazole complexes[J]. Trans Met Chem, 2001, 26(1): 76-80. |
| 31 | IMAWAKA K, SUGITA M, TAKEWAKI T, et al. Mechanochemical synthesis of bimetallic CoZn-ZIFs with sodalite structure[J]. Polyhedron, 2019, 158: 290-295. |
| 32 | WANG W, CHAI M, BIN-ZULKIFLI M Y, et al. Metal-organic framework composites from a mechanochemical process[J]. Mol Systems Design Eng, 2023, 8(5): 560-579. |
| 33 | MANDAL S, NATARAJAN S, MANI P, et al. Post-synthetic modification of metal-organic frameworks toward applications[J]. Adv Funct Mater, 2021, 31(4): 2006291. |
| 34 | 元宁, 杜冰洁, 贾晓霞, 等. 双金属金属有机骨架材料的制备及性能研究进展[J]. 应用化学, 2018, 35(5): 500-510. |
| YUAN N, DU B J, JIA X X, et al. Research progress in preparation technology and application of bimetal metal-organic frameworks materials[J]. Chin J Appl Chem 2018, 35(5): 500-510. | |
| 35 | FU J Y, LIN Z Y, XIE J Y, et al. Post-modified FeNi metal-organic frameworks assisted by microwave for oxygen evolution reaction[J]. Mater Lett, 2022, 324: 132748. |
| 36 | HE X, CHEN D R, WANG W N. Bimetallic metal-organic frameworks (MOFs) synthesized using the spray method for tunable CO2 adsorption[J]. Chem Eng J, 2020, 382: 122825. |
| 37 | HE X, WANG W N. Rational design of efficient semiconductor-based photocatalysts via microdroplets: a review[J]. KONA Powder Particle J, 2019, 36: 201-214. |
| 38 | DONG S, LI Z, FU Y, et al. Bimetal-organic framework Cu-Ni-BTC and its derivative CuO@NiO: construction of three environmental small-molecule electrochemical sensors[J]. J Electroanal Chem, 2020, 858: 113785. |
| 39 | WANG R, XU H, ZHANG K, et al. High-quality Al@Fe-MOF prepared using Fe-MOF as a micro-reactor to improve adsorption performance for selenite[J]. J Hazard Mater, 2019, 364: 272-280. |
| 40 | CHENG W, WANG Y, GE S, et al. One-step microwave hydrothermal preparation of Cd/Zr-bimetallic metal-organic frameworks for enhanced photochemical properties[J]. Adv Composites Hybrid Mater, 2021, 4(1): 150-161. |
| 41 | GAIKWAD R, GAIKWAD S, HAN S. Bimetallic UTSA-16 (Zn, X; X=Mg, Mn, Cu) metal organic framework developed by a microwave method with improved CO2 capture performances[J]. J Ind Eng Chem, 2022, 111: 346-355. |
| 42 | HE H, LI L, LIU Y, et al. Rapid room-temperature synthesis of a porphyrinic MOF for encapsulating metal nanoparticles[J]. Nano Res, 2021, 14(2): 444-449. |
| 43 | LIU S, QIU Y, LIU Y, et al. Recent advances in bimetallic metal-organic frameworks (BMOFs): synthesis, applications and challenges[J]. New J Chem, 2022, 46(29): 13818-13837. |
| 44 | MUBULA Y, YU M, YANG D et al. Recovery of valuable elements from solid waste with the aid of external electric field: a review[J]. J Environ Chem Eng, 2023, 11(6): 111237. |
| 45 | LI J, HUANG W, WANG M, et al. Low-crystalline bimetallic metal-organic framework electrocatalysts with rich active sites for oxygen evolution[J]. ACS Energy Lett, 2019, 4(1): 285-292. |
| 46 | MONDOL M M H, JHUNG S H. Adsorptive removal of pesticides from water with metal-organic framework-based materials[J]. Chem Eng J, 2021, 421: 129688. |
| 47 | DOS R, SANTOS L F S, NAVICKIENE S, et al. Evaluation of metal-organic framework as low-cost adsorbent material in the determination of pesticide residues in soursop exotic fruit (annona muricata) by liquid chromatography[J]. Food Anal Methods, 2015, 8(2): 446-451. |
| 48 | JIA Y, WANG Y, YAN M, et al. Fabrication of iron oxide@MOF-808 as a sorbent for magnetic solid phase extraction of benzoylurea insecticides in tea beverages and juice samples[J]. J Chromatogr, 2020, 1615: 460766. |
| 49 | JANJANI P, BHARDWAJ U, GUPTA R, et al. Bimetallic Mn/Fe MOF modified screen-printed electrodes for non-enzymatic electrochemical sensing of organophosphate[J]. Anal Chim Acta, 2022, 1202: 339676. |
| 50 | ZHANG T, WANG J, ZHANG W, et al. Amorphous Fe/Mn bimetal-organic frameworks: outer and inner structural designs for efficient arsenic(Ⅲ) removal[J]. J Mater Chem A, 2019, 7(6): 2845-2854. |
| 51 | MA J, FANG S, SHI P, et al. Hydrazine-functionalized guar-gum material capable of capturing heavy metal ions[J]. Carbohydr Polym, 2019, 223: 115137. |
| 52 | EL-YAZEED A W S, EL-REASH A Y G, ELATWY L A, et al. Facile fabrication of bimetallic Fe-Mg MOF for the synthesis of xanthenes and removal of heavy metal ions[J]. RSC Adv, 2020, 10(16): 9693-9703. |
| 53 | KOPPULA S, JAGASIA P, PANCHANGAM M K, et al. Synthesis of bimetallic metal-organic frameworks composite for the removal of copper(Ⅱ), chromium(Ⅵ), and uranium(Ⅵ) from the aqueous solution using fixed-bed column adsorption[J]. J Solid State Chem, 2022, 312: 123168. |
| 54 | ALSHORIFI F T, DAFRAWY E S M, AHMED A I. Fe/Co-MOF nanocatalysts: greener chemistry approach for the removal of toxic metals and catalytic applications[J]. ACS Omega, 2022, 7(27): 23421-23444. |
| 55 | PARIDA K, MISHRA K G, DSAH S K. Adsorption of toxic metal ion Cr(Ⅵ) from aqueous state by TiO2-MCM-41: equilibrium and kinetic studies[J]. J Hazard Mater, 2012, 241/242: 395-403. |
| 56 | HALLING-SORENSEN B, SENGELOV G, TJORNELUND J. Toxicity of tetracyclines and tetracycline degradation products to environmentally relevant bacteria, including selected tetracycline-resistant bacteria[J]. Arch Environ Contam Toxicol, 2002, 42(3): 263-271. |
| 57 | WEI F, LIANG X, REN Q, et al. The application of Bimetallic metal-organic frameworks for antibiotics adsorption[J]. J Saudi Chem Soci, 2022, 26(6): 101562. |
| 58 | NGUYEN V T, NGUYEN T B, CHEN C W, et al. Influence of pyrolysis temperature on polycyclic aromatic hydrocarbons production and tetracycline adsorption behavior of biochar derived from spent coffee ground[J]. Bioresource Technol, 2019, 284: 197-203. |
| 59 | WANG Z, WU C W, ZHANG Z, et al. Bimetallic Fe/Co-MOFs for tetracycline elimination[J]. J Mater Sci, 2021, 56(28): 15684-15697. |
| 60 | YANG Z H, CAO J, CHEN Y P, et al. Mn-doped zirconium metal-organic framework as an effective adsorbent for removal of tetracycline and Cr(Ⅵ) from aqueous solution[J]. Microporous Mesoporous Mater, 2019, 277: 277-285. |
| 61 | JI Y, ZANG X, CHEN Z, et al. Silk Sericin enrichment through electrodeposition and carbonous materials for the removal of methylene blue from aqueous solution[J]. Int J Mol Sci, 2022, 23(3): 1668. |
| 62 | AHMADIJOKANI F, MOHAMMADKHANI R, AHMADIPOUYA S, et al. Superior chemical stability of UiO-66 metal-organic frameworks (MOFs) for selective dye adsorption[J]. Chem Eng J, 2020, 399: 125346. |
| 63 | SUN H, ZHANG H, MAO H, et al. Facile synthesis of the magnetic metal-organic framework Fe3O4/Cu3(BTC)2 for efficient dye removal[J]. Environ Chem Lett, 2019, 17: 1091-1096. |
| 64 | SINGH H, RAJ S, RATHOUR R K S, et al. Bimetallic Fe/Al-MOF for the adsorptive removal of multiple dyes: optimization and modeling of batch and hybrid adsorbent-river sand column study and its application in textile industry wastewater[J]. Environ Sci Pollut Res Int, 2022, 29(37): 56249-56264. |
| 65 | FAR H S, HASANZADEH M, NAJAFI M, et al. Highly porous organoclay-supported bimetal-organic framework (CoNi-MOF/OC) composite with efficient and selective adsorption of organic dyes[J]. Environ Sci Pollut Res Int, 2023, 30(15): 43714-43725. |
| 66 | LIU Y, QIU G, LIU Y, et al. Fabrication of CoFe-MOF materials by different methods and adsorption properties for Congo red[J]. J Mol Liquids, 2022, 360: 119405. |
| 67 | SUN Q, LIU M, LI K, et al. Facile synthesis of Fe-containing metal-organic frameworks as highly efficient catalysts for degradation of phenol at neutral pH and ambient temperature[J]. CrystEngComm, 2015, 17(37): 7160-7168. |
| 68 | YAO B, LUA S K, LIM H S, et al. Rapid ultrasound-assisted synthesis of controllable Zn/Co-based zeolitic imidazolate framework nanoparticles for heterogeneous catalysis[J]. Microporous Mesoporous Mater, 2021, 314: 110777. |
| 69 | LUO Q, HUANG X F, DENG Q L, et al. Novel 3D cross-shaped Zn/Co bimetallic zeolite imidazolate frameworks for simultaneous removal Cr(Ⅵ) and Congo red[J]. Environ Sci Pollut Res, 2022, 29(26): 40041-40052. |
| [1] | AN Liancai, HAN Jiufang, ZHANG Yinghui, BU Xianhe. Research and Application Progress on Porous Organic Polymers for Adsorption and Separation of Organic Pollutants in Water System [J]. Chinese Journal of Applied Chemistry, 2018, 35(9): 1019-1025. |
| [2] | CHAI Fanfan,LI Keyan,GUO Xinwen. Composition and Structure Design for High Performance Heterogeneous Fenton Catalysts [J]. Chinese Journal of Applied Chemistry, 2016, 33(2): 133-143. |
| [3] | BU Xiaoyang1, WU Ming1, ZHOU Jiahong2, YANG Xiaodi1, LU Tianhong1, LI Huihui1*. Spectrometric Study on the Interactions Between Organic Pesticide Pollutant and DNA [J]. Chinese Journal of Applied Chemistry, 2011, 28(04): 458-463. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
